Abstract
Background We have described an unusual sinonasal neoplasm which is a histological mimic of renal cell carcinoma (RCC) and coined the nosological classification “sinonasal renal cell-like adenocarcinoma” (SRCLA) to describe this unusual entity. Since the original description (Zur et al. Otolaryngol Head Neck Surg 128:441–7, 2002), we have reviewed the case reported by Moh’d Hadi et al. (Rhinology 40:44–7, 2002) and have seen two new cases in consultation. Our purpose here is to describe the additional cases and to extend the reported outcome for these patients. Design Four patients were identified. Slides and immunohistochemistry results were reviewed in consultation. Updated clinical follow-up was obtained from the respective clinicians. Results This group consisted of three women, one man, 22–69 years, and mean 46. Three tumors were in the nasal cavity and one was in the nasopharynx. Histologically, these tumors were uniformly composed of clear cells, forming either solid or glandular patterns. The tumor cells were cuboidal to polyhedral; transition to short spindle cells was seen in one case. One case revealed moderate nuclear pleomorphism. No perineural or vascular invasion, or necrosis was seen. No mucin-producing or squamous elements were seen. Immunohistochemistry (IHC) revealed the following staining profile: CK7 + (4/4), CK20 + (focal 1/4), S100 + (1/4), and CD10 + (1/2). No staining was seen for vimentin (0/4), RCC (0/2), thyroglobulin (0/2), actin (0/2), or calponin (0/2). Three patients were treated primarily with surgery, two patients also received adjuvant radiotherapy (RT); the fourth patient was treated with primary RT. All patients are disease-free, based on endoscopy and/or radiography, 2, 4, 5 and 8 years after diagnosis. Renal cell carcinoma has not been identified in any patient. Conclusion Sinonasal renal cell-like adenocarcinoma is a rare and distinct entity noteworthy in its resemblance to RCC. Immunohistochemistry can easily distinguish between these two tumors. No patient developed recurrent or metastatic disease, or was found to have RCC. Greater experience will allow us to fully understand its long-term behavior and arrive at more standardized therapeutic recommendations.
Keywords: Sinonasal, Renal cell carcinoma, Renal cell-like carcinoma, Vermeer
Introduction
Sinonasal renal cell-like adenocarcinoma (SRCLA) is a rare tumor first described by Zur et al. [1] and Hadi et al. [2]. The most striking feature is its remarkable resemblance to the clear cell type of renal cell carcinoma (RCC). Indeed, in the absence of a history of RCC, a renal primary neoplasm still needs to be ruled out. Since 2002, we have reviewed two additional cases in consultation. We now report on pathologic features of the group of tumors, as well as updated clinical outcome data for each patient.
Methods
Four patients with SRCLA were identified. Hematoxylin-and-eosin (H&E) stained slides were reviewed in consultation, along with immunohistochemistry. Where possible, tissue blocks or unstained slides were obtained from the respective medical centers and additional immunohistochemical stains were performed. Clinical follow-up information was obtained for each patient from the respective clinicians.
Results
Clinical, Treatment and Outcome
The patient population consisted of three women and one man, ages 22–69 (mean 46), originating from the United States (2), England (1), and Lebanon (1). All patients presented with epistaxis, two complained of nasal obstruction, and two complained of headaches. One patient reported nasal and orbital pressure with epiphora, and one noted slightly impaired olfaction.
Three tumors were located in the nasal cavity and one was in the nasopharynx. The tumors grossly appeared as red polypoid masses (two patients), a submucosal elevation in the nasopharynx (one patient) and a spherical soft tissue mass on CT scan (one patient).
Two patients were treated with surgical resection or endoscopic nasal polypectomy followed by adjuvant radiation therapy. One patient underwent tumor embolization followed by surgical excision the following day, and the last received intensity-modulated radiation therapy alone.
All four patients are well with no disease progression at 2–8 years (mean 4.75) based on endoscopic and/or radiographic findings. One patient developed radiation sequelae with the incidental radiographic and endoscopic findings of prevertebral and parapharyngeal space fibrosis. All patients have had renal imaging studies either at the time of, or following, initial diagnosis. No evidence of renal neoplasia was found.
