Bile acids have long been known to facilitate digestion and absorption of lipids in the small intestine as well as regulate cholesterol homeostasis (1,2). Over the last decade, however, it has become clear that bile acids are not simply digestive detergents and the primary route governing cholesterol catabolism. Bile acids are now recognized as hormones involved in the regulation of various metabolic processes (3). Through activation of various signaling pathways, bile acids regulate not only their own synthesis and enterohepatic circulation, but also triglyceride, cholesterol, glucose, and energy homeostasis (2).
Manipulation of bile acid enterohepatic circulation by bile acid sequestration with nonsystemically absorbed resins can modulate the processes regulated by bile acids. Whereas bile acid sequestrants (BASs) have been used for over 40 years in the treatment of dyslipidemia (1), more recent data have emerged that have expanded their role in the treatment of dysglycemia in type 2 diabetes (4–9). The initial data suggesting such an effect were derived from post hoc analysis of a clinical trial for dyslipidemia that determined that BASs lowered glucose, particularly when compared with other lipid-lowering drugs (4). This concept was subsequently proven in several studies showing that BASs, such as colesevelam, lower glucose (5–8).
This review examines recent data exploring possible mechanisms involved in regulation of glucose metabolism by bile acids and the potential impact of disruption of their enterohepatic circulation on diabetes. We will also summarize the available clinical trial data that supported the regulatory approval of colesevelam for the treatment of hyperglycemia in type 2 diabetes.
TRADITIONAL ROLE OF BILE ACIDS: DIGESTION, EXCRETION, AND AUTOREGULATION
Bile acids are potent “digestive surfactants” that promote absorption of lipids (including fat-soluble vitamins), acting as emulsifiers (1,2).
Bile acids represent the primary pathway for cholesterol catabolism and account for ∼50% of the daily turnover of cholesterol (1). The synthesis of bile acids occurs exclusively in the liver in a series of enzymatic reactions in the hepatocyte that convert hydrophobic cholesterol into more water-soluble amphiphatic compounds (2). The production of bile acids is localized primarily in the perivenous hepatocytes, that is, the cells surrounding the central hepatic vein (10).
The immediate products of the bile acid synthetic pathways are referred to as primary bile acids. Cholic acid and chenodeoxycholic acid are the primary bile acids formed in humans. The action of intestinal bacterial flora on primary bile acids results in the formation of secondary bile acid species: deoxycholic and lithocholic acids, derived from cholic acid and chenodeoxycholic acid, respectively (2).
The steps leading to formation of primary bile acids include hydroxylation of cholesterol, catalyzed by the cytochrome P450 enzyme cholesterol 7α-hydroxylase (CYP7A1), the first and rate-limiting step of the so-called classic or neutral pathway of bile acid biosynthesis (1,2,11,12). The activity of CYP7A1 is subject to complex modes of control. The conversion of cholesterol to bile acids is primarily determined by this pathway (2).
Bile acid synthesis can also occur by an “alternative” or “acidic” pathway, which is governed by the enzyme CYP27A1 and converts oxysterols to bile acids (1,2). Unlike CYP7A1, CYP27A1 is not regulated by bile acids (2). It is estimated that only 6% of bile acid synthesis occurs via this pathway (13), but data also suggest that under certain conditions, such as fetal development (14) and chronic liver disease (13), this pathway may contribute more significantly to bile acid synthesis. The subsequent conversion of bile acid intermediates from either the classical or alternative pathways to cholic acid or chenodeoxycholic acid is governed by CYP8B1; interaction of these intermediates with this enzyme determines the amount of cholic acid versus chenodeoxycholic acid formed. Hydroxylation via CYP8B1 results in the formation of the more hydrophilic cholic acid molecule. Thus, the cholic acid/chenodeoxycholic acid ratio determines the overall hydrophobicity (and biological properties) of the bile acids pool (2).
Before their secretion into the bile canalicular lumen for storage in the gallbladder as mixed micelles with phospholipids and cholesterol, primary bile acids are conjugated with taurine or glycine, further enhancing their hydrophilicity (2). Upon ingestion of a meal, gallbladder contraction releases micellar bile acids into the intestinal lumen to aid digestion. Enterohepatic circulation enables 95% of bile acids to be reabsorbed from the distal ileum and transported back to the liver via the portal circulation. Interestingly, the perivenous hepatocytes, which account for the production of bile acids, are not involved in the reuptake of bile acids; bile acids are taken up and transported primarily by pericentral hepatocytes that surround the portal triads, where portal blood enters the liver acinus (15). The zonation differences accounting for where bile acids are produced and reenter the liver are relatively unexplored; thus, the (patho)physiological relevance of these observations is unknown at this time (2). Only ∼5% of bile acids are not reabsorbed and are eliminated in the feces. This small amount of loss is replenished via de novo synthesis of bile acids in the liver (1,2).
The size of the bile acid pool is tightly regulated within the liver and intestine to prevent cytotoxic accumulation of bile acids (2). As the bile acid pool size increases, a feedback mechanism, governed by the interplay of several nuclear receptors, is activated to inhibit de novo bile acid synthesis. In the liver, the nuclear receptor living receptor homolog (LRH)-1 activates gene transcription of the CYP7A1 gene (2). In 1999, bile acids were identified as the natural ligands for the farnesoid X receptor (FXR). By binding to the nuclear receptor FXR, bile acids mediate control of their own synthesis (16,17). FXR is thus a “bile acid sensor.” FXR can be activated by both primary and secondary conjugated bile acids, but chenodeoxycholic acid appears to be the most potent natural bile acid ligand (16,17). FXR functions as a biological regulator of bile acid synthesis through its transcriptional induction of the inhibitory nuclear receptor SHP (2). In the liver, small heterodimer partner (SHP) exerts its inhibitory effect by interacting with LRH-1 and subsequently repressing CYP7A1 transcription activation by LRH-1 (2). Bile acids can also inhibit transcription of CYP7A1 by repressing another nuclear receptor, hepatocyte nuclear factor (HNF)-4α (2). Intestinal FXR activation due to transintestinal bile acid flux after a meal also induces the expression of fibroblast growth factor (FGF)-19, which is released by small intestine epithelial cells and circulates to bind to hepatocyte FGF receptor 4 (FGFR4) receptors that signal a reduction in bile acid synthesis via c-Jun NH2-terminal kinase (JNK) pathway activation (2). Repression of CYP7A1 results in decreased synthesis of bile acids from intrahepatic cholesterol in response to the daily feeding-fasting cycle. Finally, emerging evidence suggests that expression of intestinal bile acid–binding protein (IBABP), which may be involved in the shuttling of bile acids from the apical to basolateral side of enterocytes on reabsorption, as well as Na+ taurocholate cotransporting polypeptide (NTCP), which uptakes bile acids returning to the liver, may also be under partial FXR control (2). Thus, FXR activation serves as a critical modulator of the enterohepatic circulation and de novo synthesis of bile acids to provide tight regulation of the bile acid pool (2).
