Abstract
We tested the hypothesis that endogenous N-acetylaspartylglutamate (NAAG) presynaptically inhibits glutamate release at mossy fiber-CA3 synapses. For this purpose, we made use of 2-(3-mercaptopropyl)pentanedioic acid (2-MPPA), an inhibitor of glutamate carboxypeptidase II [GCP II; also known as N-acetylated alpha-linked acidic dipeptidase (NAALADase)], the enzyme that hydrolyzes NAAG into N-acetylaspartate and glutamate. Application of 2-MPPA (1–20 μM) had no effect on intrinsic membrane properties of CA3 pyramidal neurons recorded in vitro in whole cell current- or voltage-clamp mode. Bath application of 10 μM 2-MPPA suppressed evoked excitatory postsynaptic current (EPSC) amplitudes. Attenuation of EPSC amplitudes was accompanied by a significant increase in paired-pulse facilitation (50-ms interpulse intervals), suggesting that a presynaptic mechanism is involved. The group II metabotropic glutamate receptor (mGluR) antagonist 2S-2-amino-2-(1S,2S-2-carboxycyclopropyl-1-yl)-3-(xanth-9-y l) propanoic acid (LY341495) prevented the 2-MPPA-dependent suppression of EPSC amplitudes. 2-MPPA reduced the frequencies of TTX-insensitive miniature EPSCs (mEPSC), without affecting their amplitudes, further supporting a presynaptic action for GCP II inhibition. 2-MPPA-induced reduction of mEPSC frequencies was prevented by LY341495, reinforcing the role of presynaptic group II mGluR. Because GCP II inhibition is thought to increase NAAG levels, these results suggest that NAAG suppresses synaptic transmission at mossy fiber-CA3 synapses through presynaptic activation of group II mGluRs.
INTRODUCTION
N-acetylaspartylglutamate (NAAG) is a neuropeptide ubiquitously distributed in millimolar concentrations in the mammalian brain (Coyle 1997; Neale et al. 2000). In the extracellular space, NAAG is enzymatically hydrolyzed into glutamate and N-acetylaspartate by glutamate carboxypeptidase II [GCP II; also known as N-acetylated alpha-linked acidic dipeptidase (NAALADase)] (Riveros and Orrego 1984; Stauch et al. 1989). This enzyme is expressed mainly in glial cells, where it is membrane bound (Cassidy and Neale 1993). Additional peptidases having a high affinity to NAAG were recently identified (Bacich et al. 2002). NAAG was originally considered to be an intermediate metabolite in the biosynthesis of glutamate (Coyle 1997; Curatolo et al. 1967). However, more recent data support a role for NAAG as a classical neurotransmitter (Coyle 1997; Neale et al. 2000). NAAG is concentrated in synaptic vesicles (Renno et al. 1997; Williamson and Neale 1988) and is co-localized with several neurotransmitters including GABA, glutamate, acetylcholine, and dopamine and is released in a calcium-dependent manner from synaptic terminals (Gafurov et al. 2001; Moffett et al. 1994; Tsai et al. 1993; Urazaev et al. 2001; Zollinger et al. 1988). NAAG binds to N-methyl-D-aspartate (NMDA) receptors (IC50: NAAG, 8.8 μM, vs. glutamate, 0.4 μM) but not to AMPA/kainate receptors (Valivullah et al. 1994). NAAG is a low-affinity agonist of the NMDA receptor (EC50: NAAG, 666 μM, vs. NMDA, 29 μM) at olfactory bulb neurons (Trombley and Westbrook 1990).
In contrast to its weak effects at NMDA receptors, NAAG binds to group II metabotropic glutamate receptors (mGluRs) with high affinity (IC50 <5 μM) (Schaffhauser et al. 1998; Shave et al. 2001). In transfected cell lines, NAAG activates the group II mGluR, mGluR3 (EC50: 65 μM for NAAG vs. 58 μM for trans-ACPD) (Wroblewska et al. 1997). Because activation of group II mGluRs results in presynaptic inhibition of transmitter release (Kamiya et al. 1996) NAAG may act as a potent presynaptic inhibitor. In support of this hypothesis are findings that exogenous NAAG reduces climbing fiber-mediated excitatory postsynaptic potentials (EPSPs) in Purkinje cells (Sekiguchi et al. 1989). In cortical cultures, exogenous NAAG selectively and potently inhibits GABA release via activation of mGluR3 receptors (EC50 = 28.6 μM) (Zhao et al. 2001).
The recent introduction of potent GCP II inhibitors (Jackson et al. 1996; Rong et al. 2002; Slusher et al. 1999; Vornov et al. 1999) provides a means to increase endogenous NAAG and study the subsequent effects on synaptic transmission. We took advantage of the GCP II inhibitor 2-(3-mercaptopropyl)pentanedioic acid (2-MPPA) (Majer et al. 2003) to test the hypothesis that NAAG acts to presynaptically inhibit glutamate release at hippocampal mossy fiber-CA3 pyramidal cell synapses. The CA3 region was selected because it expresses important components of NAAGergic transmission, including NAAG and GCP II (Passani et al. 1997a; Slusher et al. 1992). In addition, mGluR II receptors are highly expressed specifically at presynaptic mossy fiber terminals, the presumptive locus of NAAG’s actions (Blumcke et al. 1996; Shigemoto et al. 1997; Tamaru et al. 2001).
METHODS
Hippocampal slice preparations
Animal protocols used in this study complied with all pertinent institutional and federal regulations. Wistar male rats (21–30 day old) were anesthetized with ketamine (50 mg/kg) and decapitated. Brains were rapidly removed and placed in cold (4°C) sucrose-artificial cerebrospinal fluid (ACSF) containing (in mM) 75 sucrose, 3 KCl, 2 CaCl2, 5 MgSO4, 0.15 BES (N,N-bis(2-hydroxyethyl)2-amino-ethanesulfonic acid), NaHCO3, and 25 glucose, aerated with 95% O2-5% CO2 (pH 7.4; 305 mosM). Transverse hippocampal slices (400 μm) were cut with a Vibroslicer (OTS-4000, EMS, Fort Washington, PA). Slices were allowed to recover in standard ACSF (see following text) at 34°C for ≥20 min and then transferred to room temperature before being placed in the recording chamber. In the heated recording chamber (32°C) slices were continuously perfused (at 1.5 ml/min) with ACSF containing (in mM)125 NaCl, 3 KCl, 2 CaCl2, 2 MgSO4, BES, and 15 D-glucose, aerated with 95% O2-5% CO2, pH 7.4.
Electrophysiology
Recordings were made from visually identified pyramidal neurons in the CA3 region of the hippocampus. All cells were initially identified on the basis of somata shapes using differential interface contrast optics. Recording pipettes pulled from borosilicate glass had a resistance of 3–5 MΩ. To record miniature excitatory postsynaptic currents (mEPSCs), patch electrodes were filled with a solution comprising (in mM) 120 K-gluconate, 10 KCl, 1 MgCl2, 2.5 Mg-ATP, 10 HEPES, 0.25 Na2-GTP, 0.1 BAPTA, and biocytin 0.1% (pH 7.2; 287 mosM). Evoked EPSCs were recorded with pipettes containing a fluoride-based solution that intracellularly blocks GABAA-mediated inhibitory currents (Nelson et al. 1994). This solution contained (in mM) 135 KF, 10 KCl, 2 MgCl2, 0.4 HEPES, 0.5 CaCl2, and 0.1 BAPTA (pH 7.2) (Brager et al. 2002).
In experiments involving analysis of evoked EPSCs, 5 mM N-(2,6-dimethylphenylcarbamoyl-methyl) triethylammonium bromide (QX-314) was added to the intracellular solution to prevent Na+-dependent action potentials. Miniature mEPSCs were recorded in the presence of tetrodotoxin (TTX, 0.5 μM).
Intrinsic membrane properties were recorded in current-clamp mode using Axoclamp-2B amplifier. Currents and EPSC data were recorded with either an Axopatch1-D or Axoclamp-2B. Data were digitized at 10 kHz with a DIGIDATA 1300 A/D board (Axon Instruments Foster City, CA) and stored on a PC running pClamp8 software (Axon Instruments). Pyramidal neurons were voltage clamped at −70 mV. Series and access resistance were monitored continually, and cells were discarded if access resistance changed by >20%.
