Abstract
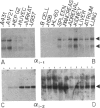
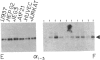
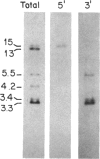
The guanine nucleotide-binding proteins (G proteins), which mediate hormonal regulation of many membrane functions, are composed of alpha, beta, and gamma subunits. We have cloned and characterized cDNA from a human T-cell library encoding a form of alpha i that is different from the human alpha i subtypes previously reported [Didsbury, J. R., Ho, Y.-S. & Snyderman, R. (1987) FEBS Lett. 211, 160-164 and Bray, P., Carter, A., Guo, V., Puckett, C., Kamholz, J., Spiegel, A. & Nirenberg, M. (1987) Proc. Natl. Acad. Sci. USA 84, 5115-5119]. alpha i is the alpha subunit of a class of G proteins that inhibits adenylate cyclase and regulates other enzymes and ion channels. This cDNA encodes a polypeptide of 354 amino acids and is assigned to encode the alpha i-3 subtype of G proteins on the basis of its similarity to other alpha i-like cDNAs and the presence of a predicted site for ADP ribosylation by pertussis toxin. We have determined the expression of mRNA for this and two other subtypes of human alpha i (alpha i-1 and alpha i-2) in a variety of human fetal tissues and in human cell lines. All three alpha i subtypes were present in the tissues tested. However, analysis of individual cell types reveals specificity of alpha i-1 expression. mRNA for alpha i-1 is absent in T cells, B cells, and monocytes but is present in other cell lines. The finding of differential expression of alpha i-1 genes may permit characterization of distinct physiological roles for this alpha i subunit. mRNA for alpha i-2 and alpha i-3 was found in all the primary and transformed cell lines tested. Thus, some cells contain all three alpha i subtypes. This observation raises the question of how cells prevent cross talk among receptors that are coupled to effectors through such similar alpha proteins.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bell G. I., Karam J. H., Rutter W. J. Polymorphic DNA region adjacent to the 5' end of the human insulin gene. Proc Natl Acad Sci U S A. 1981 Sep;78(9):5759–5763. doi: 10.1073/pnas.78.9.5759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bokoch G. M., Gilman A. G. Inhibition of receptor-mediated release of arachidonic acid by pertussis toxin. Cell. 1984 Dec;39(2 Pt 1):301–308. doi: 10.1016/0092-8674(84)90008-4. [DOI] [PubMed] [Google Scholar]
- Bray P., Carter A., Guo V., Puckett C., Kamholz J., Spiegel A., Nirenberg M. Human cDNA clones for an alpha subunit of Gi signal-transduction protein. Proc Natl Acad Sci U S A. 1987 Aug;84(15):5115–5119. doi: 10.1073/pnas.84.15.5115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bray P., Carter A., Simons C., Guo V., Puckett C., Kamholz J., Spiegel A., Nirenberg M. Human cDNA clones for four species of G alpha s signal transduction protein. Proc Natl Acad Sci U S A. 1986 Dec;83(23):8893–8897. doi: 10.1073/pnas.83.23.8893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Didsbury J. R., Ho Y. S., Snyderman R. Human Gi protein alpha-subunit: deduction of amino acid structure from a cloned cDNA. FEBS Lett. 1987 Jan 26;211(2):160–164. doi: 10.1016/0014-5793(87)81428-x. [DOI] [PubMed] [Google Scholar]
- Didsbury J. R., Snyderman R. Molecular cloning of a new human G protein. Evidence for two Gi alpha-like protein families. FEBS Lett. 1987 Jul 13;219(1):259–263. doi: 10.1016/0014-5793(87)81228-0. [DOI] [PubMed] [Google Scholar]
- Gilman A. G. G proteins: transducers of receptor-generated signals. Annu Rev Biochem. 1987;56:615–649. doi: 10.1146/annurev.bi.56.070187.003151. [DOI] [PubMed] [Google Scholar]
- Gubler U., Hoffman B. J. A simple and very efficient method for generating cDNA libraries. Gene. 1983 Nov;25(2-3):263–269. doi: 10.1016/0378-1119(83)90230-5. [DOI] [PubMed] [Google Scholar]