Pathologic Findings
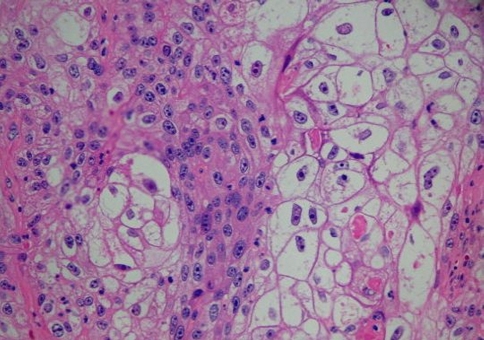
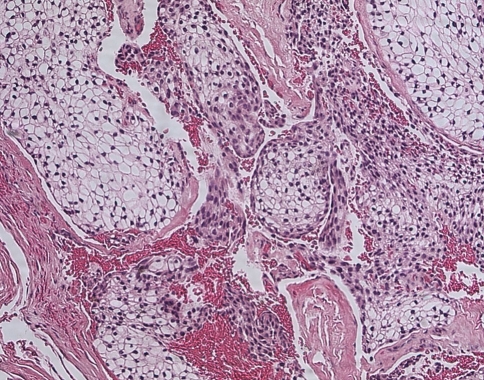
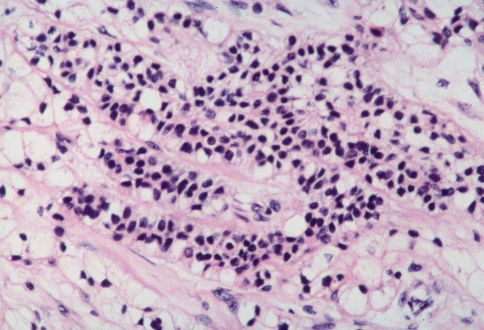
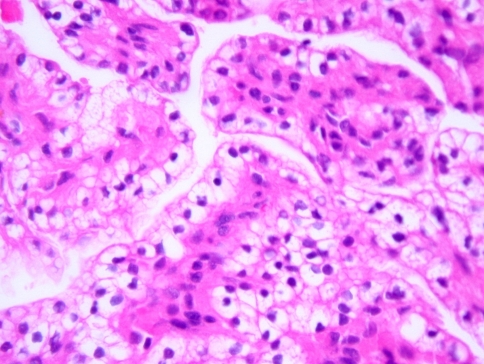
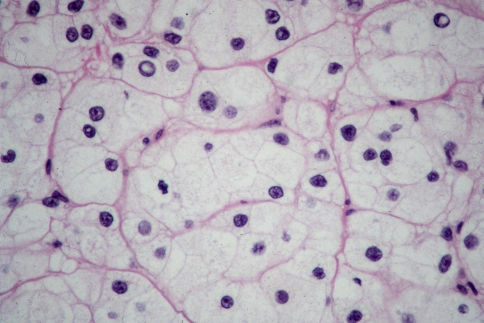
Histologically, all four tumors closely resembled clear cell renal cell carcinoma. The predominant architecture was a solid to nested, glandular growth pattern with intervening fibrous septae and prominent vascularity (Fig. 1). The tumor cells were uniform, cuboidal to polyhedral shaped with abundant clear cytoplasm (Fig. 2). The nuclear:cytoplasmic ratio was low, and the nuclei were small. One case showed mild pleomorphism including some variation in nuclear size and shape; this case also exhibited some spindle cells (Fig. 3). Intranuclear holes were prominent in one case (Fig. 4). No increased mitotic activity, necrosis, or perineural invasion was identified in any tumor.
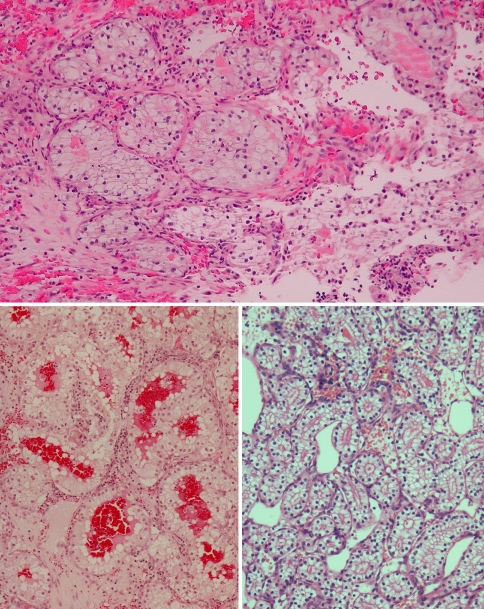
Fig. 1.
