Abstract
Background
Steroid hormones can influence neuronal excitability and subsequent seizure susceptibility through genomic and nongenomic mechanisms. For example, there are proconvulsant steroids such as estradiol and corticosterone and anticonvulsant steroids such as testosterone, progesterone, and their GABAergic metabolites. Recent findings indicated that a single, acute administration of ethanol increased levels of GABAergic steroids and that the source of this increase was peripheral organs such as the adrenals and gonads. Thus, the purpose of the present study was to determine the impact of removal of the adrenals and/or gonads on withdrawal severity following a single high dose of ethanol in 2 genotypes that differ in ethanol withdrawal severity.
Method
Male and female C57BL/6J (B6) and DBA/2J (D2) mice were either left intact (SHAM), adrenalectomized (ADX), gonadectomized (GDX), or underwent ADX/GDX surgery. Seven days following surgery, baseline handling-induced convulsions (HICs) were measured prior to administration of a 4 g/kg dose of ethanol. HICs were assessed following the ethanol injection, then hourly for 12 hours and at 24 hours. A separate group of mice were used to measure the impact of surgical status on ethanol metabolism at 30, 60, 120, and 240 minutes after a single 4 g/kg dose of ethanol.
Results
ADX and ADX/GDX treatments in male B6 and D2 mice increased ethanol withdrawal severity following a single dose of ethanol, measured by area under the withdrawal curve and peak HIC scores. Acute ethanol withdrawal also was increased in female D2 mice that had undergone ADX/GDX. In contrast, surgical status did not alter ethanol withdrawal severity in female B6 mice. Surgical status had only minor effects on ethanol metabolism.
Conclusions
Removal of peripherally derived steroids with anticonvulsant properties significantly increased HIC scores during acute ethanol withdrawal following a single dose of ethanol in male and female D2 mice and in male B6 mice. These increases were not due to changes in ethanol metabolism.
Keywords: Alcohol, GABAA Receptors, Convulsions, Neuroactive Steroids, Steroid Hormones
For over half A decade, it has been known that sex steroids can influence neuronal excitability (Seyle, 1942). More specifically, it has been shown that the hormones progesterone, deoxycorticosterone, and testosterone and some of their metabolites are protective against several types of seizures (Belelli et al., 1989; Reddy, 2004b), whereas estrogens and corticosterone are proconvulsant (Reddy, 2004b; Roberts et al., 1992). While steroids are traditionally thought of as affecting gene transcription through nuclear binding to hormone response elements, the rapid steroid effect on neuronal excitability has been shown to be mediated through interactions with membrane bound receptors (Purdy et al., 1992).
The metabolism of deoxycorticosterone, testosterone, and progesterone with 5α-reductase and 3α-hydroxysteroid dehydrogenase yields steroids that have actions at membrane receptors and that have been termed neuroactive steroids (Paul and Purdy, 1992; Rupprecht and Holsboer, 1999). The 3 major sites of neurosteroid production are the gonads (Mellon and Griffin, 2002b), the adrenals (Holzbauer et al., 1985), and the brain (Baulieu, 1998; Mellon and Griffin, 2002a). For example, a variety of evidence has shown that some neuroactive steroids, such as the progesterone derivative allopregnanolone (ALLO), are potent positive modulators of the GABAA receptor at low nanomolar concentrations (Gee, 1988; Morrow et al., 1987). Additionally, neuroactive steroids have low affinity at traditional steroid nuclear receptors (Belelli et al., 1990).
Neuroactive steroid-induced potentiation of the action of GABA at GABAA receptors can cause anticonvulsant, anxiolytic, sedative, ataxic, and cognitive impairing properties (Gasior et al., 1999), much like the effects of ethanol (EtOH; Khisti et al., 2003). While the exact mechanism of EtOH-induced potentiation of GABAA receptor function remains controversial, it is widely accepted that some of the effects of EtOH may be due to potentiation of GABA at GABAA receptors (Grobin et al., 1998). Recent work has found that acute EtOH intoxication causes a rise in GABAergic neuroactive steroids in both plasma and brain (Barbaccia et al., 1999; Finn et al., 2004b; VanDoren et al., 2000) and that the effect of EtOH on steroidogenesis produced an indirect effect on GABAA receptor function (Sanna et al., 2004). Thus, it is possible that an interaction of EtOH and GABAergic neuroactive steroids at GABAA receptors could influence sensitivity to some of the behavioral effects of EtOH. Consistent with this idea, blocking the formation of ALLO with the 5α-reductase inhibitor finasteride decreased the anticonvulsant properties of an acute dose of EtOH at 40 minutes, but not at 10 minutes, post-EtOH injection (VanDoren et al., 2000). Likewise, removal of the peripheral sources of neurosteroids eliminated the EtOH-induced increase in ALLO levels (O’Dell et al., 2004) and altered several behavioral aspects of EtOH intoxication such as anxiety, depression, and loss of righting reflex (LORR) (Hirani et al., 2002, 2005; Khisti et al., 2003). Collectively, these finding suggest that removal of the peripheral sources of GABAergic neuroactive steroids may alter behaviors associated with acute EtOH withdrawal following a single dose of EtOH.
The majority of data indicate that corticosterone administration is proconvulsant. Acute doses of EtOH increased corticosterone levels in both DBA/2 (D2) and C57BL/6 (B6) mice, which are 2 inbred strains with well-documented differences in EtOH withdrawal severity (Crabbe, 1998; Crabbe et al., 1983; Roberts et al., 1992). However, the corticosterone response in the seizure prone D2 animals was significantly higher at 60 minutes postinjection than in B6 mice (Roberts et al., 1992). Administration of high levels of corticosterone (100 mg/day) to rats accelerated the presentation of tonic-clonic seizures in animals undergoing kindling epileptogenesis (Karst et al., 1999). Corticosterone administration has also been shown to increase convulsions due to acute withdrawal from several drugs, including EtOH, in mice (Roberts et al., 1994). While corticosterone may be proconvulsant, it has been suggested that the precursor to corticosterone, deoxycorticosterone, may be seizure protective, along with other GABAergic deoxycorticosterone metabolites (Reddy and Rogawski, 2002).
Data indicated that testosterone can be both anti- and proconvulsant, depending on its metabolism. Testosterone replacement in gonadectomized (GDX) animals increased seizure susceptibility in rats administered kainic acid, indicating that testosterone can be proconvulsant (Mejias-Aponte et al., 2002). However, studies examining testosterone metabolism have shown that when testosterone was aromatized into 17β-estradiol, it was proconvulsant. In contrast, when testosterone was reduced into 5α-dihydrotestosterone and 3α-androstanediol, it was anticonvulsant (Frye and Reed, 1998; Reddy, 2004a). When the formation of estrogen was inhibited following testosterone administration, both rats and mice were protected against picrotoxin (a GABAA receptor channel blocker) induced seizures, but not against kainic acid-induced seizures (Reddy, 2004a,b). These data indicate that testosterone’s reduced metabolites are anticonvulsant via a GABAA receptor-mediated pathway.
While mentioned briefly above as proconvulsant, it is important to note that estrogen’s convulsant actions depend upon several variables, notably treatment duration, dose, and mode of administration (Veliskova, 2007). In ovariectomized (OVX) animals, chronic replacement of estrogen was seizure protective against picrotoxin-induced seizures (Schwartz- Giblin et al., 1989). However, in a similar paradigm, estrogen replaced OVX animals took fewer daily amygdala stimulations to develop seizures and developed more intense seizures to repeated pentylenetetrazol injections. Thus, estrogen can exhibit proconvulsant and anticonvulsant properties.
Some of the more potent anticonvulsant steroids are the reduced metabolites of progesterone. It was first shown in 1942 that progesterone had anticonvulsant properties (Seyle, 1942). Recent studies determined that progesterone’s anticonvulsant properties were due to its metabolism to 5α-dihydroprogesterone (5α-DHP) and ALLO (Frye et al., 2002). 5α-DHP has been shown to be seizure protective in very low doses and at time points as soon as 15 minutes postinjection (Lonsdale and Burnham, 2003).
In addition to steroid hormones and their metabolites, the adrenal glands are a significant source of epinephrine, which also affects seizure susceptibility. Injecting systemic epinephrine retards the development of kindling-induced seizures in rats (Welsh and Gold, 1986), and it has been shown that this effect is modulated through the vagus nerve (Krahl et al., 2000). While epinephrine is produced in the adrenal medulla in response to stress, it is important to note that following ADX, epinephrine can still be produced by other organs (Ricordi et al., 1988) as well as in the brain (Santibañez et al., 2005).