FXR: BEYOND BILE ACID METABOLISM/HOMEOSTASIS
Lipid metabolism
Whereas manipulation of bile acid metabolism by bile acid sequestration has been recognized as a means to control systemic lipid concentrations since the 1960s, the underlying molecular mechanisms linking bile acids and lipid metabolism have only begun to be unraveled over the last decade. BASs, as well as ileal resection, which both interrupt the enterohepatic circulation of bile acids, decrease plasma total and LDL cholesterol while increasing levels of HDL cholesterol, apolipoprotein (apo)-AI, and triglycerides (18–21).
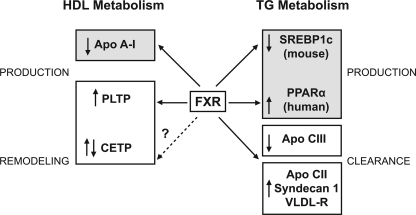
As a direct consequence of interrupting the return of bile acids to the liver, CYP7A1 expression becomes de-repressed, and conversion of cholesterol into bile acids is stimulated. The depletion of hepatic cholesterol due to increased diversion to bile acid synthesis leads to increased hepatic LDL receptor expression to harvest cholesterol from the systemic circulation (18). It is this indirect effect on LDL receptor expression that accounts for the decline in total and LDL cholesterol produced by BASs or ileal resection. However, the increase in HDL cholesterol and triglyceride levels observed with interruption of the enterohepatic circulation of bile acids cannot be explained by changes in LDL receptor expression. Animal data have revealed an independent regulatory role for FXR in both HDL cholesterol and triglyceride metabolism. With regard to HDL cholesterol, FXR represses apoAI expression (22) and plays a role in HDL particle remodeling through induction of phospholipid transfer protein (23). FXR activation increases clearance of triglycerides by influencing lipoprotein lipase (LPL) activity through induction of apoC-II expression (24) and repression of apoC-III (25) and by inducing peroxisome proliferator–activated receptor-α expression (2) (Fig. 1).
Figure 1.
Summary of the impact of FXR on lipid metabolism. FXR activation by bile acids induces the expression of sterol regulatory element–binding protein-1c (SREBP1c) in mice and peroxisome proliferator–activated receptor-α (PPARα) in humans, which both will modulate triglyceride (TG) production. However, the increase of apoC-II, VLDL receptor, and syndecan 1 gene expression together with the repression of apoC-III will increase LPL activity and therefore triglyceride clearance. FXR also represses apoAI expression and therefore HDL levels but also enhances the remodeling of HDL particles by induction of phospholipid transfer protein (PLTP) and perhaps cholesteryl ester transfer protein (CETP) expression. Used with permission from Claudel et al. (10).
Glucose metabolism
The first clinical indication that manipulation of the bile acid pool plays a role in glucose homeostasis resulted from observations made in a small study conducted by Garg and Grundy (4). In this study, the efficacy of 8 g b.i.d. of cholestyramine or placebo was evaluated in a crossover fashion over 12 weeks in 21 patients with type 2 diabetes stabilized on insulin or glyburide, with a baseline LDL cholesterol >3 mmol/l (>130 mg/dl) and triglycerides <8 mmol/l (<300 mg/dl). Unexpectedly, cholestyramine was associated with a modest improvement in glycemic control, with mean plasma glucose values lowered by 13% and a median reduction in urinary glucose excretion of 0.22 g/day (P < 0.001) and a trend toward lower glycated hemoglobin concentrations. These changes occurred without a dosage adjustment for insulin or glyburide. These results were later corroborated with colesevelam and colestimide (5–9). Similar data on the glucose-lowering effects of BASs and ileal biliary diversion have been observed in animal studies as well (26).
Mechanistic data on the influence of bile acids on glucose metabolism have been mounting. Effects on bile acid pool composition, FXR-mediated alterations in hepatic glucose production and intestinal glucose absorption, influences on peripheral insulin sensitivity, incretin effects, and energy use may all contribute to glucose regulation.
There is evidence that the bile acid pool size and composition are altered in animal models of either type 1 or type 2 diabetes (27–29) as well as in humans with type 1 or 2 diabetes (30,31). The mechanisms underlying these observations are unclear, but preliminary evidence suggests a role for insulin (32) and glucose (33) in modulation of bile acid synthesis. In the most extensive evaluation of bile acid kinetics to date in age- and BMI-matched subjects with type 2 diabetes (31), a higher rate of total bile acids synthesis, driven by an elevated rate of cholic acid synthesis and subsequent conversion to deoxycholic acid, was noted. Whether this alteration in bile acid pool composition could play a role in abnormal metabolism in diabetes remains speculative, but it is an intriguing possibility.
The type 1 diabetes rat model induced by streptozotocin is associated with an increased bile acid pool size due to enhanced synthesis. Hepatic FXR expression in this model is decreased. As FXR negatively regulates CYP7A1 activity, a molecular link between decreased expression of FXR and increased bile acid pool size in this model is suggested, since CYP7A1 mRNA levels are increased. It is also noteworthy that insulin represses FXR gene expression, whereas glucose produces the opposite effect (33). Together, these results suggest that diabetes is associated with a dysregulation of FXR expression.
More recent work in genetically modified FXR knockout mice has begun to clarify the role of FXR in glucose metabolism and carbohydrate use. FXR appears to play a role in modifying carbohydrate-induced gene expression as well as hepatic glucose production during postprandial and fasting hepatic glucose utilization, respectively. The precise mechanisms underlying FXR's regulation of glucose metabolism in the liver are only beginning to be elucidated, but FXR appears to be involved in the regulation of a complex array of gluconeogenic genes (2), such as those encoding phosphoenolpyruvate carboxykinase, fructose-1,6-biphosphatase-1, and glucose-6-phosphatase in vitro (34). FXR appears to be necessary for preventing fasting hypoglycemia through maintenance of postprandial hepatic glucose production and glycogen storage (35). Moreover, FXR activation inhibits the induction of glucose-responsive genes, such as L-type pyruvate kinase (L-PK), in the postprandial state (36). Because bile acid release is stimulated by meal ingestion, it is likely that FXR is activated during the enterohepatic recycling of bile acids (36). How diabetic alterations in the bile acid pool affect FXR activation remains to be elucidated and is an area of active investigation.
