Abstract
Background and Aims
Because plants protect each other from wind, stand density affects both the light climate and the amount of mechanical stress experienced by plants. But the potential interactive effects of mechanical stress and canopy shading on plant growth have rarely been investigated and never in stoloniferous plants which, due to their creeping growth form, can be expected to respond differently to these factors than erect plants.
Methods
Plants of ten genotypes of the stoloniferous species Potentilla reptans were subjected to two levels of mechanical stress (0 or 40 daily flexures) and two levels of spectral shading (15 % of daylight with a red:far red ratio of 0·3 vs. 50 % daylight and a red:far red ratio of 1·2).
Key Results
Mechanically stressed plants produced more leaves with shorter more flexible petioles, more roots, and more but less massive stolons. Responses to spectral shading were mostly in the opposite direction to thigmomorphogenesis, including the production of thinner, taller petioles made of more rigid tissue. The degree of thigmomorphogenesis was either independent of light climate or stimulated by spectral shading. At the genotypic level there were no clear correlations between responses to shade and mechanical stress.
Conclusions
These results suggest that in stoloniferous plants mechanical stress results in clones with a more compact, shorter shoot structure and more roots. This response does not appear to be suppressed by canopy shading, which suggests that wind shielding (reduced mechanical stress) by neighbours in dense vegetation serves as a cue that induces shade avoidance responses such as increased petiole elongation.
Key words: Allometry, biomechanics, clonal plants, phenotypic plasticity, Potentilla reptans, thigmomorphogenesis, shade avoidance, Young's modulus
INTRODUCTION
When plants are exposed to mechanical stimuli, such as wind, touching or rubbing, they typically produce shorter and thicker stems (Biro et al., 1980; Telewski, 1990; Jaffe and Forbes, 1993; Henry and Thomas, 2002; Anten et al., 2005), more roots (Crook and Ennos, 1994; Niklas, 1998; Henry and Thomas, 2002; Anten et al., 2006), and increased flexural rigidity of stems and roots (Telewski, 1994; Goodman and Ennos, 1996; Anten et al., 2005). These responses, termed thigmomorphogenesis (Jaffe, 1973) increase the resistance of plants to mechanical failure (Niklas, 1992, 1998).
Most studies on thigmomorphogenesis have been conducted with erect plants (e.g. Telewski, 1990; Niklas, 1998; Henry and Thomas, 2002; Anten et al., 2005) and as far as is known there has been no comprehensive study of the responses of stoloniferous plants to mechanical stress. Erect plants generally produce one or a few stems that act as vertical support structure, usually bearing both branches and a large number of leaves. In vegetation stands, stems are therefore the primary structure by which these plants position their leaves in the light gradient, but these stems need to be designed such that they can carry relatively large static loads (Niklas, 1992). Horizontal canopy expansion is achieved through increased branch or petiole length and these structures are designed as horizontal cantilever arms that must be sufficiently rigid to support their own weight (Niklas, 1992). Conversely in stoloniferous plants, leaf petioles generally serve as vertical support structures. Each leaf is separately supported, which, in terms of biomass use for vertical support, is less efficient than producing a single stem. This is because every petiole needs to carry its own weight in addition to the weight of the leaf lamina that it supports. On the other hand, these plants produce horizontal stems (stolons), which due to their creeping growth form, are not subject to the constraints of having to carry their own weight, and thus require a smaller mass investment in mechanical support. Stoloniferous plants might therefore be more flexible in regulating the number and horizontal placement of leaves. These differences in architecture probably have consequences for the way in which these two groups of plants respond to mechanical stress.
Many of the characteristics involved in thigmomorphogenesis are also associated with shade avoidance, a syndrome including the production of taller, more slender stems, reduced branching, and a reduction in root allocation that plants exhibit when growing in the proximity of neighbours (Smith, 1982; Weiner and Thomas, 1992; Nagashima et al., 1995; Schmitt et al., 1999). Reductions in the red:far-red ratio (R/FR) of light is generally considered to be the primary cue that induces these responses (Smith, 1982; Schmitt et al., 1999) though other factors such as blue light perceived by cryptochromes (Ballaré, 1999) and neighbour-produced ethylene (Pierik et al., 2003) are also believed to be involved. Interestingly thigmomorphogenesis and shade avoidance responses are often in the opposite direction (Anten et al., 2005).
Exposure to mechanical stress and stand density are not independent (Holbrook and Putz, 1989). In crowded vegetation, plants shield each other from wind and the amount of mechanical stress to which plants are exposed is therefore considerably lower than in open habitats (Goudriaan, 1977; Speck, 2003; Anten et al., 2005). Few studies (Holbrook and Putz, 1989; Henry and Thomas, 2002; Mitchell, 2003; Anten et al., 2005), however, have investigated the interactive effects of canopy shading and mechanical stress on plant growth. When Abutilon theoprasti plants from dense and open stands were exposed to equal wind speeds those from dense stands exhibited a much weaker inhibition of stem elongation (Henry and Thomas, 2002). The authors argued that in dense vegetation, where plants compete for light, thigmomorphogenesis should be suppressed because any reduction in height growth associated with thigmomorphogenesis would lead to reduced fitness. However, a study by Anten et al. (2005) did not reveal any suppression of thigmomorphogenesis for plants growing in dense stands. They argued that plants in dense vegetation should maintain sensitivity to variation in mechanical stress as the additional stem elongation, that would thus be induced by wind shielding, would have important consequences for lifetime growth and seed production. In stoloniferous plants the interaction between stand density, light competition, shade avoidance and wind exposure is further complicated because a plant, through stolon growth and branching pattern, at least partially determines its own shoot density and associated light climate and wind shielding. Moreover neighbouring shoots often belong to the same genetic individual, which probably reduces the competitive benefit of increased height growth (Anten, 2005).
The above studies on the effects of canopy shading and mechanical stress on plant growth were conducted with single genotypes. In fact very few studies (Emery et al., 1994) have compared thigmomorphogenesis between different genotypes of a species. If, as argued by Henry and Thomas (2002), thigmomorphogenesis should be suppressed under canopy shading, then it could be expected that genotypes that respond more plastically to low R/FR light associated with canopy shading should be less responsive to mechanical stress. These genotypes are likely to come from areas with dense vegetation (see Dudley and Schmitt, 1995), where strong thigmorphogenesis would then be selected against.
