Abstract
The purpose of this study is to investigate the efficacy and the mechanism of Hsp90 inhibition of Withaferin A (WA), a steroidal lactone occurring in Withania somnifera, in pancreatic cancer in vitro and in vivo. Withaferin A exhibited potent antiproliferative activity against pancreatic cancer cells in vitro (with IC50s of 1.24, 2.93 and 2.78 μM) in pancreatic cancer cell lines Panc-1, MiaPaca2 and BxPc3, respectively. Annexin V staining showed that WA induced significant apoptosis in Panc-1 cells in a dose dependent manner. Western blotting demonstrated that WA inhibited Hsp90 chaperone activity to induce degradation of Hsp90 client proteins (Akt, Cdk4 and glucocorticoid receptor), which was reversed by the proteasomal inhibitor, MG132. WA-Biotin pull-down assay of Hsp90 using Panc-1 cancer cell lysates and purified Hsp90 showed that WA-biotin binds to C-terminus of Hsp90, which was competitively blocked by unlabeled WA. Co-immunoprecipitation exhibited that WA (10 μM) disrupted Hsp90-Cdc37 complexes from 1–24 hour post treatment, while it neither blocked ATP binding to Hsp90, nor changed Hsp90-P23 association. WA (3, 6 mg/kg) inhibited tumor growth in pancreatic Panc-1 xenografts by 30% and 58%, respectively. These data demonstrate that Withaferin A binds Hsp90, inhibits Hsp90 chaperone activity through an ATP independent mechanism, results in Hsp90 client protein degradation, and exhibits in vivo anticancer activity against pancreatic cancer.
Keywords: Withaferin A, Pancreatic cancer, Hsp90, Reactive cysteine, Client protein, Cdc37
1. Introduction
Pancreatic cancer is the fourth leading cause of cancer deaths in the United States [1, 2] with one- and five-year survival of 23% and 4% [1]. The mortality rates associated with pancreatic cancer are almost equal to its incidence rates. The underlying mechanism of pancreatic tumor formation is rather complex. A number of biochemical and genetic abnormalities have been reported, which include mutations or overexpression of oncogenes (such as KRAS, ERBB2, and AKT) and tumor-suppressor genes (such as P53, BRCA2) [3–6]. In addition, over-expression of growth factors and their receptors, such as TGF-beta, VEGF, and EGFR [6], have also been linked to pancreatic cancer.
The treatment regimens for pancreatic cancer have no substantial improvement over the past few decades [7]. Currently, surgery is the main therapeutic option since chemotherapy and radiation only achieve minimal effects due to rapid progression, late diagnosis, and drug resistance of pancreatic cancer [1]. Unfortunately, only 15–20% of pancreatic cancer patients are amenable to curative resection while 80% of patients generally have nonresectable advanced or metastatic tumors [8]. Furthermore, even in patients with resectable disease, the overall 5-year survival is 15%. Currently, gemcitabine is the standard therapeutic drug for treatment of pancreatic cancer. However, it only improves the disease symptoms with no significant survival benefits. Thus, novel agents for prevention and treatment of pancreatic cancers are highly desired.
Natural products appear to be promising sources of drugs for cancer treatment [9]. Withaferin A (WA), a major active constituent purified from the Indian medicinal plant Withania somnifera, was shown to have antitumor, antiangiogenesis and radiosensitizing activity [10, 11]. The anticancer activity of Withaferin A has been demonstrated in prostate cancer cells [12, 13], breast cancer cells [14], leukemia cells [15], and melanoma cells [16]. It was shown that WA inhibits nuclear factor-κB (NF-κB) activation [11], induces apoptosis in prostate cancer cells through Par-4 induction [12], inhibits IκB kinase activation via a thioalkylation-sensitive redox mechanism [17], inhibits the chymotrypsin-like activity of proteasome [13], and targets the intermediate filament protein vimentin by covalently modifying the cysteine residue [18]. In addition, WA also targets annexin II to induce Actin microfilament aggregation [19].
In our preliminary study, we found that Withaferin A exhibited Hsp90 inhibition characteristics in pancreatic cancer cells by decreasing the levels of Hsp90 client proteins. Thus we intend to investigate the efficacy of WA and Hsp90 inhibition mechanisms in this study. Hsp90 is a molecular chaperone which mediates the folding, assembly, and maturation of many client proteins, including HER-2, EGFR, Akt, Raf-1, Cdk4, mutated p53, which are directly involved in the malignancy [20]. Hsp90 has three distinct domains. The N-terminal domain has the ATP binding site, the middle domain may interact with clients, and the C-terminal domain is responsible for dimerization of Hsp90. An additional ATP binding site is also found to be present in the C-terminus. The ATP binding sites act as a conformational switch to regulate Hsp90 chaperone activity [21]. In cancer cells, the newly synthesized oncogenic client proteins bind to Hsp90 to form an intermediate complex with other co-chaperones (such as Cdc37, Hop, Hsp70, Hsp40, and Hip). Upon ATP binding to Hsp90, client proteins and Hsp90 form a mature complex. This mature superchaperone complex catalyzes the conformational maturation of client proteins [22]. These oncogenic client proteins stimulate cancer cell proliferation and survival. Several Hsp90 inhibitors, which block the ATP binding sites of Hsp90, have been developed and tested in preclinical and clinical models for their anticancer activity [23–26]. Geldanamycin (GA, and its derivatives 17-AAG, IPI-504, 17-DMAG), radicicol and derivatives, purines and derivatives, pyrazoles and isoxazoles, sulfanyl analogues, resorcinol-bearing compounds, and 2-aminopyrimidine-bearing derivatives block the N-terminal ATP binding pocket, whereas novobiocin (and its derivatives) and cisplatin block the C-terminal ATP binding pocket [23, 27–31]. To date, many of these Hsp90 inhibitors have entered preclinical or phase I/II clinical studies [24, 25, 32, 33].
In this study, we investigate the efficacy and mechanism of Withaferin A (WA) for Hsp90 inhibition and its use against pancreatic cancer. Our data suggest that Withaferin A exhibits potent cytotoxicity against pancreatic cancer cells both in vitro and in vivo xenograft models. The anticancer activity of WA is partially due to its direct binding to Hsp90 C-terminus, and inhibiting Hsp90 chaperone activity, inducing Hsp90 client protein degradation through an ATP independent mechanism.
2. Materials and Methods
2.1. Cell culture and reagents
Human pancreatic cancer cell lines Panc-1, BxPC3 and MiaPaCa-2 were cultured in 10% FBS RPMI-1640 or 10% FBS DMEM at 37°C and 5% CO2. Withaferin A was purchased from Calbiochem, Inc. (San Diego, CA). The following antibodies were used for western blot: Akt (Cell Signaling, Beverly, MA), Hsp70 and Hop (StressGen, Victoria, BC, Canada), Cdk4, Cdc37 and Hsp90 (Santa Cruz, Santa Cruz, CA), Actin and p23 (Abcam, Cambridge, MA). Monoclonal Hsp90 antibody H9010 for immunoprecipitation was purchased from Alexis Biochemicals (San Diego, CA), and purified Hsp90β protein for ATP binding assay was a kind gift of Dr. David Toft (Mayo Clinic, Rochester, MN).
2.2. MTS Assay
Pancreatic cancer cells were seeded in 96-well plates at a density of 5000 cells per well. 24 hours later the cells were treated with increasing concentrations of WA as indicated. MTS assay was performed to assess cell viability after 48 h incubation. The IC50 value for cytotoxicity was estimated by WinNonlin software (Pharsight, Mountain View, CA).
2.3. Apoptosis study
The Annexin V-EGFP Apoptosis Detection Kit was purchased from BioVision Research Products (Mountain View, CA) and used as recommended by manufacturer. Human pancreatic Panc-1 cells were treated with 1, 5, and 10 μM WA for 12 h, and stained with Annexin V-EGFP to analyze the phosphoserine inversion. Early apoptotic cells were observed with a fluorescence microscopy.