The above data illustrate that hormones and their metabolites can have either pro- or anticonvulsant properties and that these steroids may be important for some of the behavioral effects of EtOH, such as seizure susceptibility. Whereas these steroids can be produced both peripherally and de novo in the brain, little attention has been paid to which source of steroid hormones is important in modulating specific effects of EtOH. The few studies that have examined these effects have shown an interesting relationship between peripherally derived steroids and sensitivity to EtOH withdrawal-induced seizures. Adrenalectomized (ADX) mice that were exposed to 14 days of chronic EtOH treatment showed a marked decrease in audiogenic-induced seizures, but these seizures were reinstated with administration of glucocorticoids (Sze et al., 1974). In a similar fashion, it was again shown that ADX was seizure protective in rats against harmine (a β-carboline tremorogenic agent) -induced seizures after withdrawal from 3 weeks of exposure to vaporized EtOH (Lamblin et al., 1996). However, these investigations were undertaken in chronic EtOH-exposed animals, a procedure that can drastically affect receptor sensitivity (Follesa et al., 2006) and the steroid profile from acute exposure (Finn et al., 2004b; Romeo et al., 1996), leaving one unable to extend the conclusions from these studies to similar undertakings in an acute EtOH withdrawal model (i.e. withdrawal from a single high dose of EtOH).
The current experiments investigated the role of peripherally derived steroids on acute EtOH withdrawal severity following a single dose of EtOH, measured by handling-induced convulsions (HICs). We hypothesized that peripheral sources of steroids, the adrenals and gonads, are important during withdrawal from a single EtOH exposure and that the direction of change in acute withdrawal severity would provide insight into the relative contribution of pro- versus anticonvulsant steroids to acute EtOH withdrawal severity. That is, an increase in EtOH withdrawal severity following removal of the adrenals and gonads would suggest that endogenous anticonvulsant steroids were protective against HICs. In contrast, a decrease in acute EtOH withdrawal severity following removal of the adrenals and gonads would suggest that endogenous proconvulsant steroids were increasing HICs. These effects were investigated in B6 and D2 male and female mice, based on the several lines of evidence. First, it is well established that acute and chronic EtOH withdrawal (Crabbe, 1998; Crabbe et al., 1983; Roberts et al., 1992) is significantly greater in D2 than B6 mice. Additionally, sensitivity to the anticonvulsant effect of ALLO is significantly greater in EtOH-naïve B6 versus D2 mice (Finn et al., 1997) and sensitivity to the anticonvulsant effect of ALLO is enhanced in B6 and reduced in D2 mice during chronic EtOH withdrawal (Finn et al., 2000). Finally, endogenous ALLO levels are higher in female than in male mice (Finn et al., 2004a) and acute EtOH withdrawal is lower in female than in male B6 and D2 mice (Gorin-Meyer et al., 2007). An additional consideration was that ADX and GDX have been reported to have varying effects on EtOH metabolism (Becker et al., 1985; Bude et al., 2002; Mezey et al., 1980; Powis et al., 1977; Wallis et al., 1984). Thus, a final study examined the effect of ADX and GDX on acute EtOH metabolism in order to account for changes in metabolism on acute EtOH withdrawal severity.
MATERIALS AND METHODS
Subjects
Eight- to 12-week-old drug naïve, sexually mature, male and female C57BL/6J (B6) and DBA/2J (D2) mice were obtained from Jackson West Laboratories (Davis, CA). Mice were separated by sex and strain; group housed 4 to a cage and were allowed free access to rodent chow (Labdiet 5001 rodent diet; PMI International, Richmond, IN) and water. Mice were maintained on a 12-hour (6 AM to 6 PM) light/dark cycle in polycarbonate cages (Thorens, Hazleton, PA) in a room kept at 21 ± 2°C with humidity control. Mice were allowed to acclimate to the facility for at least 1 week before any experimental manipulations were undertaken.
Procedure
Male and female B6 and D2 mice were randomly assigned to 1 of 4 groups: ADX, GDX, both surgeries (ADX/GDX), or no organs removed (SHAM), and had the appropriate surgery performed (details below). Seven to 14 days after surgery, baseline HICs were assessed in the mice. This time period allowed the animals to recover from the surgery and clear all endogenous steroids (Khisti et al., 2003). The mice were then given an intraperitoneal (IP) injection of 4 g/kg of EtOH (Aaper alcohol and chemical company, Shelbyville, KY; 20% v/v in saline). HIC severity was monitored hourly for 12 hours following injection and again at 24 hours. Following HIC scoring, animals were decapitated and dissected to confirm organ removal. Due to the large number of animals, several experiments were conducted; animals from each sex and genotype were tested at the same time. All procedures were conducted in accordance with the Guide for the Care and Use of Laboratory Animals as adopted by the U.S. National Institutes of Health and were approved by the local Institutional Animal Care and Use Committee.
Surgery
All surgeries were adapted from “The Biology and Medicine of Rabbits and Rodents” (Harkness and Wagner, 1995). Anesthesia was induced with 5% isoflurane (Abbott Laboratories, North Chicago, IL) and maintained at 2% throughout surgery. Briefly, to remove the adrenal glands, a dorsal midline incision was made. This incision was shifted to either side to expose the areas lateral of the midline. Two more incisions were made to the muscle wall just behind the last rib on both sides in order to expose the anterior pole of the kidney and the adrenal gland. The adrenal gland was removed by separating the gland from the surrounding tissue with tweezers and then gently pulling the gland through the flank incision; 4-0 chromic gut sutures (Davis & Geck, Danbury, CT) were used to close the incisions in the muscle walls, and metal clips were used to close the flank incision. To remove the testicles, the peritoneal cavity was entered via a small cranial pubic incision. The testicular vessels were severed using a cauterizing gun causing the testes, epididymis, and surrounding fat to be separated from the body. The body wall and cutaneous wound was closed with tissue adhesive. To remove the ovaries, a dorsal midline incision was made. Like the ADXs, this incision was used to expose the muscle wall lateral to the midline. Two incisions were made 4 to 5 cm below the last rib on both sides and the ovary and oviduct were removed through the flank incision; 4-0 gut sutures were used to close the incision in the muscle wall and metal clips were used to close the flank. The mice in the ADX/GDX group received both surgeries during the same session. In females, the adrenals and the ovaries were removed through the same incisions. SHAM animals were anesthetized and had the appropriate incisions made, but no organs were removed. After surgery, all animals were allowed to recover on a heating pad, were administered a subcutaneous dose of 3 mg/kg keterolac (Sigma-Aldrich, St Louis, MO) for 2 days and had unlimited access to 0.9% NaCl (in order to maintain sodium balance (Beers and Berkow, 2005); Baxter, Deerfield, IL), water and chow for the remainder of the experiment. The weight of all animals was monitored to insure weight loss after surgery was minimal and that all animals recovered to SHAM levels.
HIC Scoring
Handling-induced convulsions were scored according to a previously published scale (Crabbe et al., 1991). Briefly, a mild convulsion can be elicited by gently lifting a mouse by the tail and turning it 180° if necessary. The HIC scoring ranges from 0 to 7; a score of 0 indicates no convulsions, a score including 1 to 3 indicates tonic or clonic convulsions obtained by a gentle turn, a score including 4 to 6 indicates convulsions elicited by only lifting the mouse by the tail, and a score of 7 indicates a spontaneous convulsion. Specifically, the animal is first briefly observed in the home cage for spontaneous convulsions, which would indicate a score of 7. If no spontaneous seizures are observed, the animal is lifted by the tail and briefly observed. If a seizure is elicited by lifting the animal alone, the HIC is scored as a 4 to 6, depending upon the severity of the convulsion (4 indicates a tonic convulsion, 5 indicates a tonic clonic convulsion, and 6 indicates a severe tonic clonic convulsion) and the animal is returned to its home cage. If no seizure is elicited from lifting the animal, the animal is gently turned 180° by the tail. Seizures elicited by this turn are scored as 1 to 3, depending upon the severity (1 indicates a facial grimace, 2 indicates a tonic convulsion, and 3 indicates a tonic clonic convulsion). If no seizure is observed after one 180° spin, the HIC is scored as a zero and the animal is returned to the cage.
Blood Ethanol Concentrations
A separate group of animals (B6 and D2 males and females) underwent surgeries, as described above, in another experiment. After 7 to 14 days of recovery, these animals were administered a 4 g/kg IP dose of EtOH. Immediately following EtOH administration, animals were tested for ethanol clearance rate using previously published methods (Gorin-Meyer et al., 2007; Shen et al., 1995). Briefly, retro-orbital blood samples were collected from each animal at 30, 60, 120, and 240 minutes post-EtOH injection. Mice were restrained by hand while a 20-μl sample of blood was collected; eyes were alternated for each time point to minimize trauma. Between sampling, the mouse was released back to its home cage. Samples were used to determine blood ethanol concentrations (BECs) at each time point. The blood samples were diluted into 500 μl of a matrix of 4 mMn-propanol in deionized water. The 2.0 ml crimp top vial containing the blood sample in matrix was capped and vortexed thoroughly before analysis. Analysis was performed via ambient headspace sampling gas chromatography (Agilent 6890N GC, using a DB-ALC1 column, Wilmington, DE) on a 30 μl aliquot. Six pairs of ethanol standards (0.5 to 5.0 mg/ml), which included n-propanol (internal standard), were run before the samples (Finn et al., 2007). Recent work suggests that multiple retro-orbital blood sampling did not significantly alter BECs relative to animals that had received a single blood sampling (Kamens et al., 2006).
