Abstract
Subnormal plasma levels of high-density lipoprotein cholesterol (HDL-C) constitute a major cardiovascular risk factor; raising low HDL-C levels may therefore reduce the residual cardiovascular risk that frequently presents in dyslipidaemic subjects despite statin therapy. Cholesteryl ester transfer protein (CETP), a key modulator not only of the intravascular metabolism of HDL and apolipoprotein (apo) A-I but also of triglyceride (TG)-rich particles and low-density lipoprotein (LDL), mediates the transfer of cholesteryl esters from HDL to pro-atherogenic apoB-lipoproteins, with heterotransfer of TG mainly from very low-density lipoprotein to HDL. Cholesteryl ester transfer protein activity is elevated in the dyslipidaemias of metabolic disease involving insulin resistance and moderate to marked hypertriglyceridaemia, and is intimately associated with premature atherosclerosis and high cardiovascular risk. Cholesteryl ester transfer protein inhibition therefore presents a preferential target for elevation of HDL-C and reduction in atherosclerosis. This review appraises recent evidence for a central role of CETP in the action of current lipid-modulating agents with HDL-raising potential, i.e. statins, fibrates, and niacin, and compares their mechanisms of action with those of pharmacological agents under development which directly inhibit CETP. New CETP inhibitors, such as dalcetrapib and anacetrapib, are targeted to normalize HDL/apoA-I levels and anti-atherogenic activities of HDL particles. Further studies of these CETP inhibitors, in particular in long-term, large-scale outcome trials, will provide essential information on their safety and efficacy in reducing residual cardiovascular risk.
Keywords: HDL, Atherosclerosis, Cholesteryl ester transfer protein, Cholesteryl ester transfer protein inhibitor, Triglycerides, Reverse cholesterol transport
Introduction
Despite the widespread use of statin therapy, the incidence of cardiovascular morbidity and mortality remains elevated in many patients with dyslipidaemia, and particularly in those exhibiting metabolic disease and insulin resistance.1 In large landmark trials, reduction in low-density lipoprotein cholesterol (LDL-C) levels with statins has been shown to decrease the incidence of major cardiovascular events by 25–45%.2–4 Nonetheless, considerable residual cardiovascular risk, which includes a high frequency of recurrent events, remains even with an aggressive statin treatment regimen.5–9 New therapeutic options are clearly needed to further improve the treatment of atherogenic dyslipidaemia by reducing residual cardiovascular risk, especially with a view to reduction in lifetime risk.
Several cross-sectional and prospective epidemiological studies have demonstrated that high-density lipoprotein cholesterol (HDL-C) is a strong, independent, inverse predictor of risk of coronary heart disease (CHD).10–14 More recently, elevated circulating levels of the major apolipoproteins (apo) of HDL, apoA-I and A-II, have been shown to predict decreased risk of CHD.15,16 A recent meta-analysis17 suggested, however, that increasing HDL-C does not reduce the risk of cardiovascular events, and that such risk reduction is attributable to LDL-C raising alone. This finding is inconsistent with the weight of epidemiological and experimental evidence, and may reflect several major limitations in both design and methodology, including (i) the use of aggregated rather than individual subject data, (ii) lack of consideration of baseline triglyceride (TG) levels, (iii) inclusion of a majority of statin-driven studies in which differences between on-treatment and control levels of HDL-C were <3%, (iv) the risk of bias by confounding as this analysis describes an observational association, (v) insensitivity to measurement errors, and finally (vi) the combination of data from trials involving agents with significant distinctions in their action on HDL. Some prudence should therefore be applied when interpreting potentially spurious conclusions.17 In contrast, a 16-year follow-up of the Bezafibrate Infarction Prevention Study demonstrated that HDL-C-raising therapy was associated with a reduction in risk of long-term mortality.18 Moreover, large-scale prospective clinical studies have shown that therapeutic raising of HDL-C levels is associated with attenuated progression of intima-media thickening in the carotid artery, slowed progression of coronary artery atherosclerosis, and reduced cardiovascular risk.3,19–22
It is equally relevant that infusion of the HDL mimetic ETC-216, a lipidated form of recombinant apoA-I Milano, induced regression of coronary atherosclerosis in a small cohort of patients with acute coronary syndromes as evaluated by intravascular ultrasound (IVUS).23 More recently, a single infusion of reconstituted HDL particles induced acute changes in plaque composition and structure in a placebo-controlled study in patients exhibiting symptomatic atherosclerotic vascular disease in the superficial femoral artery.24 Specifically, a 20% increment in HDL-C was associated with reduction in lipid content, macrophage size, and the intra-plaque expression of vascular cell adhesion molecule (VCAM-1; –22%), consistent with reduction in intra-plaque inflammation. Finally, meta-analysis of statin-mediated lipid changes in IVUS trials in patients with incident coronary disease revealed that targets of LDL-C ≤ 87.5 mg/dL, together with HDL-C elevation ≥7.5%, are required in order to stop atherosclerosis progression, induce plaque regression, or both.21
Significantly, a recent post hoc analysis of the ‘Treating to New Targets’ trial demonstrated that low HDL-C is predictive of major cardiovascular events in patients receiving aggressive statin therapy.25 Even among patients with LDL-C < 70 mg/dL, those in the lowest quintile of HDL-C displayed an increased risk of major cardiovascular events compared with those in the highest quintile (P = 0.03).
Circulating HDL particles are highly heterogeneous in structure, intravascular metabolism, and anti-atherogenic activity, consisting primarily of two major subpopulations: large, light, cholesteryl ester (CE)-rich HDL2; and small, dense, CE-poor, protein-rich HDL3.26,27 Such subpopulations may however be further fractionated into multiple particle species by several methodologies, including bi-dimensional electrophoresis, isopycnic density gradient ultracentrifugation, immunoaffinity chromatography, and isotachophoresis; the structural, metabolic, and functional significance of such particle species, which are defined principally by their physicochemical properties and/or apo content, remains the subject of ongoing research.27 Indeed, recent proteomic analyses of HDL have revealed the presence of up to 75 distinct proteins.28,29 Moreover, all human HDL subpopulations display biological activities in which apoA-I is intimately involved; these include cellular cholesterol efflux capacity, and anti-oxidative, anti-inflammatory, anti-apoptotic, vasodilatory, anti-thrombotic, and anti-infectious actions.27,30,31 It is as a result of this spectrum of anti-atherogenic, cardioprotective activities that therapeutic elevation in plasma HDL concentration has become a major pharmacological target in patients with metabolic disease and subnormal HDL-C levels who typically exhibit high cardiovascular risk. In addition, the finding that the anti-atherogenic activities of HDL are defective in metabolic disease27,32 has identified the normalization of HDL functionality as a complementary therapeutic target.
The current options available for therapeutic elevation of HDL-C include statins, fibrates, and niacin (nicotinic acid), with development of cholesteryl ester transfer protein (CETP) inhibitors ongoing. Of these, niacin is the most effective, raising HDL-C by 20–30%.7,33 However, the therapeutic potential of niacin has been limited by its adverse effects; flushing occurs in 70–80% of the patients, although this may be attenuated by the use of extended-release niacin (ERN) formulations.33–35 Flushing may also be reduced by combining ERN with a new prostaglandin D2 receptor 1 antagonist, laropiprant (MK-0524).36 Indeed, in a recent Phase II study, significant reductions in flushing were observed in patients with dyslipidaemia treated with ERN plus laropiprant compared with ERN alone (P < 0.001), with no alterations in the beneficial lipid effects of ERN.36 In early 2008, the combination ERN/laropiprant formulation received approval for marketing authorization from the European Medicines Agency, but approval was delayed by the US Food and Drug Administration until findings in the Heart Protection Study 2—Treatment of HDL to Reduce the Incidence of Vascular Events (HPS2-THRIVE) become available.
Although statins efficaciously reduce LDL-C levels, they are not normally adequate as monotherapy to raise HDL-C, nor to correct HDL-associated cardiovascular risk in low HDL-C subjects, due to their modest effect on HDL-C levels (up to 16%).7,19,37,38 Fibric acid derivatives (fibrates) may increase HDL-C by up to 20%,39 but their efficacy may depend upon several factors.37,40 Like niacin, fibrates may be used in combination with statins, provided creatine kinase levels are monitored.7 In the Veterans Affairs High-Density Lipoprotein Intervention Trial (VA-HIT), which evaluated gemfibrozil treatment on cardiovascular morbi-mortality, circulating levels of small dense HDL3 (but not the larger HDL2 subfraction) at baseline and percentage change during treatment were significantly related to the incidence of CHD events.41 Moreover, niacin, statins, and fibrates modify other components of the lipid profile, often to a greater degree than their impact on HDL-C but clinical benefit associated with changes in individual lipoprotein classes is difficult to establish. For example, statins induce a marked reduction in the entire cascade of apoB-containing lipoproteins;42,43 the contribution of the substantially smaller effect on HDL-C towards cardiovascular risk reduction is therefore difficult to assess. Furthermore, it is unclear as to whether large or small HDL subfractions are distinct with respect to the degree of atheroprotection potentially conferred, although small, CE-poor, dense HDL3 are particularly active in vitro.31,33 Indeed, the hypothesis that all subfractions of HDL particles exert atheroprotection through one or more mechanisms appears both plausible and attractive at the present time.
The clinical benefits of raising low HDL-C levels observed in lipid intervention trials and the limitations of available therapies have stimulated the search to identify new, more efficacious HDL-raising agents. The marked increase in HDL-C associated with human deficiency of CETP44 suggested CETP inhibition as a novel and potentially effective approach to elevate HDL-C. Indeed, we interpret available evidence from prospective and cross-sectional epidemiological studies to support the overall contention that reduction of CETP activity, particularly when supranormal as typically occurs in dyslipidaemic subjects at high cardiovascular risk,45 constitutes a potential strategy for decreasing atherosclerosis and cardiovascular disease.3,46–49
This critical and timely review provides an integrated view of the role of CETP in cholesterol homeostasis and metabolism in man, identifies CETP as a central actor in the mechanisms of action of the major anti-dyslipidaemic agents which are currently available, and finally compares the principal features of pharmacological agents in development that directly target CETP. To ensure thorough identification of relevant publications, the PubMed database was searched (2002–present) using pre-defined keywords: cholesteryl ester transfer protein, CETP inhibitor, reverse cholesterol transport, TGs, HDL, statins, and fibrates.
The role of high-density lipoprotein and cholesteryl ester transfer protein in cholesterol metabolism
Although HDL exhibits a number of anti-atherosclerotic activities that appear to contribute to the cardiovascular benefits afforded by raising HDL levels, the major contribution is thought to be due to the key role of HDL particles in the atheroprotective reverse cholesterol transport (RCT) process. This anti-atherogenic pathway has been reviewed extensively and is summarized schematically in Figure 1;31,50 it involves the HDL-mediated efflux of cholesterol from peripheral tissues, including cholesterol-loaded monocyte-derived macrophages and foam cells in the arterial wall, with subsequent transport to the liver either for excretion as biliary cholesterol and bile acids, or for recycling.
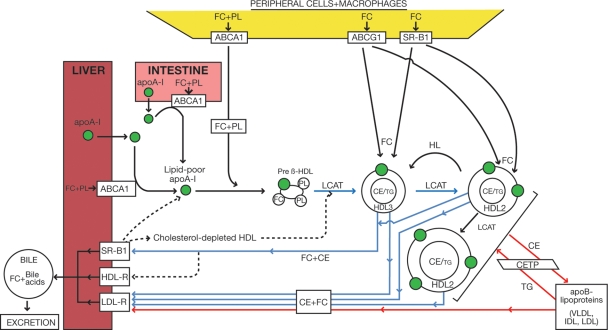
Figure 1.
Pathways of reverse cholesterol transport in man. The reverse cholesterol transport system involves lipoprotein-mediated transport of cholesterol from peripheral, extra-hepatic tissues, and arterial tissue (potentially including cholesterol-loaded foam cell macrophages of the atherosclerotic plaque) to the liver for excretion, either in the form of biliary cholesterol or bile acids. The ATP-binding cassette transporters, ABCA1 and ABCG1, and the scavenger receptor B1, are all implicated in cellular cholesterol efflux mechanisms to specific apoA-I/HDL acceptors. The progressive action of lecithin:cholesterol acyl transferase on free cholesterol in lipid-poor, apolipoprotein A-I-containing nascent high-density lipoproteins, including pre-β-HDL, gives rise to the formation of a spectrum of mature, spherical high-density lipoproteins with a neutral lipid core of cholesteryl ester and triglyceride. Mature high-density lipoproteins consist of two major subclasses, large cholesteryl ester-rich HDL2 and small cholesteryl ester-poor, protein-rich HDL3 particles; the latter represent the intravascular precursors of HDL2. The reverse cholesterol transport system involves two key pathways: (a) the direct pathway (blue lines), in which the cholesteryl ester content (and potentially some free cholesterol) of mature high-density lipoprotein particles is taken up primarily by a selective uptake process involving the hepatic scavenger receptor B1, and: (b) an indirect pathway (red lines) in which cholesteryl ester originating in high-density lipoprotein is deviated to potentially atherogenic very low-density lipoprotein, intermediate-density lipoprotein, and low-density lipoprotein particles by cholesteryl ester transfer protein. Both the cholesteryl ester and free cholesterol content of these particles are taken up by the liver predominantly via the low-density lipoprotein receptor which binds their apoB100 component. This latter pathway may represent up to 70% of cholesteryl ester delivered to the liver per day.51 The hepatic low-density lipoprotein receptor is also responsible for the direct uptake of high-density lipoprotein particles containing apoE; apoE may be present as a component of both HDL2 and HDL3 particles, and may be derived either by transfer from triglyceride-rich lipoproteins, or from tissue sources (principally liver and monocyte-macrophages). Whereas high-density lipoprotein uptake by the low-density lipoprotein receptor results primarily in lysosomal-mediated degradation of both lipids and apolipoproteins, interaction of high-density lipoprotein with scavenger receptor B1 regenerates lipid-poor apoA-I and cholesterol-depleted high-density lipoproteins, both of which may re-enter the HDL/apoA-I cycle.27 LPL, lipoprotein lipase; PL, phospholipids; HDL-R, holo HDL receptor; HL, hepatic lipase.
