Abstract
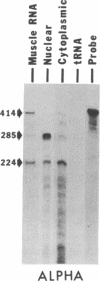
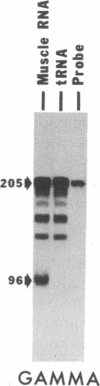
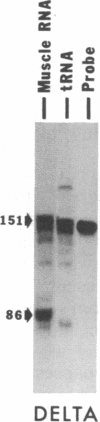
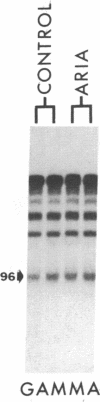
A 42-kDa glycoprotein isolated from chicken brain, referred to as acetylcholine receptor-inducing activity (ARIA), that stimulates the rate of incorporation of acetylcholine receptors into the surface of chicken myotubes may play a role in the nerve-induced accumulation of receptors at developing neuromuscular synapses. Using nuclease-protection assays, we have found that ARIA causes a 2- to 16-fold increase in the level of mRNA encoding the alpha subunit of the receptor, with little or no change in the levels of gamma- and delta-subunit messengers. ARIA also increases the amount of a putative nuclear precursor of alpha-subunit mRNA, consistent with an activation of gene transcription. These results suggest that the concentration of alpha subunit may limit the rate of biosynthesis of the acetylcholine receptors in chicken myotubes. They also indicate that neuronal factors can regulate the expression of receptor subunit genes in a selective manner. Tetrodotoxin, 8-bromo-cAMP, and forskolin also increase the amount of alpha-subunit mRNA, with little change in the amount of gamma- and delta-subunit mRNAs. Unlike, ARIA, however, these agents have little effect on the concentration of the alpha-subunit nuclear precursor.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson M. J., Cohen M. W. Nerve-induced and spontaneous redistribution of acetylcholine receptors on cultured muscle cells. J Physiol. 1977 Jul;268(3):757–773. doi: 10.1113/jphysiol.1977.sp011880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ballivet M., Nef P., Stalder R., Fulpius B. Genomic sequences encoding the alpha-subunit of acetylcholine receptor are conserved in evolution. Cold Spring Harb Symp Quant Biol. 1983;48(Pt 1):83–87. doi: 10.1101/sqb.1983.048.01.011. [DOI] [PubMed] [Google Scholar]
- Bauer H. C., Daniels M. P., Pudimat P. A., Jacques L., Sugiyama H., Christian C. N. Characterization and partial purification of a neuronal factor which increases acetylcholine receptor aggregation on cultured muscle cells. Brain Res. 1981 Mar 30;209(2):395–404. doi: 10.1016/0006-8993(81)90161-x. [DOI] [PubMed] [Google Scholar]
- Beam K. G., Caldwell J. H., Campbell D. T. Na channels in skeletal muscle concentrated near the neuromuscular junction. Nature. 1985 Feb 14;313(6003):588–590. doi: 10.1038/313588a0. [DOI] [PubMed] [Google Scholar]
- Betz H., Changeux J. P. Regulation of muscle acetylcholine receptor synthesis in vitro by cyclic nucleotide derivatives. Nature. 1979 Apr 19;278(5706):749–752. doi: 10.1038/278749a0. [DOI] [PubMed] [Google Scholar]
- Bloch R. J., Hall Z. W. Cytoskeletal components of the vertebrate neuromuscular junction: vinculin, alpha-actinin, and filamin. J Cell Biol. 1983 Jul;97(1):217–223. doi: 10.1083/jcb.97.1.217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buc-Caron M. H., Nystrom P., Fischbach G. D. Induction of acetylcholine receptor synthesis and aggregation: partial purification of low-molecular-weight activity. Dev Biol. 1983 Feb;95(2):378–386. doi: 10.1016/0012-1606(83)90039-8. [DOI] [PubMed] [Google Scholar]
- Buonanno A., Mudd J., Shah V., Merlie J. P. A universal oligonucleotide probe for acetylcholine receptor genes. Selection and sequencing of cDNA clones for the mouse muscle beta subunit. J Biol Chem. 1986 Dec 15;261(35):16451–16458. [PubMed] [Google Scholar]
- Cathala G., Savouret J. F., Mendez B., West B. L., Karin M., Martial J. A., Baxter J. D. A method for isolation of intact, translationally active ribonucleic acid. DNA. 1983;2(4):329–335. doi: 10.1089/dna.1983.2.329. [DOI] [PubMed] [Google Scholar]