Recently, FXR was found to modulate glucose absorption in the proximal intestine, since FXR-deficient mice displayed delayed glucose absorption (37).
Although FXR is primarily localized in the liver and intestine, it should also be noted that it is expressed in peripheral tissues, including adipose tissue, adrenal glands, and skin (2). Whether minute concentrations of bile acids escaping the first pass from the liver, which are measurable in plasma, activate peripheral FXR is unclear. The physiologic role for peripheral FXR is only beginning to be understood, but in adipocytes, for example, FXR appears to play a role in differentiation and maturation (38). There are also emerging data to suggest a role in peripheral insulin sensitivity, which may be under partial regulation by FXR as well (38–40). Nevertheless, these data may suggest a future role for peripheral FXR activation in the treatment of insulin resistance and diabetes.
Bile acids may play an additional role in modulating incretin release through binding to a recently identified G-protein–coupled cell surface receptor known as TGR5 (41). This receptor is expressed in multiple tissues, including the gallbladder, liver, intestine, brown adipose tissue, central nervous system, and monocytes/macrophages (41). Lithocholic acid appears to be the most potent bile acid agonist for the receptor (41). TGR5 biology is incompletely understood, but it may play a role in immune modulation and hepatocyte protection from the cytotoxic effects of bile acids (41). Bile acid activation of TGR5 was also recently shown to induce intestinal glucagon-like peptide (GLP)-1 secretion (42). In type 2 diabetic patients, bile acid sequestration with colestimide may also increase GLP-1 release (43). This observation has lent support to the involvement of bile acids in mediating the enteroinsular response to feeding.
Bile acids may also play a role in metabolic regulation through modulation of energy expenditure. This effect appears to be mediated through modulation of thermogenesis. For example, bile acids given to high fat–fed mice increase energy expenditure in brown adipose tissue, preventing obesity and insulin resistance. This effect appears to be mediated by induction of the cAMP-dependent thyroid hormone–activating enzyme type 2 iodothyronine deiodinase (D2); bile acids increase D2 activity and oxygen consumption in brown adipose tissue, an effect believed to be mediated by TGR5, not FXR (44). Support for bile acids mediating energy expenditure has been provided by a study in FXR knockout mice. Intriguingly, when these mice were fasted, they exhibited accelerated entry into torpor; this appeared to be associated with an impaired ability to mobilize energy substrates (glucose and free fatty acids) (45). These results suggest that bile acids may also play a complementary role in thermogenesis through FXR-mediated regulation of energy substrate mobilization and storage (45).
CLINICAL UTILITY OF BILE ACID SEQUESTRATION: LEVERAGING THE METABOLIC EFFECTS OF BILE ACIDS THROUGH MANIPULATION OF THE BILE ACID POOL
As discussed previously, manipulation of the bile acid pool through bile acid sequestration to alter bile acid metabolism has been used since the 1960s to treat dyslipidemia (1). BASs deplete the bile acid pool by ∼40% and can increase bile acid synthesis over 15-fold (1). This increases diversion of hepatic cholesterol to bile acid formation, which indirectly lowers LDL cholesterol by enhancing hepatic LDL receptor expression. Lipid alterations produced through bile acid sequestration have been shown to reduce cardiovascular morbidity and mortality in high-risk males (46) and have also been shown to induce atherosclerotic plaque regression alone or in combination with other dyslipidemia treatments. A summary of these data are provided in Table 1 (1, used with permission).
Table 1.
Bile acid sequestrant therapy: summary of cardiovascular outcome and plaque regression clinical trials
| Study | Agents | n | Men (%) | Study duration (years) | LDL cholesterol reduction (%) | Patients with cardiovascular events (%) | Patients with coronary artery disease progression (%)* | Patients with coronary artery disease regression (%)* |
|---|---|---|---|---|---|---|---|---|
| BAS monotherapy | ||||||||
| Dorr et al. (47) | Colestipol | 1,149 | 48 | 2† | −12‡ | 4§‖ | ND | ND |
| Placebo | 1,129 | 48 | −2‡ | 9‖ | ND | ND | ||
| Lipid Research Clinics Coronary Primary Prevention Trial (46,48) | Cholestyramine | 1,906 | 100 | 7.4† | −20§ | 8§¶ | ND | ND |
| Placebo | 1,900 | 100 | −8 | 10¶ | ND | ND | ||
| National Heart, Lung, and Blood Institute Type II Coronary Intervention Study (20,21) | Cholestyramine | 59 | 81 | 5 | −26§ | ND | 32§# | 7 |
| Placebo | 57 | 81 | −5 | ND | 49# | 7 | ||
| St Thomas' Atherosclerosis Regression Study (49) | Cholestyramine + diet | 24 | 100 | 3.3† | −36**†† | 4§ | 12§ | ND |
| Diet | 26 | 100 | −16**†† | 11‡ | 15 | ND | ||
| Usual care | 24 | 100 | 0 | 36 | 46 | ND | ||
| BAS combination therapy | ||||||||
| Cholesterol Lowering Atherosclerosis Study (50) | Colestipol + niacin | 94 | 100 | 2 | −43§** | 25 | 10§‡‡ | ND |
| Placebo | 94 | 100 | −5** | 25 | 22‡‡ | ND | ||
| Familial Atherosclerosis Treatment Study (51) | Colestipol + niacin | 36 | 100 | 2.5 | −32**†† | 4§ | 25§# | 39§# |
| Colestipol + lovastatin | 38 | 100 | −46**†† | 7§ | 21§# | 32§# | ||
| Usual care | 46 | 100 | −7** | 19 | 46# | 11# | ||
| Kane et al. (52) | Colestipol + niacin + lovastatin | 40 | 45 | 2.2 | −38§ | ND | 20§§ | 33 |
| Control (plus low-dose colestipol [14/32 patients]) | 32 | 41 | −11 | ND | 41§§ | 13 | ||
| Harvard Atherosclerosis | ||||||||
| Reversibility Project (53) | Stepwise: pravastatin + niacin + cholestyramine + gemfibrozil | 40 | 90 | 2.5 | −38§ | 14 | 33# | 13# |
| Placebo | 30 | 87 | +3 | 21 | 38# | 15# | ||
| Probucol Quantitative | ||||||||
| Regression Swedish Trial (54) | Cholestyramine + probucol | 138 | 57 | 3 | −3§ | 28 | ND | 0.6, 3‖‖ |
| Cholestyramine + placebo | 136 | 58 | +8 | 21 | ND | 4**, 4‖‖ | ||
| Armed Forces Regression | ||||||||
| Study (55) | Cholestyramine + niacin + gemfibrozil | 71 | 90 | 2.5 | −22§ | 13§ | 30¶¶ | 52 |
| Placebo | 72 | 94 | +5 | 26 | 50¶¶ | 42 | ||
| Partial ileal bypass | ||||||||
| Program on the Surgical Control of the Hyperlipidemias (19) | Partial ileal bypass | 421 | 91 | 10 | −39†† | 19§##*** | 55§††† | 6†† |
| Control | 417 | 91 | −6†† | 30 | 85 | 4†† |
*Includes patients who may have also had regression/progression, except where indicated.