Data Analysis
Data are expressed as the mean ± SE. Analyses were conducted in animals with verified organ removal. Withdrawal severity was quantified by calculating the area under the curve (AUC) for each animal (hours 0 to 24), using the trapezoidal method, as previously described (Crabbe et al., 1983;Metten and Crabbe, 2005). Peak withdrawal was found for each subject by taking the highest HIC score for each individual animal and averaging the peak score with the surrounding 2 scores. A multifactorial ANOVA analyzing time as a within subjects factor and sex, genotype, and surgery as between subjects factors was conducted. When appropriate (due to significant interactions), data were separated by sex and genotype and analyzed with a two-way ANOVA (surgery by time) with repeated measures and a Greenhouse–Geiser correction. Each time point was analyzed separately when appropriate using a one-way ANOVA. AUC was analyzed with a three-way ANOVA (sex by genotype by surgery) and then followed by a one-way ANOVA analyzing each sex and strain separately. Peak withdrawal was analyzed similarly. Baseline HIC scores were analyzed with a multifactorial ANOVA and if necessary, a one-way ANOVA due to interactions.
Linear regression analysis was performed on the retro-orbital BEC time course data for each repeatedly sampled animal. The linear portion of the regression line (60 to 240 minutes) was used to obtain an estimate of clearance rate (mg/ml/h). An estimate of volume of distribution (ml) and volume of distribution accounting for body weight (ml/g) was determined by dividing the milligrams of EtOH that each animal received by the estimated BEC at time = 0 (based on the regression slope and the y-intercept of the regression line). An estimate for total clearance time (minutes) was determined by the x-intercept of the regression line. A repeated measures ANOVA was used to assess the effects of strain, sex, surgery, and time on BECs, while a three-way ANOVA was used to assess the effects of strain, sex, and surgery on several of the clearance parameters. When appropriate, clearance parameters where analyzed with a one-way ANOVA on each sex and strain.
Tukey’s post hoc test was used when appropriate. Significance was set at p ≤ 0.05 for all analyses. All statistics were conducted with the SPSS statistical package (version 11; SPSS, Chicago, IL).
RESULTS
HIC Scores
A multifactorial ANOVA revealed that there were significant main effects of genotype (D2 > B6) [F(1,201) =353.22, p < 0.001] and surgery on hourly HIC score [F(3,201) = 10.15, p < 0.001], but no main effect of sex. However, this analysis also revealed that there was a trend for a significant four-way interaction between time, sex, genotype, and surgery [F(14,25.35) = 1.45, p = 0.067] and that all other interactions were significant [F’s(14,25.357) > 2.07, p’s ≤ 0.001]. Due to these highly significant interactions, further analysis on the hourly HIC scores was conducted separately on groups divided by sex and genotype.
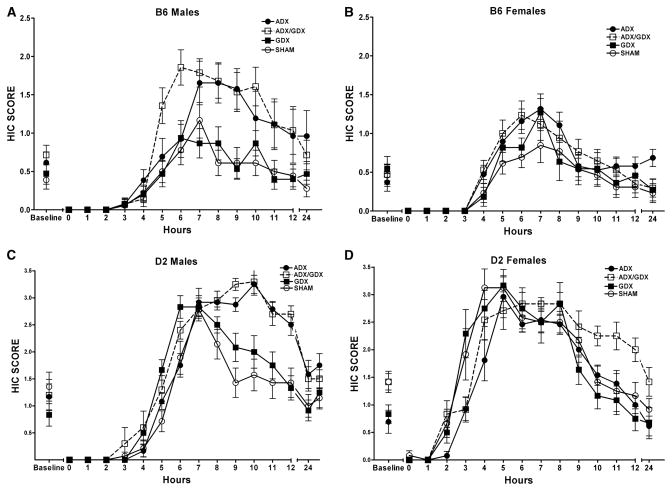
Removal of the peripheral sources of steroids significantly increased the acute withdrawal profile from a 4 g/kg dose of ethanol in D2 male and female animals and in B6 male animals, measured by hourly HIC scores. In contrast, surgical status did not alter acute withdrawal severity in B6 female mice (See Fig. 1A–D). In D2 males, both ADX and ADX/GDX mice had significantly elevated withdrawal profiles, when compared with SHAM and GDX mice. This conclusion is supported by the main effect of surgery [F(3,43) = 10.01, p < 0.001], main effect of time [F(6,288) = 142.61, p < 0.001], a significant interaction [F(20,288) = 1.94, p < 0.001] and post hoc tests. In D2 females, only ADX/GDX mice had an elevated withdrawal profile, when compared with the other surgical groups (main effect of surgery [F(3,45) = 3.40, p = 0.024], main effect of time [F(7,307) = 85.46, p < 0.001], a significant interaction [F(20,307) = 2.64, p < 0.001], and post hoc tests). Similar to D2 males, B6 males had increased withdrawal in both ADX/GDX and ADX groups, when compared with SHAM and GDX mice. This conclusion is supported by the main effect of surgery [F(3,56) = 6.75, p = 0.001], main effect of time [F(11,616) = 23.99, p < 0.001] a significant interaction between surgical status and time [F(33,616) = 2.14, p < 0.001] and post hoc tests. However, there was no effect of surgical status on the acute EtOH withdrawal profile in B6 females (main effect of time [F(7,395) = 39.07 p < 0.001], but no effect of surgery and no interaction).
Fig. 1.
The effect of surgical status on acute ethanol (EtOH) withdrawal severity in male and female C57BL/6J (B6) and DBA/2J (D2) mice, measured by hourly handling-induced convulsions (HICs). Overall, male B6 (panel A) and D2 (panel C) mice that had undergone adrenalectomy (ADX) or ADX and gonadectomy (GDX) had a more severe withdrawal response from a high dose of EtOH. In D2 females (panel D) only ADX/GDX mice had a more severe withdrawal from an acute EtOH exposure, while there was no effect in B6 female mice (panel B). Values represent the mean ± SEM for 10 to 15/group. Note differences in y-axis between D2 and B6 mice.
Analysis was carried out on the baseline HIC scores in order to ascertain if baseline HIC scores were affected by surgical status. A univariate ANOVA revealed that there were significant main effects of genotype on the baseline HIC score (D2 > B6) [F(1,198) = 354.35, p < 0.001] and a main effect of surgery [F(3,198) = 2.86, p = 0.038]. However, there was also an interaction of genotype by surgery [F(3,198), p = 0.047]. In order to be comparable with the analysis on HICs during withdrawal, groups were separated by sex and genotype. In B6 males and females and D2 males, there were no effects of surgical status on baseline HIC score. There were effects of surgery on baseline HIC scores in D2 females [F(3,42) = 5.334, p = 0.003], with post hoc tests revealing that baseline HIC score was elevated in ADX/GDX and SHAM-operated groups. Due to this finding, the previous analysis on HIC scores during withdrawal was repeated with baseline as a covariate. This analysis produced results similar to that of the analysis run without baseline as a covariate, indicating that the differences in baseline HIC did not significantly alter the acute EtOH withdrawal response measured by HIC.
To follow up on the analysis of the HIC time course with each sex and genotype, separate analyses were conducted at each time point to assess the effect of surgical status on HIC scores. In D2 male mice, there were significant effects of surgery on HIC score at hours 6, 8, 9, 10, and 12 [F’s(3,47) > 3.9, p’s ≤ 0.05]. Post hoc tests revealed that at hour 6, GDX animals were elevated above SHAM and ADX animals and that at hours 8, 9, 10, and 12, ADX and ADX/GDX animals were elevated over SHAM and GDX animals. In D2 female mice, there were effects of surgery at the baseline time point and at hours 3, 10, 11, and 12 [F’s(3,48) > 3.61, p’s ≤ 0.05]. Post hoc’s revealed that at the baseline, ADX/GDX and SHAM animals were elevated, when compared with ADX animals. At hour 3, GDX animals were elevated over ADX and ADX/GDX animals, at hours 10 and 11, ADX/GDX animals were elevated over both ADX and GDX animals, and at hour 12, ADX/GDX animals were elevated over all other groups. In B6 males there was a significant effect of surgery at hours 5 through 10 [F’s(3,59) > 3.43, p’s ≤ 0.05]. Post hoc’s revealed that at hour 5, ADX/GDX animals were elevated over GDX and SHAM and at hour 6, ADX/GDX animals were elevated over all groups. At hour 7, ADX/GDX animals were elevated over GDX animals, at hour 8, ADX/GDX animals were elevated over GDX and both ADX/GDX and ADX animals were higher than SHAM. At hour 9, ADX and ADX/GDX animals were elevated over both GDX and SHAM animals and finally, at hour 10 ADX/GDX animals were elevated over SHAM animals. There were no significant effects of surgery on HIC scores at any time point in B6 females.