Mossy fiber stimulation
To evoke mossy fiber synaptic responses, stimuli were delivered using Teflon-coated tungsten bipolar electrodes (10- to 20-μm tips) positioned in the hilar region of the dentate gyrus at the border of stratum lucidum (Jonas et al. 1993). Constant current pulses (100–150 μs) were delivered through a constant-current isolation unit (Isoflex, AMPI, Jerusalem). EPSCs were elicited by electrical stimulation of the mossy fiber pathway at a frequency of 0.05–0.033 Hz. In some experiments, stimulation frequency was increased ≤20 Hz to explore saturation of responses. Stimulus strengths were adjusted to evoke reproducible response amplitudes (~70% of the maximal amplitude). Stimulation intensities ranged from 20 to 200 μA. Paired pulse protocols (50-ms interpulse interval) were used to study short-term plasticity.
Chemicals
GCP II inhibitor 2-MPPA was obtained from Guilford Pharmaceuticals. TTX, gabazine, 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX), and D-2-amino-5-phosphonopentanoic acid (D-AP-5) were obtained from Sigma (St. Louis, MO). 2S-2-amino-2-(1S,2S-2-carboxycyclopropyl-1-yl)-3-(xanth-9-yl) propanoic acid (LY341495), 2S,3S,4S-CCG/(2S,1′S,2′S)-2-(carboxycyclopropyl)glycine (L-CCG-I), and 2S,2′R,3′R-2-(2′,3′-dicarboxycyclopropyl)glycine (DCG-IV) were obtained from Tocris Cookson, (Ellisville, MO). Other agents include biocytin (Molecular Probes, Eugene, OR) and QX-134 (Alomone labs, Jerusalem, Israel).
Analyses
Responses were analyzed off-line with MiniAnalysis software (Synaptosoft, Decatur, GA) and pCLAMP8 (Axon Instruments). Paired-pulse ratio (PPR) was calculated as the meanEPSC1/meanEPSC2 (Kim and Alger 2001), where EPSC1 is the amplitude of first evoked current and EPSC2 is the amplitude of the second synaptic current. Dose-response relationships for 2-MPPA were analyzed by fitting the data to the function f(x) = A2 + [(A1 − A2)/1 + (x/x0)p], where x is the 2-MPPA concentration, A1 is a constant representing asymptotic maximum inhibition as determined by the percent of the control data, A2 is the asymptotic minimum, p is the slope. Data are presented as means ± SE. Statistical comparisons were performed with unpaired or paired (for the same cell) Student’s t-test, the nonparametric Kolgomorov-Smirnov statistic (K-S test), one-way ANOVA with post hoc comparisons by Tukey’s honest statistical difference; P < 0.05 was considered significant.
RESULTS
Inhibition of the enzyme GCP II produces an increase in endogenous NAAG concentrations (Jackson and Slusher 2001). This increase in NAAG may impact NMDA receptors at which NAAG acts as a low-affinity agonist and partial antagonist (see Introduction). To focus on the hypothesized group II mGluR-mediated presynaptic actions of NAAG, we performed all experiments in the presence of the NMDA receptor antagonist AP5 (50 μM).
Intrinsic membrane properties are not affected by GCP II inhibition
We first evaluated the effects of GCP II inhibition on intrinsic membrane properties, by recording from CA3 pyramidal neurons in current-clamp (action potential properties) or voltage-clamp mode (whole cell currents). As summarized in Table 1, application of 2-MPPA (0.1–20 μM) had no significant effects on passive membrane properties, including resting membrane potential, membrane time constant, or input resistance (n = 10 cells). Similarly, 2-MPPA did not affect action potential properties, including firing threshold and amplitude, rheobase, or the amplitudes of afterdepolarizing and fast after-hyperpolarizing potentials (Table 1). In addition, 2-MPPA had no significant effects on responses to intra-somatic injections of hyperpolarizing and depolarizing currents (Fig. 1a).
TABLE 1.
Effect of GCP II inhibition on intrinsic membrane properties
| Properties | Control | 2-MPPA | P |
|---|---|---|---|
| Vm, mV | −64.2 ± 1.9 | −64.7 ± 1.7 | 0.59 |
| Rm, MΩ | 223.8 ± 7.1 | 230.5 ± 8.7 | 0.64 |
| τm, ms | 45.8 ± 7.1 | 49.9 ± 8.7 | 0.25 |
| AP threshold, mV | −42.4 ± 3.1 | −41.2 ± 3.0 | 0.24 |
| Rheobase, pA | 98.9 ± 1.8 | 98.1 ± 2.0 | 0.15 |
| AP amplitude, mV | 105.7 ± 5.3 | 106.9 ± 5.6 | 0.65 |
| ADP, mV | 3.3 ± 0.9 | 3.4 ± 1.0 | 0.82 |
| fAHP, mV | 4.5 ± 0.5 | 4.9 ± 0.6 | 0.52 |
Recordings from CA3 pyramidal cells (n = 7) before (control) and after application of the glutamate carboxypeptidase II (GCP II) inhibitor, 2-(3-mercaptopropyl)-pentanedioic acid (2-MPPA). Values are expressed as means ± SE. Vm, resting membrane potential; Rm, input resistance; τm, membrane time constant; AP, action potential; ADP, afterdepolarization; fAHP, fast afterhyperpolarizing potential. Statistical comparisons performed by unpaired Student’s t-test, P as indicated.
FIG. 1.
The glutamate carboxypeptidase II [GCPII, N-acetylated alpha-linked acidic dipeptidase (NAALADase)] inhibitor 2-(3-mercaptopropyl)pentanedioic acid (2-MPPA, 10 μM) does not affect intrinsic membrane properties of CA3 pyramidal neurons. A: voltage recordings of responses to current pulses (400-ms duration; −120–140 pA) before (control) and 10 min after drug application (2-MPPA). B: whole cell currents recorded in voltage-clamp mode. Numbered triangles depict time points analyzed in the I-V plots below. 2-MPPA has no effect on transient outward currents (B1), inwardly rectifying currents (B2 and B3), or sustained currents (B3).
To study the effects of the 2-MPPA on whole cell currents we recorded, in voltage-clamp mode, from 25 CA3 pyramidal neurons in the presence of TTX (0.5 μM; Fig. 1B). 2-MPPA (0.1–20 μM) had no effect on fast, rapidly inactivating inward currents (including IA current; Fig. 1B1), on hyperpolarization-activated anomalous rectifying currents (including IKir and Ih; Fig. 1B, 2 and 3), or on sustained outward currents (reflecting Ca2+ and K+ conductances; Fig. 1B3).
FIG. 2.
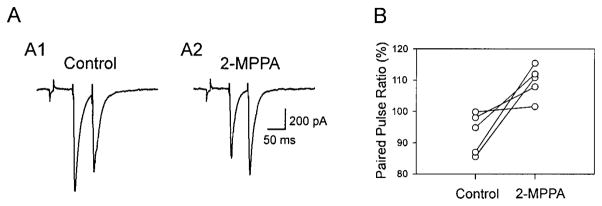
2-MPPA suppresses mossy fiber-CA3 evoked excitatory postsynaptic currents (EPSCs) and enhances paired-pulse facilitation. A1: time course of amplitudes of the first of a pair of EPSC recorded in response to a pair of stimuli (20 Hz) before, during, and after application of 2-MPPA (10 μM). Representative traces are depicted above. Bottom graph plots the time course of the normalized paired-pulse ratios. A2: 2-MPPA does not affect input resistance. Superimposed responses recorded before (—) and after (- - -) drug application, in response to 2-mV hyperpolarizing pulses applied before stimuli (◂, stimulus artifact). A3: superimposed traces, scaled to the amplitude of the 1st control EPSC, demonstrate the 2-MPPA-evoked enhancement of paired-pulse facilitation. Roman numerals correspond to time points indicated on amplitude graph. A4: dose-response curve for EPSC depression (% control) produced by 2-MPPA, showing saturation at concentrations <10 μM, IC50 = 1.33. Values are means ± SE (n = 4– 8 cells). B: group data (n = 8 cells) of normalized EPSC amplitudes and paired-pulse ratios (average of 5 consecutive traces). C: mean ± SE changes in EPSC amplitudes and paired-pulse rations (PPR). *, P < 0.001 (ANOVA).
FIG. 3.
EPSCs recorded in the presence of [Ca2+]/[Mg2+] ratio = 3:1. Under this condition, paired-pulse facilitation (PPF) was converted to paired-pulse depression (PPD, A1). 2-MPPA reversed the PPD to PPF (A2). B: Summary data of effect of 2-MPPA on paired pulse ratio in 5 experiments.