2.4. Withaferin A-Biotin pull down assay
Withaferin A-Biotin (WA-biotin) was prepared and used in the pull down assay as described previously [19]. Briefly, 500 μg of Panc-1 pancreatic cancer cell whole cell extracts or 5 μg of purified human Hsp90 beta, N-terminus Hsp90 beta, C-terminus Hsp90 beta and yeast Hsp90 were incubated with immobilized WA-Biotin for 2 h at 4 °C in TNEK buffer (5 mM Tris, pH 7.4; NP-40 1%; EDTA 2 mM; KCl 200 mM) supplemented with protease inhibitors. To perform competition assay, the samples were preincubated with 100 μM WA for 1 hr before add with WA-Biotin. The beads were then washed with TNEK buffer for 3 times, and were boiled in loading buffer for 4 min to isolate the bound proteins. Western blot was carried out to analyze the levels of Hsp90 proteins.
2.5. ATP-Sepharose Binding Assay
The assay was performed as previously described [34, 35]. Total of 5 μg of human hsp90β protein with DMSO, WA or 17-AAG were incubated on ice in 200 μl incubation buffer consisting of 10 mM Tris-HCl, 50 mM KCl, 5 mM MgCl2, 2 mM DTT, 20 mM Na2MoO4, 0.01% Nonidet P-40, pH 7.5. After 30 min, 25 μl of pre-equilibrated γ-phosphate-linked ATP-Sepharose (Jena Bioscience GmbH, Jena, Germany) was added to tubes, which were then incubated at 37 °C for another 30 min with frequent mixing to resuspend the resin. Following incubation, the sepharose was washed, pelleted and analyzed by SDS-PAGE.
2.6. Coimmunoprecipitation and Western Blotting assay
The general procedure for coimmunoprecipitation was described as follows. 500 μg of whole cell extracts was incubated with 5 μl H9010 anti-Hsp90 antibody or anti-P23 antibody for 1 h at 4°C, rotating. 30 μl protein G agarose (Pierce, Rockford, IL) was added to each sample, and incubated for another 2 h at 4°C. The beads were washed 3 times with PBS plus protease inhibitors. The beads were boiled in loading buffer for 4 min to isolate the bound proteins. Western blot was carried out to analyze the levels of coimmunoprecipitated proteins. Western blot was performed as previously described [35, 36]. Non-reducing SDS-PAGE was used to analyze the disulfide-bonded protein as described previously [37]. Isolation of triton-soluble and triton-insoluble proteins was performed as described by Chen et al [37].
2.7. Real-time PCR assay
RT-PCR is carried out as described previously [36]. Briefly, Panc-1 cancer cells are treated with 5 μM WA for 12 hrs. TRIzol reagents (Invitrogen, Carlsbad, CA) are used to extract total cellular RNAs as described in protocol provided by manufacturer. Superscript III first strand synthesis kit from Invitrogen is used to reverse transcribe the cDNA. Then the real-time PCR is carried out in ABI PRISM 7900T real-time PCR system (PerkinElmer, Branchburg, NJ) with SYBR Green PCR Master Mix (Applied Biosystems, Foster City, CA). The primers used in RT-PCR are as follows: Akt, forward, 5′-TCT ATG GCG CTG AGA TTG TG-3′, reverse, 5′-CTT AAT GTG CCC GTC CTT GT-3′; Cdk4, forward, 5′-GAA ACT CTG AAG CCG ACC AG-3′, reverse, 5′-GCC CTC TCA GTG TCC AGA AG-3′; glucocorticoid receptor (GR), forward, 5′-GAG AGG GGA GAT GTG ATG GA-3′, reverse, 5′-GTT TTC ACT TGG GGC AGT GT-3′. Internal standard -actin, forward, 5′-GCT CGT CGT CGA CAA CGG CTC-3′; reverse, 5′-CAA ACA TGC TCT GGG TCA TCT TCT C-3′. mRNA levels are calculated as fold change of control. After completion of the RT-PCR, Ct values (cycle numbers in which signal intensity equal to the threshold value) will be obtained from the software. For each samples, ΔCt is calculated as ΔCt = CtAkt/Cdk4/GR − Ctactin. Then ΔΔCt is calculated as ΔΔCt = ΔCttreatment − ΔCtcontrol. The fold change of the Akt/Cdk4/GR mRNA levels relative to control group are calculated as 2−ΔΔCt.
2.8. Pancreatic Tumor Xenograft
The pancreatic tumor xenograft mouse model was used to test anticancer activity of WA similar to previous reports [35, 36]. Briefly, 4 to 6-week old nu/nu athymic female mice were obtained from Charles River Laboratories (Charles River, Wilmington, MA). Pancreatic cancer Panc-1 cells (5–10×106) were mixed with reconstituted basement membrane (Collaborative Research, Bedford, MA) and inoculated s.c. to the right and left flanks of the mice. When the tumors became palpable (~100 mm3), mice were randomly divided into different groups for treatment (n= 6/group). WA was dissolved in the vehicle (10% DMSO, 40% Cremophor/ethanol (3:1), and 50% PBS) [13], and administered at 6 mg/kg or 3 mg/kg by i.p. injection for two continuous days. Then the dosing schedule was changed to two injections per week for 4 weeks. Tumor sizes and body weights were measured twice a week. After 30 days’ drug treatment, and tumor sizes and body weights were monitored until 70 days.
3. Results
3.1. Withaferin A inhibits proliferation in pancreatic cancer cells
The antiproliferative effect of WA (Figure 1A) against human pancreatic cancer cell lines was examined by MTS assay. A 48-h exposure to different concentrations of WA induced a dose-dependent inhibition in cell proliferation. WA exhibited high cytotoxicity against Panc-1 cells with an IC50 of 1.24 μM (Figure 1B), whereas, WA showed relatively lower cytotoxicity against MiaPaca2 and BxPc3, with IC50s of 2.93 μM and 2.78 μM (Figure 1C and 1D). Overall, WA showed potent antiproliferative effect against these three human pancreatic cancer cell lines.
Figure 1.
A. Chemical structure of WA.
B. Effect of WA treatment on viability of Panc-1 cells. Panc-1 cells were seeded in 96-well plates at a density of 5000 cells per well. 24 hours later the cells were subjected to WA treatment with concentrations of 0.05, 0.1, 0.5, 1.0, 2.5, 5, 10, and 20 μM. MTS assay was performed to assess cell viability after 48 h incubation.
C. Effect of WA treatment on viability of MiaPaCa2 cells. Viability of MiaPaCa2 cells after WA treatment was assessed similar to B.
D. Effect of WA treatment on viability of BxPc3 cells. Viability of BxPc3 cells after WA treatment was assessed similar to B.
3.2. Withaferin A induces apoptosis in pancreatic cancer cells
To illustrate that WA induces apoptosis in pancreatic cancer cells, annexin-V staining was conducted in Panc-1 cells with WA treatment. As shown in Figure 2, the Annexin-V positive staining cells accounted for 18.5±1.68, 46.8±5.22 and 68.1±7.14 of the overall cell population in Panc-1 cells treated with 1, 5, and 10 μM WA for 12 h, respectively. In contrast, only marginal apoptotic cells were observed in control Panc-1 cells.
Figure 2. WA induces apoptosis in Panc-1 cells as determined by Annexin V staining.
A. Representative images from one of four independent experiments. Panc-1 cells were treated with 1, 5, and 10 μM WA for 12 h. Cells were stained with Annexin V-EGFP (green). Apoptotic cells were observed under fluorescent microscope.
B. Quantification of WA induced apoptotic cells. The percentage of apoptotic cells were calculated as Annexin-EGFP positive cells divided by the total cancer cells.
3.3. Withaferin A induces Hsp90 client protein degradation
WA exhibited potent cytotoxicity against pancreatic cancer cells and induced apoptosis in Panc-1 cells. To investigate the underlying mechanism, we screened a panel of protein level changes in Panc-1 cells in response to WA treatment. Hsp90 client proteins (Akt, Cdk4 and Glucocorticoid receptor (GR)) were observed to be decreased. These proteins exhibited time- and dose- dependent degradation in response to WA treatment (Figure 3A and 3B). After exposure to 10 μM WA for 6 h, Akt and Cdk4 protein levels started to decrease by 1.88- and 1.95- fold. After 24 h treatment, the protein levels were undetectable. GR protein levels decreased even faster and became undetectable as early as 2 h after exposure to 10 μM WA (Figure 3C). Previous studies have shown that these proteins are clients of Hsp90, and inhibiting Hsp90 chaperone activity would lead to the degradation of these proteins [35, 38]. These data suggest that inhibition of Hsp90 chaperone activity might contribute to the anticancer activity of WA.