†Mean follow-up time reported.
‡Total cholesterol levels reported because LDL cholesterol levels not available.
§P < 0.05 compared with placebo, usual care, or control.
‖Percentage represents only the men enrolled in the study (n = 1,094). Differences were nonsignificant for women.
¶Only coronary artery disease deaths and nonfatal myocardial infarctions are included. Risk reduction was 19% relative to the incidence of cardiovascular events in the placebo-treated group.
#Definite or probable progression with no regression or regression with no progression.
**P < 0.05 compared with baseline.
††Statistical comparisons not conducted between treatment groups.
‡‡Number represents percentage of patients with new lesions in native vessels.
§§Although not statistically significant, there was a strong trend towards favoring active treatment over control.
‖‖Numbers represent % increase in femoral artery lumen volume from baseline and % decrease in roughness of arterial edge.
¶¶Statistical comparisons were only conducted for the percentage of patients who had “controlled” coronary artery disease (that is, patients who had regression or no change). That comparison (70 vs. 50% for drug therapy vs. placebo) was significant (P < 0.05).
##Only coronary artery disease deaths and nonfatal myocardial infarctions are included.
***Represents a 35% risk reduction.
†††Data on 10 years of follow-up reported; a significant difference was also observed after 3, 5, and 7 years of follow-up. ND, not determined (values were not determined or not reported). Used with permission from Insull (1).
The precise molecular mechanisms involved in BAS modulation of the bile acid pool and subsequent effects on glucose metabolism in diabetes are only beginning to be understood and are the subject of ongoing investigation. In a recently reported study of subjects with type 2 diabetes, colesevelam-associated reductions in A1C and fasting glucose did not appear to be related to an improvement in peripheral glucose disposal rate, but were associated with improvements in total-body insulin sensitivity, as determined by an improvement in the Matsuda index. Moreover, data from meal tolerance tests showed that colesevelam treatment resulted in reductions in fasting glucose and systemic glucose exposure (area under the curve for glucose [AUCg]) without significant changes in insulin levels. These results suggest that colesevelam may have effects on both insulin sensitivity and insulin secretion (Schwartz SL et al. Effect of colesevelam on postprandial glucose levels in subjects with type 2 diabetes mellitus. Abstract. Sixth Annual World Congress on the Insulin Resistance Syndrome, Los Angeles, CA, 25–27 September 2008). In another preliminary study in db/db diabetic mice, colesevelam administration improved insulin secretory response in the presence of hyperglycemia. Moreover, microarray analysis of genes abnormally expressed in the ileum, liver, skeletal muscle, and adipose tissue of db/db diabetic mice showed a partial or complete normalization of expression in the db/db diabetic mice relative to controls, particularly in the ileum, after treatment with colesevelam (Forman BM et al. Colesevelam affects gene expression in metabolic tissues in db/db mice. Abstract. American Diabetes Association 68th Annual Scientific Sessions, San Francisco, CA, 6–10 June 2008). Whether the genes dysregulated and subsequently normalized in the db/db diabetic mice reflect disruption of FXR and TGR5 pathways (among other bile acid–influenced and non-bile acid–influenced pathways) requires further study.
It is possible that alterations in bile acid pool composition produced by BASs may be involved in the effects observed with colesevelam on gene regulation and metabolism described above. The binding characteristics of a given BAS may have long-term effects on the bile acids pool by selectively depleting certain bile acid species through fecal elimination, resulting in an altered ratio of bile acid species in the pool (56). Preliminary animal data suggest that alteration of the bile acids pool can induce profound alterations in bile acid synthesis, which in turn may modulate multiple metabolic processes. In a study of CYP8B1 knockout mice, changing the composition of the bile acids pool by preventing the generation of cholic acid resulted in numerous alterations in bile acid metabolism that included a striking elevation in CYP7A1 expression and reduced SHP expression, but no alteration in expression of the bile salt export protein (57). Conventional BASs (cholestyramine and colestipol) have been shown to markedly alter the composition of bile. For example, cholestyramine preferentially binds the more hydrophobic bile acids chenodeoxycholic acid and deoxycholic acid over the more hydrophilic cholic acid and, therefore, over time causes a shift in the bile acids pool to one that is depleted in chenodeoxycholic acid and deoxycholic acid and enriched in cholic acid (56). The altered bile acid ratio also affects the relative degree of hydrophilicity of the bile acid pool; cholic acid enrichment results in a more hydrophilic bile acid pool. In addition to binding chenodeoxycholic acid and deoxycholic acid, colesevelam differs from cholestyramine and colestipol in that it binds cholic acid more effectively (58). Thus, the bile composition produced by colesevelam may differ from first-generation BASs, such as cholestyramine. The impact of bile acid sequestration on further modification (or normalization) of bile acid pool composition is currently under investigation (29).
Based on the preliminary observations of Garg and Grundy (4), BASs have also been discovered to have utility in the treatment of type 2 diabetes; colesevelam specifically received a labeled indication as adjunctive treatment of this condition in 2008. In addition to its well-characterized effects on lipid metabolism, colesevelam also produced consistent modest incremental reductions in A1C of up to 0.8% in addition to LDL cholesterol reductions of up to 17% when given to type 2 diabetic subjects inadequately controlled on stable regimens of metformin, sulfonylureas, or insulin (6–8). Reductions in A1C resulting from the addition of colesevelam are similar to changes observed with other adjunctive antidiabetic therapies in subjects with comparable baseline A1C values of 8–9%, with the added advantage of a neutral effect on weight. Side effects across the pivotal trials were mild and included constipation, nausea, and dyspepsia. A detailed summary of these data are provided in Table 2. Another BAS, colestimide, has also demonstrated effects on glucose metabolism in an animal model of diet-induced obesity and insulin resistance, suggesting that BASs might also find clinical application in the treatment of metabolic syndrome or pre-diabetes (60). This represents a potentially attractive therapeutic role for these drugs, considering that they are not systemically absorbed and have a good safety record, which may be attractive for long-term preventive use.
