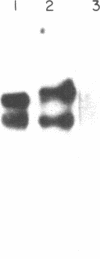
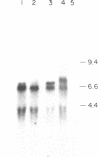
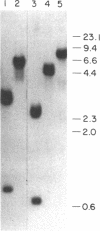
Abstract
Cytotactin is an extracellular glycoprotein that influences neuron-glia interactions. It has been shown to appear in multiple forms that are differentially expressed in neural and non-neural tissues during vertebrate development. We report here the isolation and characterization of a cytotactin cDNA clone (lambda C801) that encodes 933 amino acids, equivalent to about half of a cytotactin polypeptide. Clone lambda C801 is an authentic cytotactin cDNA: it encodes a polypeptide that reacts with a monoclonal anti-cytotactin antibody and its deduced amino acid sequence is identical for 15 amino acids to the directly determined sequence of a CNBr fragment that reacted with the same antibody. Southern blot analyses with fragments of lambda C801 suggested that there may be only one cytotactin gene, but RNA transfer blots detected multiple mRNAs ranging in size from 6.5 to 8.0 kilobases. An 8.0-kilobase message and a Mr 240,000 cytotactin polypeptide were present in embryonic gizzard but not brain, while a 7.2-kilobase message and a Mr 220,000 polypeptide were present in brain but not gizzard. These results indicate that differential splicing of primary transcripts of the cytotactin gene yields various site-specific polypeptides. Sequence analyses of lambda C801 indicated that it specifies a region with extensive similarities to other proteins: the sequence begins with four consecutive epidermal growth factor-like repeats that are followed by eight segments that closely resemble each other and the type III repeats in fibronectin, and it ends with a 66 amino acid sequence similar to part of the beta and gamma chains of fibrinogen. One fibronectin-like repeat contains a single Arg-Gly-Asp sequence. The similarities with all three of these apparently unrelated proteins are extensive, suggesting that cytotactin has an evolutionary and possibly a functional relationship to each.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chiquet-Ehrismann R., Mackie E. J., Pearson C. A., Sakakura T. Tenascin: an extracellular matrix protein involved in tissue interactions during fetal development and oncogenesis. Cell. 1986 Oct 10;47(1):131–139. doi: 10.1016/0092-8674(86)90374-0. [DOI] [PubMed] [Google Scholar]
- Chuong C. M., Crossin K. L., Edelman G. M. Sequential expression and differential function of multiple adhesion molecules during the formation of cerebellar cortical layers. J Cell Biol. 1987 Feb;104(2):331–342. doi: 10.1083/jcb.104.2.331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crossin K. L., Hoffman S., Grumet M., Thiery J. P., Edelman G. M. Site-restricted expression of cytotactin during development of the chicken embryo. J Cell Biol. 1986 May;102(5):1917–1930. doi: 10.1083/jcb.102.5.1917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doolittle R. F. Fibrinogen and fibrin. Annu Rev Biochem. 1984;53:195–229. doi: 10.1146/annurev.bi.53.070184.001211. [DOI] [PubMed] [Google Scholar]
- Edelman G. M. CAMs and Igs: cell adhesion and the evolutionary origins of immunity. Immunol Rev. 1987 Dec;100:11–45. doi: 10.1111/j.1600-065x.1987.tb00526.x. [DOI] [PubMed] [Google Scholar]
- Edelman G. M. Cell adhesion molecules in the regulation of animal form and tissue pattern. Annu Rev Cell Biol. 1986;2:81–116. doi: 10.1146/annurev.cb.02.110186.000501. [DOI] [PubMed] [Google Scholar]