A major quantitative route for delivery of cholesterol to the liver is represented by the CETP-mediated transfer of CE from HDL to apoB-containing particles, mainly very low-density lipoprotein (VLDL) and LDL, with subsequent uptake primarily by hepatic LDL receptors;26,45,49,51,52 this pathway is frequently referred to as the indirect RCT pathway and accounts for some 70% of CE delivery to the liver in man (Figure 1). Cholesteryl ester transfer protein is secreted primarily by the liver and adipose tissue, and circulates in plasma associated principally with HDL.45,53 It promotes the transfer of CE from HDL to VLDL and LDL, in exchange for TG which moves in the opposite direction (Figure 2); the endogenous plasma activity of CETP is modulated to a major degree by the magnitude of triglyceridaemia.49 Indeed the rapid intravascular turnover of VLDL (half-life <30 min) is consistent with maintenance of a non-steady state in the plasma CE pool, with net mass transfer of CE from HDL to VLDL by CETP.51
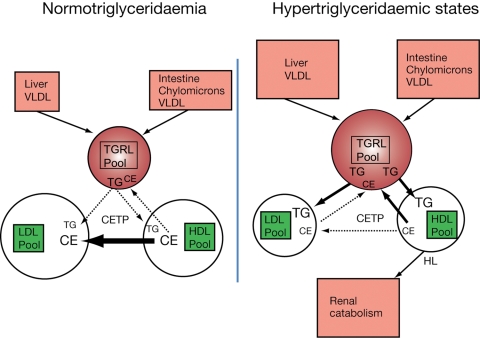
Figure 2.
Comparison of pathways of cholesteryl ester transfer protein-mediated heterotransfer of neutral core lipids between lipoprotein particles in normotriglyceridaemia vs. mixed dyslipidaemia involving moderate to marked hypertriglyceridaemia and subnormal levels of triglyceride-enriched high-density lipoprotein. In normotriglyceridaemia, net cholesteryl ester transfer from high-density lipoprotein to low-density lipoprotein predominates, with minor transfer to triglyceride-rich lipoproteins. In hypertriglyceridaemic states, increased numbers of very low-density lipoprotein particles constitute preferential cholesteryl ester acceptors giving rise to elevated acceptor capacity for cholesteryl ester transfer protein; high net mass transfer rates of cholesteryl ester from high-density lipoprotein to triglyceride-rich lipoproteins and of triglyceride from triglyceride-rich lipoproteins to both high- and low-density lipoproteins result. Triglyceride enrichment of both high- and low-density lipoproteins by this mechanism gives rise to formation of small dense low-density lipoprotein and small dense high-density lipoprotein.45 Modified from Barter et al.49 (with permission from Lippincott Williams and Wilkins).
Critically, CETP may exert both pro-atherogenic and anti-atherogenic actions.3 In its pro-atherogenic dimension, CETP-mediated CE transfer may effectively reduce the flux of cholesterol through HDL to hepatic scavenger receptor B1 (SR-B1) and HDL receptors in the direct RCT pathway,51 concomitantly enhancing the mass of cholesterol transported by atherogenic VLDL, intermediate-density lipoprotein (IDL), remnants, and LDL. In this way, the cholesterol burden of these particles is increased, potentially resulting in enhanced deposition in peripheral tissues and the arterial wall.54 As we and others have proposed, this mechanism may be of special relevance in the post-prandial state.45,55 In moderate to marked hypertriglyceridaemia, a second major CETP-mediated, pro-atherogenic pathway is of critical importance. Thus, under such conditions, elevated levels of apoB-containing acceptor particles for CETP drive enhanced transfer of TG from VLDL to HDL, leading to TG enrichment of HDL with abnormal intravascular metabolism involving reduction in particle size and fall in HDL-C and apoA-I levels due to accelerated renal catabolism (see below).45,56
In contrast, however, CETP may exert anti-atherogenic impact as it promotes the flux of CE to the liver via indirect RCT, with hepatic CE uptake predominantly through the anti-atherogenic LDL receptor pathway. Furthermore, CETP is critical to optimization of LDL particle structure and apoB100 conformation for high affinity binding to LDL receptors.45,57
As indicated above, CETP is centrally implicated in post-prandial hypertriglyceridaemia, an independent risk factor for CHD.3,58–61 The post-prandial state is characterized by the transient accumulation of intestinally derived chylomicrons (CM) and hepatically derived VLDL and their remnants, which may infiltrate and undergo retention in the arterial wall.62 During the lipolytic process, surface components (mainly phospholipids and free cholesterol) of CM and VLDL are sequestered to HDL due in part to the action of phospholipid transfer protein (PLTP). In post-prandial hypertriglyceridaemia, CETP-mediated transfer of CE and TG between plasma lipoprotein particles is accelerated as a direct consequence both of increase in the absolute number of apoB-containing acceptor particles for CE, and of major increase in the cumulative surface area under the curve for these particles during the 8 h post-prandial phase, thereby favouring CE enrichment of TG-rich lipoproteins with concomitant transformation of CE-enriched HDL into TG-rich HDL particles (Figure 2).45,53,59,63–65 Triglyceride enrichment of HDL is deleterious, as it leads to a loss of apoA-I from the HDL particle; in addition, hepatic lipase-mediated hydrolysis of HDL phospholipids and TG leads to reduction in HDL particle size.56,66 Accelerated catabolism of HDL and apoA-I ensues via the renal pathway, with decrease in plasma levels of both HDL-C and apoA-I.56,66
The action of CETP during the post-prandial phase has been shown to differ in normolipidaemic subjects when compared with that in patients with the mixed dyslipidaemic phenotype typical of premature coronary artery disease, type 2 diabetes, and the metabolic syndrome.45,59,65 In the post-prandial phase, CETP-mediated net CE transfer flux from HDL to potentially atherogenic TG-rich lipoproteins (especially large VLDL1) is markedly enhanced in mixed (type IIB) dyslipidaemia compared with normolipidaemic controls (Figure 2); such enhanced CE mass transfer occurs concomitantly with elevated levels of TG-rich particles which are maintained over the 8 h post-prandial phase and act as preferential acceptors of CE. In contrast, the area under the curve for triglyceridaemia is up to four-fold lower over the post-prandial phase in normolipidaemic controls, who typically display peak TG levels at 2–4 h of less than ∼150 mg/dL.59
The nature of the assay employed for evaluation of CETP activity in plasma is of special relevance to the above discussion; indeed, in vitro assays of CETP activity provide contrasting data depending on whether endogenous or exogenous substrate(s) are employed. Assays involving the addition of exogenous CE donors (HDL) or acceptors (VLDL and/or LDL) are most frequently used. Such assays reflect the maximal transfer capacity of CETP protein present in a given plasma sample as substrate concentrations are not rate-limiting. Under these conditions, the biological activity quantified is not the same as that occurring endogenously in plasma. Thus endogenous assays of CETP activity do not involve addition of exogenous CE acceptors or donors, and measure the net mass transfer of CE from HDL to acceptor VLDL and LDL particles at their plasma concentrations; such endogenous activity is modulated primarily by the relative concentrations of CE donor and acceptor particles, their lipid and protein composition, circulating CETP protein levels, and finally, the plasma half-life of the respective particles. For clinical studies of CETP activity, when the status of the integrated CETP system in the plasma of a given subject is to be evaluated, then the endogenous assay is most appropriate, as it uniquely respects endogenous levels of all the components of the CETP system in the sample.45
Lipid-modulating agents that act to modify the atherogenic lipid profile via indirect or direct action on cholesteryl ester transfer protein
Abundant evidence from in vivo and in vitro studies reveals that the current pharmacological agents (i.e. statins, fibrates, and niacin) commonly used in the treatment of atherogenic dyslipidaemia share the characteristic that their mechanisms of action involve—to varying degrees—direct and/or indirect reduction in plasma CETP activity. Such pharmacologically mediated modulation of CETP typically occurs either through reduction in numbers of apoB-containing lipoprotein acceptor particles (CM, VLDL, remnants, and LDL) for CETP-mediated CE transfer during the fasting and/or post-prandial phases, or through effects on CETP gene expression with resulting alteration in circulating concentrations of CETP protein, or both. These effects are especially relevant to the atherogenic lipid profile typical of type 2 diabetes and metabolic syndrome; such dysmetabolic states not only feature the atherogenic lipid triad, i.e. elevated levels of TG-rich lipoproteins and small dense LDL, together with subnormal levels of HDL-C, but also elevated levels of endogenous CETP activity, a key driver of this lipid phenotype.3,45 Indeed, supranormal CETP activity equally favours the qualitative abnormalities in HDL particles discussed above, which are intimately associated with defective anti-atherogenic function.3,27
Statins
In all common forms of atherogenic dyslipidaemia, notably hypercholesterolaemia and mixed dyslipidaemia, therapy to attenuate atherosclerosis and cardiovascular risk is firmly focused on marked reduction of circulating concentrations of atherogenic lipoproteins (LDL, VLDL, and remnants) with inhibitors of endogenous cholesterol synthesis, i.e. statins.37 A number of clinical trials have however revealed that statins typically induce modest and sustained elevation in HDL-C of up to 16%;37,67 most frequently, such elevations are in the range of 5–10% as revealed in the recent VOYAGER meta-analysis.38 The mechanism(s) underlying the statin-mediated increase in HDL-C is unclear, but appears to be multiple. Significantly, both in vitro and in vivo studies, in addition to post hoc analyses from large statin-related outcomes studies, have identified key factors which may contribute to the HDL response and facilitate deduction of putative mechanisms. In sum, these studies have revealed that statins reduce supranormal rates of endogenous CETP-mediated CE transfer from HDL to atherogenic particles in dyslipidaemic subjects.3,42,43,45 This effect, whether in normolipidaemic or dyslipidaemic subjects, or in animal models, involves several mechanisms which include reduction in the number of apoB-containing lipoprotein particles available to accept CE from HDL, and down-regulation of hepatic CETP mRNA expression with subsequent reduction of circulating plasma CETP concentration. As the absolute degree of reduction in baseline levels of apoB-containing particles by statins is largely dose-dependent for each statin, it is predictable that incremental statin-mediated reduction in atherogenic lipoprotein acceptor levels drives concomitant reduction in CETP activity (Table 1). Thus, the most potent statin, rosuvastatin, at its highest dose (40 mg/day), induced decrements of 12 and 59%, respectively, in CETP activity in hypercholesterolaemic and in mixed dyslipidaemic subjects, together with reductions in plasma CETP mass of 33–37%.42 The superior reduction in CETP activity seen in mixed dyslipidaemia reflects potent reduction in TG-rich lipoproteins, notably the VLDL1 subfraction (–46%), the most avid CE acceptor particle.42,43,71 Indeed, earlier studies with atorvastatin (10 mg/day) in a similar mixed lipid phenotype revealed that decrease in CETP activity was significantly correlated with statin-mediated reduction in VLDL1 levels.43 Clearly then, the effects of statins on lipoprotein profile and CETP activity are intimately related and are at least in part dependent on baseline lipid phenotype. Statins equally appear to moderately enhance hepatic apoA-I production (10–15%) and reduce CETP gene expression by inhibiting cholesterol biosynthesis in the liver;72 the cholesterol response element in the promoter of the CETP gene presumably underlies this latter effect.73,74 Finally, statin-induced increase in HDL-C may in part be attributable to enhanced peroxisome proliferator-activated receptor (PPAR) α activity, which may stimulate both hepatic apoA-I synthesis and HDL formation.75
Table 1.
Effect of statins and fibrates on endogenous plasma cholesteryl ester transfer protein activity, cholesteryl ester transfer protein mass, and the atherogenic lipid profile in dyslipidaemic subjects
| Lipid-lowering agent | Lipid phenotype | Patient status | TG (mg/dL) | VLDL-C (mg/dL) | LDL-C (mg/dL) | HDL-C (mg/dL) | ApoB (mg/dL) | ApoA-I (mg/dL) | Reduction in CE transfer rates from HDL to apoB-lipoproteins (μg CE/h/mL plasma) | CETP mass (μg/mL) |
|---|---|---|---|---|---|---|---|---|---|---|
| Statins | ||||||||||
| Pravastatin 40 mg/day68 | HFH | Baseline | 108 | 10 | 258 | 52 | 192 | 149 | –18% | ND |
| On-treatment | 71 (–34%) | 10 (0%) | 167 (–35%) | 52 (0%) | 133 (–31%) | 139 (–7%) | ND | |||
| Atorvastatin 10 mg/day43 | Mixed (combined) hyperlipidaemia (IIb) | Baseline | 197 | 46 | 175 | 46 | 144 | 132 | −21% | ND |
| On-treatment | 144 (–27%) | 26 (–43%) | 111 (–37%) | 46 (0%) | 99 (–31%) | 135 (+2%) | ND | |||
| Rosuvastatin 40 mg/day42 | Hypercholesterolaemia (IIa) | Baseline | 121 | 15 | 172 | 57 | 127 | 125 | −12% | 1.8 |
| On-treatment | 89 (–26%) | 10 (–36%) | 68 (–60%) | 62 (+9%) | 65 (–49%) | 144 (+15%) | 1.2 (–33%) | |||
| Mixed (combined) hyperlipidaemia (IIb) | Baseline | 234 | 36 | 164 | 42 | 134 | 124 | −59% | 1.9 | |
| On-treatment | 157 (–33%) | 18 (–50%) | 72 (–56%) | 46 (+11%) | 69 (–49%) | 133 (+7%) | 1.2 (–37%) | |||
| Fibrates | ||||||||||
| Fenofibrate 200 mg/day69 | Mixed (combined) hyperlipidaemia (IIb) | Baseline | 289 | 48 | 185 | 37 | 157 | 132 | −30% | ND |
| On-treatment | 161 (–44%) | 23 (–52%) | 159 (–14%) | 44 (+19%) | 133 (–15%) | 148 (+12%) | ND | |||
| Ciprofibrate 100 mg/day70 | Mixed (combined) hyperlipidaemia (IIb) | Baseline | 198 | 43 | 186 | 37 | 147 | 150 | −25% | ND |
| On-treatment | 108 (–45%) | 25 (–42%) | 149 (–20%) | 42 (+14%) | 109 (–26%) | 156 (+5%) | ND | |||
Mixed (combined) hyperlipidaemia is alternatively referred to as mixed or combined dyslipidaemia. Apo, apolipoprotein; CE, cholesteryl ester; CETP, cholesteryl ester transfer protein; HDL, high-density lipoprotein; HFH, heterozygous familial hypercholesterolaemia; ND, not determined; TG, triglyceride; VLDL-C, very low-density lipoprotein cholesterol.