These findings, and the current-clamp data in the preceding text, indicate that application of 2-MPPA and the presumed consequent increases in NAAG concentrations have no discernible effects on intrinsic membrane properties or whole cell conductances of CA3 neurons.
GCP II inhibition depressed mossy fiber-CA3 synaptic transmission
Next we examined the effects of the GCP II inhibitor 2-MPPA on stimulus-evoked AMPA/kainate-mediated EPSCs at mossy fiber-CA3 synapses. Stimulation in the hilar region of the dentate gyrus may evoke in CA3 pyramidal neurons EPSCs mediated by mossy fiber inputs or by other inputs, including those mediated by antidromically activated axon collaterals of CA3 cells, or polysynaptic inputs from neighboring CA3 neurons (for review, see Henze et al. 2000). We therefore included in our analyses only evoked EPSCs that satisfied criteria previously established as diagnostic of mossy-fiber-evoked EPSCs (Claiborne et al. 1993). These criteria include responses having a single component with a rapid rise time (<2.5 ms at 10–90%) and a constant, short latency (<3.5 ms) (Jonas et al. 1993; Xiang et al. 1994). In addition, mossy-fiber-evoked EPSCs are characterized by large paired-pulse facilitation (PPF), (Kobayashi et al. 1999; Salin et al. 1996; Xiang et al. 1994). We therefore accepted for analyses only responses that exhibited a PPF ≥130%, in response to a pair of stimuli delivered at 50-ms interstimulus interval. Finally, mossy-fiber-evoked EPSCs can be diagnosed by their sensitivity to agonists of group II mGluRs, which produce presynaptic inhibition in these synapses (Kamiya et al. 1996; Yoshino et al. 1996). In preliminary experiments, we confirmed that the group II mGluR agonists L-CCG-1 (10 μM; n = 3) or DCG-IV (0.5–10 μM; n = 3) resulted in a significant suppression of EPSC satisfying the preceding criteria (data not shown).
Bath application of 2-MPPA (10 μM for 10–15 min) induced a gradual and long-lasting depression of EPSC amplitudes in these synapses (Fig. 2A). In 8 of 10 cells, this effect was long-lasting, and mean EPSC amplitudes—measured 45 min after drug application—were reduced to 62.6 ± 8.6% of control amplitudes (ANOVA, P < 0.001; Fig. 2, B and C). In the remaining two cells, 2-MPPA significantly reduced EPSC amplitudes, but this effect was partially reversed within 20 min after drug washout (data not shown). Changes in EPSC amplitude were not accompanied by a significant change in input resistance (Fig. 2A2). Moreover, 2-MPPA-induced depression of EPSCs was accompanied by a twofold increase in the coefficient of variation (CV) of the responses (CVcontrol = 0.072 ± 0.01 vs. CV2-MPPA = 0.15 ± 0.01, n = 8, paired t-test, P < 0.001) consistent with a presynaptic mechanism of action. In contrast to its effect on EPSC amplitudes, 2-MPPA had no effect on EPSC rise or decay kinetics. This is shown in Fig. 2A3, depicting scaled and superimposed EPSCs recorded during the control, drug application, and washout periods. The 10–90% rise time of evoked EPSCs during the control periods (2.07 ± 0.4 ms) was not significantly different (P > 0.05, paired t-test, n = 10) from that recorded 10 min after 2-MPPA application (1.95 ± 0.5 ms). Similarly, the 10–90% decay time constants before (15.2 ± 3.2 ms) and during drug application (14.9 ± 2.4 ms) were indistinguishable (P > 0.05).
We studied dose-response relationships by analyzing the effects of varying the concentrations of 2-MPPA on inhibition of evoked EPSC. Bath application of 1 μM 2-MPPA produced a significant reduction (86.0 ± 4.2%, P < 0.001, ANOVA) of EPSC amplitudes measured 45 min after application. Higher 2-MPPA concentrations (20 and 50 μM) also induced a significant depression of synaptic responses (67.7 ± 2.7 and 64.8 ± 1.0%). However, the effect at 20 and 50 μM was not significant different that depression produced by 10 μM 2-MPPA, suggesting that 10 μM is a saturating dose to inhibit synaptic responses at mossy fiber-CA3 synapses. The relation between dose and EPSC inhibition was well fit with a sigmoidal function (Fig. 2A4, r2 = 0.99, see Methods). From this sigmoidal function we estimated IC50 to be 1.33 μM.
GCP II inhibition increases PPF
The mechanisms by which 2-MPPA affects mossy fiber synaptic transmission may involve a decrease in presynaptic glutamate release, an alteration in postsynaptic responsiveness or a combination of both effects. Short-term plasticity is one approach to distinguish between these mechanisms. In response to a pair of stimuli, mossy fiber-CA3 synapses display short-term plasticity, in which the response to a second stimulus is facilitated (Xiang et al. 1994). This PPF is induced by presynaptic mechanisms (Zucker and Regehr 2002). We therefore reasoned that if GCP II inhibition-mediated increases in NAAG act presynaptically, then GCP II inhibition itself should affect PPF. In all cells examined (n = 10), pairs of stimuli (50-ms interval; 20 Hz) resulted in PPF ranging from 120 to 200% (145.8 ± 12.6%; Fig. 2A). Application of 2-MPPA (10 μM) resulted in a significant enhancement of PPF (Fig. 2A). Analysis of group data (n = 8; Fig. 2b) revealed a significant increase in PPF (paired t-test, P < 0.001, Fig. 2C), measured 45 min after drug application (183.7 ± 16.5%). Because an enhancement in PPF at these excitatory synapses depends on initial release probability (Dobrunz and Stevens 1997), these findings suggest that 2-MPPA increases [NAAG] which acts presynaptically at mossy fiber synapses.
GCP II inhibition in high [Ca2+]
As a further test for NAAG’s presynaptic locus of action, we tested the effects of GCP II inhibition under conditions of increased release probability, by increasing the ratio of extracellular [Ca2+]/[Mg2+] from 2:1.3 to 3:1. This results in increased [Ca2+] in presynaptic terminals (Heidelberger et al. 1994) and a subsequent enhanced transmitter release probability (Thomson 2000; Tong and Jahr 1994). As a result, and as previously documented (Canepari and Cherubini 1998), evoked EPSCs exhibited larger amplitudes (by 35 ± 3% relative to control ACSF, n = 5). The enhanced release probability results in rapid vesicle depletion, such that the PPF recorded in normal ACSF was reverted to paired pulse depression (PPD, 5 of 5 cells; Fig. 3A1). We reasoned that if NAAG acts presynaptically to suppress glutamate release, GCP II inhibition should attenuate the effects of high [Ca2+] by suppressing responses to the first stimulus in a train and by suppressing the reduction in PPF. Indeed, 2-MPPA significantly reduced the amplitude of the first EPSC in the train to 69 ± 5.5% of predrug levels (t-test, P < 0.01; Fig. 3A2) and prevented the conversion of PPF to PPD in four of five cells (t-test, P < 0.01, Fig. 3B).
Taken together, these findings indicate that GCP II inhibition, likely resulting in increases in NAAG, exerts a potent modulation of presynaptic glutamate release mechanisms at mossy fiber synapses.
