Abstract
Indolent T-lymphoblastic proliferation has been rarely reported in the upper aerodigestive tract. The lymphoid cells associated with this condition have the morphological and phenotypical features of immature thymocytes. However, their pathogenesis and biology are unknown. We present an unusual type of tumor infiltrating lymphocytes in a case with hepatocellular carcinoma, presumed to be a T-lymphoblastic proliferation. A 58-yr-old female patient presented with indigestion and a palpable epigastric mass. The abdominal computed tomography revealed a mass in the S6 region of the liver. A hepatic segmentectomy was performed. Microscopic examination showed dense isolated nests of monomorphic lymphoid cells within the tumor. Immunohistochemically, the lymphoid cells were positive for CD3, terminal deoxymucleotide transferase (TdT) and CD1a. In addition, they showed dual expression of CD4 and CD8. The polymerase chain reaction used to examine the T-cell antigen receptor gamma gene rearrangement showed polyclonal T-cell proliferation. This is the second case of hepatocellular carcinoma combined with indolent T-lymphoblastic proliferation identified by an unusual tumor infiltrating lymphocytes.
Keywords: Lymphocytes, Tumor-Infiltrating; Carcinoma, Hepatocellular; indolent T-lymphoblastic proliferation; Immunohistochemistry
INTRODUCTION
Tumor-infiltrating lymphocytes (TILs) can be found in most cancers with a variable intensity. They are mainly composed of CD8+ cytotoxic T-cells, CD4+ helper T-cells and NK cells (1). Although these cells exert antitumor activity, they usually fail to control tumor growth due to other factors such as the presence of regulatory cells and the expression of inhibitory ligands of the tumor cells. The association between TILs and a favorable prognosis has been demonstrated in a few tumors including melanoma and colon cancer (2).
Likewise, in hepatocellular carcinoma, it is known that CD8+ TILs may play a role in the occurrence of tumor cell apoptosis (3), however, CD4+CD25+ regulatory T-cells impair the effector function of them (4, 5). Although there are a few reports that describe hepatocellular carcinoma with lymphoid rich stroma, so called lymphoepithelioma-like hepatocellular carcinoma (6, 7), the phenotypic analysis of TILs in 28 cases of hepatocellular carcinoma revealed that hepatocellular carcinoma tissues had less intensity of lymphocyte infiltration than the corresponding non-tumor liver tissues with increased CD4+CD25+ regulatory T-cells.
Here we report an unusual type of TIL composed of immature T-cells in a patient with hepatocellular carcinoma. Because almost TILs in hepatocellular carcinoma are activated type and expressed antigen-experienced phenotypes (8) this finding is very exceptional and up to now only one similar case was previously reported. This case further suggests that hepatocellular carcinoma could occur in association with indolent T-lymphoblastic proliferation.
CASE REPORT
A 58-yr-old female patient presented with indigestion and a palpable epigastric mass for two months. The patient was diagnosed as being a hepatitis B s-antigen (HBsAg) carrier 15 yr previously. However, she received no medical treatment. The patient had no history of excessive alcohol intake. The serum alpha-1-fetoprotein measured 17,400 ng/mL. The white blood cell count was 2,600/µL, and the platelet count was 86,000/µL. The abdominal computed tomography (CT) scan revealed an exophytic mass in the S6 region of the liver. The tumor was confined to the liver; the other organs showed no abnormalities. The patient received hepatic segmentectomy.
