Cytokinesis is the last step in the cell cycle, where daughter cells finally separate. Cytokinesis is precisely regulated in both time and space to ensure that each daughter cell receives an equal share of DNA and other cellular materials. Successful completion of cytokinesis, with the cleavage furrow exactly bisecting the axis of chromosome segregation, involves the coordinated action of the cytoskeleton, cell cycle and membrane machineries of cells (1). Chemical biology approaches have been used very successfully to study the mechanism of cytokinesis. In this review, we will discuss the use of small molecule probes to perturb cytokinesis as well as on role that naturally occurring small molecule metabolites such as lipids play during cytokinesis.
The mechanism of cytokinesis
Cytokinesis occurs immediately after mitosis, the division of DNA. The mechanism of cytokinesis has been reviewed in detail elsewhere (1–3). Here, we will briefly introduce key steps to provide context for this review. During mitosis, chromosomes line up in a metaphase plate at the center of the mitotic spindle. As the chromosomes are dividing, a dramatic and poorly understood rearrangement of microtubules takes place and a contractile ring begins to assemble (Figure 1). Contact of the cellular cortex (and plasma membrane?) with microtubule structures is essential for the correct positioning of the contractile ring. Several possible mechanisms have been proposed, although recent studies suggest that multiple mechanisms may occur simultaneously (2, 4, 5). There are several main signaling pathways, for example those centered around Aurora B and Polo kinases and the small GTPase Rho, that operate and are essential throughout cytokinesis (1). As the location of the cleavage furrow is being specified, the contractile ring begins to assemble. The temporal sequence of protein recruitment to the ring is a significant, but poorly understood mechanistic feature of early cytokinesis. The complete composition of the ring is not known, but the plasma membrane, actin, myosin II and Anillin are clearly important (Figure 1) (6–8). It is not known how the contractile ring is linked to the plasma membrane during ring assembly or ingression. The contractile ring then ingresses, with actin and myosin providing much of the mechanical force (1). We have partial information for the stages of cytokinesis described so far, but the last step, called completion or abscission, remains the least understood. At the end of ingression, a small structure, the midbody, forms. Severing between the daughter cells, which has to be very carefully controlled to avoid the generation of a hole in the cell, occurs in this structure (9). Targeted vesicle transport is thought to play a role both during contractile ring ingression and in generating a membrane barrier that seals the daughter cells during severing (10–15).
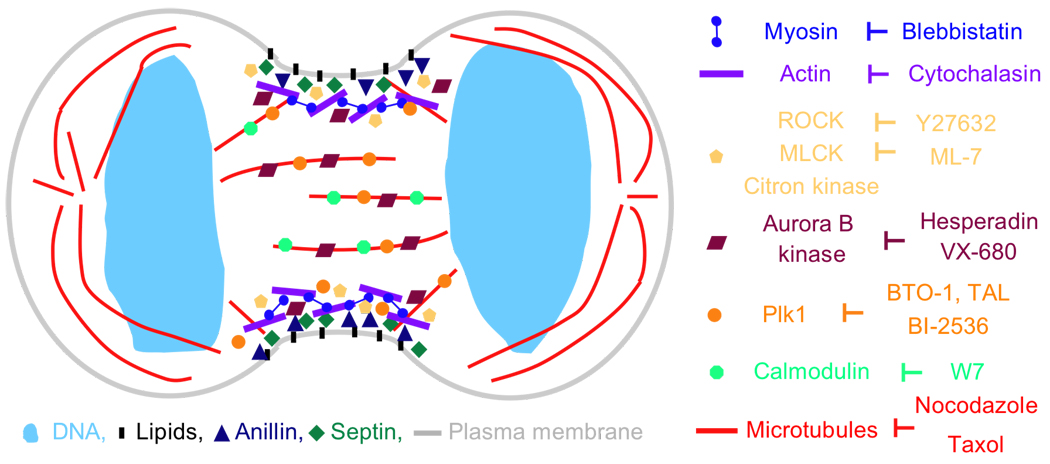
Figure 1. Cartoon of a cell during early stages of cytokinesis.
During cytokinesis, a contractile ring assembles. It is located just beneath the plasma membrane and includes a network of actin and myosin II filaments as well as septins and Anillin. Phosphorylation and activation of myosin II at the cleavage furrow is regulated by several kinases, including ROCK, MLCK and Citron kinase. The mitotic kinases Plk1 and Aurora B both localize to the cleavage furrow as well as midzone microtubules where they function as key regulators of cytokinesis. An expanding collection of small molecules has been used to dissect cytokinesis regulation in both time and space. Key cytokinesis protein and their corresponding small molecule inhibitors are shown. For clarity, only localizations at the cleavage furrow and midzone microtubules are shown. Not drawn to scale.
One of the interesting features of cytokinesis is that there can be considerable mechanistic variation between cell types (1). Often multiple pathways operate in parallel and the importance of individual pathways varies across different systems. This mechanistic plasticity may have evolved to allow cytokinesis in cells with very different characteristics, ranging from very large embryos with a large cell surface area to smaller cells. Some aspects of cytokinesis have been extensively studied in the popular model organisms budding (S. cerevisiae) and especially fission yeast (S. pombe) (16, 17). Insights into the timing of contractile ring assembly during cytokinesis have come from studies in S. pombe (18). Since fungi have many organism-specific features, however, we will focus mostly on cytokinesis in higher eukaryotes.
SMALL MOLECULE PROBES OF CYTOKINESIS
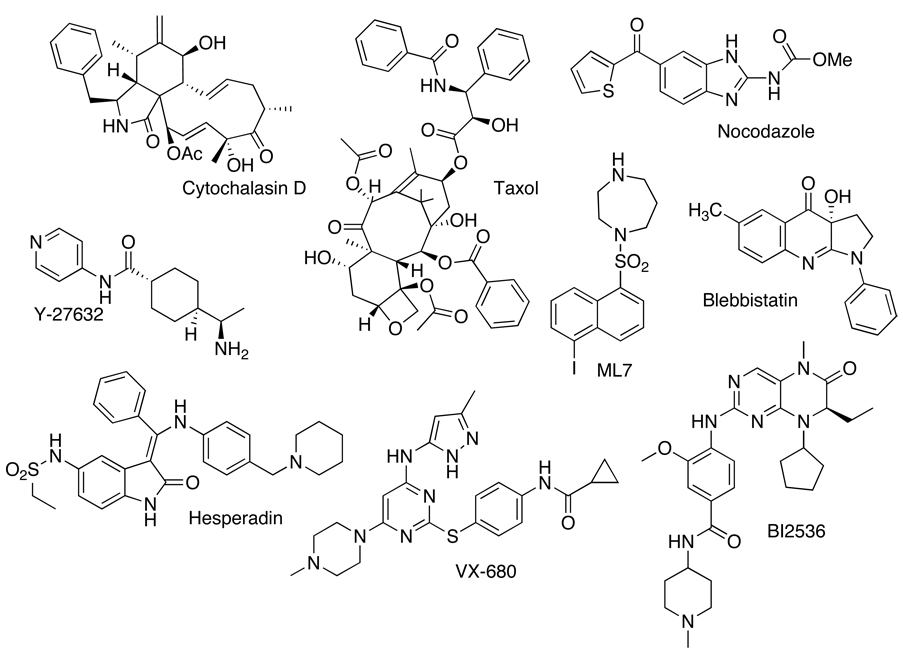
Small molecules are versatile tools to probe dynamic cellular processes like cytokinesis because they enable spatial and temporal control. It has been challenging to study cytokinesis by traditional methods because it is a rapid and dynamic process that occupies only a small portion of the cell cycle. Many key cytokinesis proteins also perform important functions earlier in the cell cycle, especially during mitosis, and their removal leads to an arrest prior to cytokinesis. Small molecules act rapidly and can be added at specific points in the cell cycle. In addition, the effects of small molecules are often reversible upon washout, allowing even greater control because treatments can be combined sequentially. Although the collection of small molecules that are available to study cytokinesis is being expanded (19), it is still small at present (Figure 2), but has been used to great effect.
Figure 2. Chemical structures of small molecules that have been used to probe the mechanism of cytokinesis.
The role of the cytoskeleton in cytokinesis
Precise positioning of the cleavage furrow is specified by the mitotic apparatus and allows ingression to occur along a plane that bisects segregating chromosomes (20). Thus, microtubules as well as the actin-based cytoskeleton are essential for cytokinesis and small molecules have been used to probe their involvement. In particular, small molecules targeting actin have a distinguished history in cytokinesis research (21). For example, natural products in the cytochalasin family cause the depolymerization of actin filaments (22). Since the cytochalasins inhibited multiple processes such as cell migration, ruffling and division (23), they were used to show that the same protein, actin, was involved in these processes. Cytochalasin inhibited cellular division, but not nuclear division, providing evidence, for the first time, that the two processes could be decoupled from each other (23).
