Abstract
Thymosin β4 (Tβ4) is a polypeptide involved in cellular proliferation, differentiation, and migration, over-expressed in several tumor entities. We evaluated its expression and function in 298 newly diagnosed multiple myeloma patients and the murine 5TMM model. Mean Tβ4 expression was significantly lower in myeloma cells compared to normal plasma cells (P<0.001). The same observation can be made in the 5TMM-mouse model by qRT-PCR and ELISA. Here, Tβ4 overexpression by lentiviral transduction of 5T33MMvt-cells led to significantly decreased proliferative and migratory capacities and increased sensitivity to apoptosis-induction. Mice injected with Tβ4 over-expressing myeloma cells showed a longer survival compared to mice injected with controls (88,9 vs. 65,9 days, P<0.05). In 209 MM patients treated with high-dose therapy and autologous stem cell transplantation, expression of Tβ4 below the median was associated with a significantly shorter event free survival (37.6 vs. 26.2 months, P<0.05). In conclusion, our results indicate a possible tumor suppressive function of Tβ4.
Keywords: thymosin β4, cellular proliferation, multiple myeloma
Introduction
β-thymosins are a family of small peptides that were originally proposed to be thymic hormones.1 They were identified as actin monomer binding proteins, controlling the availability of actin for polymerization. They may, therefore, have a crucial role in regulating cellular functions involving actin polymerization/depolymerization cycles. Currently, 15 β-thymosins have been identified and characterized as highly conservative 5-kDa peptides containing 40 to 44 amino acid residues. In most mammalian tissues, thymosin-β 4 (Tβ4), the most abundant thymosin peptide, Tβ10 and Tβ15, have been studied as important members of the β-thymosin family.2 Several studies reported that these genes are over-expressed in solid tumors, which could be correlated to the angiogenic and metastatic potential of the studied tumors.3
Multiple myeloma (MM) is a hematologic malignancy characterized by the accumulation of monoclonal plasma cells (PC) in the bone marrow (BM). MM cell biology can be dissected into the interactions of MM cells with their surrounding stroma (matrix proteins, cytokines and BM cells) and in the acquisition of essential changes in cell behavior, such as self-sufficiency in growth signals, evasion of apoptosis and acquisition of invasive and spreading capacities.4 Earlier reports indicated that Tβ4 was down-regulated in RNA from primary human MM cells and cell lines.5
This observation is in contrast to the results obtained in most solid tumors where an upregulation is seen in malignant cells compared to their normal counterparts. Cha et al. showed that overexpression of Tβ4 resulted in an increased metastatic capacity of lung cancer cells and increased angiogenic response.6
Since migration, invasion and associated angiogenesis are key features in MM biology, we were interested in studying Tβ4 expression in a large panel of MM patients and its functionality in the murine 5TMM model.
Design and Methods
Gene expression analysis on human myeloma cells
Tβ4 expression was analyzed in purified PCs from BM samples obtained from 14 healthy donors, 11 patients with monoclonal gammopathy of unknown significance (MGUS) and 298 previously untreated multiple MM patients at the University Hospitals of Heidelberg or Montpellier.7 Of these, 209 MM patients were treated by high-dose therapy and autologous stem cell transplantation (ASCT). Biotinylated complementary RNA (cRNA) was amplified according to the Affymetrix labeling protocol (Affymetrix, Santa Clara, CA, USA). cRNA from a first group of patients (7 normal donors, 7 MGUS and 65 MM patients) was hybridized to the human U133 A and B. This group will be referred to as the HM1-group. A second independent validation group of patients (7 normal donors, 16 MGUS and 233 MM patients) was named the HM2 group. For these patients, the U133 2.0 GeneChip was used. These micro-array data had been previously used for several analyses, but thymosin β4 expression had never been analyzed before.8,9 HM2 data were corrected for batch effect due to the usage of different labeling kits according to Johnson et al.10 Expression data were gcrma-normalized and analyzed by the bioconductor packages for R. For patients’ characteristic see Online Supplementary Table S1.