Here the following questions are addressed: (a) What are the effects of mechanical stress on growth, mass allocation and petiole and stolon structure of individual genotypes of a stoloniferous plant? (b) Is thigmorphogenesis in these plants suppressed by canopy shading, and are individual genotypes that exhibit greater levels of shade avoidance consequently less sensitive to mechanical stress? To this end, ten genotypes of the stoloniferous plant Potentilla reptans were subjected to two levels of spectral shading and two levels of mechanical stress in a factorial design. Previous studies (Henry and Thomas, 2002; Anten et al., 2005) used combinations of stand density and mechanical stress, which had the disadvantage that interaction between the two treatments through wind shielding, could not be entirely ignored. Here, therefore, spectral shading is used to simulate the light climate in dense vegetation without changing plant density itself. Potentilla reptans was chosen because of its distinguishable rosette structure, its simple leaves with one leaf being born at the tip of the petiole and its high plasticity in petiole length and stolon architecture (Huber, 1996; Stuefer and Huber, 1998).
MATERIALS AND METHODS
Plant material
The stoloniferous, rosette-forming herb Potentilla reptans L. (Rosaceae) occurs in sunny habitats. It commonly occurs in dense herbaceous vegetation, in mixed grasslands and sometimes it may form dense monospecific stands. In addition this species can frequently be found in more open, disturbed sites such as river shores and roadsides (Huber and Stuefer, 1997). Established rosettes produce horizontally growing stems (i.e. primary stolons) which may root at their nodes and thereby give rise to potentially independent offspring rosettes (Stuefer et al., 1994). For a drawing of this species see Huber (1996). During the spring of 1997, rosettes of ten genotypes of P. reptans were collected separately from natural populations in different habitats of The Netherlands and allowed to proliferate in monocultural stands in the experimental garden of Utrecht University, Utrecht, The Netherlands.
Experimental set-up
In May 2005, similar-sized rosettes of the ten genotypes were taken from these genotype stands and transplanted into plastic trays (15 cm×15 cm×100 cm) filled with river sand. These trays were placed in the greenhouse, at a light availability of 50% of natural daylight achieved by the shading the greenhouse roof. After 4 weeks, offspring rosettes of the ten genotypes were excavated and size-standardized by removing all but the youngest unfolded leaf, and by cutting roots to a length of 3 cm. Rosettes were then planted into plastic pots (13 cm in depth and diameter) filled with river sand. At this time we added 2 g of slow-release fertilizer (Osmocote, 10 % N+10 P+10 % K+3 % Mg+trace elements) to each pot (for a total of 0·2 g N plant–1). Plants were watered daily throughout the experiment using a wash bottle to avoid mechanical stimulation of petioles (see Henry and Thomas, 2002).
After 2 weeks, 40 similar-sized plants of each genotype were randomly divided into five groups of eight replicate plants. The first of the five groups was immediately used for the first harvest (see below). The others were subjected to a combination of shading and mechanical stress treatments. Two levels of shading were applied in which plants were exposed to either 15 % of daylight with an R/FR of 0·3 (denoted ‘shade’) or 50 % daylight and R/FR=1·2 (denoted ‘non-shade’). Light was measured with a LI190 quantum sensor (LiCor, Lincoln, NE, USA) connected to an LI1000 data logger. Red (655–665 nm) and far-red (725–735 nm) light were measured with an LI1800 spectrometer. The ‘shade’ treatment was meant to simulate the light conditions that plants experience under a leaf canopy and was applied by creating cages covered with one layer of a plastic film (no. 122; Lee Colortran International, Andover, UK). Below the film 0·2 m of open space was left to allow for free air movement. Microclimatic measurements revealed no differences in temperature and air humidity between the two shading treatments. Mechanical stress (MS) was also applied at two levels (0 or 40 daily brushes with a duster, denoted ‘low-MS’ or ‘high-MS’, respectively, hereafter). This type of stress was chosen as it simulates the mechanical effect of wind without affecting the plants' microclimate. Every week, positions of differently treated plants and of the different genotypes were randomized to minimize possible effects of position in the greenhouse.
Growth and mechanical measurements
The first harvest was conducted in mid-July, before initiation of treatments, to determine the baseline biomass distribution. The eight selected plants of each genotype were cut at ground level and divided into petioles, leaves and stolons. The length of the longest petiole of each plant was measured with a digital calliper to the nearest 0·1 mm and its basal diameter was measured in two perpendicular directions (see eqn 1b) with a leaf thickness meter to the nearest 0·01 mm. The mean of the two measurements was then calculated. Leaf lamina area was measured with a leaf area meter (LI 3100), and the root system was carefully washed. Dry mass of all plant parts was determined after oven drying for at least 72 h at 70 °C.
A second harvest was conducted 30 d after the initial harvest, to determine growth rates. Plants were cut at ground level and divided into petioles. Leaves and stolons and roots were washed carefully. The following traits were measured using the same methodology as described above: basal diameters, leaf lamina area, petiole length, and the fresh and dry weights of petiole and lamina of the tallest leaf; total leaf number, leaf area and petiole, lamina and root dry weight of the parent rosette; the number of stolons and their combined length and dry weight; and the petiole and lamina dry weights of all offspring rosettes combined. For clarity, the rosette is defined to be without stolons and the parent rosette is considered to be the one that was planted initially. Also a leaf is defined as the petiole and lamina together.
In addition, Young's elastic modulus (E, MN m−2), a measure for the rigidity of the tissue (Niklas, 1992), of the petiole of the tallest leaf was measured. To prevent water loss, petioles were immediately packed in wet tissue paper, placed in a polythene bag and transported to the laboratory. E was then measured with a universal testing machine (Instron Model 5542, Canton, USA) using a three-point bending method. This method has the advantage that it keeps the force perpendicular to the petiole. Sections of the basal part of the petiole were placed horizontally over two supports that were 2–3 cm apart. The distance was adjusted such that it was two-thirds of the length of the petiole segment. Vertical applied forces (F, N) and resulting deflections (δ, m) were recorded. Young's modulus was calculated as follows (Gere and Timoshenko, 1999):
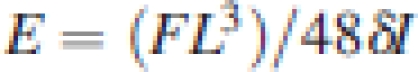 |
1a |
where L is the length between the supports (m) and I the second moment of area (m4) (Gere and Timoshenko, 1999). I was calculated from the cross-sectional dimensions of the petiole assuming it to have a parabolic shape (see fig. 3·3 in Niklas, 1992):
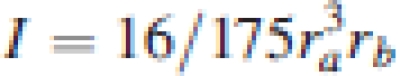 |
1b |
with a length equal to ra and a width equal to 2*rb (Niklas, 1992). Also the flexural stiffness of the petiole was calculated as the product of E and I (EI, MN m2).