GCP II inhibition acts via group II mGluRs
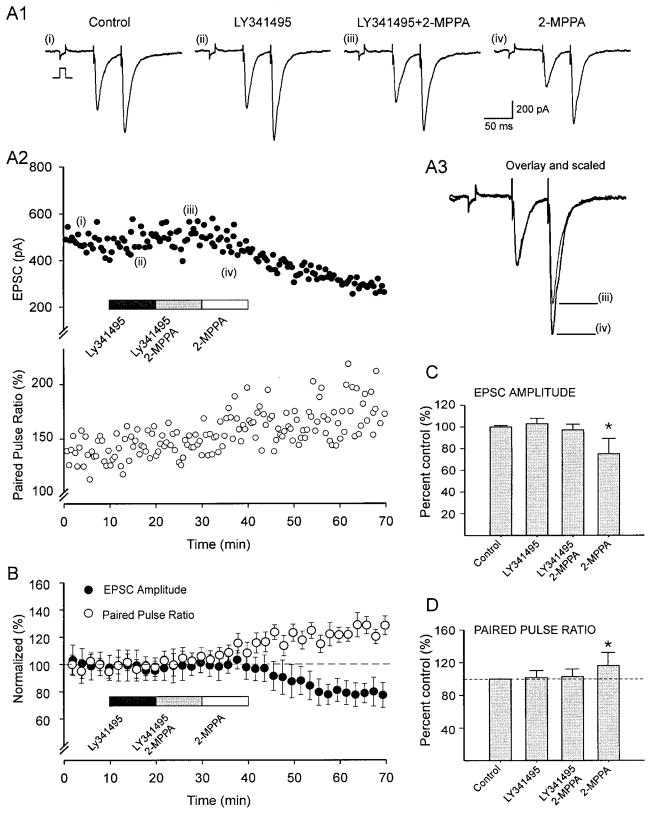
The suppression of presynaptic glutamate release by NAAG is likely due to activation of presynaptic, group II mGluRs, which are expressed by mossy fiber terminals (Petralia et al. 1996; Tamaru et al. 2001). NAAG is a selective agonist at these receptors, mainly of the mGluR3 subtype (Wroblewska et al. 1997; Zhao et al. 2001). To test this hypothesis, we assessed the ability of a group II mGluR antagonist, LY341495, to competitively prevent the effect of GCP II inhibition. LY341495 is a selective and potent group II mGluR antagonist at nanomolar concentrations (20–100 nM, (Kingston et al. 1998). As shown in Fig. 4A, LY341495 (100 nM) had no significant effects on normalized EPSC amplitudes (102.8 ± 5.0%) or on PPF ratios (101.4 ± 8.2%). This is consistent with previous reports that LY341495 has little effect on basal activation of mGluRs (Schoepp 2001). However, LY341495 completely suppressed the presynaptic effects of 2-MPPA (10 μM; Fig. 4, A and B). Neither the amplitudes of mossy fiber evoked EPSCs (97.1 ± 5.2%) (Fig. 4, A and B) nor PPF ratios (101.9 ± 8.3%) were significantly different from controls (t-test, P = 0.12). These effects were reversible on washout of LY341495 when 2-MPPA produced the previously described suppression of EPSC amplitudes and enhancement of PPF ratios (Fig. 4, A and B). Analyses of group data (n = 10) reveal that both the amplitude suppression (74.8 ± 14.2%, 1-way ANOVA, F = 47.2, P < 0.001) and the enhancement of PPF ratios (115.3 ± 9.4%, F = 70.3, P < 0.001) were statistically significant (Fig. 4, B–D). These findings indicate that GCP II inhibition depresses mossy fiber synapses by a mechanism involving activation of group II mGluRs.
FIG. 4.
GCP II inhibition presynaptically suppresses transmitter release by acting on type II mGluRs. A1: representative EPSCs demonstrate that preincubation with the mGluR antagonist 2S-2-amino-2-(1S,2S-2-carboxycyclopropyl-1-yl)-3-(xanth-9-y l) propanoic acid (LY341495, 100 nM) reversibly suppressed the effects of 2-MPPA. Roman numerals refer to time points in A2. Time courses of EPSC amplitudes and paired-pulse ratio (PPR) are depicted in A2, and superimposed responses, scaled to the 1st EPSC, are shown in A3. B: time course of group data for normalized mean ± SE EPSC amplitudes and PPR (n = 10 cells). Mean ± SE (10 cells) normalized EPSC amplitudes (B) and PPR (C; ANOVA, *: P < 0.001).
2-MPPA reduces mEPSC frequency but not their amplitude
Processes that affect the frequency but not the amplitude of spontaneous, TTX-insensitive mEPSCs involve presynaptic, but not postsynaptic mechanisms (Scanziani et al. 1995). Therefore a finding that 2-MPPA results in a reduction in mEPSC frequencies, but has no effect on their amplitudes, would support a presynaptic action of NAAG. We tested this postulate in experiments performed in whole cell voltage-clamp mode (−70 mV) in the presence of TTX (0.5 μM), AP5 (50 μM), and gabazine (10 μM). As previously reported (Jonas et al. 1993), the 10–90% rise time of mEPSC recorded from CA3 pyramidal neurons varied considerably (0.3–12.0 ms). To focus our analysis on spontaneous events mediated by mossy fiber synapses, we restricted our analysis to mEPSCs having a rapid rise time ≤3 ms (see Methods). Application of non-NMDA antagonist CNQX (10–20 μM) completely suppressed all mEPSCs, indicating that they were mediated by AMPA/kainate receptors (data not shown).
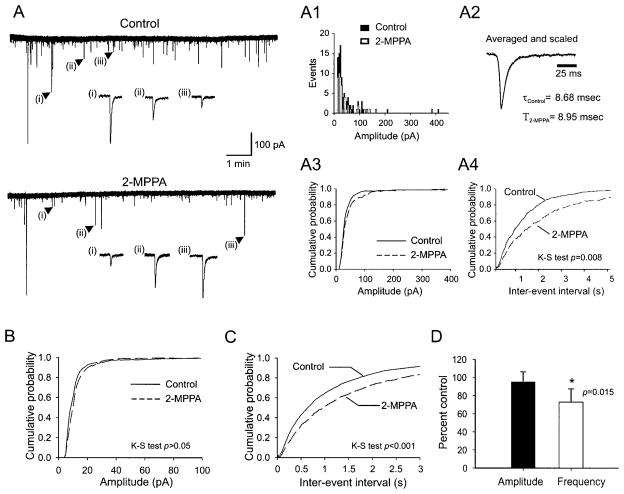
Figure 5A depicts a representative sample of mEPSCs recorded in control ACSF (top) and 10 min after application of 2-MPPA (10 μM; bottom). To test for potential effects of 2-MPPA on mEPSC kinetics, we averaged, scaled, and superimposed mEPSCs recorded from a single neuron before and during drug application (Fig. 5A2). In this example, and from analyses of group data (n = 10 neurons), we conclude that 2-MPPA had no significant effects on mEPSC kinetics, including the 10–90% rise time (ACSF = 2.85 ± 0.07 ms; 2-MPPA = 2.69 ± 0.09 ms, t-test, P = 0.15) and decay time constant (13.1 ± 1.7 vs. 13.5 ± 1.4 ms, t-test, P = 0.31). This finding confirms that 2-MPPA had no discernible effect on postsynaptic properties.
FIG. 5.
GCP II inhibition reduces miniature EPSC (mEPSC) frequency without affecting their amplitudes. A: mEPSC recorded during control artificial cerebrospinal fluid (ACSF; top) and 10 min after 2-MPPA application (10 μM; bottom). Representative EPSCs are shown at higher scale. The amplitude distribution of these mEPSCs is shown in A1 (n = 148 events for control and 91 events for 2-MPPA). Superimposed and scaled average traces (A2) and cumulative probability plots (A3; K-S test, P > 0.05) demonstrate that 2-MPPA had no effect on mEPSC kinetics. By contrast, 2-MPPA significantly suppressed the frequency of mEPSCs (A and A4; K-S test, P < 0.01). Group data (n = 10 cells; B–D) support this conclusion.
The effects of 2-MPPA on the amplitudes and frequencies of mEPSCs recorded from a single CA3 pyramidal neuron are depicted in Fig. 5A. The amplitude histogram (Fig. 5A1) and the cumulative distribution of amplitudes (Fig. 5A3) revealed no significant changes in mEPSC amplitudes after 2-MPPA application (K-S test P = 0.09). By contrast, 2-MPPA resulted in a rightward shift in the cumulative distribution of interevent intervals, demonstrating a significant decrease in mEPSC frequencies (Fig. 5A4, K-S test, P = 0.008). Analyses of group data (n = 10 neurons) are consistent with these findings, demonstrating that 2-MPPA significantly decreased mEPSC frequencies (Fig. 5C, K-S test, P < 0.001) or mean frequency (Fig. 5D, mean = 72.7 ± 14.5% relative to control, P < 0.05), while having no significant effects on their mean amplitudes (91.9 ± 12.3% relative to control; P > 0.05; Fig. 5D) or their distribution (K-S test, P > 0.05, Fig. 5, B and D).
These findings further strengthen our conclusion that inhibition of GCP II, and subsequent increases in NAAG, acts presynaptically to decrease the probability of glutamate release from the mossy fiber nerve synapses.
2-MPPA-induced changes in mEPSC are mediated by group II mGluRs
We next asked whether the 2-MPPA-mediated presynaptic suppression of mEPSCs—like the effects on evoked EPSCs described in the preceding text—are mediated via activation of group II mGluRs. Bath application of the mGluR antagonist LY351495 (100 nM) had no significant effect on the amplitude or frequency of mEPSCs (Fig. 6, A, B, and D). However, LY341495 completely prevented 2-MPPA-induced reduction in mEPSC frequencies (n = 6; Fig. 6, A, C, and D). This effect was reversible, as after LY341495 washout, 2-MPPA produced a significant reduction in mEPSC frequencies but had no effect on their amplitudes (P > 0.05; Fig. 6). Analysis of group data from these six CA3 pyramidal cells demonstrated that 2-MPPA produced a significant rightward shift in the cumulative distribution of interevent intervals (Fig. 6C, K-S test, P < 0.001) and produced a significant reduction in mean mEPSC frequency (65.4 ± 4.6% relative to control, ANOVA P < 0.01; Fig. 6D). These findings support the conclusion that GCP II inhibition increases NAAG, which acts presynaptically by activating group II mGluRs, to suppress glutamate release from mossy fiber synapses.
