Abstract
Duplex DNA oligomer constructs (32 base pairs) were prepared that contained a single benzo[a]pyrene (BP) adduct at a specific deoxyadenosine or deoxyguanosine site in either one or both strands. These constructs were inserted into M13 replicative form viral DNA, and the DNA from progeny virus generated by transfection of Escherichia coli was examined by sequence analysis at the site of oligomer insertion. With nonalkylated constructs, and with constructs containing only one BP adduct, no sequence alterations were found in progeny viral DNAs. With constructs containing two BP adducts, one in each strand and closely spaced, some progeny DNAs showed the original oligomer sequence, whereas others exhibited large deletions and illegitimate (nonhomologous) recombination, both of which removed the damaged construct. Increasing the distance between BP adducts in the construct reduced the frequency of recombinant events. These sequence alterations occurred in both recA+ and recA- host cells. We speculate that the closely spaced adducts in opposite construct strands cause a rare distortion in DNA structure, which activates the recombinant machinery, and that mutagenic and carcinogenic agents other than polycyclic aromatic hydrocarbons may cause similar DNA distortions, which induce illegitimate recombination.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Agarwal K. L., Riftina F. Chemical synthesis of a self-complementary octanucleotide, dG-G-T-T-A-A-C-C by a modified triester method. Nucleic Acids Res. 1978 Aug;5(8):2809–2823. doi: 10.1093/nar/5.8.2809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson P. Anecdotal, historical and critical commentaries on genetics twenty years of illegitimate recombination. Genetics. 1987 Apr;115(4):581–583. doi: 10.1093/genetics/115.4.581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birnboim H. C., Doly J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 1979 Nov 24;7(6):1513–1523. doi: 10.1093/nar/7.6.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boles T. C., Hogan M. E. High-resolution mapping of carcinogen binding sites on DNA. Biochemistry. 1986 May 20;25(10):3039–3043. doi: 10.1021/bi00358a045. [DOI] [PubMed] [Google Scholar]
- Duesberg P. H. Cancer genes: rare recombinants instead of activated oncogenes (a review). Proc Natl Acad Sci U S A. 1987 Apr;84(8):2117–2124. doi: 10.1073/pnas.84.8.2117. [DOI] [PMC free article] [PubMed] [Google Scholar]
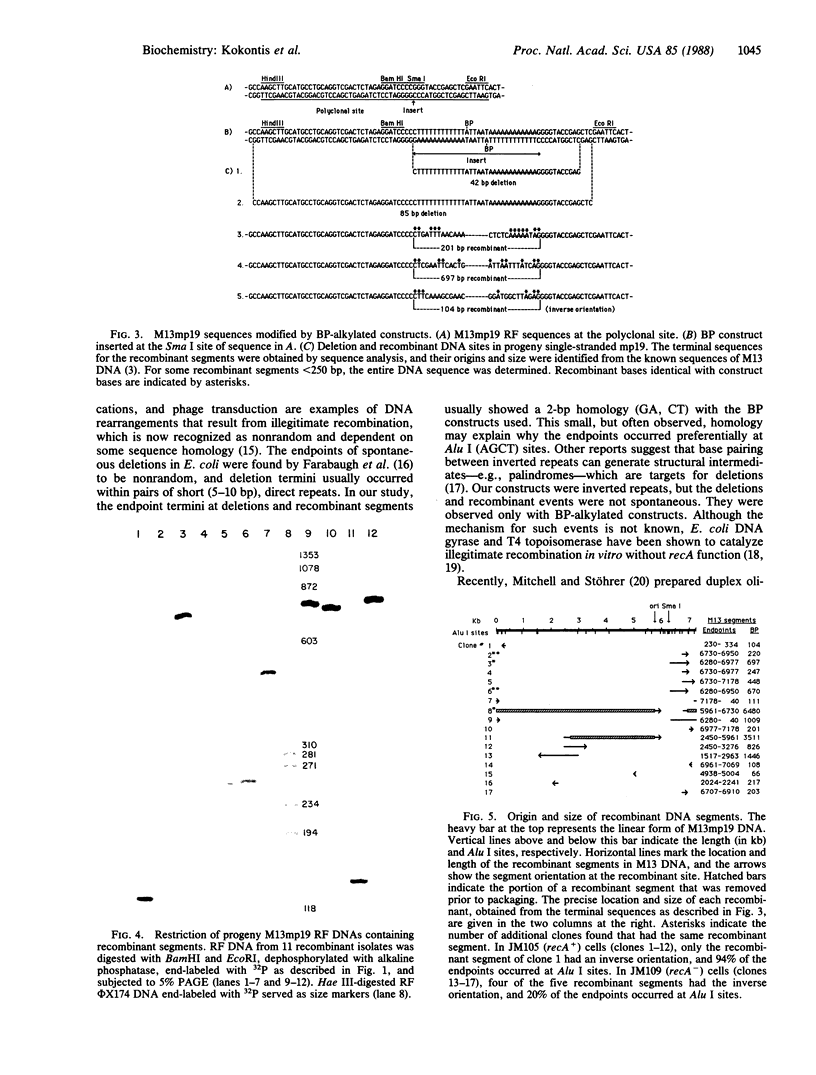
- Farabaugh P. J., Schmeissner U., Hofer M., Miller J. H. Genetic studies of the lac repressor. VII. On the molecular nature of spontaneous hotspots in the lacI gene of Escherichia coli. J Mol Biol. 1978 Dec 25;126(4):847–857. doi: 10.1016/0022-2836(78)90023-2. [DOI] [PubMed] [Google Scholar]