Statistical analysis and calculation of plasticity
A three-way ANOVA was used to test for differences in response parameters, with canopy shading (d.f.=1) and mechanical stress (d.f.=1) as fixed factors and genotype (d.f.=9) as a random factor. Data transformation was based first on Levene's test for equality of variances and second on the Shapiro–Wilk test of normality (Table 1).
Table 1.
Leaf, rosette, stolon and whole-plant properties of P. reptans plants subjected to mechanical stress and spectral shading
| No shade |
Shade |
Significance levels |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Low-MS | High-MS | Low-MS | High-MS | G | L | M | G×L | G×M | L×M | G×L×M | |
| Properties of tallest leaf | |||||||||||
| Petiole length (cm) | 10·2 (0·75) | 6·5 (0·32) | 26·7 (1·9) | 16·8 (1·2) | *** | *** | *** | n.s. | n.s. | n.s. | n.s. |
| Petiole diameter (cm) | 0·153 (0·005) | 0·154 (0·006) | 0·109 (0·004) | 0·117 (0·003) | *** | *** | n.s. | *** | n.s. | n.s. | n.s. |
| Elastic modulus (MPa) | 128·9 (6·5) | 78·9 (3·8) | 152·7 (11·6) | 117·9 (7·0) | ** | *** | *** | n.s. | n.s. | n.s. | n.s. |
| Total mass | 0·113 (0·012) | 0·090 (0·009) | 0·067 (0·007) | 0·052 (0·006) | *** | *** | ** | ** | n.s. | n.s. | n.s. |
| Petiole mass | 0·026 (0·003) | 0·016 (0·002) | 0·032 (0·004) | 0·022 (0·003 | *** | ** | ** | n.s. | ** | n.s. | n.s. |
| I (mm4) | 0·0110 (0·014) | 0·113 (0·05) | 0·032 (0·005) | 0·043 (0·005) | *** | *** | ** | n.s. | n.s. | n.s. | n.s. |
| EI (N m2) | 13·8 (1·8) | 9·0 (0·78) | 4·7 (0·40) | 5·2 (0·53) | *** | *** | n.s. | n.s. | n.s. | ** | n.s. |
| Dry weight/fresh weight | 0·168 (0·006) | 0·157 (0·004) | 0·105 (0·003) | 0·102 (0·002) | n.s. | *** | n.s. | n.s. | n.s. | n.s. | n.s. |
| Properties of parent rosette | |||||||||||
| Total mass (g) | 1·03 (0·09) | 0·95 (0·08) | 0·32 (0·03) | 0·32 (0·03) | *** | *** | n.s. | n.s. | n.s. | n.s. | n.s. |
| Leaf mass (g) | 0·628 (0·059) | 0·526 (0·049) | 0·260 (0·027) | 0·245 (0·024) | *** | *** | * | n.s. | n.s. | n.s. | n.s. |
| Root mass (g) | 0·404 (0·034) | 0·421 (0·030) | 0·058 (0·004) | 0·072 (0·006) | *** | *** | ** | n.s. | n.s. | * | n.s. |
| Leaf no. | 7·66 (0·34) | 7·94 (0·27) | 5·15 (0·17) | 6·48 (0·40) | *** | *** | *** | n.s. | n.s. | * | n.s. |
| Root:shoot ratio | 0·655 (0·035) | 0·825 (0·043) | 0·267 (0·018) | 0·319 (0·014) | *** | *** | *** | * | n.s. | n.s. | n.s. |
| Stolon properties | |||||||||||
| Stolon number | 4·52 (0·28) | 5·19 (0·30) | 0·88 (0·23) | 1·36 (0·23) | *** | *** | ** | n.s. | n.s. | n.s. | n.s. |
| Total stolon length (cm) | 163·1 (14·2) | 143·2 (11·9) | 19·4 (4·1) | 30·4 (6·2) | *** | *** | n.s. | n.s. | n.s. | n.s. | n.s. |
| Total stolon mass (g) | 0·623 (0·060) | 0·56618375 | 0·043744875 | 0·046 (0·009) | *** | *** | n.s. | * | n.s. | n.s. | n.s. |
| Whole-plant properties | |||||||||||
| Total mass (g) | 1·98 (0·17) | 1·84 (0·13) | 0·40 (0·03) | 0·40 (0·04) | *** | *** | n.s. | n.s. | n.s. | n.s. | n.s. |
| SLA (g m–2) | 0·029 (0·001) | 0·028 (0·001) | 0·047 (0·001) | 0·046 (0·001) | ** | *** | n.s. | * | n.s. | n.s. | n.s. |
| RGR (g kg–1) | 80·2 (2·5) | 78·1 (2·9) | 27·7 (2·4) | 27·1 (2·6) | *** | *** | n.s. | * | n.s. | n.s. | n.s. |
| Mother mass ratio | 0·568 (0·024) | 0·546 (0·024) | 0·841 (0·027) | 0·825 (0·027) | *** | *** | n.s. | n.s. | n.s. | n.s. | n.s. |
Values presented are the treatment means for the data of ten genotypes combined and values in parentheses indicate the standard error of the mean (n=10, for the number of genotypes).
n.s., *, ** and *** indicate significance levels (non-significant, P>0·05, 0·01, 0·001 and 0·0001, respectively) of different effects in an analysis of variance (ANOVA) with genotype (G) as a random factor, and light (L) and mechanical stress (M) as fixed factors.
I and EI indicate the second moment of area and the petiole flexural stiffness, respectively.