Upper panel: A clear cell neoplasm in a hemorrhagic background brings to mind metastatic renal cell carcinoma. Lower panels: Sinonasal renal cell-like carcinoma (SNRCLC) forms follicular and glandular structures; this would not be seen with salivary clear cell carcinoma
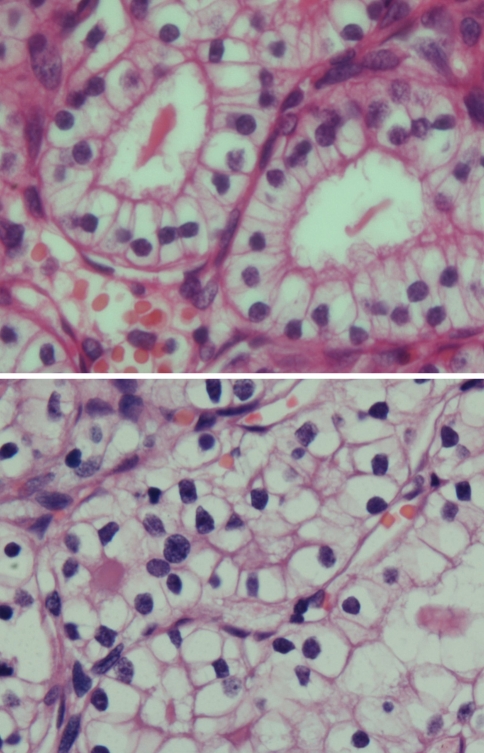
Fig. 2.
The tumor cells are cuboidal to polyhedral, with abundant clear cytoplasm and irregularly shaped nuclei. Here they form glandular spaces containing eosinophilic secretions
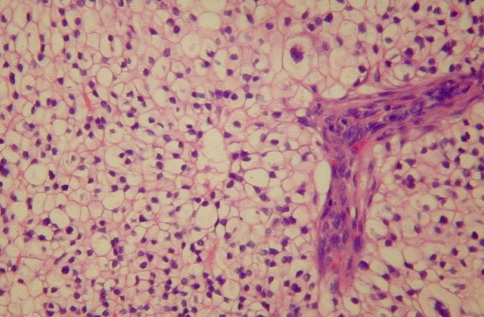
Fig. 3.
Solid pattern of SNRCLC with greater pleomorphism. The nuclei are peripheral, shrunken, and more irregularly shaped. The nuclear/cytoplasmic ratio remains low; necrosis and mitotic activity are not seen
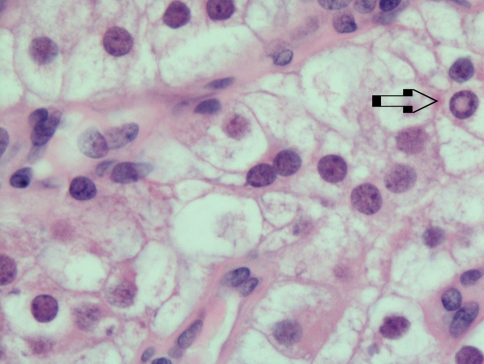
Fig. 4.
Intranuclear holes (arrow)
Immunohistochemistry
The tumor cells were uniformly positive for CK7 + (4/4) and negative for vimentin (0/4), RCC (0/2), thyroglobulin (0/2), actin (0/2), and calponin (0/2). Discordance in immunohistochemical staining was observed for the following markers: CK20 + (focal 1/4), S100 + (1/4), CD10 + (1/2). A limited amount of tissue was available for one case, and a full immunohistochemical profile could not be obtained (Table 1).
Table 1.