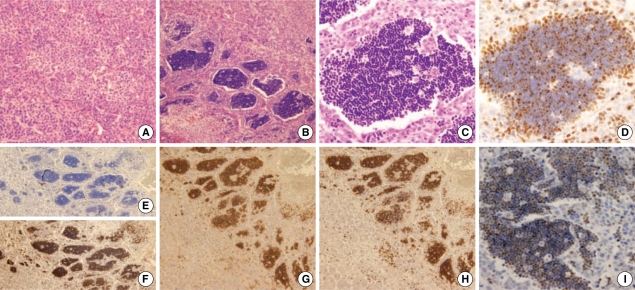
Grossly the tumor measured 5 cm in diameter and had a multinodular confluent pattern with a yellow-tan color. The microscopic examination revealed a poorly differentiated hepatocellular carcinoma (Edmonson grade 3) with a macrotrabecular and acinar pattern (Fig. 1A). At the periphery, the tumor showed poorer differentiation (Edmonson grade 4) and many giant cells. The surrounding non-neoplastic liver parenchyma was replaced by cirrhotic nodules, 3-5 mm in diameter. Notably, there were foci of extensive infiltrations of lymphoid cells within the tumor. The lymphoid cells occasionally formed dense isolated nests between the cords of tumor cells (Fig. 1B). The lymphoid cells were small with a round nucleus and had scanty cytoplasm. The nucleus was basophilic with indistinct nucleoli. They showed monomorphic features with little cytologic atypism (Fig. 1C). On the immunohistochemical staining, the monomorphic lymphoid cells were positive for CD3 (Fig. 1F), terminal deoxynucleotide transferase (TdT) (Fig. 1I) and CD1a. They showed dual expression of CD4 and CD8 (Fig. 1G, H). The above morphological and immunohistochemical features of the lymphoid cells were consistent with the phenotype of immature T-lymphoblasts. They were all negative for L26 (Fig. 1E), CD79a, granzyme, CD68, CD5, and c-kit. They had a relatively high Ki-67 labeling index (approximately more than 40%) (Fig. 1D). The polymerase chain reaction to examine the T-cell antigen receptor gamma gene rearrangement, revealed polyclonal T-cell proliferation.
Fig. 1.
Histological and immunohistochemical features. (A) The hepatocellular carcinoma is composed of poorly differentiated cells with trabecular pattern (H&E, ×200). (B) Aggregates of monomorphic lymphoid cells are observed within the hepatocellular carcinoma (H&E, ×100). (C) The lymphocytes show a morphology similar to immature thymocytes (H&E, ×400). (D) Ki67 labeling index is approximately 40% (H&E, ×400). (E) The lymphoid cells are negative for L26 (H&E, ×100). (F) The lymphoid cells are positive for CD3 (H&E, ×100). (G, H) The lymphoid cells show dual expression of CD4 and CD8 (G, CD4; H, CD8) (H&E, ×100). (I) TdT are expressed in lymphoid cells (H&E, ×400).
On the other hand, in the peripheral portion of the tumor, small numbers of polymorphic TILs composed of mature T or B lymphocytes or plasma cells were observed. Some of the polymorphic TILs showed limited infiltration into the portal area and lobules of the hepatic parenchyma around the tumor.
Two months later, the patient presented with a chest wall mass. The radiological examination showed recurrence of the tumor in the liver with portal vein involvement, multiple lung and rib metastases. Although biopsy was not performed, the CT scan favored the possibility of aggravation of hepatocellular carcinoma and the serum alpha-1-fetoprotein measured 15,000 ng/mL at that time. The operability of the tumor was very low so the patient is receiving conservative management.
DISCUSSION
The porcine species is known to have a large percentage of CD4 and CD8 dual expressing peripheral T-cells (9). However, in humans, CD4 and CD8 dual expressing T-cells can be found in thymic cortex in which reside the immature thymic T-lymphocytes, and extrathymic precursor T-cells and T-lymphoblastic lymphoma/leukemia. Positivity for TdT is observed in cortical thymocytes and immature bone marrow T and B cell precursors undergoing antigen receptor gene rearrangement. The immunophenotype found in the lymphocytes of the present case was the same as that of precursor (immature) T-cell or T-lymphoblastic lymphoma/leukemia. In addition, they showed a high Ki-67 proliferation index, which made us to considered the possibility of neoplastic precursor T cell proliferation.
T-lymphoblastic lymphoma/leukemia is a high-grade neoplasm composed of small to medium sized blastic cells. Patients with T-lymphoblastic lymphoma/leukemia frequently present with a mediastinal mass and rapidly progressive disease if untreated. In our case, the proliferating lymphocytes were morphologically and immunohistochemically T lymphoblasts. However, the presentation was apart from the usual T-lymphoblastic lymphoma. In addition, the polymerase chain reaction study did not show monoclonal T-cell proliferation. Therefore, the possibility of T-lymphoblastic lymphoma/leukemia was excluded.
