Abstract
Background
Robot-assisted therapy offers a promising approach to neurorehabilitation, particularly for severely to moderately impaired stroke patients. The objective of this study was to investigate the effects of intensive arm training on motor performance in four chronic stroke patients using the robot ARMin II.
Methods
ARMin II is an exoskeleton robot with six degrees of freedom (DOF) moving shoulder, elbow and wrist joints. Four volunteers with chronic (≥ 12 months post-stroke) left side hemi-paresis and different levels of motor severity were enrolled in the study. They received robot-assisted therapy over a period of eight weeks, three to four therapy sessions per week, each session of one hour.
Patients 1 and 4 had four one-hour training sessions per week and patients 2 and 3 had three one-hour training sessions per week. Primary outcome variable was the Fugl-Meyer Score of the upper extremity Assessment (FMA), secondary outcomes were the Wolf Motor Function Test (WMFT), the Catherine Bergego Scale (CBS), the Maximal Voluntary Torques (MVTs) and a questionnaire about ADL-tasks, progress, changes, motivation etc.
Results
Three out of four patients showed significant improvements (p < 0.05) in the main outcome. The improvements in the FMA scores were aligned with the objective results of MVTs. Most improvements were maintained or even increased from discharge to the six-month follow-up.
Conclusion
Data clearly indicate that intensive arm therapy with the robot ARMin II can significantly improve motor function of the paretic arm in some stroke patients, even those in a chronic state. The findings of the study provide a basis for a subsequent controlled randomized clinical trial.
Background
Stroke remains the leading cause of permanent disability. Recent studies estimate that it affects more than 1 million people in the EU [1,2] and more than 0.7 million in the U.S. each year [3]. The major symptom of stroke is severe sensory and motor hemiparesis of the contralesional side of the body [4]. The degree of recovery highly depends on the severity and the location of the lesion [5]. However, only 18% of stroke survivors regain full motor function after six months [6]. Restoration of arm and hand functions is essential [6] to cope with tasks of daily living and regain independence in life.
There is evidence that the rehabilitation plateau can be prolonged beyond six months post-stroke and that improvements in motor functions can be achieved even in a chronic stage with appropriate therapy [7,8]. For this to occur, effective therapy must comprise key factors containing repetitive, functional, and task-specific exercises performed with high intensity and duration [9-12]. Enhancing patients' motivation, cooperation, and satisfaction can reinforce successful therapy [13]. Robot-assisted training can provide such key elements for inducing long-term brain plasticity and effective recovery [14-19].
Robotic devices can objectively and quantitatively monitor patients' progress - an additional benefit since clinical assessments are often subjective and suffer from reliability issues [20]. Patient-cooperative control algorithms [21,22] can support patients' efforts only as much as needed, thus allowing for intensive robotic intervention.
Several clinical studies have been successfully conducted with endeffector based robots [14,16,17,23]. In these robots, the human arm is connected to the robot at a single (distal) limb only. Consequently, endeffector based robots are easy to use but do not allow single joint torque control over large ranges of motion. In general, they provide less guidance and support than exoskeleton robots [24]. In this study we propose using an exoskeleton-type robot for the intervention. Such a type of robot provides superior guidance and permits individual joint torque control [24]. The device used here is called ARMin and has been developed over the last six years [21,25].
A first pilot study with three chronic stroke patients showed significant improvements in motor functions with intensive training using the first prototype ARMin I. Since ARMin I provided therapy only to the shoulder and elbow, there were no improvements in distal arm functions [25]. Consequently, the goal was to develop a robot, which enables a larger variability of different (also more complex and functional) training modalities involving proximal and distal joint axes [26,27].
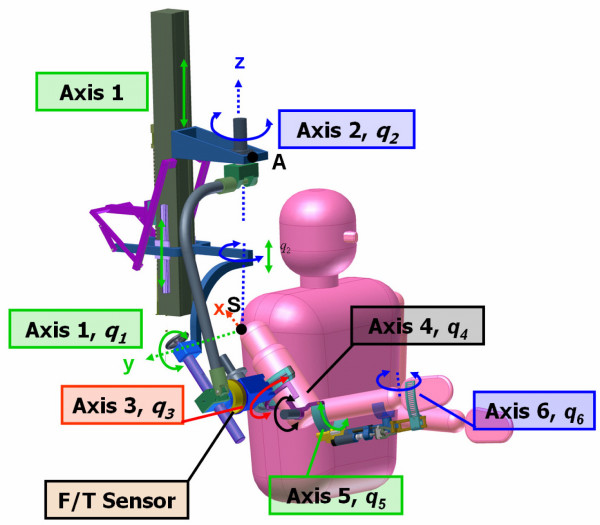
For this study we used an enhanced prototype, ARMin II, with six independently actuated degrees of freedom (DOF) and one coupled DOF (Figure 1). The robot trains both proximal joints (horizontal and vertical shoulder rotation, arm inner - outer rotation, and elbow flexion - extension) and distal joints (pro - supination of lower arm and wrist flexion - extension). Together with an audiovisual display, ARMin II provides a wide variety of training modes with complex exercises and the possibility of performing motivating games.
Figure 1.
Mechanical structure of the exoskeleton robot ARMin II. Axis 1: Vertical shoulder rotation, Axis 2: Horizontal shoulder rotation, Axis 3: Internal/external shoulder rotation, Axis 4: Elbow flexion/extension, Axis 5: Pro/supination of the lower arm, Axis 6: Wrist flexion/extension.
The goal of this study was to investigate the effects of ARMin II training on motor function, strength and use in everyday life.
Methods
Participants
Four patients (three male, one female) met the inclusion criteria and volunteered in the study. The inclusion criteria were i) diagnosis of a single ischemic stroke on the right brain hemisphere with impairment of the left upper extremity and ii) that stroke occurred at least twelve months before study entrance.
Study exclusion criteria were 1) pain in the upper limb, so that the study protocol could not be followed, 2) mental illness or insufficient cognitive or language abilities to understand and follow instructions, 3) cardiac pacemaker, and 4) body weight greater than 120 kg.
