Abstract
OBJECTIVE
We investigated whether palmitoleate, which prevents insulin resistance in mice, predicts insulin sensitivity in humans.
RESEARCH DESIGN AND METHODS
The fasting fatty acid pattern in the plasma free fatty acid (FFA) fraction was determined in 100 subjects at increased risk for type 2 diabetes. Insulin sensitivity was estimated during an oral glucose tolerance test (OGTT) at baseline and after 9 months of lifestyle intervention and measured during the euglycemic-hyperinsulinemic clamp (n = 79).
RESULTS
Circulating palmitoleate (OGTT:F ratio = 8.2, P = 0.005; clamp:F ratio = 7.8, P = 0.007) but not total FFAs (OGTT:F ratio = 0.6, P = 0.42; clamp:F ratio = 0.7, P = 0.40) correlated positively with insulin sensitivity, independently of age, sex, and adiposity. High baseline palmitoleate predicted a larger increase in insulin sensitivity. For 1-SD increase in palmitoleate, the odds ratio for being in the highest versus the lowest tertile of adjusted change in insulin sensitivity was 2.35 (95% CI 1.16–5.35).
CONCLUSIONS
Circulating palmitoleate strongly and independently predicts insulin sensitivity, suggesting that it plays an important role in the pathophysiology of insulin resistance in humans.
Free fatty acids (FFAs) are considered to link obesity with insulin resistance and type 2 diabetes (1,2). Mechanisms include intracellular accumulation of lipotoxic metabolites, such as long-chain fatty acyl-CoA, ceramides, and diacylglycerol, which interfere with insulin signaling (2) and signaling via membrane toll-like receptor 4 (3). However, a significant relationship between total FFA levels and insulin resistance is not found in all studies (4–6).
Most recently, the fatty acid palmitoleate (C16:1n7) was found to increase insulin action in skeletal muscle and to prevent hepatosteatosis in mice, thus representing a link between adipose tissue and systemic metabolism (7). In the present study, we investigated whether palmitoleate may also be a determinant of insulin sensitivity in humans.
RESEARCH DESIGN AND METHODS
Data from 100 Caucasians of the Tübingen Lifestyle Intervention Program (8) were analyzed. Subjects underwent measurements at baseline and after 9 months of lifestyle intervention (≥3 h of moderate sports per week, intake of calories from fat <30%, intake of fibers ≥15 g/1,000 kcal, intake of saturated fat ≤10%). Informed written consent was obtained from all participants, and an ethics committee approved the protocol. Body fat was measured by bioelectrical impedance, total and visceral fat by magnetic resonance tomography, and liver fat by 1H magnetic resonance spectroscopy (8). Insulin sensitivity was estimated from a 75-g oral glucose tolerance test (OGTT), as proposed by Matsuda and DeFronzo [10,000/√(Insmean · Glucmean · Ins0 · Gluc0)] and additionally measured by the euglycemic clamp (8). The fatty acids in the plasma FFA fraction were measured as previously described (supplemental Table 1 in the online appendix at http://care.diabetesjournals.org/cgi/content/full/dc09-0544/DC1) (9). Forward stepwise multivariate linear and logistic regressions were performed.
RESULTS
Subject characteristics are shown in supplemental Table 2. At baseline, insulin sensitivity correlated inversely with body weight, BMI, waist circumference, total body fat, visceral fat, and liver fat (all P < 0.0001). Total fasting FFA levels were lower in males (P = 0.0002). They were not associated with age or with measures of adiposity, liver fat (all adjusted P > 0.09), or insulin sensitivity (OGTT:F ratio = 0.6, P = 0.42; clamp:F ratio = 0.7; P = 0.40) independently of sex, age, and body fat.
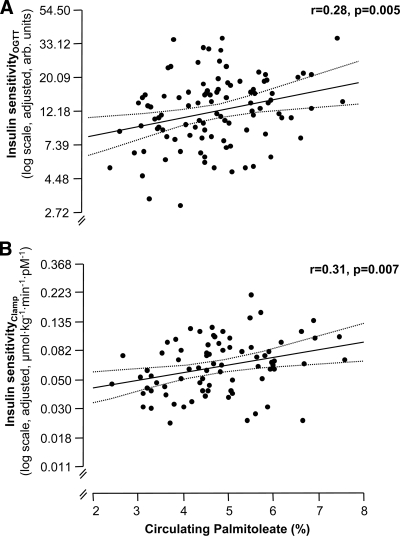
Circulating palmitoleate was lower in males (4.38 ± 0.19 vs. 5.03 ± 0.14%, P = 0.007) and not associated with age, or with measures of adiposity such as body weight, BMI, waist circumference, or total and visceral fat, adjusted for sex and age (all P > 0.23). Because palmitoleate in serum cholesteryl esters correlated with high-sensitivity C-reactive protein (hsCRP) levels (10), we also investigated the relationship between circulating palmitoleate and adjusted hsCRP levels. No significant relationship was found (P = 0.44). A weak negative correlation between palmitoleate and liver fat was observed after adjustment for age, sex, and body fat (F ratio = 3.7; P = 0.057). In contrast, palmitoleate correlated positively with insulin sensitivity (OGTT:F Ratio = 8.2, Fig. 1A; clamp:F ratio = 7.8, Fig. 1B) independently of sex, age, and body fat. Additional adjustment for visceral fat only moderately affected these relationships (P = 0.017 and P = 0.019).