Relative differences in parameter values between treatments were used rather than absolute ones to compare plasticity between genotypes, as the latter partly depends on inherent genotypic differences (Huber, 1996). Thus the plasticity in the response to spectral shade under conditions of either low [PSHADE(low-MS)] or high mechanical stress [PSHADE(high-MS)] was calculated as:
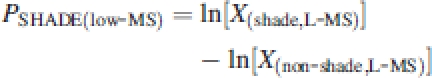 |
2a |
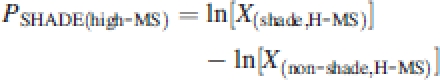 |
2b |
with X the treatment combination means for each genotype of a given parameter and L-MS and H-MS the two treatments, low MS and high MS. Similarly, degrees of plasticity in the response to mechanical stress were calculated under both shaded PMS(shade) and un-shaded conditions PMS(non-shade) as:
| 2c |
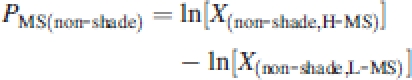 |
2d |
Using these values it was possible to analyse whether at the genotypic level plasticity to spectral shading correlated with plasticity to MS simply by means of a Pearson's correlation analysis of PSHADE values against PMS. Because thigmomorphogenesis and shade avoidance were in the opposite direction, and to allow for a convenient analysis of possible correlations, negative PMS values were converted to positive ones and visa versa so that a positive correlation coefficient implied that genotypes that exhibited stronger shade avoidance responses were also exhibiting greater thigmomorphogenesis. To avoid the data being used both on the x and y axes of these analyses, thus creating autocorrelation, the eight replicate plants per treatment combination were randomly split into two groups of four, where one was used for the calculation of PSHADE values and the other in the calculation of PMS.
RESULTS
Individual leaf properties
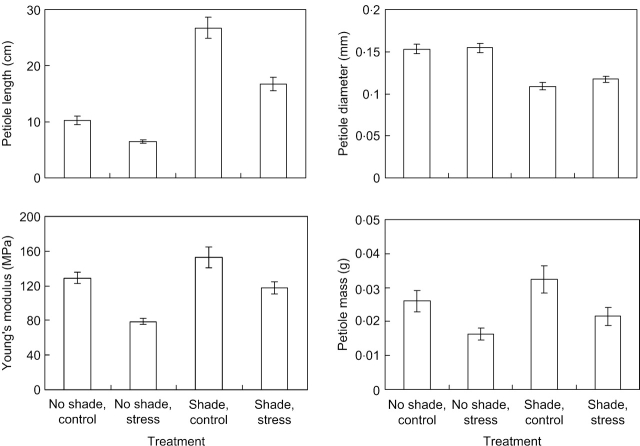
The tallest leaves on the parent rosette of plants grown under mechanical stress (MS) had shorter petioles of lower dry mass (Table 1 and Fig. 1). With the exception of genotypes 4, 5 and 10 grown without shade, these petioles also had a lower Young's elastic modulus (E); they were made of less rigid tissue (Fig. 1). This reduction in E was smaller under spectral shade (22 %) than without shade (39%), but the light×MS interactive effect was only marginally significant (P=0·065, Table 1). MS did not significantly affect petiole diameter nor the second moment of area (I, eqn 1b), which is calculated as a function of diameter. At high light, the flexural stiffness, the product of E and I, was lower in MS plants than in unstressed ones, but at low light there was no difference (Table 1).
Fig. 1.
Mean petiole characteristics (length, basal diameter, Young's elastic modulus and dry mass) of the tallest leaves on the parent rosette for plants of ten genotypes of Potentilla reptans subjected to spectral shading and mechanical stress treatments. Only treatment means are shown for which the data from the ten genotypes were combined. Bars indicate standard errors (n=10, for the number of genotypes). Further statistical analysis is provided in Table 1.
Spectral shading induced the production of taller, thinner and more massive petioles (Fig. 1 and Table 1). These results were consistent across all genotypes. Petioles of shaded plants also tended to have higher Young's elastic modules (E, Table 1), though this was not the case for all genotypes (Fig. 1C). Since a smaller petiole diameter is reflected in a lower second moment of area (I), petioles of shaded leaves also had a lower flexural stiffness (EI). Finally, the dry:fresh mass ratio was significantly reduced, indicating that shaded leaves retained relatively more water in their petioles (Table 1). Overall there was no significant interactive effect of mechanical stress and shading on any of the petiole characteristics except flexural stiffness (see above).
The genotypes differed significantly with respect to all leaf traits, except the fresh:dry mass ratio of the petiole (Table 1). There was a significant interactive genotype×light effect on total leaf mass and petiole diameter, indicating that with respect to these traits the ten genotypes differed in their response to spectral shading. On the other hand, there were no similar interactive effects on petiole length. Also a significant genotype×MS interactive effect was observed on petiole mass and marginally significant effects on leaf mass and petiole length (P=0·054 and 0·058, respectively), indicating that, at the leaf-level, there were differences in thigmomorphogenic responses between the ten genotypes (Table 1).
Parent rosette, stolon and whole-plant characteristics
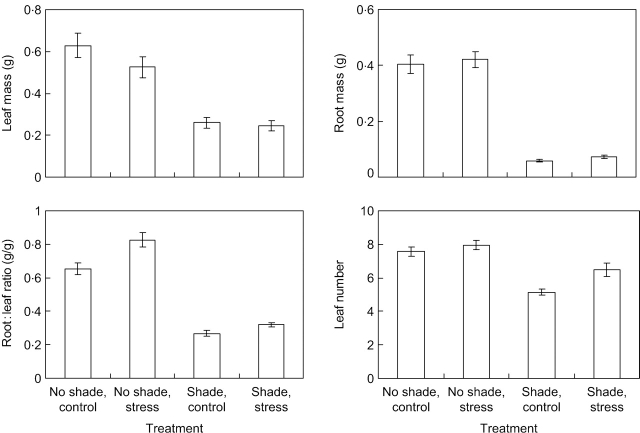
Mechanical stress had a significantly negative effect (P=0·025) on leaf mass of the parent rosette (Fig. 2 and Table 1). Root growth was stimulated by MS under spectral shade but not under unshaded conditions. Overall the parent rosettes of MS plants had higher root:leaf ratios than untreated plants. Shading had a negative effect on both leaf and root mass (Fig. 2 and Table 1). The reduction in root mass was stronger than the reduction in leaf mass, resulting in a reduced root:leaf ratio. The degree of this reduction differed between genotypes. The number of leaves on the parent rosette tended to be larger in mechanically stressed plants than in unstressed ones (Fig. 2 and Table 1). This effect was much stronger in the shading than in the non-shading treatment, as indicated by the significant MS×light interaction (Table 1). Shading on the other hand reduced the number of leaves.
