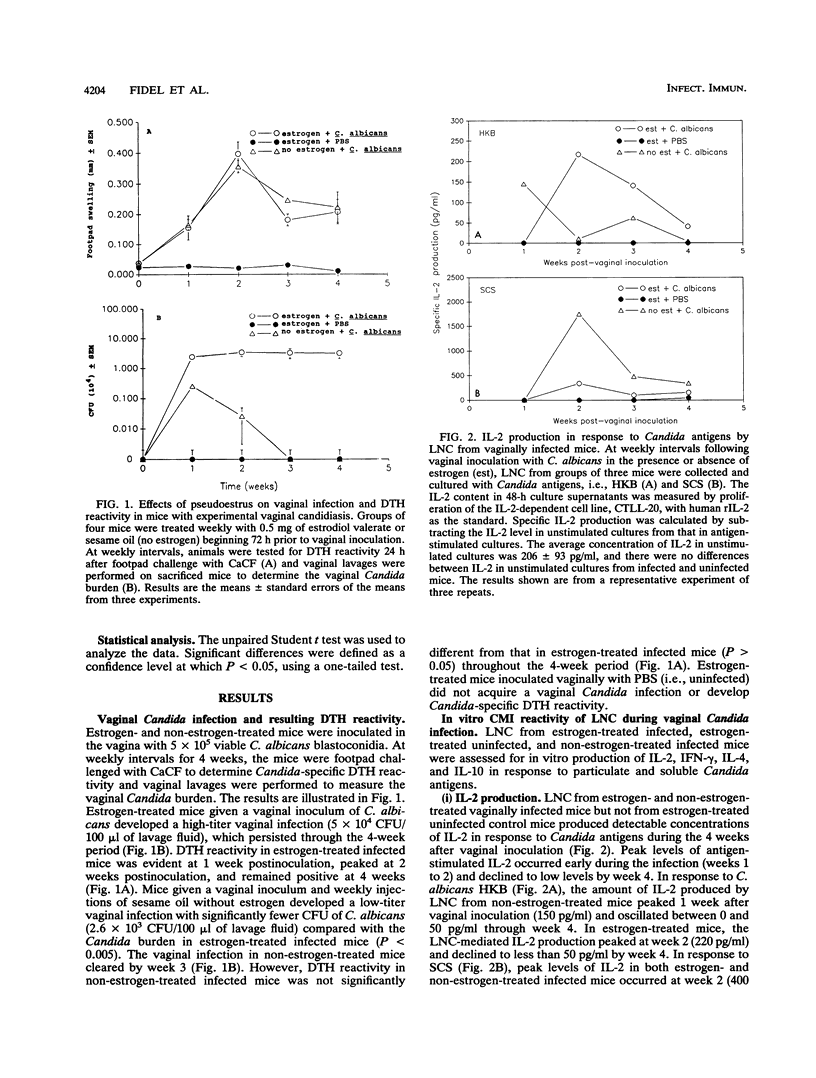
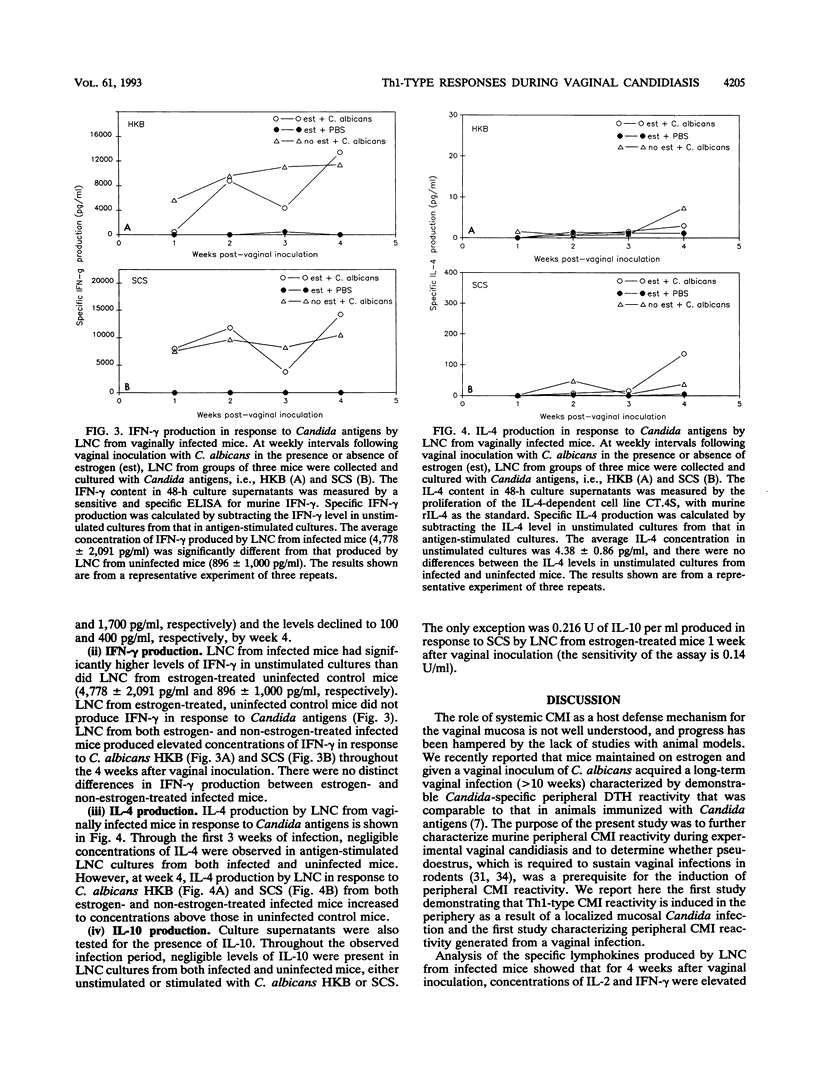
Abstract
The role of systemic cell-mediated immunity (CMI) as a host defense mechanism in the vagina is poorly understood. Using a murine pseudoestrus model of experimental vaginal candidiasis, we previously found that animals given a vaginal inoculum of viable Candida albicans blastoconidia acquired a persistent vaginal infection and developed Candida-specific delayed-type hypersensitivity (DTH) responses. The present study was designed to characterize the peripheral CMI reactivity generated from the vaginal infection in mice and to determine whether pseudoestrus is a prerequisite for the induction of peripheral CMI reactivity. Mice treated or not treated with estrogen and given a vaginal inoculum of C. albicans blastoconidia were examined for 4 weeks for their vaginal Candida burden and peripheral CMI reactivity, including DTH responsiveness and in vitro Th1 (interleukin-2 [IL-2], gamma interferon [IFN-gamma]/Th2 (IL-4, IL-10)-type lymphokine production in response to Candida antigens. Results showed that although mice not treated with estrogen before being given a vaginal inoculum of C. albicans blastoconidia developed only a short-lived vaginal infection and harbored significantly fewer Candida CFU in the vagina compared with those given estrogen and then infected; DTH reactivity was equivalent in both groups. In vitro measurement of CMI reactivity further showed that lymph node cells from both estrogen- and non-estrogen-treated infected mice produced elevated levels of IL-2 and IFN-gamma in response to Candida antigens during the 4 weeks after vaginal inoculation. In contrast, lymph node cells from the same vaginally infected mice showed no IL-10 production and only small elevations of IL-4 during week 4 of infection. These results suggest that mice with experimental vaginal candidiasis develop predominantly Th1-type Candida-specific peripheral CMI reactivity and that similar patterns of Th1-type reactivity occur in mice regardless of the persistence of infection and the estrogen status of the infected mice.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aronson I. K., Rieger C. H., Soltani K., Tkalcevic V., Chan W. C., Lorincz A. L., Matz G. Late onset chronic mucocutaneous candidiasis with lymphoma and specific serum inhibitory factor. Cancer. 1979 Jan;43(1):101–108. doi: 10.1002/1097-0142(197901)43:1<101::aid-cncr2820430116>3.0.co;2-6. [DOI] [PubMed] [Google Scholar]
- Boom W. H., Liebster L., Abbas A. K., Titus R. G. Patterns of cytokine secretion in murine leishmaniasis: correlation with disease progression or resolution. Infect Immun. 1990 Dec;58(12):3863–3870. doi: 10.1128/iai.58.12.3863-3870.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bottomly K. A functional dichotomy in CD4+ T lymphocytes. Immunol Today. 1988 Sep;9(9):268–274. doi: 10.1016/0167-5699(88)91308-4. [DOI] [PubMed] [Google Scholar]