Comparison of immunohistochemical staining patterns of SRCLA and RCC
| CK7 | CK20 | CD10 | Vim | RCC | S100 | CK HMW | AE13 | Cam5.2 | CK LMW | TTF1 | Calponin | Actin | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Patient 1 | Pos | Neg | Neg | Neg | Pos | Neg | Neg | ||||||
| Patient 2 | Pos | Neg | Neg | Neg | Neg | Pos | Pos | Pos | Pos | Neg | Neg | ||
| Patient 3 | Pos | Pos (focal) | Pos | Neg | Neg | Neg | Neg | ||||||
| Patient 4 | Pos | Neg | Neg | Neg | |||||||||
| Renal cell carcinoma | Variable | Neg | Pos | Pos | Pos | Variable | Neg | Variable | Pos | Pos | Neg | Neg | Neg |
Discussion
SRCLA represents an extremely rare neoplasm; the diagnosis necessitates an exclusionary approach. Foremost, metastatic RCC needs to be ruled out. Metastatic RCC has a predilection for the sinonasal tract [3–5] and sinonasal metastatic disease may be the initial presentation [6–8]. RCC is the most common carcinoma to metastasize to the sinonasal tract [9]. Overall, metastatic RCC comprises approximately 6% of all metastatic tumors to the head and neck [10].
SRCLA can be distinguished from RCC by immunohistochemistry; a useful distinguishing panel includes vimentin, RCC, and CK7. Vimentin and RCC are usually positive in conventional clear cell RCC; both markers are negative in all four cases of SRCLA. Only a minority of conventional clear cell RCC express CK7 [11–13] whereas all cases of SRCLA are uniformly positive for CK7. Table 1 details the immunohistochemical distinctions between conventional clear cell RCC and our cases of SRCLA. Despite differences in the immunohistochemical expression profile, it remains important to exclude radiographically a renal neoplasm.
A number of other entities must be also considered in the differential diagnosis of SRCLA: squamous cell carcinoma with clear cell change, clear cell predominant mucoepidermoid carcinoma, salivary clear cell carcinoma, endolymphatic sac tumor, metastatic clear cell variant of follicular thyroid carcinoma, and “balloon cell” melanoma.
Squamous cell carcinoma (SCC) is the most common sinonasal malignancy; clear cell change can be prominent in SCC either due to glycogen accumulation or secondary to treatment effect (Fig. 5). Evidence of keratinization, intercellular bridges, and an infiltrative growth pattern distinguish SCC with clear cell change from SRCLA.
Fig. 5.
SCC with prominent clear cell change
Mucoepidermoid carcinoma (MEC) should be considered next in the differential diagnosis. Although it is one of the most common salivary malignancies, it remains relatively uncommon to the sinonasal tract [14–18]. Clear cell-predominant variant of MEC resembles SRCLA [19] and can occur in the sinonasal tract [18]. Careful evaluation will reveal mucin-producing cells and foci of keratinizing squamous cells in clear cell-predominant MEC (Fig. 6).
Fig. 6.
Clear cell predominant MEC, careful searching will confirm the presence of at least focal mucinous and squamous elements
Salivary clear cell carcinoma (CCC) is a low-grade carcinoma most often arising in the palate. CCC mimics SRCLA with low-grade nuclei, occasional pleomorphism, clear cytoplasm, and well-defined cell borders. SRCLA is best distinguished from CCC by histomorphology, as the immunohistochemical staining patterns can be quite similar between these two tumors. The most distinguishing feature is that CCC does not form glandular structures, but rather grows as sheets of cells, whereas SRCLA can form true glandular/follicular structures (Fig. 7). The site of predilection for CCC (oral cavity, especially palate) is very different from SRCLA; however, one case of CCC of the maxilla has been reported [20].
Fig. 7.
The tumor cells of salivary clear cell carcinoma are generally smaller than those of SRCLA. No follicular or glandular spaces are seen in salivary clear cell carcinoma
Endolymphatic sac tumors can enter into the differential diagnosis of clear cell neoplasia, but their site of occurrence (inner ear/skull base) is anatomically distinct from the sinonasal tract/nasopharynx. Patients with endolymphatic sac tumors often have characteristic signs and symptoms referable to the inner ear (hearing loss, tinnitus, and vertigo). Endolymphatic sac tumors have clear cytoplasm, uniform nuclei, limited pleomorphism, no necrosis, and limited mitotic activity. Architecturally, endolymphatic sac tumors form distinctive papillary fronds, which are not seen in SRCLA (Fig. 8). Imaging studies evaluating the skull base are important, especially if there is a history of Von Hippel-Lindau syndrome.