Further lines of evidence support an effect of statins on CETP activity; first, the degree of change in HDL-C is directly related to the degree of reduction in TG and LDL-C,37 and secondly, a shift in the HDL particle distribution towards larger, relatively cholesterol- and apoA-I-rich HDL particles typical of HDL2 observed in statin-treated populations, including patients displaying heterozygous familial hypercholesterolaemia.43,68,72,76,77 Furthermore, lifestyle factors known to influence plasma CETP activity, such as alcohol intake, body mass index, and reduction in plasma TG, are also independent contributors to statin-induced change in HDL-C.78,79
The activity of hepatic lipase, an enzyme which hydrolyses both lipoprotein phospholipids and TG, may be moderately attenuated (up to –22%) on a dose-dependent basis by statin treatment.80 This effect favours maintenance of HDL/apoA-I lipidation—and thus prolonged apoA-I plasma residence time—and may indeed amplify the effect of statins in up-regulating apoA-I production. Further studies are needed, however, not only to determine how the above mechanisms mutually interact to favour elevation in circulating HDL-C and apoA-I levels, but also to establish whether statin-mediated effects on CETP activity, HDL-C, and apoA-I levels independently contribute to cardiovascular benefit in dyslipidaemic patients.
Fibrates
Early prospective trials of fibrates and of niacin promoted the concept that raising HDL-C levels by therapeutic means81–83 would translate into clinical benefit in dyslipidaemic patients at high cardiovascular risk.
Fibrates are a chemically heterogeneous class of agents, among which the most widely clinically used, fenofibrate, is primarily a PPARα-agonist of moderate affinity.84 Fibrates bind to PPARα by mimicking the structure of free fatty acids85 and may increase HDL-C by up to 20% as a function of baseline lipid phenotype.39,86 Fibrates appear to increase HDL-C levels in part by reducing plasma CETP activity, an action associated primarily with the potent ability of these agents to lower levels of TG-rich acceptor lipoproteins for CE, mainly VLDL, in both the fasting and post-prandial phases45,58,69,70 (Table 1). The capacity of fibrates to reduce (endogenous) plasma CETP concentration by up to −26% in patients with mixed dyslipidaemia69 appears related at least in part to CETP gene expression, suggesting that fibrates may modulate CETP gene expression through activation of PPARα.45 Reduction in VLDL, and specifically in the VLDL1 subfraction, following treatment with fenofibrate or ciprofibrate in patients with mixed dyslipidaemia was associated with a significant decrease (up to –35%) in the CETP-mediated transfer and targeting of CE from HDL to these particles.69,70 Reduction in the CETP-mediated flux of CE from HDL to VLDL therefore represents a common feature of the impact of statins and fibrates on the perturbed intravascular cholesterol metabolism characteristic of mixed dyslipidaemia.3,37,45
Fibrates also mediate modification in qualitative features of HDL and LDL particles.40,87 Thus, fenofibrate induced increases in the mass of light HDL subspecies at the expense of dense HDL3 particles in mixed dyslipidaemia, and equally shifted the dense LDL profile to a normalized distribution in which particles of lower density predominated; reduction in CETP activity is readily implicated in each of these effects.69,70,87 Fibrates preferentially enhance concentrations of apoA-I plus apoA-II-containing HDL particles with physicochemical properties intermediate between those of large HDL2 and small dense HDL3.88,89 Such action is in contrast to that of statins, however, which induce increase in the apoA-I-rich HDL subpopulation of largest size (α-1-HDL particles).48,76,77 The effect of fibrates on HDL particle subspecies result in part from fibrate-mediated up-regulation of apoA-I and apoA-II gene expression, although the increment in their plasma levels of apoA-I is minor as their fractional catabolic rate in the plasma compartment is accelerated.90
Other documented effects of fibrates on HDL metabolism result from PPARα-mediated up-regulation of lipoprotein lipase activity with enhanced lipolysis of CM and VLDL, resulting in release of surface fragments containing phospholipid and free cholesterol that sequester to the HDL pool under the action of PLTP; this latter effect may be amplified by PPARα-mediated attenuation of the hepatic synthesis and production of apoC-III.89,91,92 The potent TG-lowering action of fibrates is of course central to the attenuation of elevated basal levels of CETP activity in dyslipidaemic subjects, as it effects marked reduction in numbers of TG-rich particle acceptors with high avidity for CETP. Finally, enhanced cholesterol efflux from macrophages to HDL/apoA-I acceptors subsequent to PPARα-mediated up-regulation of SR-B1 and ABCA1 expression may impact on plasma HDL-C levels to a minor degree.93
As emphasized earlier, the impact of fibrates is largely a function of baseline lipid levels;40,91 the effects of both gemfibrozil and fenofibrate on plasma HDL-C levels are most pronounced when fasting levels of TG and TG-rich lipoproteins are elevated, and when baseline HDL-C levels are low.91,94 As with statins, the question can be legitimately raised as to the relative contribution of HDL-raising to cardiovascular benefit by fibrates, particularly given the wide range of anti-inflammatory actions of these agents.91,92
In regard to the impact of fibrates on cardiovascular disease, fenofibrate reduced angiographic progression of CHD in patients with type 2 diabetes,95,96 whereas gemfibrozil significantly reduced the frequency of non-fatal myocardial infarction or death attributable to CHD by 22% relative to placebo in the VA-HIT Trial.83 The FIELD trial, however, failed to show this in type 2 diabetes patients in a primary prevention context.97 In the Helsinki Heart Study, the observed reduction in major coronary events in subjects without CHD, but with non-HDL-C > 200 mg/dL, was attributed in part to the gemfibrozil-induced increase in HDL-C.82 Similarly, in men with known CHD and low HDL-C in the VA-HIT study, cardiovascular event reduction was shown to be inversely related to HDL-C level, and particularly that of HDL3, but not to change in either TG or LDL-C.41,83 It is noteworthy, however, that absolute increments in HDL-C in these studies were 11 and 6%, respectively, and that reductions in TG levels were at least three-fold greater (35 and 31%, respectively).
Importantly, a pooled meta-analysis of long-term randomized placebo-controlled clinical trails with fibrates has revealed that these agents significantly reduce the occurrence of non-fatal myocardial infarction, but are without significant effect on other adverse cardiovascular outcomes.98 Recent subgroup analyses have however revealed that subjects displaying the lipid triad in conjunction with a metabolic syndrome phenotype appear to benefit significantly from fibrate therapy; the mechanistic basis of such findings is indeterminate, but suggests that in addition to their effects on the lipid profile, fibrates may beneficially attenuate vascular and systemic inflammation due to PPARα-mediated down-regulation of a wide spectrum of pro-inflammatory genes.91,92,99,100
In summary, statins and fibrates act in part by similar mechanisms to attenuate supranormal CETP activity in atherogenic dyslipidaemia by reducing acceptor particle numbers for HDL CE. Other aspects of the actions of fibrates which influence the concentrations and qualitative aspects of HDL particles (notably those focused on TG-rich particles involving the lipolytic pathway) appear to be distinct from those not only of statins, but also of niacin and CETP inhibitors (see below).
Niacin
The broad spectrum action and efficacy of niacin (nicotinic acid; vitamin B3) in markedly lowering elevated concentrations of TG-rich lipoproteins, IDL, LDL, and Lp(a), together with its capacity to raise HDL-C, are especially notable. Indeed, niacin is presently the most effective agent available for raising HDL-C, typically increasing levels by up to 30% on a dose-dependent basis.3,7,24,33,101 The clinical benefits associated with niacin treatment, both as monotherapy or in combination with a statin, feature attenuation of atherosclerosis progression and/or induction of plaque regression in addition to reduction in cardiovascular risk, and have been reviewed elsewhere.22 The mechanisms underlying the action of niacin in reducing plasma VLDL, LDL, and apoB levels in vivo involve enhanced clearance of TG-rich lipoproteins containing either apoB100 or B48,102 although evidence is equally available to support decreased rates of VLDL production; such discrepancies may depend upon the metabolic background.103 Only recently has attention been focused on delineating the mechanisms which underlie the HDL-raising action of niacin.102,104,105 Four key processes are considered to contribute to niacin-mediated elevation in apoA-I and HDL-C levels: (i) up-regulation of apoA-I production rate (+24%) relative to placebo without change in fractional catabolic rate,102 with no change in either the concentration of or kinetic parameters for apoA-II; (ii) the ability of niacin to exert transient inhibition of hormone-sensitive TG lipase in adipose tissue and attenuate liberation of free fatty acids via TG lipolysis, with consequent reduction in hepatic VLDL-TG production, plasma VLDL levels, and thence in CETP-mediated depletion of HDL-CE; (iii) reduction in plasma CETP activity as a result of the combined effect of reduction in hepatic CETP gene expression, plasma CETP mass, and numbers of apoB-containing acceptor particles available for HDL-CE (see below);104,106 and (iv) reduction in the hepatic uptake of HDL, potentially by the holo-particle uptake pathway.107,108 Considered together, these processes would feasibly increase the plasma residence time of HDL and apoA-I and thus increase HDL-C levels. Such action is entirely consistent with recent findings in low HDL-C human subjects with established CAD who were treated with a niacin/statin combination, and in whom abnormalities in the HDL proteome were partially reversed.109 Finally, the potential role of niacin in enhancing cholesterol efflux via ABCA1 from macrophages to HDL acceptors, with positive impact on HDL-C levels, cannot be excluded.110 The above observations concur to place CETP firmly at the centre of the processes mediated by niacin treatment which directly lead to efficacious elevation of both HDL-C and apoA-I.
Cholesteryl ester transfer protein inhibitors
Several efficacious chemical CETP inhibitors have been identified; these include torcetrapib (Pfizer, New York, NY, USA), dalcetrapib (previously referred to as RO4607381/JTT-705, Roche/Japan Tobacco, Basel, Switzerland), and anacetrapib (MK-0859, Merck & Co., Whitehouse Station, NJ, USA). Molecular insight into the mechanism of action of these inhibitors has become possible as a result of the definition of the crystal structure of CETP.111 Thus, the identification of a hydrophobic substrate-binding tunnel in the three-dimensional structure of CETP which can accommodate two molecules of neutral lipid (either CE or TG, or one of each) is especially relevant to the mechanisms of action of these inhibitors, and notably to that of torcetrapib.111,112
Torcetrapib
Torcetrapib is a potent inhibitor of CETP activity (IC50 value ≈50 nM),112 enhancing the association between CETP and HDL to form a complex that inhibits the transfer of lipids between HDL and other lipoproteins.47 Data from a number of clinical studies performed in dyslipidaemic patients indicate that torcetrapib has beneficial effects on an atherogenic lipoprotein profile.113–115 However, in late 2006, The Investigation of Lipid Level Management to Understand its Impact in Atherosclerotic Events (ILLUMINATE) trial, which investigated the effects of atorvastatin (10 mg/day) plus torcetrapib (60 mg/day) or placebo in patients at high risk of CHD,116 was prematurely halted by the Data and Safety Monitoring Board. Despite the highly favourable changes in lipid profile (HDL-C levels +72%, LDL-C –25%), a significant increase in all-cause mortality [hazard ratio (HR), 1.58; 95% confidence interval (CI), 1.14–2.19; P = 0.006] and cardiovascular events (HR, 1.25; 95% CI, 1.09–1.44; P = 0.001) was seen for torcetrapib plus atorvastatin therapy for 12 months compared with atorvastatin plus placebo. As in small clinical trials with torcetrapib,113,117 elevation in blood pressure was observed in ILLUMINATE (mean systolic blood pressure increment of 5.4 mmHg) along with changes in electrolyte levels and elevated aldosterone levels, suggesting that torcetrapib may have off-target effects unrelated to HDL-raising. Studies in rats support a relationship between torcetrapib-mediated changes in blood pressure and aldosterone level, revealing that torcetrapib raised blood pressure with concomitant increase in expression of component genes of the renin–angiotensin–aldosterone system (RAAS) in adrenal tissue.118 Structure–activity investigations have provided further evidence that the hypertensive effects of torcetrapib are unrelated to CETP inhibition.119
Three prospective clinical trials of torcetrapib120–122 reported increments in systolic blood pressure of 2.8–5.4 mmHg; a pooled analysis of two of the trials121,122 reported elevation in plasma sodium and bicarbonate levels and reduction in potassium levels, supporting the contention that an off-target mineralocorticoid excess in patients administered torcetrapib contributed to the adverse outcome in ILLUMINATE.123 These trials used imaging modalities to evaluate atherosclerosis progression (ILLUSTRATE)120 and carotid intima-media thickening (CIMT; RADIANCE 1, RADIANCE 2).121,122 Although substantial increases in HDL-C (54–63%) and reductions in LDL-C (18–20%) from baseline were observed, torcetrapib plus atorvastatin failed to diminish maximum CIMT in patients with familial hypercholesterolaemia121 and in mixed dyslipidaemia;122 equally, this combination did not significantly decrease IVUS-assessed atheroma volume in patients with CHD.120,124 Although overall findings in the ILLUSTRATE trial did not reveal a beneficial impact of torcetrapib treatment on the progression of coronary atheroma, a post hoc analysis showed that patients exhibiting the greatest HDL-raising response (HDL-C > 87 mg/dL) displayed the lowest rate of progression of per cent atheroma volume (–0.7 vs. +0.7%, P = 0.0003).124
It has nonetheless been hypothesized that HDL function may be impaired by torcetrapib, either by a direct mechanism or indirectly by CETP inhibition.32 Torcetrapib-associated HDL dysfunction might result directly from the formation of non-productive complexes in which torcetrapib binds to CETP in a 1:1 ratio, forming a larger complex with HDL particles.112,125 However, calculation shows that for plasma concentrations of HDL 6–10 µM, and CETP 20–60 nM, only up to 1% of HDL particles could contain a single molecule of torcetrapib bound to CETP—at this level potential HDL particle dysfunction resulting from direct binding of torcetrapib would be undetectable unless inactive complexes were purified; in addition, any torcetrapib in excess of that bound to CETP-HDL complexes appears to partition preferentially into TG-rich lipoproteins (R. Clark, personal communication).