Small molecules’ ability to rapidly disrupt cytoskeletal organization was also used to establish the importance of microtubules in cleavage furrow positioning and ingression. Studies using the microtubule depolymerizer nocodazole to disrupt microtubule organization have demonstrated the importance of midzone microtubules in defining the cleavage plane (24). Disruption of microtubule dynamics during anaphase with the microtubule stabilizing agent taxol led to a reduction in the rate of furrowing (25). Work by Shannon et al. showed that taxol-stabilized microtubules were sufficient to initiate furrow formation, however the taxol-mediated disruption of microtubule dynamics delayed ingression and prevented cytokinesis completion (26). Using a combination of small molecules including taxol and the historically important hexylene glycol and urethane (20), Strickland et al. differentially perturbed the dynamic state of microtubules. They determined that successful furrow initiation was positively regulated by the physical contact between the astral microtubule plus end and the cell cortex, rather than the dynamic state of the microtubules (27).
Using small molecules to study the regulation of cytokinesis
In addition to helping elucidate the critical role of actin and microtubules, small molecules have provided insight into the function of key regulators of cytokinesis, especially by taking advantage of their high temporal control and often reversibility. For example, washout studies following blebbistatin (see below), nocodazole, or cytochalasin treatment have been used to identify the approximately one hour timing window in which cells remain competent to carry out cytokinesis, termed C-phase (28, 29).
The phosphorylation and activation of myosin II regulatory light chain (MRLC) by several kinases including Rho kinase (ROCK), myosin light chain kinase (MLCK – see the section on calcium for a discussion on the activation of MLCK) and Citron kinase is important for proper ingression of the cleavage furrow (Figure 3). Small molecule inhibitors of ROCK and MLCK have been used to dissect their respective functions and possible functional redundancies during cytokinesis (30–32). HeLa cells treated with the ROCK inhibitor Y-27632 exhibited a reduced MRLC phosphorylation, enhanced accumulation of ROCK at the cleavage furrow and a significantly prolonged ingression time (31). In contrast, acute inhibition of MLCK with ML7 redistributed myosin and actin away from the equatorial cortex and caused subsequent furrow regression (32). Lucero et al. used ML7 and the ROCK inhibitor H-1152 to tease apart the temporal requirements for ROCK and MLCK activities during cytokinesis in sea urchin eggs (33). While both MLCK and ROCK were required for furrow ingression, only MLCK was necessary for the global activation of myosin II at the metaphase-anaphase transition. Thus, treatment with ROCK and MLCK inhibitors leads to different cytokinesis defects, suggesting they have different functions and cannot compensate for each other. Adding even more complexity is Citron kinase. RNAi and overexpression studies suggest that Citron kinase induces the di-phosphorylation of MRLC and is important during the later stages of cytokinesis (34, 35). Currently there are no small molecule inhibitors of Citron kinase. Future studies using small molecules will continue to provide information about how these kinases differentially regulate myosin phosphorylation at the cleavage furrow.
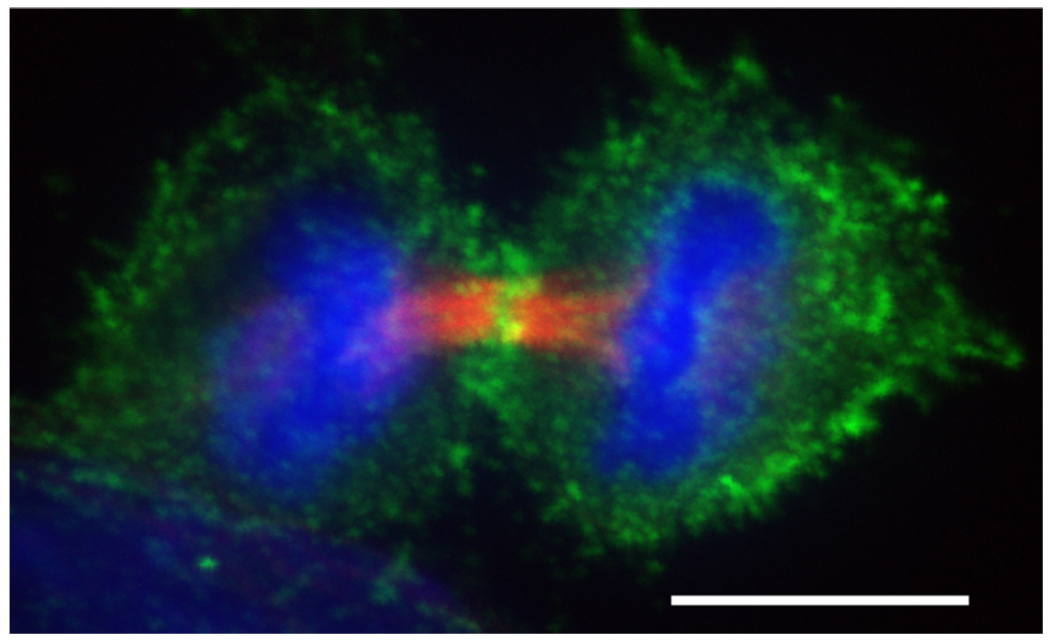
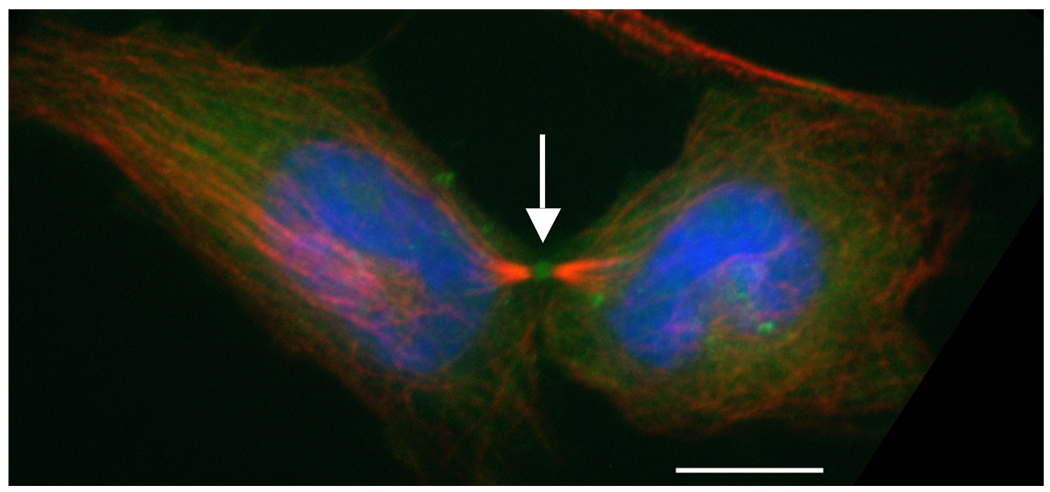
Figure 3. Dividing HeLa cell during early cytokinesis.
Phosphorylated myosin II accumulates at the cleavage furrow.  (red) were stained with anti-tubulin (Sigma T6199),
(red) were stained with anti-tubulin (Sigma T6199),  (green) was stained with Phospho-Myosin Light Chain 2 (Ser19) antibody (Cell Signalling, 3671L),
(green) was stained with Phospho-Myosin Light Chain 2 (Ser19) antibody (Cell Signalling, 3671L),  (blue) with DAPI. Scalebar represents 10 µM.
(blue) with DAPI. Scalebar represents 10 µM.
Polo-like kinase (Plk1) regulates several aspects of cell division including mitotic entry, bipolar spindle formation, chromosome segregation and cytokinesis (36). Perturbing Plk1 function in animal cells leads to a failure in bipolar spindle assembly and arrest in early mitosis. The lack of mitotic progression upon Plk1 disruption greatly complicated studies on the role of Plk1 during late mitosis and cytokinesis. Only recently was the role of Plk1 in cytokinesis dissected in more detail through the use of chemical genetics and small molecule inhibitors, where Plk1 could be rapidly inactivated in synchronized cells (37–40). Inhibition of Plk1 activity during anaphase disrupted Plk1-mediated phosphorylation of HsCyk-4 RhoGAP and subsequent recruitment of Ect2 RhoGEF to the central spindle, events necessary to initiate contractile ring assembly and to activate Rho GTPase, one of the master regulators of cytokinesis (41, 42). Future experiments to characterize additional Plk1 targets will be important to further elucidate the role of Plk1 during cytokinesis.
Like Plk1, Aurora B kinase regulates a variety of critical events during mitosis, including chromosome alignment, bipolar and central spindle assembly and cytokinesis (43). Hesperadin, a small molecule inhibitor of Aurora B, has been used to elucidate the role of Aurora B during cytokinesis (44, 45). Miyauchi et al. treated HeLa cells with Hesperadin and observed mis-localization of a number of key cytokinesis proteins, as well as disorganized astral and midzone microtubules. Hesperadin treatment during anaphase/telophase was used to establish a role for Aurora B in the phosphorylation and activation of MKLP1, a kinesin motor protein needed during cytokinesis (46). Plk1 can also phosphorylate MKLP1, so it will be interesting to determine if and how coordinated phosphorylation of MKLP1 by both kinases is necessary for central spindle function and proper cytokinesis.
The Rho GTPase pathway is another key regulator of cytokinesis, but no specific inhibitors of the Rho pathway exist apart from the ROCK inhibitors mentioned above. We have developed small molecules that target the Rho pathway in cytokinesis, using a chemical variant of a classical modifier screen (A. Castoreno and U. Eggert, unpublished data). Our compounds inhibit cellular functions of the Rho pathway and likely target different proteins within the Rho pathway.