Fig. 2.
Characteristics of the parent rosette (its leaf and root mass, the root:leaf mass ratio and the number of leaves) for plants of ten genotypes of Potentilla reptans subjected to spectral shading and mechanical stress treatments. Only treatment means are shown for which the data from the ten genotypes were combined. Bars indicate standard errors (n=10, for the number of genotypes). Further statistical analysis is provided in Table 1.
Mechanical stress had a significantly positive effect on stolon number but no effect on total stolon mass, suggesting that individual stolons tended to be smaller in MS plants (Fig. 3 and Table 1). The stolon number results were consistent across all genotypes and both shading treatments, except for genotypes 3, 5 and 10 under spectral shading. The combined stolon length increased under MS at low but not at high light, which was reflected in the significant MS×light interaction (Table 1). Shaded plants produced fewer stolons, with a shorter combined length and accumulated mass (Fig. 3 and Table 1).
Fig. 3.
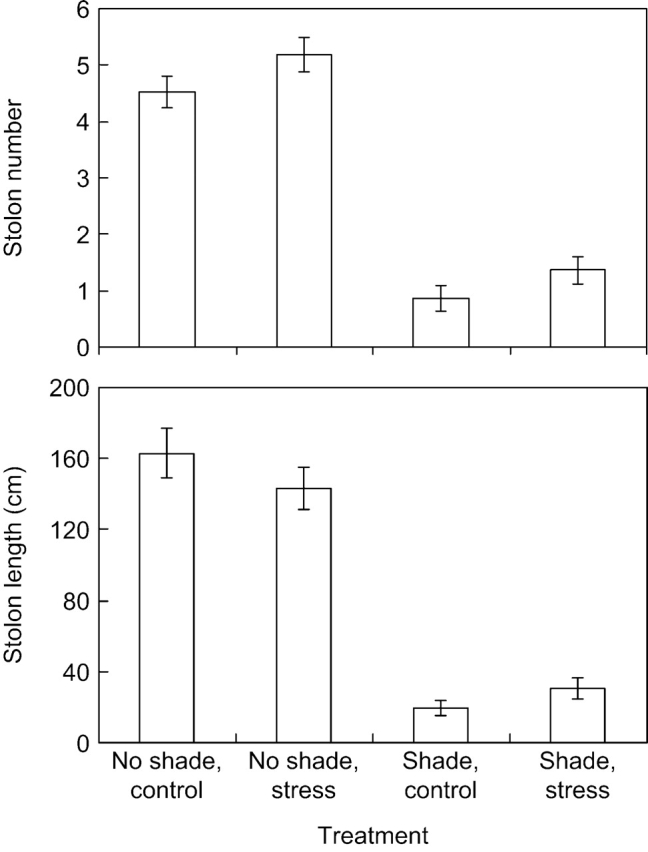
Stolon characteristics (stolon number and combined length of all stolons per plant) for plants of ten genotypes of Potentilla reptans subjected to spectral shading and mechanical stress treatments. Only treatment means are shown for which the data from the ten genotypes were combined. Bars indicate standard errors (n=10, for the number of genotypes). Further statistical analysis is provided in Table 1.
At the whole-plant level, mechanical stress had no effect on either relative growth rates (RGR) of plants or their final standing mass (Fig. 4 and Table 1). Shading on the other hand, had a strong negative effect on these parameters. The effect of shade on RGR differed between genotypes (Table 1); for example, genotype 5 exhibited a >80 % reduction in RGR while in genotypes 7 and 10 RGR was halved.
Fig. 4.
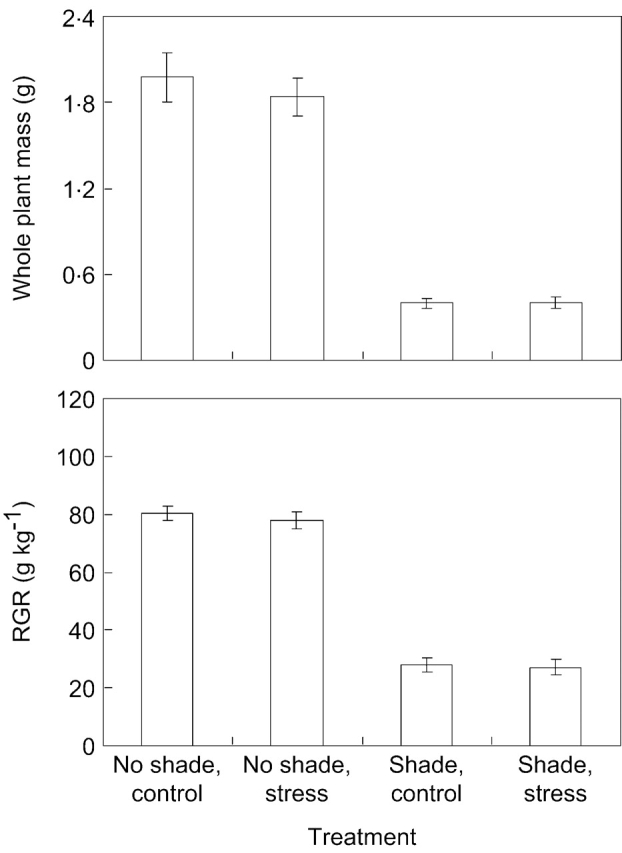
Total plant dry mass and mean growth per unit mass [relative growth rate (RGR)] for ten genotypes of Potentilla reptans subjected to spectral shading and mechanical stress treatments. Only treatment means are shown for which the data from the ten genotypes were combined. Bars indicate standard errors (n=10, for the number of genotypes). Further statistical analysis is provided in Table 1.
Genotypic differences in plasticity
For three traits – petiole length, petiole Young' modulus and the root:leaf mass ratio of the parent rosette – plasticity was calculated in response to spectral shading (PSHADE) and mechanical stress (PMS) following eqn (2). In most cases PSHADE was not significantly correlated with PMS. The exceptions were in the case of petiole length responses to spectral shade for mechanically stressed plants [PSHADE(high-MS)] against thigmomorphogenesis under unshaded conditions [PMS(non-shade)] and in the case of root:leaf ratio responses to spectral shade for non-stressed plants [PSHADE(low-MS)] against PMS(non-shade) (Table 2). In both cases the correlation was positive, indicating that genotypes which were more responsive to shading were also more responsive to mechanical stress.