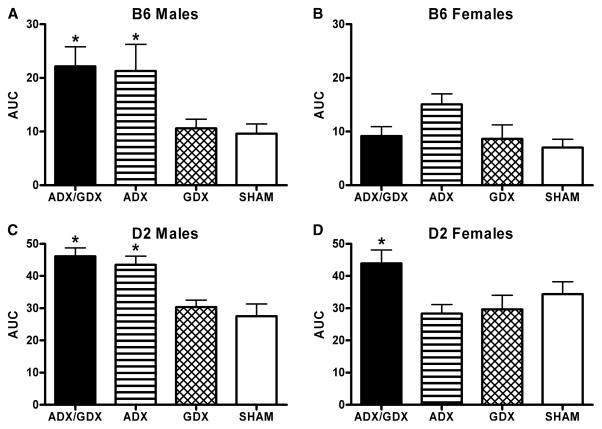
In order to assess the effect of removal of peripheral sources of steroids on total withdrawal severity, the area under the withdrawal curve was calculated for each group of animals (see Fig. 2). A three-way ANOVA revealed a main effect of genotype (D2 > B6) [F(1,216) = 220.85, p < 0.001], sex (male > female) [F(1,216) = 7.25, p < 0.005], and surgery [F(3,216) = 12.85, p < 0.001] on AUC. There was also a significant interaction of sex and surgery [F(3,216) = 4.25, p < 0.005] and a trend for an interaction between genotype and surgery [F(1,216) = 2.361, p < 0.075]. Due to these interactions, further analysis was conducted on groups, separated by sex and genotype. In D2 male mice, AUC was increased significantly in ADX/GDX and ADX mice, when compared with values in SHAM and GDX animals [F(3,43) = 10.01, p < 0.001 and post hoc tests]. The same pattern was observed in B6 male mice, with ADX/GDX and ADX mice having increased overall withdrawal when compared with SHAM and GDX animals [F(3,56) = 6.751, p < 0.001 and post hoc tests]. In D2 female mice, AUC was increased significantly only in the ADX/GDX mice when compared with SHAM animals [F(3,45) = 3.40, p < 0.05 and post hoc test]. There was no significant effect of surgery on AUC in B6 female mice [F(3, 56) = 2.4, p = 0.09].
Fig. 2.
Surgical status significantly alters acute ethanol (EtOH) withdrawal severity, measured by area under the curve (AUC). Tukey post hoc tests confirmed that AUC in the adrenalectomy (ADX) and the ADX/GDX groups were significantly higher than in the gonadectomy (GDX) and no surgery (SHAM) groups in the C57BL/6J (B6; panel A) and DBA/2J (D2; panel C) males. In the D2 females (panel D), AUC was significantly increased only in the ADX/GDX group, while in the B6 females (panel B), there were no group differences in AUC. Values represent the mean ± SEM for the animals depicted in Fig. 1. *Indicates significance of p ≤ 0.05 versus SHAM. Note differences in y-axis between D2 and B6 mice.
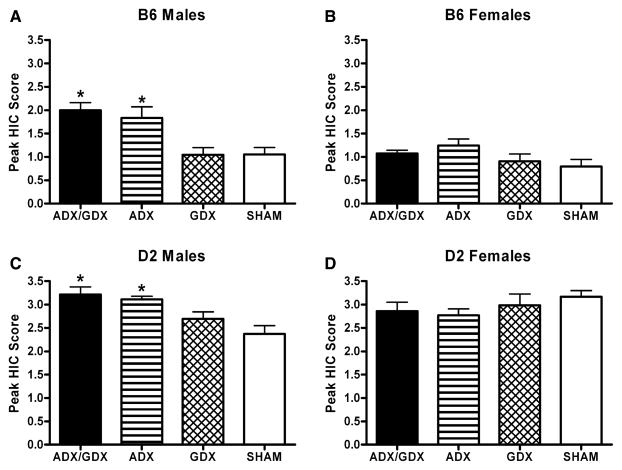
One way in which surgery could be increasing overall withdrawal severity is through increasing the peak HIC score that each animal reached. In order to investigate this possibility, average peak withdrawal was compared between surgical groups in each sex and genotype (see Fig. 3). In each animal, the high dose of ethanol used in this experiment causes the animals to be completely sedated for several hours. As the EtOH is metabolized, withdrawal occurs with animals reaching peak withdrawal ~4 to 6 hours following EtOH administration. ANOVA revealed that in male D2 and B6 mice, surgical status significantly altered peak withdrawal severity [F(3,56) = 8.46, p < 0.05 and F(3,44) = 6.95, p < 0.05, respectively]. There was no main effect of surgery in either female B6 or D2 mice. Tukey’s post hoc analysis in the 2 male groups revealed that both ADX and ADX/GDX surgical treatment significantly increased peak withdrawal when compared with the SHAM group.
Fig. 3.
Surgical status significantly affects peak withdrawal scores in male mice, but not in female mice. Peak withdrawal is defined as the peak handling-induced convulsion score averaged with the 2 surrounding scores for each animal. In C5BL/6J (B6) males (panel A), peak withdrawal was significantly increased in groups that had undergone adrenalectomy (ADX) and ADX/gonadectomy (GDX) surgeries. The same pattern was true for DBA/2J (D2) males (panel C). There was no difference in peak withdrawal scores in D2 (panel D) or B6 (panel B) female mice. Values represent the mean ± SEM for the animals depicted in Fig. 1. *Indicates significance of p ≤ 0.05 versus respective SHAM.
BEC Determination
A univariate ANOVA on body weight revealed a main effect of sex (male > female) [F(1,73) = 110.88, p < 0.001] and genotype (D2 > B6) [F(1,73) = 95.58, p < 0.001] with significant interactions between genotype and sex [F(1,73) = 9.72, p < 0.01] and between surgery, genotype, and sex [F(3,73) = 3.27, p < 0.05] on body weight. When divided by sex and genotype, there were no effects of surgery in male and female D2 or female B6 mice. There was a significant effect of surgery on body weight in B6 males [F(3,24) = 5.75, p < 0.005) with the ADX/GDX group weighing significantly less when compared with SHAM animals (see Table 1).
Table 1.
Ethanol Clearance Parameters
| ADX | GDX | ADX/GDX | SHAM | ADX | GDX | ADX/GDX | SHAM | |
|---|---|---|---|---|---|---|---|---|
| Clearance parameters | C57BL/6J Males | C57BL/6J Females | ||||||
| Weight (g) | 25.18 ± 0.59 | 24.03 ± 0.59 | 23.15 ± 0.36 | 25.97 ± 0.49 | 20.00 ± 0.32 | 19.68 ± 1.31 | 20.82 ± 0.40 | 19.65 ± 0.24 |
| BEC at time = 0 (mg/ml) | 4.87 ± 0.19 | 4.78 ± 0.15 | 4.79 ± 0.12 | 4.86 ± 0.21 | 5.28 ± 0.23 | 5.24 ± 0.20 | 5.92 ± 0.28 | 5.08 ± 0.12 |
| Clearance rate (mg/ml/h) | 1.00 ± 0.03 | 0.86 ± 0.03 | 0.99 ± 0.02 | 0.95 ± 0.03 | 1.10 ± 0.10 | 1.28 ± 0.19 | 1.44 ± 0.15 | 1.01 ± 0.11 |
| Vd (ml) | 20.87 ± 1.16 | 20.29 ± 0.66 | 19.38 ± 0.42 | 21.76 ± 1.07 | 15.49 ± 0.92 | 15.80 ± 0.73 | 14.23 ± 0.57 | 15.47 ± 0.37 |
| Vd/body weight (ml/g) | 0.83 ± 0.04 | 0.85 ± 0.03 | 0.84 ± 0.02 | 0.84 ± 0.04 | 0.77 ± 0.04 | 0.82 ± 0.08 | 0.68 ± 0.03 | 0.79 ± 0.02 |
| Total clearance time (minutes) | 296.22 ± 20.81 | 335.19 ± 10.18 | 289.81 ± 4.85 | 306.66 ± 12.47 | 297.98 ± 20.81 | 250.50 ± 17.25 | 253.12 ± 13.70 | 304.62 ± 8.94 |
| DBA 2/J Males | DBA 2/J Females | |||||||
| Weight (g) | 25.55 ± 0.378 | 25.64 ± 0.33 | 25.53 ± 0.32 | 25.90 ± 0.20 | 22.67 ± 1.09 | 24.05 ± 0.41 | 21.97 ± 0.42 | 24.07 ± 0.95 |
| BEC at time = 0 (mg/ml) | 4.73 ± 0.29 | 5.51 ± 0.17 | 5.12 ± 0.34 | 5.01 ± 0.07 | 5.25 ± 0.17 | 4.92 ± 0.23 | 5.40 ± 0.12 | 4.83 ± 0.17 |
| Clearance rate (mg/ml/h) | 0.90 ± 0.08 | 1.23 ± 0.02 | 1.10 ± 0.06 | 1.20 ± 0.03 | 1.22 ± 0.08 | 1.13 ± 0.05 | 1.34 ± 0.05 | 1.23 ± 0.04 |
| Vd (ml) | 21.84 ± 0.89 | 18.70 ± 0.47 | 20.66 ± 1.77 | 20.71 ± 0.53 | 17.31 ± 0.65 | 19.72 ± 0.98 | 16.37 ± 0.49 | 20.00 ± 0.77 |
| Vd/body weight (ml/g) | 0.85 ± 0.03 | 0.82 ± 0.02 | 0.81 ± 0.07 | 0.80 ± 0.01 | 0.77 ± 0.02 | 0.73 ± 0.04 | 0.74 ± 0.01 | 0.83 ± 0.03 |
| Total clearance time (minutes) | 393.49 ± 17.84 | 293.96 ± 9.08 | 283.48 ± 24.53 | 250.56 ± 7.67 | 262.41 ± 11.70 | 241.58 ± 10.11 | 242.44 ± 8.02 | 236.22 ± 10.02 |
Values represent the mean ± SEM for 12 to 15/group. ADX, adrenalectomy; BEC, blood ethanol concentration; GDX, gonadectomy.