All four patients received written and verbal information about the study and gave written informed consent. The protocol of the study was approved by the local ethics committee.
Procedure
To investigate the effects of training with the rehabilitation device ARMin II, four single-case studies with A-B design were applied. Clinical evaluations of the Fugl-Meyer Score of the upper extremity Assessment (FMA), the Wolf Motor Function Test (WMFT), the Catherine Bergego Scale (CBS), and the Maximal Voluntary Torques (MVTs) were administered twice during a baseline period of three weeks (A). A training phase of eight weeks (B) followed. The same evaluation tools were applied every two weeks. Patients 1 and 4 executed three training hours per week (totally 24 hours over entire training period), patients 2 and 3 completed four training hours per week (totally 32 hours). A single training session comprised approximately 15 minutes passive mobilization and approximately 45 minutes active training. Training sessions were always led by the same therapist.
Robotic therapy
ARMin II [21] allows for complex proximal and distal motions in the functional 3-D workspace of the human arm (Figure 1). The patient sits in a wheelchair (wheels locked) and the arm is placed into an orthotic shell, which is fixed and connected by three cuffs to the exoskeletal structure of the robot. Position and force sensors support active and passive control modes. Two types of therapy modes were applied: a passive 'teach and repeat' mobilizing mode and a game mode with active training modalities.
For the passive therapy, the therapist can carry out a patient-specific mobilization sequence adapted to individual needs and deficits, using the robot's 'teach and repeat' mode. The therapist guides the mobilization ('teach') by moving the patient's arm in the orthotic shell. The trajectory of this guided mobilization is recorded by the robot, so that the same mobilization can be repeated several times ('repeat'). The patient receives visual feedback from an avatar on the screen, that performs the same movements in real-time. During the teaching sessions, the robot is controlled by a zero-impedance mode, in which the robot does not add any resistance to the movement, so that the therapist consequently only feels the resistance of the human arm. During the 'repeat' mode, the robot is position-controlled and repeats the motion that has been recorded before.
For the active part of the therapy, a ball game and a labyrinth scenario were selected (see Figure 2). In the ball game, the patient moves a virtual handle on the screen. The aim is to catch a ball that is rolling down a virtual ramp by shifting the handle. When a patient is unable to succeed, the robot provides support by directing the handle to the ball (ARMin II in impedance-control mode). To give the patient visual feedback, the color of the handle turns from green to red when robot-support is delivered. Acoustic feedback is provided when a ball is precisely caught. The difficulty level of the ball game can be modified and adjusted to the patient's need by the therapist, i.e. the number of joint axes involved, the starting arm position, the range of motion, the robotic assistance, resistance or opposing force, and speed.
Figure 2.
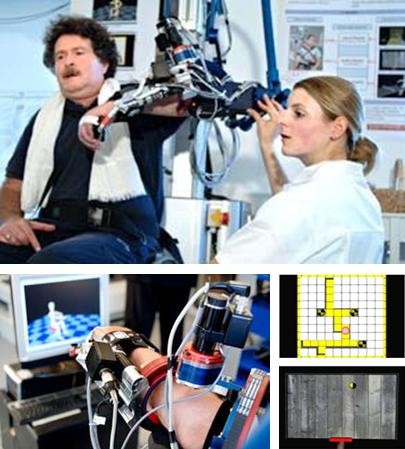
Subject in the robot ARMin II with labyrinth and ball game scenario.
In the labyrinth game, a red ball (cursor) moves according to the patient's arm motions. The objective is to direct the ball from the bottom to the top of the labyrinth. The cursor must be moved accurately. If the ball touches the wall too hard, it drops to the bottom and the game restarts. Like the ball game, the labyrinth provides various training modalities by changing the settings, such as the amount of arm weight compensation, vertical support, number of joint axes involved, working space and sensitivity of the wall [28].
Outcome measurements
To ensure reproducibility and consistency of the testing procedure, all measurements were executed by the same person and with the same settings for each patient. Evaluations were always completed before training sessions. Clinical assessments were filmed and later evaluated by an independent "blinded" therapist from "Charité, Median Clinic Berlin, Department Neurological Rehabilitation".
The main clinical outcome was the Fugl-Meyer Assessment (FMA) of the upper-limb. This impairment-based test consists of 33 items with a total maximum score of 66. The test records the degree of motor deficits and reflexes, the ability to perform isolated movements at each joint and the influence of abnormal synergies on motion [29]. It shows good quality factors (reliability and validity) [30,31] and it is widely used for clinical and research assessments [32].
The Wolf Motor Function Test (WMFT) is a 15-item instrument to quantify disability and to assess performance of simple and complex movements as well as functional tasks [33]. This test has high interrater reliability, internal consistency, and test-retest reliability [34]. The WMFT is responsive to patients with mild to moderate stroke impairments. However, for severely affected patients it has low sensitivity due to a floor effect (when single test items are too difficult).
Severity of neglect was evaluated with the Catherine Bergego Scale (CBS), a test that shows good reliability, validity [35], and sensitivity [36].
To assess sensory functions of the upper limb, the American Spinal Injury Association (ASIA) scoring system was used [37]. The degree of sensation to pinprick (absent = 0, impaired = 1, normal = 2) was determined at the key sensory points of the C4 to T1 dermatomes. The single scores were summed.
In addition, a questionnaire was designed, referring to ADL-tasks, progress, changes, motivation etc. The patients then had to rate the different questions on a scale from 1 to 10, and furthermore, add a comment, expressing their subjective experiences and impressions.
Measurements with ARMin II
With the ARMin II robot, maximal voluntary torques (MVTs) were determined for six isometric joint actions including vertical shoulder flexion and extension, horizontal shoulder abduction and adduction, as well as elbow flexion and extension. Patients were seated in a locked wheelchair with the upper body fixed by three belts (two crosswise diagonal torso belts and one belt over the waist) to prevent the torso from assisting the movements. The starting position was always the same. The shoulder was flexed 70° and transversally abducted 20°, the rotation of the upper and lower arm was neutral (0°), and the elbow was flexed 90°. Patients were instructed to generate maximal isometric muscle contractions against the resistance of ARMin II for at least two seconds before relaxing. During the effort, verbal encouragement was given in each case.