Figure 1.
Cross-sectional relationships of circulating palmitoleate with insulin sensitivity estimated from the OGTT (A) and measured by the clamp (B) at baseline. Insulin sensitivity was adjusted for age, sex, and body fat in multivariate linear regression models (regression line and 95% CI). arb. units, arbitrary units.
During the 9 months of lifestyle intervention, insulin sensitivity and circulating palmitoleate increased (P < 0.0001, supplemental Tables 1 and 2). Change in palmitoleate did not correlate with changes in insulin sensitivity in all subjects (n = 95, OGTT P = 0.67, and n = 38, clamp P = 0.56). However, a positive relationship was observed in subjects in the upper tertile of insulin sensitivity (OGTT) at baseline (r = 0.41, P = 0.03), suggesting that in subjects starting with low insulin sensitivity, the multiple benefits of lifestyle interaction such as reduction in adiposity or increased exercise intensity have a stronger impact on insulin sensitivity than change in palmitoleate levels. In agreement with the analyses at baseline, at the follow-up visit, palmitoleate levels also correlated positively with adjusted insulin sensitivity (n = 95, OGTT:F ratio = 5.0; P = 0.029).
In forward stepwise linear regression analyses including palmitoleate at baseline, age, sex, insulin sensitivity at baseline, and body fat at baseline and at follow-up, high palmitoleate levels predicted a larger increase in insulin sensitivity (P = 0.02, supplemental Table 3). After dividing subjects into tertiles by the observed change in insulin sensitivity, for 1-SD increase in circulating palmitoleate at baseline, the odds ratio of subjects for being in the highest versus the lowest tertile of change in insulin sensitivity was 2.35 (95% CI 1.16–5.35).
CONCLUSIONS
We found that circulating palmitoleate was a determinant of insulin sensitivity both estimated from the OGTT and measured by the clamp. Furthermore, in subjects with high palmitoleate at baseline, there was a higher chance to observe an increase in insulin sensitivity, independently of the change in adiposity, compared with subjects with low levels. These novel data strongly support that palmitoleate may also be involved in the regulation of insulin sensitivity in humans.
In animals, palmitoleate infusion decreased expression of hepatic lipogenic enzymes, thus possibly regulating liver fat (7), an important determinant of insulin sensitivity in humans (11,12). We observed a weak negative relationship between circulating palmitoleate and liver fat, suggesting that, in humans, the effects of palmitoleate on the regulation of insulin sensitivity may be more pronounced than on hepatic steatosis.
What mechanisms are involved in the determination of circulating palmitoleate? The lipid chaperons fatty acid–binding protein (FABP)-4 and -5 were found to suppress the biosynthesis of palmitoleate (7). FABPs induce lipolysis in adipocytes and inflammatory pathways in macrophages, particularly in visceral obesity (13). However, FABP expression was not elevated in visceral obesity (14) and, in the present study, palmitoleate levels did not correlate with visceral fat mass, suggesting that increase in this fat compartment does not appreciably determine palmitoleate levels. Another regulator of palmitoleate may be diet. However, dietary levels of palmitoleate are very low. Therefore, palmitoleate levels are most probably not largely affected by the diet, but mainly by de novo lipogenesis in adipose tissue (7). In addition, stearoyl-CoA desaturase-1, the key enzyme in the biosynthesis of palmitoleate from palmitate, may determine palmitoleate levels. This is supported by data showing that thiazolidinedione treatment increased insulin sensitivity, circulating palmitoleate, and expression and activity of stearoyl-CoA desaturase-1 in adipose tissue (15).
In conclusion, particularly the cross-sectional analyses with measurements of insulin sensitivity from both the OGTT and the clamp suggest that palmitoleate strongly and independently of adiposity determines insulin sensitivity. Therefore, circulating palmitoleate may also play an important role in the pathophysiology of insulin resistance in humans.