Figure 3. Protein level changes after WA treatment in Panc-1 cells.
A and B. WA induces Hsp90 client protein Akt and Cdk4 degradation in Panc-1 cells in a time- and dose-dependent manner. Panc-1 cells were treated with different concentrations of WA for different times. Cell lysates (50 μg protein in each lane) were analyzed by western blot analysis with specific antibodies to Akt, Cdk4 and Actin. Actin was served as internal standard.
C. WA induces Hsp90 client protein GR degradation. Panc-1 cells were treated with10 μM WA for different times. Cell lysates (50 μg protein in each lane) were analyzed by western blot analysis with specific antibodies to GR and Actin. Actin was served as internal standard.
D. WA induces expression of Hsp70. Panc-1 cells were treated with10 μM WA for different times. Cell lysates (50 μg protein in each lane) were analyzed by western blot analysis with specific antibodies to Hsp70 and Actin. Actin was served as internal standard.
E. WA does not change the Cdk2 protein level. Panc-1 cells were treated with10 μM WA for different times. Cell lysates (50 μg protein in each lane) were analyzed by western blot analysis with specific antibodies to Cdk2 and Actin. Actin was served as internal standard.
To further confirm inhibition of Hsp90 by WA, we examined two additional protein expression level changes, Hsp70 and Cdk2. The induction of Hsp70 is another molecular signature in response to Hsp90 inhibition [39]. As shown in Fig 3D, 10 μM WA increased the protein level of Hsp70 by 13.47-fold after 6 h while without affecting the Hsp90 protein level. To demonstrate WA specifically inhibits Hsp90, a non-Hsp90 client protein Cdk2 was examined. Indeed, Cdk2 levels were not significantly altered after WA treatment (Fig 3E). These data demonstrate that WA inhibited Hsp90 chaperone activity.
3.4. Withaferin A directly binds to Hsp90
Previous study reported that Hsp90 is sensitive to cellular redox conditions and tend to form disulfide bond under oxidative stress [37, 40]. In addition, susceptible cysteine residues in C-terminal Hsp90 were revealed including Cys-521, Cys-589/590 and Cys597 [41, 42]. WA was demonstrated to be highly reactive with cysteine residues in proteins [43, 44], such as annexin II [19] and vimentin [18] to form covalent bonds. Therefore, we tested whether WA will bind to Hsp90 using WA-biotin pull down assay. The results show that WA-biotin successfully pulled down Hsp90 both from the cell lysate and purified full length human hsp90 (Figure 4A). To further illustrate the binding domains of Hsp90 with WA-biotin, we performed the pull down assay against Hsp90 fragments N-terminus Hsp90 (without cysteine residue), C-terminus Hsp90 (with cysteine residue), and full length yeast Hsp90 (without cysteine residues). As shown in Figure 4A, WA-biotin can only pull down C-terminus Hsp90 (with cysteine residues), but not N-terminus Hsp90 nor yeast Hsp90. Hence, WA-Biotin binding to Hsp90 is dependent on cysteine residues on Hsp90. In addition, the WA-biotin binding to Hsp90 was in a competitive manner since 100 μM unlabeled WA preincubation for 1 hr would significantly decrease the WA-biotin binding to Hsp90 both in cell lysate and purified full length Hsp90 as well as C-terminus Hsp90 (Figure 4B).
Figure 4.
A. WA-Biotin Hsp90 pull down assay.1 mg of Panc-1 cell lysates (Lysate), 5 μg of purified full length human Hsp90β (Full Hsp90) 5 μg of N-terminus human Hsp90β (N-Hsp90), 5 μg of C-terminus human Hsp90β (C-Hsp90) and 5 μg of yeast Hsp90 (Y-Hsp90) were used to carry out the WA-Biotin pull down assay. The WA-Biotin pull down protein were subjected to western blot analysis with specific antibodies to Hsp90.
B. WA competes with WA-Biotin binding to Hsp90. The samples (Lysate, Full Hsp90 and C-Hsp90) were preincubated with 100 μM WA for 1 h before subject to WA-Biotin binding assay. The WA-Biotin pull down protein were subjected to western blot analysis with specific antibodies to Hsp90.
C. WA induces Hsp90 aggregation in a dose dependent manner. Panc-1 cells were treated with 1, 5 and 10 μM WA for 24 h. Cell lysates (50 μg protein in each lane) were subjected to non-reducing gel electrophoresis and then analyzed by western blot with specific antibodies to Hsp90.
Next we examined the aggregation of Hsp90 after WA treatment. Non-reducing gel electrophoresis was performed to detect the formation of Hsp90 aggregation, which would exhibit a slower migration pattern and appeared as higher molecular weight bands. As shown in Fig 4C, WA induced Hsp90 aggregation in a dose-dependent manner.
3.5. Withaferin A induces Hsp90 client protein degradation through proteasome
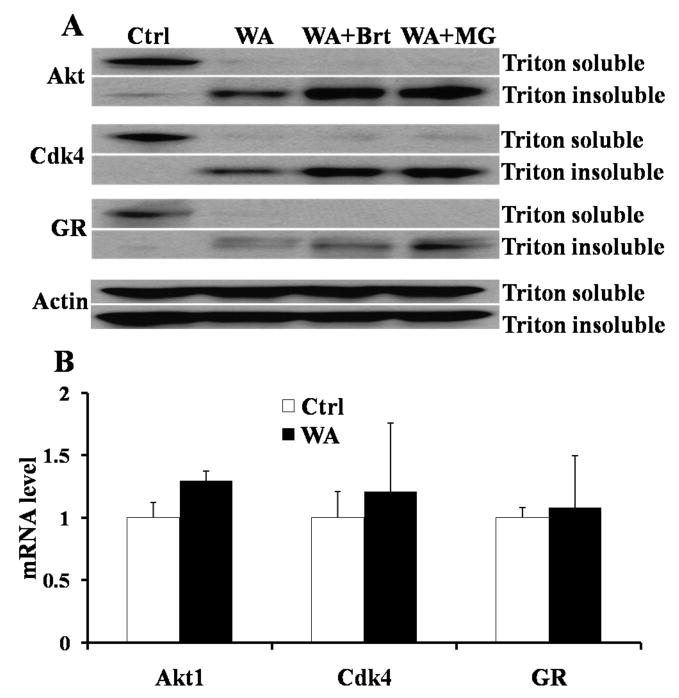
Since classical Hsp90 inhibitor induced Hsp90 client protein degradation was proteasome mediated, we further investigated whether WA induced Hsp90 client protein degradation was also proteasome-dependent. Two proteasome inhibitors Bortezomib and MG132 were used to reverse the protein degradation. As shown in Fig 5A, preincubation with 10 μM Bortezomib and MG132 could rescue the degradation of Hsp90 client proteins. All of the three Hsp90 client proteins (Akt, Cdk4, and GR) under investigation were found to accumulate in the triton-insoluble fraction after combination treatment of WA and proteasomal inhibitors, while WA alone decreased the levels of Akt, Cdk4 and GR. In addition, we carried out RT-PCR to examine whether WA affects the mRNA levels of these three genes. Fig 5B shows that WA treatment did not change the mRNA levels of these three genes. These data suggest that WA induced Hsp90 client protein degradation is proteasome-dependent and WA did not affect the transcriptional level of Hsp90 client proteins.
Figure 5.
A. WA induced Hsp90 client protein degradation is proteasome-dependent. Panc-1 cells were preincubated with 10 μM Bortezomib (Brt) or MG132 (MG) for 1 h, and then were treated with 5 μM WA for another 12 h. Cells were collected and proteins were isolated as triton soluble part and triton insoluble part. Proteins (both triton soluble and triton insoluble parts) were subjected to western blot analysis with specific antibodies to Akt, Cdk4, GR and Actin. Actin was served as internal standard.