Fig. 8.
Endolymphatic space tumor has papillary features and the tumor is negative for CD10
The diagnosis of clear cell variant of follicular thyroid carcinoma (FTC) should also be considered. This well-differentiated tumor has a clear cell appearance similar to SRCLA; both may produce follicular structures. Although metastatic clear cell variant of FTC to the head and neck has been described [21], no sinonasal metastases have thus been reported. IHC for thyroglobulin and TTF-1 will be positive for clear cell variant of FTC.
Balloon cell melanoma is a rare variant of melanoma composed predominantly or entirely of cells containing large amounts of clear, vacuolated cytoplasm. Because of its rarity and resemblance to other clear cell tumors, it presents a unique diagnostic challenge (Fig. 9). However, Baehner [22] described a case of metastatic balloon cell melanoma in a cervical lymph node with no known primary lesion, requiring parotidectomy and lymph node dissection. In such cases, an appropriate immunohistochemical panel including HMB-45, Melan-A and S100 is helpful in excluding this diagnosis.
Fig. 9.
Balloon cell melanoma
In summary, additional experience is needed regarding SRCLA with respect to optimal treatment. Fortunately, the apparently favorable prognosis of this low-grade carcinoma may preclude the need for multimodality therapeutic algorithms. More cases are required, however, to better define short- and long-term clinical behavior, risk factors (if any), pathogenesis, cytogenetic abnormalities, and other relevant information; this necessitates continued surveillance to identify additional cases. In addition, further use of special stains, immunohistochemical testing and investigation of ultrastructural features would also be of interest in defining this group of tumors. The actual prevalence of SRCLC is unknown, however, it is quite rare. Perhaps as SRCLC gains recognition, more lesions will be diagnosed allowing for collection of more accurate clinicopathologic data.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
References
- 1.Zur KB, Brandwein M, Wang B, et al. Primary description of a new entity, renal cell-like carcinoma of the nasal cavity: van Meegeren in the house of Vermeer. Arch Otolaryngol Head Neck Surg. 2002;128:441–7. doi: 10.1001/archotol.128.4.441. [DOI] [PubMed] [Google Scholar]
- 2.Moh’d Hadi U, Kahwaji GJ, Mufarrij AA, et al. Low grade primary clear cell carcinoma of the sinonasal tract. Rhinology. 2002;40:44–7. [PubMed] [Google Scholar]
- 3.Yee LL, Keng CG. A rare case of renal cell carcinoma metastatic to the sinonasal area. Ear Nose Throat J. 2001;80:462–7. [PubMed] [Google Scholar]
- 4.Ziari M, Shen S, Amato RJ, Teh BS. Metastatic renal cell carcinoma to the nose and ethmoid sinus. Urology. 2006;67:199. doi: 10.1016/j.urology.2005.07.052. [DOI] [PubMed] [Google Scholar]
- 5.Cheng ET, Greene D, Koch RJ. Metastatic renal cell carcinoma to the nose. Otolaryngol Head Neck Surg. 2000;122:464. doi: 10.1016/S0194-5998(00)70069-6. [DOI] [PubMed] [Google Scholar]
- 6.Lee S, Whipple M. Metastatic renal cell carcinoma to the nasal cavity. Am J Otolaryngol. 2004;25:255–7. doi: 10.1016/j.amjoto.2004.02.003. [DOI] [PubMed] [Google Scholar]
- 7.Singh I, Khaitan A. Diplopia—an unusual primary manifestation of metastatic renal cell carcinoma. Urol Int. 2004;73:285–6. doi: 10.1159/000080845. [DOI] [PubMed] [Google Scholar]
- 8.Lee HM, Kang HJ, Lee SH. Metastatic renal cell carcinoma presenting as epistaxis. Eur Arch Otorhinolaryngol. 2005;262:69–71. doi: 10.1007/s00405-003-0671-2. [DOI] [PubMed] [Google Scholar]
- 9.Barnes L, Tse LLY, Hunt J. Secondary tumors. In chapter 1: nasal cavity and paranasal sinuses. World Health Organization classification of tumors. Pathology and genetics, head and neck tumors. Lyon, France: IARC; 2005.