- Canales L., Middlemas R. O., 3rd, Louro J. M., South M. A. Immunological observations in chronic mucocutaneous candidiasis. Lancet. 1969 Sep 13;2(7620):567–571. doi: 10.1016/s0140-6736(69)90264-5. [DOI] [PubMed] [Google Scholar]
- Domer J. E., Garner R. E., Befidi-Mengue R. N. Mannan as an antigen in cell-mediated immunity (CMI) assays and as a modulator of mannan-specific CMI. Infect Immun. 1989 Mar;57(3):693–700. doi: 10.1128/iai.57.3.693-700.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durandy A., Fischer A., Le Deist F., Drouhet E., Griscelli C. Mannan-specific and mannan-induced T-cell suppressive activity in patients with chronic mucocutaneous candidiasis. J Clin Immunol. 1987 Sep;7(5):400–409. doi: 10.1007/BF00917018. [DOI] [PubMed] [Google Scholar]
- Fidel P. L., Jr, Lynch M. E., Sobel J. D. Candida-specific cell-mediated immunity is demonstrable in mice with experimental vaginal candidiasis. Infect Immun. 1993 May;61(5):1990–1995. doi: 10.1128/iai.61.5.1990-1995.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiorentino D. F., Bond M. W., Mosmann T. R. Two types of mouse T helper cell. IV. Th2 clones secrete a factor that inhibits cytokine production by Th1 clones. J Exp Med. 1989 Dec 1;170(6):2081–2095. doi: 10.1084/jem.170.6.2081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer A., Ballet J. J., Griscelli C. Specific inhibition of in vitro Candida-induced lymphocyte proliferation by polysaccharidic antigens present in the serum of patients with chronic mucocutaneous candidiasis. J Clin Invest. 1978 Nov;62(5):1005–1013. doi: 10.1172/JCI109204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fleury F. J. Adult vaginitis. Clin Obstet Gynecol. 1981 Jun;24(2):407–438. doi: 10.1097/00003081-198106000-00008. [DOI] [PubMed] [Google Scholar]
- Fong I. W., McCleary P., Read S. Cellular immunity of patients with recurrent or refractory vulvovaginal moniliasis. Am J Obstet Gynecol. 1992 Mar;166(3):887–890. doi: 10.1016/0002-9378(92)91356-f. [DOI] [PubMed] [Google Scholar]
- Gajewski T. F., Fitch F. W. Anti-proliferative effect of IFN-gamma in immune regulation. I. IFN-gamma inhibits the proliferation of Th2 but not Th1 murine helper T lymphocyte clones. J Immunol. 1988 Jun 15;140(12):4245–4252. [PubMed] [Google Scholar]
- Gatenby P., Basten A., Adams E. Thymoma and late onset mucocutaneous candidiasis associated with a plasma inhibitor of cell-mediated immune function. J Clin Lab Immunol. 1980 May;3(3):209–216. [PubMed] [Google Scholar]
- Gillis S., Ferm M. M., Ou W., Smith K. A. T cell growth factor: parameters of production and a quantitative microassay for activity. J Immunol. 1978 Jun;120(6):2027–2032. [PubMed] [Google Scholar]