Equally, CETP inhibition could potentially result in the generation of HDL particles with deficient anti-atherogenic properties despite absence of bound torcetrapib; for example, large HDL particles enriched in apoA-I and CE might exert deleterious effects on the direct or indirect RCT pathways and on steroid metabolism.32,126 Further evidence for the functionality of HDL particles formed under torcetrapib treatment has recently been reported127 in mixed dyslipidaemic subjects with low HDL-C and elevated TG levels at baseline; CETP inhibition favoured modification towards normalization of the abnormally low neutral core lipid ratio (CE/TG) in all HDL particles including HDL2 and HDL3 subfractions. These findings support the contention that selective CETP inhibition favourably modulates the abnormal physicochemical properties of HDL2 and HDL3 particles in mixed dyslipidaemia, concomitantly enhancing both cholesterol efflux and selective hepatic uptake of HDL-CE (Figure 1).
In summary, available evidence indicates that torcetrapib-mediated inhibition of CETP does not induce dysfunction in HDL particles, but rather modifies their metabolism, structure, and physicochemical properties favouring normalization of anti-atherogenic functionality.
Dalcetrapib
Dalcetrapib is distinct from torcetrapib in the nature of its interaction with the CETP protein. Indeed, depending on the assay used, IC50 values for CETP activity have been estimated to be 0.4–10 µM for dalcetrapib compared with 19–79 nM for torcetrapib, clearly suggesting that plasma concentrations reached in clinical studies with dalcetrapib are unlikely to achieve complete inhibition of CETP.128 Dalcetrapib interacts with cysteine 13 residue in CETP, with high specificity for CETP over other SH-containing enzymes.129 Furthermore, unlike torcetrapib, dalcetrapib does not appear to induce the formation of a CETP–HDL complex at therapeutic plasma concentrations.128
The efficacy of dalcetrapib was initially demonstrated in cholesterol-fed rabbits.129 After 6-month treatment, dalcetrapib (mean dose 255 mg/kg/day) significantly increased HDL-C (+90%), with elevation in HDL2-C (+170%), HDL3-C (+59%), and apoA-I (+78%) (P < 0.01 for comparison of on-treatment levels vs. baseline). In addition, dalcetrapib treatment effected a 70% reduction in aortic arch lesions compared with controls.129 In a subsequent similar study, dalcetrapib elevated HDL-C levels but atheromatous area was not correlated with HDL-C levels or CETP activity.130
A Phase II, placebo-controlled, randomized study evaluated the efficacy and safety of dalcetrapib in 198 healthy subjects with mild hyperlipidaemia (HDL-C ≤ 60 mg/dL and TG ≤ 400 mg/dL).131 After 4 weeks, dalcetrapib (900 mg/day) significantly reduced CETP activity (–37%, P < 0.0001), increased HDL-C (+34%, P < 0.0001), and decreased LDL-C (–7%, P = 0.02), and in addition exerted a non-significant effect on apoA-I (+16%). Dalcetrapib was well tolerated, with no clinically significant changes in blood pressure. The efficacy and safety of dalcetrapib at doses of 300 and 600 mg/day were also assessed in a randomized, Phase II study conducted in 155 patients with type II hypercholesterolaemia (LDL-C > 160 mg/dL, HDL-C < 60 mg/dL, and TG < 400 mg/dL) receiving pravastatin (40 mg/day).132 After 4 weeks, dalcetrapib (600 mg/day) significantly reduced CETP activity by 30%, compared with baseline (P < 0.001). Significant increases in HDL-C were observed (up to 28%), reflecting significant elevations in HDL2-C and HDL3-C relative to baseline (P < 0.001).132 The combination of agents was well tolerated, with no clinically significant changes in blood pressure. Furthermore, in a recent analysis of four 4-week Phase IIa studies (including the two studies mentioned above) and one 12-week Phase IIb study in patients with type II hyperlipidaemia, CHD, or CHD risk equivalents, dalcetrapib was generally well tolerated and was not associated with clinically relevant elevations in blood pressure or cardiovascular adverse events at the doses studied.133 Finally, in a CETP-deficient rat model, dalcetrapib did not increase blood pressure or expression of RAAS-related genes.118
Several clinical trials are ongoing with the objective of evaluating the clinical efficacy and safety of dalcetrapib. One of these, dal-VESSEL, is focused on modulation of vascular function by CETP inhibition and will shed further light on the mechanisms implicated in the improved endothelial function which was recently observed in hypercholesterolaemic subjects with low baseline HDL-C subsequent to dalcetrapib treatment.134 The impact of dalcetrapib on atherosclerotic plaque development (dal-PLAQUE) has been initiated in some 100 patients with CHD using positron emission tomography/computerized tomography and magnetic resonance imaging.135 Finally, in order to evaluate the effects of dalcetrapib on mortality and morbidity, >15 600 high-risk CHD patients considered to have stable disease after a recent acute coronary syndrome event will be recruited into the ongoing dal-OUTCOMES trial.136 Patients will receive dalcetrapib on a background of optimized therapy for LDL-C reduction; importantly, no inclusion criterion for HDL-C level was set in this trial, thereby allowing assessment of the potential clinical benefit of HDL raising via CETP inhibition to be evaluated across a range of baseline HDL-C levels (Table 2).
Table 2.
Overview of the dal-OUTCOMES trial: a randomized, double-blind, placebo-controlled study assessing the effect of RO4607381 (dalcetrapib) 600 mg/d on cardiovascular mortality and morbidity in clinically stable patients with a recent acute coronary syndrome135
| Design | Criteria | Main outcomes |
|---|---|---|
| Phase III | Inclusion | Primary |
| Treatment (interventional) | Male/female adult patients ≥45 years of age | Time to first occurrence of any component of the composite cardiovascular event, cardiovascular mortality/morbidity (event driven) |
| Randomized | Recently hospitalized for acute coronary syndrome | Secondary |
| Double-blind (subject, investigator) | Clinically stable | Composite endpoint: all-cause mortality (event driven) |
| Placebo controlled | Receiving evidence-based medical and dietary management of dyslipidaemia | Change from baseline in blood lipids and lipoprotein levels (throughout study) |
| Parallel assignment | Exclusion | Adverse events, laboratory safety, vital signs, ECG (throughout study) |
| International | Uncontrolled diabetes | |
| Clinically unstable | ||
| Severe anaemia | ||
| Uncontrolled hypertension | ||
| Concomitant treatment with any other HDL-C-raising drug (e.g. niacin, fibrate) | ||
| Healthy volunteers |
ECG, electrocardiogram; HDL-C, high-density lipoprotein cholesterol.
Anacetrapib
Anacetrapib, like torcetrapib, induces tight reversible binding of CETP to HDL, with IC50 values for CETP of 15–57 nM.137
A Phase I randomized, placebo-controlled study assessed the efficacy and safety of anacetrapib in 50 patients with dyslipidaemia (LDL-C, 100–190 mg/dL).138 After 28-day treatment, anacetrapib produced dose-dependent lipid-altering effects; at the maximal dose tested, anacetrapib (300 mg/day) induced marked increments in HDL-C and apoA-I levels (+129 and +47%, respectively), with significant reduction in LDL-C (−38%) compared with placebo. In a second Phase I study of the effects of anacetrapib on 24 h ambulatory blood pressure over 10 days in 22 healthy individuals,138 similar profiles for systolic and diastolic blood pressure were observed for anacetrapib and placebo. These Phase I studies were short, involved a small number of patients, and were not powered to detect a difference in blood pressure of <6 mmHg. More recently, the efficacy and safety of anacetrapib were evaluated as monotherapy and in co-administration with atorvastatin (20 mg/day) in patients (n = 589) displaying either hypercholesterolaemia or mixed dyslipidaemia over an 8-week period; some 54% exhibited low HDL-C at baseline.139 For anacetrapib monotherapy, a dose-titration design revealed incremental reduction in LDL-C levels to –39% at the maximal 300 mg dose, with progressive elevation in HDL-C to +133% at this same dose. Co-administration of the CETP inhibitor with background statin therapy produced major incremental reductions in LDL-C attaining a maximal value at –70%; moreover, HDL-C elevations mediated by anacetrapib alone were maintained on co-administration of the two agents. Triglyceride levels at baseline exerted little effect on the dose-dependent reductions seen in LDL-C either in monotherapy or co-administration modes. The CETP inhibitor was well tolerated, no changes in blood pressure were noted, and the incidence of adverse effects was similar in placebo and active treatment groups. Further studies are now required to evaluate the long-term efficacy and safety of anacetrapib, both in monotherapy and in association with a statin. Indeed, the DEFINE study is ongoing and was designed to evaluate the lipid-lowering efficacy, tolerability, and safety of anacetrapib (100 mg/day) in normotriglyceridaemic subjects (n = 1623) with LDL-C < 100 mg/dL and HDL-C < 60 mg/dL on statin treatment over an 18-month period; here, the combination of statin background plus CETP inhibitor treatment is being compared with statin monotherapy.
Residual cardiovascular risk: validity of cholesteryl ester transfer protein as a therapeutic target
Despite recent genome-wide association scans identifying genetic variants influencing plasma lipid concentrations, and in the case of HDL-C those concerning the CETP gene,140 the use of gene therapy to improve the management of dyslipidaemia and reduce cardiovascular risk remains elusive. In the meantime, residual cardiovascular risk remains high even in patients treated with aggressive statin therapy,5–7 highlighting the need for add-on treatment to reduce the considerable cardiovascular event rate (Figure 3). Among risk factors other than LDL-C that are associated with atherogenic dyslipidaemia, a low level of HDL-C is now most recognized, especially as it is a key feature of common metabolic diseases (Figure 3).3,32,52 Moreover, the defective anti-atherogenic function of HDL particles in metabolic disease is now established and has become recognized as a therapeutic target of similar significance to that of HDL-C level.27,32,141 Such defective HDL function is intimately linked to the abnormal metabolism of TG-rich lipoproteins and is consistent with concomitant therapeutic correction of both these anomalies in order to reduce residual risk.142
Figure 3.
Cardiovascular risk remains high despite aggressive statin therapy. Statin treatment across a wide range of lipid phenotypes in patients at high cardiovascular risk has been highly successful in reducing relative risk by up to 45%. Nonetheless, major residual cardiovascular risk remains, part of which is due to non-modifiable risk factors but equally to modifiable risk factors. Atherogenic mixed dyslipidaemia is a frequent component of the latter, thereby suggesting that therapeutic attenuation of risk in this phenotype, which involves elevated levels of triglyceride-rich lipoproteins and small dense low-density lipoprotein, with subnormal levels of high-density lipoprotein cholesterol and apoA-I, would contribute to further reduction in residual risk across a wide range of metabolic disease states. ACS, acute coronary syndrome; CHD, coronary heart disease; LDL-C, LDL cholesterol.
The critical appraisal presented herein of the mechanisms of action implicated in the HDL-raising action of statins, fibrates, and niacin not only highlights but equally validates the central role of CETP in the modulation of perturbed lipid and cholesterol metabolism in dyslipidaemic subjects by these agents, particularly as it relates to HDL. Indeed, this evidence base substantiates the argument that CETP constitutes a preferential pharmacological target for HDL-raising therapies.
The direct and/or indirect actions of statins, fibrates, and niacin on the CETP system impact, to a significant degree, both the quantitative and the qualitative features not only of the atherogenic lipoproteins, but equally of their anti-atherogenic counterparts, the high-density particles. As discussed, these agents favour normalization of HDL and apoA-I levels to varying degrees as a function of baseline lipid phenotype, but may exert distinct structural, metabolic, and functional effects on the heterogeneous population of HDL particles. In addition to raising HDL levels, they equally may potentiate at least partial normalization of defective HDL function,27,127,144 but this question remains only partially resolved.
Who may benefit clinically from treatment with cholesteryl ester transfer protein inhibitors?
The pharmacological signature of CETP inhibitors and their impact on dysmetabolism characteristic of mixed dyslipidaemia, hypertriglyceridaemia, and hypercholesterolaemia suggests potential utility in treating common forms of dyslipidaemia associated with premature atherosclerosis.115,126,127,131,132,143,144 In particular, metabolic syndrome and type 2 diabetes may be ideal targets for intervention with CETP inhibitors, given the quantitative and qualitative anomalies of HDL particles in these insulin-resistant disease states (Figure 3).31,32,145
From a quantitative viewpoint, it is established that the relation of cardiovascular risk to HDL-C levels is especially steep in the range of 20–40 mg/dL, clearly indicating that therapeutic approaches targeted to HDL-C elevation may be critically important in many low HDL-C patients.146–148 Thus, the potent HDL-raising action of the CETP inhibitors would allow the clinician to efficaciously attain a potential HDL-C target of 40 mg/dL or higher in such patients, potentially affording major clinical benefit.
Qualitatively, and as a consequence of hypertriglyceridaemia and elevated CETP activity, functionally deficient HDL particles enriched in core TG and depleted in CE and apoA-I are formed intravascularly in both type 2 diabetic and metabolic syndrome patients.24,31,149,150 Thus, therapeutic normalization of both the quantity and the quality of HDL particles by CETP inhibitors constitutes a key target to efficaciously attenuate atherosclerosis in dyslipidaemic individuals with metabolic disease.
Statins, fibrates, and niacin attenuate plasma CETP activity principally by indirect mechanisms, and such effects are associated with favourable impact on both cholesterol homeostasis and the atherogenic process. In contrast, we do not fully understand the potential impact of partial, direct CETP inhibition on cholesterol homeostasis and atherosclerosis. Indeed, the therapeutic impact of such agents may vary as a function of individual metabolic phenotypes associated—or not—with insulin resistance. Long-term, large-scale morbi-mortality outcome trials are therefore essential to provide critical information on their efficacy, clinical benefit, and safety. Such clinical investigations are eagerly awaited, as the CETP inhibitors remain by far the most efficacious agents to raise HDL-C levels above the risk threshold range of ∼40–50 mg/dL across a wide range of lipid phenotypes.146
Funding
Drs T. Haigh and C.V. Felton (Prime Healthcare) conducted the literature search and provided excellent editorial assistance supported by an educational grant from F. Hoffmann La-Roche Ltd, Basel, Switzerland. Funding to pay the Open Access charge was provided by an educational grant from F. Hoffmann La-Roche Ltd. M.J.C. and A.K. gratefully acknowledge the award of a Contrat d'Interface of the Assistance Publique-Hopitaux de Paris/INSERM. The studies of the authors were supported by INSERM.