B. mRNA levels of Hsp90 client protein in Panc-1 cells after WA treatment. Panc-1 cells were treated with 5 μM WA for 12 h, and the total mRNAs were isolated. RT-PCR was carried out to examine the mRNA levels of Akt, Cdk4 and GR using the specific primers of Akt, Cdk4, GR and Actin. Actin was served as internal standard.
3.6. Withaferin A does not block ATP binding to Hsp90
Most of current Hsp90 inhibitors, including geldanamycin, 17-AAG, IPI-504 and 17-DMAG, bind to the Hsp90 ATP binding pocket which prevents client protein refolding [23, 27]. To investigate whether WA also change the ATP binding to Hsp90, an ATP-sepharose beads pull down assay was performed. As shown in Fig 6, 5 μM 17-AAG completely blocked ATP beads binding to Hsp90; in contrast, 5, 10, and 20 μM WA did not block ATP beads binding to Hsp90. As a negative control, 20 μM celastrol did not block ATP beads binding to Hsp90, which is consistent with previous study [35].
Figure 6. WA does not inhibit ATP binding to Hsp90.
5 μg of purified human Hsp90β was incubated with WA, 17-AAG and Celastrol (Cel) for 30 min. 25 μl ATP-sepharose beads were added to the samples to pull down Hsp90. Western blot was used to detect Hsp90 using specific antibody to Hsp90.
3.7. Withaferin A dissociates Hsp90-Cdc37 complex in pancreatic cancer cells
Since Hsp90 forms a superchaperone complex with other cochaporones, including Hsp70, P23, Cdc37, Hop and immunophilins, we further tested the alteration of Hsp90 superchaperone complexes in response to WA treatment by using co-immunoprecipitation (coIP) assay in Panc-1 cells. Panc-1 cells were treated with 10 μM WA for 1, 6, 12, and 24 h, coIP Hsp90 was carried out. The coIP samples were then immunobloted with anti-Cdc37 antibody. Fig 7A shows that 10 μM WA completely disrupted the Hsp90-Cdc37 complex as early as 1 h post treatment. In contrast, the Hsp90-Hop complex was not decreased by WA treatment. Fig 7B shows the dose-dependence of WA on disrupting the Hsp90-Cdc37 complex. After exposure to WA for 24 h, 1 μM WA decreased the amount of Cdc37 by 2.04-folds, which was pulled down by Hsp90. WA (5 μM) completely blocked the Hsp90-Cdc37 complex. To investigate whether the decreased levels of Cdc37 in the coIP results were due to the expression level alteration of Cdc37, western blotting was carried out to examine the protein level of Cdc37 without coIP. Fig 7C shows that WA did not change the Cdc37 protein level. These data further confirmed that the decreased level of Cdc37 by WA in coIP Hsp90 samples was due to the dissociation of Hsp90-Cdc37 complex.
Figure 7. WA disrupts Hsp90-Cdc37 complex in Panc-1 cells.
A and B. Co-immunoprecipitation (coIP) Hsp90. Cell lysates (500 μg total protein) were immunoprecipitated with Hsp90 antibody. Western blot was performed to detect Cdc37, Hop and Hsp90 using specific antibodies to Cdc37, Hop and Hsp90. A. Panc-1 cells were treated with 10 μM WA for 0 to 24 h. B. Panc-1 cells were treated with 1, 5, and 10 μM WA for 24 h. Input, total cell lysate; -IgG, without adding antibody.
C. Western blot analysis of Cdc37 expression level. Panc-1 cells were treated with 10 μM WA for different times. Cell lysates (50 μg protein in each lane) were analyzed by western blot with specific antibodies to Cdc37 and Actin. Actin was served as internal standard.
D. CoIP P23. Cell lysates (500 μg total protein) were immunoprecipitated with P23 antibody, then western blot was performed to detect Hsp90 and P23 using specific antibodies to Hsp90 and P23.
P23 has been demonstrated to bind directly to Hsp90 when it is in the ATP bound conformation [45]. Classical Hsp90 inhibitors like geldanamycin (GA) and PU24FCl bind to the ATP binding pocket and lock the Hsp90 in the intermediate superchaperone complex [46, 47], hence Hsp90 will no longer be available to bind to p23. As shown in Fig 7D, coIP with P23 showed that WA did not change Hsp90-P23 association, resulting in equal amount of Hsp90 pulled down by P23. In contrast, 17-AAG decreased the Hsp90-P23 interaction dramatically, which was consistent with ATP binding assay (Fig 7D).
3.8. Withaferin A exhibits anticancer activity in pancreatic cancer xenografts
The data described above showed that WA is an Hsp90 inhibitor by binding to Hsp90, which resulted in Hsp90 client protein degradation and apoptosis in pancreatic cancer cells in vitro. We next examined WA’s therapeutic efficacy in vivo. Pancreatic cancer (Panc-1) xenografts were generated in female nude mice. When the tumors reached 100 mm3, the mice were randomly divided into three groups (n=6). The mice were treated i.p. with either vehicle control or WA at 3.0 mg/kg or 6.0 mg/kg. After 70 days, control tumors grew to an average size of 1014±176 mm3. In contrast, tumors from 3.0 mg/kg and 6.0 mg/kg WA-treatment group grew to an average size of 701±268 and 422±95 mm3, corresponding to 30% and 58% inhibition, respectively (P<0.001; Fig 8A). Systemic toxicity of WA was analyzed by measuring the animal weights. As shown in Fig 8B, the higher dose (6 mg/kg) WA treatment group had negligible weight loss (<10%) during the first week treatment, regained their weight from the second week, and had a 12% increase in body weight increase after 70 days. Meanwhile, mice in control group and the 3 mg/kg WA treatment group did not display weight loss. These data demonstrated WA shows potential anticancer effect against pancreatic cancer in vivo without significant toxicity.
Figure 8. Antitumor effect of WA in Panc-1 xenografts.
A. Tumor growth curves. The pancreatic tumor xenograft mouse model was generated by injecting the Panc-1 cancer cells s.c. to the right and left flanks of the nude mice. When the tumors reached 100 mm3, mice were divided randomly into three groups (n= 6/group) to receive vehicle, 3 mg/kg or 6 mg/kg WA treatment as scheduled. Tumor sizes and body weights were measured twice a week. Drug treatment was stopped after 30 days’ treatment, and tumor sizes and body weights were monitored until 70 days. Arrows indicate the date that stops treatment.
B. Body weight of mice. Body weights of mice were monitored twice per week.
4. Discussion
In the present study, we evaluated the anticancer efficacy of WA against pancreatic cancer. WA treatment significantly induced antiproliferative effects against Panc-1, BxPc3 and MiaPaca2 pancreatic cancer cells in cell culture and exhibited potent tumor growth inhibition of pancreatic cancer xenografts. Our data showed that WA directly binds to Hsp90 and leads to the degradation of Hsp90 client proteins through an ATP independent mechanism.
Hsp90 is crucial for maintaining the native conformation of proteins. It was found to be highly expressed in various cancerous tissues compared to the non-cancerous tissue [48, 49], which provides cancer cells selectivity by Hsp90 inhibitor [22, 50]. For instance, 17-AAG binding affinity to Hsp90 in cancer cells is 100-fold higher than that to Hsp90 in normal cells [22]. Therefore, inhibiting the Hsp90 chaperone activity emerged as a new molecular target for developing anticancer agents because of its high selectivity and simultaneous knockdown of various oncogenic proteins. Several Hsp90 inhibitors have been developed and tested in preclinical and clinical models for their anticancer activity including 17-AAG, 17-DMAG and IPI-504 [23–26].
In addition to ATP binding blockage of Hsp90, researchers have also identified other Hsp90 inhibition mechanisms. For example, the histone deacetylase inhibitors (hydroxamic acid analogue, LAQ 824 and LBH589) were shown to induce the hyperacetylation of Hsp90, resulting in inhibition of ATP binding and attenuation of chaperone activity [51]. In this study, we demonstrated that WA inhibited Hsp90 chaperone activity to induce Hsp90 client protein degradation. However, unlike the classical Hsp90 inhibitor, WA directly binds to Hsp90 C-terminus. These were evidenced by the pull-down assay of WA-Biotin to C-terminus Hsp90 containing cysteine residues but not N-terminus Hsp90 or yeast Hsp90 (without cysteine residues). In addition, the binding of WA-biotin to Hsp90 could be competitively inhibited by unlabeled WA. Furthermore, ATP-sepharose beads pull down assay did not show any inhibition of WA to ATP binding to Hsp90. These data also further suggest that WA inhibition of Hsp90 is not through an ATP dependent mechanism, which is different from the classical Hsp90 inhibitors.