The 5T2MM and 5T33MM murine models of myeloma
The 5TMM models originated in elderly C57Bl/KaLwRij mice.11 The 5T33MMvivo (5T33MMvv) cells grow in vitro stroma-dependently with a limited survival while the 5T33MMvitro (5T33MMvt) cell line is a clonally identical variant that originated from an in vitro culture of 5T33MMvv cells, growing BM stroma-independently in RPMI-1640 supplemented with 10% bovine serum 1% natriumpyruvate, 100 U/mL penicillin, 100 μg/mL streptomycin and 2 mM L-glutamine (all from Biowhittaker, Verviers, Belgium).12
Quantification of intracellular protein levels of Tβ4 and F-Actin G-Actin
Enzyme-Linked Immunosorbent Assays (ELISA) for measuring Tβ4 concentrations were performed according to the manufacturer’s instructions (Immundiagnostik, Bensheim, Germany). Cells (107) were lyzed in a phosphate buffer containing 0.14 M NaCl, 2.6 M KCl, 8 mM Na2HPO4, 1.4 M KH2PO4 and 1% Triton X100 and sonicated with an ultrasound finger. Protein levels and ratios between F-Actin and G-Actin were determined using the G-actin/F-actin in vivo assay kit (Cytoskeleton Inc, Denver, USA).
Quantitative real-time PCR
Quantitative real-time PCR (qRT-PCR) was performed using the ABI Prism 7700 Sequence Detection System. For the detection of both human and mouse Tβ4 mRNA and the endogenous reference gene GUS, Assays on Demand (Applied Biosystems) were used. To verify the results obtained with the microarrays studies, Tβ4 expression was measured in 3 cell lines and in 3 patient samples and their correlations statistically verified using a Spearman correlation test.
Generation of 5T33MMvt cells over-expressing Tβ4
A lentiviral transferplasmid encoding mouse Tβ4 (m Tβ4) was constructed. The mTβ4 gene was obtained from HJ Cha (NIDCR, NIH, Bethesda, USA)6 and inserted into the transferplasmid pHR’tripCMV-IRES-tNGFR-SIN.13 mTβ4-encoding lentiviral vector particles were produced in 293T cells, collected, ultracentrifugated and their viral titer determined.14 After transduction, 5T33MMvt cells were surface stained using an in-house biotinylated anti-tNGFR antibody and purified by FACS sorting into a 6-well plate (Becton Dickinson, FACSVantage). Next, they were analyzed for Tβ4 expression by RT-PCR. The 5T33MMvt cells over-expressing Tβ4 will be referred to as 5T33MMvtTβ4+.
In vitro and in vivo effects of Tβ4 overexpression
In vitro proliferation was assessed by measuring DNA synthesis using a 3H-thymidine incorporation assay, as described earlier.15 Apoptosis sensitivity of the MM cells was analyzed by staining with FITC labeled-annexin V and propidium iodide according to the manufacturers’ instructions (BD Biosciences, Erembodegem, Belgium). In vitro migration studies were performed using Transwell chambers and 10% fetal calf serum as chemoattractant and were quantified through flow cytometry. To determine the effect of Tβ4 overexpression on survival, groups of 10 C57BLKaLwRij mice were intravenously injected with either 5×105 5T33MMvtTβ4+ or wild-type 5T33MMvt cells. Animals were sacrificed when they showed signs of morbidity, namely hind limb paralysis. Kaplan-Meier analysis was used to determine a difference in the survival.