Table 2.
Results of Pearson's correlation analysis of plasticity in the response to spectral shading (PSHADE) against plasticity in the response to mechanical stress (PMS) with r indicating the correlation coefficient
| Case 1 |
Case 2 |
Case 3 |
Case 4 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Trait | Sign | r | P | Sign | r | P | Sign | r | P | Sign | r | P |
| Petiole length | NEG | 0·224 | 0·535 | NEG | 0·245 | 0·834 | POS | 0·729 | 0·017 | POS | 0·141 | 0·696 |
| Youngs modulus | NEG | 0·430 | 0·215 | POS | 0·032 | 0·916 | NEG | 0·084 | 0·815 | POS | 0·173 | 0·631 |
| Root:shoot ratio | POS | 0·712 | 0·021 | NEG | 0·589 | 0·073 | POS | 0·441 | 0·202 | POS | 0·148 | 0·683 |
Four cases are considered: Case 1, PSHADE at low mechanical stress [PSHADE(low-MS)] against PMS under un-shaded conditions [PMS(non-shade)], Case 2, PSHADE(low-MS) vs. PMS under shaded conditions [PMS(shade)], Case 3, PSHADE at high mechanical stress [PSHADE(high-MS)] vs. PMS(non-shade) and Case 4 PSHADE(high-MS) vs. PMS(shade).
DISCUSSION
Effects of mechanical stress on the structure, growth and mass allocation of a stoloniferous plant
Mechanically stressed plants produced more leaves with shorter petioles made of more flexible tissue (lower Young's modulus, E), relatively more roots and more but less massive stolons. While thigmomorphogenesis in erect plants has been well documented at the level of leaves, stems and roots (see the Introduction), as far as is known this is the first study to give a comprehensive description of these responses in a stoloniferous plant. From a biomechanical perspective, stoloniferous plants such as P. reptans are different from erect plants. As noted in the Introduction this is because leaf petioles and not stems are the vertical support structures, the creeping growth form of stolons provides the possibility of efficient horizontal spread and these plants are therefore more flexible in regulating horizontal placement of leaves, which in turn influences the amount of wind stress on each leaf.
The reductions in petiole length and Young's modulus in response to MS is a result that is consistent with most studies on stems (e.g. Telewski, 1990; Jaffe and Forbes, 1993; Anten et al., 2005, 2006) and petioles (Niklas, 1996, 1999) of erect plants. These responses probably lead to increased mechanical resilience as shorter more flexible petioles are less prone to fracture or buckling (Niklas, 1992). Similarly, the increase in root growth and reduction in leaf growth was also observed in other studies (Crook and Ennos, 1994; Niklas, 1996), and there are indications that the phytohormone ethylene is involved in this shift in allocation (Anten et al., 2006). It has been demonstrated that larger root systems increase the anchorage strength of plants (Goodman and Ennos, 1996) thus preventing plants from being uprooted under mechanical stress. However, in stoloniferous plants such as P. reptans where above-ground structures are short and flexible, being uprooted is probably not likely to occur; buckling or rupture of petioles are more likely causes of mechanical failure.
Under unshaded conditions, mechanically stressed plants produced petioles with both greater flexibility (lower Young's modulus E) and lower flexural rigidity (EI) values. This result is contrary to the findings for stems, where EI usually increases in mechanically stressed plants (Jaffe et al., 1984; Telewski, 1994; Niklas, 1996), but consistent with the results for leaf petioles on erect plants (Niklas, 1996, 1999). Niklas (1996) argued that this difference in response between stems and leaf petioles reflects their overall different mechanical behaviour. Stems are central support structures and, as the plant grows, they support increasingly heavy loads. They should be sufficiently rigid to fulfil this function and are probably restricted in the extent to which they can reduce EI without buckling. Thus, they tend to respond to mechanical stress by increasing stem girth, which increases the maximum drag force that they can resist (see Anten et al., 2005). Petioles, by contrast, only carry the lamina. A reduced EI enables them to more easily reconfigure in response to wind drag (Vogel, 1994), thus avoiding drag, without compromising their ability to maintain their position. The results for the non-shaded plants suggest that leaves of rosette plants behave similarly even though they act as vertical support structures.
Contrary to the results above, under spectral shading EI was not affected by mechanical stress. This was mostly due to the fact that MS had a much smaller negative effect on Young's modulus (E) than without spectral shading (Table 1). Interestingly, spectral shading itself increased E. Tissue rigidity (E) not only depends on its composition – relative amounts of parenchyma, collenchyma and vascular tissue – but also on cell turgor; there appears to be a strong correlation between tissue water content and rigidity (Greenberg et al., 1989; Niklas, 1989, 1999). Plants grown under spectral shade had an almost 10 % higher water content than those grown without shade. In dense vegetation, spectral shading induces petiole elongation but shading simultaneously reduces assimilate availability for petiole growth through reduction in carbon gain. In a vertical light gradient, maintenance of a vertical stature is crucial for light interception. An increased turgor pressure might then be an energy efficient way of achieving the required stiffness to maintain vertical stature. Possibly this mechanism is less sensitive to mechanical stress such that E is not reduced as much and EI is conserved for MS plants under spectral shade.
Mechanically stressed plants produced more but less massive stolons, which indicates increased branching and reduced apical dominance. Similarly to the present study, Pigliucci (2002) found that wind exposure stimulates basal branching in the annual rosette plant Arabidopsis thaliana. But, as far as is known, stolon production in response to mechanical stress has been analysed in no other study. In clonal plants, the degree of stolon branching is often associated with resource availability. Branching is reduced in low resource areas to induce more linear growth and reduce the amount of mass needed to cross an unfavourable patch (e.g. Hutchings, 1988; Hutchings and de Kroon, 1994; but see Huber and Stuefer, 1997). However, stolon architecture also determines the placement of offspring rosettes and thus the above-ground structure of the clone; more but shorter stolons produce a denser canopy. As noted in the Introduction, plants can shield each other against wind drag forces and this shielding probably increases with shoot density. For example, it has been shown that attenuation of hydrodynamic drag forces within patches of the rhizomatous Spartina anglica in salt marshes increases considerably with shoot density (Bouma et al., 2005). Thus increased stolon production in response to mechanical stress might be an adaptation to reduce the impact of wind drag forces.