- 10.Flocks RH, Boatman DL. Incidence of head and neck metastases from genito-urinary neoplasms. Laryngoscope. 1973;83:1527–39. doi: 10.1288/00005537-197309000-00011. [DOI] [PubMed] [Google Scholar]
- 11.Allory Y, Bazille C, Vieillefond A, et al. Profiling and classification tree applied to renal epithelial tumours. Histopathology. 2008;52:158–66. doi: 10.1111/j.1365-2559.2007.02900.x. [DOI] [PubMed] [Google Scholar]
- 12.Pan CC, Chen PC, Tsay SH, Ho DM. Differential immunoprofiles of hepatocellular carcinoma, renal cell carcinoma, and adrenocortical carcinoma: a systemic immunohistochemical survey using tissue array technique. Appl Immunohistochem Mol Morphol. 2005;13:347–52. doi: 10.1097/01.pai.0000146525.72531.19. [DOI] [PubMed] [Google Scholar]
- 13.Ozolek JA, Bastacky SI, Myers EN, Hunt JL. Immunophenotypic comparison of salivary gland oncocytoma and metastatic renal cell carcinoma. Laryngoscope. 2005;115:1097–100. doi: 10.1097/01.MLG.0000163497.61332.77. [DOI] [PubMed] [Google Scholar]
- 14.Brandwein MS, Ivanov K, Wallace DI, et al. Mucoepidermoid carcinoma: a clinicopathologic study of 80 patients with special reference to histological grading. Am J Surg Pathol. 2001;25:835–45. doi: 10.1097/00000478-200107000-00001. [DOI] [PubMed] [Google Scholar]
- 15.Triantafillidou K, Dimitrakopoulos J, Iordanidis F, Koufogiannis D. Mucoepidermoid carcinoma of minor salivary glands: a clinical study of 16 cases and review of the literature. Oral Dis. 2006;12:364–70. doi: 10.1111/j.1601-0825.2005.01166.x. [DOI] [PubMed] [Google Scholar]
- 16.Kokemueller H, Brueggemann N, Swennen G, Eckardt A. Mucoepidermoid carcinoma of the salivary glands—clinical review of 42 cases. Oral Oncol. 2005;41:3–10. doi: 10.1016/j.oraloncology.2004.01.017. [DOI] [PubMed] [Google Scholar]
- 17.Thomas GR, Regalado JJ, McClinton M. A rare case of mucoepidermoid carcinoma of the nasal cavity. Ear Nose Throat J. 2002;81:519–22. [PubMed] [Google Scholar]
- 18.Peison B, Benisch B, Schwartz IS, Gordon RE. Clear-cell mucoepidermoid carcinoma arising in the nasal cavity: case report with ultrastructural observations. Mt Sinai J Med. 1988;55:417–20. [PubMed] [Google Scholar]
- 19.Brandwein M, Hille JJ, Gnepp D, et al. The many faces of mucoepidermoid carcinoma. Pathol Case Rev. 2000;5:214–20. doi: 10.1097/00132583-200005040-00007. [DOI] [Google Scholar]
- 20.Sahasrabudhe NS, Puranik SC, Holla VV. Clear cell adenocarcinoma of the maxillary sinus: a case report. Ind J Pathol Microbiol. 2003;46:93–95. [PubMed] [Google Scholar]
- 21.Kelessis NG, Prassas EP, Dascalopoulou DV, et al. Unusual metastatic spread of follicular thyroid carcinoma: report of a case. Surg Today. 2005;35:300–3. doi: 10.1007/s00595-004-2922-2. [DOI] [PubMed] [Google Scholar]
- 22.Baehner FL, Ng B, Sudilovsky D. Metastatic balloon cell melanoma: a case report. Acta Cytol. 2005;49:543–8. doi: 10.1159/000326203. [DOI] [PubMed] [Google Scholar]