As we discuss below, membranes, and also membrane trafficking, are important in cytokinesis, but poorly understood (10–15). Unfortunately, there are fewer small molecules available that target membrane trafficking than there are inhibitors of the cytoskeleton or signaling cascades. Brefeldin A, an inhibitor of post-Golgi trafficking, inhibits cytokinesis in some (but not all) cell types, and has been used to show that membrane accumulation at the abscission point is important for completion (13, 15, 47).
Using sequential small molecule treatments to study cytokinesis
A major advantage of small molecules is that they can be added sequentially or combined to dissect different steps within a process. Using small molecules to synchronize cells at different stages of the cell cycle is an important application in many different areas of research. Several of the studies described in the previous section used a variety of small molecules to synchronize cells in mitosis, both to increase the pool of cells about to enter cytokinesis and to focus specifically on the role of proteins such as Aurora B and Plk-1 in cytokinesis rather than mitosis. In addition to dissecting the role of these kinases, combined small molecule treatments have also proven useful in to studying contractile ring assembly.
Blebbistatin, a reversible inhibitor of myosin II ATPase activity, has enabled a detailed study of contractile ring assembly and dynamics. In the presence of blebbistatin, components of the cleavage furrow including microtubules, myosin II and Anillin (a contractile ring scaffolding protein) organize properly, but contractile ring ingression is blocked (29). Straight et al. combined blebbistatin treatment with additional small molecules to probe the signaling pathways necessary to maintain components at the cleavage furrow following assembly. The addition of cytochalasin or nocodazole to blebbistatin-treated cells caused the delocalization of myosin and Anillin, while small molecule inhibition of ROCK (using Y27632) or Aurora B kinase functions perturbed myosin but not Anillin localization, indicating alternate pathways for the maintenance of myosin and Anillin at the cleavage furrow (29).
Combined small molecule treatments have also been used to study of how cells with monopolar spindles are able to form furrows, which can be used as a lower-complexity model to dissect contractile ring assembly and ingression. Work by Canman et al. showed that cytokinesis could occur in cells without a bipolar spindle (48). To extend these studies, Hu et al. developed a drug synchronization method with two small molecules to trigger monopolar cytokinesis in HeLa cells. After arresting cells in mitosis using the kinesin-5 inhibitor S-trityl-L-cysteine and then forcing cells into cytokinesis using the Cdk1 inhibitor purvanalol A, they observed a strong polarization of microtubules and a large cellular protrusion. Subsequently, a furrow formed between the protrusion and the cell body (49). To probe the requirements for polarization in monopolar cells, a third inhibitor was introduced. The addition of nocodazole, latrunculin B, blebbistatin, or the Aurora B inhibitor VX680 disrupted polarization and furrowing in monopolar cells, demonstrating a requirement for microtubules, actin, myosin II and Aurora B during monopolar cytokinesis.
Using small molecules to study spatial regulation in cytokinesis
The ability to locally perfuse small molecules to spatially distinct areas of a dividing cell has provided a deeper understanding into how equatorial and polar regions regulate furrow ingression.
Local application of cytochalasin D to the polar region, not the equatorial region, of dividing epithelial cells blocked cytokinesis, indicating a global coordination of contractive and cortical activities for proper ingression (50). Following up on these results, Guha et al. applied blebbistatin to the equator close to the cell cortex and observed inhibited ingression in most cells, along with a strong accumulation of equatorial actin filaments. In contrast, application of blebbistatin to the polar region caused minimal cytokinesis failure. Many cells, however, displayed problems with furrow ingression and positioning, suggesting a coordinating role for myosin activity in both regions.
In complementary studies, Murthy et al. performed FRAP experiments in GFP-actin expressing cells and observed decreased actin turnover in the assembled contractile ring the presence of blebbistatin. These results provide evidence for a second role for myosin in regulating actin dynamics at the cleavage furrow (32, 51).
SMALL MOLECULE METABOLITES AND CYTOKINESIS
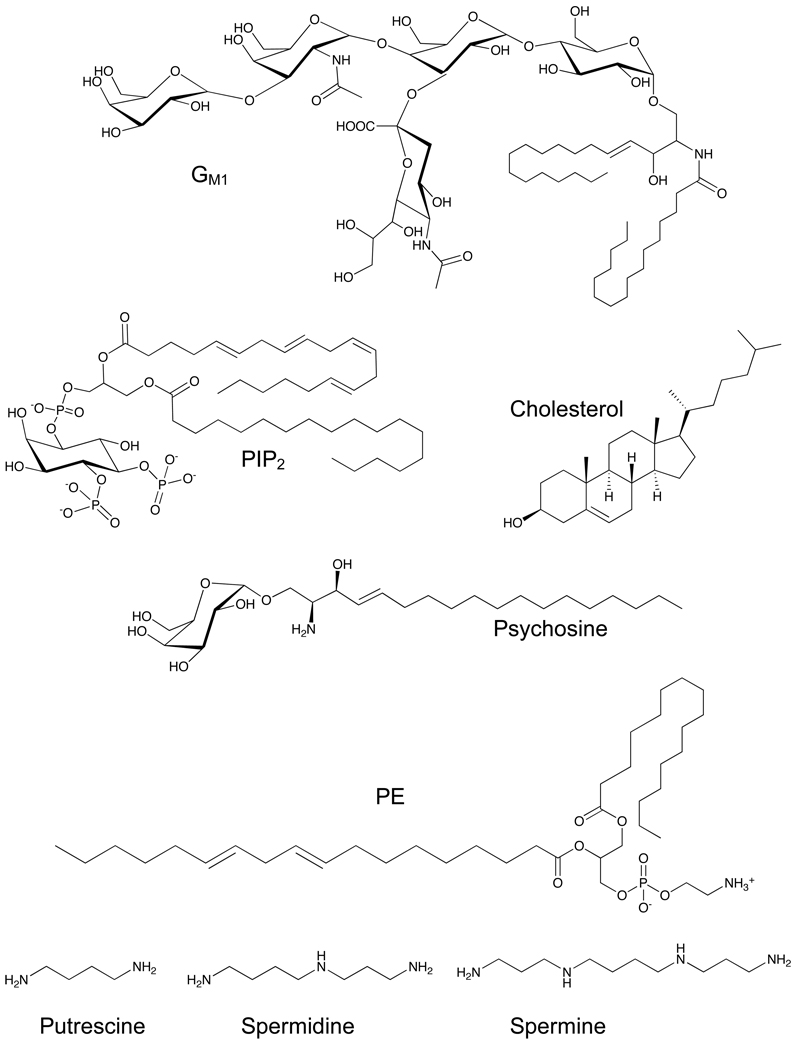
Given the complexity of cytokinesis, it is not surprising that naturally occurring small molecule metabolites are important participants in this process. Much less is known about the functions of small molecule metabolites than about key proteins, in part because it can be difficult to detect many metabolites in their cellular context. Here, we will focus mostly on the roles of lipids and of calcium in cytokinesis (Figure 4), mainly because the role of other metabolites has not yet been explored.
Figure 4. Chemical structures of lipid and polyamine metabolites that are thought to participate in cytokinesis (www.lipidlibrary.co.uk).
The role of lipids in cytokinesis
Surprisingly, given the important link between the membrane and the actin cortex, little is known about the role of the plasma membrane and lipids in cytokinesis. It is known that some lipids localize to the cleavage site and that membrane transport is required to relieve the cortical tension that occurs during separation. However, the precise roles of these lipids are still unclear, leaving a number of important outstanding questions: Are any lipids actively involved in cytokinesis and is their role structural or do they have a signaling function? Some early clues suggest that both of these scenarios are true (52, 53). How do membranes influence cell shape changes that occur during cytokinesis? How are the actin cortex and more specifically the contractile ring linked to the plasma membrane? How are different lipids transported to the cleavage furrow? How are they maintained there? What is the role of membranes in the midbody and how do they seal the hole in the cell wall that inevitably occurs during cleavage? We will discuss first steps that have been made to address some of these questions.
It has been reported that cholesterol (54–56), phosphatidyl ethanolamine (PE) (57, 58), phosphatidylinositol 4,5-bisphosphate (PIP2) (59, 60), and ganglioside GM1 (54) localize to the cleavage furrow, suggesting an involvement in cytokinesis. In addition, the sphingolipid psychosine has also been found to participate in cytokinesis (61). It has been possible to link these lipids to cytokinesis because they can be visualized with fluorescent probes. The development of new tools to detect lipids both in live and fixed cells would be extremely useful and would contribute significantly to unraveling which lipids are involved in cytokinesis. It is very likely that there are other undiscovered lipids that play significant roles in the process since recent RNAi screens targeting cytokinesis identified lipid modifying enzymes (19, 62, 63).
Phosphatidylinositols
Phosphatidylinositols (PtdIns) are important signaling molecules that regulate the actin cytoskeleton, for example during cell polarization and division (53, 64). PIP2 is the best-studied lipid in cytokinesis. PIP2 was first shown to be involved in cytokinesis when injection of X. laevis embryos with PIP2 antibodies inhibited furrow ingression (65). Similarly, the treatment of crane fly spermatocytes with inhibitors of PtdIns recycling, particularly PI3 kinase and Phospholipase C (PLC), slowed down or inhibited furrow ingression, demonstrating that PtdIns metabolism is required for cleavage (66).