- Cohen S. A., Fischbach G. D. Regulation of muscle acetylcholine sensitivity by muscle activity in cell culture. Science. 1973 Jul 6;181(4094):76–78. doi: 10.1126/science.181.4094.76. [DOI] [PubMed] [Google Scholar]
- Devreotes P. N., Fambrough D. M. Acetylcholine receptor turnover in membranes of developing muscle fibers. J Cell Biol. 1975 May;65(2):335–358. doi: 10.1083/jcb.65.2.335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans S., Goldman D., Heinemann S., Patrick J. Muscle acetylcholine receptor biosynthesis. Regulation by transcript availability. J Biol Chem. 1987 Apr 5;262(10):4911–4916. [PMC free article] [PubMed] [Google Scholar]
- Fischbach G. D., Frank E., Jessell T. M., Rubin L. L., Schuetze S. M. Accumulation of acetylcholine receptors and acetylcholinesterase at newly formed nerve-muscle synapses. Pharmacol Rev. 1978 Dec;30(4):411–428. [PubMed] [Google Scholar]
- Fontaine B., Klarsfeld A., Changeux J. P. Calcitonin gene-related peptide and muscle activity regulate acetylcholine receptor alpha-subunit mRNA levels by distinct intracellular pathways. J Cell Biol. 1987 Sep;105(3):1337–1342. doi: 10.1083/jcb.105.3.1337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Froehner S. C., Gulbrandsen V., Hyman C., Jeng A. Y., Neubig R. R., Cohen J. B. Immunofluorescence localization at the mammalian neuromuscular junction of the Mr 43,000 protein of Torpedo postsynaptic membranes. Proc Natl Acad Sci U S A. 1981 Aug;78(8):5230–5234. doi: 10.1073/pnas.78.8.5230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Godfrey E. W., Nitkin R. M., Wallace B. G., Rubin L. L., McMahan U. J. Components of Torpedo electric organ and muscle that cause aggregation of acetylcholine receptors on cultured muscle cells. J Cell Biol. 1984 Aug;99(2):615–627. doi: 10.1083/jcb.99.2.615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jessell T. M., Siegel R. E., Fischbach G. D. Induction of acetylcholine receptors on cultured skeletal muscle by a factor extracted from brain and spinal cord. Proc Natl Acad Sci U S A. 1979 Oct;76(10):5397–5401. doi: 10.1073/pnas.76.10.5397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klarsfeld A., Changeux J. P. Activity regulates the levels of acetylcholine receptor alpha-subunit mRNA in cultured chicken myotubes. Proc Natl Acad Sci U S A. 1985 Jul;82(13):4558–4562. doi: 10.1073/pnas.82.13.4558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knaack D., Shen I., Salpeter M. M., Podleski T. R. Selective effects of ascorbic acid on acetylcholine receptor number and distribution. J Cell Biol. 1986 Mar;102(3):795–802. doi: 10.1083/jcb.102.3.795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuromi H., Brass B., Kidokoro Y. Formation of acetylcholine receptor clusters at neuromuscular junction in Xenopus cultures. Dev Biol. 1985 May;109(1):165–176. doi: 10.1016/0012-1606(85)90357-4. [DOI] [PubMed] [Google Scholar]
- Markelonis G. J., Oh T. H., Eldefrawi M. E., Guth L. Sciatin: a myotrophic protein increases the number of acetylcholine receptors and receptor clusters in cultured skeletal muscle. Dev Biol. 1982 Feb;89(2):353–361. doi: 10.1016/0012-1606(82)90324-4. [DOI] [PubMed] [Google Scholar]
- Melton D. A., Krieg P. A., Rebagliati M. R., Maniatis T., Zinn K., Green M. R. Efficient in vitro synthesis of biologically active RNA and RNA hybridization probes from plasmids containing a bacteriophage SP6 promoter. Nucleic Acids Res. 1984 Sep 25;12(18):7035–7056. doi: 10.1093/nar/12.18.7035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merlie J. P., Smith M. M. Synthesis and assembly of acetylcholine receptor, a multisubunit membrane glycoprotein. J Membr Biol. 1986;91(1):1–10. doi: 10.1007/BF01870209. [DOI] [PubMed] [Google Scholar]