- Hobbs J. R., Brigden D., Davidson F., Kahan M., Oates J. K. Immunological aspects of candidal vaginitis. Proc R Soc Med. 1977;70 (Suppl 4):11–14. doi: 10.1177/00359157770700S404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu-Li J., Ohara J., Watson C., Tsang W., Paul W. E. Derivation of a T cell line that is highly responsive to IL-4 and IL-2 (CT.4R) and of an IL-2 hyporesponsive mutant of that line (CT.4S). J Immunol. 1989 Feb 1;142(3):800–807. [PubMed] [Google Scholar]
- Hurley R. Candidal vaginitis. Proc R Soc Med. 1977;70 (Suppl 4):1–2. doi: 10.1177/00359157770700S401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurley R., De Louvois J. Candida vaginitis. Postgrad Med J. 1979 Sep;55(647):645–647. doi: 10.1136/pgmj.55.647.645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurley R. Recurrent Candida infection. Clin Obstet Gynaecol. 1981 Apr;8(1):209–214. [PubMed] [Google Scholar]
- Mills K. H., Barnard A., Watkins J., Redhead K. Cell-mediated immunity to Bordetella pertussis: role of Th1 cells in bacterial clearance in a murine respiratory infection model. Infect Immun. 1993 Feb;61(2):399–410. doi: 10.1128/iai.61.2.399-410.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mond J. J., Carman J., Sarma C., Ohara J., Finkelman F. D. Interferon-gamma suppresses B cell stimulation factor (BSF-1) induction of class II MHC determinants on B cells. J Immunol. 1986 Dec 1;137(11):3534–3537. [PubMed] [Google Scholar]
- Morton R. S., Rashid S. Candidal vaginitis: natural history, predisposing factors and prevention. Proc R Soc Med. 1977;70 (Suppl 4):3–6. doi: 10.1177/00359157770700S402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosmann T. R., Cherwinski H., Bond M. W., Giedlin M. A., Coffman R. L. Two types of murine helper T cell clone. I. Definition according to profiles of lymphokine activities and secreted proteins. J Immunol. 1986 Apr 1;136(7):2348–2357. [PubMed] [Google Scholar]
- Paterson P. Y., Semo R., Blumenschein G., Swelstad J. Mucocutaneous candidiasis, anergy and a plasma inhibitor of cellular immunity: reversal after amphotericin B therapy. Clin Exp Immunol. 1971 Nov;9(5):595–602. [PMC free article] [PubMed] [Google Scholar]
- Perito S., Calabresi A., Romani L., Puccetti P., Bistoni F. Involvement of the Th1 subset of CD4+ T cells in acquired immunity to mouse infection with Trypanosoma equiperdum. Cell Immunol. 1992 Sep;143(2):261–271. doi: 10.1016/0008-8749(92)90024-j. [DOI] [PubMed] [Google Scholar]
- Piccolella E., Lombardi G., Morelli R. Generation of suppressor cells in the response of human lymphocytes to a polysaccharide from Candida albicans. J Immunol. 1981 Jun;126(6):2151–2155. [PubMed] [Google Scholar]
- Reynolds D. S., Boom W. H., Abbas A. K. Inhibition of B lymphocyte activation by interferon-gamma. J Immunol. 1987 Aug 1;139(3):767–773. [PubMed] [Google Scholar]