Data analysis
From the main baseline measurements - FMA, WMFT, CBS, and MVT - the mean values and standard deviations were calculated. Data recorded during the intervention phases were evaluated by using the least square linear regression model with applied bootstrap resampling technique [38]. For the statistical analysis, the programs SYSTAT 12 and Matlab 6.1 were used. The significance level p ≤ 0.05 of the slope of the regression line was considered to indicate a statistically significant improvement.
Results
The results of the FMA are presented in Table 1. From baseline to discharge, patients 1, 2, and 3 increased their scores significantly (p < 0.05). They continued to improve in the FMA at the six-month follow-up (see Figure 3).
Table 1.
Overview of the Fugl-Meyer Assessment
| FMA: Total§ sh/e§ w/h§ |
Baseline | Post- therapy |
Difference† | Follow up (6 mt) |
Difference‡ | Total change |
R2 | p |
|---|---|---|---|---|---|---|---|---|
| S1: Total | 21 | 38.6 | +17.6 | 50 | +11.4 | +29 | 0.943 | 0.001* |
| sh/e | 20 | 24.0 | +4.0 | 28 | +4.0 | +8 | ||
| w/h | 1 | 14.6 | +13.6 | 22 | +7.4 | +21 | ||
| S2: Total | 24 | 27.1 | +3.1 | 29 | +1.9 | +5 | 0.800 | 0.041* |
| sh/e | 21 | 23.1 | +2.1 | 24 | +0.9 | +3 | ||
| w/h | 3 | 4.0 | +1.0 | 5 | +1.0 | +2 | ||
| S3: Total | 11 | 17.8 | +6.8 | 19 | +1.2 | +8 | 0.908 | 0.003* |
| sh/e | 10 | 15.8 | + 5.8 |
18 | +2.2 | +8 | ||
| w/h | 1 | 2.0 | +1.0 | 1 | -1.0 | +0 | ||
| S4: Total | 10 | 12.1 | +2.1 | 13 | +0.9 | +3 | 0.408 | 0.172 |
| sh/e | 10 | 12.1 | +2.1 | 12 | -0.1 | +2 | ||
| w/h | 0 | 0 | +0 | 1 | +1.0 | +1 |
Note: An increase in score indicates improvement. S1 - S4 means subject 1 to 4.
§Fugl-Meyer (FMA), total score, maximum = 66; score for shoulder/elbow (sh/e), max. = 36; score for wrist/hand (w/h), max. = 30
†Difference of score between baseline and post-test.
‡Difference of score between post-test and follow up.
*Indicate significant p-values < 0.05
Figure 3.
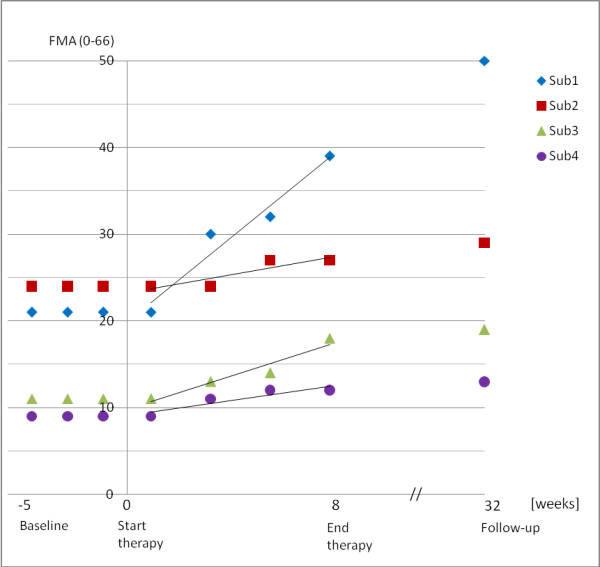
Clinical FMA scores across evaluation sessions.
Patient 1 gained +17.6 points in the FMA (from 21 to 38.6 points), while at the follow-up, six months later, he demonstrated even further impressive progress, without having received additional therapy in the mean time. Overall, patient 1 showed an absolute improvement of +29 points (from 21 to 50 points), particularly due to high recovery in distal arm functions (+21 points).
The FMA gains of patients 2 and 3 were +5 points (from 24 to 29 points) and +8 points (from 11 to 19 points). These findings were in line with other investigations about the effects of robot-assisted therapy in chronic stroke patients that demonstrated changes between 3.2 and 6.8 points [14,23,39-43]. However, one must note that such comparisons have to be done with care since studies often differ in methods and criteria (e.g. intervention time, number of training sessions per week, duration of training sessions, type of stroke, affected brain side, time post-stroke, and severity of lesion). Patient 4 showed an increase of +3 points (from 10 to 13 points) in the FMA; however, this increase was statistically not significant.
Typical arm functions that are relevant for activities of daily life can be expressed by the WMFT (Table 2). During the therapy, the WMFT scores of patients 1, 2 and 3 increased by +1.00, +0.5, and +0.86 points, respectively. Patients 2 and 3 slightly diminished at follow-up. Nevertheless, these three patients achieved significant progress (p < 0.05), in contrast to patient 4, who showed no significant improvement. However, at the follow-up examination, patient 4 was the only one who further improved in the WMFT (see Figure 4).
Table 2.
Overview of the Wolf Motor Function Test
| WMFT§ | Baseline | Post- therapy |
Difference† | Follow up (6 mt) |
Difference‡ | Total change |
R2 | p |
|---|---|---|---|---|---|---|---|---|
| S1: | 1.86 | 2.86 | +1.00 | 2.86 | 0 | +1.00 | 0.911 | 0.003* |
| S2: | 2.07 | 2.57 | +0.50 | 2.50 | -0.07 | +0.43 | 0.891 | 0.005* |
| S3: | 1.07 | 1.93 | +0.86 | 1.79 | -0.14 | +0.72 | 0.831 | 0.011* |
| S4: | 0.93 | 1.07 | +0.14 | 1.29 | +0.22 | +0.36 | 0.577 | 0.080 |
Note: An increase in score indicates functional improvement. S1 - S4 means subject 1 to 4.