To further confirm that WA binds to Hsp90 reactive cysteine residues, we examined whether exogenous thiols in cell culture would rescue the WA-inhibition to Hsp90. N-acetylcysteine (NAC) is a natural sulfur-containing amino acid derivative and is a thiol antioxidant [52]. Preincubation of Panc-1 cells with NAC reversed WA induced Hsp90 aggregation (data not shown), whereas NAC failed to reverse a mild oxidant, hydrogen peroxide (H2O2) induced Hsp90 aggregation. Although geldanamycin and its derivatives were shown to be able to produce ROS [53], 17-AAG was unable to induce Hsp90 aggregation. In addition, WA induced Hsp90 client protein degradation and Hsp70 induction were also rescued by NAC pretreatment (Data not shown). Regardless the mechanism of NAC in reverse WA effects (chemical reaction or cellular response), these data suggest that WA may inhibit Hsp90 function through cysteine of the C-terminal Hsp90. However, the specific cysteine residues that WA binds to need to be further elucidated.
One interesting phenomenon we observed is that WA disrupts Hsp90-Cdc37 complex. Cdc37 is believed to play a central role in regulating kinase client proteins in the intermediate Hsp90 superchaperone complex. A client protein first binds the Hsp70/Hsp40 chaperone complex and then interacts with Cdc37. Hsp90 is subsequently recruited to the complex via p60/Hop [54]. Similar to Hsp90, Cdc37 is also upregulated in cancer cells. Cdc37 is highly expressed in all prostate tumors and absent from normal prostate epithelium. Transgenic mice expressing Cdc37 in the prostate epithelium have displayed dramatic proliferative disorders in the prostate, including epithelial hyperplasia and dysplasia [55]. Hepatocellular carcinoma also overexpresses Cdc37 and Hsp90 compared to normal and surrounding tissues [56]. Previous study showed that Celastrol inhibited Hsp90 chaperone activity by blocking Hsp90-Cdc37 interactions and led to Hsp90 client protein degradation for its anticancer activity [35]. Celastrol also binds to C-terminus of Hsp90 (Zhang et al., unpublished data). In the present study, we found that WA disrupts Hsp90-Cdc37 complex. WA blockage of Hsp90-Cdc37 complex might be a result of WA binding-induced conformational change of Hsp90 or other unknown mechanisms. Further study is warranted to elucidate these mechanisms. It is worth noting that WA inhibition of Hsp90 chaperone activity should not be solely due to the disruption of Hsp90-Cdc37 association. As described earlier, Cdc37 helps load its client proteins onto the Hsp90 chaperone complex. The client proteins of Cdc37 include many kinases, such as Raf-1, Akt and Src family kinases, and steroid receptors such as androgen receptor but not the closely related glucocorticoid receptor (GR) [57]. However, our data showed that WA treatment could also induce the degradation of GR. These data suggest that WA inhibits Hsp90 chaperone activity might be also due to the direct binding of WA to Hsp90 [42].
Furthermore, P23 and Cdc37 were demonstrated to bind to the same sites on the N-terminus of Hsp90, and binding of P23 and Cdc37 to Hsp90 is mutually exclusive [35, 58]. Cdc37 and P23 bind to Hsp90 at different stages of the chaperone cycle: Cdc37 binds to Hsp90 in the intermediate complex; whereas P23 binds to Hsp90 in the mature complex in which Hsp90 adopts a different conformation from the intermediate complex. Classical Hsp90 inhibitors such as geldanamycin and 17-AAG lock Hsp90 in the intermediate complex, hence preventing P23 binding to Hsp90 [35]. Our data showed that WA did not interfere with the Hsp90-P23 interaction, while 17-AAG induced Hsp90-P23 dissociation. There are two possible mechanisms for WA induced Hsp90-Cdc37 dissociation but not Hsp90-P23 dissociation: one is that WA binds directly to the cysteine residues on the C-terminus of Hsp90, resulting in a conformational change of Hsp90 which preventing Cdc37 but not P23 from binding; another is that besides Hsp90, WA also reacts with cysteines in Cdc37 which contributes to the disruption of Hsp90-Cdc37 interaction, however, although cysteine residues are present in P23, they are not accessible to WA, thus providing a selectivity of action. It is important to note that this is rather speculative and needs to be further confirmed.
Previous studies have shown that the 4β-hydroxy-5β, 6β-epoxy-2-en-1-one moiety and unsaturated lactone are critical for WA’s biological function [59]. The epoxide within B ring and the unsaturated lactone ring were demonstrated to be involved in Michael addition thioalkylation reactions [43, 44, 60]. In addition, the ketone containing unsaturated A-ring could also react with thiol-nucleophiles and act as Michael acceptor [59]. Thus these three functional groups could be crucial for the interaction between WA and Hsp90. Gedunin and aforementioned Celastrol are two natural products exhibiting Hsp90 inhibitory activity. They modulate Hsp90 activity by a mechanism different from classical Hsp90 inhibitors such as GA, since they do not competitively bind to the ATP-binding pocket of Hsp90 [47]. The exact mechanism how Gedunin inhibits Hsp90 is still unknown. Gedunin also possesses a ketone containing unsaturated A ring and an additional epoxide which are similar to WA. Although the preliminary structure-activity studies of Gedunin revealed that the, α, β-unsaturated ketone within the A ring of Gedunin does not behave as a Michael acceptor [61], the epoxide on Gedunin might act as a Michael acceptor and thus Gedunin might also inhibit Hsp90 in a way similar to WA. On the other hand, Celastrol also has electrophilic sites within the unsaturated A and B ring and was shown to react with thiols in proteins [62, 63]. Our unpublished data by Zhang et al. indicate that Celastrol binds to C-terminus Hsp90; hence, it would not be surprised if Celastrol inhibits Hsp90 via binding to reactive cysteine residues of Hsp90.
Withaferin A belongs to a large family of natural products steroidal lactone triterpenoids, the withanolides, which are major constituents purified from medicinal plant Withania somnifera and its related solanaceae species such as Physalis, Nicandra, Dunalia, Datura, Jaborosa, and Acnistus [64]. The withanolides are demonstrated to have antitumor, antibacterial, anti-inflammatory, antidepressant, antioxidant, antiulcer, cytotoxic, quinone reductase induction, antileishmanial, antitrypanosomal, immunosuppressive, cognition-enhancing and memory-improving effects, as well as hypotensive, bradycardic and respiratory-stimulant action [65–67]. As a prototype of the withanolides, Withaferin A has been studied extensively. Up to date, over 130 withanolides are known and more than 40 withanolides are isolated [68]. The individual withanolides were purified and evaluated for their biological functions. For instance, some newly isolated withanolides have been shown to have cytotoxic activity, including Withangulatin B, Withangulatin C, Withangulatin G, Withangulatin H, and Withangulatin I [66, 67]. The structure-activity studies further confirmed that the unsaturated A ring and epoxide are important for the cytotoxic activity of withanolides [66]. Another Withanolide, Withangulatin A was shown to inhibit topoisomerase II and induce heat shock response [69, 70]. Although DNA damage mediated by topoisomerase II inhibitors has been shown to induce heat shock response [71], another topoisomerase II inhibitor VM-26 could not induce heat shock response in the same cell line as Withangulatin A [70]. Therefore, Withangulatin A induces heat shock response through a way other than topoisomerase II inhibition mediated DNA damage. Considering the quasi identical structure (the same unsaturated ketone containing A ring, epoxide containing B ring and unsaturated lactone ring) of Withangulatin A to Withaferin A, Withangulatin A might induce heat shock response by inhibition of Hsp90 [72]. In addition, Tubocapsenolide A (TA), another withanolide possessing the three key functional groups, showed high cytotoxicity against cancer cells and exhibited Hsp90 inhibitory activity [37]. TA induces Hsp90 client protein degradation and induces Hsp90 dimer formation. Although there’s no direct evidence that TA binds to Hsp90, TA could inhibit Hsp90 through binding to the reactive cysteine residues of Hsp90 like WA.