Supplementary Material
Acknowledgments
The study was supported by grants from the Deutsche Forschungsgemeinschaft (KFO 114 and a Heisenberg-Grant to N.S., STE 1096/1–1) and the European Community's FP6 EUGENE2 (LSHM-CT-2004-512013).
No potential conflicts of interest relevant to this article were reported.
The authors thank all participants for their cooperation.
Footnotes
The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. Section 1734 solely to indicate this fact.
References
- 1.Risérus U, Willett WC, Hu FB. Dietary fats and prevention of type 2 diabetes. Prog Lipid Res 2009; 48: 44– 51 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Savage DB, Petersen KF, Shulman GI. Disordered lipid metabolism and the pathogenesis of insulin resistance. Physiol Rev 2007; 87: 507– 520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Shi H, Kokoeva MV, Inouye K, Tzameli I, Yin H, Flier JS. TLR4 links innate immunity and fatty acid-induced insulin resistance. J Clin Invest 2006; 116: 3015– 3025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Perseghin G, Scifo P, De Cobelli F, Pagliato E, Battezzati A, Arcelloni C, Vanzulli A, Testolin G, Pozza G, Del Maschio A, Luzi L. Intramyocellular triglyceride content is a determinant of in vivo insulin resistance in humans: a 1H-13C nuclear magnetic resonance spectroscopy assessment in offspring of type 2 diabetic parents. Diabetes 1999; 48: 1600– 1606 [DOI] [PubMed] [Google Scholar]
- 5.Stefan N, Kantartzis K, Machann J, Schick F, Thamer C, Rittig K, Balletshofer B, Machicao F, Fritsche A, Häring HU. Identification and characterization of metabolically benign obesity in humans. Arch Intern Med 2008; 168: 1609– 1616 [DOI] [PubMed] [Google Scholar]
- 6.Roden M. How free fatty acids inhibit glucose utilization in human skeletal muscle. News Physiol Sci 2004; 19: 92– 96 [DOI] [PubMed] [Google Scholar]
- 7.Cao H, Gerhold K, Mayers JR, Wiest MM, Watkins SM, Hotamisligil GS. Identification of a lipokine, a lipid hormone linking adipose tissue to systemic metabolism. Cell 2008; 134: 933– 944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Stefan N, Hennige AM, Staiger H, Machann J, Schick F, Kröber SM, Machicao F, Fritsche A, Häring HU. Alpha2-Heremans-Schmid glycoprotein/fetuin-A is associated with insulin resistance and fat accumulation in the liver in humans. Diabetes Care 2006; 29: 853– 857 [DOI] [PubMed] [Google Scholar]
- 9.Stefan N, Wahl HG, Fritsche A, Haring H, Stumvoll M. Effect of the pattern of elevated free fatty acids on insulin sensitivity and insulin secretion in healthy humans. Horm Metab Res 2001; 33: 432– 438 [DOI] [PubMed] [Google Scholar]
- 10.Petersson H, Basu S, Cederholm T, Risérus U. Serum fatty acid composition and indices of stearoyl-CoA desaturase activity are associated with systemic inflammation: longitudinal analyses in middle-aged men. Br J Nutr 2008; 99: 1186– 1189 [DOI] [PubMed] [Google Scholar]
- 11.Stefan N, Kantartzis K, Häring HU. Causes and metabolic consequences of fatty liver. Endocr Rev 2008; 29: 939– 960 [DOI] [PubMed] [Google Scholar]
- 12.Roden M. Mechanisms of disease: hepatic steatosis in type 2 diabetes–pathogenesis and clinical relevance. Nat Clin Pract Endocrinol Metab 2006; 2: 335– 348 [DOI] [PubMed] [Google Scholar]
- 13.Furuhashi M, Hotamisligil GS. Fatty acid-binding proteins: role in metabolic diseases and potential as drug targets. Nat Rev Drug Discov 2008; 7: 489– 503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fisher RM, Eriksson P, Hoffstedt J, Hotamisligil GS, Thörne A, Rydén M, Hamsten A, Arner P. Fatty acid binding protein expression in different adipose tissue depots from lean and obese individuals. Diabetologia 2001; 44: 1268– 1273 [DOI] [PubMed] [Google Scholar]
- 15.Risérus U, Tan GD, Fielding BA, Neville MJ, Currie J, Savage DB, Chatterjee VK, Frayn KN, O'Rahilly S, Karpe F. Rosiglitazone increases indexes of stearoyl-CoA desaturase activity in humans: link to insulin sensitization and the role of dominant-negative mutation in peroxisome proliferator-activated receptor-gamma. Diabetes 2005; 54: 1379– 1384 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.