Results and Discussion
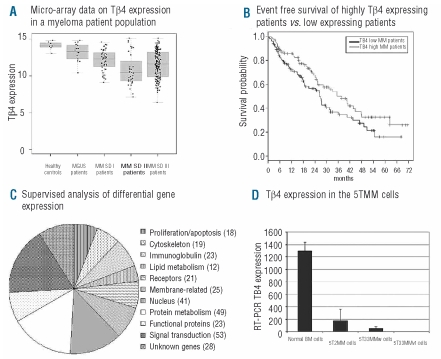
Different studies indicated a pivotal role of Tβ4 in the metastatic process of solid tumors.16,17 An adenoviral-based overexpression of Tβ4 was applied in a colon cancer and melanoma model showing increased growth, motility and invasive capacities in vitro and a larger tumor load in vivo.18,19 Since proliferation, migration and invasion are part of the hallmarks of the biology of MM, we were interested in investigating an involvement of Tβ4 in this disease. We first investigated the Tβ4 expression pattern in 298 primary MM-cell samples and 14 normal plasma cell samples from healthy donors. Tβ4 expression is significantly lower in MM cells of the HM1 group (P<0.05) and HM2 group (P<0.001) compared to normal plasma cells. This holds true for a significantly lower Tβ4 expression in its pre-malignant stage (MGUS), its early (Durie Salmon stage I) or late stage (Durie Salmon II and III) in both HM1 and HM2 groups (P<0.001) (Figure 1A). No relevant correlation could be found between Tβ4 expression and percentage of plasma cell infiltration in the bone marrow smear. Gene expression assessed by DNA-microarray correlates well with qRT-PCR performed on MM patient samples (coefficient of correlation r=0.993, P<0.001). These data are in agreement with results from Gondo et al. showing a decrease in Tβ4 expression in a small number of MM samples by Northern blot analysis.5
Figure 1.
(A) The micro-array data obtained for the Tβ4 expression in CD138+ sorted BM plasma cells from healthy donors and MM patients. These results were validated by quantitative RT-PCR. MGUS: monoclonal gammopathy of undetermined significance, MM multiple MM, SD: Salmon and Durie Stage. (B) The event free survival of Tβ4high and Tβ4low patients. Patients with Tβ4low MM had statistically significantly decreased event free survivals compared to patients with Tβ4high MM (P<0.001), while also their overall survival tended to be shorter. (C) The differently expressed genes between Tβ4high and Tβ4low patient groups. After identification of the gene, these were grouped into similar biological function. A complete list of the genes can be found in Online Supplementary Table S2. (D) A similar gene expression pattern was observed in the murine 5TMM models where Tβ4 mRNA expression in 5T33MM and 5T2MM invaded BM was lowered compared to normal BM cells.
Given the differential Tβ4 expression in MM patients, we subsequently investigated a possible prognostic value and influence on event free (EFS) and overall survival (OS) in our patient population. As Tβ4 was expressed in all MM cells, we examined the survival of 209 patients by comparing patients with Tβ4 expression above (Tβ4high) and below (Tβ4low) the median (Figure 1B). Patients with Tβ4high compared with Tβ4low show a significantly longer median EFS (n=209, 37.6 months vs. 26,2, P<0.05), but only a trend regarding OS (P=0.1). Concerning EFS, multivariate analyses on Tβ4 expression with either ISS or β2-microglobulin indicated an independent (P=0.04) prognostic value of Tβ4 expression regarding ISS (P<0.001), but not (P=0.07) regarding β2-microglobulin (P=0.003) levels. In multi-variate analyses for OS, ISS and β2-microglobulin appear as significant (P=<0.001) variables, whereas Tβ4 expression fails to reach independence (P=0.09 and P=0.01, respectively).
A supervized analysis of expression data comparing the Tβ4high to the Tβ4low group identified over 300 significantly differentially expressed genes. These genes are listed in Online Supplementary Table S2. Analysis of their biological function allowed them to be divided into main functional categories and this distribution is illustrated in Figure 1C. Signal transduction, protein metabolism and nuclear functions were the largest categories, but 19 genes were implicated in cytoskeletal organization, and 32 genes in lymphoid differentiation and immunoglobulin processing. In general, these gene clusters indicate a biological difference between MM cells of the two patient groups.
The data obtained in MM patients were also seen in the 5T33MM murine MM model by qRT-PCR demonstrating a decreased mRNA expression in 5TMM cells compared to normal BM cells (P<0.001, Figure 1D). Competitive ELISA confirmed these results on protein level (results not shown). To study functional effects of Tβ4, the gene was over-expressed using a lentiviral expression vector. The 5T33MMvt cell line was stably transduced and after sub-cloning, a 99% pure clone with strong t-NGFR expression was obtained. qRT-PCR confirmed the overexpression of Tβ4 compared to control cells (Figure 2A).
