Abstract
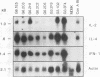
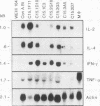
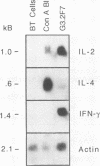
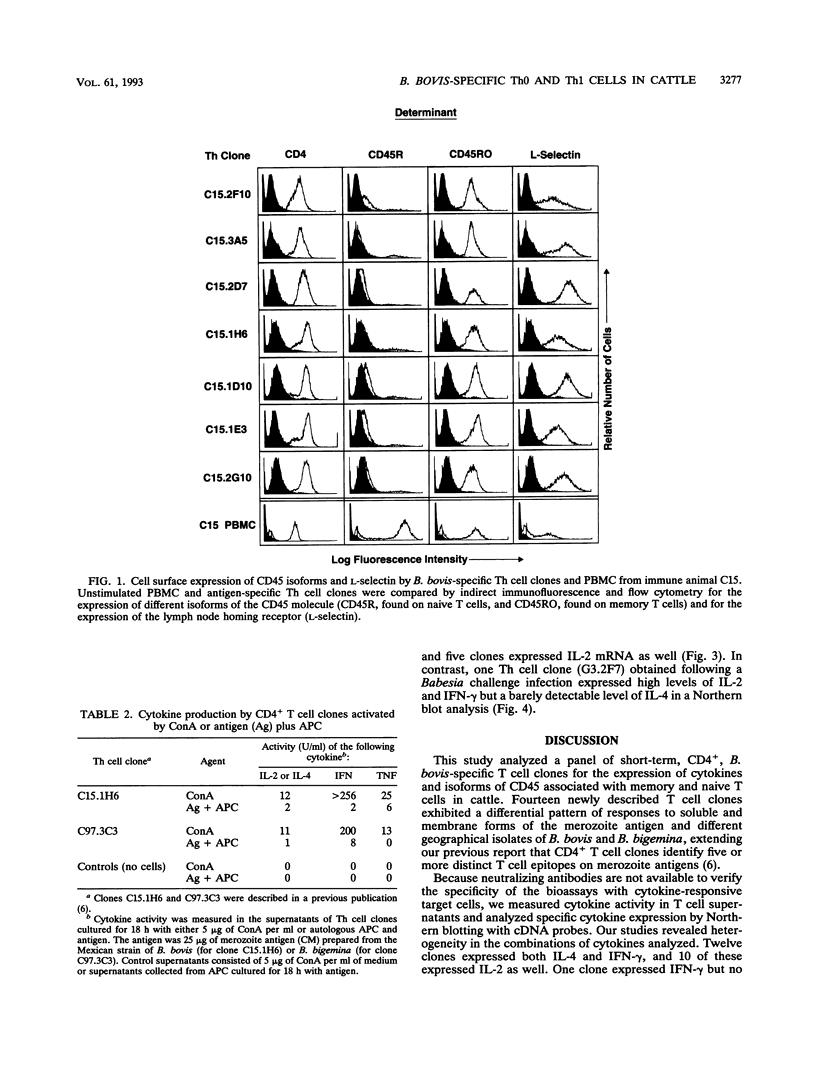
The central role of T cells in the immune response against hemoprotozoan parasites, both as helper cells for T cell-dependent antibody production and as effector cells acting on intracellular parasites through the elaboration of cytokines, has prompted an investigation of the bovine cellular immune response against Babesia bovis antigens. CD4+ T helper (Th) cell clones generated from four B. bovis-immune cattle by in vitro stimulation with a soluble or membrane-associated merozoite antigen were characterized for reactivity against various forms of antigen and against different geographical isolates of B. bovis and B. bigemina and analyzed for cytokine production following mitogenic stimulation with concanavalin A. Biological assays to measure interleukin-2 (IL-2), IL-4, gamma interferon (IFN-gamma), and tumor necrosis factor alpha or tumor necrosis factor beta and Northern (RNA) blot analysis to verify the expression of IL-2, IL-4, IFN-gamma, and tumor necrosis factor alpha revealed differential production of cytokines by the Th cell clones. The majority of clones expressed the Th0 pattern of cytokines: IFN-gamma, IL-4, and IL-2. One clone expressed the Th1 profile (IFN-gamma and IL-2 but not IL-4), whereas none of the clones expressed the Th2 profile. All of the Th cell clones examined expressed the low-molecular-weight isoform of the leukocyte common antigen associated with a memory cell phenotype (CD45RO), and all expressed the lymph node homing receptor (L-selectin). These results extend our previous finding of differential cytokine expression by B. bovis-specific Th cell clones and confirm the identity of the specific cytokines produced, showing that a Th0 response is preferentially induced in a panel of 20 CD4+ T cell clones obtained from immune cattle.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brown W. C., Grab D. J. Biological and biochemical characterization of bovine interleukin 2. Studies with cloned bovine T cells. J Immunol. 1985 Nov;135(5):3184–3190. [PubMed] [Google Scholar]
- Brown W. C., Logan K. S. Babesia bovis: bovine helper T cell lines reactive with soluble and membrane antigens of merozoites. Exp Parasitol. 1992 Mar;74(2):188–199. doi: 10.1016/0014-4894(92)90046-d. [DOI] [PubMed] [Google Scholar]
- Brown W. C., Logan K. S., Wagner G. G., Tetzlaff C. L. Cell-mediated immune responses to Babesia bovis merozoite antigens in cattle following infection with tick-derived or cultured parasites. Infect Immun. 1991 Jul;59(7):2418–2426. doi: 10.1128/iai.59.7.2418-2426.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown W. C., Zhao S., Rice-Ficht A. C., Logan K. S., Woods V. M. Bovine helper T cell clones recognize five distinct epitopes on Babesia bovis merozoite antigens. Infect Immun. 1992 Oct;60(10):4364–4372. doi: 10.1128/iai.60.10.4364-4372.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown W. C., Zhao S., Woods V. M., Tripp C. A., Tetzlaff C. L., Heussler V. T., Dobbelaere D. A., Rice-Ficht A. C. Identification of two Th1 cell epitopes on the Babesia bovis-encoded 77-kilodalton merozoite protein (Bb-1) by use of truncated recombinant fusion proteins. Infect Immun. 