FIG. 6.
The effects of GCP II inhibition on mEPSCs are mediated by presynaptic, type II mGluRs. Representative records of mEPSCs (A) and group data (B–D; n = 6 neurons) demonstrate that the mGluR antagonist LY341495 (100 nM) reversibly suppressed the effects of 2-MPPA on the frequency of mEPSCs. B and C: cumulative probability plots of mESPC amplitudes and frequencies. D: the mean ± SE of mEPSC amplitudes and frequency (ANOVA, *: P < 0.01).
DISCUSSION
Our aim was to test the hypothesis that endogenous NAAG acts presynaptically, via activation of mGluRs, to presynaptically suppress glutamate release. For this purpose, we took advantage of 2-MPPA, an inhibitor of GCP II, NAAG’s catalytic enzyme. 2-MPPA was recently demonstrated to potently inhibit NAALADase activity with an IC50 of 90 nM and a Ki of 30 nM (Majer et al. 2003). Further, 2-MPPA was recently evaluated for its selectivity for GCP II over >100 other potentially relevant proteins, such as metalloproteases and glutamate receptors (Majer et al. 2003; Wozniak and Slusher, unpublished observations). Even at a 100-fold higher concentration than its IC50 for GCP II (10 μM), 2-MPPA did not exhibit significant activity against any of the proteins examined. Further, preliminary studies demonstrate that a different GCP II inhibitor 2-(phosphonomethyl)pentanedioic acid (2-PMPA) (Witkin et al. 2002) also produces presynaptic suppression of glutamate release in the CA1 region of the hippocampus (Garrido Sanabria and Keller, unpublished observations), further supporting our conclusion that GCP II inhibition results in a specific increase in NAAG concentration. Thus the high affinity of 2-MPPA for GCP II, and its selective action on this enzyme, suggest that the effects we report are indeed due to an increase in endogenous NAAG concentrations and allow us to assay the roles of endogenous NAAG in regulating synaptic transmission.
GCP II inhibition significantly suppressed the amplitudes of mossy-fiber-evoked EPSCs and resulted in a significant enhancement of PPF, a presynaptically mediated phenomenon of short-term plasticity. Further supporting a presynaptic mechanism is the finding that 2-MPPA suppressed the conversion of PPF to PPD in high [Ca2+]. In addition, GCP II inhibition suppressed the frequency but not the amplitude of mEPSCs, consistent with a presynaptic action. In agreement with our findings, NAAG inhibits KCl-mediated GABA release from cortical neurons by acting via presynaptic mGluR3 receptors (Zhao et al. 2001). We found no evidence for postsynaptic effects of GCP II inhibition: there were no discernible effects on the intrinsic membrane properties of CA3 pyramidal neurons or on the decay kinetics of EPSCs evoked by trains of stimuli. In support of the postulate that NAAG acts via activation of group II mGluRs are findings that antagonists of these receptors suppressed the effects of 2-MPPA.
The hydrophilic structure of 2-MPPA predicts a rapid wash-out of the drug. However, in most experiments the effects of 2-MPPA did not reverse, even after 45 min of washout. It is possible that this reflects an irreversible inhibition of NAAG cleavage by GCP II, and a requirement for de novo NAAG synthesis, which may be suppressed in the slice. Additionally, it would be possible that 2-MPPA may act as a slow off rate inhibitor. Another possibility is that NAAG binding to presynaptic mGluRs activates a cascade of intracellular cascades that produce long-term, irreversible effects. Support for this comes from reports that other mGluR II agonists also produce irreversible suppression of synaptic transmission at mossy fiber-CA3 terminals (Kamiya et al. 1996; Manzoni et al. 1995).
NAAG and GCPII expression in hippocampus
The finding that GCP II inhibition strongly depresses synaptic transmission at mossy fiber-CA3 synapses is in agreement with neuroanatomical findings on the expression of NAAG and GCP II in this region. Mossy fiber axon terminals express high levels of GCP II (Slusher et al. 1992). Biochemical studies detect high concentrations of NAAG (300 μM) and GCP II in the hippocampus (Fuhrman et al. 1994). The immunocytochemistry results of Passani et al. (1997b) argue that NAAG is localized in dendritic structures in the molecular layer of the dentate gyrus and in granule cells, suggesting that NAAG may be localized in mossy fibers. These results are contradicted by findings from another study using a high-affinity NAAG antibody which does not cross-react with N-acetyl-aspartate (Moffett and Namboodiri 1995). The latter study concluded that NAAG is expressed in large nonpyramidal neurons in the stratum pyramidale of CA1 and CA3. A selective localization of NAAG in dentate and CA3 interneurons is also supported by the immunocytochemistry study of Anderson et al. (1986).
These anatomical findings, and our present finding, suggest that NAAG is released after activation of mossy fiber axons. NAAG may be released from a subpopulation of NAAG-containing neurons (putative interneurons) postsynaptic to these axons. Based on the distribution patterns of NAAG (see preceding text), it is unlikely that NAAG is directly released from mossy fiber terminals. After its release, NAAG activates presynaptic group II mGluR receptors (mGluR2 and mGluR3), which are expressed by mossy fiber axon terminals and by granule cell bodies (Blumcke et al. 1996; Shigemoto et al. 1997; Tamaru et al. 2001). Indeed, recent studies using [3H]-NAAG revealed strong labeling of NAAG binding sites in the hippocampus that was inhibited by DCG-IV, an agonist of group II mGluRs (Shave et al. 2001). NAAG is a potent mGluR3 agonist (Ghose et al. 1997; Lea et al. 2001; Wroblewska et al. 1997; Zhao et al. 2001) and also binds to mGluR2 with a lower affinity (Schaffhauser et al. 1998).
In addition to GCP II, a novel membrane-bound NAAG peptidase activity was recently identified in the brain, spinal cord, and kidney of GCP II knockout mice (Bacich et al. 2002). The affinity of 2-MPPA to these peptidases has not yet been determined. One possibility is that 2-MPPA inhibits all peptidases, including the novel variants. Alternatively, 2-MPPA may spare the novel peptidases. As a result, our findings may underestimate the effects of NAAG on presynaptic inhibition of transmitter release.
NAAG as a modulator of synaptic transmission
Our findings are consistent with those reported in previous studies, in which the effects of exogenous NAAG were studied. Exogenous NAAG reduces the release of [H3]norepinephrine in rat hippocampal slices (Puttfarcken et al. 1993) and depresses climbing fiber-Purkinje cell excitatory synaptic transmission in cerebellar slices (Sekiguchi et al. 1989). NAAG—acting via mGluR3—reduces GABA release from cortical neurons (Zhao et al. 2001)
In contrast, other studies report a postsynaptic action of exogenous NAAG. In the hippocampal medial perforant pathway, exogenous NAAG (50–200 μM) suppressed LTP by acting on postsynaptic mGluR3 (Lea et al. 2001). That study also reported that NAAG had no effect on PPD, arguing against a presynaptic mechanism (see also (Huang et al. 1999). These postsynaptic actions of NAAG are related to the high density of group II mGluRs on granule cells postsynaptic to medial perforant path-axon terminals (Petralia et al. 1996; Shigemoto et al. 1997). By contrast, the CA3 pyramidal neurons we studied do not express these receptors (Tamaru et al. 2001), a finding that may explain the absence of postsynaptic NAAG effects in our study.
In our study, we suppressed the potential postsynaptic effects of NAAG on NMDA receptors (Koenig et al. 1994; Pai and Ravindranath 1991; Sekiguchi et al. 1992; Valivullah et al. 1994; Westbrook et al. 1986). We did this for technical reasons and because NAAG’s affinity for NMDA receptors is 23-fold lower than that of NMDA (Trombley and Westbrook 1990), suggesting that NAAG’s actions at NMDA receptors may not be physiologically relevant. However, endogenously released NAAG may have a physiological effect as a precursor for glutamate, which has a high affinity for NMDA receptors. Indeed, Thomas et al. (2000) found that GCP II inhibition prevents NMDA-dependent neurotoxicity by suppressing NAAG cleavage to glutamate.