Table 2.
Clinical studies showing glucose-lowering effects of BASs in patients with type 2 diabetes
| Reference | Study design | Treatment | Duration | n | Background antidiabetic therapy | A1C (%) |
Treatment Difference | Fasting plasma glucose (mmol/l) |
Treatment Difference | LDL cholesterol (% Δ) |
Treatment Difference |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Δ from Baseline | Δ from Baseline | Δ from Baseline | |||||||||
| Cholestyramine | |||||||||||
| Garg and Grundy (4) | R, B, PC, and crossover | Placebo | 6 weeks | 21 | Added to insulin (n = 9) or glyburide (n = 12) | Week 6: −0.4* | −0.5* | Week 6: +0.3† | NR | Week 6: −1.9 | −28‡ |
| Cholestyramine 16 g/day | 19 | Week 6: −0.9* | Week 6: −0.8†‡ | Week 6: −28.9 | |||||||
| Colesevelam HCl | |||||||||||
| Zieve et al. (5) | R, DB, PC, and PG | Placebo | 12 weeks | 34 | Added to SU alone (n = 27), MET alone (n = 9), SU + MET (n = 29) | Week 12: +0.2 | −0.5‡ | Week 12: +0.12 | −0.78 | Week 12: +2.1‖ | −11.7‡ |
| Colesevelam 3.75 g/day | 31 | Week 12: −0.3 | Week 12: −0.28 | Week 12: −9.6 | |||||||
| Goldberg et al. (8) | R, DB, PC, and PG | Placebo | 16 weeks | 147 | Added to insulin alone (n = 116) or in combination with OAD(s) (n = 171) | Week 16: +0.1 | −0.5‡ Total cohort −0.6‡ Insulin alone cohort −0.4‡ Insulin + OAD(s) cohort | Week 16: +1.1 | −0.81 | Week 16: +0.5 | −12.8‡ |
| Colesevelam 3.75 g/day | 140 | Week 16: −0.4 | Week 16: −0.22 | Week 16: −12.3‖ | |||||||
| Fonseca et al. (7) | R, DB, PC, and PG | Placebo | 26 weeks | 231 | Added to SU alone (n = 156) or in combination with other OAD(s) (n = 304) | Week 26: +0.2 | −0.5‡ Total cohort −0.8‡ SU alone cohort −0.4‡ SU + OAD(s) cohort | Week 26: +0.39 | −0.75§ | Week 26: +0.6 | −16.7‡ |
| Colesevelam 3.75 g/day | 230 | Week 26: −0.3 | Week 26: −0.31 | Week 26: −16.1‖ | |||||||
| Bays et al. (6) | R, DB, PC, and PG | Placebo | 26 weeks | 157 | Added to MET alone (n = 159) or in combination with other OAD(s) (n = 157) | Week 26: +0.2 | −0.5‡ Total cohort −0.5‡ MET alone cohort −0.6‡ MET + OAD(s) cohort | Week 26: +0.64 | −0.77§ | Week 26: +4 | −15.9‡ |
| Colesevelam 3.75 g/day | 159 | Week 26: −0.4 | Week 26: −0.26 | Week 26: −12‖ | |||||||
| Colestimide | |||||||||||
| Yamakawa et al. (9) | R, open label, and PG | Pravastatin 10 mg/day | 3 months | 35 | Added to Diet (n = 4), OAD monotherapy (n = 17), OAD combination therapy (n = 34), and insulin (n = 17) | NS | NR | NS | NR | Month 3: −16.2‖ | NR |
| Colestimide 6 g/day | 35 | Month 3: −0.9‖ | Month 3: −0.8‖ | Month 3: −22.9‖ | |||||||
| Suzuki et al. (59) | R, open label, and PG | Acarbose 150 mg/day | 2 weeks | 16 | Diet alone (n = 18), diet + OAD (n = 15) | Week 2: NR# | NR | Week 2: −0.44‖ | NR | NR** | NR |
| Colestimide 3 g/day | 17 | Week 2: NR# | Week 2: −0.48 | NR** |
*Glycosylated hemoglobin; values represent the mean of two determinations on days 28 and 38 during the study periods.
†Mean plasma glucose; for each patient, a mean value was calculated from plasma glucose concentrations measured at 3:00, 7:00, and 11:00 a.m. and at 4:00 and 8:00 p.m. each day for 5 consecutive days.
‡P ≤ 0.007 vs. placebo.
§P < 0.05 vs. placebo.
‖P < 0.05 vs. baseline.
¶No significant change from baseline; values are not reported.
#Because of the short duration of the study, A1C values were not examined, but M value and J index decreased significantly in both groups (P < 0.0001 vs. baseline).
**LDL cholesterol levels not reported, but total cholesterol levels decreased significantly in both groups versus baseline (P ≤ 0.002), with no significant change in HDL cholesterol, suggesting a reduction in LDL cholesterol. DB, double-blind; MET, metformin; NR, not reported; OAD, oral antidiabetic; PC, placebo controlled; PG, parallel group; R, randomized; SU, sulfonylurea.
CONCLUSIONS
Knowledge of bile acid physiology has dramatically evolved from the concept of digestive detergents to an elegant story of bile acid functioning as hormones involved in the modulation of a variety of metabolic processes. Manipulation of bile acids composition and pool size through bile acid sequestration takes advantage of this physiology and has found clinical application for dyslipidemia and, more recently, type 2 diabetes. Further research will continue to refine our knowledge of bile acid physiology and will contribute to potential additional therapeutic applications for these complex molecules.
Acknowledgments
B.S. was funded by grants from Agence Nationale de la Recherche (A05056GS) and Hepatic and Adipose Tissue and Functions in the Metabolic Syndrome (HEPADIP) (018734). Funding for manuscript preparation was provided by Daiichi Sankyo, Inc.
No other potential conflicts of interest relevant to this article were reported.
Editorial assistance was provided by Eleanor O'Rangers, PharmD.
Footnotes
The publication of this supplement was made possible in part by unrestricted educational grants from Eli Lilly, Ethicon Endo-Surgery, Generex Biotechnology, Hoffmann-La Roche, Johnson & Johnson, LifeScan, Medtronic, MSD, Novo Nordisk, Pfizer, sanofi-aventis, and WorldWIDE.