1993 Jan;61(1):236–244. doi: 10.1128/iai.61.1.236-244.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavacini L. A., Parke L. A., Weidanz W. P. Resolution of acute malarial infections by T cell-dependent non-antibody-mediated mechanisms of immunity. Infect Immun. 1990 Sep;58(9):2946–2950. doi: 10.1128/iai.58.9.2946-2950.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colina K. F., Perler F. B., Matsumura I., Meda M., Nutman T. B. The identification of an Onchocerca-specific recombinant antigen containing a T cell epitope. J Immunol. 1990 Sep 1;145(5):1551–1556. [PubMed] [Google Scholar]
- Degen J. L., Neubauer M. G., Degen S. J., Seyfried C. E., Morris D. R. Regulation of protein synthesis in mitogen-activated bovine lymphocytes. Analysis of actin-specific and total mRNA accumulation and utilization. J Biol Chem. 1983 Oct 25;258(20):12153–12162. [PubMed] [Google Scholar]
- Del Prete G. F., De Carli M., Mastromauro C., Biagiotti R., Macchia D., Falagiani P., Ricci M., Romagnani S. Purified protein derivative of Mycobacterium tuberculosis and excretory-secretory antigen(s) of Toxocara canis expand in vitro human T cells with stable and opposite (type 1 T helper or type 2 T helper) profile of cytokine production. J Clin Invest. 1991 Jul;88(1):346–350. doi: 10.1172/JCI115300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Del Prete G., Maggi E., Parronchi P., Chrétien I., Tiri A., Macchia D., Ricci M., Banchereau J., De Vries J., Romagnani S. IL-4 is an essential factor for the IgE synthesis induced in vitro by human T cell clones and their supernatants. J Immunol. 1988 Jun 15;140(12):4193–4198. [PubMed] [Google Scholar]
- Ellis J. A., Davis W. C., MacHugh N. D., Emery D. L., Kaushal A., Morrison W. I. Differentiation antigens on bovine mononuclear phagocytes identified by monoclonal antibodies. Vet Immunol Immunopathol. 1988 Oct;19(3-4):325–340. doi: 10.1016/0165-2427(88)90118-3. [DOI] [PubMed] [Google Scholar]
- Firestein G. S., Roeder W. D., Laxer J. A., Townsend K. S., Weaver C. T., Hom J. T., Linton J., Torbett B. E., Glasebrook A. L. A new murine CD4+ T cell subset with an unrestricted cytokine profile. J Immunol. 1989 Jul 15;143(2):518–525. [PubMed] [Google Scholar]
- Goeddel D. V., Aggarwal B. B., Gray P. W., Leung D. W., Nedwin G. E., Palladino M. A., Patton J. S., Pennica D., Shepard H. M., Sugarman B. J. Tumor necrosis factors: gene structure and biological activities. Cold Spring Harb Symp Quant Biol. 1986;51(Pt 1):597–609. doi: 10.1101/sqb.1986.051.01.072. [DOI] [PubMed] [Google Scholar]
- Heussler V. T., Eichhorn M., Dobbelaere D. A. Cloning of a full-length cDNA encoding bovine interleukin 4 by the polymerase chain reaction. Gene. 1992 May 15;114(2):273–278. doi: 10.1016/0378-1119(92)90587-f. [DOI] [PubMed] [Google Scholar]
- Howard C. J., Sopp P., Parsons K. R. L-selectin expression differentiates T cells isolated from different lymphoid tissues in cattle but does not correlate with memory. Immunology. 1992 Oct;77(2):228–234. [PMC free article] [PubMed] [Google Scholar]