Functional implications
The CA3 region contains a highly interconnected network of pyramidal neurons, rendering it susceptible to recurrent excitation and pathological hyper-excitability (Bains et al. 1999; Christian and Dudek 1988). Presynaptic NAAGergic modulation of synaptic efficacies may be essential for preventing pathological processes associated with excessive glutamate release. Thus pharmacological tools that enhance NAAG activity, such as the GCP II inhibitor 2-MPPA we used in this study, may be an effective therapeutic tool to treat disorders such as epilepsy and neurotoxicity. Indeed, GCP II inhibitors have been successfully used to inhibit the expression and development of cocaine-induced seizures (Witkin et al. 2002). Furthermore, GCP II inhibition exerts a neuroprotective action in animal models of degeneration such as ischemic-hypoxic damage and long-term diabetic neuropathy (Cai et al. 2002; Jackson and Slusher 2001; Lu et al. 2000; Zhang et al. 2002).
Acknowledgments
We are indebted to Dr. Bradley Alger for insightful comments. We thank Dr. Darrell Henze for helpful technical suggestions.
GRANTS
This work was supported by National Institute of Neurological Disorders and Stroke Grants NS-31078 and NS-35360 to A. Keller.
References
- Anderson KJ, Monaghan DT, Cangro CB, Namboodiri MA, Neale JH, Cotman CW. Localization of N-acetylaspartylglutamate-like immunoreactivity in selected areas of the rat brain. Neurosci Lett. 1986;72:14–20. doi: 10.1016/0304-3940(86)90610-5. [DOI] [PubMed] [Google Scholar]
- Bacich DJ, Ramadan E, O’Keefe DS, Bukhari N, Wegorzewska I, Ojeifo O, Olszewski R, Wrenn CC, Bzdega T, Wroblewska B, Heston WD, Neale JH. Deletion of the glutamate carboxypeptidase II gene in mice reveals a second enzyme activity that hydrolyzes N-acetylaspartylglutamate. J Neurochem. 2002;83:20–29. doi: 10.1046/j.1471-4159.2002.01117.x. [DOI] [PubMed] [Google Scholar]
- Bains JS, Longacher JM, Staley KJ. Reciprocal interactions between CA3 network activity and strength of recurrent collateral synapses. Nat Neurosci. 1999;2:720–726. doi: 10.1038/11184. [DOI] [PubMed] [Google Scholar]
- Blumcke I, Behle K, Malitschek B, Kuhn R, Knopfel T, Wolf HK, Wiestler OD. Immunohistochemical distribution of metabotropic glutamate receptor subtypes mGluR1b, mGluR2/3, mGluR4a, and mGluR5 in human hippocampus. Brain Res. 1996;736:217–226. doi: 10.1016/0006-8993(96)00697-x. [DOI] [PubMed] [Google Scholar]
- Brager DH, Capogna M, Thompson SM. Short-term synaptic plasticity, simulation of nerve terminal dynamics, and the effects of protein kinase C activation in rat hippocampus. J Physiol. 2002;541:545–559. doi: 10.1113/jphysiol.2001.015842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai Z, Lin S, Rhodes PG. Neuroprotective effects of N-acetylaspartylglutamate in a neonatal rat model of hypoxia-ischemia. Eur J Pharmacol. 2002;437:139–145. doi: 10.1016/s0014-2999(02)01289-x. [DOI] [PubMed] [Google Scholar]
- Canepari M, Cherubini E. Dynamics of excitatory transmitter release: analysis of synaptic responses in CA3 hippocampal neurons after repetitive stimulation of afferent fibers. J Neurophysiol. 1998;79:1977–1988. doi: 10.1152/jn.1998.79.4.1977. [DOI] [PubMed] [Google Scholar]
- Cassidy M, Neale JH. N-acetylaspartylglutamate catabolism is achieved by an enzyme on the cell surface of neurons and glia. Neuropeptides. 1993;24:271–278. doi: 10.1016/0143-4179(93)90015-3. [DOI] [PubMed] [Google Scholar]
- Christian EP, Dudek FE. Characteristics of local excitatory circuits studied with glutamate microapplication in the CA3 area of rat hippocampal slices. J Neurophysiol. 1988;59:90–109. doi: 10.1152/jn.1988.59.1.90. [DOI] [PubMed] [Google Scholar]
- Claiborne BJ, Xiang Z, Brown TH. Hippocampal circuitry complicates analysis of long-term potentiation in mossy fiber synapses. Hippocampus. 1993;3:115–121. doi: 10.1002/hipo.450030202. [DOI] [PubMed] [Google Scholar]
- Coyle JT. The nagging question of the function of N-acetylaspartylglutamate. Neurobiol Dis. 1997;4:231–238. doi: 10.1006/nbdi.1997.0153. [DOI] [PubMed] [Google Scholar]
- Curatolo A, Marchetti M, Brancati A, Salleo A. Action of N-acetylaspartic and N-acetyl-aspartyl-glutamic acids on the spontaneous electrical activity of the cat cerebral cortex. Arch Sci Biol (Bologna) 1967;51:98–103. [PubMed] [Google Scholar]
- Dobrunz LE, Stevens CF. Heterogeneity of release probability, facilitation, and depletion at central synapses. Neuron. 1997;18:995–1008. doi: 10.1016/s0896-6273(00)80338-4. [DOI] [PubMed] [Google Scholar]
- Fuhrman S, Palkovits M, Cassidy M, Neale JH. The regional distribution of N-acetylaspartylglutamate (NAAG) and peptidase activity against NAAG in the rat nervous system. J Neurochem. 1994;62:275–281. doi: 10.1046/j.1471-4159.1994.62010275.x. [DOI] [PubMed] [Google Scholar]
- Gafurov B, Urazaev AK, Grossfeld RM, Lieberman EM. N-acetylaspartylglutamate (NAAG) is the probable mediator of axon-to-glia signaling in the crayfish medial giant nerve fiber. Neuroscience. 2001;106:227–235. doi: 10.1016/s0306-4522(01)00271-8. [DOI] [PubMed] [Google Scholar]
- Ghose S, Wroblewska B, Corsi L, Grayson DR, De Blas AL, Vicini S, Neale JH. N-acetylaspartylglutamate stimulates metabotropic glutamate receptor 3 to regulate expression of the GABA(A) alpha6 subunit in cerebellar granule cells. J Neurochem. 1997;69:2326–2335. doi: 10.1046/j.1471-4159.1997.69062326.x. [DOI] [PubMed] [Google Scholar]
- Heidelberger R, Heinemann C, Neher E, Matthews G. Calcium dependence of the rate of exocytosis in a synaptic terminal. Nature. 1994;371:513–515. doi: 10.1038/371513a0. [DOI] [PubMed] [Google Scholar]
- Henze DA, Urban NN, Barrionuevo G. The multifarious hippocampal mossy fiber pathway: a review. Neuroscience. 2000;98:407–527. doi: 10.1016/s0306-4522(00)00146-9. [DOI] [PubMed] [Google Scholar]
- Huang L, Rowan MJ, Anwyl R. Induction of long-lasting depression by (+)-alpha-methyl-4-carboxyphenylglycine and other group II mGlu receptor ligands in the dentate gyrus of the hippocampus in vitro. Eur J Pharmacol. 1999;366:151–158. doi: 10.1016/s0014-2999(98)00918-2. [DOI] [PubMed] [Google Scholar]
- Jackson PF, Cole DC, Slusher BS, Stetz SL, Ross LE, Donzanti BA, Trainor DA. Design, synthesis, and biological activity of a potent inhibitor of the neuropeptidase N-acetylated alpha-linked acidic dipeptidase. J Med Chem. 1996;39:619–622. doi: 10.1021/jm950801q. [DOI] [PubMed] [Google Scholar]
- Jackson PF, Slusher BS. Design of NAALADase inhibitors: a novel neuroprotective strategy. Curr Med Chem. 2001;8:949–357. doi: 10.2174/0929867013372797. [DOI] [PubMed] [Google Scholar]