Two recent studies focused on the role of PIP2 in mammalian cells (59, 60). Both studies showed that PIP2 localizes to the cleavage furrow during ingression (Figure 5) and, more importantly, that interference with PIP2 results in cytokinetic defects. Field et al. investigated the effects of PLC-PH overexpression in mammalian cells. The PH domain of PLC, an enzyme that hydrolyses PIP2, binds to PIP2. GFP-PLC-PH can be used to study the localization of PIP2 and to sequester its function when overexpressed. Ten-fold overexpression of GFP-PLC-PH resulted in separation of actin from the plasma membrane, indicating that PIP2 connects actin to the plasma membrane at the cleavage furrow. Furthermore, a decrease in PIP2 levels by overexpression of PLC-PH or a kinase-inactive form of PI(4)P-5-kinase, the enzyme that synthesizes PIP2, induces the formation of binucleated cells. Field et al. suggest a model in which PIP2 provides the connection between the contractile ring and the plasma membrane by binding to plasma membranes and/or different components of the contractile ring.
Figure 5. Dividing HeLa cell during the final stage of cytokinesis.
PIP2 localizes to the midbody (white arrow). HeLa cells were transfected with  (vector kindly provided by Tamas Balla, NIH).
(vector kindly provided by Tamas Balla, NIH).  (red) were stained with anti-tubulin (Sigma T6199),
(red) were stained with anti-tubulin (Sigma T6199),  (blue) with DAPI. Scalebar represents 10 µM.
(blue) with DAPI. Scalebar represents 10 µM.
Giotto, a class I PITP (PtdIns transfer protein), is enriched at the cleavage furrow and the Endoplasmic Reticulum. It mediates the transfer of PtdIns monomers to the equatorial membrane in Drosophila, leading to a local increase in the levels of PtdIns. Giotto mutants fail cytokinesis and show abnormal localization of vesicle-like structures. The contractile ring forms but can not constrict. The cytokinetic arrest phenotypes in this mutant are similar to that of the fwd (PI4 kinase) and fws (four way stop, a Cog5 homolog in Drosophila) mutants. Based on these results, the authors suggested that changes in membrane composition might contribute to vesicle formation, which results in targeted delivery of proteins and/or membrane addition (67).
PIP2 binds to septins and ERM (Ezrin/Radixin/Moesin) family proteins in vitro (68). These proteins are thought to be mainly scaffolding proteins that may be involved in linking the actin cortex to the plasma membrane. It was shown recently that PIP2 levels are directly correlated to the amount of membrane-bound ERM proteins in cells, possibly through regulation of PLC (69). An intriguing study in S. cerevisiae has shown that PIP2 binding is a mechanism by which Rho GTPase can localize to the bud neck, further supporting the notion that PIP2 is an important signaling molecule in cytokinesis (70).
Cholesterol and phosphatidyl ethanolamine
It has been possible to hypothesize that cholesterol is involved in cytokinesis because it can be visualized in cells with the fluorescent macrolide filipin (71). Filipin binds to sterols and sequesters their activity. Using filipin, it was shown that cholesterol is involved in cytokinesis in S. pombe (56). In a similar study in sea urchin eggs, the Burgess group has shown that the formation of an equatorial band enriched with GM1 and cholesterol is coupled to contractile ring formation and assembly. The same study also demonstrated that filipin addition during cleavage furrow formation results in the retraction of the furrow (54). Methyl-β-cyclodextrin has also been used to investigate the effects of cholesterol depletion in various cellular events. With a hydrophilic external and a hydrophobic internal face, cyclodextrins are able to extract cholesterol from the plasma membrane. Cholesterol depletion in zebrafish blastomeres with methyl-β-cyclodextrin inhibits endocytosis and prevents completion of cytokinesis, eventually resulting multinucleated cells at high concentrations (72). Another study in human leukemia cells, focusing on the role of cholesterol in mitosis, revealed that cholesterol starvation and inhibition of cholesterol synthesis induced cytokinesis failure. Reintroduction of cholesterol to the growth medium abolished this effect, reinforcing the idea that the abnormalities were due to cholesterol deficiency (55). These data suggest an involvement of cholesterol in cytokinesis. Its precise role, be it structural or signaling, however, is not known.
PE is an abundant lipid in most cells. It has also been reported to participate in cytokinesis, although its function is not clear. The cell surface localization of PE, which is normally localized at the inner leaflet of the plasma membrane, appears to be important for the completion of cytokinesis. Reduced PE can lead to cytokinetic arrest, which can be reversed by the addition of PE or ethanolamine, a precursor of PE (57). A study investigating the localization of PE using a peptide that recognizes PE showed that its surface exposure at the cleavage furrow is specific to the late telophase of cytokinesis. Cell surface immobilization of PE with a streptavidin analog of the peptide blocked cytokinesis (58).
Very-long-chain fatty acids
Very-long-chain fatty acids (VLCFA), with aliphatic tails consisting of 22 carbons or longer, are thought to participate in the stabilization of highly curved membrane domains (73). They are mostly found in sphingolipids (see next section) and are essential for the formation and the function of these lipids. Elovl enzymes are involved in the biosynthesis of VLCFAs. A loss of function mutation in bond, an Elovl protein in Drosophila, causes cytokinetic defects in spermatocytes (74). Szafer-Glusman and colleagues investigated this phenotype and showed that VLCFAs are crucial in early cytokinesis. Although contractile ring assembly was normal, ring constriction was impaired, leading to halted furrow ingression, regression, and finally failed cytokinesis. In addition, the authors observed collapse and detachment of the contractile ring from the membrane during late cytokinesis. These mutants were unable to form an organized midzone which is directly related to furrow regression (75). These results suggest that defects in the elongation of the VLCFAs affect the contraction of the ring as well as the stabilization of the midzone microtubules by the ring during cytokinesis.
Sphingolipids
Sphingolipids, ubiquitous components of the cell membrane, play a variety of roles in cellular environments. A few studies, mostly using small molecule probes, have found tentative links between sphingolipids and cytokinesis. Serine palmitoyl transferase (SPT) catalyzes the first reaction during sphingolipid biosynthesis. Myriocin, an inhibitor of SPT, causes cytokinetic defects in some cell types, which can be rescued by the addition of sphingosine (76, 77).
Psychosine, a metabolite of the sphingolipid pathway, induces multinucleate cell formation in many cell types (61, 78). This was an exciting finding because it showed, for the first time, that changes in membrane composition could somehow cause cell division failure. Other sphingolipid analogs glucopsychosine, sphingosylphosphorylcholine and lysosulfatide also induced the formation of multinuclear globoid-like cells (61). Psychosine accumulates and multinuclear globular giant cells are observed in the brains of patients with Globoid Cell Leukodystrophy, a neurological disorder (79).
Psychosine’s mechanism of cytokinesis inhibiton is not clear. TDAG8 (T cell death associated gene 8), a G protein-coupled receptor (GPCR), is a target of psychosine. Expression of TDAG8 in some cell types in the presence of psychosine induced the formation of multinuclear cells. Overexpression of TDAG8 or psychosine treatment of cells that did not express TDAG8 did not induce the formation of multinuclear cells (78). Subsequent papers, however, suggested that it is likely that psychosine acts through additional targets (80). The inhibitory binding constant of psychosine for TDAG8 at physiological conditions is 3 µM, which is signficantly higher than the binding constants of other lysolipids to their GPCR receptors. Also, TDAG8 is now thought to be proton-sensing GPCR. Psychosine shifts TDAG8’s (and other proton–sensing GPCRs from the same family) proton response towards a more basic pH, suggesting that protons and psychosine may bind the GPCRs simultaneously (81). Once psychosine’s cell membrane receptor(s) have been confirmed, the next challenge is to understand how it signals to the cell division machinery in the cell’s interior.
GM1 is another sphingolipid that has been connected to cytokinesis (54). Together with their findings on the localization of sterols, Ng, Chang and Burgess studied the localization of GM1 by using a fluorophore conjugate of cholera toxin subunit B. In sea urchin eggs, they found a 7-fold increase in GM1 levels in furrowing cells compared to metaphase cells as well as a localization to the equatorial band during contractile ring formation (54). These data suggest a possible involvement, but no functional analyses are available at this point.
Are polyamines involved in regulating cytokinesis?
Polyamines are another class of naturally occurring small molecules that appear to be involved in cytokinesis, but their functional connections are not entirely clear. Small molecule inhibition of polyamine biosynthesis, particularly spermine, spermidine and putrescine (Figure 4) synthesis, causes cytokinesis defects that can be rescued when the polyamine is added back (82). Biophysical experiments showed that polyamines can affect actin polymerization in vitro and actin staining patterns in cells are disrupted when polyamine biosynthesis is inhibited (82–84). Thus, it is possible that the role of polyamines on cell division is through their specific interaction with actin filaments.
The role of calcium signaling in cytokinesis
It has long been thought that calcium signaling plays important roles in cytokinesis, although the precise molecular functions are still only partially understood. Several studies conducted in embryos of different species have detected transient calcium signals at different stages of cytokinesis. Embryos have been used as a model system because they undergo several division cycles and are larger than cultured cells, allowing better differentiation of events that are tightly regulated in time and space.