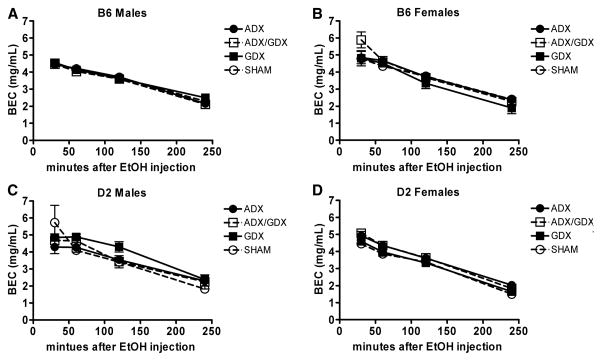
Repeated measures ANOVA of BECs measured 30, 60, 120, and 240 minutes after administration of a 4 g/kg dose of EtOH indicated that that BECs changed significantly across time [F(3,216) = 1003.31, p < 0.001]. There was a significant interaction between time and sex [F(3,126) = 12.07, p < 0.001], but no interaction between time and surgery or time and genotype. When divided by sex and genotype, there was no effect of surgery on BEC in B6 or D2 male or female mice at any time point (see Fig. 4A–D).
Fig. 4.
Surgical status has no significant effect on EtOH metabolism. In C57BL/6J (B6) males (panel A), there was no significant effect of surgery on blood ethanol concentration (BEC) at 30, 60, 120, or 240 minutes after EtOH administration. The same pattern was true for B6 females (panel B) and DBA/2J (D2) male and female mice (Panels C and D, respectively). Values represent the mean ± SEM for 12 to 15/group.
Clearance rates were based on the linear portion of the curve (60 to 240 minutes post-EtOH injection). These estimated clearance rates were used to calculate several other clearance parameters (See Table 1). A three-way ANOVA revealed that clearance (mg/ml/h) was significantly affected by genotype (D2 > B6) [F(1,88) = 11.718, p < 0.001], sex (female > male) [F(1,88) = 34.36, p < 0.001], and surgery (ADX > SHAM) [F(1,88) = 3.30, p < 0.05 and post hoc]. These changes in clearance rates represent a decrease of 9% in B6 compared with D2 mice, an 18% decrease in clearance in male compared with females and a 10% decrease in SHAM compared with ADX mice.
Other clearance parameters that were affected by both sex and genotype included the volume of distribution [F’s(1,88) > 7.5, p’s < 0.01] and total clearance time [F’s(1,88) > 17.31, p’s < .001]. There were no effects of surgery on either of these parameters.
DISCUSSION
The current experiments were undertaken in an effort to elucidate the effect of peripherally derived steroids on acute EtOH withdrawal severity. These experiments revealed that in male D2 and B6 mice, the removal of the adrenal glands significantly increased the withdrawal profile following an acute exposure to EtOH, measured by hourly HIC scores, AUC and peak withdrawal. These measures were not significantly affected by GDX in male mice. In female D2 mice, the concurrent removal of the adrenals and the gonads significantly increased the withdrawal profile from an acute exposure to EtOH. Whereas hourly HIC score and AUC were increased in the ADX/GDX group, organ removal had no effect on peak withdrawal. The removal of the adrenals and/or gonads had no effect on acute withdrawal severity in B6 female mice. In general, these findings suggest that peripherally derived steroids are important during withdrawal from a high dose of EtOH. The fact that removal of the sources of these steroids significantly increased acute withdrawal severity in 3 of 4 sexes/strains suggests that an endogenous anticonvulsant steroid (or steroids) contributes to the withdrawal profile exhibited in intact animals.
While removing the peripheral sources of steroids increased withdrawal from acute EtOH exposure in both D2 and B6 male mice and in D2 female mice, the method of increase was different between the sexes. In the male groups, the severity of peak withdrawal in addition to the duration of withdrawal were both significantly increased in the ADX and ADX/GDX groups. In contrast, the female D2 group that received ADX/GDX treatment exhibited a significant increase in the duration of withdrawal, with no change the peak withdrawal severity. Withdrawal in D2 female mice started sooner and lasted longer when compared with male mice. It is interesting to note that withdrawal started in D2 female animals several hours before the D2 and B6 males (See Fig. 1). This is consistent with the fact that D2 females had a faster clearance time, when compared with the D2 and B6 males. This cannot, however, account for the fact that in D2 females, the AUC was greater in ADX/GDX animals. Nonetheless, these data suggest that removal of at least 1 anticonvulsant steroid significantly increased acute EtOH withdrawal severity and that male mice were more sensitive than the female mice to this manipulation (i.e. increases in peak and duration of withdrawal in D2 and B6 males versus increases in duration in D2 females).
The differences between the male and female groups in the effect of surgical status may give a clue as to which peripherally derived (anticonvulsant) steroids are important in modulating withdrawal from an acute EtOH exposure. In males, only the removal of the adrenal gland was effective at increasing withdrawal severity, as there were no differences between the ADX and ADX/GDX groups or the GDX and SHAM groups. These findings suggest that testosterone and its neuroactive steroid metabolites, 5α-dihydrotestosterone and 3α-androstanediol, are not mediating the increase in withdrawal severity even though these neuroactive steroids are potent positive modulators of GABAA receptors and have seizure protective properties (Reddy, 2004a). As both progesterone and deoxycorticosterone are produced in the adrenals, it is possible that GABAergic metabolites of either or both of these steroids (i.e. 5α-tetrahydrodeoxycorticosterone or ALLO) are contributing to the present findings in male mice. Additionally, epinephrine is produced in the adrenals and removal of epinephrine could be contributing to increased seizure susceptibility. This may be less likely, however, because as previously mentioned, epinephrine is still produced in the brain following ADX.
In contrast to the male group differences, withdrawal severity was only increased when both the adrenals and the ovaries were removed in the female D2 mice. The female gonads produce a significant amount of progesterone and estrogen in sexually mature mice such as those used in this study. While estrogen is proconvulsant (Reddy, 2004b), progesterone metabolites are anticonvulsant (Belelli et al., 1989) and represent some of the most potent endogenous positive GABAA receptor modulators (Gee, 1988; Morrow et al., 1987). Additionally, the adrenal glands also significantly contribute to the levels of progesterone in females. An acute dose of EtOH can markedly increase progesterone levels in OVX rats, suggesting that the adrenals represent an important contribution to hormone levels (Budec et al., 2002). Increases in progesterone levels following an acute dose of EtOH also is seen in male rats and is attenuated by removal of the adrenals (O’Dell et al., 2004). Taken together, these data provide indirect evidence that progesterone or a progesterone metabolite, such as ALLO, may be important in mediating EtOH withdrawal severity in male and female mice and that these steroids are peripheral in origin.
Several studies have demonstrated that modest, acute doses of EtOH increased neuroactive steroid levels in both the brain and plasma of rodents and that this increase was dose dependent (Barbaccia et al., 1999; Morrow et al., 1999; VanDoren et al., 2000). More recent studies have provided evidence that this rise could be attributed to peripheral origins (Budec et al., 2002; O’Dell et al., 2004), as removal of the adrenals and gonads attenuated the rise in neuroactive steroids or their precursors following an acute dose of EtOH. Due to the time course of the rise and fall of neuroactive steroids in response to a dose of EtOH, it has been hypothesized that neuroactive steroids may be modulating some of EtOH behavioral effects such as its anxiolytic (Hirani et al., 2005), anticonvulsant (VanDoren et al., 2000) and antidepressant (Hirani et al., 2002) properties. The anticonvulsant effect of EtOH can be prevented by administration of finasteride, which blocks the metabolism of progesterone, deoxycorticosterone, and testosterone, providing evidence that GABAergic neuroactive steroids can modulate some of EtOH’s effects (VanDoren et al., 2000). Finasteride also reduced the antidepressant effects of EtOH in the forced swim test (Hirani et al., 2002). Additionally, it was recently shown that animals that had undergone ADX had reduced EtOH-induced LORR and that administering 5α-DHP (a precursor of ALLO) restored this behavior (Khisti et al., 2003). Finally, removal of the adrenal glands in male rats attenuated the rise in ALLO following an acute dose of EtOH (O’Dell et al., 2004). Thus, limited data suggest that neuroactive steroid synthesis, especially that of ALLO, is important for some EtOH-related behaviors and that this synthesis may occur in peripheral organs.