The liver plays an important role in innate and adaptive immunity through the production of acute phase proteins, phagocytosis and removal of activated T-cells (11). Extrathymic T-cell development and proliferation also could be occur in the mammalian liver, and this function becomes more important with aging (10-12). Therefore, the hepatic T-cell population is quite different from those of other organs. For example, unlike the peripheral blood, the conventional CD4+ or CD8+ T-cells account only 40% of hepatic CD3+ T-cells. However, CD4+CD8+ dual expressing T-cells and CD4-CD8- dual negative T-cells account for a significant proportion of hepatic T-cells (13). IL-7 induced proliferation of T-cells with intermediate levels of TCR and CD4/CD8 dual negative or CD4 positive phenotype in the murine liver (14). In the human liver, the hepatic microenvironment including cytokines might influence the distribution of T-cell populations, and increased production of certain cytokines might result in selective expansion of the resident population of T-cells under pathological conditions. In the present case, it would be possible that certain cytokines produced by transformed hepatocytes of hepatocellular carcinoma, might have stimulated the expansion of resident CD4+CD8+ dual expressing immature T-cells, or recruited them from the thymic cortex or bone marrow.
Wang et al. previously reported one similar case. They presented a case with hepatocellular carcinoma accompanied by TdT positive T lymphocyte proliferation in the tumor. They suggested that their patient had indolent T-lymphoblastic proliferation combined with hepatocellular carcinoma (15). Indolent T-lymphoblastic proliferation was first described by Velankar et al. in 1999 in the upper aerodigestive tract (16). Two years later Strauchen reported a similar phenomenon in the oropharynx in a myasthenia gravis patient (17). In this newly introduced disorder, the infiltrating lymphocytes showed lymphoblastic morphology and expressed TdT, and both CD4 and CD8. Although the two patients suffered from multiple recurrences over a long time, they showed no systemic dissemination with or without chemotherapy. Whether this disorder is a neoplastic proliferation or true hyperplasia of T-lymphoblasts remains unclear. Strauchen reported that the tropism of the T-lymphoblasts of the oropharynx due to retained thymic potential of the pharyngeal epithelium might cause the indolent T-lymphoblastic proliferation (17). Wang suggested that hepatocellular carcinoma could recruit immature T-lymphocytes into the tumor from the thymic cortex or bone marrow through unknown mechanisms (15).
The clinical course of the present case was more aggressive than that of Wang's case and showed multiple metastases two months after the initial diagnosis and treatment. If the immature T-cell proliferation accompanying the hepatocellular carcinoma was related to a poor patient prognosis should be clarified by further study.
In conclusion, we have reported a second case of hepatocellular carcinoma with T-cell lymphoblastic proliferation as a type of unusual TIL. The precise mechanism of the T lymphoblastic proliferation in the hepatocellular carcinoma and the clinicopathological significance require further elucidation.
References
- 1.Chiou SH, Sheu BC, Chang WC, Huang SC, Hong-Nerng H. Current concepts of tumor-infiltrating lymphocytes in human malignancies. J Reprod Immunol. 2005;67:35–50. doi: 10.1016/j.jri.2005.06.002. [DOI] [PubMed] [Google Scholar]
- 2.Liakou CI, Narayanan S, Ng Tang D, Logothetis CJ, Sharma P. Focus on TILs: prognostic significance of tumor infiltrating lymphocytes in human bladder cancer. Cancer Immun. 2007;7:10–15. [PMC free article] [PubMed] [Google Scholar]