Conflict of interest: M.J.C. has received grant support from Pfizer, AstraZeneca, and MSD, and has been a consultant and member of the Speakers bureau of Pfizer, Abbott, AstraZeneca, MSD, and Merck-Schering Plough. A.K. was a recipient of an International HDL Award from Pfizer and grant funding from AstraZeneca. M.G. has received research grant funding from Pfizer and Sanofi-Aventis. W.L.G. has received grant support from MSD.
References
- 1.Libby P. The forgotten majority: unfinished business in cardiovascular risk reduction. J Am Coll Cardiol. 2005;46:1225–1228. doi: 10.1016/j.jacc.2005.07.006. [DOI] [PubMed] [Google Scholar]
- 2.Baigent C, Keech A, Kearney PM, Blackwell L, Buck G, Pollicino C, Kirby A, Sourjina T, Peto R, Collins R, Simes R Cholesterol Treatment Trialists' (CTT) Collaborators. Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet. 2005;366:1267–1278. doi: 10.1016/S0140-6736(05)67394-1. [DOI] [PubMed] [Google Scholar]
- 3.Chapman MJ. Therapeutic elevation of HDL-cholesterol to prevent atherosclerosis and coronary heart disease. Pharmacol Ther. 2006;111:893–908. doi: 10.1016/j.pharmthera.2006.02.003. [DOI] [PubMed] [Google Scholar]
- 4.Ridker PM, Danielson E, Fonseca FA, Genest J, Gotto AM, Jr, Kastelein JJ, Koenig W, Libby P, Lorenzatti AJ, MacFadyen JG, Nordestgaard BG, Shepherd J, Willerson JT, Glynn RJ JUPITER Study Group. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med. 2008;359:2195–2207. doi: 10.1056/NEJMoa0807646. [DOI] [PubMed] [Google Scholar]
- 5.Cannon CP, Braunwald E, McCabe CH, Rader DJ, Rouleau JL, Belder R, Joyal SV, Hill KA, Pfeffer MA, Skene AM Pravastatin or Atorvastatin Evaluation Infection Therapy-Thrombolysis in Myocardial Infarction 22 Investigators. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med. 2004;350:1495–1504. doi: 10.1056/NEJMoa040583. [DOI] [PubMed] [Google Scholar]
- 6.LaRosa JC, Grundy SM, Waters DD, Shear C, Barter P, Fruchart JC, Gotto AM, Greten H, Kastelein JJ, Shepherd J, Wenger NK Treating to New Targets (TNT) Investigators. Intensive lipid lowering with atorvastatin in patients with stable coronary disease. N Engl J Med. 2005;352:1425–1435. doi: 10.1056/NEJMoa050461. [DOI] [PubMed] [Google Scholar]
- 7.Singh IM, Shishehbor MH, Ansell BJ. High-density lipoprotein as a therapeutic target: a systematic review. JAMA. 2007;298:786–798. doi: 10.1001/jama.298.7.786. [DOI] [PubMed] [Google Scholar]
- 8.Miller M, Cannon CP, Murphy SA, Qin J, Ray KK, Braunwald E PROVE IT-TIMI 22 Investigators. Impact of triglyceride levels beyond low-density lipoprotein cholesterol after acute coronary syndrome in the PROVE IT-TIMI 22 trial. J Am Coll Cardiol. 2008;51:724–730. doi: 10.1016/j.jacc.2007.10.038. [DOI] [PubMed] [Google Scholar]
- 9.Wolfram RM, Brewer HB, Xue Z, Satler LF, Pichard AD, Kent KM, Waksman R. Impact of low high-density lipoproteins on in-hospital events and one-year clinical outcomes in patients with non-ST-elevation myocardial infarction acute coronary syndrome treated with drug-eluting stent implantation. Am J Cardiol. 2006;98:711–717. doi: 10.1016/j.amjcard.2006.04.006. [DOI] [PubMed] [Google Scholar]
- 10.Gordon T, Castelli WP, Hjortland MC, Kannel WB, Dawber TR. High density lipoprotein as a protective factor against coronary heart disease. The Framingham Study. Am J Med. 1977;62:707–714. doi: 10.1016/0002-9343(77)90874-9. [DOI] [PubMed] [Google Scholar]
- 11.Gordon DJ, Probstfield JL, Garrison RJ, Neaton JD, Castelli WP, Knoke JD, Jacobs DR, Jr, Bangdiwala S, Tyroler HA. High-density lipoprotein cholesterol and cardiovascular disease. Four prospective American studies. Circulation. 1989;79:8–15. doi: 10.1161/01.cir.79.1.8. [DOI] [PubMed] [Google Scholar]
- 12.Assmann G, Schulte H, von Eckardstein A, Huang Y. High-density lipoprotein cholesterol as a predictor of coronary heart disease risk. The PROCAM experience and pathophysiological implications for reverse cholesterol transport. Atherosclerosis. 1996;124(Suppl.):S11–S20. doi: 10.1016/0021-9150(96)05852-2. [DOI] [PubMed] [Google Scholar]
- 13.Sharrett AR, Ballantyne CM, Coady SA, Heiss G, Sorlie PD, Catellier D, Patsch W Atherosclerosis Risk in Communities Study Group. Coronary heart disease prediction from lipoprotein cholesterol levels, triglycerides, lipoprotein(a), apolipoproteins A-I and B, and HDL density subfractions: the Atherosclerosis Risk in Communities (ARIC) Study. Circulation. 2001;104:1108–1113. doi: 10.1161/hc3501.095214. [DOI] [PubMed] [Google Scholar]
- 14.Luc G, Bard JM, Ferrières J, Evans A, Amouyel P, Arveiler D, Fruchart JC, Ducimetière P. Value of HDL cholesterol, apolipoprotein A-I, lipoprotein A-I, and lipoprotein A-I/A-II in prediction of coronary heart disease: the PRIME Study. Prospective Epidemiological Study of Myocardial Infarction. Arterioscler Thromb Vasc Biol. 2002;22:1155–1161. doi: 10.1161/01.atv.0000022850.59845.e0. [DOI] [PubMed] [Google Scholar]
- 15.Birjmohun RS, Dallinga-Thie GM, Kuivenhoven JA, Stroes ESG, Otvos JD, Wareham NJ, Luben R, Kastelein JJP, Khaw K-T, Boekholdt SM. Apolipoprotein A-II is inversely associated with risk of future coronary artery disease. Circulation. 2007;116:2029–2035. doi: 10.1161/CIRCULATIONAHA.107.704031. [DOI] [PubMed] [Google Scholar]
- 16.van der Steeg WA, Holme I, Boekholdt SM, Larsen ML, Lindahl C, Stroes ES, Tikkanen MJ, Wareham NJ, Faergeman O, Olsson AG, Pedersen TR, Khaw KT, Kastelein JJ. High-density lipoprotein cholesterol, high-density lipoprotein particle size, and apolipoprotein A-I: significance for cardiovascular risk: the IDEAL and EPIC-Norfolk studies. J Am Coll Cardiol. 2008;51:634–642. doi: 10.1016/j.jacc.2007.09.060. [DOI] [PubMed] [Google Scholar]
- 17.Briel M, Ferreira-Gonzalez I, You JJ, Karanicolas PJ, Akl EA, Wu P, Blechacz B, Bassler D, Wei X, Sharman A, Whitt I, Alves da Silva S, Khalid Z, Nordmann AJ, Zhou Q, Walter SD, Vale N, Bhatnagar N, O'Regan C, Mills EJ, Bucher HC, Montori VM, Guyatt GH. Association between change in high density lipoprotein cholesterol and cardiovascular disease morbidity and mortality: systematic review and meta-regression analysis. BMJ. 2009;338:92. doi: 10.1136/bmj.b92. doi:10.1136/bmj.b92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Goldenberg I, Boyko V, Tennenbaum A, Tanne D, Behar S, Guetta V. Long-term benefit of high-density lipoprotein cholesterol-raising therapy with bezafibrate. Arch Intern Med. 2009;169:508–514. doi: 10.1001/archinternmed.2008.584. [DOI] [PubMed] [Google Scholar]
- 19.Nissen SE, Nicholls SJ, Sipahi I, Libby P, Raichlen JS, Ballantyne CM, Davignon J, Erbel R, Fruchart JC, Tardif JC, Schoenhagen P, Crowe T, Cain V, Wolski K, Goormastic M, Tuzcu EM ASTEROID Investigators. Effect of very high-intensity statin therapy on regression of coronary atherosclerosis: the ASTEROID trial. JAMA. 2006;295:1556–1565. doi: 10.1001/jama.295.13.jpc60002. [DOI] [PubMed] [Google Scholar]
- 20.Taylor AJ, Lee HJ, Sullenberger LE. The effect of 24 months of combination statin and extended-release niacin on carotid intima-media thickness: ARBITER 3. Curr Med Res Opin. 2006;22:2243–2250. doi: 10.1185/030079906x148508. [DOI] [PubMed] [Google Scholar]
- 21.Nicholls SJ, Tuzcu EM, Sipahi I, Grasso AW, Schoenhagen P, Hu T, Wolski K, Crowe T, Desai MY, Hazen SL, Kapadia SR, Nissen SE. Statins, high-density lipoprotein cholesterol, and regression of coronary atherosclerosis. JAMA. 2007;297:499–508. doi: 10.1001/jama.297.5.499. [DOI] [PubMed] [Google Scholar]
- 22.Brown BG, Zhao XQ. Nicotinic acid, alone and in combinations, for reduction of cardiovascular risk. Am J Cardiol. 2008;101:58B–62B. doi: 10.1016/j.amjcard.2008.02.039. [DOI] [PubMed] [Google Scholar]
- 23.Nissen SE, Tsunoda T, Tuzcu EM, Schoenhagen P, Cooper CJ, Yasin M, Eaton GM, Lauer MA, Sheldon WS, Grines CL, Halpern S, Crowe T, Blankenship JC, Kerensky R. Effect of recombinant ApoA-I Milano on coronary atherosclerosis in patients with acute coronary syndromes: a randomized controlled trial. JAMA. 2003;5:2292–2300. doi: 10.1001/jama.290.17.2292. [DOI] [PubMed] [Google Scholar]
- 24.Shaw JA, Bobik A, Murphy A, Kanellakis P, Blombery P, Mukhamedova N, Woollard K, Lyon S, Sviridov D, Dart AM. Infusion of reconstituted high-density lipoprotein leads to acute changes in human atherosclerotic plaque. Circ Res. 2008;103:1084–1091. doi: 10.1161/CIRCRESAHA.108.182063. [DOI] [PubMed] [Google Scholar]
- 25.Barter P, Gotto AM, LaRosa JC, Maroni J, Szarek M, Grundy SM, Kastelein JJ, Bittner V, Fruchart JC Treating to New Targets Investigators. HDL cholesterol, very low levels of LDL cholesterol, and cardiovascular events. N Engl J Med. 2007;57:1301–1310. doi: 10.1056/NEJMoa064278. [DOI] [PubMed] [Google Scholar]
- 26.Tall AR. Plasma high density lipoproteins. Metabolism and relationship to atherogenesis. J Clin Invest. 1990;86:379–384. doi: 10.1172/JCI114722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kontush A, Chapman MJ. Functionally defective high-density lipoprotein: a new therapeutic target at the crossroads of dyslipidemia, inflammation, and atherosclerosis. Pharmacol Rev. 2006;58:342–374. doi: 10.1124/pr.58.3.1. [DOI] [PubMed] [Google Scholar]
- 28.Davidson WS, Silva RA, Chantepie S, Lagor WR, Chapman MJ, Kontush A. Proteomic analysis of defined HDL subpopulations reveals particle-specific protein clusters: relevance to antioxidative function. Arterioscler Thromb Vasc Biol. 2009;29:870–876. doi: 10.1161/ATVBAHA.109.186031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Vaisar T, Pennathur S, Green PS, Gharib SA, Hoofnagle AN, Cheung MC, Byun J, Vuletic S, Kassim S, Singh P, Chea H, Knopp RH, Brunzell J, Geary R, Chait A, Zhao XQ, Elkon K, Marcovina S, Ridker P, Oram JF, Heinecke JW. Shotgun proteomics implicates protease inhibition and complement activation in the antiinflammatory properties of HDL. J Clin Invest. 2007;117:746–756. doi: 10.1172/JCI26206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Assmann G, Nofer JR. Atheroprotective effects of high-density lipoproteins. Annu Rev Med. 2003;54:321–341. doi: 10.1146/annurev.med.54.101601.152409. [DOI] [PubMed] [Google Scholar]
- 31.Kontush A, Chapman MJ. Antiatherogenic small, dense HDL-guardian angel of the arterial wall? Nat Clin Pract Cardiovasc Med. 2006;3:144–153. doi: 10.1038/ncpcardio0500. [DOI] [PubMed] [Google Scholar]
- 32.Joy T, Hegele RA. Is raising HDL a futile strategy for atheroprotection? Nat Rev Drug Discov. 2008;7:143–155. doi: 10.1038/nrd2489. [DOI] [PubMed] [Google Scholar]
- 33.Chapman MJ, Assmann G, Fruchart JC, Shepherd J, Sirtori C European Consensus Panel on HDL-C. Raising high-density lipoprotein cholesterol with reduction of cardiovascular risk: the role of nicotinic acid—a position paper developed by the European Consensus Panel on HDL-C. Curr Med Res Opin. 2004;20:1253–1268. doi: 10.1185/030079904125004402. [DOI] [PubMed] [Google Scholar]
- 34.McGovern M. Niaspan®: creating a new concept for raising HDL-cholesterol. Eur Heart J Suppl. 2005;7:F41–F47. [Google Scholar]
- 35.Gille A, Bodor ET, Ahmed K, Offermanns S. Nicotinic acid: pharmacological effects and mechanisms of action. Annu Rev Pharmacol Toxicol. 2008;48:79–106. doi: 10.1146/annurev.pharmtox.48.113006.094746. [DOI] [PubMed] [Google Scholar]
- 36.Paolini JF, Mitchel YB, Reyes R, Kher U, Lai E, Watson DJ, Norquist JM, Meehan AG, Bays HE, Davidson M, Ballantyne CM. Effects of laropiprant on nicotinic acid-induced flushing in patients with dyslipidemia. Am J Cardiol. 2008;101:625–630. doi: 10.1016/j.amjcard.2007.10.023. [DOI] [PubMed] [Google Scholar]