- Mishina M., Takai T., Imoto K., Noda M., Takahashi T., Numa S., Methfessel C., Sakmann B. Molecular distinction between fetal and adult forms of muscle acetylcholine receptor. Nature. 1986 May 22;321(6068):406–411. doi: 10.1038/321406a0. [DOI] [PubMed] [Google Scholar]
- Nef P., Mauron A., Stalder R., Alliod C., Ballivet M. Structure linkage, and sequence of the two genes encoding the delta and gamma subunits of the nicotinic acetylcholine receptor. Proc Natl Acad Sci U S A. 1984 Dec;81(24):7975–7979. doi: 10.1073/pnas.81.24.7975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neugebauer K., Salpeter M. M., Podleski T. R. Differential responses of L5 and rat primary muscle cells to factors in rat brain extract. Brain Res. 1985 Oct 28;346(1):58–69. doi: 10.1016/0006-8993(85)91094-7. [DOI] [PubMed] [Google Scholar]
- Olson E. N., Glaser L., Merlie J. P., Lindstrom J. Expression of acetylcholine receptor alpha-subunit mRNA during differentiation of the BC3H1 muscle cell line. J Biol Chem. 1984 Mar 10;259(5):3330–3336. [PubMed] [Google Scholar]
- Olson E. N., Glaser L., Merlie J. P., Sebanne R., Lindstrom J. Regulation of surface expression of acetylcholine receptors in response to serum and cell growth in the BC3H1 muscle cell line. J Biol Chem. 1983 Nov 25;258(22):13946–13953. [PubMed] [Google Scholar]
- Role L. W., Matossian V. R., O'Brien R. J., Fischbach G. D. On the mechanism of acetylcholine receptor accumulation at newly formed synapses on chick myotubes. J Neurosci. 1985 Aug;5(8):2197–2204. doi: 10.1523/JNEUROSCI.05-08-02197.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salpeter M. M. Electron microscope radioautography as a quantitative tool in enzyme cytochemistry. II. The distribution of DFP-reactive sties at motor endplates of a vertebrate twitch muscle. J Cell Biol. 1969 Jul;42(1):122–134. doi: 10.1083/jcb.42.1.122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salpeter M. M., Loring R. H. Nicotinic acetylcholine receptors in vertebrate muscle: properties, distribution and neural control. Prog Neurobiol. 1985;25(4):297–325. doi: 10.1016/0301-0082(85)90018-8. [DOI] [PubMed] [Google Scholar]
- Sanes J. R., Chiu A. Y. The basal lamina of the neuromuscular junction. Cold Spring Harb Symp Quant Biol. 1983;48(Pt 2):667–678. doi: 10.1101/sqb.1983.048.01.070. [DOI] [PubMed] [Google Scholar]
- Schuetze S. M., Role L. W. Developmental regulation of nicotinic acetylcholine receptors. Annu Rev Neurosci. 1987;10:403–457. doi: 10.1146/annurev.ne.10.030187.002155. [DOI] [PubMed] [Google Scholar]
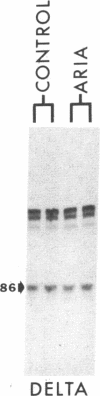
- Shieh B. H., Ballivet M., Schmidt J. Quantitation of an alpha subunit splicing intermediate: evidence for transcriptional activation in the control of acetylcholine receptor expression in denervated chick skeletal muscle. J Cell Biol. 1987 May;104(5):1337–1341. doi: 10.1083/jcb.104.5.1337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Usdin T. B., Fischbach G. D. Purification and characterization of a polypeptide from chick brain that promotes the accumulation of acetylcholine receptors in chick myotubes. J Cell Biol. 1986 Aug;103(2):493–507. doi: 10.1083/jcb.103.2.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziskind-Conhaim L., Geffen I., Hall Z. W. Redistribution of acetylcholine receptors on developing rat myotubes. J Neurosci. 1984 Sep;4(9):2346–2349. doi: 10.1523/JNEUROSCI.04-09-02346.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]