In addition to supporting the hypothesis that peripherally derived steroids can modulate withdrawal severity to an acute dose of EtOH, the present finding that acute ethanol withdrawal was significantly greater in D2 than in B6 mice is consistent with previous work (Gorin-Meyer et al., 2007; Roberts et al., 1992). The study by Roberts et al. (1992) suggested that corticosterone could be playing a role in the strain difference in acute withdrawal severity, as D2 mice had higher levels of corticosterone in response to an acute dose of EtOH and administration of high doses of corticosterone could mimic the acute withdrawal effect. One possibility is that the balance of steroid metabolism was shifted towards corticosterone as opposed to ALLO production in intact D2 mice and that D2 mice have higher withdrawal seizures because of lower production of ALLO in response to an acute dose of EtOH. However, the strain difference in withdrawal severity was maintained upon removal of the adrenals and gonads, suggesting the contribution of additional factors to the strain difference in acute withdrawal severity. Another possibility was that estrous cycle-related differences in seizure susceptibility (Finn and Gee, 1994) at the time of testing contributed to the strain differences in withdrawal severity in the female D2 and B6 animals. While estrous cycle was not monitored in these animals, it is well documented that group-housed mice cycle together (Turner and Babnara, 1976). In our experience, upwards of 70% of group-housed mice have synchronized estrous cycles (unpublished observation). Importantly, the strain difference in acute withdrawal severity between the female genotypes was maintained after OVX, suggesting that estrous cycle-related fluctuations in seizure susceptibility did not contribute to the strain difference in acute EtOH withdrawal severity in female D2 and B6 mice.
Additional studies were conducted to ensure that the present findings on withdrawal severity were not due to an indirect effect of surgical status on ethanol metabolism. The current study represents the first report of the effects of ADX/GDX on EtOH metabolism in these strains of mice. While surgery caused significant changes in body weight only in B6 males, EtOH administration was based on body weight, and changes in body weight did not affect EtOH metabolism. These results agree with previous findings in our laboratory that an acute 4 g/kg dose of EtOH produces consistent BECs across male and female D2 and B6 mice (Gorin-Meyer et al., 2007). However, it was previously reported that GDX can increase the rate of EtOH elimination through altering the activity of liver alcohol dehydrogenase. If GDX was increasing the rate of EtOH elimination, one would expect a decrease in withdrawal severity or for animals to reach peak withdrawal sooner. However, neither of these effects was seen in the current study. In support of this conclusion, our study found no effect of surgery on total elimination time. Additional work indicated that metabolism of the 4 g/kg dose of EtOH was not affected in either OVX rats (Budec et al., 2002) or OVX B6 mice (Becker et al., 1985), a conclusion supported by the current findings. With regard to ADX, there is contradictory data about the effects of ADX on EtOH metabolism. A study in which male rats were administered 0.8 g/kg of EtOH showed that ADX increased peak EtOH concentration without altering the rate of metabolism (Powis et al., 1977). In another study, female C3H mice were administered 1.5 g/kg of EtOH after ADX and it was shown that these mice had a lowered peak BEC and unaltered rate of clearance (Wallis et al., 1984). The current study showed that while ADX alone did not affect clearance rates, the overall effect of ADX in combination with GDX significantly increased clearance rates by 10% when compared with SHAM animals (although there was no change in overall clearance time). Collectively, the current results in concert with previous studies indicate that the effects of surgery on EtOH metabolism cannot explain the changes in acute withdrawal severity.
The fact that removal of peripheral sources of neurosteroids did not seem to affect acute EtOH withdrawal in female B6 mice needs further exploration. It is possible to modulate the withdrawal profile of seizure resistant strains, as has been shown in the selected line of mice with picrotoxin and pentylenetetrazol (Crabbe et al., 1991), suggesting that HICs can be increased under specific circumstances in resistant genotypes. However, in the present procedure, it is possible that B6 female mice are insensitive to neuroactive steroid modulation. It has been shown that sensitivity to neuroactive steroids differs between genotypes in male B6 and D2 mice (Finn et al., 1997, 2000), but this effect has not been investigated systematically in female animals. However, it has been shown that there is an increase in whole brain ALLO levels in male B6 mice in response to a voluntary drinking paradigm, while the same paradigm elicited no change in brain ALLO levels in female B6 mice (Finn et al., 2004b) and that injections of ALLO increased EtOH consumption in male B6 mice (Ford et al., 2005; Sinnott et al., 2002), but had no effect on consumption in female B6 mice (Ford et al., 2007). Taken together, these data suggest that female B6 mice may be relatively insensitive to neuroactive steroid modulation.
A limitation to the current findings is that hormone levels have not been analyzed in these animals. However, the removal of the adrenals and gonads was confirmed by visual inspection upon termination of the experiment, and animals with incomplete organ removal (<5%) were removed from all analyses. Additionally, measuring ALLO levels in both the brain and the periphery of these animals would lend strength to the idea that the effect of ADX and ADX/GDX on acute EtOH withdrawal severity was being modulated by ALLO or another progesterone metabolite. Although we were unable to measure ALLO levels in the present study due to decreased sensitivity of the antibody used in the ALLO assay after long-term storage, we are exploring alternate methods of ALLO analysis for future studies.
In conclusion, the present findings provide important evidence that peripherally derived anticonvulsant steroids are modulating withdrawal severity from an acute dose of EtOH. These experiments represent important elucidation of factors involved in neuroadaptation and neuroexcitability from a high dose of EtOH. Notably, recent work indicated that acute (single dose) and chronic withdrawal have similar effects on GABAA receptor plasticity, with differences in the persistence of these changes (Spigelman et al., 2007). These findings lend support to the idea that examination of neuroadaptation following withdrawal from a single high dose of ethanol can provide insight regarding neuroadaptaion following chronic ethanol withdrawal. Therefore, neuroactive steroid synthesis may represent a potential important site for therapeutic intervention in the treatment of alcohol dependence. The interaction of ethanol and neuroactive steroids warrants further investigation.
Acknowledgments
The support for this work was contributed by AA10760, AA12439, 5-T32- DA07262-14, the Tartar Fellowship and theDepartment of Veterans Affairs.
This research was supported by USPHS grants AA10760 and AA12439 from the National Institute on Alcohol Abuse and Alcoholism (NIAAA), a training grant stipend from 5-T32-DA07262-14, an NL Tartar Fellowship and the Department of Veterans Affairs. I also thank Amy Beadles- Bohling, PhD, Naomi Yoneyama, Michelle Tanchuck, Moriah Strong, and Christopher Snelling for their significant technical assistance, Debra Hickman, DVM for her patience while teaching me surgical techniques, and Kathleen Grant, PhD for her assistance in preparation of this manuscript and invaluable mentorship skills.