§Wolf Motor Function Test (WMFT), 5 = normal motor functions.
*Indicate significant p-values < 0.05
Figure 4.
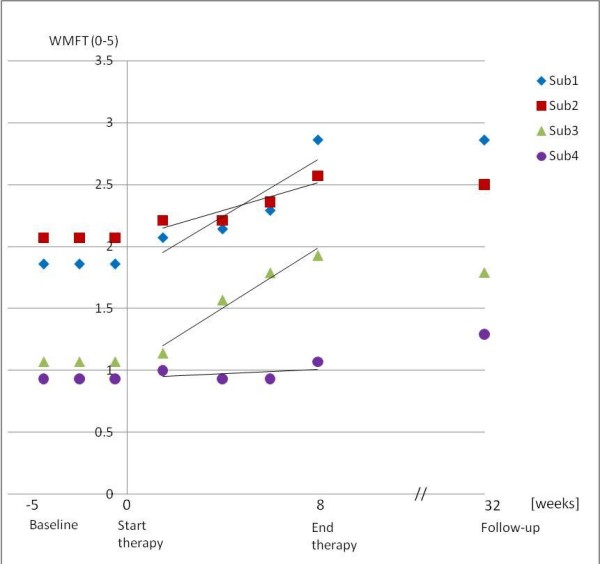
Clinical WMFT scores across evaluation sessions.
A questionnaire was used to obtain further information about patient status. The patients reported progress of the affected upper extremity in everyday life activities (e.g. the arm can be lifted higher and better, is more integrated, feels lighter and is less stiff, able to lift glass, fold laundry, use index finger, and control motions better). The grades of patients 1 to 4 regarding the use of their impaired arm during ADLs after the intervention (scale range 1 to 10, no better use = 1, much better use = 10) were 5, 7, 4 and 3, respectively. Furthermore, they described to be more motivated and willing to try to engage their arm in diverse daily activities.
An overview of the MVTs, consisting of six different torque measurements, is presented in Table 3. At the follow-up, improvement in muscle strength increased in patient 1, while it slightly diminished in patients 2 and 3. In patient 4, muscle strength returned to the base level at the follow-up.
Table 3.
Overview of Total Maximal Voluntary Torques (MVTs)
| MVT§ (Nm) | Baseline | Post- therapy |
Difference† | Follow up (6 mt) |
Difference‡ | Total change |
|---|---|---|---|---|---|---|
| S1: | 17.6 ± 7.5 | 31.6 ± 9.0 | +14.0 | 36.4 ± 10.9 | +4.8 | +18.8 |
| S2: | 6.1 ± 5.2 | 14.4 ± 3.4 | +8.3 | 12.9 ± 5.2 | -1.5 | +6.8 |
| S3: | 9.6 ± 6.7 | 22.4 ± 10.7 | +12.7 | 19.2 ± 13.3 | -3.2 | +9.6 |
| S4: | 2.6 ± 2.0 | 8.3 ± 3.4 | +5.7 | 3.4 ± 2.9 | -4.9 | +0.8 |
Note: An increase in Nm indicates improvement in muscle torque. S1 - S4 means subject 1 to 4.
§Total of maximal voluntary torques (Nm): Consists of 6 different torque measurements (shoulder flexion - extension; horizontal shoulder rotation; elbow flexion - extension).
†Difference of Nm between baseline and post-test.
‡Difference of Nm between post-test and follow up.
The demographic data and clinical characteristics of the four patients are summarized in Table 4. None of the patients reported any adverse effects from robot-mediated therapy. In contrast, patients 3 and 4 described reduced hardening and pain of their neck and shoulder muscles.
Table 4.
Data on the Subjects at Admission
| S1 | S2 | S3 | S4 | |
|---|---|---|---|---|
| Gender | Male | female | male | male |
| Age | 39 | 60 | 54 | 58 |
| Handedness (before stroke) | Right | right | right | right |
| Hemisphere of unilateral stroke | Right | right | right | right |
| Diagnosis of stroke | ischemic media insult right, bleeding into Ncl. Lentiformis |
ischemic media insult right, in the temporal dorsal brain |
ischemic insult in the right PCA* |
ischemic media insult right |
| Months post-stroke (at entrance) | 12 | 131 | 22 | 16 |
| Reflex Status (0/+/++/+++) | ++ | +++ | +++ | +++ |
| Sensation, pin prick, C4-T1 (0-24) | 20 | 22 | 24 | 7 |
| F. Independence Measure (18-126) | 103 | 121 | 112 | 90 |
| Fugl-Meyer Assessment UL (0-66) | 21 | 24 | 11 | 9 |
| Wolf Motor Function Test (0-5) | 1.80 | 2.07 | 1.01 | 0.35 |
| Catherine Bergego Scale (0-30) | 5 | 4 | 12 | 16 |
| Modified Asworth Scale (0-5) | ||||
| Elbow | 0 | 3 | 3 | 3 |
| Wrist | 0 | 3 | 3 | 3 |
| Finger | 2 | 3 | 3 | 3 |
Note: Tests refer to the impaired body side. Functional Independence Measure: 18 = being completely dependent, 126 = acting completely independent. Wolf Motor Function Test: 0 = no motor functions, 75 = normal motor functions. Fugl-Meyer Assessment: 0 = no motor functions, 66 = normal motor functions. Catherine Bergego Scale: Neglect: 0 = normal, 30 = severe neglect. Modified Asworth: 0 =no spasticity, 5 = severe spasticity. Reflex Status: 0 = no reflex, '+' = moderate reflex,
'++' = normal reflex, '+++' = vigorous reflex. Sensation, pin prick C4 to T1: absent = 0, impaired = 1, normal = 2, max 24.
Patients 1, 2 and 3 completed measurements and therapy sessions, except for patient 4, who missed one measurement date and two therapy sessions for reasons that are not related to the study.