Spectral shading does not suppress thigmomorphogenesis; however, thigmomorphogenesis seems to constrain some shade avoidance responses
The effects of mechanical stress were either similar for both light treatments (almost all traits), or greater under spectral shading (leaf number and root mass) (Table 1). The exception was the change in the flexural stiffness of petioles which was greater under high light (see the Discussion). Apparently, thigmomorphogenesis does not appear to be suppressed under a spectral shading treatment that simulates the light climate experienced by plants in dense canopies. These findings are contrary to the notion that in dense vegetation thigmomorphogenesis should be suppressed because a thigmomorphogenic reduction in shoot height would cause plants to be shaded by neighbours (Henry and Thomas, 2002). They are, however, consistent with the proposition that sensitivity to mechanical stress should be maintained also in dense vegetation (Anten et al., 2005). In dense vegetation plants shield each other from wind and are thus exposed to less mechanical stress. A response to this lower level of mechanical stress, e.g. through increased petiole or stem elongation, can give plants an added competitive advantage.
Interestingly, some of the responses to spectral shading, reductions in root growth and stolon length, were weaker in mechanically stressed than in unstressed ones. This suggests that expression of shade avoidance can be constrained by mechanical stress, which as far as is known has not been reported previously, and indicates that the two responses do not act independently. There are indications in the physiological literature that responses to shade and mechanical stress indeed involve partially overlapping signal transduction pathways (Braam et al., 1996). Microarray and quantitative RT-PCR analyses showed that, in Arabidopsis thaliana, 67 % of the genes that were up-regulated at least 2-fold by mechanical stress were also up-regulated in darkness (Lee et al., 2005). However, it should be noted that responses to darkness are different from responses to spectral shade and that there is no study that has analysed the potential overlap in gene expression under MS and spectral shade.
At the individual genotype level there was no indication that spectral shading suppressed thigmomorphogenesis, but the question remains whether there is an inter-genotypic trade-off in these responses. Are genotypes that exhibit greater shade avoidance more or less plastic in response to mechanical stress? As noted in the Introduction, based on the argument that thigmomorphogenesis should be suppressed when plants grow in dense stands (Henry and Thomas, 2002), one would predict a negative correlation between the degrees of shade avoidance and thigmomorphogensis at the genotypical level. The present results give no indication of this. In most comparisons, no significant correlation was found between shade avoidance and thigmomorphogenesis (Table 2) except two where the correlation was positive. However, two significant cases out of 12 regression analyses might easily have occurred purely due to chance. Thus the present data do not give a clear indication of there being a correlation between the two responses.
Conclusions
The present data indicate that in ten genotypes of the stoloniferous species P. reptans, mechanical stress results in distribution of mass over more but smaller units, which produces clones with a more compact, shorter shoot structure and relatively more roots. These thigmomorphogenic responses are opposite in nature to shade avoidance but are not suppressed under spectral shading, though expression of shade avoidance can be constrained by mechanical stress. This in turn suggests that in addition to changes in light quality and neighbour-produced ethylene (see Introduction), wind shielding (lack of mechanical stress) may act as an extra cue that induces shade avoidance responses of plants in dense stands.
ACKNOWLEDGEMENTS
We thank Heinjo During, Yusuke Onoda and Marinus Werger for valuable comments on the manuscript, and are grateful to Henri Noordman, Sander van Hal, Betty Verduyn and Fred Siesling for providing technical assistance. This work was supported by Southwest University Postdoctoral Grants, China to Y.L.
LITERATURE CITED
- Anten NPR. Optimal characteristics of individual plants in vegetation stands and implications for species coexistence. Annals of Botany. 2005;95:495–506. doi: 10.1093/aob/mci048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anten NPR, Casado-Garcia R, Nagashima H. Effects of mechanical stress and plant density on mechanical characteristics, growth and lifetime reproduction of tobacco plants. American Naturalist. 2005;166:650–660. doi: 10.1086/497442. [DOI] [PubMed] [Google Scholar]
- Anten NPR, Casado-Garcia R, Pierik R, Pons TL. Ethylene sensitivity affects changes in growth patterns, but not stem properties, in response to mechanical stress in tobacco. Physiologia Plantarum. 2006;128:274–282. [Google Scholar]
- Ballaré CL. Keeping up with neighbors: phytochrome sensing and other signalling mechanisms. Trends in Plant Science. 1999;4:97–102. doi: 10.1016/s1360-1385(99)01383-7. [DOI] [PubMed] [Google Scholar]
- Biro RL, Hunt ER, Erner Y, Jaffe MJ. Thigmomorphogenesis: changes in cell division and elongation in the internodes of mechanically-perturbed or ethrel-treated bean plants. Annals of Botany. 1980;45:655–664. [Google Scholar]
- Bouma TJ, De Vries MB, Low E, Peralta G, Tanczos C, Van de Koppel J. Trade-offs related to ecosystem engineering: a case study on stiffness of emerging macrophytes. Ecology. 2005;86:2187–2199. [Google Scholar]
- Braam J, Sistrunk ML, Polysensky DH, Xu W, Puruganan MM, Antosiewicz DM. Life in a changing world: TCH gene regulation of expression and response to environmental signals. Physiologia Plantarum. 1996;98:909–916. [PubMed] [Google Scholar]
- Crook MJ, Ennos AR. Stem and root characteristics associated with lodging resistance in four winter wheat cultivars. Journal of Agricultural Science. 1994;123:167–174. [Google Scholar]
- Dudley SA, Schmitt J. Genetic differentiation in morphological responses to simulated foliage shade between populations of Impatiens capensis from open and woodland sites. Functional Ecology. 1995;9:655–666. [Google Scholar]