Contributor Information
Katherine R. Gililland, Department of Behavioral Neuroscience, Oregon Health & Science University, Portland, Oregon
Deborah A. Finn, Portland Alcohol Research Center, Department of Veterans Affairs Medical Center, Portland, Oregon
References
- Barbaccia ML, Affricano D, Trabucchi M, Purdy RH, Colombo G, Agabio R, Gessa GL. Ethanol markedly increases “GABAergic” neurosteroids in alcohol-preferring rats. Eur J Pharmacol. 1999;384:R1–R2. doi: 10.1016/s0014-2999(99)00678-0. [DOI] [PubMed] [Google Scholar]
- Baulieu EE. Neurosteroids: a novel function of the brain. Psychoneuroendocrinology. 1998;23:963–987. doi: 10.1016/s0306-4530(98)00071-7. [DOI] [PubMed] [Google Scholar]
- Becker HC, Anton RF, De Trana C, Randall CL. Sensitivity to ethanol in female mice: effects of ovariectomy and strain. Life Sci. 1985;37:1293–1300. doi: 10.1016/0024-3205(85)90244-9. [DOI] [PubMed] [Google Scholar]
- Beers M, Berkow R. The Merk Manual. 2. Merck & Co; White-house Station, NJ: 2005. [Google Scholar]
- Belelli D, Bolger MB, Gee KW. Anticonvulsant profile of the progesterone metabolite 5 α-pregnan-3 α-ol-20-one. Eur J Pharmacol. 1989;166:325–329. doi: 10.1016/0014-2999(89)90077-0. [DOI] [PubMed] [Google Scholar]
- Belelli D, Lan NC, Gee KW. Anticonvulsant steroids and the GABA/benzodiazepine receptor-chloride ionophore complex. Neurosci Biobehav Rev. 1990;14:315–322. doi: 10.1016/s0149-7634(05)80041-7. [DOI] [PubMed] [Google Scholar]
- Budec M, Koko V, Milovanović T, Balint-Perić L, Petković A. Acute ethanol treatment increases level of progesterone in ovariectomized rats. Alcohol. 2002;26:173–178. doi: 10.1016/s0741-8329(02)00197-0. [DOI] [PubMed] [Google Scholar]
- Crabbe JC. Provisional mapping of quantitative trait loci for chronic ethanol withdrawal severity in BXD recombinant inbred mice. J Pharmacol Exp Ther. 1998;286:263–271. [PubMed] [Google Scholar]
- Crabbe JC, Merrill CD, Belknap JK. Effects of convulsants on handling- induced convulsions in mice selected for ethanol withdrawal severity. Brain Res. 1991;550:1–6. doi: 10.1016/0006-8993(91)90397-e. [DOI] [PubMed] [Google Scholar]
- Crabbe JC, Young ER, Kosobud A. Genetic correlations with ethanol withdrawal severity. Pharmacol Biochem Behav. 1983;18(Suppl 1):541–547. doi: 10.1016/0091-3057(83)90233-2. [DOI] [PubMed] [Google Scholar]
- Finn DA, Ford MM, Wiren KM, Roselli CE, Crabbe JC. The role of pregnane neurosteroids in ethanol withdrawal: behavioral genetic approaches. Pharmacol Ther. 2004a;101:91–112. doi: 10.1016/j.pharmthera.2003.10.006. [DOI] [PubMed] [Google Scholar]
- Finn DA, Gallaher EJ, Crabbe JC. Differential change in neuroactive steroid sensitivity during ethanol withdrawal. J Pharmacol Exp Ther. 2000;292:394–405. [PubMed] [Google Scholar]
- Finn DA, Gee KW. The estrus cycle, sensitivity to convulsants and the anticonvulsant effect of a neuroactive steroid. J Pharmacol Exp Ther. 1994;271:164–170. [PubMed] [Google Scholar]
- Finn DA, Roberts AJ, Lotrich F, Gallaher EJ. Genetic differences in behavioral sensitivity to a neuroactive steroid. J Pharmacol Exp Ther. 1997;280:820–828. [PubMed] [Google Scholar]
- Finn DA, Sinnott RS, Ford MM, Long SL, Tanchuck MA, Phillips TJ. Sex differences in the effect of ethanol injection and consumption on brain allopregnanolone levels in C57BL/6 mice. Neuroscience. 2004b;123:813–819. doi: 10.1016/j.neuroscience.2003.11.017. [DOI] [PubMed] [Google Scholar]
- Finn DA, Snelling C, Fretwell AM, Tanchuck MA, Underwood L, Cole M, Crabbe JC, Roberts AJ. Increased drinking during withdrawal from intermittent ethanol exposure is blocked by the CRF receptor antagonist d- Phe-CRF(12–41) Alcohol Clin Exp Res. 2007;31:939–949. doi: 10.1111/j.1530-0277.2007.00379.x. [DOI] [PubMed] [Google Scholar]
- Follesa P, Biggio F, Talani G, Murru L, Serra M, Sanna E, Biggio G. Neurosteroids, GABAA receptors, and ethanol dependence. Psychopharmacology. 2006;186:267–280. doi: 10.1007/s00213-005-0126-0. [DOI] [PubMed] [Google Scholar]
- Ford MM, Nickel JD, Eddy S, Beckley EH, Finn DA. Ethanol intake patterns in female C57BL/6J mice: influence of allopregnanolone and the inhibition of its synthesis. Alcohol Clin Exp Res. 2007;31:26A. doi: 10.1016/j.drugalcdep.2008.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ford MM, Nickel JD, Finn DA. Treatment with and withdrawal from finasteride alters ethanol intake patterns in male C57BL/6J mice: potential role of endogenous neurosteroids? Alcohol. 2005;37:23–33. doi: 10.1016/j.alcohol.2005.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frye CA, Reed TA. Androgenic neurosteroids: anti-seizure effects in an animal model of epilepsy. Psychoneuroendocrinology. 1998;23:385–399. doi: 10.1016/s0306-4530(98)00009-2. [DOI] [PubMed] [Google Scholar]
- Frye CA, Rhodes ME, Walf A, Harney J. Progesterone reduces pentylenetetrazol- induced ictal activity of wild-type mice but not those deficient in type I 5 α-reductase. Epilepsia. 2002;43(Suppl 5):14–17. doi: 10.1046/j.1528-1157.43.s.5.19.x. [DOI] [PubMed] [Google Scholar]
- Gasior M, Carter RB, Witkin JM. Neuroactive steroids: potential therapeutic use in neurological and psychiatric disorders. Trends Pharmacol Sci. 1999;20:107–112. doi: 10.1016/s0165-6147(99)01318-8. [DOI] [PubMed] [Google Scholar]
- Gee KW. Steroid modulation of the GABA/benzodiazepine receptor-linked chloride ionophore. Mol Neurobiol. 1988;2:291–317. doi: 10.1007/BF02935636. [DOI] [PubMed] [Google Scholar]
- Gorin-Meyer RE, Wiren KM, Tanchuck MA, Long SL, Yoneyama N, Finn DA. Sex differences in the effect of finasteride on acute ethanol withdrawal severity in C57BL/6J and DBA/2J mice. Neuroscience. 2007;146:1302–1315. doi: 10.1016/j.neuroscience.2007.02.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grobin AC, Matthews DB, Devaud LL, Morrow AL. The role of GABAA receptors in the acute and chronic effects of ethanol. Psychopharmacology. 1998;139:2–19. doi: 10.1007/s002130050685. [DOI] [PubMed] [Google Scholar]
- Harkness J, Wagner J. Chapter 3: clinical procedures, in The Biology and Medicine of Rabbits and Rodents. 4. Williams and Wilkins, Media; PA: 1995. p. 126. [Google Scholar]
- Hirani K, Khisti RT, Chopde CT. Behavioral action of ethanol in Porsolt’s forced swim test: modulation by 3alpha-hydroxy-5alpha-pregnan-20- one. Neuropharmacology. 2002;43:1339–1350. doi: 10.1016/s0028-3908(02)00330-1. [DOI] [PubMed] [Google Scholar]
- Hirani K, Sharma AN, Jain NS, Ugale RR, Chopde CT. Evaluation of GABAergic neuroactive steroid 3α-hydroxy-5α-pregnane-20-one as a neurobiological substrate for the anti-anxiety effect of ethanol in rats. Psychopharmacology. 2005;180:267–278. doi: 10.1007/s00213-005-2169-7. [DOI] [PubMed] [Google Scholar]
- Holzbauer M, Birmingham MK, De Nicola AF, Oliver JT. In vivo secretion of 3 α-hydroxy-5 α-pregnan-20-one, a potent anaesthetic steroid, by the adrenal gland of the rat. J Steroid Biochem. 1985;22:97–102. doi: 10.1016/0022-4731(85)90147-5. [DOI] [PubMed] [Google Scholar]
- Kamens HM, Burkhart-Kasch S, McKinnon CS, Li N, Reed C, Phillips TJ. Ethanol-related traits in mice selectively bred for differential sensitivity to methamphetamine-induced activation. Behav Neurosci. 2006;120:1356–1366. doi: 10.1037/0735-7044.120.6.1356. [DOI] [PubMed] [Google Scholar]
- Karst H, de Kloet ER, Joels M. Episodic corticosterone treatment accelerates kindling epileptogenesis and triggers long-term changes in hippocampal CA1 cells, in the fully kindled state. Eur J Neurosci. 1999;11:889–898. doi: 10.1046/j.1460-9568.1999.00495.x. [DOI] [PubMed] [Google Scholar]
- Khisti RT, VanDoren MJ, O’Buckley T, Morrow AL. Neuroactive steroid 3α-hydroxy-5α-pregnan-20-one modulates ethanol-induced loss of righting reflex in rats. Brain Res. 2003;980:255–265. doi: 10.1016/s0006-8993(03)02978-0. [DOI] [PubMed] [Google Scholar]