Discussion
In this study, intensive therapy using the robot ARMin II was administered to four chronic stroke patients during eight weeks of training. Patients 1 and 4 received 32 and 30 hours of therapy respectively, while patients 2 and 3 received 24 hours.
The results of these single-case series underline prior findings with robotic therapy, namely that intensive, repetitive, task-specific, and goal-directed training can significantly improve motor functions in chronic stroke patients - even years post-stroke [14,23,44]. All four patients demonstrated improvements in motor and functional activities, but to various degrees. Overall, they sustained their functional gains at the six-month follow-up or even continued to improve after the end of treatment, indicating potential long-term benefits of robot-assisted therapy.
Patient 4 had the lowest motor functions at study entrance and hardly any sensation in the clinical pinprick test. Such neurological deficits can make functional therapy very difficult as feedback functions are not, or hardly, available. This might explain why patient 4 could only profit little from the training with ARMin II. For stroke individuals with little sensory functions, as e.g. patient 4, a sensory intervention is suggested to be a more effective approach [45]. In general, it can be said that stroke patients with severe sensory loss benefited less from treatment than moderately impaired patients [10,46].
The gains in the WMFT likely reflect increased motor performance levels that are suggested to facilitate use of the impaired upper limb in daily activities. These changes seemed to be clinically significant from the patients' perspective. However, the analysis suggested that the impaired upper limb was mainly involved as an assist in bimanual ADLs after intervention. In addition, one must note that not only gains in motor abilities were achieved, but also positive impacts on concentration, neglect, physical capacity, well-being, body balance and posture were noticed. Patient 4, for example, diminished twelve points in the CBS, indicating a reduced neglect (Table 5).
Table 5.
Overview of Catherine Bergego Scale
| Catherine Bergego Scale: Neglect | ||||
|---|---|---|---|---|
| Baseline | Post-therapy | 6-mo Follow up | Change | |
| S1: | 5 | 3 | 4 | -1 |
| S2: | 4 | 3 | 2 | -2 |
| S3: | 12 | 2 | 3 | -9 |
| S4: | 16 | 11 | 4 | -12 |
Note: Lower score of CBS means a reduction of neglect (0 = normal, 30 = severe neglect).
The different responses of this pilot research could be explained by patients' heterogeneity, as patients differed in terms of age, time post-stroke, affected brain areas, sensation, muscle tone, etc. (Table 4) - all factors that influence motor relearning. The highest motor recoveries were experienced by patient 1, the youngest and least chronic patient. But note that patient 1 like patient 4 received more intensive intervention than the other two patients. It seems that treatment, with additional therapy hours, is primarily fruitful and beneficial for patients with a certain level of remaining sensory functions and motor abilities.
In a comparable study that has been conducted with the robot ARMin I, patients I and II received 24 hours of therapy, while patient III received 40 hours. The improvement of patients I, II and III were +3.1, +3.0, and +4.2 points (initial FMA scores were 14, 26 and 15).
The reason for the less distinctive improvements with the former version ARMin I and other previous robots might be due to the limited movement capabilities (only proximal [14,47,48] or distal [49] arm involvement) or the missing control of proximal joints (endeffector-based robots). In contrast, ARMin II allows for authentic motion sequences, including coordinated interactions between wrist, elbow and shoulder joints. This seems to be an important feature since most everyday activities are composed of inter-joint coordination. ARMin II is an exoskeleton-based robot and, in general, better suited to train ADL-tasks than an endeffector-based robot. This is because, in an exoskeleton robot, the human arm is very well supported and guided by the robot, movements with large ROM can be trained, and the interaction torques that the robot apply to each joint of the human arm can be controlled individually.
Complex movements also enable patients to break abnormal synergy patterns that are limiting arm motor functions [50-52]. As ARMin II provides support against gravity, abnormal synergy patterns in hemiparetic limbs can progressively be learned to be overcome, a matter that was observed in patients 1, 2 and 3. For this therapy issue, the labyrinth scenario seemed to be particularly suitable, as the parameters can be highly varied and adapted individually to the patient's needs. Similar to these findings, Sukal et al. [53] have shown in their research that a reduced range of motion in stroke patients was a result of pathologic synergies during arm lifting. By de-weighting and supporting the impaired arm in training sessions, the effect of gravity could be overcome.
Another approach by Ellis et al. [52] demonstrated that abnormal joint torque coupling could be modified by 'multi-DOF progressive resistance training' in severely impaired chronic stroke individuals. Patients gained strength and simultaneously improved in multi-DOF joint torque combinations. The same relationship could also be observed in the patients that participated in this pilot study. An increase in muscle strength was associated with a larger active range of motion as well as improved muscle coordination. Patients dissociated from synergistic co-activation in the FMA and WMFT.
A distinct finding of the ARMin II study was that post-treatment further progress was achieved in the FMA. These continuing improvements might be due to therapy of distal arm functions since neither the ARMin I study showed such additional effects at follow-up nor any other proximal robotic study in literature that the authors are aware of. Krebs et al. [41] found that training of the more distal limb segments led to twice as much carryover effect to the proximal segments than vice-versa. Moreover, they observed that improvement in more distal segments continued significantly even without further training for that particular limb segment. This finding supports our assumption that the patients were better able to use their arm in daily activities after robotic treatment, allowing them to further improve at the six-month follow up.
With eight weeks of robot training, patient 1 enhanced his performance to such an extent (from initially 21 points to 50 points in the follow-up in the FMA) that he reached a higher functional state - opening up new therapy approaches like constrained induced movement therapy CIMT.
In the present study the two game scenarios (labyrinth and ball game), were particularly suitable to create an enjoyable, efficient and motivating intervention. Patients' interests could be incorporated into therapy by choosing different game settings and levels with miscellaneous arm positions and various joint axes. Nevertheless, complementary therapy modes focusing on specific ADL-tasks and/or virtual reality scenarios might additionally help to facilitate a transfer to ADLs [26,27]. An additional hand module for opening and closing hand function and/or single finger functions would enable more specific and individualized therapy of hand and fingers, allowing for the implementation of more authentic ADL-tasks.