References
- 1. Insull W, Jr: Clinical utility of bile acid sequestrants in the treatment of dyslipidemia: a scientific review. South Med J 2006; 99: 257– 273 [DOI] [PubMed] [Google Scholar]
- 2. Lefebvre P, Cariou B, Lien F, Kuipers F, Staels B: Role of bile acids and bile acid receptors in metabolic regulation. Physiol Rev 2009; 89: 147– 191 [DOI] [PubMed] [Google Scholar]
- 3. Houten SM, Watanabe M, Auwerx J: Endocrine function of bile acids. EMBO J 2006; 25: 1419– 1425 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Garg A, Grundy SM: Cholestyramine therapy for dyslipidemia in non-insulin-dependent diabetes mellitus: a short-term, double-blind, crossover trial. Ann Intern Med 1994; 121: 416– 422 [DOI] [PubMed] [Google Scholar]
- 5. Zieve FJ, Kalin MF, Schwartz SL, Jones MR, Bailey WL: Results of the Glucose-Lowering Effect of WelChol Study (GLOWS): a randomized, double-blind, placebo-controlled pilot study evaluating the effect of colesevelam hydrochloride on glycaemic control in subjects with type 2 diabetes. Clin Ther 2007; 29: 74– 83 [DOI] [PubMed] [Google Scholar]
- 6. Bays HE, Goldberg RB, Truitt KE, Jones MR: Colesevelam hydrochloride therapy in patients with type 2 diabetes mellitus treated with metformin. Arch Intern Med 2008; 168: 1975– 1983 [DOI] [PubMed] [Google Scholar]
- 7. Fonseca VA, Rosenstock J, Wang AC, Truitt KE, Jones MR: Colesevelam HCl improves glycemic control and reduces LDL cholesterol in patients with inadequately controlled type 2 diabetes on sulfonylurea-based therapy. Diabetes Care 2008; 31: 1479– 1484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Goldberg RB, Fonseca VA, Truitt KE, Jones MR: Efficacy and safety of colesevelam in patients with type 2 diabetes mellitus and inadequate glycemic control receiving insulin-based therapy. Arch Intern Med 2008; 168: 1531– 1540 [DOI] [PubMed] [Google Scholar]
- 9. Yamakawa T, Takano T, Utsunomiya H, Kadonosono K, Okamura A: Effect of colestimide therapy for glycemic control in type 2 diabetes mellitus with hypercholesterolemia. Endocr J 2007; 54: 53– 58 [DOI] [PubMed] [Google Scholar]
- 10. Twisk J, Hoekman MF, Mager WH, Moorman AF, de Boer PA, Scheja L, Princen HM, Gebhardt R: Heterogeneous expression of cholesterol 7α-hydroxylase and sterol 27-hydroxylase genes in the rat liver lobulus. J Clin Invest 1995; 95: 1235– 1243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Claudel T, Staels B, Kuipers F: The farnesoid X receptor: a molecular link between bile acid and lipid and glucose metabolism. Arterioscler Thromb Vasc Biol 2005; 25: 2020– 2031 [DOI] [PubMed] [Google Scholar]
- 12. Pullinger CR, Eng C, Salen G, Shefer S, Batta AK, Erickson SK, Verhagen A, Rivera CR, Mulvihill SJ, Malloy MJ, Kane JP: Human cholesterol 7α-hydroxylase (CYP7A1) deficiency has a hypercholesterolemic phenotype. J Clin Invest 2002; 110: 109– 117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Crosignani A, Del Puppo M, Longo M, De Fabiani E, Caruso D, Zuin M, Podda M, Javitt NB, Kienle MG: Changes in classic and alternative pathways of bile acid synthesis in chronic liver disease. Clinica Chimica Acta 2007; 382: 82– 88 [DOI] [PubMed] [Google Scholar]
- 14. Back P, Walter K: Developmental pattern of bile acid metabolism as revealed by bile acid analysis of meconium. Gastroenterology 1980; 78: 671– 678 [PubMed] [Google Scholar]
- 15. Groothuis GM, Hardonk MJ, Keulemans KP, Nieuwenhuis P, Meijer DK: Autoradiographic and kinetic demonstration of acinar heterogeneity of taurocholate transport. Am J Physiol 1982; 243: G455– G462 [DOI] [PubMed] [Google Scholar]
- 16. Makishima M, Okamoto AY, Repa JJ, Tu H, Learned RM, Luk A, Hull MV, Lustig KD, Mangelsdorf DJ, Shan B: Identification of a nuclear receptor for bile acids. Science 1999; 284: 1362– 1365 [DOI] [PubMed] [Google Scholar]
- 17. Parks DJ, Blanchard SG, Bledsoe RK, Chandra G, Consler TG, Kliewer SA, Stimmel JB, Willson TM, Zavacki AM, Moore DD, Lehmann JM: Bile acids: natural ligands for an orphan nuclear receptor. Science 1999; 284: 1365– 1368 [DOI] [PubMed] [Google Scholar]
- 18. Shepherd J, Packard CJ, Bicker S, Lawrie TD, Morgan HG: Cholestyramine promotes receptor-mediated low-density-lipoprotein catabolism. N Engl J Med 1980; 302: 1219– 1222 [DOI] [PubMed] [Google Scholar]
- 19. Buchwald H, Varco RL, Matts JP, Long JM, Fitch LL, Campbell GS, Pearce MB, Yellin AE, Edmiston WA, Smink RD, Jr, et al. : Effect of partial ileal bypass surgery on mortality and morbidity from coronary heart disease in patients with hypercholesterolemia: report of the Program on the Surgical Control of the Hyperlipidemias (POSCH). N Engl J Med 1990; 323: 946– 955 [DOI] [PubMed] [Google Scholar]
- 20. Brensike JF, Levy RI, Kelsey SF, Passamani ER, Richardson JM, Loh IK, Stone NJ, Alrich RF, Battaglini JW, Moriarty DJ, Fisher MR, Friedman L, Friedewald W, Detre KM, Epstein SE: Effects of therapy with cholestyramine on progression of coronary arteriosclerosis: results of the NHLBI Type II Coronary Intervention Study. Circulation 1984; 69: 313– 324 [DOI] [PubMed] [Google Scholar]