- Romani L., Mencacci A., Cenci E., Spaccapelo R., Mosci P., Puccetti P., Bistoni F. CD4+ subset expression in murine candidiasis. Th responses correlate directly with genetically determined susceptibility or vaccine-induced resistance. J Immunol. 1993 Feb 1;150(3):925–931. [PubMed] [Google Scholar]
- Romani L., Mencacci A., Grohmann U., Mocci S., Mosci P., Puccetti P., Bistoni F. Neutralizing antibody to interleukin 4 induces systemic protection and T helper type 1-associated immunity in murine candidiasis. J Exp Med. 1992 Jul 1;176(1):19–25. doi: 10.1084/jem.176.1.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryley J. F., McGregor S. Quantification of vaginal Candida albicans infections in rodents. J Med Vet Mycol. 1986 Dec;24(6):455–460. [PubMed] [Google Scholar]
- Sobel J. D., Muller G., McCormick J. F. Experimental chronic vaginal candidosis in rats. Sabouraudia. 1985 Jun;23(3):199–206. doi: 10.1080/00362178585380301. [DOI] [PubMed] [Google Scholar]
- Sobel J. D. Pathogenesis and epidemiology of vulvovaginal candidiasis. Ann N Y Acad Sci. 1988;544:547–557. doi: 10.1111/j.1749-6632.1988.tb40450.x. [DOI] [PubMed] [Google Scholar]
- Syverson R. E., Buckley H., Gibian J., Ryan G. M., Jr Cellular and humoral immune status in women with chronic Candida vaginitis. Am J Obstet Gynecol. 1979 Jul 15;134(6):624–627. doi: 10.1016/0002-9378(79)90641-0. [DOI] [PubMed] [Google Scholar]
- Tsukada H., Kawamura I., Arakawa M., Nomoto K., Mitsuyama M. Dissociated development of T cells mediating delayed-type hypersensitivity and protective T cells against Listeria monocytogenes and their functional difference in lymphokine production. Infect Immun. 1991 Oct;59(10):3589–3595. doi: 10.1128/iai.59.10.3589-3595.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Twomey J. J., Waddell C. C., Krantz S., O'reilly R., L'esperance P., Good R. A. Chronic mucocutaneous candidiasis with macrophage dysfunction, a plasma inhibitor, and co-existent aplastic anemia. J Lab Clin Med. 1975 Jun;85(6):968–977. [PubMed] [Google Scholar]
- Witkin S. S., Hirsch J., Ledger W. J. A macrophage defect in women with recurrent Candida vaginitis and its reversal in vitro by prostaglandin inhibitors. Am J Obstet Gynecol. 1986 Oct;155(4):790–795. doi: 10.1016/s0002-9378(86)80022-9. [DOI] [PubMed] [Google Scholar]
- Witkin S. S. Inhibition of Candida-induced lymphocyte proliferation by antibody to Candida albicans. Obstet Gynecol. 1986 Nov;68(5):696–699. [PubMed] [Google Scholar]
- Yssel H., Shanafelt M. C., Soderberg C., Schneider P. V., Anzola J., Peltz G. Borrelia burgdorferi activates a T helper type 1-like T cell subset in Lyme arthritis. J Exp Med. 1991 Sep 1;174(3):593–601. doi: 10.1084/jem.174.3.593. [DOI] [PMC free article] [PubMed] [Google Scholar]