- Halliday K. R. Regional homology in GTP-binding proto-oncogene products and elongation factors. J Cyclic Nucleotide Protein Phosphor Res. 1983;9(6):435–448. [PubMed] [Google Scholar]
- Holden C. Forensic Scientists Go to Argentina' s Aid: New government seeks to identify bodies of those who "disappeared" under military regime. Science. 1984 Jul 27;225(4660):397–397. doi: 10.1126/science.225.4660.397. [DOI] [PubMed] [Google Scholar]
- Itoh H., Kozasa T., Nagata S., Nakamura S., Katada T., Ui M., Iwai S., Ohtsuka E., Kawasaki H., Suzuki K. Molecular cloning and sequence determination of cDNAs for alpha subunits of the guanine nucleotide-binding proteins Gs, Gi, and Go from rat brain. Proc Natl Acad Sci U S A. 1986 Jun;83(11):3776–3780. doi: 10.1073/pnas.83.11.3776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Itoh H., Okajima F., Ui M. Conversion of adrenergic mechanism from an alpha- to a beta-type during primary culture of rat hepatocytes. Accompanying decreases in the function of the inhibitory guanine nucleotide regulatory component of adenylate cyclase identified as the substrate of islet-activating protein. J Biol Chem. 1984 Dec 25;259(24):15464–15473. [PubMed] [Google Scholar]
- Jelsema C. L., Axelrod J. Stimulation of phospholipase A2 activity in bovine rod outer segments by the beta gamma subunits of transducin and its inhibition by the alpha subunit. Proc Natl Acad Sci U S A. 1987 Jun;84(11):3623–3627. doi: 10.1073/pnas.84.11.3623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones D. T., Reed R. R. Molecular cloning of five GTP-binding protein cDNA species from rat olfactory neuroepithelium. J Biol Chem. 1987 Oct 15;262(29):14241–14249. [PubMed] [Google Scholar]
- Jurnak F. Structure of the GDP domain of EF-Tu and location of the amino acids homologous to ras oncogene proteins. Science. 1985 Oct 4;230(4721):32–36. doi: 10.1126/science.3898365. [DOI] [PubMed] [Google Scholar]
- Kim S., Mellor J., Kingsman A. J., Kingsman S. M. Multiple control elements in the TRP1 promoter of Saccharomyces cerevisiae. Mol Cell Biol. 1986 Dec;6(12):4251–4258. doi: 10.1128/mcb.6.12.4251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lerea C. L., Somers D. E., Hurley J. B., Klock I. B., Bunt-Milam A. H. Identification of specific transducin alpha subunits in retinal rod and cone photoreceptors. Science. 1986 Oct 3;234(4772):77–80. doi: 10.1126/science.3529395. [DOI] [PubMed] [Google Scholar]
- Lochrie M. A., Hurley J. B., Simon M. I. Sequence of the alpha subunit of photoreceptor G protein: homologies between transducin, ras, and elongation factors. Science. 1985 Apr 5;228(4695):96–99. doi: 10.1126/science.3856323. [DOI] [PubMed] [Google Scholar]
- Logothetis D. E., Kurachi Y., Galper J., Neer E. J., Clapham D. E. The beta gamma subunits of GTP-binding proteins activate the muscarinic K+ channel in heart. Nature. 1987 Jan 22;325(6102):321–326. doi: 10.1038/325321a0. [DOI] [PubMed] [Google Scholar]
- Masters S. B., Stroud R. M., Bourne H. R. Family of G protein alpha chains: amphipathic analysis and predicted structure of functional domains. Protein Eng. 1986 Oct-Nov;1(1):47–54. [PubMed] [Google Scholar]
- Medynski D. C., Sullivan K., Smith D., Van Dop C., Chang F. H., Fung B. K., Seeburg P. H., Bourne H. R. Amino acid sequence of the alpha subunit of transducin deduced from the cDNA sequence. Proc Natl Acad Sci U S A. 1985 Jul;82(13):4311–4315. doi: 10.1073/pnas.82.13.4311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michel T., Winslow J. W., Smith J. A., Seidman J. G., Neer E. J. Molecular cloning and characterization of cDNA encoding the GTP-binding protein alpha i and identification of a related protein, alpha h. Proc Natl Acad Sci U S A. 1986 Oct;83(20):7663–7667. doi: 10.1073/pnas.83.20.7663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minamino N., Sudoh T., Kangawa K., Matsuo H. Neuromedins: novel smooth-muscle stimulating peptides identified in porcine spinal cord. Peptides. 1985;6 (Suppl 3):245–248. doi: 10.1016/0196-9781(85)90381-x. [DOI] [PubMed] [Google Scholar]