- Jonas P, Major G, Sakmann B. Quantal components of unitary EPSCs at the mossy fiber synapse on CA3 pyramidal cells of rat hippocampus. J Physiol. 1993;472:615–663. doi: 10.1113/jphysiol.1993.sp019965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamiya H, Shinozaki H, Yamamoto C. Activation of metabotropic glutamate receptor type 2/3 suppresses transmission at rat hippocampal mossy fiber synapses. J Physiol. 1996;493:447–455. doi: 10.1113/jphysiol.1996.sp021395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J, Alger BE. Random response fluctuations lead to spurious paired-pulse facilitation. J Neurosci. 2001;21:9608–9618. doi: 10.1523/JNEUROSCI.21-24-09608.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kingston AE, Ornstein PL, Wright RA, Johnson BG, Mayne NG, Burnett JP, Belagaje R, Wu S, Schoepp DD. LY341495 is a nanomolar potent and selective antagonist of group II metabotropic glutamate receptors. Neuropharmacology. 1998;37:1–12. doi: 10.1016/s0028-3908(97)00191-3. [DOI] [PubMed] [Google Scholar]
- Kobayashi K, Manabe T, Takahashi T. Calcium-dependent mechanisms involved in presynaptic long-term depression at the hippocampal mossy fiber-CA3 synapse. Eur J Neurosci. 1999;11:1633–1638. doi: 10.1046/j.1460-9568.1999.00578.x. [DOI] [PubMed] [Google Scholar]
- Koenig ML, Rothbard PM, DeCoster MA, Meyerhoff JL. N-acetylaspartyl-glutamate (NAAG) elicits rapid increase in intraneuronal Ca2+ in vitro. Neuroreport. 1994;5:1063–1068. doi: 10.1097/00001756-199405000-00012. [DOI] [PubMed] [Google Scholar]
- Lea PMt, Wroblewska B, Sarvey JM, Neale JH. beta-NAAG rescues LTP from blockade by NAAG in rat dentate gyrus via the type 3 metabotropic glutamate receptor. J Neurophysiol. 2001;85:1097–1106. doi: 10.1152/jn.2001.85.3.1097. [DOI] [PubMed] [Google Scholar]
- Lu XM, Tang Z, Liu W, Lin Q, Slusher BS. N-acetylaspartylglutamate protects against transient focal cerebral ischemia in rats. Eur J Pharmacol. 2000;408:233–239. doi: 10.1016/s0014-2999(00)00762-7. [DOI] [PubMed] [Google Scholar]
- Majer P, Jackson PF, Delahanty G, Grella BS, Ko YS, Li W, Liu Q, Maclin KM, Polakova J, Shaffer KA, Stoermer D, Vitharana D, Wang EY, Zakrzewski A, Rojas C, Slusher BS, Wozniak KM, Burak E, Limsakun T, Tsukamoto T. Synthesis and biological evaluation of thiol-based inhibitors of glutamate carboxypeptidase II: discovery of an orally active GCP II inhibitor. J Med Chem. 2003;46:1989–1996. doi: 10.1021/jm020515w. [DOI] [PubMed] [Google Scholar]
- Manzoni OJ, Castillo PE, Nicoll RA. Pharmacology of metabotropic glutamate receptors at the mossy fiber synapses of the guinea pig hippocampus. Neuropharmacology. 1995;34:965–971. doi: 10.1016/0028-3908(95)00060-j. [DOI] [PubMed] [Google Scholar]
- Moffett JR, Namboodiri MA. Differential distribution of N-acetylaspartylglutamate and N-acetylaspartate immunoreactivities in rat forebrain. J Neurocytol. 1995;24:409–433. doi: 10.1007/BF01181604. [DOI] [PubMed] [Google Scholar]
- Moffett JR, Palkovits M, Namboodiri A, Neale JH. Comparative distribution of N-acetylaspartylglutamate and GAD67 in the cerebellum and precerebellar nuclei of the rat utilizing enhanced carbodiimide fixation and immunohistochemistry. J Comp Neurol. 1994;347:598–618. doi: 10.1002/cne.903470410. [DOI] [PubMed] [Google Scholar]
- Neale JH, Bzdega T, Wroblewska B. N-Acetylaspartylglutamate: the most abundant peptide neurotransmitter in the mammalian central nervous system. J Neurochem. 2000;75:443–452. doi: 10.1046/j.1471-4159.2000.0750443.x. [DOI] [PubMed] [Google Scholar]
- Nelson S, Toth L, Sheth B, Sur M. Orientation selectivity of cortical neurons during intracellular blockade of inhibition. Science. 1994;265:774–777. doi: 10.1126/science.8047882. [DOI] [PubMed] [Google Scholar]
- Pai KS, Ravindranath V. Toxicity of N-acetylaspartylglutamate and its protection by NMDA and non-NMDA receptor antagonists. Neurosci Lett. 1991;126:49–51. doi: 10.1016/0304-3940(91)90368-4. [DOI] [PubMed] [Google Scholar]
- Passani LA, Vonsattel JP, Carter RE, Coyle JT. N-acetylaspartylglutamate, N-acetylaspartate, and N-acetylated alpha- linked acidic dipeptidase in human brain and their alterations in Huntington and Alzheimer’s diseases. Mol Chem Neuropathol. 1997a;31:97–118. doi: 10.1007/BF02815236. [DOI] [PubMed] [Google Scholar]
- Passani LA, Vonsattel JP, Coyle JT. Distribution of N-acetylaspartylglutamate immunoreactivity in human brain and its alteration in neurodegenerative disease. Brain Res. 1997b;772:9–22. doi: 10.1016/s0006-8993(97)00784-1. [DOI] [PubMed] [Google Scholar]
- Petralia RS, Wang YX, Niedzielski AS, Wenthold RJ. The metabotropic glutamate receptors, mGluR2 and mGluR3, show unique postsynaptic, presynaptic and glial localizations. Neuroscience. 1996;71:949–976. doi: 10.1016/0306-4522(95)00533-1. [DOI] [PubMed] [Google Scholar]
- Puttfarcken PS, Handen JS, Montgomery DT, Coyle JT, Werling LL. N-acetylaspartylglutamate modulation of N-methyl-D-aspartate-stimulated [3H]norepinephrine release from rat hippocampal slices. J Pharmacol Exp Ther. 1993;266:796–803. [PubMed] [Google Scholar]
- Renno WM, Lee JH, Beitz AJ. Light and electron microscopic immunohistochemical localization of N-acetylaspartylglutamate (NAAG) in the olivocerebellar pathway of the rat. Synapse. 1997;26:140–154. doi: 10.1002/(SICI)1098-2396(199706)26:2<140::AID-SYN5>3.0.CO;2-8. [DOI] [PubMed] [Google Scholar]
- Riveros N, Orrego F. A study of possible excitatory effects of N-acetylaspartylglutamate in different in vivo and in vitro brain preparations. Brain Res. 1984;299:393–395. doi: 10.1016/0006-8993(84)90727-3. [DOI] [PubMed] [Google Scholar]
- Rong SB, Zhang J, Neale JH, Wroblewski JT, Wang S, Kozikowski AP. Molecular modeling of the interactions of glutamate carboxypeptidase II with its potent NAAG-based inhibitors. J Med Chem. 2002;45:4140–4152. doi: 10.1021/jm010561g. [DOI] [PubMed] [Google Scholar]
- Salin PA, Scanziani M, Malenka RC, Nicoll RA. Distinct short-term plasticity at two excitatory synapses in the hippocampus. Proc Natl Acad Sci USA. 1996;93:13304–13309. doi: 10.1073/pnas.93.23.13304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scanziani M, Gahwiler BH, Thompson SM. Presynaptic inhibition of excitatory synaptic transmission by muscarinic and metabotropic glutamate receptor activation in the hippocampus: are Ca2+ channels involved? Neuropharmacology. 1995;34:1549–1557. doi: 10.1016/0028-3908(95)00119-q. [DOI] [PubMed] [Google Scholar]
- Schaffhauser H, Richards JG, Cartmell J, Chaboz S, Kemp JA, Klingelschmidt A, Messer J, Stadler H, Woltering T, Mutel V. In vitro binding characteristics of a new selective group II metabotropic glutamate receptor radioligand, [3H] LY354740, in rat brain. Mol Pharmacol. 1998;53:228–233. [PubMed] [Google Scholar]
- Schoepp DD. Unveiling the functions of presynaptic metabotropic glutamate receptors in the central nervous system. J Pharmacol Exp Ther. 2001;299:12–20. [PubMed] [Google Scholar]