- Krahl SE, Senanayake SS, Handforth A. Seizure suppression by systemic epinephrine is mediated by the vagus nerve. Epilepsy Res. 2000;38:171–175. doi: 10.1016/s0920-1211(99)00089-3. [DOI] [PubMed] [Google Scholar]
- Lamblin T, Meert F, Witte PD. Adrenalectomy protects ethanol-withdrawn rats from harmine-induced tremor. Alcohol Alcohol. 1996;31:175–181. doi: 10.1093/oxfordjournals.alcalc.a008129. [DOI] [PubMed] [Google Scholar]
- Lonsdale D, Burnham WM. The anticonvulsant effects of progesterone and 5α-dihydroprogesterone on amygdala-kindled seizures in rats. Epilepsia. 2003;44:1494–1499. doi: 10.1111/j.0013-9580.2003.59402.x. [DOI] [PubMed] [Google Scholar]
- Mejias-Aponte CA, Jimenez-Rivera CA, Segarra AC. Sex differences in models of temporal lobe epilepsy: role of testosterone. Brain Res. 2002;944:210–218. doi: 10.1016/s0006-8993(02)02691-4. [DOI] [PubMed] [Google Scholar]
- Mellon SH, Griffin LD. Neurosteroids: biochemistry and clinical significance. Trends Endocrinol Metab. 2002a;13:35–43. doi: 10.1016/s1043-2760(01)00503-3. [DOI] [PubMed] [Google Scholar]
- Mellon SH, Griffin LD. Synthesis, regulation, and function of neurosteroids. Endocr Res. 2002b;28:463. doi: 10.1081/erc-120016823. [DOI] [PubMed] [Google Scholar]
- Metten P, Crabbe JC. Alcohol withdrawal severity in inbred mouse (Mus musculus) strains. Behav Neurosci. 2005;119:911–925. doi: 10.1037/0735-7044.119.4.911. [DOI] [PubMed] [Google Scholar]
- Mezey E, Potter JJ, Harmon SM, Tsitouras PD. Effects of castration and testosterone administration on rat liver alcohol dehydrogenase activity. Biochem Pharmacol. 1980;29:3175. doi: 10.1016/0006-2952(80)90582-1. [DOI] [PubMed] [Google Scholar]
- Morrow AL, Janis GC, VanDoren MJ, Matthews DB, Samson HH, Janak PH, Grant KA. Neurosteroids mediate pharmacological effects of ethanol: a new mechanism of ethanol action? Alcohol Clin Exp Res. 1999;23:1933–1940. doi: 10.1111/j.1530-0277.1999.tb04094.x. [DOI] [PubMed] [Google Scholar]
- Morrow AL, Suzdak PD, Paul SM. Steroid hormone metabolites potentiate GABA receptor-mediated chloride ion flux with nanomolar potency. Eur J Pharmacol. 1987;142:483–485. doi: 10.1016/0014-2999(87)90094-x. [DOI] [PubMed] [Google Scholar]
- O’Dell LE, Alomary AA, Vallee M, Koob GF, Fitzgerald RL, Purdy RH. Ethanol-induced increases in neuroactive steroids in the rat brain and plasma are absent in adrenalectomized and gonadectomized rats. Eur J Pharmacol. 2004;484:241–247. doi: 10.1016/j.ejphar.2003.11.031. [DOI] [PubMed] [Google Scholar]
- Paul SM, Purdy RH. Neuroactive steroids. FASEB J. 1992;6:2311–2322. [PubMed] [Google Scholar]
- Powis G, Cummings J, Morgan E. The effect of adrenalectomy upon the absorption, distribution and metabolism of ethanol in the rat. Life Sci. 1977;21:1033–1036. doi: 10.1016/0024-3205(77)90271-5. [DOI] [PubMed] [Google Scholar]
- Purdy RH, Moore PH, Jr, Morrow AL, Paul SM. Neurosteroids and GABAA receptor function. Adv Biochem Psychopharmacol. 1992;47:87–92. [PubMed] [Google Scholar]
- Reddy DS. Anticonvulsant activity of the testosterone-derived neurosteroid 3α-androstanediol. Neuroreport. 2004a;15:515–518. doi: 10.1097/00001756-200403010-00026. [DOI] [PubMed] [Google Scholar]
- Reddy DS. Testosterone modulation of seizure susceptibility is mediated by neurosteroids 3α-androstanediol and 17β-estradiol. Neuroscience. 2004b;129:195–207. doi: 10.1016/j.neuroscience.2004.08.002. [DOI] [PubMed] [Google Scholar]
- Reddy DS, Rogawski MA. Stress-induced deoxycorticosterone-derived neurosteroids modulate GABAA receptor function and seizure susceptibility. J Neurosci. 2002;22:3795–3805. doi: 10.1523/JNEUROSCI.22-09-03795.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ricordi C, Shah SD, Lacy PE, Clutter WE, Cryer PE. Delayed extra-adrenal epinephrine secretion after bilateral adrenalectomy in rats. Am J Physiol Endocrinol Metab. 1988;254:E52–E53. doi: 10.1152/ajpendo.1988.254.1.E52. [DOI] [PubMed] [Google Scholar]
- Roberts AJ, Crabbe JC, Keith LD. Genetic differences in hypothalamic- pituitary-adrenal axis responsiveness to acute ethanol and acute ethanol withdrawal. Brain Res. 1992;579:296–302. doi: 10.1016/0006-8993(92)90064-g. [DOI] [PubMed] [Google Scholar]
- Roberts AJ, Crabbe JC, Keith LD. Corticosterone increases severity of acute withdrawal from ethanol, pentobarbital, and diazepam in mice. Psychopharmacology. 1994;115:278–284. doi: 10.1007/BF02244784. [DOI] [PubMed] [Google Scholar]
- Romeo E, Brancati A, de Lorenzo A, Fucci P, Furnari C, Pompili E, Sasso GF, Spalletta G, Troisi A, Pasini A. Marked decrease of plasma neuroactive steroids during alcohol withdrawal. Clin Neuropharmacol. 1996;19:366–369. doi: 10.1097/00002826-199619040-00011. [DOI] [PubMed] [Google Scholar]
- Rupprecht R, Holsboer F. Neuropsychopharmacological properties of neuroactive steroids. Steroids. 1999;64:83–91. doi: 10.1016/s0039-128x(98)00101-9. [DOI] [PubMed] [Google Scholar]
- Sanna E, Talani G, Busonero F, Pisu MG, Purdy RH, Serra M, Biggio G. Brain steroidogenesis mediates ethanol modulation of GABAA receptor activity in rat hippocampus. J Neurosci. 2004;24:6521–6530. doi: 10.1523/JNEUROSCI.0075-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santibañez M, Gysling K, Forray MI. Adrenalectomy decreases corticotropin- releasing hormone gene expression and increases noradrenaline and dopamine extracellular levels in the rat lateral bed nucleus of the stria terminalis. J Neurosci Res. 2005;81:140–152. doi: 10.1002/jnr.20538. [DOI] [PubMed] [Google Scholar]
- Schwartz-Giblin S, Korotzer A, Pfaff DW. Steroid hormone effects on picrotoxin-induced seizures in female and male rats. Brain Res. 1989;476:240–247. doi: 10.1016/0006-8993(89)91244-4. [DOI] [PubMed] [Google Scholar]
- Seyle H. The antagonism between anesthetic steroid hormones and pentamethylenetetrazol (metrazol) J Lab Clin Med. 1942;27:1051–1053. [Google Scholar]
- Shen EH, Harland RD, Crabbe JC, Phillips TJ. Bidirectional selective breeding for ethanol effects on locomotor activity: characterization of FAST and SLOW mice through selection generation 35. Alcohol Clin Exp Res. 1995;19:1234–1245. doi: 10.1111/j.1530-0277.1995.tb01606.x. [DOI] [PubMed] [Google Scholar]
- Sinnott RS, Phillips TJ, Finn DA. Alteration of voluntary ethanol and saccharin consumption by the neurosteroid allopregnanolone in mice. Psychopharmacology. 2002;162:438–447. doi: 10.1007/s00213-002-1123-1. [DOI] [PubMed] [Google Scholar]
- Spigelman I, Liang J, Suyanarayanon A, Olsen RW, Snyder B, Abrian A. Contrasting effect of chronic intermittent ethanol treatment vs. single dosing on GABAA receptor plasticity. Alcohol Clin Exp Res. 2007;31:315A. [Google Scholar]
- Sze PY, Yanai J, Ginsburg BE. Adrenal glucocorticoids as a required factor in the development of ethanol withdrawal seizures in mice. Brain Res. 1974;80:155–159. doi: 10.1016/0006-8993(74)90735-5. [DOI] [PubMed] [Google Scholar]
- Turner C, Babnara J. General Endocrinology. 6. W.B. Saunders Compnay; Philadelphia, PA: 1976. [Google Scholar]
- VanDoren MJ, Matthews DB, Janis GC, Grobin AC, Devaud LL, Morrow AL. Neuroactive steroid 3α-hydroxy-5α-pregnan-20-one modulates electrophysiological and behavioral actions of ethanol. J Neurosci. 2000;20:1982–1989. doi: 10.1523/JNEUROSCI.20-05-01982.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Veliskova J. Estrogens and epilepsy: why are we so excited? Neuroscientist. 2007;13:77–88. doi: 10.1177/1073858406295827. [DOI] [PubMed] [Google Scholar]
- Wallis CJ, Anton RF, Randall CL. Adrenalectomy reduces alcohol-stimulated activity: blood and brain alcohol content. Pharmacol Biochem Behav. 1984;20:883–886. doi: 10.1016/0091-3057(84)90012-1. [DOI] [PubMed] [Google Scholar]
- Welsh KA, Gold PE. Epinephrine proactive retardation of amygdala-kindled epileptogenesis. Behav Neurosci. 1986;100:236–245. doi: 10.1037//0735-7044.100.2.236. [DOI] [PubMed] [Google Scholar]