- Howard C. J., Sopp P., Parsons K. R., McKeever D. J., Taracha E. L., Jones B. V., MacHugh N. D., Morrison W. I. Distinction of naive and memory BoCD4 lymphocytes in calves with a monoclonal antibody, CC76, to a restricted determinant of the bovine leukocyte-common antigen, CD45. Eur J Immunol. 1991 Sep;21(9):2219–2226. doi: 10.1002/eji.1830210933. [DOI] [PubMed] [Google Scholar]
- Ju S. T., Ruddle N. H., Strack P., Dorf M. E., DeKruyff R. H. Expression of two distinct cytolytic mechanisms among murine CD4 subsets. J Immunol. 1990 Jan 1;144(1):23–31. [PubMed] [Google Scholar]
- Kelso A., Troutt A. B., Maraskovsky E., Gough N. M., Morris L., Pech M. H., Thomson J. A. Heterogeneity in lymphokine profiles of CD4+ and CD8+ T cells and clones activated in vivo and in vitro. Immunol Rev. 1991 Oct;123:85–114. doi: 10.1111/j.1600-065x.1991.tb00607.x. [DOI] [PubMed] [Google Scholar]
- Kumaratilake L. M., Ferrante A. IL-4 inhibits macrophage-mediated killing of Plasmodium falciparum in vitro. A possible parasite-immune evasion mechanism. J Immunol. 1992 Jul 1;149(1):194–199. [PubMed] [Google Scholar]
- Kumaratilake L. M., Ferrante A., Rzepczyk C. The role of T lymphocytes in immunity to Plasmodium falciparum. Enhancement of neutrophil-mediated parasite killing by lymphotoxin and IFN-gamma: comparisons with tumor necrosis factor effects. J Immunol. 1991 Jan 15;146(2):762–767. [PubMed] [Google Scholar]
- Lahesmaa R., Yssel H., Batsford S., Luukkainen R., Möttönen T., Steinman L., Peltz G. Yersinia enterocolitica activates a T helper type 1-like T cell subset in reactive arthritis. J Immunol. 1992 May 15;148(10):3079–3085. [PubMed] [Google Scholar]
- Langhorne J., Meding S. J., Eichmann K., Gillard S. S. The response of CD4+ T cells to Plasmodium chabaudi chabaudi. Immunol Rev. 1989 Dec;112:71–94. doi: 10.1111/j.1600-065x.1989.tb00553.x. [DOI] [PubMed] [Google Scholar]
- Liew F. Y., Millott S. M., Schmidt J. A. A repetitive peptide of Leishmania can activate T helper type 2 cells and enhance disease progression. J Exp Med. 1990 Nov 1;172(5):1359–1365. doi: 10.1084/jem.172.5.1359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mackay C. R. T-cell memory: the connection between function, phenotype and migration pathways. Immunol Today. 1991 Jun;12(6):189–192. doi: 10.1016/0167-5699(91)90051-T. [DOI] [PubMed] [Google Scholar]
- Maggi E., Del Prete G., Macchia D., Parronchi P., Tiri A., Chrétien I., Ricci M., Romagnani S. Profiles of lymphokine activities and helper function for IgE in human T cell clones. Eur J Immunol. 1988 Jul;18(7):1045–1050. doi: 10.1002/eji.1830180712. [DOI] [PubMed] [Google Scholar]
- Maggi E., Parronchi P., Manetti R., Simonelli C., Piccinni M. P., Rugiu F. S., De Carli M., Ricci M., Romagnani S. Reciprocal regulatory effects of IFN-gamma and IL-4 on the in vitro development of human Th1 and Th2 clones. J Immunol. 1992 Apr 1;148(7):2142–2147. [PubMed] [Google Scholar]
- Mosmann T. R., Cherwinski H., Bond M. W., Giedlin M. A., Coffman R. L. Two types of murine helper T cell clone. I. Definition according to profiles of lymphokine activities and secreted proteins. J Immunol. 1986 Apr 1;136(7):2348–2357. [PubMed] [Google Scholar]