- 3.Ikeguchi M, Oi K, Hirooka Y, Kaibara N. CD8+ lymphocyte infiltration and apoptosis in hepatocellular carcinoma. Eur J Surg Oncol. 2004;30:53–57. doi: 10.1016/j.ejso.2003.10.001. [DOI] [PubMed] [Google Scholar]
- 4.Fu J, Xu D, Liu Z, Shi M, Zhao P, Fu B, Zhang Z, Yang H, Zhang H, Zhou C, Yao J, Jin L, Wang H, Yang Y, Fu YX, Wang FS. Increased regulatory T cells correlate with CD8 T-cell impairment and poor survival in hepatocellular carcinoma patients. Gastroenterology. 2007;132:2328–2339. doi: 10.1053/j.gastro.2007.03.102. [DOI] [PubMed] [Google Scholar]
- 5.Unitt E, Rushbrook SM, Marshall A, Davies S, Gibbs P, Morris LS, Coleman N, Alexander GJ. Compromised lymphocytes infiltrate hepatocellular carcinoma: the role of T-regulatory cells. Hepatology. 2005;41:722–730. doi: 10.1002/hep.20644. [DOI] [PubMed] [Google Scholar]
- 6.Chen CJ, Jeng LB, Huang SF. Lymphoepithelioma-like hepatocellular carcinoma. Chang Gung Med J. 2007;30:172–177. [PubMed] [Google Scholar]
- 7.Emile JF, Adam R, Sebagh M, Marchadier E, Falissard B, Dussaix E, Bismuth H, Reynès M. Hepatocellular carcinoma with lymphoid stroma: a tumour with good prognosis after liver transplantation. Histopathology. 2000;37:523–529. doi: 10.1046/j.1365-2559.2000.00952.x. [DOI] [PubMed] [Google Scholar]
- 8.Chen CH, Lee HS, Huang GT, Yang PM, Yu WY, Cheng KC, Lee PH, Jeng YM, Chen DS, Sheu JC. Phenotypic analysis of tumor-infiltrating lymphocytes in hepatocellular carcinoma. Hepatogastroenterology. 2007;54:1529–1533. [PubMed] [Google Scholar]
- 9.Pescovitz MD, Sakopoulos AG, Gaddy JA, Husmann RJ, Zuckermann FA. Porcine peripheral blood CD4+/CD8+ dual expressing T-cells. Vet Immunol Immunopathol. 1994;43:53–62. doi: 10.1016/0165-2427(94)90120-1. [DOI] [PubMed] [Google Scholar]
- 10.Parker GA, Picut CA. Liver immunobiology. Toxicol Pathol. 2005;33:52–62. doi: 10.1080/01926230590522365. [DOI] [PubMed] [Google Scholar]
- 11.Sato K, Ohtsuka K, Hasegawa K, Yamagiwa S, Watanabe H, Asakura H, Abo T. Evidence for extrathymic generation of intermediate T cell receptor cells in the liver revealed in thymectomized, irradiated mice subjected to bone marrow transplantation. J Exp Med. 1995;182:759–767. doi: 10.1084/jem.182.3.759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ohteki T, Seki S, Abo T, Kumagai K. Liver is a possible site for the proliferation of abnormal CD3+4-8- double-negative lymphocytes in autoimmune MRL-lpr/lpr mice. J Exp Med. 1990;172:7–12. doi: 10.1084/jem.172.1.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Doherty DG, O'Farrelly C. Innate and adaptive lymphoid cells in the human liver. Immunol Rev. 2000;174:5–20. doi: 10.1034/j.1600-0528.2002.017416.x. [DOI] [PubMed] [Google Scholar]
- 14.Miyaji C, Watanabe H, Osman Y, Kuwano Y, Abo T. A comparison of proliferative response to IL-7 and expression of IL-7 receptors in intermediate TCR cells of the liver, spleen, and thymus. Cell Immunol. 1996;169:159–165. doi: 10.1006/cimm.1996.0106. [DOI] [PubMed] [Google Scholar]
- 15.Wang ZM, Xiao WB, Zheng SS, Sun K, Wang LJ. Hepatocellular carcinoma with Indolent T-lymphoblastic proliferation. Leuk Lymphoma. 2006;47:2424–2426. doi: 10.1080/10428190600822151. [DOI] [PubMed] [Google Scholar]
- 16.Velankar MM, Nathwani BN, Schlutz MJ, Bain LA, Arber DA, Slovak ML, Weiss LM. Indolent T-lymphoblastic proliferation: report of a case with a 16-year course without cytotoxic therapy. Am J Surg Pathol. 1999;23:977–981. doi: 10.1097/00000478-199908000-00017. [DOI] [PubMed] [Google Scholar]
- 17.Strauchen JA. Indolent T-lymphoblastic proliferation: report of a case with an 11-year history and association with myasthenia gravis. Am J Surg Pathol. 2001;25:411–415. doi: 10.1097/00000478-200103000-00018. [DOI] [PubMed] [Google Scholar]