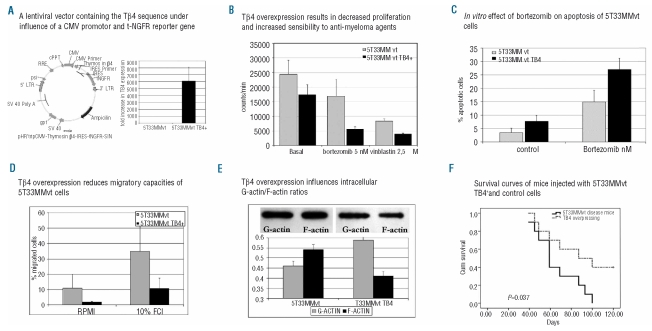
Figure 2.
(A) Schematic representation of the modified lentiviral transfer plasmid and results of RT-PCR and qRT-PCR indicating the presence of the inserted Tβ4 gene in cultured 5T33MMvtTβ4+ cells. (B) 3H thymidine uptake revealed a decreased DNA synthesis rate in 5T33MMvtTβ4+ cells compared to wild-type cells. Incubation with the anti-MM agent bortezomib (5 nM) or the micro-tubuli inhibitor vinblastine (2,5 μM) had significantly (P<0.001) stronger effects on 5T33MMvtTβ4+ cells than on control cells. A similar observation was made in apoptosis studies (C), where 5nM of bortezomib resulted in a significantly (P<0.05) increased apoptotic cell population after 18h incubation. (D) The effects of Tβ4 overexpression on migration of 5T33MMvt cells: using 10% fetal calf serum as chemo-attractant, only 10.8% (SD 6.6%) of 5T33MMvtTβ4+ cells migrated compared to 34.7% (SD 15.9%) of the control 5T33MMvt cells (P<0.05). (E) (Upper) The F-actin and G-actin bands of 5T33MMvt and 5T33MMvtTβ4+ cells. The graph illustrates the ratios of quantified F-actin and G-actin. In 5T33MMt cells actin is present in its polymerized form, whereas Tβ4 overexpression results in decreased F-actin formation and a greater pool of G-actin. (F) C57Bl/KaLwRij mice were injected with 5T33MMvt wild-type and 5T33MMvtTβ4+ cells. Kaplan-Meier analysis showed a significantly different survival between these 2 groups with a mean survival of mice injected with 5T33MMvt wild type of 65.9 days (SD 6.6 days), compared to 88.9 days (SD 9.3 days) for mice injected with 5T33MMvtTβ4+ cells. (P<0.05). LTR: long terminal repeat; gag: frame-shifted gag gene; RRE: rev-responsive element; CMV: cytomegalovirus promotor trip: central polypurine tract + termination sequence; Ires: internal ribosomal entry site; tNGFR: truncated form of the nerve growth factor receptor.
To assess the functional involvement of differential Tβ4 expression we used the 5T33MMvt and 5T33MMvtTβ4+ cell lines. In a 3H thymidine assay, 5T33MMvtTβ4+ cells showed a significant decrease in DNA synthesis compared to control cells (P<0.05). 5T33MMvtTβ4+ cells showed a significantly increased sensitivity to vinca-alkaloids (vinblastin) and bortezomib (Figure 2B; P<0.001 for both bortezomib and vinblastin).
Likewise, bortezomib induced apoptosis was higher in 5T33MMvtTβ4+ compared with 5T33MMvt cells (P<0.05; Figure 2C). In addition to affect survival pathways, Tβ4 overexpression reduced migratory capacities of 5T3MM cells; the percentages of cells that migrated in basal conditions and in 10%FCI was significantly lower in 5T33MMvtTβ4+ compared to control cells (P<0.05; Figure 2D). The relative increase after stimulation (compared to basal conditions) was, however, similar in both populations. We further examined the effects of Tβ4 expression on tumor development and survival of diseased mice by injecting mice intravenously with 5T33MMvtTβ4+ or control cells. In this study, the mean survival of mice injected with control cells was significantly shorter 65.9 days (SD 6.6 days), compared to 88.9 days (SD 9.3 days) for mice injected with 5T33MMvtTβ4+ cells (P<0.05; Figure 2F). These in vivo results confirm data obtained using the in vitro proliferation and apoptosis assays.