So far, treatment with robotic devices [17,19,54] shows no consistent improvement in functional abilities of daily activities. Although high functional improvements and a transfer to ADLs were achieved in this investigation, these findings are limited to single cases. The pilot study included only four, rather heterogeneous chronic stroke patients. Despite the fact that functional stability could be verified in all patients at baseline, no separate control group was used. All patients continued with their conventional outpatient therapies (maximum 1 hour of physical and occupational therapy per week, focusing on the gait and posture only). However, the patients were encouraged to continue their standard therapies on a constant level, so that possible improvements due to this small amount of conventional therapy can be excluded from the study. Overall, these encouraging results definitely justify starting a large randomized clinical trial.
Conclusion
This paper presents preliminary results of a pilot study with four heterogeneous, chronic stroke patients using the robotic device ARMin II. The findings support the assumption that robot therapy can significantly influence therapy outcomes. A robot that includes proximal and distal joints, such as ARMin II, allows for a wide field of specific training modalities with natural and complex motions.
In this study it was noted that a subgroup of patients could achieve a transfer to ADLs after performing the training phase. This finding is of great importance since treatment with robotic devices to date did not result in consistent improvement in activities of daily living. A transfer to everyday life should be indeed a central intention of rehabilitation as it brings independence and improve quality of life.
Competing interests
Tobias Nef and Robert Riener are inventors of patents describing the ARMin invention (WO2006058442, EP1827349, EP07020795). The owners of the patents are the ETH Zurich and the University of Zurich.
Authors' contributions
PS developed the study design, performed subject recruitment, data acquisition and statistical analysis. She was the primary composer of this manuscript. TN provided feedback and expert guidance throughout this study and was also involved in data analysis. TN and RR designed and built the robotic device ARMin II used in this work. All four authors contributed significantly to the intellectual content of the manuscript and have approved the final version to be published.
Contributor Information
Patricia Staubli, Email: patricia.staubli@alumni.ethz.ch.
Tobias Nef, Email: nef@cua.edu.
Verena Klamroth-Marganska, Email: verena.klamroth@mavt.ethz.ch.
Robert Riener, Email: riener@mavt.ethz.ch.
Acknowledgements
This project was supported by the National Centre of Competence in Research, Neural Plasticity and Repair (subprojects 7 and 8). A special thank you goes to Oliver Maric, MD, for his accomplishment of the physical examinations of all patients. Furthermore, we want to express our gratitude to Stefanie van Kaick, MSc OT, and Cordula Werner, MSc OT, for their blinded ratings of the clinical assessments. Special thanks also go to Volker Dietz, MD and Claudia Rudhe-Link, MSc OT for their assistance as scientific advisors. Finally, we would like to thank all patients who kindly participated in this time consuming study.
References
- Thorvaldsen P, Asplund K, Kuulasmaa K, Rajakangas AM, Schroll M. Stroke incidence, case fatality, and mortality in the WHO MONICA project. World Health Organization Monitoring Trends and Determinants in Cardiovascular Disease. Stroke. 1995;26:361–367. doi: 10.1161/01.str.26.3.361. [DOI] [PubMed] [Google Scholar]
- Brainin M, Bornstein N, Boysen G, Demarin V. Acute neurological stroke care in Europe: results of the European Stroke Care Inventory. Eur J Neurol. 2000;7:5–10. doi: 10.1046/j.1468-1331.2000.007001005.x. [DOI] [PubMed] [Google Scholar]
- Rosamond W, Flegal K, Friday G, Furie K, Go A, Greenlund K, Haase N, Ho M, Howard V, Kissela B. Heart disease and stroke statistics--2007 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2007;115:e69–171. doi: 10.1161/CIRCULATIONAHA.106.179918. [DOI] [PubMed] [Google Scholar]
- Maeurer HC, Diener HC. Der Schlaganfall. Stuttgart: Georg Thieme Verlag; 1996. [Google Scholar]
- Rossini PM, Calautti C, Pauri F, Baron JC. Post-stroke plastic reorganisation in the adult brain. Lancet Neurol. 2003;2:493–502. doi: 10.1016/S1474-4422(03)00485-X. [DOI] [PubMed] [Google Scholar]
- Nakayama H, Jorgensen HS, Raaschou HO, Olsen TS. Recovery of upper extremity function in stroke patients: the Copenhagen Stroke Study. Arch Phys Med Rehabil. 1994;75:394–398. doi: 10.1016/0003-9993(94)90161-9. [DOI] [PubMed] [Google Scholar]
- Carey JR, Kimberley TJ, Lewis SM, Auerbach EJ, Dorsey L, Rundquist P, Ugurbil K. Analysis of fMRI and finger tracking training in subjects with chronic stroke. Brain. 2002;125:773–788. doi: 10.1093/brain/awf091. [DOI] [PubMed] [Google Scholar]
- Lee J van der, Wagenaar R, Lankhorst G, Vogelaar T, Deville W, Bouter L. Forced Use of the Upper Extremity in Chronic Stroke Patients: Results From a Single-Blind Randomized Clinical Trial. Stroke. 1999;30:2369–2375. doi: 10.1161/01.str.30.11.2369. [DOI] [PubMed] [Google Scholar]