- 21. Levy RI, Brensike JF, Epstein SE, Kelsey SF, Passamani ER, Richardson JM, Loh IK, Stone NJ, Aldrich RF, Battaglini JW, Moriarty DJ, Fisher ML, Friedman L, Friedewald W, Detre KM: The influence of changes in lipid values induced by cholestyramine and diet on progression of coronary artery disease: results of the NHLBI Type II Coronary Intervention Study. Circulation 1984; 69: 325– 337 [DOI] [PubMed] [Google Scholar]
- 22. Claudel T, Sturm E, Duez H, Pineda Torra I, Sirvent A, Kosykh V, Fruchart JC, Dallongeville J, Hum DW, Kuipers F, Staels B: Bile acid-activated nuclear receptor FXR suppresses apolipoprotein A-I transcription via a negative FXR response element. J Clin Invest 2002; 109: 961– 971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Urizar NL, Dowhan DH, Moore DD: The farnesoid X-activated receptor mediates bile acid activation of phospholipid transfer protein gene expression. J Biol Chem 2000; 275: 39313– 39317 [DOI] [PubMed] [Google Scholar]
- 24. Kast HR, Nguyen CM, Sinal CJ, Jones SA, Laffitte BA, Reue K, Gonzalez FJ, Willson TM, Edwards PA: Farnesoid X-activated receptor induces apolipoprotein C-II transcription: a molecular mechanism linking plasma triglyceride levels to bile acids. Mol Endocrinol 2001; 15: 1720– 1728 [DOI] [PubMed] [Google Scholar]
- 25. Claudel T, Inoue Y, Barbier O, Duran-Sandoval D, Kosykh V, Fruchart J, Fruchart JC, Gonzalez FJ, Staels B: Farnesoid X receptor agonists suppress hepatic apolipoprotein CIII expression. Gastroenterology 2003; 125: 544– 555 [DOI] [PubMed] [Google Scholar]
- 26. Staels B, Kuipers F: Bile acid sequestrants and the treatment of type 2 diabetes mellitus. Drugs 2007; 67: 1383– 1392 [DOI] [PubMed] [Google Scholar]
- 27. Hassan AS, Ravi Subbiah MT, Thiebert P: Specific changes of bile acid metabolism in spontaneously diabetic Wistar rats. Proc Soc Exp Biol Med 1980; 164: 449– 452 [DOI] [PubMed] [Google Scholar]
- 28. Nervi FO, Severin CH, Valdivieso VD: Bile acid pool changes and regulation of cholate synthesis in experimental diabetes. Biochim Biophys Acta 1978; 529: 212– 223 [DOI] [PubMed] [Google Scholar]
- 29. Uchida K, Makino S, Akiyoshi T: Altered bile acid metabolism in nonobese, spontaneously diabetic (NOD) mice. Diabetes 1985; 34: 79– 83 [DOI] [PubMed] [Google Scholar]
- 30. Andersen E, Karlaganis G, Sjovalll J: Altered bile acid profiles in duodenal bile and urine in diabetic subjects. Eur J Clin Invest 1988; 18: 166– 172 [DOI] [PubMed] [Google Scholar]
- 31. Brufau G, Kuipers F, Prado K, Abbey S, Jones M, Schwartz S, Stellaard F, Murphy E: Altered bile salt metabolism in type 2 diabetes mellitus (T2DM) (Abstract). Diabetes 2008; 57 ( Suppl. 1); A435 [Google Scholar]
- 32. Michael MD, Kulkarni RN, Postic C, Previs SF, Shulman GI, Magnuson MA, Kahn CR: Loss of insulin signaling in hepatocytes leads to severe insulin resistance and progressive hepatic dysfunction. Mol Cell 2000; 6: 87– 97 [PubMed] [Google Scholar]
- 33. Duran-Sandoval D, Mautino G, Martin G, Percevault F, Barbier O, Fruchart JC, Kuipers F, Staels B: Glucose regulates the expression of the farnesoid X receptor in liver. Diabetes 2004; 53: 890– 898 [DOI] [PubMed] [Google Scholar]
- 34. Yamagata K, Daitoku H, Shimamoto Y, Matsuzaki H, Hirota K, Ishida J, Fukamizu A: Bile acids regulate gluconeogenic gene expression via small heterodimer partner-mediated repression of hepatocyte nuclear factor 4 and Foxo1. J Biol Chem 2004; 279: 23158– 23165 [DOI] [PubMed] [Google Scholar]
- 35. Cariou B, van Harmelen K, Duran-Sandoval D, van Dijk T, Grefhorst A, Bouchaert E, Fruchart JC, Gonzalez FJ, Kuipers F, Staels B: Transient impairment of the adaptive response to fasting in FXR-deficient mice. FEBS Lett 2005; 579: 4076– 4080 [DOI] [PubMed] [Google Scholar]
- 36. Duran-Sandoval D, Cariou B, Percevault F, Hennuyer N, Grefhorst A, van Dijk TH, Gonzalez FJ, Fruchart JC, Kuipers F, Staels B: The farnesoid X receptor modulates hepatic carbohydrate metabolism during the fasting/refeeding transition. J Biol Chem 2005; 280: 29971– 29979 [DOI] [PubMed] [Google Scholar]
- 37. van Dijk TH, Grefhorst A, Oosterveer MH, Blok VW, Staels B, Reijingoud DJ, Kuipers F: An increased flux through the glucose-6-phosphate pool in enterocytes delays glucose absorption in Fxr-/- mice. J Biol Chem 2009; 284: 10315– 10323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Cariou B, van Harmelen K, Duran-Sandoval D, van Dijk TH, Grefhorst A, Abdelkarim M, Caron S, Torpier G, Fruchart JC, Gonzalez FJ, Kuipers F, Staels B: The farnesoid X receptor modulates adiposity and peripheral insulin sensitivity in mice. J Biol Chem 2006; 281: 11039– 11049 [DOI] [PubMed] [Google Scholar]
- 39. Zhang Y, Lee FY, Barrera G, Lee H, Vales C, Gonzalez FJ: Activation of the nuclear receptor FXR improves hyperglycemia and hyperlipidemia in diabetic mice. Proc Natl Acad Sci U S A 2006; 103: 1006– 1011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Ma K, Saha PK, Chan L, Moore DD: Farnesoid X receptor is essential for normal glucose homeostasis. J Clin Invest 2006; 116: 1102– 1109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Thomas C, Auwerx J, Schoonjans K: Bile acids and the membrane bile acid receptor TGR5: connecting nutrition and metabolism. Thyroid 2008; 18: 167– 174 [DOI] [PubMed] [Google Scholar]