In conclusion, our data suggest that withaferin A represents a new type of Hsp90 inhibitor. It directly binds Hsp90, inhibits Hsp90 with an ATP-independent mechanism, induces Hsp90 client protein degradation and disrupts the Hsp90-Cdc37 interaction. These Hsp90 inhibition mechanisms of withaferin A may partially contribute to its anticancer activity in vitro pancreatic cancer cell lines and in vivo pancreatic cancer xenografts. These data provide a potential of withaferin A as a novel Hsp90 inhibitor for use against pancreatic cancers.
Acknowledgments
This study is partially supported by NIH funding RO1 CA 120023, University of Michigan Cancer Center Research Grant (Munn), University of Michigan Cancer Center Core Grant to DS. We thank Dr. Thomas Ratajczak (University of Western Australia, Australia) for the generous gifts of the purified N-Hsp90β and expression plasmids, pET15b-hHsp90β and pET28a(+)-hHsp90β (530–724), respectively. We also thank Dr. Dan Bolon (University of Massachusetts, Massachusetts) for the kind gifts of the purified yeast Hsp90, human full length Hsp90, N-terminus Hsp90 and C-terminus Hsp90. We thank Luke Whitesell (Whitehead Institute for Biomedical Research, Nine Cambridge Center, Cambridge, MA 02142) for reagents and insightful suggestions.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Bardeesy N, DePinho RA. Pancreatic cancer biology and genetics. Nat Rev Cancer. 2002;2:897–909. doi: 10.1038/nrc949. [DOI] [PubMed] [Google Scholar]
- 2.Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ. Cancer statistics. CA Cancer J Clin. 2007;57:43–66. doi: 10.3322/canjclin.57.1.43. [DOI] [PubMed] [Google Scholar]
- 3.Di Costanzo F, Carlini P, Doni L, Massidda B, Mattioli R, Iop A, et al. Gemcitabine with or without continuous infusion 5-FU in advanced pancreatic cancer: a randomised phase II trial of the Italian oncology group for clinical research (GOIRC) British Journal of Cancer. 2005;93:185–9. doi: 10.1038/sj.bjc.6602640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kindler HL, Friberg G, Singh DA, Locker G, Nattam S, Kozloff M, et al. Phase II trial of bevacizumab plus gemcitabine in patients with advanced pancreatic cancer. J Clin Oncol. 2005;23:8033–40. doi: 10.1200/JCO.2005.01.9661. [DOI] [PubMed] [Google Scholar]
- 5.Xiong HQ, Rosenberg A, LoBuglio A, Schmidt W, Wolff RA, Deutsch J, et al. Cetuximab, a monoclonal antibody targeting the epidermal growth factor receptor, in combination with gemcitabine for advanced pancreatic cancer: a multicenter phase II Trial. J Clin Oncol. 2004;22:2610–6. doi: 10.1200/JCO.2004.12.040. [DOI] [PubMed] [Google Scholar]
- 6.Buchler P, Reber HA, Buchler M, Shrinkante S, Buchler MW, Friess H, et al. Hypoxia-inducible factor 1 regulates vascular endothelial growth factor expression in human pancreatic cancer. Pancreas. 2003;26:56–64. doi: 10.1097/00006676-200301000-00010. [DOI] [PubMed] [Google Scholar]
- 7.Michaud DS. Epidemiology of pancreatic cancer. Minerva Chir. 2004;59:99–111. [PubMed] [Google Scholar]
- 8.Schnall SF, Macdonald JS. Chemotherapy of adenocarcinoma of the pancreas. Semin Oncol. 1996;23:220–8. [PubMed] [Google Scholar]
- 9.Newman DJ, Cragg GM, Snader KM. Natural products as sources of new drugs over the period 1981–2002. J Nat Prod. 2003;66:1022–37. doi: 10.1021/np030096l. [DOI] [PubMed] [Google Scholar]
- 10.Shohat B, Gitter S, Abraham A, Lavie D. Antitumor activity of withaferin A (NSC-101088) Cancer chemotherapy reports. 1967;51:271–6. [PubMed] [Google Scholar]
- 11.Mohan R, Hammers HJ, Bargagna-Mohan P, Zhan XH, Herbstritt CJ, Ruiz A, et al. Withaferin A is a potent inhibitor of angiogenesis. Angiogenesis. 2004;7:115–22. doi: 10.1007/s10456-004-1026-3. [DOI] [PubMed] [Google Scholar]
- 12.Srinivasan S, Ranga RS, Burikhanov R, Han SS, Chendil D. Par-4-dependent apoptosis by the dietary compound withaferin A in prostate cancer cells. Cancer Res. 2007;67:246–53. doi: 10.1158/0008-5472.CAN-06-2430. [DOI] [PubMed] [Google Scholar]
- 13.Yang H, Shi G, Dou QP. The tumor proteasome is a primary target for the natural anticancer compound Withaferin A isolated from “Indian winter cherry”. Mol Pharmacol. 2007;71:426–37. doi: 10.1124/mol.106.030015. [DOI] [PubMed] [Google Scholar]
- 14.Stan SD, Hahm ER, Warin R, Singh SV. Withaferin A causes FOXO3a- and Bim-dependent apoptosis and inhibits growth of human breast cancer cells in vivo. Cancer Res. 2008;68:7661–9. doi: 10.1158/0008-5472.CAN-08-1510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Malik F, Kumar A, Bhushan S, Khan S, Bhatia A, Suri KA, et al. Reactive oxygen species generation and mitochondrial dysfunction in the apoptotic cell death of human myeloid leukemia HL-60 cells by a dietary compound withaferin A with concomitant protection by N-acetyl cysteine. Apoptosis. 2007;12:2115–33. doi: 10.1007/s10495-007-0129-x. [DOI] [PubMed] [Google Scholar]
- 16.Devi PU, Kamath R, Rao BS. Radiosensitization of a mouse melanoma by withaferin A: in vivo studies. Indian J Exp Biol. 2000;38:432–7. [PubMed] [Google Scholar]
- 17.Kaileh M, Vanden Berghe W, Heyerick A, Horion J, Piette J, Libert C, et al. Withaferin a strongly elicits IkappaB kinase beta hyperphosphorylation concomitant with potent inhibition of its kinase activity. J Biol Chem. 2007;282:4253–64. doi: 10.1074/jbc.M606728200. [DOI] [PubMed] [Google Scholar]
- 18.Bargagna-Mohan P, Hamza A, Kim YE, Khuan Abby Ho Y, Mor-Vaknin N, Wendschlag N, et al. The tumor inhibitor and antiangiogenic agent withaferin A targets the intermediate filament protein vimentin. Chem Biol. 2007;14:623–34. doi: 10.1016/j.chembiol.2007.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Falsey RR, Marron MT, Gunaherath GM, Shirahatti N, Mahadevan D, Gunatilaka AA, et al. Actin microfilament aggregation induced by withaferin A is mediated by annexin II. Nat Chem Biol. 2006;2:33–8. doi: 10.1038/nchembio755. [DOI] [PubMed] [Google Scholar]
- 20.Rowlands MG, Newbatt YM, Prodromou C, Pearl LH, Workman P, Aherne W. High-throughput screening assay for inhibitors of heat-shock protein 90 ATPase activity. Anal Biochem. 2004;327:176–83. doi: 10.1016/j.ab.2003.10.038. [DOI] [PubMed] [Google Scholar]
- 21.Prodromou C, Roe SM, O’Brien R, Ladbury JE, Piper PW, Pearl LH. Identification and structural characterization of the ATP/ADP-binding site in the Hsp90 molecular chaperone. Cell. 1997;90:65–75. doi: 10.1016/s0092-8674(00)80314-1. [DOI] [PubMed] [Google Scholar]