For example, in zebrafish embryos, Ca2+ elevation along the equator of the cleavage furrow precedes the initiation of furrow contraction, followed by additional Ca2+ elevations at different time points during subsequent cleavages (85). Transient Ca2+ waves have also been observed in Xenopus embryos, although their timing and positioning differs from zebrafish embryos (86, 87). While the details are unclear, it is likely that there are functional relationships between calcium waves and cytokinesis because calcium sequestration by Ca2+ chelators such as BAPTA (1,2-bis(aminophenoxy)ethane-N,N,N',N'-tetraacetic acid) (85, 87, 88) caused cytokinesis failure and because extra furrows were formed when calcium release was stimulated (89). Variations in the characteristics of the Ca2+ signals and their presence during diverse events of cytokinesis might not only be due to differences in the studied systems. Using different imaging techniques and most importantly different Ca2+-sensitive indicators might result in opposing or partial complete results (90, 91).
If calcium signaling is important in cytokinesis, the origin of the calcium needs to be examined. There is evidence that Ca2+ is released from internal stores through binding of IP3 (inositol 1,4,5 triphosphate) to the IP3 receptor. Manipulations of the IP3 receptor or of IP3 levels have an effect on cytokinesis (85, 86, 88, 92, 93) and endoplasmic reticulum proteins together with the IP3 receptor co-localize to both sides of the ingressing cleavage furrow in zebrafish embryos (94). IP3 is the hydrolysis product of the signaling lipid PIP2, which is also important for cytokinesis. The IP3 receptor can be phosphorylated by the key cytokinesis regulator Plk1, leading to an increased sensitivity of the receptor towards IP3 and therefore enhanced Ca2+ release (95). Ca2+ release from IP3 mediated stores might also be regulated through the actin binding protein Cofilin, which plays a role in cytokinesis (96, 97). During maturation and fertilization of Starfish oocytes Cofilin modulates Ca2+ release through IP3 and other Ca2+-releasing messengers (97).
It is not clear which proteins are involved in calcium regulation during cytokinesis. Some of the functions of calcium seem to be mediated by Calmodulin. Calmodulin is a receptor for intracellular calcium and activates a diverse array of target proteins in a calcium-dependent manner (98). Calmodulin localizes to interzonal microtubules and the intercellular bridge during cytokinesis in HeLa cells (99). Loss of Calmodulin or application of calmodulin inhibitors disrupted or prevented cytokinesis (100, 101). Calmodulin activates MLCK, one of the proteins that activate myosin regulatory light chain (see discussion above for a dissection of MLCK signaling using small molecules) (102). To study upstream modulation of MLCK activity during cytokinesis, Wong et al. inhibited PIP2 hydrolysis to the second messengers DAG (diacylglycerol) and IP3, resulting in a redistribution of actin away from the cleavage furrow and disrupted ingression (88, 102). When the authors used ML7 and ML9 to inhibit MLCK activity, they observed a similar cytokinesis defect compared to inhibition of PIP2 hydrolysis and proposed a model whereby PIP2 hydrolysis regulates myosin activation and furrow ingression via calcium signaling and MLCK activity. Proteins such as dyneins or other with microtubule-associated proteins (101, 103) have also been suggested as possible Calmodulin effectors. However, more work is needed to understand their precise nature or functions.
Several other possible downstream functions of Ca2+ signaling in cytokinesis have been described. Calcium-dependent vesicle transport and membrane remodeling are important in some systems (15, 92). Interfering with the calcium concentration inhibited accumulation of new membranes during cytokinesis, using the extracellular matrix protein hyalin as a membrane marker in sea urchin eggs (15). VAMP-2 vesicle fusion at the ingressing furrow in zebrafish embryos depends on Ca2+ released from IP3 receptor mediated stores (92). The Ca2+-dependent Calpain family might play a role in cytokinesis (92, 104, 105) as well as Annexin XI, a Ca2+-dependent phospholipid-binding protein. Annexin XI is recruited to the midbody in late telophase in some cell types and seems to be necessary for the formation of the midbody and the completion of cytokinesis, possibly by facilitating the interaction between the plasma membrane and the microtubules of the midbody (106).
A role for calcium signaling during cytokinesis in different systems seems to be apparent, but the molecular details, the spatio-temporal correlation and functions are less clear. New imaging methods and more precise and sensitive Ca2+ reporters will help to shed light in the role of calcium in cytokinesis.
Can cytokinesis be a target for cancer chemotherapy?
This review discusses how chemical biology approaches have been very useful in understanding different aspects of the mechanism of cytokinesis. In the first part, we focused on small molecules as biological probes. In addition to their use as probe compounds, however, small molecules also have therapeutic potential. One would expect small molecules that target cytokinesis to be important in the development of cancer therapeutics because improperly regulated cell division can be a cause or a consequence of cancer. Mitosis failure as a cause of cancer progression has been well documented, and a recent report also linked cytokinesis failure to carcinogenesis (107).
Since cancer cells proliferate (and hence divide) rapidly, drugs that target cell division have been very successful for therapeutic intervention in cancer treatment. The overwhelming majority of these drugs inhibit mitosis, for example by binding tubulin directly like taxol, or by targeting mitotic kinesins, which are promising targets for clinical development (108). Given the success of anti-mitotic drugs in cancer therapy, it is surprising that cytokinesis has not been targeted on a wide scale. Aurora kinase inhibitors, which target both mitosis and cytokinesis, are currently in clinical trials (109), although the mechanism by which they kill cancer cells is poorly understood. As we improve our understanding of the mechanism of cytokinesis and develop more small molecule probes targeting different proteins within cytokinesis, we anticipate that it will soon be possible to explore the potential “druggability” of cytokinesis.
Acknowledgements
Cytokinesis research in the Eggert lab is supported by NIH grant R01 GM082834, a Human Frontier Science Program Young Investigator Grant, the Claudia Adams Barr Program and the Dana-Farber Cancer Institute.
References
- 1.Eggert US, Mitchison TJ, Field CM. ANIMAL CYTOKINESIS: From Parts List to Mechanisms. Annu Rev Biochem. 2006;75:543–566. doi: 10.1146/annurev.biochem.74.082803.133425. [DOI] [PubMed] [Google Scholar]
- 2.D'Avino PP, Savoian MS, Glover DM. Cleavage furrow formation and ingression during animal cytokinesis: a microtubule legacy. J Cell Sci. 2005;118:1549–1558. doi: 10.1242/jcs.02335. [DOI] [PubMed] [Google Scholar]
- 3.Glotzer M. The molecular requirements for cytokinesis. Science. 2005;307:1735–1739. doi: 10.1126/science.1096896. [DOI] [PubMed] [Google Scholar]
- 4.Glotzer M. Cleavage furrow positioning. J Cell Biol. 2004;164:347–351. doi: 10.1083/jcb.200310112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Maddox AS, Oegema K. Deconstructing cytokinesis. Nat Cell Biol. 2003;5:773–776. doi: 10.1038/ncb0903-773b. [DOI] [PubMed] [Google Scholar]
- 6.Mabuchi I, Okuno M. The effect of myosin antibody on the division of starfish blastomeres. J Cell Biol. 1977;74:251–263. doi: 10.1083/jcb.74.1.251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schroeder TE. Actin in dividing cells: contractile ring filaments bind heavy meromyosin. Proc Natl Acad Sci U S A. 1973;70:1688–1692. doi: 10.1073/pnas.70.6.1688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Finger FP, White JG. Fusion and fission: membrane trafficking in animal cytokinesis. Cell. 2002;108:727–730. doi: 10.1016/s0092-8674(02)00668-2. [DOI] [PubMed] [Google Scholar]
