Abstract
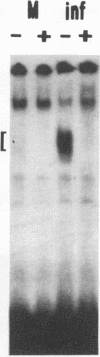
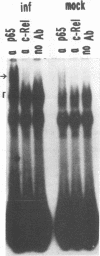
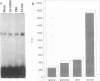
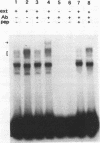
Although information about the genetic basis and mechanisms of Shigella flexneri cellular invasion is accumulating, little is known about changes in cell signaling and their consequences following bacterium-host cell interactions. A general result of signal transduction is alterations in the levels and/or activities of transcription factors. Alterations in transcription factor binding activities were observed following challenge with S. flexneri. Changes in the DNA-binding activities of cellular transcription factors to AP1, AP2, cyclic AMP response element, CTF1/NF1, NF-kappa B/Rel, OCT1, and SP1 DNA-binding sites were investigated by electrophoretic mobility shift assays. NF-kappa B/Rel DNA-binding activity was enhanced more than 11-fold by cellular invasion; noninvasive S. flexneri strains induced low levels of kappa B DNA binding. Both subunits of the NF-kappa B transcription factor, p50 and p65, but not c-Rel (p85), are components of the kappa B DNA-binding activity. These data suggest that changes in cellular transcription factor binding activity are a consequence of S. flexneri invasion, and these changes could play a role in the initial host response or in the pathogenesis of the disease.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arima N., Molitor J. A., Smith M. R., Kim J. H., Daitoku Y., Greene W. C. Human T-cell leukemia virus type I Tax induces expression of the Rel-related family of kappa B enhancer-binding proteins: evidence for a pretranslational component of regulation. J Virol. 1991 Dec;65(12):6892–6899. doi: 10.1128/jvi.65.12.6892-6899.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arnold J. W., Niesel D. W., Annable C. R., Hess C. B., Asuncion M., Cho Y. J., Peterson J. W., Klimpel G. R. Tumor necrosis factor-alpha mediates the early pathology in Salmonella infection of the gastrointestinal tract. Microb Pathog. 1993 Mar;14(3):217–227. doi: 10.1006/mpat.1993.1021. [DOI] [PubMed] [Google Scholar]
- Bachelerie F., Alcami J., Arenzana-Seisdedos F., Virelizier J. L. HIV enhancer activity perpetuated by NF-kappa B induction on infection of monocytes. Nature. 1991 Apr 25;350(6320):709–712. doi: 10.1038/350709a0. [DOI] [PubMed] [Google Scholar]
- Baeuerle P. A. The inducible transcription activator NF-kappa B: regulation by distinct protein subunits. Biochim Biophys Acta. 1991 Apr 16;1072(1):63–80. doi: 10.1016/0304-419x(91)90007-8. [DOI] [PubMed] [Google Scholar]
- Baudry B., Kaczorek M., Sansonetti P. J. Nucleotide sequence of the invasion plasmid antigen B and C genes (ipaB and ipaC) of Shigella flexneri. Microb Pathog. 1988 May;4(5):345–357. doi: 10.1016/0882-4010(88)90062-9. [DOI] [PubMed] [Google Scholar]
- Baudry B., Maurelli A. T., Clerc P., Sadoff J. C., Sansonetti P. J. Localization of plasmid loci necessary for the entry of Shigella flexneri into HeLa cells, and characterization of one locus encoding four immunogenic polypeptides. J Gen Microbiol. 1987 Dec;133(12):3403–3413. doi: 10.1099/00221287-133-12-3403. [DOI] [PubMed] [Google Scholar]
- Blank V., Kourilsky P., Israël A. NF-kappa B and related proteins: Rel/dorsal homologies meet ankyrin-like repeats. Trends Biochem Sci. 1992 Apr;17(4):135–140. doi: 10.1016/0968-0004(92)90321-y. [DOI] [PubMed] [Google Scholar]
- Clerc P., Sansonetti P. J. Entry of Shigella flexneri into HeLa cells: evidence for directed phagocytosis involving actin polymerization and myosin accumulation. Infect Immun. 1987 Nov;55(11):2681–2688. doi: 10.1128/iai.55.11.2681-2688.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cullen B. R. Regulation of HIV-1 gene expression. FASEB J. 1991 Jul;5(10):2361–2368. doi: 10.1096/fasebj.5.10.1712325. [DOI] [PubMed] [Google Scholar]