- Kwakkel G, Wagenaar RC, Koelman TW, Lankhorst GJ, Koetsier JC. Effects of intensity of rehabilitation after stroke. A research synthesis. Stroke. 1997;28:1550–1556. doi: 10.1161/01.str.28.8.1550. [DOI] [PubMed] [Google Scholar]
- Sunderland A, Tinson DJ, Bradley EL, Fletcher D, Langton Hewer R, Wade DT. Enhanced physical therapy improves recovery of arm function after stroke. A randomised controlled trial. J Neurol Neurosurg Psychiatry. 1992;55:530–535. doi: 10.1136/jnnp.55.7.530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butefisch C, Hummelsheim H, Denzler P, Mauritz KH. Repetitive training of isolated movements improves the outcome of motor rehabilitation of the centrally paretic hand. J Neurol Sci. 1995;130:59–68. doi: 10.1016/0022-510X(95)00003-K. [DOI] [PubMed] [Google Scholar]
- Kwakkel G, van Peppen R, Wagenaar RC, Wood Dauphinee S, Richards C, Ashburn A, Miller K, Lincoln N, Partridge C, Wellwood I, Langhorne P. Effects of augmented exercise therapy time after stroke: a meta-analysis. Stroke. 2004;35:2529–2539. doi: 10.1161/01.STR.0000143153.76460.7d. [DOI] [PubMed] [Google Scholar]
- Maclean N, Pound P, Wolfe C, Rudd A. Qualitative analysis of stroke patients' motivation for rehabilitation. BMJ. 2000;321:1051–1054. doi: 10.1136/bmj.321.7268.1051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lum PS, Burgar CG, Shor PC, Majmundar M, Loos M Van der. Robot-assisted movement training compared with conventional therapy techniques for the rehabilitation of upper-limb motor function after stroke. Arch Phys Med Rehabil. 2002;83:952–959. doi: 10.1053/apmr.2001.33101. [DOI] [PubMed] [Google Scholar]
- Reinkensmeyer DJ, Emken JL, Cramer SC. Robotics, motor learning, and neurologic recovery. Annu Rev Biomed Eng. 2004;6:497–525. doi: 10.1146/annurev.bioeng.6.040803.140223. [DOI] [PubMed] [Google Scholar]
- Riener R, Nef T, Colombo G. Robot-aided neurorehabilitation of the upper extremities. Med Biol Eng Comput. 2005;43:2–10. doi: 10.1007/BF02345116. [DOI] [PubMed] [Google Scholar]
- Prange GB, Jannink MJ, Groothuis-Oudshoorn CG, Hermens HJ, Ijzerman MJ. Systematic review of the effect of robot-aided therapy on recovery of the hemiparetic arm after stroke. J Rehabil Res Dev. 2006;43:171–184. doi: 10.1682/JRRD.2005.04.0076. [DOI] [PubMed] [Google Scholar]
- Nelles G. Cortical reorganization--effects of intensive therapy. Restor Neurol Neurosci. 2004;22:239–244. [PubMed] [Google Scholar]
- Kwakkel G, Kollen BJ, Krebs HI. Effects of robot-assisted therapy on upper limb recovery after stroke: a systematic review. Neurorehabil Neural Repair. 2008;22:111–121. doi: 10.1177/1545968307305457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wade DT. Evaluating outcome in stroke rehabilitation (quality control and clinical audit) Scand J Rehabil Med Suppl. 1992;26:97–104. [PubMed] [Google Scholar]
- Nef T, Mihelj M, Riener R. ARMin: a robot for patient-cooperative arm therapy. Med Biol Eng Comput. 2007;45:887–900. doi: 10.1007/s11517-007-0226-6. [DOI] [PubMed] [Google Scholar]
- Mihelj M, Nef T, Riener R. A novel paradigm for patient-cooperative control of upper limb rehabilitation robots. Advanced Robotics. 2007;21:843–867. doi: 10.1163/156855307780851975. [DOI] [Google Scholar]
- Fasoli SE, Krebs HI, Stein J, Frontera WR, Hughes R, Hogan N. Robotic therapy for chronic motor impairments after stroke: Follow-up results. Arch Phys Med Rehabil. 2004;85:1106–1111. doi: 10.1016/j.apmr.2003.11.028. [DOI] [PubMed] [Google Scholar]
- Nef T, Guidali M, Riener R. ARMin III - Arm therapy exoskeleton with an ergonomic shoulder actuation. Applied Bionics and Biomechanics. 2009;6:127–142. doi: 10.1080/11762320902840179. [DOI] [Google Scholar]
- Nef T, Quinter G, Muller R, Riener R. Effects of Arm Training with the Robotic Device ARMin I in Chronic Stroke: Three Single Cases. Neurodegener Dis. 2009. in press . [DOI] [PubMed]
- Bayona NA, Bitensky J, Salter K, Teasell R. The role of task-specific training in rehabilitation therapies. Top Stroke Rehabil. 2005;12:58–65. doi: 10.1310/BQM5-6YGB-MVJ5-WVCR. [DOI] [PubMed] [Google Scholar]
- Carey JR, Bhatt E, Nagpal A. Neuroplasticity promoted by task complexity. Exerc Sport Sci Rev. 2005;33:24–31. [PubMed] [Google Scholar]
- Carr JH, Shepherd RB. Movement science: foundations for physical therapy in rehabilitation. Rockville (MD): Aspen; 1987. A motor learning model for rehabilitation; pp. 31–91. [Google Scholar]
- Fugl-Meyer AR, Jaasko L, Leyman I, Olsson S, Steglind S. The post-stroke hemiplegic patient. 1. a method for evaluation of physical performance. Scand J Rehabil Med. 1975;7:13–31. [PubMed] [Google Scholar]
- Duncan PW, Propst M, Nelson SG. Reliability of the Fugl-Meyer assessment of sensorimotor recovery following cerebrovascular accident. Phys Ther. 1983;63:1606–1610. doi: 10.1093/ptj/63.10.1606. [DOI] [PubMed] [Google Scholar]
- Gladstone DJ, Danells CJ, Black SE. The fugl-meyer assessment of motor recovery after stroke: a critical review of its measurement properties. Neurorehabil Neural Repair. 2002;16:232–240. doi: 10.1177/154596802401105171. [DOI] [PubMed] [Google Scholar]
- Amirabdollahian F, Loureiro R, Gradwell E, Collin C, Harwin W, Johnson G. Multivariate analysis of the Fugl-Meyer outcome measures assessing the effectiveness of GENTLE/S robot-mediated stroke therapy. J Neuroengineering Rehabil. 2007;4:4. doi: 10.1186/1743-0003-4-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf SL, Catlin PA, Ellis M, Archer AL, Morgan B, Piacentino A. Assessing Wolf motor function test as outcome measure for research in patients after stroke. Stroke. 2001;32:1635–1639. doi: 10.1161/01.str.32.7.1635. [DOI] [PubMed] [Google Scholar]