- Emery RJN, Reid DM, Chinnappa CC. Phenotypic plasticity of stem elongation in two ecotypes of Stellaria longipes: the role of ethylene and responses to wind. Plant, Cell and Environment. 1994;17:691–700. [Google Scholar]
- Gere JM, Timoshenko SP. Mechanics of materials. Cheltenham: Stanley Thornton; 1999. [Google Scholar]
- Goodman AM, Ennos AR. A comparative study of the response of the roots and shoots of sunflower and maize to mechanical stimulation. Journal of Experimental Botany. 1996;47:1499–1507. [Google Scholar]
- Goudriaan J. Crop micrometeorology: a simulation study. Wageningen: Pudoc; 1977. [Google Scholar]
- Greenberg AR, Mehling A, Lee M, Bock JH. Tensile behavior of grass. Journal of Material Science. 1989;24:2549–2554. [Google Scholar]
- Henry HAL, Thomas SC. Interactive effects of lateral shade and wind on stem allometry, biomass allocation and mechanical stability in Abutilon theophrasti (Malvaceae) American Journal of Botany. 2002;89:1609–1615. doi: 10.3732/ajb.89.10.1609. [DOI] [PubMed] [Google Scholar]
- Holbrook NM, Putz FE. Influence of neighbors on tree form: effects of lateral shade and prevention of sway on the allometry of Lyquidambar styraciflua. American Journal of Botany. 1989;76:1740–1749. [Google Scholar]
- Huber H. Plasticity of internodes and petioles in prostrate and erect Potentilla species. Functional Ecology. 1996;10:401–409. [Google Scholar]
- Huber H, Stuefer JF. Shade-induced changes in the branching pattern in a stoloniferous herb: functional response or allometric effect? Oecologia. 1997;110:478–486. doi: 10.1007/s004420050183. [DOI] [PubMed] [Google Scholar]
- Hutchings MJ. Differential foraging for resources and structural plasticity in plants. Trends in Ecology and Evolution. 1988;3:200–204. doi: 10.1016/0169-5347(88)90007-9. [DOI] [PubMed] [Google Scholar]
- Hutchings MJ, de Kroon H. Foraging in plants: the role of morphological plasticity in resource acquisition. Advances in Ecological Research. 1994;25:159–238. [Google Scholar]
- Jaffe MJ. Thigmomorphogenesis: the response of plant growth and development to mechanical stimulation. Planta. 1973;114:143–157. doi: 10.1007/BF00387472. [DOI] [PubMed] [Google Scholar]
- Jaffe MJ, Forbes S. Thigmomorphogenesis: the effect of mechanical perturbation on plants. Plant Growth Regulation. 1993;12:313–324. doi: 10.1007/BF00027213. [DOI] [PubMed] [Google Scholar]
- Jaffe MJ, Telewski FW, Cooke PW. Thigmomorphogenesis: on the mechanical properties of mechanically perturbed bean plants. Physiologia Plantarum. 1984;62:73–78. doi: 10.1111/j.1399-3054.1984.tb05925.x. [DOI] [PubMed] [Google Scholar]
- Lee D, Polinsky DH, Braam J. Genome-wide identification of touch- and darkness-regulated Arabidopisis genes: a focus on calmodulin-like and XTH genes. New Phytologist. 2005;165:429–444. doi: 10.1111/j.1469-8137.2004.01238.x. [DOI] [PubMed] [Google Scholar]
- Mitchell SJ. Effects of mechanical stimulus, shade and nitrogen fertilization on morphology and bending resistance in Douglas-fir seedlings. Canadian Journal of Forest Research. 2003;33:1602–1609. [Google Scholar]
- Nagashima H, Terashima I, Katoh S. Effects of plant density on frequency distributions of plant height in Chenopodium album stands: analysis based on continuous monitoring of height growth of individual plants. Annals of Botany. 1995;75:173–180. [Google Scholar]
- Niklas KJ. Mechanical behaviour of plant tissues as inferred from the theory of pressurized cellular solids. American Jounral of Botany. 1989;76:929–937. [Google Scholar]
- Niklas KJ. Plant biomechanics: an engineering approach to plant form and function. Chicago, IL: University of Chicago Press; 1992. [Google Scholar]
- Niklas KJ. Differences between Acer saccharum leaves from open and wind-protected sites. Annals of Botany. 1996;78:61–66. [Google Scholar]
- Niklas KJ. Effects of vibration on mechanical properties and biomass allocation pattern of Capsella bursa-pastoris (Cruciferae) Annals of Botany. 1998;82:147–156. [Google Scholar]
- Niklas KJ. A mechanical perspective on foliage leaf form and function. New Phytologist. 1999;143:19–31. [Google Scholar]
- Pierik R, Visser EJW, de Kroon H, Voesenek LACJ. Ethylene is required in tobacco to successfully compete with proximate neighbors. Plant, Cell and Environment. 2003;26:1229–1234. [Google Scholar]
- Pigliucci M. Touchy and bushy: phenotypic plasticity and integration in response to wind stimulation in Arabidopsis thaliana. International Journal of Plant Science. 2002;163:399–408. [Google Scholar]
- Schmitt J, Dudley SA, Pigliucci M. Manipulative approaches to testing adaptive plasticity: phytochrome-mediated shade-avoidance responses in plants. American Naturalist. 1999;154(suppl.):S43–S54. doi: 10.1086/303282. [DOI] [PubMed] [Google Scholar]
- Smith H. Light quality photoreception and plant strategy. Annual Review of Plant Physiology. 1982;33:481–518. [Google Scholar]
- Speck O. Field measurements of wind speed and reconfiguration in Arundo donax (Poaceae) with estimates of drag forces. American Journal of Botany. 2003;90:1253–1256. doi: 10.3732/ajb.90.8.1253. [DOI] [PubMed] [Google Scholar]
- Stuefer JF, During HJ, de Kroon H. High benefits of clonal integration in two stoloniferous species, in response to heterogeneous light environments. Journal of Ecology. 1994;82:511–518. [Google Scholar]
- Stuefer JF, Huber H. Differential effects of light quantity and spectral light quality on growth, morphology and development of two stoloniferous Potentilla species. Oecologia. 1998;117:1–8. doi: 10.1007/s004420050624. [DOI] [PubMed] [Google Scholar]
- Telewski FW. Growth, wood density and ethylene production in response to mechanical perturbation in Pinus taeda. Canadian Journal of Forest Research. 1990;20:1277–1282. [Google Scholar]
- Telewski FW. Wind-induced physiological and developmental responses in trees. In: Coutts MP, Grace J, editors. Wind and trees. Cambridge: Cambridge University Press; 1994. pp. 237–263. [Google Scholar]
- Vogel S. Life in moving fluids. Princeton, NJ: Princeton University Press; 1994. [Google Scholar]
- Weiner J, Thomas SC. Competition and allometry in three species of annual plants. Ecology. 1992;73:648–656. [Google Scholar]