- Galán J. E., Pace J., Hayman M. J. Involvement of the epidermal growth factor receptor in the invasion of cultured mammalian cells by Salmonella typhimurium. Nature. 1992 Jun 18;357(6379):588–589. doi: 10.1038/357588a0. [DOI] [PubMed] [Google Scholar]
- Garcia-Blanco M. A., Cullen B. R. Molecular basis of latency in pathogenic human viruses. Science. 1991 Nov 8;254(5033):815–820. doi: 10.1126/science.1658933. [DOI] [PubMed] [Google Scholar]
- Gedrich R. W., Bayley S. T., Engel D. A. Induction of AP-1 DNA-binding activity and c-fos mRNA by the adenovirus 243R E1A protein and cyclic AMP requires domains necessary for transformation. J Virol. 1992 Oct;66(10):5849–5859. doi: 10.1128/jvi.66.10.5849-5859.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ginocchio C., Pace J., Galán J. E. Identification and molecular characterization of a Salmonella typhimurium gene involved in triggering the internalization of salmonellae into cultured epithelial cells. Proc Natl Acad Sci U S A. 1992 Jul 1;89(13):5976–5980. doi: 10.1073/pnas.89.13.5976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green P. L., Chen I. S. Regulation of human T cell leukemia virus expression. FASEB J. 1990 Feb 1;4(2):169–175. doi: 10.1096/fasebj.4.2.2404818. [DOI] [PubMed] [Google Scholar]
- Hale T. L., Bonventre P. F. Shigella infection of Henle intestinal epithelial cells: role of the bacterium. Infect Immun. 1979 Jun;24(3):879–886. doi: 10.1128/iai.24.3.879-886.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hale T. L., Morris R. E., Bonventre P. F. Shigella infection of henle intestinal epithelial cells: role of the host cell. Infect Immun. 1979 Jun;24(3):887–894. doi: 10.1128/iai.24.3.887-894.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haseltine W. A., Sodroski J., Rosen C. A. Control of gene expression and the replication and pathogenesis of retroviruses. Important Adv Oncol. 1986:159–172. [PubMed] [Google Scholar]
- Hess C. B., Niesel D. W., Klimpel G. R. The induction of interferon production in fibroblasts by invasive bacteria: a comparison of Salmonella and Shigella species. Microb Pathog. 1989 Aug;7(2):111–120. doi: 10.1016/0882-4010(89)90030-2. [DOI] [PubMed] [Google Scholar]
- Jones N. C., Rigby P. W., Ziff E. B. Trans-acting protein factors and the regulation of eukaryotic transcription: lessons from studies on DNA tumor viruses. Genes Dev. 1988 Mar;2(3):267–281. doi: 10.1101/gad.2.3.267. [DOI] [PubMed] [Google Scholar]
- Karin M. Signal transduction from cell surface to nucleus in development and disease. FASEB J. 1992 May;6(8):2581–2590. doi: 10.1096/fasebj.6.8.1317309. [DOI] [PubMed] [Google Scholar]
- Kretzschmar M., Meisterernst M., Scheidereit C., Li G., Roeder R. G. Transcriptional regulation of the HIV-1 promoter by NF-kappa B in vitro. Genes Dev. 1992 May;6(5):761–774. doi: 10.1101/gad.6.5.761. [DOI] [PubMed] [Google Scholar]
- Lenardo M. J., Baltimore D. NF-kappa B: a pleiotropic mediator of inducible and tissue-specific gene control. Cell. 1989 Jul 28;58(2):227–229. doi: 10.1016/0092-8674(89)90833-7. [DOI] [PubMed] [Google Scholar]
- Marriott S. J., Brady J. N. Enhancer function in viral and cellular gene regulation. Biochim Biophys Acta. 1989 Dec 17;989(2):97–110. doi: 10.1016/0304-419x(89)90037-1. [DOI] [PubMed] [Google Scholar]
- Maurelli A. T., Blackmon B., Curtiss R., 3rd Loss of pigmentation in Shigella flexneri 2a is correlated with loss of virulence and virulence-associated plasmid. Infect Immun. 1984 Jan;43(1):397–401. doi: 10.1128/iai.43.1.397-401.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maurelli A. T., Sansonetti P. J. Genetic determinants of Shigella pathogenicity. Annu Rev Microbiol. 1988;42:127–150. doi: 10.1146/annurev.mi.42.100188.001015. [DOI] [PubMed] [Google Scholar]