- 22.Kamal A, Thao L, Sensintaffar J, Zhang L, Boehm MF, Fritz LC, et al. A high-affinity conformation of Hsp90 confers tumour selectivity on Hsp90 inhibitors. Nature. 2003;425:407–10. doi: 10.1038/nature01913. [DOI] [PubMed] [Google Scholar]
- 23.Chiosis G. Targeting chaperones in transformed systems--a focus on Hsp90 and cancer. Expert Opin Ther Targets. 2006;10:37–50. doi: 10.1517/14728222.10.1.37. [DOI] [PubMed] [Google Scholar]
- 24.Adams J, Elliott PJ. New agents in cancer clinical trials. Oncogene. 2000;19:6687–92. doi: 10.1038/sj.onc.1204088. [DOI] [PubMed] [Google Scholar]
- 25.Egorin MJ, Zuhowski EG, Rosen DM, Sentz DL, Covey JM, Eiseman JL. Plasma pharmacokinetics and tissue distribution of 17-(allylamino)-17-demethoxygeldanamycin (NSC 330507) in CD2F1 mice1. Cancer Chemother Pharmacol. 2001;47:291–302. doi: 10.1007/s002800000242. [DOI] [PubMed] [Google Scholar]
- 26.Ramanathan RK, Trump DL, Eiseman JL, Belani CP, Agarwala SS, Zuhowski EG, et al. Phase I pharmacokinetic-pharmacodynamic study of 17-(allylamino)-17-demethoxygeldanamycin (17AAG, NSC 330507), a novel inhibitor of heat shock protein 90, in patients with refractory advanced cancers. Clin Cancer Res. 2005;11:3385–91. doi: 10.1158/1078-0432.CCR-04-2322. [DOI] [PubMed] [Google Scholar]
- 27.Neckers L. Development of small molecule Hsp90 inhibitors: utilizing both forward and reverse chemical genomics for drug identification. Curr Med Chem. 2003;10:733–9. doi: 10.2174/0929867033457818. [DOI] [PubMed] [Google Scholar]
- 28.Janin YL. Heat shock protein 90 inhibitors. A text book example of medicinal chemistry? J Med Chem. 2005;48:7503–12. doi: 10.1021/jm050759r. [DOI] [PubMed] [Google Scholar]
- 29.Taldone T, Sun W, Chiosis G. Discovery and development of heat shock protein 90 inhibitors. Bioorg Med Chem. 2009;17:2225–35. doi: 10.1016/j.bmc.2008.10.087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Powers MV, Workman P. Inhibitors of the heat shock response: biology and pharmacology. FEBS Lett. 2007;581:3758–69. doi: 10.1016/j.febslet.2007.05.040. [DOI] [PubMed] [Google Scholar]
- 31.Whitesell L, Lindquist SL. HSP90 and the chaperoning of cancer. Nat Rev Cancer. 2005;5:761–72. doi: 10.1038/nrc1716. [DOI] [PubMed] [Google Scholar]
- 32.Amin K, Ip C, Jimenez L, Tyson C, Behrsing H. In vitro detection of differential and cell-specific hepatobiliary toxicity induced by geldanamycin and 17-allylaminogeldanamycin using dog liver slices. Toxicol Sci. 2005;87:442–50. doi: 10.1093/toxsci/kfi254. [DOI] [PubMed] [Google Scholar]
- 33.Schnur RC, Corman ML, Gallaschun RJ, Cooper BA, Dee MF, Doty JL, et al. erbB-2 oncogene inhibition by geldanamycin derivatives: synthesis, mechanism of action, and structure-activity relationships. Journal of Medicinal Chemistry. 1995;38:3813–20. doi: 10.1021/jm00019a011. [DOI] [PubMed] [Google Scholar]
- 34.Grenert JP, Sullivan WP, Fadden P, Haystead TA, Clark J, Mimnaugh E, et al. The amino-terminal domain of heat shock protein 90 (hsp90) that binds geldanamycin is an ATP/ADP switch domain that regulates hsp90 conformation. Journal of Biological Chemistry. 1997;272:23843–50. doi: 10.1074/jbc.272.38.23843. [DOI] [PubMed] [Google Scholar]
- 35.Zhang T, Hamza A, Cao X, Wang B, Yu S, Zhan CG, et al. A novel Hsp90 inhibitor to disrupt Hsp90/Cdc37 complex against pancreatic cancer cells. Mol Cancer Ther. 2008;7:162–70. doi: 10.1158/1535-7163.MCT-07-0484. [DOI] [PubMed] [Google Scholar]
- 36.Cao X, Bloomston M, Zhang T, Frankel WL, Jia G, Wang B, et al. Synergistic antipancreatic tumor effect by simultaneously targeting hypoxic cancer cells with HSP90 inhibitor and glycolysis inhibitor. Clin Cancer Res. 2008;14:1831–9. doi: 10.1158/1078-0432.CCR-07-1607. [DOI] [PubMed] [Google Scholar]
- 37.Chen WY, Chang FR, Huang ZY, Chen JH, Wu YC, Wu CC. Tubocapsenolide A, a novel withanolide, inhibits proliferation and induces apoptosis in MDA-MB-231 cells by thiol oxidation of heat shock proteins. J Biol Chem. 2008;283:17184–93. doi: 10.1074/jbc.M709447200. [DOI] [PubMed] [Google Scholar]
- 38.Blagosklonny MV. Hsp-90-associated oncoproteins: Multiple targets of geldanamycin and its analogs. Leukemia. 2002;16:455–62. doi: 10.1038/sj.leu.2402415. [DOI] [PubMed] [Google Scholar]
- 39.Banerji U, Walton M, Raynaud F, Grimshaw R, Kelland L, Valenti M, et al. Pharmacokinetic-pharmacodynamic relationships for the heat shock protein 90 molecular chaperone inhibitor 17-allylamino, 17-demethoxygeldanamycin in human ovarian cancer xenograft models. Clin Cancer Res. 2005;11:7023–32. doi: 10.1158/1078-0432.CCR-05-0518. [DOI] [PubMed] [Google Scholar]
- 40.Cumming RC, Andon NL, Haynes PA, Park M, Fischer WH, Schubert D. Protein disulfide bond formation in the cytoplasm during oxidative stress. J Biol Chem. 2004;279:21749–58. doi: 10.1074/jbc.M312267200. [DOI] [PubMed] [Google Scholar]
- 41.Nardai G, Sass B, Eber J, Orosz G, Csermely P. Reactive cysteines of the 90-kDa heat shock protein, Hsp90. Arch Biochem Biophys. 2000;384:59–67. doi: 10.1006/abbi.2000.2075. [DOI] [PubMed] [Google Scholar]
- 42.Martinez-Ruiz A, Villanueva L, Gonzalez de Orduna C, Lopez-Ferrer D, Higueras MA, Tarin C, et al. S-nitrosylation of Hsp90 promotes the inhibition of its ATPase and endothelial nitric oxide synthase regulatory activities. Proc Natl Acad Sci U S A. 2005;102:8525–30. doi: 10.1073/pnas.0407294102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Fuska J, Fuskova A, Rosazza JP, Nicholas AW. Novel cytotoxic and antitumor agents. IV. Withaferin A: relation of its structure to the in vitro cytotoxic effects on P388 cells. Neoplasma. 1984;31:31–6. [PubMed] [Google Scholar]
- 44.Oh JH, Lee TJ, Park JW, Kwon TK. Withaferin A inhibits iNOS expression and nitric oxide production by Akt inactivation and down-regulating LPS-induced activity of NF-kappaB in RAW 264.7 cells. Eur J Pharmacol. 2008;599:11–7. doi: 10.1016/j.ejphar.2008.09.017. [DOI] [PubMed] [Google Scholar]
- 45.Sullivan W, Stensgard B, Caucutt G, Bartha B, McMahon N, Alnemri ES, et al. Nucleotides and two functional states of hsp90. J Biol Chem. 1997;272:8007–12. doi: 10.1074/jbc.272.12.8007. [DOI] [PubMed] [Google Scholar]
- 46.Johnson JL, Toft DO. A novel chaperone complex for steroid receptors involving heat shock proteins, immunophilins, and p23. J Biol Chem. 1994;269:24989–93. [PubMed] [Google Scholar]
- 47.Hieronymus H, Lamb J, Ross KN, Peng XP, Clement C, Rodina A, et al. Gene expression signature-based chemical genomic prediction identifies a novel class of HSP90 pathway modulators. Cancer Cell. 2006;10:321–30. doi: 10.1016/j.ccr.2006.09.005. [DOI] [PubMed] [Google Scholar]