- Mosmann T. R., Coffman R. L. TH1 and TH2 cells: different patterns of lymphokine secretion lead to different functional properties. Annu Rev Immunol. 1989;7:145–173. doi: 10.1146/annurev.iy.07.040189.001045. [DOI] [PubMed] [Google Scholar]
- Paliard X., de Waal Malefijt R., Yssel H., Blanchard D., Chrétien I., Abrams J., de Vries J., Spits H. Simultaneous production of IL-2, IL-4, and IFN-gamma by activated human CD4+ and CD8+ T cell clones. J Immunol. 1988 Aug 1;141(3):849–855. [PubMed] [Google Scholar]
- Palmer G. H., McElwain T. F., Perryman L. E., Davis W. C., Reduker D. R., Jasmer D. P., Shkap V., Pipano E., Goff W. L., McGuire T. C. Strain variation of Babesia bovis merozoite surface-exposed epitopes. Infect Immun. 1991 Sep;59(9):3340–3342. doi: 10.1128/iai.59.9.3340-3342.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parronchi P., Macchia D., Piccinni M. P., Biswas P., Simonelli C., Maggi E., Ricci M., Ansari A. A., Romagnani S. Allergen- and bacterial antigen-specific T-cell clones established from atopic donors show a different profile of cytokine production. Proc Natl Acad Sci U S A. 1991 May 15;88(10):4538–4542. doi: 10.1073/pnas.88.10.4538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reeves R., Spies A. G., Nissen M. S., Buck C. D., Weinberg A. D., Barr P. J., Magnuson N. S., Magnuson J. A. Molecular cloning of a functional bovine interleukin 2 cDNA. Proc Natl Acad Sci U S A. 1986 May;83(10):3228–3232. doi: 10.1073/pnas.83.10.3228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruddle N. H., Bergman C. M., McGrath K. M., Lingenheld E. G., Grunnet M. L., Padula S. J., Clark R. B. An antibody to lymphotoxin and tumor necrosis factor prevents transfer of experimental allergic encephalomyelitis. J Exp Med. 1990 Oct 1;172(4):1193–1200. doi: 10.1084/jem.172.4.1193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salgame P., Abrams J. S., Clayberger C., Goldstein H., Convit J., Modlin R. L., Bloom B. R. Differing lymphokine profiles of functional subsets of human CD4 and CD8 T cell clones. Science. 1991 Oct 11;254(5029):279–282. doi: 10.1126/science.254.5029.279. [DOI] [PubMed] [Google Scholar]
- Salmon M., Kitas G. D., Bacon P. A. Production of lymphokine mRNA by CD45R+ and CD45R- helper T cells from human peripheral blood and by human CD4+ T cell clones. J Immunol. 1989 Aug 1;143(3):907–912. [PubMed] [Google Scholar]
- Sanders M. E., Makgoba M. W., Shaw S. Human naive and memory T cells: reinterpretation of helper-inducer and suppressor-inducer subsets. Immunol Today. 1988 Jul-Aug;9(7-8):195–199. doi: 10.1016/0167-5699(88)91212-1. [DOI] [PubMed] [Google Scholar]
- Schofield L., Ferreira A., Altszuler R., Nussenzweig V., Nussenzweig R. S. Interferon-gamma inhibits the intrahepatocytic development of malaria parasites in vitro. J Immunol. 1987 Sep 15;139(6):2020–2025. [PubMed] [Google Scholar]
- Schofield L., Villaquiran J., Ferreira A., Schellekens H., Nussenzweig R., Nussenzweig V. Gamma interferon, CD8+ T cells and antibodies required for immunity to malaria sporozoites. Nature. 1987 Dec 17;330(6149):664–666. doi: 10.1038/330664a0. [DOI] [PubMed] [Google Scholar]