- Neer E. J., Lok J. M., Wolf L. G. Purification and properties of the inhibitory guanine nucleotide regulatory unit of brain adenylate cyclase. J Biol Chem. 1984 Nov 25;259(22):14222–14229. [PubMed] [Google Scholar]
- Neer E. J., Michel T., Eddy R., Shows T., Seidman J. G. Genes for two homologous G-protein alpha subunits map to different human chromosomes. Hum Genet. 1987 Nov;77(3):259–262. doi: 10.1007/BF00284481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nukada T., Tanabe T., Takahashi H., Noda M., Haga K., Haga T., Ichiyama A., Kanagawa K., Hiranaga M., Matsuo Primary structure of the alpha-subunit of bovine adenylate cyclase-inhibiting G-protein deduced from the cDNA sequence. FEBS Lett. 1986 Mar 3;197(1-2):305–310. doi: 10.1016/0014-5793(86)80347-7. [DOI] [PubMed] [Google Scholar]
- Robishaw J. D., Russell D. W., Harris B. A., Smigel M. D., Gilman A. G. Deduced primary structure of the alpha subunit of the GTP-binding stimulatory protein of adenylate cyclase. Proc Natl Acad Sci U S A. 1986 Mar;83(5):1251–1255. doi: 10.1073/pnas.83.5.1251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robishaw J. D., Smigel M. D., Gilman A. G. Molecular basis for two forms of the G protein that stimulates adenylate cyclase. J Biol Chem. 1986 Jul 25;261(21):9587–9590. [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sigal I. S., Gibbs J. B., D'Alonzo J. S., Scolnick E. M. Identification of effector residues and a neutralizing epitope of Ha-ras-encoded p21. Proc Natl Acad Sci U S A. 1986 Jul;83(13):4725–4729. doi: 10.1073/pnas.83.13.4725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sternweis P. C., Robishaw J. D. Isolation of two proteins with high affinity for guanine nucleotides from membranes of bovine brain. J Biol Chem. 1984 Nov 25;259(22):13806–13813. [PubMed] [Google Scholar]
- Sullivan K. A., Liao Y. C., Alborzi A., Beiderman B., Chang F. H., Masters S. B., Levinson A. D., Bourne H. R. Inhibitory and stimulatory G proteins of adenylate cyclase: cDNA and amino acid sequences of the alpha chains. Proc Natl Acad Sci U S A. 1986 Sep;83(18):6687–6691. doi: 10.1073/pnas.83.18.6687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanabe T., Nukada T., Nishikawa Y., Sugimoto K., Suzuki H., Takahashi H., Noda M., Haga T., Ichiyama A., Kangawa K. Primary structure of the alpha-subunit of transducin and its relationship to ras proteins. Nature. 1985 May 16;315(6016):242–245. doi: 10.1038/315242a0. [DOI] [PubMed] [Google Scholar]
- Van Meurs K. P., Angus C. W., Lavu S., Kung H. F., Czarnecki S. K., Moss J., Vaughan M. Deduced amino acid sequence of bovine retinal Go alpha: similarities to other guanine nucleotide-binding proteins. Proc Natl Acad Sci U S A. 1987 May;84(10):3107–3111. doi: 10.1073/pnas.84.10.3107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winslow J. W., Bradley J. D., Smith J. A., Neer E. J. Reactive sulfhydryl groups of alpha 39, a guanine nucleotide-binding protein from brain. Location and function. J Biol Chem. 1987 Apr 5;262(10):4501–4507. [PubMed] [Google Scholar]
- Yatsunami K., Khorana H. G. GTPase of bovine rod outer segments: the amino acid sequence of the alpha subunit as derived from the cDNA sequence. Proc Natl Acad Sci U S A. 1985 Jul;82(13):4316–4320. doi: 10.1073/pnas.82.13.4316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young R. A., Davis R. W. Efficient isolation of genes by using antibody probes. Proc Natl Acad Sci U S A. 1983 Mar;80(5):1194–1198. doi: 10.1073/pnas.80.5.1194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- la Cour T. F., Nyborg J., Thirup S., Clark B. F. Structural details of the binding of guanosine diphosphate to elongation factor Tu from E. coli as studied by X-ray crystallography. EMBO J. 1985 Sep;4(9):2385–2388. doi: 10.1002/j.1460-2075.1985.tb03943.x. [DOI] [PMC free article] [PubMed] [Google Scholar]