- 9.Mullins JM, Biesele JJ. Terminal phase of cytokinesis in D-98s cells. J Cell Biol. 1977;73:672–684. doi: 10.1083/jcb.73.3.672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Albertson R, Riggs B, Sullivan W. Membrane traffic: a driving force in cytokinesis. Trends Cell Biol. 2005;15:92–101. doi: 10.1016/j.tcb.2004.12.008. [DOI] [PubMed] [Google Scholar]
- 11.Albertson R, Cao J, Hsieh TS, Sullivan W. Vesicles and actin are targeted to the cleavage furrow via furrow microtubules and the central spindle. J Cell Biol. 2008;181:777–790. doi: 10.1083/jcb.200803096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gromley A, Yeaman C, Rosa J, Redick S, Chen CT, Mirabelle S, Guha M, Sillibourne J, Doxsey SJ. Centriolin anchoring of exocyst and SNARE complexes at the midbody is required for secretory-vesicle-mediated abscission. Cell. 2005;123:75–87. doi: 10.1016/j.cell.2005.07.027. [DOI] [PubMed] [Google Scholar]
- 13.Montagnac G, Echard A, Chavrier P. Endocytic traffic in animal cell cytokinesis. Curr Opin Cell Biol. 2008;20:454–461. doi: 10.1016/j.ceb.2008.03.011. [DOI] [PubMed] [Google Scholar]
- 14.Goss JW, Toomre DK. Both daughter cells traffic and exocytose membrane at the cleavage furrow during mammalian cytokinesis. J Cell Biol. 2008;181:1047–1054. doi: 10.1083/jcb.200712137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shuster CB, Burgess DR. Targeted new membrane addition in the cleavage furrow is a late, separate event in cytokinesis. Proc Natl Acad Sci U S A. 2002;99:3633–3638. doi: 10.1073/pnas.052342699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pollard TD. Progress towards understanding the mechanism of cytokinesis in fission yeast. Biochem Soc Trans. 2008;36:425–430. doi: 10.1042/BST0360425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Balasubramanian MK, Bi E, Glotzer M. Comparative analysis of cytokinesis in budding yeast, fission yeast and animal cells. Curr Biol. 2004;14:R806–R818. doi: 10.1016/j.cub.2004.09.022. [DOI] [PubMed] [Google Scholar]
- 18.Wu JQ, Pollard TD. Counting cytokinesis proteins globally and locally in fission yeast. Science. 2005;310:310–314. doi: 10.1126/science.1113230. [DOI] [PubMed] [Google Scholar]
- 19.Eggert US, Kiger AA, Richter C, Perlman ZE, Perrimon N, Mitchison TJ, Field CM. Parallel chemical genetic and genome-wide RNAi screens identify cytokinesis inhibitors and targets. PLoS Biol. 2004;2:e379. doi: 10.1371/journal.pbio.0020379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rappaport R. Cytokinesis in Animal Cells. Cambridge: Cambridge University Press; 1996. [Google Scholar]
- 21.Peterson JR, Mitchison TJ. Small molecules, big impact: a history of chemical inhibitors and the cytoskeleton. Chem Biol. 2002;9:1275–1285. doi: 10.1016/s1074-5521(02)00284-3. [DOI] [PubMed] [Google Scholar]
- 22.MacLean-Fletcher S, Pollard TD. Mechanism of action of cytochalasin B on actin. Cell. 1980;20:329–341. doi: 10.1016/0092-8674(80)90619-4. [DOI] [PubMed] [Google Scholar]
- 23.Dixon B. The cytocholasins and cell mechanics. New Sci. 1966;32:170–171. [Google Scholar]
- 24.Wheatley SP, Wang Y. Midzone microtubule bundles are continuously required for cytokinesis in cultured epithelial cells. J Cell Biol. 1996;135:981–989. doi: 10.1083/jcb.135.4.981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Snyder JA, McLelland SL. Taxol reduces the rate of cytokinesis in PtK1 cells. Cell Biol Int. 1996;20:573–578. doi: 10.1006/cbir.1996.0075. [DOI] [PubMed] [Google Scholar]
- 26.Shannon KB, Canman JC, Ben Moree C, Tirnauer JS, Salmon ED. Taxol-stabilized microtubules can position the cytokinetic furrow in mammalian cells. Mol Biol Cell. 2005;16:4423–4436. doi: 10.1091/mbc.E04-11-0974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Strickland LI, Donnelly EJ, Burgess DR. Induction of Cytokinesis Is Independent of Precisely Regulated Microtubule Dynamics. Mol Biol Cell. 2005 doi: 10.1091/mbc.E05-04-0305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Canman JC, Hoffman DB, Salmon ED. The role of pre- and post-anaphase microtubules in the cytokinesis phase of the cell cycle. Curr Biol. 2000;10:611–614. doi: 10.1016/s0960-9822(00)00490-5. [DOI] [PubMed] [Google Scholar]
- 29.Straight AF, Cheung A, Limouze J, Chen I, Westwood NJ, Sellers JR, Mitchison TJ. Dissecting temporal and spatial control of cytokinesis with a myosin II Inhibitor. Science. 2003;299:1743–1747. doi: 10.1126/science.1081412. [DOI] [PubMed] [Google Scholar]
- 30.Ishizaki T, Uehata M, Tamechika I, Keel J, Nonomura K, Maekawa M, Narumiya S. Pharmacological properties of Y-27632, a specific inhibitor of rho-associated kinases. Mol Pharmacol. 2000;57:976–983. [PubMed] [Google Scholar]
- 31.Kosako H, Yoshida T, Matsumura F, Ishizaki T, Narumiya S, Inagaki M. Rho-kinase/ROCK is involved in cytokinesis through the phosphorylation of myosin light chain and not ezrin/radixin/moesin proteins at the cleavage furrow. Oncogene. 2000;19:6059–6064. doi: 10.1038/sj.onc.1203987. [DOI] [PubMed] [Google Scholar]
- 32.Murthy K, Wadsworth P. Myosin-II-dependent localization and dynamics of F-actin during cytokinesis. Curr Biol. 2005;15:724–731. doi: 10.1016/j.cub.2005.02.055. [DOI] [PubMed] [Google Scholar]
- 33.Lucero A, Stack C, Bresnick AR, Shuster CB. A global, myosin light chain kinase-dependent increase in myosin II contractility accompanies the metaphase-anaphase transition in sea urchin eggs. Mol Biol Cell. 2006;17:4093–4104. doi: 10.1091/mbc.E06-02-0119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Naim V, Imarisio S, Di Cunto F, Gatti M, Bonaccorsi S. Drosophila citron kinase is required for the final steps of cytokinesis. Mol Biol Cell. 2004;15:5053–5063. doi: 10.1091/mbc.E04-06-0536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yamashiro S, Totsukawa G, Yamakita Y, Sasaki Y, Madaule P, Ishizaki T, Narumiya S, Matsumura F. Citron kinase, a Rho-dependent kinase, induces di-phosphorylation of regulatory light chain of myosin II. Mol Biol Cell. 2003;14:1745–1756. doi: 10.1091/mbc.E02-07-0427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Barr FA, Sillje HH, Nigg EA. Polo-like kinases and the orchestration of cell division. Nat Rev Mol Cell Biol. 2004;5:429–440. doi: 10.1038/nrm1401. [DOI] [PubMed] [Google Scholar]
- 37.Brennan IM, Peters U, Kapoor TM, Straight AF. Polo-like kinase controls vertebrate spindle elongation and cytokinesis. PLoS One. 2007;2:e409. doi: 10.1371/journal.pone.0000409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Burkard ME, Randall CL, Larochelle S, Zhang C, Shokat KM, Fisher RP, Jallepalli PV. Chemical genetics reveals the requirement for Polo-like kinase 1 activity in positioning RhoA and triggering cytokinesis in human cells. Proc Natl Acad Sci U S A. 2007;104:4383–4388. doi: 10.1073/pnas.0701140104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Petronczki M, Glotzer M, Kraut N, Peters JM. Polo-like kinase 1 triggers the initiation of cytokinesis in human cells by promoting recruitment of the RhoGEF Ect2 to the central spindle. Dev Cell. 2007;12:713–725. doi: 10.1016/j.devcel.2007.03.013. [DOI] [PubMed] [Google Scholar]
- 40.Santamaria A, Neef R, Eberspacher U, Eis K, Husemann M, Mumberg D, Prechtl S, Schulze V, Siemeister G, Wortmann L, Barr FA, Nigg EA. Use of the novel Plk1 inhibitor ZK-thiazolidinone to elucidate functions of Plk1 in early and late stages of mitosis. Mol Biol Cell. 2007;18:4024–4036. doi: 10.1091/mbc.E07-05-0517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Burkard ME, Maciejowski J, Rodriguez-Bravo V, Repka M, Lowery DM, Clauser KR, Zhang C, Shokat KM, Carr SA, Yaffe MB, Jallepalli PV. Plk1 self-organization and priming phosphorylation of HsCYK-4 at the spindle midzone regulate the onset of division in human cells. PLoS Biol. 2009;7:e1000111. doi: 10.1371/journal.pbio.1000111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wolfe BA, Takaki T, Petronczki M, Glotzer M. Polo-like kinase 1 directs assembly of the HsCyk-4 RhoGAP/Ect2 RhoGEF complex to initiate cleavage furrow formation. PLoS Biol. 2009;7:e1000110. doi: 10.1371/journal.pbio.1000110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ruchaud S, Carmena M, Earnshaw WC. Chromosomal passengers: conducting cell division. Nat Rev Mol Cell Biol. 2007;8:798–812. doi: 10.1038/nrm2257. [DOI] [PubMed] [Google Scholar]