In solid tumors, Tβ4 expression is frequently upregulated in malignant and metastatic cells. In these cancers, higher Tβ4 expression resulted in increased metastatic and invasive capacities of tumor cells, while proliferation remained unaffected.6 In hematologic disorders, malignant plasma cell disorders, such as plasma cell leukemia and MM were the rare disorders that showed a decreased Tβ4 expression.5,20 In contrast to solid tumors, publications on the function of Tβ4 in hematologic conditions are scanty but indicate some inhibitory activity. Tβ4 was initially isolated and purified from a thymic protein preparation, called thymosin fraction-5. Addition of this protein fraction to different leukemic cell lines resulted in a decrease in growth responses.21 Similar inhibitory effects were recently described for Tβ4 on hematopoietic stem cells,22 bone marrow derived mast cells23 and human promyelocytic leukemia cells,24 in agreement with the results presented here. Whereas a mechanistic explanation of this discrepancy is beyond the scope of this paper, further investigations are clearly merited.
Since Tβ4 has been shown to bind G-actin in a 1:1 manner and thus affects the polymerization of G-Actin into F-Actin, we analyzed in a semi-quantitative way, intracellular G-actin and F-Actin. This quantification showed a lowered G-Actin-F-Actin ratio after Tβ4 overexpression (Figure 2E). F-Actin is of particular importance in cytoskeleton changes involved in cellular migration and in microtubuli organization controlling the mitotic spindle.25,26 In line with these results, vinca-alkaloids (e.g. vinblastine used here) with micro-tubulin (polymerization) inhibitory activity, had more affect on the proliferation capacities of 5T33MMvtTβ4+ cells than on control cells (Figure 2B). Since immunohistochemical studies also showed a nuclear staining of Tβ4 in 5TMM cells (results not shown), involvement of other pathways might also be implicated. Supervized gene analysis comparing Tβ4high with Tβ4low found different groups of genes differently expressed, including genes involved in cytoskeleton organization, nuclear homeostasis, lymphocyte differentiation and protein metabolism, which might indicate that the role of Tβ4 is more complicated than initially supposed.
In conclusion, our results propose a tumor suppressive function of Tβ4 expression in MM with impact on survival. Tβ4 was down-regulated in MM cells of patients compared to the normal BM plasma cells and studies with the murine 5T33MM model show a decreased in vitro and in vivo tumor growth for cells over-expressing the Tβ4 gene.
Supplementary Material
Acknowledgments
we thank Hae-Jae Cha (Research Center for Genetic Medicine, Children’s National Medical Center, Washington DC, USA) for providing the plasmid containing the murine thymosin β4 sequence. We also thank Angelo Willems, Carlo Heirman, Elsy Vaeremans, Erik Quartier, and Carine Seynaeve for excellent technical assistance.
Footnotes
Funding: this work was financially supported by the Fund for Scientific Research-Vlaanderen (FWO-Vlaanderen), the Association for International Cancer Research and by a grant from the European Commission FP6 to MSCNET. EM is a post-doctoral fellow of FWO-Vlaanderen.
The online version of this article has a supplementary appendix.
Authorship and Disclosures
JC and DH were the principal investigators and took primary responsibility for the paper. JC, DH, IK, TJB, EDB and EM participated in the laboratory work for this study. BVC, EVV, BK and KV coordinated the research. HG and BK were responsible for patient recruitment and patient data. JC, DH, TJB, BK and KV wrote the paper.
The authors reported no potential conflicts of interest.
