Abstract
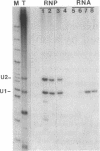
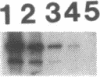
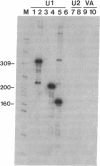
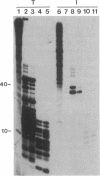
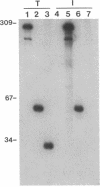
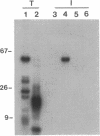
An autoantibody from a patient with lupus-overlap syndrome was found to bind a specific region of U1 RNA. By using RNA sequence analysis, immunoprecipitation, and competition experiments with in vitro synthesized fragments of U1 RNA, a region of 40 nucleotides representing a stem-loop secondary structure was found to be an immunoreactive domain. This antibody recognized a conformational epitope because neither the RNA stem nor the RNA loop alone was immunoprecipitable. Antisense U1 RNA, U1 DNA, and other small RNAs were not reactive with the antibody. While the origins of nucleic acid-binding antibodies are unknown, this RNA-specific autoantibody probably originated by direct presentation to the immune system or as an anti-idiotype against a more common U1 small nuclear ribonucleoprotein-specific autoantibody. Thus, these findings have implications for the mechanisms of autoimmune recognition and provide an immunological approach to probing RNA structure and protein-RNA interactions.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Branlant C., Krol A., Ebel J. P., Gallinaro H., Lazar E., Jacob M. The conformation of chicken, rat and human U1A RNAs in solution. Nucleic Acids Res. 1981 Feb 25;9(4):841–858. doi: 10.1093/nar/9.4.841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bunn C. C., Bernstein R. M., Mathews M. B. Autoantibodies against alanyl-tRNA synthetase and tRNAAla coexist and are associated with myositis. J Exp Med. 1986 May 1;163(5):1281–1291. doi: 10.1084/jem.163.5.1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carey J., Cameron V., de Haseth P. L., Uhlenbeck O. C. Sequence-specific interaction of R17 coat protein with its ribonucleic acid binding site. Biochemistry. 1983 May 24;22(11):2601–2610. doi: 10.1021/bi00280a002. [DOI] [PubMed] [Google Scholar]
- Chambers J. C., Kurilla M. G., Keene J. D. Association between the 7 S RNA and the lupus La protein varies among cell types. J Biol Chem. 1983 Oct 10;258(19):11438–11441. [PubMed] [Google Scholar]
- Eilat D., Ben Sasson S. A., Laskov R. A ribonucleic acid-specific antibody produced during autoimmune disease: evidence for nucleotide sequence specificity. Eur J Immunol. 1980 Nov;10(11):841–845. doi: 10.1002/eji.1830101108. [DOI] [PubMed] [Google Scholar]
- Epstein P., Reddy R., Busch H. Site-specific cleavage by T1 RNase of U-1 RNA in u-1 ribonucleoprotein particles. Proc Natl Acad Sci U S A. 1981 Mar;78(3):1562–1566. doi: 10.1073/pnas.78.3.1562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frappier L., Price G. B., Martin R. G., Zannis-Hadjopoulos M. Monoclonal antibodies to cruciform DNA structures. J Mol Biol. 1987 Feb 20;193(4):751–758. doi: 10.1016/0022-2836(87)90356-1. [DOI] [PubMed] [Google Scholar]
- Fresco L. D., Kurilla M. G., Keene J. D. Rapid inhibition of processing and assembly of small nuclear ribonucleoproteins after infection with vesicular stomatitis virus. Mol Cell Biol. 1987 Mar;7(3):1148–1155. doi: 10.1128/mcb.7.3.1148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hernandez N. Formation of the 3' end of U1 snRNA is directed by a conserved sequence located downstream of the coding region. EMBO J. 1985 Jul;4(7):1827–1837. doi: 10.1002/j.1460-2075.1985.tb03857.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isenberg D. A., Shoenfeld Y., Madaio M. P., Rauch J., Reichlin M., Stollar B. D., Schwartz R. S. Anti-DNA antibody idiotypes in systemic lupus erythematosus. Lancet. 1984 Aug 25;2(8400):417–422. doi: 10.1016/s0140-6736(84)92904-0. [DOI] [PubMed] [Google Scholar]
- Jerne N. K., Roland J., Cazenave P. A. Recurrent idiotopes and internal images. EMBO J. 1982;1(2):243–247. doi: 10.1002/j.1460-2075.1982.tb01154.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jerne N. K. Towards a network theory of the immune system. Ann Immunol (Paris) 1974 Jan;125C(1-2):373–389. [PubMed] [Google Scholar]