- 42. Katsuma S, Hirasawa A, Tsujimoto G: Bile acids promote glucagon-like peptide-1 secretion through TGR5 in a murine enteroendocrine cell line STC-1. Biochem Biophys Res Commun 2005; 329: 386– 390 [DOI] [PubMed] [Google Scholar]
- 43. Suzuki T, Oba K, Igari Y, Matsumura N, Watanabe K, Futami-Suda S, Yasuoka H, Ouchi M, Suzuki K, Kigawa Y, Nakano H: Colestimide lowers plasma glucose levels and increases plasma glucagon-like peptide-1 (7-36) levels in patients with type 2 diabetes mellitus complicated by hypercholesterolemia. J Nippon Med Sch 2007; 74: 338– 343 [DOI] [PubMed] [Google Scholar]
- 44. Watanabe M, Houten SM, Mataki C, Christoffolete MA, Kim BW, Sato H, Messadeq N, Harney JW, Ezaki O, Kodama T, Schoonjans K, Bianco AC, Auwerx J: Bile acids induce energy expenditure by promoting intracellular thyroid hormone activation. Nature 2006; 439: 484– 489 [DOI] [PubMed] [Google Scholar]
- 45. Cariou B, Bouchaert E, Abdelkarim M, Dumont J, Caron S, Fruchart JC, Burcelin R, Kuipers F, Staels B: FXR-deficiency confers increased susceptibility to torpor. FEBS Lett 2007; 581: 5191– 5198 [DOI] [PubMed] [Google Scholar]
- 46. The Lipid Research Clinics Coronary Primary Prevention Trial results. I. Reduction in incidence of coronary heart disease. JAMA 1984; 251: 351– 364 [DOI] [PubMed] [Google Scholar]
- 47. Dorr AE, Gundersen K, Schneider JC, Jr, Spencer TW, Martin WB: Colestipol hydrochloride in hypercholesterolemic patients: effect on serum cholesterol and mortality. J Chronic Dis 1978; 31: 5– 14 [DOI] [PubMed] [Google Scholar]
- 48. Lipid Research Clinics Coronary Primary Prevention Trial results. II. The relationship of reduction in incidence of coronary heart disease to cholesterol lowering. JAMA 1984; 251: 365– 374 [PubMed] [Google Scholar]
- 49. Watts GF, Lewis B, Brunt JN, Lewis ES, Coltart DJ, Smith LD, Mann JI, Swan AV: Effects on coronary artery disease of lipid-lowering diet, or diet plus cholestyramine, in the St Thomas' Atherosclerosis Regression Study (STARS). Lancet 1992; 339: 563– 569 [DOI] [PubMed] [Google Scholar]
- 50. Blankenhorn DH, Nessim SA, Johnson RL, Sanmarco ME, Azen SP, Cashin-Hemphill L: Beneficial effects of combined colestipol-niacin therapy on coronary atherosclerosis and coronary venous bypass grafts. JAMA 1987; 257: 3233– 3240 [PubMed] [Google Scholar]
- 51. Brown G, Albers JJ, Fisher LD, Schaefer SM, Lin JT, Kaplan C, Zhao XQ, Bisson BD, Fitzpatrick VS, Dodge HT: Regression of coronary artery disease as a result of intensive lipid-lowering therapy in men with high levels of apolipoprotein B. N Engl J Med 1990; 323: 1289– 1298 [DOI] [PubMed] [Google Scholar]
- 52. Kane JP, Malloy MJ, Ports TA, Phillips NR, Diehl JC, Havel RJ: Regression of coronary atherosclerosis during treatment of familial hypercholesterolemia with combined drug regimens. JAMA 1990; 264: 3007– 3012 [PubMed] [Google Scholar]
- 53. Sacks FM, Pasternak RC, Gibson CM, Rosner B, Stone PH: Effect on coronary atherosclerosis of decrease in plasma cholesterol concentrations in normocholesterolaemic patients. Lancet 1994; 344: 1182– 1186 [DOI] [PubMed] [Google Scholar]
- 54. Walldius G, Erikson U, Olsson AG, Bergstrand L, Hadell K, Johansson J, Kaijser L, Lassvik C, Molgaard J, Nilsson S, et al. : The effect of probucol on femoral atherosclerosis: the Probucol Quantitative Regression Swedish Trial (PQRST). Am J Cardiol 1994; 74: 875– 883 [DOI] [PubMed] [Google Scholar]
- 55. Whitney EJ, Krasuski RA, Personius BE, Michalek JE, Maranian AM, Kolasa MW, Monick E, Brown BG, Gotto AM, Jr: A randomized trial of a strategy for increasing high-density lipoprotein cholesterol levels: effects on progression of coronary heart disease and clinical events. Ann Intern Med 2005; 142: 95– 104 [DOI] [PubMed] [Google Scholar]
- 56. Benson GM, Haynes C, Blanchard S, Ellis D: In vitro studies to investigate the reasons for the low potency of cholestyramine and colestipol. J Pharm Sci 1993; 82: 80– 86 [DOI] [PubMed] [Google Scholar]
- 57. Li-Hawkins J, Gafvels M, Olin M, Lund EG, Andersson U, Schuster G, Bjorkhem I, Russell DW, Eggertsen G: Cholic acid mediates negative feedback regulation of bile acid synthesis in mice. J Clin Invest 2002; 110: 1191– 1200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Braunlin W, Zhorov E, Smisek D, Guo A, Appruzese W, Xu Q, Hook P, Holmes-Farley R, Mandeville H: In vitro comparison of bile acid binding to colesevelam HCl and other bile acid sequestrants. Polymer Preprints 2000; 41: 708– 709 [Google Scholar]
- 59. Suzuki T, Oba K, Futami S, Suzuki K, Ouchi M, Igari Y, Matsumura N, Watanabe K, Kigawa Y, Nakano H: Blood glucose-lowering activity of colestimide in patients with type 2 diabetes and hypercholesterolemia: a case-control study comparing colestimide with acarbose. J Nippon Med Sch 2006; 73: 277– 284 [DOI] [PubMed] [Google Scholar]
- 60. Kobayashi M, Ikegami H, Fujisawa T, Nojima K, Kawabata Y, Noso S, Babaya N, Itoi-Babaya M, Yamaji K, Hiromine Y, Shibata M, Ogihara T: Prevention and treatment of obesity, insulin resistance, and diabetes by bile acid-binding resin. Diabetes 2007; 56: 239– 247 [DOI] [PubMed] [Google Scholar]