- 44.Hauf S, Cole RW, LaTerra S, Zimmer C, Schnapp G, Walter R, Heckel A, van Meel J, Rieder CL, Peters JM. The small molecule Hesperadin reveals a role for Aurora B in correcting kinetochore-microtubule attachment and in maintaining the spindle assembly checkpoint. J Cell Biol. 2003;161:281–294. doi: 10.1083/jcb.200208092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Miyauchi K, Zhu X, Foong C, Hosoya H, Murata-Hori M. Aurora B kinase activity is required to prevent polar cortical ingression during cytokinesis. Cell Cycle. 2007;6:2549–2553. doi: 10.4161/cc.6.20.4817. [DOI] [PubMed] [Google Scholar]
- 46.Guse A, Mishima M, Glotzer M. Phosphorylation of ZEN-4/MKLP1 by aurora B regulates completion of cytokinesis. Curr Biol. 2005;15:778–786. doi: 10.1016/j.cub.2005.03.041. [DOI] [PubMed] [Google Scholar]
- 47.Skop AR, Bergmann D, Mohler WA, White JG. Completion of cytokinesis in C. elegans requires a brefeldin A-sensitive membrane accumulation at the cleavage furrow apex. Curr Biol. 2001;11:735–746. doi: 10.1016/s0960-9822(01)00231-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Canman JC, Cameron LA, Maddox PS, Straight A, Tirnauer JS, Mitchison TJ, Fang G, Kapoor TM, Salmon ED. Determining the position of the cell division plane. Nature. 2003;424:1074–1078. doi: 10.1038/nature01860. [DOI] [PubMed] [Google Scholar]
- 49.Hu CK, Coughlin M, Field CM, Mitchison TJ. Cell polarization during monopolar cytokinesis. J Cell Biol. 2008;181:195–202. doi: 10.1083/jcb.200711105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.O'Connell CB, Warner AK, Wang Y. Distinct roles of the equatorial and polar cortices in the cleavage of adherent cells. Curr Biol. 2001;11:702–707. doi: 10.1016/s0960-9822(01)00181-6. [DOI] [PubMed] [Google Scholar]
- 51.Guha M, Zhou M, Wang YL. Cortical actin turnover duing cytokinesis requires myosin II. Curr Biol. 2005;15:732–736. doi: 10.1016/j.cub.2005.03.042. [DOI] [PubMed] [Google Scholar]
- 52.Echard A. Membrane traffic and polarization of lipid domains during cytokinesis. Biochem Soc Trans. 2008;36:395–399. doi: 10.1042/BST0360395. [DOI] [PubMed] [Google Scholar]
- 53.Logan MR, Mandato CA. Regulation of the actin cytoskeleton by PIP2 in cytokinesis. Biol Cell. 2005;98:377–388. doi: 10.1042/BC20050081. [DOI] [PubMed] [Google Scholar]
- 54.Ng MM, Chang F, Burgess DR. Movement of membrane domains and requirement of membrane signaling molecules for cytokinesis. Dev Cell. 2005;9:781–790. doi: 10.1016/j.devcel.2005.11.002. [DOI] [PubMed] [Google Scholar]
- 55.Fernandez C, Lobo Md Mdel V, Gomez-Coronado D, Lasuncion MA. Cholesterol is essential for mitosis progression and its deficiency induces polyploid cell formation. Exp Cell Res. 2004;300:109–120. doi: 10.1016/j.yexcr.2004.06.029. [DOI] [PubMed] [Google Scholar]
- 56.Wachtler V, Rajagopalan S, Balasubramanian MK. Sterol-rich plasma membrane domains in the fission yeast Schizosaccharomyces pombe. J Cell Sci. 2003;116:867–874. doi: 10.1242/jcs.00299. [DOI] [PubMed] [Google Scholar]
- 57.Emoto K, Umeda M. An essential role for a membrane lipid in cytokinesis. Regulation of contractile ring disassembly by redistribution of phosphatidylethanolamine. J Cell Biol. 2000;149:1215–1224. doi: 10.1083/jcb.149.6.1215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Emoto K, Kobayashi T, Yamaji A, Aizawa H, Yahara I, Inoue K, Umeda M. Redistribution of phosphatidylethanolamine at the cleavage furrow of dividing cells during cytokinesis. Proc Natl Acad Sci U S A. 1996;93:12867–12872. doi: 10.1073/pnas.93.23.12867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Emoto K, Inadome H, Kanaho Y, Narumiya S, Umeda M. Local change in phospholipid composition at the cleavage furrow is essential for completion of cytokinesis. J Biol Chem. 2005;280:37901–37907. doi: 10.1074/jbc.M504282200. [DOI] [PubMed] [Google Scholar]
- 60.Field SJ, Madson N, Kerr ML, Galbraith KA, Kennedy CE, Tahiliani M, Wilkins A, Cantley LC. PtdIns(4,5)P2 functions at the cleavage furrow during cytokinesis. Curr Biol. 2005;15:1407–1412. doi: 10.1016/j.cub.2005.06.059. [DOI] [PubMed] [Google Scholar]
- 61.Kanazawa T, Nakamura S, Momoi M, Yamaji T, Takematsu H, Yano H, Sabe H, Yamamoto A, Kawasaki T, Kozutsumi Y. Inhibition of cytokinesis by a lipid metabolite, psychosine. J Cell Biol. 2000;149:943–950. doi: 10.1083/jcb.149.4.943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kittler R, Pelletier L, Heninger AK, Slabicki M, Theis M, Miroslaw L, Poser I, Lawo S, Grabner H, Kozak K, Wagner J, Surendranath V, Richter C, Bowen W, Jackson AL, Habermann B, Hyman AA, Buchholz F. Genome-scale RNAi profiling of cell division in human tissue culture cells. Nat Cell Biol. 2007;9:1401–1412. doi: 10.1038/ncb1659. [DOI] [PubMed] [Google Scholar]
- 63.Echard A, Hickson GR, Foley E, O'Farrell PH. Terminal cytokinesis events uncovered after an RNAi screen. Curr Biol. 2004;14:1685–1693. doi: 10.1016/j.cub.2004.08.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Janetopoulos C, Devreotes P. Phosphoinositide signaling plays a key role in cytokinesis. J Cell Biol. 2006;174:485–490. doi: 10.1083/jcb.200603156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Han JK, Fukami K, Nuccitelli R. Reducing inositol lipid hydrolysis, Ins(1,4,5)P3 receptor availability, or Ca2+ gradients lengthens the duration of the cell cycle in Xenopus laevis blastomeres. J Cell Biol. 1992;116:147–156. doi: 10.1083/jcb.116.1.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Saul D, Fabian L, Forer A, Brill JA. Continuous phosphatidylinositol metabolism is required for cleavage of crane fly spermatocytes. J Cell Sci. 2004;117:3887–3896. doi: 10.1242/jcs.01236. [DOI] [PubMed] [Google Scholar]
- 67.Giansanti MG, Bonaccorsi S, Kurek R, Farkas RM, Dimitri P, Fuller MT, Gatti M. The class I PITP giotto is required for Drosophila cytokinesis. Curr Biol. 2006;16:195–201. doi: 10.1016/j.cub.2005.12.011. [DOI] [PubMed] [Google Scholar]
- 68.Niggli V, Rossy J. Ezrin/radixin/moesin: versatile controllers of signaling molecules and of the cortical cytoskeleton. Int J Biochem Cell Biol. 2008;40:344–349. doi: 10.1016/j.biocel.2007.02.012. [DOI] [PubMed] [Google Scholar]
- 69.Hao JJ, Liu Y, Kruhlak M, Debell KE, Rellahan BL, Shaw S. Phospholipase C-mediated hydrolysis of PIP2 releases ERM proteins from lymphocyte membrane. J Cell Biol. 2009;184:451–462. doi: 10.1083/jcb.200807047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Yoshida S, Bartolini S, Pellman D. Mechanisms for concentrating Rho1 during cytokinesis. Genes Dev. 2009;23:810–823. doi: 10.1101/gad.1785209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Gimpl G, Gehrig-Burger K. Cholesterol reporter molecules. Biosci Rep. 2007;27:335–358. doi: 10.1007/s10540-007-9060-1. [DOI] [PubMed] [Google Scholar]
- 72.Feng B, Schwarz H, Jesuthasan S. Furrow-specific endocytosis during cytokinesis of zebrafish blastomeres. Exp Cell Res. 2002;279:14–20. doi: 10.1006/excr.2002.5579. [DOI] [PubMed] [Google Scholar]
- 73.Schneiter R, Brugger B, Amann CM, Prestwich GD, Epand RF, Zellnig G, Wieland FT, Epand RM. Identification and biophysical characterization of a very-long-chain-fatty-acid-substituted phosphatidylinositol in yeast subcellular membranes. Biochem J. 2004;381:941–949. doi: 10.1042/BJ20040320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Giansanti MG, Farkas RM, Bonaccorsi S, Lindsley DL, Wakimoto BT, Fuller MT, Gatti M. Genetic dissection of meiotic cytokinesis in Drosophila males. Mol Biol Cell. 2004;15:2509–2522. doi: 10.1091/mbc.E03-08-0603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Szafer-Glusman E, Giansanti MG, Nishihama R, Bolival B, Pringle J, Gatti M, Fuller MT. A role for very-long-chain fatty acids in furrow ingression during cytokinesis in Drosophila spermatocytes. Curr Biol. 2008;18:1426–1431. doi: 10.1016/j.cub.2008.08.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Nakamura S, Kozutsumi Y, Sun Y, Miyake Y, Fujita T, Kawasaki T. Dual roles of sphingolipids in signaling of the escape from and onset of apoptosis in a mouse cytotoxic T-cell line, CTLL-2. J Biol Chem. 1996;271:1255–1257. doi: 10.1074/jbc.271.3.1255. [DOI] [PubMed] [Google Scholar]