- 37.McTaggart F, Jones P. Effects of statins on high-density lipoproteins: a potential contribution to cardiovascular benefit. Cardiovasc Drugs Ther. 2008;22:321–338. doi: 10.1007/s10557-008-6113-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Barter PJ, Brandrup-Wognsen G, Palmer MK, Nicholls SJ. Effects of statins on the concentration of high-density lipoprotein cholesterol and its relation to change in low-density lipoprotein cholesterol: results from the VOYAGER database. J Am Coll Cardiol. 2009;53(Suppl. A):1021–1087. Abstract. [Google Scholar]
- 39.Birjmohun RS, Hutten BA, Kastelein JJ, Stroes ES. Efficacy and safety of high-density lipoprotein cholesterol-increasing compounds: a meta-analysis of randomized controlled trials. J Am Coll Cardiol. 2005;45:185–197. doi: 10.1016/j.jacc.2004.10.031. [DOI] [PubMed] [Google Scholar]
- 40.Chapman MJ. Fibrates in 2003: therapeutic action in atherogenic dyslipidaemia and future perspectives. Atherosclerosis. 2003;171:1–13. doi: 10.1016/s0021-9150(03)00156-4. [DOI] [PubMed] [Google Scholar]
- 41.Robins SJ, Collins D, Wittes JT, Papademetriou V, Deedwania PC, Schaefer EJ, McNamara JR, Kashyap ML, Hershman JM, Wexler LF, Rubins HB VA-HIT Study Group. Veterans Affairs High-Density Lipoprotein Intervention Trial. Relation of gemfibrozil treatment and lipid levels with major coronary events: VA-HIT: a randomized controlled trial. JAMA. 2001;285:1585–1591. doi: 10.1001/jama.285.12.1585. [DOI] [PubMed] [Google Scholar]
- 42.Caslake MJ, Stewart G, Day SP, Daly E, McTaggart F, Chapman MJ, Durrington P, Laggner P, Mackness M, Pears J, Packard CJ. Phenotype-dependent and -independent actions of rosuvastatin on atherogenic lipoprotein subfractions in hyperlipidaemia. Atherosclerosis. 2003;171:245–253. doi: 10.1016/j.atherosclerosis.2003.08.025. [DOI] [PubMed] [Google Scholar]
- 43.Guérin M, Lassel TS, Le Goff W, Farnier M, Chapman MJ. Action of atorvastatin in combined hyperlipidemia: preferential reduction of cholesteryl ester transfer from HDL to VLDL1 particles. Arterioscler Thromb Vasc Biol. 2000;20:189–197. doi: 10.1161/01.atv.20.1.189. [DOI] [PubMed] [Google Scholar]
- 44.Inazu A, Brown ML, Hesler CB, Agellon LB, Koizumi J, Takata K, Maruhama Y, Mabuchi H, Tall AR. Increased high-density lipoprotein levels caused by a common cholesteryl-ester transfer protein gene mutation. N Engl J Med. 1990;323:1234–1238. doi: 10.1056/NEJM199011013231803. [DOI] [PubMed] [Google Scholar]
- 45.Le Goff W, Guerin M, Chapman MJ. Pharmacological modulation of cholesterol ester transfer protein, a new therapeutic target in atherogenic dyslipidemia. Pharmacol Ther. 2004;101:17–38. doi: 10.1016/j.pharmthera.2003.10.001. [DOI] [PubMed] [Google Scholar]
- 46.Curb JD, Abbott RD, Rodriguez BL, Masaki K, Chen R, Sharp DS, Tall AR. A prospective study of HDL-C and cholesteryl ester transfer protein gene mutations and the risk of coronary heart disease in the elderly. J Lipid Res. 2004;45:948–953. doi: 10.1194/jlr.M300520-JLR200. [DOI] [PubMed] [Google Scholar]
- 47.Barter PJ, Kastelein JJ. Targeting cholesteryl ester transfer protein for the prevention and management of cardiovascular disease. J Am Coll Cardiol. 2006;47:492–499. doi: 10.1016/j.jacc.2005.09.042. [DOI] [PubMed] [Google Scholar]
- 48.Schaefer EJ, Asztalos BF. Increasing high-density lipoprotein cholesterol, inhibition of cholesteryl ester transfer protein, and heart disease risk reduction. Am J Cardiol. 2007;100(Suppl.):25N–31N. doi: 10.1016/j.amjcard.2007.08.010. [DOI] [PubMed] [Google Scholar]
- 49.Barter PJ, Brewer HB, Jr, Chapman MJ, Hennekens CH, Rader DJ, Tall AR. Cholesteryl ester transfer protein: a novel target for raising HDL and inhibiting atherosclerosis. Arterioscler Thromb Vasc Biol. 2003;23:160–167. doi: 10.1161/01.atv.0000054658.91146.64. [DOI] [PubMed] [Google Scholar]
- 50.Lewis GF, Rader DJ. New insights into the regulation of HDL metabolism and reverse cholesterol transport. Circ Res. 2005;96:1221–1232. doi: 10.1161/01.RES.0000170946.56981.5c. [DOI] [PubMed] [Google Scholar]
- 51.Schwartz CC, VandenBroek JM, Cooper PS. Lipoprotein cholesteryl ester production, transfer, and output in vivo in humans. J Lipid Res. 2004;45:1594–1607. doi: 10.1194/jlr.M300511-JLR200. [DOI] [PubMed] [Google Scholar]
- 52.Brewer HB., Jr High-density lipoproteins: a new potential therapeutic target for the prevention of cardiovascular disease. Arterioscler Thromb Vasc Biol. 2004;24:387–391. doi: 10.1161/01.ATV.0000121505.88326.d2. [DOI] [PubMed] [Google Scholar]
- 53.Tall AR. Plasma cholesteryl ester transfer protein. J Lipid Res. 1993;34:1255–1274. [PubMed] [Google Scholar]
- 54.Williams KJ, Tabas I. The response-to-retention hypothesis of early atherogenesis. Arterioscler Thromb Vasc Biol. 1995;15:551–561. doi: 10.1161/01.atv.15.5.551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lassel TS, Guérin M, Auboiron S, Chapman MJ, Guy-Grand B. Preferential cholesteryl ester acceptors among triglyceride-rich lipoproteins during alimentary lipemia in normolipidemic subjects. Arterioscler Thromb Vasc Biol. 1998;18:65–74. doi: 10.1161/01.atv.18.1.65. [DOI] [PubMed] [Google Scholar]
- 56.Lamarche B, Uffelman KD, Carpentier A, Cohn JS, Steiner G, Barrett PH, Lewis GF. Triglyceride enrichment of HDL enhances in vivo metabolic clearance of HDL apo A-I in healthy men. J Clin Invest. 1999;103:1191–1199. doi: 10.1172/JCI5286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lund-Katz S, Laplaud PM, Phillips MC, Chapman MJ. Apolipoprotein B-100 conformation and particle surface charge in human LDL subspecies: implication for LDL receptor interaction. Biochemistry. 1998;37:12867–12874. doi: 10.1021/bi980828m. [DOI] [PubMed] [Google Scholar]
- 58.Simpson HS, Williamson CM, Olivecrona T, Pringle S, Maclean J, Lorimer AR, Bonnefous F, Bogaievsky Y, Packard CJ, Shepherd J. Postprandial lipemia, fenofibrate and coronary artery disease. Atherosclerosis. 1990;85:193–202. doi: 10.1016/0021-9150(90)90111-u. [DOI] [PubMed] [Google Scholar]
- 59.Guérin M, Egger P, Soudant C, Le Goff W, van Tol A, Dupuis R, Chapman MJ. Cholesteryl ester flux from HDL to VLDL-1 is preferentially enhanced in type IIB hyperlipidemia in the postprandial state. J Lipid Res. 2002;43:1652–1660. doi: 10.1194/jlr.m200135-jlr200. [DOI] [PubMed] [Google Scholar]
- 60.Nordestgaard BG, Benn M, Schnohr P, Tybjaerg-Hansen A. Nonfasting triglycerides and risk of myocardial infarction, ischemic heart disease, and death in men and women. JAMA. 2007;298:299–308. doi: 10.1001/jama.298.3.299. [DOI] [PubMed] [Google Scholar]
- 61.Bansal S, Buring JE, Rifai N, Mora S, Sacks FM, Ridker PM. Fasting compared with nonfasting triglycerides and risk of cardiovascular events in women. JAMA. 2007;298:309–316. doi: 10.1001/jama.298.3.309. [DOI] [PubMed] [Google Scholar]
- 62.Williams KJ. Molecular processes that handle—and mishandle—dietary lipids. J Clin Invest. 2008;118:3247–3259. doi: 10.1172/JCI35206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Castro GR, Fielding CJ. Effects of postprandial lipemia on plasma cholesterol metabolism. J Clin Invest. 1985;75:874–882. doi: 10.1172/JCI111786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tall A, Sammett D, Granot E. Mechanisms of enhanced cholesteryl ester transfer from high density lipoproteins to apolipoprotein B-containing lipoproteins during alimentary lipemia. J Clin Invest. 1986;77:1163–1172. doi: 10.1172/JCI112417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Contacos C, Barter PJ, Vrga L, Sullivan DR. Cholesteryl ester transfer in hypercholesterolaemia: fasting and postprandial studies with and without pravastatin. Atherosclerosis. 1998;141:87–98. doi: 10.1016/s0021-9150(98)00151-8. [DOI] [PubMed] [Google Scholar]
- 66.Rashid S, Barrett PH, Uffelman KD, Watanabe T, Adeli K, Lewis GF. Lipolytically modified triglyceride-enriched HDLs are rapidly cleared from the circulation. Arterioscler Thromb Vasc Biol. 2002;22:483–487. doi: 10.1161/hq0302.105374. [DOI] [PubMed] [Google Scholar]
- 67.Brewer HB., Jr Benefit-risk assessment of rosuvastatin 10 to 40 milligrams. Am J Cardiol. 2003;92:23K–29K. doi: 10.1016/s0002-9149(03)00779-3. [DOI] [PubMed] [Google Scholar]
- 68.Guérin M, Dolphin PJ, Talussot C, Gardette J, Berthézène F, Chapman MJ. Pravastatin modulates cholesteryl ester transfer from HDL to apoB-containing lipoproteins and lipoprotein subspecies profile in familial hypercholesterolemia. Arterioscler Thromb Vasc Biol. 1995;15:1359–1368. doi: 10.1161/01.atv.15.9.1359. [DOI] [PubMed] [Google Scholar]
- 69.Guérin M, Bruckert E, Dolphin PJ, Turpin G, Chapman MJ. Fenofibrate reduces plasma cholesteryl ester transfer from HDL to VLDL and normalizes the atherogenic, dense LDL profile in combined hyperlipidemia. Arterioscler Thromb Vasc Biol. 1996;16:763–772. doi: 10.1161/01.atv.16.6.763. [DOI] [PubMed] [Google Scholar]
- 70.Guérin M, Le Goff W, Frisdal E, Schneider S, Milosavljevic D, Bruckert E, Chapman MJ. Action of ciprofibrate in type IIb hyperlipoproteinemia: modulation of the atherogenic lipoprotein phenotype and stimulation of high-density lipoprotein-mediated cellular cholesterol efflux. J Clin Endocrinol Metab. 2003;88:3738–3746. doi: 10.1210/jc.2003-030191. [DOI] [PubMed] [Google Scholar]
- 71.Guérin M, Le Goff W, Lassel TS, Van Tol A, Steiner G, Chapman MJ. Atherogenic role of elevated CE transfer from HDL to VLDL(1) and dense LDL in type 2 diabetes: impact of the degree of triglyceridemia. Arterioscler Thromb Vasc Biol. 2001;21:282–288. doi: 10.1161/01.atv.21.2.282. [DOI] [PubMed] [Google Scholar]
- 72.Schaefer JR, Schweer H, Ikewaki K, Stracke H, Seyberth HJ, Kaffarnik H, Maisch B, Steinmetz A. Metabolic basis of high density lipoproteins and apolipoprotein A-I increase by HMG-CoA reductase inhibition in healthy subjects and a patient with coronary artery disease. Atherosclerosis. 1999;144:177–184. doi: 10.1016/s0021-9150(99)00053-2. [DOI] [PubMed] [Google Scholar]
- 73.Oliveira HC, Chouinard RA, Agellon LB, Bruce C, Ma L, Walsh A, Breslow JL, Tall AR. Human cholesteryl ester transfer protein gene proximal promoter contains dietary cholesterol positive responsive elements and mediates expression in small intestine and periphery while predominant liver and spleen expression is controlled by 5′-distal sequences. Cis-acting sequences mapped in transgenic mice. J Biol Chem. 1996;271:31831–31838. doi: 10.1074/jbc.271.50.31831. [DOI] [PubMed] [Google Scholar]
- 74.Gauthier B, Robb M, Gaudet F, Ginsburg GS, McPherson R. Characterization of a cholesterol response element (CRE) in the promoter of the cholesteryl ester transfer protein gene: functional role of the transcription factors SREBP-1a, -2, and YY1. J Lipid Res. 1999;40:1284–1293. [PubMed] [Google Scholar]
- 75.Martin G, Duez H, Blanquart C, Berezowski V, Poulain P, Fruchart JC, Najib-Fruchart J, Glineur C, Staels B. Statin-induced inhibition of the Rho signaling pathway activates PPARalpha and induces HDL apoA-I. J Clin Invest. 2001;107:1423–1432. doi: 10.1172/JCI10852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Asztalos BF, Horvath KV, McNamara JR, Roheim PS, Rubinstein JJ, Schaefer EJ. Comparing the effects of five different statins on the HDL subpopulation profiles of coronary heart disease patients. Atherosclerosis. 2002;164:361–369. doi: 10.1016/s0021-9150(02)00149-1. [DOI] [PubMed] [Google Scholar]