- McMahon S. B., Monroe J. G. Role of primary response genes in generating cellular responses to growth factors. FASEB J. 1992 Jun;6(9):2707–2715. doi: 10.1096/fasebj.6.9.1612295. [DOI] [PubMed] [Google Scholar]
- Mitchell P. J., Tjian R. Transcriptional regulation in mammalian cells by sequence-specific DNA binding proteins. Science. 1989 Jul 28;245(4916):371–378. doi: 10.1126/science.2667136. [DOI] [PubMed] [Google Scholar]
- Muchardt C., Seeler J. S., Nirula A., Shurland D. L., Gaynor R. B. Regulation of human immunodeficiency virus enhancer function by PRDII-BF1 and c-rel gene products. J Virol. 1992 Jan;66(1):244–250. doi: 10.1128/jvi.66.1.244-250.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muegge K., Durum S. K. Cytokines and transcription factors. Cytokine. 1990 Jan;2(1):1–8. doi: 10.1016/1043-4666(90)90036-s. [DOI] [PubMed] [Google Scholar]
- Nigg E. A. Mechanisms of signal transduction to the cell nucleus. Adv Cancer Res. 1990;55:271–310. doi: 10.1016/s0065-230x(08)60471-6. [DOI] [PubMed] [Google Scholar]
- Nolan G. P., Baltimore D. The inhibitory ankyrin and activator Rel proteins. Curr Opin Genet Dev. 1992 Apr;2(2):211–220. doi: 10.1016/s0959-437x(05)80276-x. [DOI] [PubMed] [Google Scholar]
- Oaks E. V., Wingfield M. E., Formal S. B. Plaque formation by virulent Shigella flexneri. Infect Immun. 1985 Apr;48(1):124–129. doi: 10.1128/iai.48.1.124-129.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pace J., Hayman M. J., Galán J. E. Signal transduction and invasion of epithelial cells by S. typhimurium. Cell. 1993 Feb 26;72(4):505–514. doi: 10.1016/0092-8674(93)90070-7. [DOI] [PubMed] [Google Scholar]
- Payne S. M., Niesel D. W., Peixotto S. S., Lawlor K. M. Expression of hydroxamate and phenolate siderophores by Shigella flexneri. J Bacteriol. 1983 Sep;155(3):949–955. doi: 10.1128/jb.155.3.949-955.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reeves R., Magnuson N. S. Mechanisms regulating transient expression of mammalian cytokine genes and cellular oncogenes. Prog Nucleic Acid Res Mol Biol. 1990;38:241–282. doi: 10.1016/s0079-6603(08)60713-8. [DOI] [PubMed] [Google Scholar]
- Rosenshine I., Donnenberg M. S., Kaper J. B., Finlay B. B. Signal transduction between enteropathogenic Escherichia coli (EPEC) and epithelial cells: EPEC induces tyrosine phosphorylation of host cell proteins to initiate cytoskeletal rearrangement and bacterial uptake. EMBO J. 1992 Oct;11(10):3551–3560. doi: 10.1002/j.1460-2075.1992.tb05438.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sansonetti P. J., Mounier J. Metabolic events mediating early killing of host cells infected by Shigella flexneri. Microb Pathog. 1987 Jul;3(1):53–61. doi: 10.1016/0882-4010(87)90037-4. [DOI] [PubMed] [Google Scholar]
- Schreiber E., Matthias P., Müller M. M., Schaffner W. Rapid detection of octamer binding proteins with 'mini-extracts', prepared from a small number of cells. Nucleic Acids Res. 1989 Aug 11;17(15):6419–6419. doi: 10.1093/nar/17.15.6419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vlach J., Pitha P. M. Activation of human immunodeficiency virus type 1 provirus in T-cells and macrophages is associated with induction of inducer-specific NF-kappa B binding proteins. Virology. 1992 Mar;187(1):63–72. doi: 10.1016/0042-6822(92)90295-z. [DOI] [PubMed] [Google Scholar]
- de Vries G. E., Raymond C. K., Ludwig R. A. Extension of bacteriophage lambda host range: selection, cloning, and characterization of a constitutive lambda receptor gene. Proc Natl Acad Sci U S A. 1984 Oct;81(19):6080–6084. doi: 10.1073/pnas.81.19.6080. [DOI] [PMC free article] [PubMed] [Google Scholar]