- Scott P., Natovitz P., Coffman R. L., Pearce E., Sher A. Immunoregulation of cutaneous leishmaniasis. T cell lines that transfer protective immunity or exacerbation belong to different T helper subsets and respond to distinct parasite antigens. J Exp Med. 1988 Nov 1;168(5):1675–1684. doi: 10.1084/jem.168.5.1675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevens M. G., Wilson L. K., Gershwin L. J. Isolation and characterization of an IL-1-dependent IL-4-producing bovine CD4+ T cell clone. Cell Immunol. 1992 Apr;140(2):453–467. doi: 10.1016/0008-8749(92)90211-7. [DOI] [PubMed] [Google Scholar]
- Stevenson M. M., Ghadirian E. Human recombinant tumor necrosis factor alpha protects susceptible A/J mice against lethal Plasmodium chabaudi AS infection. Infect Immun. 1989 Dec;57(12):3936–3939. doi: 10.1128/iai.57.12.3936-3939.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Street N. E., Schumacher J. H., Fong T. A., Bass H., Fiorentino D. F., Leverah J. A., Mosmann T. R. Heterogeneity of mouse helper T cells. Evidence from bulk cultures and limiting dilution cloning for precursors of Th1 and Th2 cells. J Immunol. 1990 Mar 1;144(5):1629–1639. [PubMed] [Google Scholar]
- Swain S. L., Bradley L. M., Croft M., Tonkonogy S., Atkins G., Weinberg A. D., Duncan D. D., Hedrick S. M., Dutton R. W., Huston G. Helper T-cell subsets: phenotype, function and the role of lymphokines in regulating their development. Immunol Rev. 1991 Oct;123:115–144. doi: 10.1111/j.1600-065x.1991.tb00608.x. [DOI] [PubMed] [Google Scholar]
- Takada S., Koide J., Engleman E. G. Differences in surface phenotype between cytolytic and non-cytolytic CD4+ T cells. MHC class II-specific cytotoxic T lymphocytes lack Leu 8 antigen and express CD2 in high density. J Immunol. 1989 May 1;142(9):3038–3044. [PubMed] [Google Scholar]
- Taverne J., Tavernier J., Fiers W., Playfair J. H. Recombinant tumour necrosis factor inhibits malaria parasites in vivo but not in vitro. Clin Exp Immunol. 1987 Jan;67(1):1–4. [PMC free article] [PubMed] [Google Scholar]
- Tedder T. F., Matsuyama T., Rothstein D., Schlossman S. F., Morimoto C. Human antigen-specific memory T cells express the homing receptor (LAM-1) necessary for lymphocyte recirculation. Eur J Immunol. 1990 Jun;20(6):1351–1355. doi: 10.1002/eji.1830200622. [DOI] [PubMed] [Google Scholar]
- Umetsu D. T., Jabara H. H., DeKruyff R. H., Abbas A. K., Abrams J. S., Geha R. S. Functional heterogeneity among human inducer T cell clones. J Immunol. 1988 Jun 15;140(12):4211–4216. [PubMed] [Google Scholar]
- Vitetta E. S., Berton M. T., Burger C., Kepron M., Lee W. T., Yin X. M. Memory B and T cells. Annu Rev Immunol. 1991;9:193–217. doi: 10.1146/annurev.iy.09.040191.001205. [DOI] [PubMed] [Google Scholar]
- Wright I. G., Goodger B. V., Clark I. A. Immunopathophysiology of Babesia bovis and Plasmodium falciparum infections. Parasitol Today. 1988 Aug;4(8):214–218. doi: 10.1016/0169-4758(88)90161-5. [DOI] [PubMed] [Google Scholar]
- Yssel H., Johnson K. E., Schneider P. V., Wideman J., Terr A., Kastelein R., De Vries J. E. T cell activation-inducing epitopes of the house dust mite allergen Der p I. Proliferation and lymphokine production patterns by Der p I-specific CD4+ T cell clones. J Immunol. 1992 Feb 1;148(3):738–745. [PubMed] [Google Scholar]