- Morris DM, Uswatte G, Crago JE, Cook EW, Taub E. The reliability of the wolf motor function test for assessing upper extremity function after stroke. Arch Phys Med Rehabil. 2001;82:750–755. doi: 10.1053/apmr.2001.23183. [DOI] [PubMed] [Google Scholar]
- Azouvi P, Olivier S, de Montety G, Samuel C, Louis-Dreyfus A, Tesio L. Behavioral assessment of unilateral neglect: study of the psychometric properties of the Catherine Bergego Scale. Arch Phys Med Rehabil. 2003;84:51–57. doi: 10.1053/apmr.2003.50062. [DOI] [PubMed] [Google Scholar]
- Halligan PW, Marshall JC, Wade DT. Visuospatial neglect: underlying factors and test sensitivity. Lancet. 1989;2:908–911. doi: 10.1016/S0140-6736(89)91561-4. [DOI] [PubMed] [Google Scholar]
- Ditunno JF Jr, Young W, Donovan WH, Creasey G. The international standards booklet for neurological and functional classification of spinal cord injury. American Spinal Injury Association. Paraplegia. 1994;32:70–80. doi: 10.1038/sc.1994.13. [DOI] [PubMed] [Google Scholar]
- Parker RI. Increased reliability for single-case research results: is the bootstrap the answer? Behav Ther. 2006;37:326–338. doi: 10.1016/j.beth.2006.01.007. [DOI] [PubMed] [Google Scholar]
- Volpe BT, Lynch D, Rykman-Berland A, Ferraro M, Galgano M, Hogan N, Krebs HI. Intensive Sensorimotor Arm Training Mediated by Therapist or Robot Improves Hemiparesis in Patients With Chronic Stroke. Neurorehabil Neural Repair. 2008;22:305–310. doi: 10.1177/1545968307311102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krebs HI, Mernoff S, Fasoli SE, Hughes R, Stein J, Hogan N. A comparison of functional and impairment-based robotic training in severe to moderate chronic stroke: a pilot study. NeuroRehabilitation. 2008;23:81–87. [PMC free article] [PubMed] [Google Scholar]
- Krebs HI, Volpe BT, Williams D, Celestino J, Charles SK, Lynch D, Hogan N. Robot-aided neurorehabilitation: a robot for wrist rehabilitation. IEEE Trans Neural Syst Rehabil Eng. 2007;15:327–335. doi: 10.1109/TNSRE.2007.903899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coote S, Murphy B, Harwin W, Stokes E. The effect of the GENTLE/s robot-mediated therapy system on arm function after stroke. Clin Rehabil. 2008;22:395–405. doi: 10.1177/0269215507085060. [DOI] [PubMed] [Google Scholar]
- McCombe Waller S, Whitall J. Hand dominance and side of stroke affect rehabilitation in chronic stroke. Clin Rehabil. 2005;19:544–551. doi: 10.1191/0269215505cr829oa. [DOI] [PubMed] [Google Scholar]
- Liepert J, Bauder H, Wolfgang HR, Miltner WH, Taub E, Weiller C. Treatment-induced cortical reorganization after stroke in humans. Stroke. 2000;31:1210–1216. doi: 10.1161/01.str.31.6.1210. [DOI] [PubMed] [Google Scholar]
- Sullivan JE, Hedman LD. Sensory dysfunction following stroke: incidence, significance, examination, and intervention. Top Stroke Rehabil. 2008;15:200–217. doi: 10.1310/tsr1503-200. [DOI] [PubMed] [Google Scholar]
- Cirstea MC, Ptito A, Levin MF. Arm reaching improvements with short-term practice depend on the severity of the motor deficit in stroke. Exp Brain Res. 2003;152:476–488. doi: 10.1007/s00221-003-1568-4. [DOI] [PubMed] [Google Scholar]
- Krebs HI, Volpe BT, Aisen ML, Hogan N. Increasing productivity and quality of care: robot-aided neuro-rehabilitation. J Rehabil Res Dev. 2000;37:639–652. [PubMed] [Google Scholar]
- Reinkensmeyer DJ, Kahn LE, Averbuch M, McKenna-Cole A, Schmit BD, Rymer WZ. Understanding and treating arm movement impairment after chronic brain injury: progress with the ARM guide. J Rehabil Res Dev. 2000;37:653–662. [PubMed] [Google Scholar]
- Hesse S, Schulte-Tigges G, Konrad M, Bardeleben A, Werner C. Robot-assisted arm trainer for the passive and active practice of bilateral forearm and wrist movements in hemiparetic subjects. Arch Phys Med Rehabil. 2003;84:915–920. doi: 10.1016/S0003-9993(02)04954-7. [DOI] [PubMed] [Google Scholar]
- Beer RF, Dewald JP, Rymer WZ. Deficits in the coordination of multijoint arm movements in patients with hemiparesis: evidence for disturbed control of limb dynamics. Exp Brain Res. 2000;131:305–319. doi: 10.1007/s002219900275. [DOI] [PubMed] [Google Scholar]
- Dewald JP, Beer RF. Abnormal joint torque patterns in the paretic upper limb of subjects with hemiparesis. Muscle Nerve. 2001;24:273–283. doi: 10.1002/1097-4598(200102)24:2<273::AID-MUS130>3.0.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- Ellis MD, Holubar BG, Acosta AM, Beer RF, Dewald JP. Modifiability of abnormal isometric elbow and shoulder joint torque coupling after stroke. Muscle Nerve. 2005;32:170–178. doi: 10.1002/mus.20343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sukal TM, Ellis MD, Dewald JP. Source of work area reduction following hemiparetic stroke and preliminary intervention using the ACT3D system. Conf Proc IEEE Eng Med Biol Soc. 2006;1:177–180. doi: 10.1109/IEMBS.2006.259311. full_text. [DOI] [PubMed] [Google Scholar]
- Mehrholz J, Platz T, Kugler J, Pohl M. Electromechanical and Robot-Assisted Arm Training for Improving Arm Function and Activities of Daily Living After Stroke. Stroke. 2009;40:392–393. doi: 10.1161/STROKEAHA.108.536219. [DOI] [PubMed] [Google Scholar]