- 48.Yufu Y, Nishimura J, Nawata H. High constitutive expression of heat shock protein 90 alpha in human acute leukemia cells. Leuk Res. 1992;16:597–605. doi: 10.1016/0145-2126(92)90008-u. [DOI] [PubMed] [Google Scholar]
- 49.Ogata M, Naito Z, Tanaka S, Moriyama Y, Asano G. Overexpression and localization of heat shock proteins mRNA in pancreatic carcinoma. Journal of Nippon Medical School. 2000;67:177–85. doi: 10.1272/jnms.67.177. [DOI] [PubMed] [Google Scholar]
- 50.Plescia J, Salz W, Xia F, Pennati M, Zaffaroni N, Daidone MG, et al. Rational design of shepherdin, a novel anticancer agent. Cancer Cell. 2005;7:457–68. doi: 10.1016/j.ccr.2005.03.035. [DOI] [PubMed] [Google Scholar]
- 51.Nimmanapalli R, Fuino L, Bali P, Gasparetto M, Glozak M, Tao J, et al. Histone deacetylase inhibitor LAQ824 both lowers expression and promotes proteasomal degradation of Bcr-Abl and induces apoptosis of imatinib mesylate-sensitive or -refractory chronic myelogenous leukemia-blast crisis cells. Cancer Res. 2003;63:5126–35. [PubMed] [Google Scholar]
- 52.Deneke SM. Thiol-based antioxidants. Curr Top Cell Regul. 2000;36:151–80. doi: 10.1016/s0070-2137(01)80007-8. [DOI] [PubMed] [Google Scholar]
- 53.Fukuyo Y, Inoue M, Nakajima T, Higashikubo R, Horikoshi NT, Hunt C, et al. Oxidative stress plays a critical role in inactivating mutant BRAF by geldanamycin derivatives. Cancer Res. 2008;68:6324–30. doi: 10.1158/0008-5472.CAN-07-6602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Mandal AK, Lee P, Chen JA, Nillegoda N, Heller A, DiStasio S, et al. Cdc37 has distinct roles in protein kinase quality control that protect nascent chains from degradation and promote posttranslational maturation. J Cell Biol. 2007;176:319–28. doi: 10.1083/jcb.200604106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Stepanova L, Finegold M, DeMayo F, Schmidt EV, Harper JW. The oncoprotein kinase chaperone CDC37 functions as an oncogene in mice and collaborates with both c-myc and cyclin D1 in transformation of multiple tissues. Mol Cell Biol. 2000;20:4462–73. doi: 10.1128/mcb.20.12.4462-4473.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Pascale RM, Simile MM, Calvisi DF, Frau M, Muroni MR, Seddaiu MA, et al. Role of HSP90, CDC37, and CRM1 as modulators of P16(INK4A) activity in rat liver carcinogenesis and human liver cancer. Hepatology. 2005;42:1310–9. doi: 10.1002/hep.20962. [DOI] [PubMed] [Google Scholar]
- 57.MacLean M, Picard D. Cdc37 goes beyond Hsp90 and kinases. Cell Stress Chaperones. 2003;8:114–9. doi: 10.1379/1466-1268(2003)008<0114:cgbhak>2.0.co;2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Siligardi G, Hu B, Panaretou B, Piper PW, Pearl LH, Prodromou C. Co-chaperone regulation of conformational switching in the Hsp90 ATPase cycle. J Biol Chem. 2004;279:51989–98. doi: 10.1074/jbc.M410562200. [DOI] [PubMed] [Google Scholar]
- 59.Yokota Y, Bargagna-Mohan P, Ravindranath PP, Kim KB, Mohan R. Development of withaferin A analogs as probes of angiogenesis. Bioorg Med Chem Lett. 2006;16:2603–7. doi: 10.1016/j.bmcl.2006.02.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Liang MC, Bardhan S, Pace EA, Rosman D, Beutler JA, Porco JA, Jr, et al. Inhibition of transcription factor NF-kappaB signaling proteins IKKbeta and p65 through specific cysteine residues by epoxyquinone A monomer: correlation with its anti-cancer cell growth activity. Biochem Pharmacol. 2006;71:634–45. doi: 10.1016/j.bcp.2005.11.013. [DOI] [PubMed] [Google Scholar]
- 61.Brandt GE, Schmidt MD, Prisinzano TE, Blagg BS. Gedunin, a novel hsp90 inhibitor: semisynthesis of derivatives and preliminary structure-activity relationships. J Med Chem. 2008;51:6495–502. doi: 10.1021/jm8007486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lee JH, Koo TH, Yoon H, Jung HS, Jin HZ, Lee K, et al. Inhibition of NF-kappa B activation through targeting I kappa B kinase by celastrol, a quinone methide triterpenoid. Biochem Pharmacol. 2006;72:1311–21. doi: 10.1016/j.bcp.2006.08.014. [DOI] [PubMed] [Google Scholar]
- 63.Trott A, West JD, Klaic L, Westerheide SD, Silverman RB, Morimoto RI, et al. Activation of heat shock and antioxidant responses by the natural product celastrol: transcriptional signatures of a thiol-targeted molecule. Mol Biol Cell. 2008;19:1104–12. doi: 10.1091/mbc.E07-10-1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kuroyanagi M, Shibata K, Umehara K. Cell differentiation inducing steroids from Withania somnifera L. (DUN. ) Chem Pharm Bull (Tokyo) 1999;47:1646–9. [Google Scholar]
- 65.Mirjalili MH, Moyano E, Bonfill M, Cusido RM, Palazon J. Steroidal lactones from Withania somnifera, an ancient plant for novel medicine. Molecules. 2009;14:2373–93. doi: 10.3390/molecules14072373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Damu AG, Kuo PC, Su CR, Kuo TH, Chen TH, Bastow KF, et al. Isolation, structures, and structure - cytotoxic activity relationships of withanolides and physalins from Physalis angulata. J Nat Prod. 2007;70:1146–52. doi: 10.1021/np0701374. [DOI] [PubMed] [Google Scholar]
- 67.Lee SW, Pan MH, Chen CM, Chen ZT. Withangulatin I, a new cytotoxic withanolide from Physalis angulata. Chem Pharm Bull (Tokyo) 2008;56:234–6. doi: 10.1248/cpb.56.234. [DOI] [PubMed] [Google Scholar]
- 68.Tursunava RN, Maslennikova VA, Abubakirov NK. Withanolides in the vegetable kingdom. Chem Nat Comp. 1977;13:131–8. [Google Scholar]
- 69.Juang JK, Huang HW, Chen CM, Liu HJ. A new compound, withangulatin A, promotes type II DNA topoisomerase-mediated DNA damage. Biochem Biophys Res Commun. 1989;159:1128–34. doi: 10.1016/0006-291x(89)92226-2. [DOI] [PubMed] [Google Scholar]
- 70.Lee WC, Lin KY, Chen CM, Chen ZT, Liu HJ, Lai YK. Induction of heat-shock response and alterations of protein phosphorylation by a novel topoisomerase II inhibitor, withangulatin A, in 9L rat brain tumor cells. J Cell Physiol. 1991;149:66–76. doi: 10.1002/jcp.1041490110. [DOI] [PubMed] [Google Scholar]
- 71.Krueger JH, Walker GC. groEL and dnaK genes of Escherichia coli are induced by UV irradiation and nalidixic acid in an htpR+-dependent fashion. Proc Natl Acad Sci U S A. 1984;81:1499–503. doi: 10.1073/pnas.81.5.1499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Zaarur N, Gabai VL, Porco JA, Jr, Calderwood S, Sherman MY. Targeting heat shock response to sensitize cancer cells to proteasome and Hsp90 inhibitors. Cancer Res. 2006;66:1783–91. doi: 10.1158/0008-5472.CAN-05-3692. [DOI] [PubMed] [Google Scholar]