- Franklin N. C. Extraordinary recombinational events in Escherichia coli. Their independence of the rec+ function. Genetics. 1967 Apr;55(4):699–707. doi: 10.1093/genetics/55.4.699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glickman B. W., Ripley L. S. Structural intermediates of deletion mutagenesis: a role for palindromic DNA. Proc Natl Acad Sci U S A. 1984 Jan;81(2):512–516. doi: 10.1073/pnas.81.2.512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harvey R. G., Cho H. Efficient resolution of the dihydrodiol derivatives of benzo[a]pyrene by high-pressure liquid chromatography of the related (-)-dimenthoxyacetates. Anal Biochem. 1977 Jun;80(2):540–546. doi: 10.1016/0003-2697(77)90677-7. [DOI] [PubMed] [Google Scholar]
- Heidecker G., Messing J., Gronenborn B. A versatile primer for DNA sequencing in the M13mp2 cloning system. Gene. 1980 Jun;10(1):69–73. doi: 10.1016/0378-1119(80)90145-6. [DOI] [PubMed] [Google Scholar]
- Ikeda H., Aoki K., Naito A. Illegitimate recombination mediated in vitro by DNA gyrase of Escherichia coli: structure of recombinant DNA molecules. Proc Natl Acad Sci U S A. 1982 Jun;79(12):3724–3728. doi: 10.1073/pnas.79.12.3724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikeda H. Bacteriophage T4 DNA topoisomerase mediates illegitimate recombination in vitro. Proc Natl Acad Sci U S A. 1986 Feb;83(4):922–926. doi: 10.1073/pnas.83.4.922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LEVAN A. Chromosomes in cancer tissue. Ann N Y Acad Sci. 1956 Mar 14;63(5):774–792. doi: 10.1111/j.1749-6632.1956.tb50892.x. [DOI] [PubMed] [Google Scholar]
- Messing J. New M13 vectors for cloning. Methods Enzymol. 1983;101:20–78. doi: 10.1016/0076-6879(83)01005-8. [DOI] [PubMed] [Google Scholar]
- Miller J. H. Carcinogens induce targeted mutations in Escherichia coli. Cell. 1982 Nov;31(1):5–7. doi: 10.1016/0092-8674(82)90398-1. [DOI] [PubMed] [Google Scholar]
- Mitchell N., Stöhrer G. Mutagenesis originating in site-specific DNA damage. J Mol Biol. 1986 Sep 20;191(2):177–180. doi: 10.1016/0022-2836(86)90254-8. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Straub K. M., Meehan T., Burlingame A. L., Calvin M. Identification of the major adducts formed by reaction of benzo(a)pyrene diol epoxide with DNA in vitro. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5285–5289. doi: 10.1073/pnas.74.12.5285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker G. C. Mutagenesis and inducible responses to deoxyribonucleic acid damage in Escherichia coli. Microbiol Rev. 1984 Mar;48(1):60–93. doi: 10.1128/mr.48.1.60-93.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei S. J., Desai S. M., Harvey R. G., Weiss S. B. Use of short DNA oligonucleotides for determination of DNA sequence modifications induced by benzo[a]pyrene diol epoxide. Proc Natl Acad Sci U S A. 1984 Oct;81(19):5936–5940. doi: 10.1073/pnas.81.19.5936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanisch-Perron C., Vieira J., Messing J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene. 1985;33(1):103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]