- 77.Miyake Y, Kozutsumi Y, Nakamura S, Fujita T, Kawasaki T. Serine palmitoyltransferase is the primary target of a sphingosine-like immunosuppressant, ISP-1/myriocin. Biochem Biophys Res Commun. 1995;211:396–403. doi: 10.1006/bbrc.1995.1827. [DOI] [PubMed] [Google Scholar]
- 78.Im DS, Heise CE, Nguyen T, O'Dowd BF, Lynch KR. Identification of a molecular target of psychosine and its role in globoid cell formation. J Cell Biol. 2001;153:429–434. doi: 10.1083/jcb.153.2.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Suzuki K. Twenty five years of the "psychosine hypothesis": a personal perspective of its history and present status. Neurochem Res. 1998;23:251–259. doi: 10.1023/a:1022436928925. [DOI] [PubMed] [Google Scholar]
- 80.Mitchison TJ. Psychosine, cytokinesis, and orphan receptors. Unexpected connections. J Cell Biol. 2001;153:F1–F3. doi: 10.1083/jcb.153.2.f1. [DOI] [PubMed] [Google Scholar]
- 81.Wang JQ, Kon J, Mogi C, Tobo M, Damirin A, Sato K, Komachi M, Malchinkhuu E, Murata N, Kimura T, Kuwabara A, Wakamatsu K, Koizumi H, Uede T, Tsujimoto G, Kurose H, Sato T, Harada A, Misawa N, Tomura H, Okajima F. TDAG8 is a proton-sensing and psychosine-sensitive G-protein-coupled receptor. J Biol Chem. 2004;279:45626–45633. doi: 10.1074/jbc.M406966200. [DOI] [PubMed] [Google Scholar]
- 82.Sunkara PS, Rao PN, Nishioka K, Brinkley BR. Role of polyamines in cytokinesis of mammalian cells. Exp Cell Res. 1979;119:63–68. doi: 10.1016/0014-4827(79)90335-5. [DOI] [PubMed] [Google Scholar]
- 83.Oriol-Audit C. Polyamine-induced actin polymerization. Eur J Biochem. 1978;87:371–376. doi: 10.1111/j.1432-1033.1978.tb12386.x. [DOI] [PubMed] [Google Scholar]
- 84.Oriol-Audit C. Actin-polyamines interaction : relationship between physicochemical properties and cytokinesis induction. Biochem Biophys Res Commun. 1982;105:1096–1101. doi: 10.1016/0006-291x(82)91082-8. [DOI] [PubMed] [Google Scholar]
- 85.Webb SE, Li WM, Miller AL. Calcium signalling during the cleavage period of zebrafish development. Philos Trans R Soc Lond B Biol Sci. 2008;363:1363–1369. doi: 10.1098/rstb.2007.2253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Muto A, Kume S, Inoue T, Okano H, Mikoshiba K. Calcium waves along the cleavage furrows in cleavage-stage Xenopus embryos and its inhibition by heparin. J Cell Biol. 1996;135:181–190. doi: 10.1083/jcb.135.1.181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Noguchi T, Mabuchi I. Localized calcium signals along the cleavage furrow of the Xenopus egg are not involved in cytokinesis. Mol Biol Cell. 2002;13:1263–1273. doi: 10.1091/mbc.01-10-0501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Wong R, Hadjiyanni I, Wei HC, Polevoy G, McBride R, Sem KP, Brill JA. PIP2 hydrolysis and calcium release are required for cytokinesis in Drosophila spermatocytes. Curr Biol. 2005;15:1401–1406. doi: 10.1016/j.cub.2005.06.060. [DOI] [PubMed] [Google Scholar]
- 89.Mitsuyama F, Sawai T, Carafoli E, Furuichi T, Mikoshiba K. Microinjection of Ca2+ store-enriched microsome fractions to dividing newt eggs induces extra-cleavage furrows via inositol 1,4,5-trisphosphate-induced Ca2+ release. Dev Biol. 1999;214:160–167. doi: 10.1006/dbio.1999.9416. [DOI] [PubMed] [Google Scholar]
- 90.Creton R, Speksnijder JE, Jaffe LF. Patterns of free calcium in zebrafish embryos. J Cell Sci. 1998;111(Pt 12):1613–1622. doi: 10.1242/jcs.111.12.1613. [DOI] [PubMed] [Google Scholar]
- 91.Rudolf R, Mongillo M, Rizzuto R, Pozzan T. Looking forward to seeing calcium. Nat Rev Mol Cell Biol. 2003;4:579–586. doi: 10.1038/nrm1153. [DOI] [PubMed] [Google Scholar]
- 92.Li WM, Webb SE, Chan CM, Miller AL. Multiple roles of the furrow deepening Ca2+ transient during cytokinesis in zebrafish embryos. Dev Biol. 2008;316:228–248. doi: 10.1016/j.ydbio.2008.01.027. [DOI] [PubMed] [Google Scholar]
- 93.Ciapa B, Pesando D, Wilding M, Whitaker M. Cell-cycle calcium transients driven by cyclic changes in inositol trisphosphate levels. Nature. 1994;368:875–878. doi: 10.1038/368875a0. [DOI] [PubMed] [Google Scholar]
- 94.Lee KW, Webb SE, Miller AL. Ca2+ released via IP3 receptors is required for furrow deepening during cytokinesis in zebrafish embryos. Int J Dev Biol. 2003;47:411–421. [PubMed] [Google Scholar]
- 95.Ito J, Yoon SY, Lee B, Vanderheyden V, Vermassen E, Wojcikiewicz R, Alfandari D, De Smedt H, Parys JB, Fissore RA. Inositol 1,4,5-trisphosphate receptor 1, a widespread Ca2+ channel, is a novel substrate of polo-like kinase 1 in eggs. Dev Biol. 2008;320:402–413. doi: 10.1016/j.ydbio.2008.05.548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Bamburg JR, McGough A, Ono S. Putting a new twist on actin: ADF/cofilins modulate actin dynamics. Trends Cell Biol. 1999;9:364–370. doi: 10.1016/s0962-8924(99)01619-0. [DOI] [PubMed] [Google Scholar]
- 97.Nusco GA, Chun JT, Ercolano E, Lim D, Gragnaniello G, Kyozuka K, Santella L. Modulation of calcium signalling by the actin-binding protein cofilin. Biochem Biophys Res Commun. 2006;348:109–114. doi: 10.1016/j.bbrc.2006.07.023. [DOI] [PubMed] [Google Scholar]
- 98.Crivici A, Ikura M. Molecular and structural basis of target recognition by calmodulin. Annu Rev Biophys Biomol Struct. 1995;24:85–116. doi: 10.1146/annurev.bb.24.060195.000505. [DOI] [PubMed] [Google Scholar]
- 99.Li CJ, Heim R, Lu P, Pu Y, Tsien RY, Chang DC. Dynamic redistribution of calmodulin in HeLa cells during cell division as revealed by a GFP-calmodulin fusion protein technique. J Cell Sci. 1999;112(Pt 10):1567–1577. doi: 10.1242/jcs.112.10.1567. [DOI] [PubMed] [Google Scholar]
- 100.Liu T, Williams JG, Clarke M. Inducible expression of calmodulin antisense RNA in Dictyostelium cells inhibits the completion of cytokinesis. Mol Biol Cell. 1992;3:1403–1413. doi: 10.1091/mbc.3.12.1403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Yu YY, Dai G, Pan FY, Chen J, Li CJ. Calmodulin regulates the post-anaphase reposition of centrioles during cytokinesis. Cell Res. 2005;15:548–552. doi: 10.1038/sj.cr.7290324. [DOI] [PubMed] [Google Scholar]
- 102.Wong R, Fabian L, Forer A, Brill JA. Phospholipase C and myosin light chain kinase inhibition define a common step in actin regulation during cytokinesis. BMC Cell Biol. 2007;8:15. doi: 10.1186/1471-2121-8-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Yamamoto H, Yamauchi E, Taniguchi H, Ono T, Miyamoto E. Phosphorylation of microtubule-associated protein tau by Ca2+/calmodulindependent protein kinase II in its tubulin binding sites. Arch Biochem Biophys. 2002;408:255–262. doi: 10.1016/s0003-9861(02)00556-8. [DOI] [PubMed] [Google Scholar]
- 104.Schollmeyer JE. Calpain II involvement in mitosis. Science. 1988;240:911–913. doi: 10.1126/science.2834825. [DOI] [PubMed] [Google Scholar]
- 105.Tonami K, Kurihara Y, Aburatani H, Uchijima Y, Asano T, Kurihara H. Calpain 6 is involved in microtubule stabilization and cytoskeletal organization. Mol Cell Biol. 2007;27:2548–2561. doi: 10.1128/MCB.00992-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Tomas A, Futter C, Moss SE. Annexin 11 is required for midbody formation and completion of the terminal phase of cytokinesis. J Cell Biol. 2004;165:813–822. doi: 10.1083/jcb.200311054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Fujiwara T, Bandi M, Nitta M, Ivanova EV, Bronson RT, Pellman D. Cytokinesis failure generating tetraploids promotes tumorigenesis in p53-null cells. Nature. 2005;437:1043–1047. doi: 10.1038/nature04217. [DOI] [PubMed] [Google Scholar]
- 108.Bergnes G, Brejc K, Belmont L. Mitotic kinesins: prospects for antimitotic drug discovery. Curr Top Med Chem. 2005;5:127–145. doi: 10.2174/1568026053507697. [DOI] [PubMed] [Google Scholar]
- 109.Harrington EA, Bebbington D, Moore J, Rasmussen RK, Ajose-Adeogun AO, Nakayama T, Graham JA, Demur C, Hercend T, Diu-Hercend A, Su M, Golec JM, Miller KM. VX-680, a potent and selective small-molecule inhibitor of the Aurora kinases, suppresses tumor growth in vivo. Nat Med. 2004;10:262–267. doi: 10.1038/nm1003. [DOI] [PubMed] [Google Scholar]