- 77.Asztalos BF, Le Maulf F, Dallal GE, Stein E, Jones PH, Horvath KV, McTaggart F, Schaefer EJ. Comparison of the effects of high doses of rosuvastatin versus atorvastatin on the subpopulations of high density lipoproteins. Am J Cardiol. 2007;99:681–685. doi: 10.1016/j.amjcard.2006.09.117. [DOI] [PubMed] [Google Scholar]
- 78.Streja L, Packard CJ, Shepherd J, Cobbe S, Ford I WOSCOPS Group. Factors affecting low-density lipoprotein and high density lipoprotein cholesterol response to pravastatin in the West of Scotland Coronary Prevention Study (WOSCOPS) Am J Cardiol. 2002;90:731–736. doi: 10.1016/s0002-9149(02)02599-7. [DOI] [PubMed] [Google Scholar]
- 79.Ashen MD, Blumenthal RS Clinical practice. Low HDL cholesterol levels. N Engl J Med. 2005;353:1252–1260. doi: 10.1056/NEJMcp044370. [DOI] [PubMed] [Google Scholar]
- 80.Berk-Planken II, Hoogerbrugge N, Stolk RP, Bootsma AH, Jansen H DALI Study Group. Atorvastatin dose-dependently decreases hepatic lipase activity in type 2 diabetes: effect of sex and the LIPC promoter variant. Diabetes Care. 2003;26:427–432. doi: 10.2337/diacare.26.2.427. [DOI] [PubMed] [Google Scholar]
- 81.Coronary Drug Project Research Group. Clofibrate and niacin in coronary heart disease. JAMA. 1975;231:360–381. [PubMed] [Google Scholar]
- 82.Frick MH, Elo O, Haapa K, Heinonen OP, Heinsalmi P, Helo P, Huttunen JK, Kaitaniemi P, Koskinen P, Manninen V, Mäenpää H, Mälkönen M, Mänttäri M, Norola S, Pasternack A, Pikkarainen J, Romo M, Sjöblom T, Nikkilä EA. Helsinki Heart Study: primary-prevention trial with gemfibrozil in middle-aged men with dyslipidemia. Safety of treatment, changes in risk factors, and incidence of coronary heart disease. N Engl J Med. 1987;317:1237–1245. doi: 10.1056/NEJM198711123172001. [DOI] [PubMed] [Google Scholar]
- 83.Rubins HB, Robins SJ, Collins D, Fye CL, Anderson JW, Elam MB, Faas FH, Linares E, Schaefer EJ, Schectman G, Wilt TJ, Wittes J. Gemfibrozil for the secondary prevention of coronary heart disease in men with low levels of high-density lipoprotein cholesterol. Veterans Affairs High-Density Lipoprotein Cholesterol Intervention Trial Study Group. N Engl J Med. 1999;341:410–418. doi: 10.1056/NEJM199908053410604. [DOI] [PubMed] [Google Scholar]
- 84.Willson TM, Brown PJ, Sternbach DD, Henke BR. The PPARs: from orphan receptors to drug discovery. J Med Chem. 2000;43:527–550. doi: 10.1021/jm990554g. [DOI] [PubMed] [Google Scholar]
- 85.Duez H, Lefebvre B, Poulain P, Torra IP, Percevault F, Luc G, Peters JM, Gonzalez FJ, Gineste R, Helleboid S, Dzavik V, Fruchart JC, Fiévet C, Lefebvre P, Staels B. Regulation of human apoA-I by gemfibrozil and fenofibrate through selective peroxisome proliferator-activated receptor alpha modulation. Arterioscler Thromb Vasc Biol. 2005;25:585–591. doi: 10.1161/01.ATV.0000154140.73570.00. [DOI] [PubMed] [Google Scholar]
- 86.Hausenloy DJ, Yellon DM. Targeting residual cardiovascular risk: raising high-density lipoprotein cholesterol levels. Heart. 2008;94:706–714. doi: 10.1136/hrt.2007.125401. [DOI] [PubMed] [Google Scholar]
- 87.Packard CJ, Shepherd J. Lipoprotein heterogeneity and apolipoprotein B metabolism. Arterioscler Thromb Vasc Biol. 1997;17:3542–3556. doi: 10.1161/01.atv.17.12.3542. [DOI] [PubMed] [Google Scholar]
- 88.Schonfeld G. The effects of fibrates on lipoprotein and hemostatic coronary risk factors. Atherosclerosis. 1994;111:161–174. doi: 10.1016/0021-9150(94)90090-6. [DOI] [PubMed] [Google Scholar]
- 89.Staels B, Dallongeville J, Auwerx J, Schoonjans K, Leitersdorf E, Fruchart JC. Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation. 1998;98:2088–2093. doi: 10.1161/01.cir.98.19.2088. [DOI] [PubMed] [Google Scholar]
- 90.Watts GF, Barrett PH, Ji J, Serone AP, Chan DC, Croft KD, Loehrer F, Johnson Differential regulation of lipoprotein kinetics by atorvastatin and fenofibrate in subjects with the metabolic syndrome. Diabetes. 2003;52:803–811. doi: 10.2337/diabetes.52.3.803. [DOI] [PubMed] [Google Scholar]
- 91.Barter PJ, Rye KA. Is there a role for fibrates in the management of dyslipidemia in the metabolic syndrome? Arterioscler Thromb Vasc Biol. 2008;28:39–46. doi: 10.1161/ATVBAHA.107.148817. [DOI] [PubMed] [Google Scholar]
- 92.Zambon A, Gervois P, Pauletto P, Fruchart JC, Staels B. Modulation of hepatic inflammatory risk markers of cardiovascular diseases by PPAR-alpha activators: clinical and experimental evidence. Arterioscler Thromb Vasc Biol. 2006;26:977–986. doi: 10.1161/01.ATV.0000204327.96431.9a. [DOI] [PubMed] [Google Scholar]
- 93.Chinetti G, Lestavel S, Bocher V, Remaley AT, Neve B, Torra IP, Teissier E, Minnich A, Jaye M, Duverger N, Brewer HB, Fruchart JC, Clavey V, Staels B. PPAR-alpha and PPAR-gamma activators induce cholesterol removal from human macrophage foam cells through stimulation of the ABCA1 pathway. Nat Med. 2001;7:53–58. doi: 10.1038/83348. [DOI] [PubMed] [Google Scholar]
- 94.Poulter N. The impact of micronized fenofibrate on lipid subfractions and on reaching HDL-target levels in 7,098 patients with dyslipidaemia. Br J Cardiol. 1999;6:682–685. [Google Scholar]
- 95.Diabetes Atherosclerosis Intervention Study (DAIS) Group. Effect of fenofibrate on progression of coronary-artery disease in type 2 diabetes: the Diabetes Atherosclerosis Intervention Study, a randomised study. Lancet. 2001;357:905–910. Erratum in: Lancet 2001;357:1890. [PubMed] [Google Scholar]
- 96.Vakkilainen J, Steiner G, Ansquer JC, Aubin F, Rattier S, Foucher C, Hamsten A, Taskinen MR DAIS Group. Relationships between low-density lipoprotein particle size, plasma lipoproteins, and progression of coronary artery disease: the Diabetes Atherosclerosis Intervention Study (DAIS) Circulation. 2003;107:1733–1737. doi: 10.1161/01.CIR.0000057982.50167.6E. [DOI] [PubMed] [Google Scholar]
- 97.Keech A, Simes RJ, Barter P, Best J, Scott R, Taskinen MR, Forder P, Pillai A, Davis T, Glasziou P, Drury P, Kesäniemi YA, Sullivan D, Hunt D, Colman P, d'Emden M, Whiting M, Ehnholm C, Laakso M FIELD study investigators. Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet. 2005;366:1849–1861. doi: 10.1016/S0140-6736(05)67667-2. Erratum in: Lancet 2006;368:1415, 1420. [DOI] [PubMed] [Google Scholar]
- 98.Saha SA, Kizhakepunnur LG, Bahekar A, Arora RR. The role of fibrates in the prevention of cardiovascular disease—a pooled meta-analysis of long-term randomized placebo-controlled clinical trials. Am Heart J. 2007;154:943–953. doi: 10.1016/j.ahj.2007.07.011. [DOI] [PubMed] [Google Scholar]
- 99.Fazio S, Linton MF. The role of fibrates in managing hyperlipidemia: mechanisms of action and clinical efficacy. Curr Atheroscler Rep. 2004;6:148–157. doi: 10.1007/s11883-004-0104-8. [DOI] [PubMed] [Google Scholar]
- 100.Scott R, O'Brien R, Fulcher G, Pardy C, D'Emden M, Tse D, Taskinen MR, Ehnholm C, Keech A Fenofibrate Intervention Event Lowering in Diabetes (FIELD) Study Investigators. Effects of fenofibrate treatment on cardiovascular disease risk in 9,795 individuals with type 2 diabetes and various components of the metabolic syndrome: the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study. Diabetes Care. 2009;32:493–498. doi: 10.2337/dc08-1543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Carlson LA. Nicotinic acid: the broad-spectrum lipid drug. A 50th Anniversary review. J Intern Med. 2005;258:94–114. doi: 10.1111/j.1365-2796.2005.01528.x. [DOI] [PubMed] [Google Scholar]
- 102.Lamon-Fava S, Diffenderfer MR, Barrett PH, Buchsbaum A, Nyaku M, Horvath KV, Asztalos BF, Otokozawa S, Ai M, Matthan NR, Lichtenstein AH, Dolnikowski GG, Schaefer EJ. Extended-release niacin alters the metabolism of plasma apolipoprotein (Apo) A-I and ApoB-containing lipoproteins. Arterioscler Thromb Vasc Biol. 2008;28:1672–1678. doi: 10.1161/ATVBAHA.108.164541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Wang W, Basinger A, Neese RA, Shane B, Myong SA, Christiansen M, Hellerstein MK. Effect of nicotinic acid administration on hepatic very low density lipoprotein-triglyceride production. Am J Physiol Endocrinol Metab. 2001;280:E540–E547. doi: 10.1152/ajpendo.2001.280.3.E540. [DOI] [PubMed] [Google Scholar]
- 104.Watts GF, Chan DC. Of mice and men: blowing away the cobwebs from the mechanism of action of niacin on HDL metabolism. Arterioscler Thromb Vasc Biol. 2008;28:1892–1895. doi: 10.1161/ATVBAHA.108.175224. [DOI] [PubMed] [Google Scholar]
- 105.van der Hoorn JW, de Haan W, Berbée JF, Havekes LM, Jukema JW, Rensen PC, Princen HM. Niacin increases HDL by reducing hepatic expression and plasma levels of cholesteryl ester transfer protein in APOE*3Leiden.CETP mice. Arterioscler Thromb Vasc Biol. 2008;28:2016–2022. doi: 10.1161/ATVBAHA.108.171363. [DOI] [PubMed] [Google Scholar]
- 106.Hernandez M, Wright SD, Cai TQ. Critical role of cholesterol ester transfer protein in nicotinic acid-mediated HDL elevation in mice. Biochem Biophys Res Commun. 2007;355:1075–1080. doi: 10.1016/j.bbrc.2007.02.079. [DOI] [PubMed] [Google Scholar]
- 107.Zhang LH, Kamanna VS, Zhang MC, Kashyap ML. Niacin inhibits surface expression of ATP synthase beta chain in HepG2 cells: implications for raising HDL. J Lipid Res. 2008;49:1195–1201. doi: 10.1194/jlr.M700426-JLR200. [DOI] [PubMed] [Google Scholar]
- 108.Kamanna VS, Kashyap ML. Mechanism of action of niacin. Am J Cardiol. 2008;101:20B–26B. doi: 10.1016/j.amjcard.2008.02.029. [DOI] [PubMed] [Google Scholar]
- 109.Green PS, Vaisar T, Pennathur S, Kulstad JJ, Moore AB, Marcovina S, Brunzell J, Knopp RH, Zhao XQ, Heinecke JW. Combined statin and niacin therapy remodels the high-density lipoprotein proteome. Circulation. 2008;118:1259–1267. doi: 10.1161/CIRCULATIONAHA.108.770669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Rubic T, Trottmann M, Lorenz RL. Stimulation of CD36 and the key effector of reverse cholesterol transport ATP-binding cassette A1 in monocytoid cells by niacin. Biochem Pharmacol. 2004;67:411–419. doi: 10.1016/j.bcp.2003.09.014. [DOI] [PubMed] [Google Scholar]
- 111.Qiu X, Mistry A, Ammirati MJ, Chrunyk BA, Clark RW, Cong Y, Culp JS, Danley DE, Freeman TB, Geoghegan KF, Griffor MC, Hawrylik SJ, Hayward CM, Hensley P, Hoth LR, Karam GA, Lira ME, Lloyd DB, McGrath KM, Stutzman-Engwall KJ, Subashi AK, Subashi TA, Thompson JF, Wang IK, Zhao H, Seddon AP. Crystal structure of cholesteryl ester transfer protein reveals a long tunnel and four bound lipid molecules. Nat Struct Mol Biol. 2007;14:106–113. doi: 10.1038/nsmb1197. [DOI] [PubMed] [Google Scholar]
- 112.Clark RW, Ruggeri RB, Cunningham D, Bamberger MJ. Description of the torcetrapib series of cholesterol ester transfer protein inhibitors, including the mechanism of action. J Lipid Res. 2006;47:537–552. doi: 10.1194/jlr.M500349-JLR200. [DOI] [PubMed] [Google Scholar]
- 113.McKenney JM, Davidson MH, Shear CL, Revkin JH. Efficacy and safety of torcetrapib, a novel cholesteryl ester transfer protein inhibitor, in individuals with below-average high-density lipoprotein cholesterol levels on a background of atorvastatin. J Am Coll Cardiol. 2006;48:1782–1790. doi: 10.1016/j.jacc.2006.06.066. [DOI] [PubMed] [Google Scholar]
- 114.Millar JS, Brousseau ME, Diffenderfer MR, Barrett PH, Welty FK, Faruqi A, Wolfe ML, Nartsupha C, Digenio AG, Mancuso JP, Dolnikowski GG, Schaefer EJ, Rader DJ. Effects of the cholesteryl ester transfer protein inhibitor torcetrapib on apolipoprotein B100 metabolism in humans. Arterioscler Thromb Vasc Biol. 2006;26:1350–1356. doi: 10.1161/01.ATV.0000219695.84644.56. [DOI] [PubMed] [Google Scholar]