References
- 1.Goldstein AL, Badamchian M. Thymosins: chemistry and biological properties in health and disease. Expert Opin Biol Ther. 2004;4(4):559–73. doi: 10.1517/14712598.4.4.559. [DOI] [PubMed] [Google Scholar]
- 2.Hannappel E. beta-Thymosins. Ann NY Acad Sci. 2007;1112:21–37. doi: 10.1196/annals.1415.018. [DOI] [PubMed] [Google Scholar]
- 3.Chen C, Li M, Yang H, Chai H, Fisher W, Yao Q. Roles of thymosins in cancers and other organ systems. World J Surg. 2005;29(3):264–70. doi: 10.1007/s00268-004-7817-2. [DOI] [PubMed] [Google Scholar]
- 4.Caers J, Van Valckenborgh E, Menu E, Van Camp B, Vanderkerken K. Unraveling the biology of multiple myeloma disease: cancer stem cells, acquired intracellular changes and interactions with the surrounding micro-environment. Bull Cancer. 2008;95(3):301–13. doi: 10.1684/bdc.2008.0600. [DOI] [PubMed] [Google Scholar]
- 5.Gondo H, Kudo J, White JW, Barr C, Selvanayagam P, Saunders GF. Differential expression of the human thymosin-beta 4 gene in lymphocytes, macrophages, and granulocytes. J Immunol. 1987;139(11):3840–8. [PubMed] [Google Scholar]
- 6.Cha HJ, Jeong MJ, Kleinman HK. Role of thymosin beta4 in tumor metastasis and angiogenesis. J Natl Cancer Inst. 2003;95 (22):1674–80. doi: 10.1093/jnci/djg100. [DOI] [PubMed] [Google Scholar]
- 7.Hose D, Moreaux J, Meissner T, Seckinger A, Goldschmidt H, Benner A, et al. Induction of angiogenesis by normal and malignant plasma cells. Blood. 2009;114(1):128–43. doi: 10.1182/blood-2008-10-184226. [DOI] [PubMed] [Google Scholar]
- 8.Sprynski AC, Hose D, Caillot L, Reme T, Shaughnessy JD, Jr, Barlogie B, et al. The role of IGF-1 as a major growth factor for myeloma cell lines and the prognostic relevance of the expression of its receptor. Blood. 2009;113(19):4614–26. doi: 10.1182/blood-2008-07-170464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hose D, Reme T, Meissner T, Moreaux J, Seckinger A, Lewis J, et al. Inhibition of aurora kinases for tailored risk-adapted treatment of multiple myeloma. Blood. 2009;113(18):4331–40. doi: 10.1182/blood-2008-09-178350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Johnson WE, Li C, Rabinovic A. Adjusting batch effects in microarray expression data using empirical Bayes methods. Biostatistics. 2007;8(1):118–27. doi: 10.1093/biostatistics/kxj037. [DOI] [PubMed] [Google Scholar]
- 11.Radl J. Multiple myeloma and related disorders. Lessons from an animal model. Pathol Biol (Paris) 1999;47(2):109–14. [PubMed] [Google Scholar]
- 12.Manning LS, Berger JD, O’Donoghue HL, Sheridan GN, Claringbold PG, Turner JH. A model of multiple myeloma: culture of 5T33 murine myeloma cells and evaluation of tumorigenicity in the C57BL/KaLwRij mouse. Br J Cancer. 1992;66(6):1088–93. doi: 10.1038/bjc.1992.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Breckpot K, Dullaers M, Bonehill A, van Meirvenne S, Heirman C, de Greef C, et al. Lentivirally transduced dendritic cells as a tool for cancer immunotherapy. J Gene Med. 2003;5(8):654–67. doi: 10.1002/jgm.400. [DOI] [PubMed] [Google Scholar]
- 14.Breckpot K, Heirman C, De Greef C, van der Bruggen P, Thielemans K. Identification of new antigenic peptide presented by HLA-Cw7 and encoded by several MAGE genes using dendritic cells transduced with lentiviruses. J Immunol. 2004;172(4):2232–7. doi: 10.4049/jimmunol.172.4.2232. [DOI] [PubMed] [Google Scholar]