- Sekiguchi M, Okamoto K, Sakai Y. Low-concentration N-acetylaspartylglutamate suppresses the climbing fiber response of Purkinje cells in guinea pig cerebellar slices and the responses to excitatory amino acids of Xenopus laevis oocytes injected with cerebellar mRNA. Brain Res. 1989;482:87–96. doi: 10.1016/0006-8993(89)90545-3. [DOI] [PubMed] [Google Scholar]
- Sekiguchi M, Wada K, Wenthold RJ. N-acetylaspartylglutamate acts as an agonist upon homomeric NMDA receptor (NMDAR1) expressed in Xenopus oocytes. FEBS Lett. 1992;311:285–289. doi: 10.1016/0014-5793(92)81121-2. [DOI] [PubMed] [Google Scholar]
- Shave E, Pliss L, Lawrance ML, FitzGibbon T, Stastny F, Balcar VJ. Regional distribution and pharmacological characteristics of [3H]N-acetylaspartyl-glutamate (NAAG) binding sites in rat brain. Neurochem Int. 2001;38:53–62. doi: 10.1016/s0197-0186(00)00045-0. [DOI] [PubMed] [Google Scholar]
- Shigemoto R, Kinoshita A, Wada E, Nomura S, Ohishi H, Takada M, Flor PJ, Neki A, Abe T, Nakanishi S, Mizuno N. Differential presynaptic localization of metabotropic glutamate receptor subtypes in the rat hippocampus. J Neurosci. 1997;17:7503–7522. doi: 10.1523/JNEUROSCI.17-19-07503.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slusher BS, Tsai G, Yoo G, Coyle JT. Immunocytochemical localization of the N-acetyl-aspartyl-glutamate (NAAG) hydrolyzing enzyme N-acetylated alpha-linked acidic dipeptidase (NAALADase) J Comp Neurol. 1992;315:217–229. doi: 10.1002/cne.903150208. [DOI] [PubMed] [Google Scholar]
- Slusher BS, Vornov JJ, Thomas AG, Hurn PD, Harukuni I, Bhardwaj A, Traystman RJ, Robinson MB, Britton P, Lu XC, Tortella FC, Wozniak KM, Yudkoff M, Potter BM, Jackson PF. Selective inhibition of NAALADase, which converts NAAG to glutamate, reduces ischemic brain injury. Nat Med. 1999;5:1396–1402. doi: 10.1038/70971. [DOI] [PubMed] [Google Scholar]
- Stauch BL, Robinson MB, Forloni G, Tsai G, Coyle JT. The effects of N-acetylated alpha-linked acidic dipeptidase (NAALADase) inhibitors on [3H]NAAG catabolism in vivo. Neurosci Lett. 1989;100:295–300. doi: 10.1016/0304-3940(89)90702-7. [DOI] [PubMed] [Google Scholar]
- Tamaru Y, Nomura S, Mizuno N, Shigemoto R. Distribution of metabotropic glutamate receptor mGluR3 in the mouse CNS: differential location relative to pre- and postsynaptic sites. Neuroscience. 2001;106:481–503. doi: 10.1016/s0306-4522(01)00305-0. [DOI] [PubMed] [Google Scholar]
- Thomas AG, Vornov JJ, Olkowski JL, Merion AT, Slusher BS. N-Acetylated alpha-linked acidic dipeptidase converts N-acetylaspartylglutamate from a neuroprotectant to a neurotoxin. J Pharmacol Exp Ther. 2000;295:16–22. [PubMed] [Google Scholar]
- Thomson AM. Facilitation, augmentation, and potentiation at central synapses. Trends Neurosci. 2000;23:305–312. doi: 10.1016/s0166-2236(00)01580-0. [DOI] [PubMed] [Google Scholar]
- Tong G, Jahr CE. Multivesicular release from excitatory synapses of cultured hippocampal neurons. Neuron. 1994;12:51–59. doi: 10.1016/0896-6273(94)90151-1. [DOI] [PubMed] [Google Scholar]
- Trombley PQ, Westbrook GL. Excitatory synaptic transmission in cultures of rat olfactory bulb. J Neurophysiol. 1990;64:598–606. doi: 10.1152/jn.1990.64.2.598. [DOI] [PubMed] [Google Scholar]
- Tsai G, Slusher BS, Sim L, Coyle JT. Immunocytochemical distribution of N-acetylaspartylglutamate in the rat forebrain and glutamatergic pathways. J Chem Neuroanat. 1993;6:277–292. doi: 10.1016/0891-0618(93)90033-z. [DOI] [PubMed] [Google Scholar]
- Urazaev AK, Grossfeld RM, Fletcher PL, Speno H, Gafurov BS, Buttram JG, Lieberman EM. Synthesis and release of N-acetylaspartylglutamate (NAAG) by crayfish nerve fibers: implications for axon-glia signaling. Neuroscience. 2001;106:237–247. doi: 10.1016/s0306-4522(01)00270-6. [DOI] [PubMed] [Google Scholar]
- Valivullah HM, Lancaster J, Sweetnam PM, Neale JH. Interactions between N-acetylaspartylglutamate and AMPA, kainate, and NMDA binding sites. J Neurochem. 1994;63:1714–1719. doi: 10.1046/j.1471-4159.1994.63051714.x. [DOI] [PubMed] [Google Scholar]
- Vornov JJ, Wozniak K, Lu M, Jackson P, Tsukamoto T, Wang E, Slusher B. Blockade of NAALADase: a novel neuroprotective strategy based on limiting glutamate and elevating NAAG. Ann NY Acad Sci. 1999;890:400–405. doi: 10.1111/j.1749-6632.1999.tb08019.x. [DOI] [PubMed] [Google Scholar]
- Westbrook GL, Mayer ML, Namboodiri MA, Neale JH. High concentrations of N-acetylaspartylglutamate (NAAG) selectively activate NMDA receptors on mouse spinal cord neurons in cell culture. J Neurosci. 1986;6:3385–3392. doi: 10.1523/JNEUROSCI.06-11-03385.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williamson LC, Neale JH. Ultrastructural localization of N-acetylaspartylglutamate in synaptic vesicles of retinal neurons. Brain Res. 1988;456:375–381. doi: 10.1016/0006-8993(88)90243-0. [DOI] [PubMed] [Google Scholar]
- Witkin JM, Gasior M, Schad C, Zapata A, Shippenberg T, Hartman T, Slusher BS. NAALADase (GCP II) inhibition prevents cocaine-kindled seizures. Neuropharmacology. 2002;43:348–356. doi: 10.1016/s0028-3908(02)00124-7. [DOI] [PubMed] [Google Scholar]
- Wroblewska B, Wroblewski JT, Pshenichkin S, Surin A, Sullivan SE, Neale JH. N-acetylaspartylglutamate selectively activates mGluR3 receptors in transfected cells. J Neurochem. 1997;69:174–181. doi: 10.1046/j.1471-4159.1997.69010174.x. [DOI] [PubMed] [Google Scholar]
- Xiang Z, Greenwood AC, Kairiss EW, Brown TH. Quantal mechanism of long-term potentiation in hippocampal mossy-fiber synapses. J Neurophysiol. 1994;71:2552–2556. doi: 10.1152/jn.1994.71.6.2552. [DOI] [PubMed] [Google Scholar]
- Yoshino M, Sawada S, Yamamoto C, Kamiya H. A metabotropic glutamate receptor agonist DCG-IV suppresses synaptic transmission at mossy fiber pathway of the guinea pig hippocampus. Neurosci Lett. 1996;207:70–72. doi: 10.1016/0304-3940(96)12486-1. [DOI] [PubMed] [Google Scholar]
- Zhang W, Slusher B, Murakawa Y, Wozniak KM, Tsukamoto T, Jackson PF, Sima AA. GCPII (NAALADase) inhibition prevents long-term diabetic neuropathy in type 1 diabetic BB/Wor rats. J Neurol Sci. 2002;194:21–28. doi: 10.1016/s0022-510x(01)00670-0. [DOI] [PubMed] [Google Scholar]
- Zhao J, Ramadan E, Cappiello M, Wroblewska B, Bzdega T, Neale JH. NAAG inhibits KCl-induced [(3)H]-GABA release via mGluR3, cAMP, PKA and L-type calcium conductance. Eur J Neurosci. 2001;13:340–346. [PubMed] [Google Scholar]
- Zollinger M, Amsler U, Do KQ, Streit P, Cuenod M. Release of N-acetylaspartylglutamate on depolarization of rat brain slices. J Neurochem. 1988;51:1919–1923. doi: 10.1111/j.1471-4159.1988.tb01178.x. [DOI] [PubMed] [Google Scholar]
- Zucker RS, Regehr WG. Short-term synaptic plasticity. Annu Rev Physiol. 2002;64:355–405. doi: 10.1146/annurev.physiol.64.092501.114547. [DOI] [PubMed] [Google Scholar]