- 115.Guérin M, Le Goff W, Duchene E, Julia Z, Nguyen T, Thuren T, Shear CL, Chapman MJ. Inhibition of CETP by torcetrapib attenuates the atherogenicity of postprandial TG-rich lipoproteins in type IIB hyperlipidemia. Arterioscler Thromb Vasc Biol. 2008;28:148–154. doi: 10.1161/ATVBAHA.107.151688. [DOI] [PubMed] [Google Scholar]
- 116.Barter PJ, Caulfield M, Eriksson M, Grundy SM, Kastelein JJ, Komajda M, Lopez-Sendon J, Mosca L, Tardif JC, Waters DD, Shear CL, Revkin JH, Buhr KA, Fisher MR, Tall AR, Brewer B ILLUMINATE Investigators. Effects of torcetrapib in patients at high risk for coronary events. N Engl J Med. 2007;b 357:2109–2122. doi: 10.1056/NEJMoa0706628. [DOI] [PubMed] [Google Scholar]
- 117.Davidson MH, McKenney JM, Shear CL, Revkin JH. Efficacy and safety of torcetrapib, a novel cholesteryl ester transfer protein inhibitor, in individuals with below-average high-density lipoprotein cholesterol levels. J Am Coll Cardiol. 2006;48:1774–1781. doi: 10.1016/j.jacc.2006.06.067. [DOI] [PubMed] [Google Scholar]
- 118.Stroes ES, Kastelein JJ, Benardeau A, Blum D, Clerc RG, Campos L, Niesor EJ. Absence of effect of R1658/JTT-705 on blood pressure and tissue expression of renin-angiotensin system-related genes in rats. J Am Coll Cardiol. 2008;10(Suppl. A):A322. [Google Scholar]
- 119.DePasquale M, Knight D, Loging W, Morehouse L, Winter S, Blasi E, Keiser J. Mechanistic studies of hemodynamics with a series of cholesterol ester transfer protein inhibitors. Circ Res. 2007;101:1209–1210. (Abstract 4021) [Google Scholar]
- 120.Nissen SE, Tardif JC, Nicholls SJ, Revkin JH, Shear CL, Duggan WT, Ruzyllo W, Bachinsky WB, Lasala GP, Tuzcu EM ILLUSTRATE Investigators. Effect of torcetrapib on the progression of coronary atherosclerosis. N Engl J Med. 2007;356:1304–1316. doi: 10.1056/NEJMoa070635. [DOI] [PubMed] [Google Scholar]
- 121.Kastelein JJ, van Leuven SI, Burgess L, Evans GW, Kuivenhoven JA, Barter PJ, Revkin JH, Grobbee DE, Riley WA, Shear CL, Duggan WT, Bots ML RADIANCE 1 Investigators. Effect of torcetrapib on carotid atherosclerosis in familial hypercholesterolemia. N Engl J Med. 2007;356:1620–1630. doi: 10.1056/NEJMoa071359. [DOI] [PubMed] [Google Scholar]
- 122.Bots ML, Visseren FL, Evans GW, Riley WA, Revkin JH, Tegeler CH, Shear CL, Duggan WT, Vicari RM, Grobbee DE, Kastelein JJ RADIANCE 2 Investigators. Torcetrapib and carotid intima-media thickness in mixed dyslipidaemia (RADIANCE 2 study): a randomised, double-blind trial. Lancet. 2007;370:153–160. doi: 10.1016/S0140-6736(07)61088-5. [DOI] [PubMed] [Google Scholar]
- 123.Vergeer M, Bots ML, van Leuven SI, Basart DC, Sijbrands EJ, Evans GW, Grobbee DE, Visseren FL, Stalenhoef AF, Stroes ES, Kastelein JJ. Cholesteryl ester transfer protein inhibitor torcetrapib and off-target toxicity: a pooled analysis of the rating atherosclerotic disease change by imaging with a new CETP inhibitor (RADIANCE) trials. Circulation. 2008;118:2515–2522. doi: 10.1161/CIRCULATIONAHA.108.772665. [DOI] [PubMed] [Google Scholar]
- 124.Nicholls SJ, Tuzcu EM, Brennan DM, Tardif JC, Nissen SE. Cholesteryl ester transfer protein inhibition, high-density lipoprotein raising, and progression of coronary atherosclerosis: insights from ILLUSTRATE (Investigation of Lipid Level Management Using Coronary Ultrasound to Assess Reduction of Atherosclerosis by CETP Inhibition and HDL Elevation) Circulation. 2008;118:2506–2514. doi: 10.1161/CIRCULATIONAHA.108.790733. [DOI] [PubMed] [Google Scholar]
- 125.Clark RW, Sutfin TA, Ruggeri RB, Willauer AT, Sugarman ED, Magnus-Aryitey G, Cosgrove PG, Sand TM, Wester RT, Williams JA, Perlman ME, Bamberger MJ. Raising high-density lipoprotein in humans through inhibition of cholesteryl ester transfer protein: an initial multidose study of torcetrapib. Arterioscler Thromb Vasc Biol. 2004;24:490–497. doi: 10.1161/01.ATV.0000118278.21719.17. [DOI] [PubMed] [Google Scholar]
- 126.Kontush A, Guerin M, Chapman MJ. Spotlight on HDL-raising therapies: insights from the torcetrapib trials. Nat Clin Pract Cardiovasc Med. 2008;5:329–336. doi: 10.1038/ncpcardio1191. [DOI] [PubMed] [Google Scholar]
- 127.Catalano G, Julia Z, Frisdal E, Vedie B, Fournier N, Le Goff W, Chapman MJ, Guerin M. Torcetrapib differentially modulates the biological activities of HDL2 and HDL3 particles in the reverse cholesterol transport pathway. Arterioscler Thromb Vasc Biol. 2009;29:268–275. doi: 10.1161/ATVBAHA.108.179416. [DOI] [PubMed] [Google Scholar]
- 128.Niesor EJ, von der Marck E, Brousse M, Maugeais C. Inhibition of cholesteryl ester transfer protein (CETP): different in vitro characteristics of RO4607381/JTT-705 and torcetrapib (TOR) Atherosclerosis. 2008;199:231. [Google Scholar]
- 129.Okamoto H, Yonemori F, Wakitani K, Minowa T, Maeda K, Shinkai H. A cholesteryl ester transfer protein inhibitor attenuates atherosclerosis in rabbits. Nature. 2000;406:203–207. doi: 10.1038/35018119. [DOI] [PubMed] [Google Scholar]
- 130.Huang Z, Inazu A, Nohara A, Higashikata T, Mabuchi H. Cholesteryl ester transfer protein inhibitor (JTT-705) and the development of atherosclerosis in rabbits with severe hypercholesterolaemia. Clin Sci (Lond) 2002;103:587–594. doi: 10.1042/cs1030587. [DOI] [PubMed] [Google Scholar]
- 131.de Grooth GJ, Kuivenhoven JA, Stalenhoef AF, de Graaf J, Zwinderman AH, Posma JL, van Tol A, Kastelein JJ. Efficacy and safety of a novel cholesteryl ester transfer protein inhibitor, JTT-705, in humans: a randomized phase II dose response study. Circulation. 2002;105:2159–2165. doi: 10.1161/01.cir.0000015857.31889.7b. [DOI] [PubMed] [Google Scholar]
- 132.Kuivenhoven JA, de Grooth GJ, Kawamura H, Klerkx AH, Wilhelm F, Trip MD, Kastelein JJ. Effectiveness of inhibition of cholesteryl ester transfer protein by JTT-705 in combination with pravastatin in type II dyslipidemia. Am J Cardiol. 2005;95:1085–1088. doi: 10.1016/j.amjcard.2004.12.064. [DOI] [PubMed] [Google Scholar]
- 133.Stein EA, Stroes ESG, Steiner G, Buckley BM, Capponi AM, Burgess T, Niesor EJ, Kallend D, Kastelein JJP. Safety and tolerability of dalcetrapib. Am J Cardiol. 2009;104:82–91. doi: 10.1016/j.amjcard.2009.02.061. [DOI] [PubMed] [Google Scholar]
- 134.Hermann F, Enseleit F, Spieker LE, Périat D, Sudano I, Hermann M, Corti R, Noll G, Ruschitzka F, Lüscher TF. Cholesteryl ester transfer protein inhibition and endothelial function in Type II hyperlipidemia. Thromb Res. 2009;123:460–465. doi: 10.1016/j.thromres.2008.06.022. [DOI] [PubMed] [Google Scholar]
- 135.National Institutes of Health. A study of RO4607381 in stable coronary heart disease patients with recent acute coronary syndrome. http://clinicaltrials.gov/ct2/show/NCT00658515?term=RO4607381&rank=2. (3 July 2009) [Google Scholar]
- 136.National Institutes of Health. A study of the effect of RO4607381 on atherosclerotic plaque in patients with coronary heart disease. http://clinicaltrials.gov/ct2/show?term=RO4607381&rank=4. (3 July 2009) [Google Scholar]
- 137.O'Neill E, Sparrow CP, Chen Y, Eveland S, Frantz-Wattley B, Milot D, Sinclair PJ, Ali A, Lu Z, Smith CJ, Taylor G, Thompason CF, Anderson MS, Cumiskey A, Rosa R, Strain J, Peterson LB. Identification and characterization of MK-0859, a novel cholesteryl ester transfer protein inhibitor. J Clin Lipidol. 2007;1:367. [Google Scholar]
- 138.Krishna R, Anderson MS, Bergman AJ, Jin B, Fallon M, Cote J, Rosko K, Chavez-Eng C, Lutz R, Bloomfield DM, Gutierrez M, Doherty J, Bieberdorf F, Chodakewitz J, Gottesdiener KM, Wagner JA. Effect of the cholesteryl ester transfer protein inhibitor, anacetrapib, on lipoproteins in patients with dyslipidaemia and on 24-h ambulatory blood pressure in healthy individuals: two double-blind, randomised placebo-controlled phase I studies. Lancet. 2007;370:1907–1914. doi: 10.1016/S0140-6736(07)61813-3. [DOI] [PubMed] [Google Scholar]
- 139.Bloomfield D, Carlson GL, Sapre A, Tribble D, McKenney JM, Littlejohn TW, 3rd, Sisk CM, Mitchel Y, Pasternak RC. Efficacy and safety of the cholesteryl ester transfer protein inhibitor anacetrapib as monotherapy and coadministered with atorvastatin in dyslipidemic patients. Am Heart J. 2009;157:352–360.e2. doi: 10.1016/j.ahj.2008.09.022. [DOI] [PubMed] [Google Scholar]
- 140.Kathiresan S, Melander O, Guiducci C, Surti A, Burtt NP, Rieder MJ, Cooper GM, Roos C, Voight BF, Havulinna AS, Wahlstrand B, Hedner T, Corella D, Tai ES, Ordovas JM, Berglund G, Vartiainen E, Jousilahti P, Hedblad B, Taskinen MR, Newton-Cheh C, Salomaa V, Peltonen L, Groop L, Altshuler DM, Orho-Melander M. Six new loci associated with blood low-density lipoprotein cholesterol, high-density lipoprotein cholesterol or triglycerides in humans. Nat Genet. 2008;40:189–197. doi: 10.1038/ng.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Ansell BJ, Watson KE, Fogelman AM, Navab M, Fonarow GC. High-density lipoprotein function recent advances. J Am Coll Cardiol. 2005;46:1792–1798. doi: 10.1016/j.jacc.2005.06.080. [DOI] [PubMed] [Google Scholar]
- 142.Staels B, Maes M, Zambon A. Fibrates and future PPARalpha agonists in the treatment of cardiovascular disease. Nat Clin Pract Cardiovasc Med. 2008;5:542–553. doi: 10.1038/ncpcardio1278. [DOI] [PubMed] [Google Scholar]
- 143.Brousseau ME, Diffenderfer MR, Millar JS, Nartsupha C, Asztalos BF, Welty FK, Wolfe ML, Rudling M, Björkhem I, Angelin B, Mancuso JP, Digenio AG, Rader DJ, Schaefer EJ. Effects of cholesteryl ester transfer protein inhibition on high-density lipoprotein subspecies, apolipoprotein A-I metabolism, and fecal sterol excretion. Arterioscler Thromb Vasc Biol. 2005;25:1057–1064. doi: 10.1161/01.ATV.0000161928.16334.dd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Yvan-Charvet L, Matsuura F, Wang N, Bamberger MJ, Nguyen T, Rinninger F, Jiang XC, Shear CL, Tall AR. Inhibition of cholesteryl ester transfer protein by torcetrapib modestly increases macrophage cholesterol efflux to HDL. Arterioscler Thromb Vasc Biol. 2007;27:1132–1138. doi: 10.1161/ATVBAHA.106.138347. [DOI] [PubMed] [Google Scholar]
- 145.Semenkovich CF. Insulin resistance and atherosclerosis. J Clin Invest. 2006;116:1813–1822. doi: 10.1172/JCI29024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Sacks FM Expert Group on HDL Cholesterol. The role of high-density lipoprotein (HDL) cholesterol in the prevention and treatment of coronary heart disease: expert group recommendations. Am J Cardiol. 2002;90:139–143. doi: 10.1016/s0002-9149(02)02436-0. [DOI] [PubMed] [Google Scholar]
- 147.Genest JJ, McNamara JR, Salem DN, Schaefer EJ. Prevalence of risk factors in men with premature coronary artery disease. Am J Cardiol. 1991;67:1185–1189. doi: 10.1016/0002-9149(91)90924-a. [DOI] [PubMed] [Google Scholar]
- 148.Kannel WB. High-density lipoproteins: epidemiologic profile and risks of coronary artery disease. Am J Cardiol. 1983;52:9B–12B. doi: 10.1016/0002-9149(83)90649-5. [DOI] [PubMed] [Google Scholar]
- 149.Nobécourt E, Jacqueminet S, Hansel B, Chantepie S, Grimaldi A, Chapman MJ, Kontush A. Defective antioxidative activity of small dense HDL3 particles in type 2 diabetes: relationship to elevated oxidative stress and hyperglycaemia. Diabetologia. 2005;48:529–538. doi: 10.1007/s00125-004-1655-5. [DOI] [PubMed] [Google Scholar]
- 150.Hansel B, Giral P, Nobecourt E, Chantepie S, Bruckert E, Chapman MJ, Kontush A. Metabolic syndrome is associated with elevated oxidative stress and dysfunctional dense high-density lipoprotein particles displaying impaired antioxidative activity. J Clin Endocrinol Metab. 2004;89:4963–4971. doi: 10.1210/jc.2004-0305. [DOI] [PubMed] [Google Scholar]