- 15.Caers J, Menu E, De Raeve H, Lepage D, Van Valckenborgh E, Van Camp B, et al. Antitumour and antiangiogenic effects of Aplidin in the 5TMM syngeneic models of multiple myeloma. Br J Cancer. 2008;98 (12):1966–74. doi: 10.1038/sj.bjc.6604388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nummela P, Yin M, Kielosto M, Leaner V, Birrer MJ, Holtta E. Thymosin beta4 is a determinant of the transformed phenotype and invasiveness of S-adenosylmethionine decarboxylase-transfected fibroblasts. Cancer Res. 2006;66(2):701–12. doi: 10.1158/0008-5472.CAN-05-2421. [DOI] [PubMed] [Google Scholar]
- 17.Goldstein AL. Thymosin beta4: a new molecular target for antitumor strategies. J Natl Cancer Inst. 2003;95(22):1646–7. doi: 10.1093/jnci/djg126. [DOI] [PubMed] [Google Scholar]
- 18.Wang WS, Chen PM, Hsiao HL, Wang HS, Liang WY, Su Y. Overexpression of the thymosin beta-4 gene is associated with increased invasion of SW480 colon carcinoma cells and the distant metastasis of human colorectal carcinoma. Oncogene. 2004;23(39):6666–71. doi: 10.1038/sj.onc.1207888. [DOI] [PubMed] [Google Scholar]
- 19.Wang WS, Chen PM, Hsiao HL, Ju SY, Su Y. Overexpression of the thymosin beta-4 gene is associated with malignant progression of SW480 colon cancer cells. Oncogene. 2003;22(21):3297–306. doi: 10.1038/sj.onc.1206404. [DOI] [PubMed] [Google Scholar]
- 20.Shimamura R, Kudo J, Kondo H, Dohmen K, Gondo H, Okamura S, et al. Expression of the thymosin beta 4 gene during differentiation of hematopoietic cells. Blood. 1990;76(5):977–84. [PubMed] [Google Scholar]
- 21.Spangelo BL, Pompilius M, Farrimond DD, Stevens N, Nieva R, Shroff S, et al. Presence of a peptide component of thymosin fraction-5 manifesting discrete cytostatic properties in HL-60 human promyelocytic leukemia cells. Int Immunopharmacol. 2005;5(7–8):1317–29. doi: 10.1016/j.intimp.2005.04.001. [DOI] [PubMed] [Google Scholar]
- 22.Bonnet D, Lemoine FM, Frobert Y, Bonnet ML, Baillou C, Najman A, et al. Thymosin beta4, inhibitor for normal hematopoietic progenitor cells. Exp Hematol. 1996;24(7):776–82. [PubMed] [Google Scholar]
- 23.Leeanansaksiri W, DeSimone SK, Huff T, Hannappel E, Huff TF. Thymosin beta 4 and its N-terminal tetrapeptide, AcSDKP, inhibit proliferation, and induce dysplastic, non-apoptotic nuclei and degranulation of mast cells. Chem Biodivers. 2004;1(7):1091–100. doi: 10.1002/cbdv.200490081. [DOI] [PubMed] [Google Scholar]
- 24.Huang WQ, Wang BH, Wang QR. Thymosin beta4 and AcSDKP inhibit the proliferation of HL-60 cells and induce their differentiation and apoptosis. Cell Biol Int. 2006;30(6):514–20. doi: 10.1016/j.cellbi.2006.01.009. [DOI] [PubMed] [Google Scholar]
- 25.Menu E, Braet F, Timmers M, Van Riet I, Van Camp B, Vanderkerken K. The F-actin content of multiple myeloma cells as a measure of their migration. Ann NY Acad Sci. 2002;973:124–36. doi: 10.1111/j.1749-6632.2002.tb04620.x. [DOI] [PubMed] [Google Scholar]
- 26.Xu FL, Saunders WS. Actin and micro-tubules: working together to control spindle polarity. Cancer Cell. 2008;14(3):197–9. doi: 10.1016/j.ccr.2008.08.007. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.