- Erickson H. P., Taylor H. C. Hexabrachion proteins in embryonic chicken tissues and human tumors. J Cell Biol. 1987 Sep;105(3):1387–1394. doi: 10.1083/jcb.105.3.1387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grumet M., Hoffman S., Crossin K. L., Edelman G. M. Cytotactin, an extracellular matrix protein of neural and non-neural tissues that mediates glia-neuron interaction. Proc Natl Acad Sci U S A. 1985 Dec;82(23):8075–8079. doi: 10.1073/pnas.82.23.8075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffman S., Edelman G. M. A proteoglycan with HNK-1 antigenic determinants is a neuron-associated ligand for cytotactin. Proc Natl Acad Sci U S A. 1987 Apr;84(8):2523–2527. doi: 10.1073/pnas.84.8.2523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hynes R. O., Yamada K. M. Fibronectins: multifunctional modular glycoproteins. J Cell Biol. 1982 Nov;95(2 Pt 1):369–377. doi: 10.1083/jcb.95.2.369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornblihtt A. R., Vibe-Pedersen K., Baralle F. E. Human fibronectin: molecular cloning evidence for two mRNA species differing by an internal segment coding for a structural domain. EMBO J. 1984 Jan;3(1):221–226. doi: 10.1002/j.1460-2075.1984.tb01787.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messing J. New M13 vectors for cloning. Methods Enzymol. 1983;101:20–78. doi: 10.1016/0076-6879(83)01005-8. [DOI] [PubMed] [Google Scholar]
- Patthy L. Evolution of the proteases of blood coagulation and fibrinolysis by assembly from modules. Cell. 1985 Jul;41(3):657–663. doi: 10.1016/s0092-8674(85)80046-5. [DOI] [PubMed] [Google Scholar]
- Petersen T. E., Thøgersen H. C., Skorstengaard K., Vibe-Pedersen K., Sahl P., Sottrup-Jensen L., Magnusson S. Partial primary structure of bovine plasma fibronectin: three types of internal homology. Proc Natl Acad Sci U S A. 1983 Jan;80(1):137–141. doi: 10.1073/pnas.80.1.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierschbacher M. D., Ruoslahti E. Cell attachment activity of fibronectin can be duplicated by small synthetic fragments of the molecule. Nature. 1984 May 3;309(5963):30–33. doi: 10.1038/309030a0. [DOI] [PubMed] [Google Scholar]
- Staden R. An interactive graphics program for comparing and aligning nucleic acid and amino acid sequences. Nucleic Acids Res. 1982 May 11;10(9):2951–2961. doi: 10.1093/nar/10.9.2951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabor S., Richardson C. C. DNA sequence analysis with a modified bacteriophage T7 DNA polymerase. Proc Natl Acad Sci U S A. 1987 Jul;84(14):4767–4771. doi: 10.1073/pnas.84.14.4767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan S. S., Crossin K. L., Hoffman S., Edelman G. M. Asymmetric expression in somites of cytotactin and its proteoglycan ligand is correlated with neural crest cell distribution. Proc Natl Acad Sci U S A. 1987 Nov;84(22):7977–7981. doi: 10.1073/pnas.84.22.7977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tuszynski G. P., Rothman V., Murphy A., Siegler K., Smith L., Smith S., Karczewski J., Knudsen K. A. Thrombospondin promotes cell-substratum adhesion. Science. 1987 Jun 19;236(4808):1570–1573. doi: 10.1126/science.2438772. [DOI] [PubMed] [Google Scholar]
- Watt K. W., Takagi T., Doolittle R. F. Amino acid sequence of the beta chain of human fibrinogen: homology with the gamma chain. Proc Natl Acad Sci U S A. 1978 Apr;75(4):1731–1735. doi: 10.1073/pnas.75.4.1731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wharton K. A., Johansen K. M., Xu T., Artavanis-Tsakonas S. Nucleotide sequence from the neurogenic locus notch implies a gene product that shares homology with proteins containing EGF-like repeats. Cell. 1985 Dec;43(3 Pt 2):567–581. doi: 10.1016/0092-8674(85)90229-6. [DOI] [PubMed] [Google Scholar]
- Wilbur W. J., Lipman D. J. Rapid similarity searches of nucleic acid and protein data banks. Proc Natl Acad Sci U S A. 1983 Feb;80(3):726–730. doi: 10.1073/pnas.80.3.726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada K. M. Cell surface interactions with extracellular materials. Annu Rev Biochem. 1983;52:761–799. doi: 10.1146/annurev.bi.52.070183.003553. [DOI] [PubMed] [Google Scholar]
- Young R. A., Davis R. W. Yeast RNA polymerase II genes: isolation with antibody probes. Science. 1983 Nov 18;222(4625):778–782. doi: 10.1126/science.6356359. [DOI] [PubMed] [Google Scholar]