- Johnston M. I., Stollar B. D., Torrence P. F., Witkop B. Structural features of double-stranded polyribonucleotides required for immunological specificity and interferon induction. Proc Natl Acad Sci U S A. 1975 Nov;72(11):4564–4568. doi: 10.1073/pnas.72.11.4564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keene J. D., Schubert M., Lazzarini R. A., Rosenberg M. Nucleotide sequence homology at the 3' termini of RNA from vesicular stomatitis virus and its defective interfering particles. Proc Natl Acad Sci U S A. 1978 Jul;75(7):3225–3229. doi: 10.1073/pnas.75.7.3225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keene J. D., Schubert M., Lazzarini R. A. Terminal sequences of vesicular stomatitis virus RNA are both complementary and conserved. J Virol. 1979 Oct;32(1):167–174. doi: 10.1128/jvi.32.1.167-174.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krainer A. R., Maniatis T. Multiple factors including the small nuclear ribonucleoproteins U1 and U2 are necessary for pre-mRNA splicing in vitro. Cell. 1985 Oct;42(3):725–736. doi: 10.1016/0092-8674(85)90269-7. [DOI] [PubMed] [Google Scholar]
- Krämer A., Keller W., Appel B., Lührmann R. The 5' terminus of the RNA moiety of U1 small nuclear ribonucleoprotein particles is required for the splicing of messenger RNA precursors. Cell. 1984 Aug;38(1):299–307. doi: 10.1016/0092-8674(84)90551-8. [DOI] [PubMed] [Google Scholar]
- Lerner M. R., Steitz J. A. Snurps and scyrps. Cell. 1981 Aug;25(2):298–300. doi: 10.1016/0092-8674(81)90047-7. [DOI] [PubMed] [Google Scholar]
- Möller A., Gabriels J. E., Lafer E. M., Nordheim A., Rich A., Stollar B. D. Monoclonal antibodies recognize different parts of Z-DNA. J Biol Chem. 1982 Oct 25;257(20):12081–12085. [PubMed] [Google Scholar]
- Nordheim A., Pardue M. L., Lafer E. M., Möller A., Stollar B. D., Rich A. Antibodies to left-handed Z-DNA bind to interband regions of Drosophila polytene chromosomes. Nature. 1981 Dec 3;294(5840):417–422. doi: 10.1038/294417a0. [DOI] [PubMed] [Google Scholar]
- Oudin J., Cazenave P. A. Similar idiotypic specificities in immunoglobulin fractions with different antibody functions or even without detectable antibody function. Proc Natl Acad Sci U S A. 1971 Oct;68(10):2616–2620. doi: 10.1073/pnas.68.10.2616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Padgett R. A., Mount S. M., Steitz J. A., Sharp P. A. Splicing of messenger RNA precursors is inhibited by antisera to small nuclear ribonucleoprotein. Cell. 1983 Nov;35(1):101–107. doi: 10.1016/0092-8674(83)90212-x. [DOI] [PubMed] [Google Scholar]
- Peattie D. A., Douthwaite S., Garrett R. A., Noller H. F. A "bulged" double helix in a RNA-protein contact site. Proc Natl Acad Sci U S A. 1981 Dec;78(12):7331–7335. doi: 10.1073/pnas.78.12.7331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pettersson I., Hinterberger M., Mimori T., Gottlieb E., Steitz J. A. The structure of mammalian small nuclear ribonucleoproteins. Identification of multiple protein components reactive with anti-(U1)ribonucleoprotein and anti-Sm autoantibodies. J Biol Chem. 1984 May 10;259(9):5907–5914. [PubMed] [Google Scholar]
- Pisetsky D. S., Hoch S. O., Klatt C. L., O'Donnell M. A., Keene J. D. Specificity and idiotypic analysis of a monoclonal anti-Sm antibody with anti-DNA activity. J Immunol. 1985 Dec;135(6):4080–4085. [PubMed] [Google Scholar]
- Rauch J., Massicotte H., Tannenbaum H. Hybridoma anti-DNA autoantibodies from patients with rheumatoid arthritis and systemic lupus erythematosus demonstrate similar nucleic acid binding characteristics. J Immunol. 1985 Jan;134(1):180–186. [PubMed] [Google Scholar]
- Rauch J., Tannenbaum H., Stollar B. D., Schwartz R. S. Monoclonal anti-cardiolipin antibodies bind to DNA. Eur J Immunol. 1984 Jun;14(6):529–534. doi: 10.1002/eji.1830140609. [DOI] [PubMed] [Google Scholar]
- Romaniuk P. J., Lowary P., Wu H. N., Stormo G., Uhlenbeck O. C. RNA binding site of R17 coat protein. Biochemistry. 1987 Mar 24;26(6):1563–1568. doi: 10.1021/bi00380a011. [DOI] [PubMed] [Google Scholar]
- Stollar B. D. Antibodies to DNA. CRC Crit Rev Biochem. 1986;20(1):1–36. doi: 10.3109/10409238609115899. [DOI] [PubMed] [Google Scholar]
- Stollar B. D., Zon G., Pastor R. W. A recognition site on synthetic helical oligonucleotides for monoclonal anti-native DNA autoantibody. Proc Natl Acad Sci U S A. 1986 Jun;83(12):4469–4473. doi: 10.1073/pnas.83.12.4469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan E. M. Autoantibodies to nuclear antigens (ANA): their immunobiology and medicine. Adv Immunol. 1982;33:167–240. doi: 10.1016/s0065-2776(08)60836-6. [DOI] [PubMed] [Google Scholar]
- Wilusz J., Keene J. D. Autoantibodies specific for U1 RNA and initiator methionine tRNA. J Biol Chem. 1986 Apr 25;261(12):5467–5472. [PubMed] [Google Scholar]