Abstract
Nano- and microparticulate carriers can exert a beneficial impact on the pharmacodynamics of anticancer agents. To investigate the relationships between carrier and antitumor pharmacodynamics, paclitaxel incorporated in liposomes (L-pac) was compared with the clinical standard formulated in Cremophor-EL/ethanol (Cre-pac) in a rat model of advanced primary brain cancer. Three maximum-tolerated-dose regimens given by intravenous administration were investigated: 50 mg/kg on day 8 (d8) after implantation of 9L gliosarcoma tumors; 40 mg/kg on d8 and d15; 20 mg/kg on d8, d11, and d15. Body weight change and neutropenia were assessed as pharmacodynamic markers of toxicity. The pharmacodynamic markers of antitumor efficacy were increase in lifespan (ILS) and tumor volume progression, measured noninvasively by magnetic resonance imaging. At equivalent doses, neutropenia was similar for both formulations, but weight loss was more severe for Cre-pac. No regimen of Cre-pac extended survival, whereas L-pac at 40 mg/kg ×2 doses was well tolerated and mediated 26% ILS (p < 0.0002) compared with controls. L-pac at a lower cumulative dose (20 mg/kg ×3) was even more effective (40% ILS; p < 0.0001). In striking contrast, the identical regimen of Cre-pac was lethal. Development of a novel semimechanistic pharmacodynamic model permitted quantitative hypothesis testing with the tumor volume progression data, and suggested the existence of a transient treatment effect that was consistent with sensitization or “priming” of tumors by more frequent L-pac dosing schedules. Therefore, improved antitumor responses of carrier-based paclitaxel formulations can arise both from dose escalation, because of reduced toxicity, and from novel carrier-mediated alterations of antitumor pharmacodynamic effects.
The treatment of malignant brain tumors presents continuing challenges. Conventional therapy by surgical debulking and followed by radiation and chemotherapy generally is not curative. Chemotherapy fails for pathophysiological, pharmacological, and pharmaceutical reasons. Poor perfusion, tortuous and poorly permeable vasculature, and drug resistance are tumor properties that hinder drug penetration, deposition, and retention (Trédan et al., 2007). Suboptimal drug properties, including poor intrinsic membrane and tissue permeability, short circulating half-life, and rapid metabolism also impede therapy.
Incorporation of drugs in particulate carriers such as emulsions, nanoparticles, or liposomes may provide the means to overcome several of these factors. By limiting the drug volume of distribution, the carrier can reduce toxicity to normal tissues, permitting higher doses and thereby overcoming functional resistance (Drummond et al., 1999). In addition, carriers may extravasate through the flawed tumor microvasculature, increasing drug deposition and antitumor effects (Sharma et al., 1997; Zhou et al., 2002; Arnold et al., 2005).
Paclitaxel is a clinically important cytostatic/cytotoxic agent that also exerts potent antiangiogenic effects (Belotti et al., 1996; Wang et al., 2003). Inhibition of proliferation and migration of vascular endothelial cells, as well as alterations in microtubule dynamics, have been observed at paclitaxel concentrations 10- to 100-fold lower than those inducing mitotic arrest in nonendothelial cells (Belotti et al., 1996; Pasquier et al., 2005). Additional pharmacological effects have been observed with ultralow doses or protracted exposure (Bocci et al., 2002; Wang et al., 2003; Ling et al., 2004). However, the activity of paclitaxel against brain tumors has been disappointing (Postma et al., 2000; Chang et al., 2001); they are frequently drug-resistant, and paclitaxel is transported poorly across the blood-brain barrier.
Paclitaxel aqueous solubility is poor, and the clinical product Taxol consists of Cremophor-EL and ethanol (Cre-pac), which transforms spontaneously into a microemulsion (ten Tije et al., 2003) when prepared for administration. This vehicle is associated with severe, life-threatening hypersensitivity reactions (Weiss et al., 1990) and may interfere with taxane pharmacokinetics (PK) and antitumor activity (ten Tije et al., 2003).
Alternative formulations have been developed that permit rapid administration and possess altered pharmacodynamic properties compared with Cre-pac. ABI-007 (Abraxane) is an approved nanoparticulate formulation of paclitaxel complexed noncovalently with aggregated human albumin (A-pac). In animal and human trials, A-pac shows reduced toxicity compared with Cre-pac (Ibrahim et al., 2002; Desai et al., 2006), and phase III clinical data suggest that dose escalation is well tolerated and increases therapeutic responses (Gradishar et al., 2005). It is noteworthy that A-pac produces considerably higher levels of free drug in blood than does Cre-pac, and this observation has not yet been rationalized in terms of the reduced toxicity observed (Gardner et al., 2008).
Microparticulate liposome-based paclitaxel formulations (L-pac) also have been developed (Riondel et al., 1992; Sharma et al., 1993). They show activity in highly paclitaxel-resistant tumor models (Sharma et al., 1993), and are less toxic than Cre-pac in clinical trials (Fetterly et al., 2008). Despite their very different physicochemical characteristics, the particulate A-pac and L-pac formulations permit clinical dose escalation.
A third particulate paclitaxel formulation, consisting of a tocopherol nanoemulsion (T-pac), also has progressed from promising animal studies to clinical trial (Constantinides et al., 2006; Bulitta et al., 2009a; Bulitta et al., 2009b). However, T-pac failed to meet its clinical objective of noninferiority of response rates compared with Cre-pac in a phase III pivotal trial in women with metastatic breast cancer (Bulitta et al., 2009b). A detailed pharmacodynamic study of neutropenia showed deeper nadir neutrophil counts with T-pac compared with Cre-pac, and the investigators concluded that relative exposure to unbound paclitaxel at the site of toxicity was twice as large for T-pac compared with Cre-pac (Bulitta et al., 2009b).
These disparate effects of formulation on paclitaxel efficacy suggest the need for greater understanding of carrier-mediated effects on taxane pharmacodynamics (PD). Here, we investigated L-pac and Cre-pac pharmacodynamics in a multidrug-resistant orthotopic brain tumor model. Body weight and neutropenia were used as toxicity markers. Increase in lifespan (ILS) and tumor volume progression, quantified by magnetic resonance (MR) imaging, were used as endpoints for therapeutic efficacy. Robust, semimechanistic, quantitative systems pharmacological models (Lobo and Balthasar, 2002; Simeoni et al., 2004; Yang et al., 2009) were developed to evaluate mechanistic hypotheses, based on the data and the literature, that might provide insight into the differential therapeutic responses observed.
Materials and Methods
Crystalline paclitaxel was obtained from the National Cancer Institute (Bethesda, MD), Cremophor-EL (polyethoxylated castor oil) was from BASF (Parsippany, NJ), l-α-phosphatidylglycerol was from Avanti Polar Lipids (Birmingham, AL) and l-α-phosphatidylcholine was from Genzyme (Cambridge, MA). Male Fisher 344 rats (160–200 g) were from Harlan (Indianapolis, IN). The 9L rat gliosarcoma cell line (9L-72) was obtained from Dr. D. Deen (University of California, San Francisco) and maintained in RPMI 1640 medium containing 10% fetal bovine serum.
Stereotaxic Tumor Implantation.
Intracranial tumors were established in the caudate/putamen as described previously (Zhou et al., 2002; Arnold et al., 2005). A suspension of 4 × 104 9L tumor cells in 4 μl was injected stereotaxically 4.5 mm below the exposed dura at 1.5 mm anterior and 2.4 mm lateral to Bregma.
Paclitaxel Formulations.
To administer Cre-pac at the necessary doses, a stock solution was prepared at a higher concentration than in paclitaxel. Crystalline drug was dissolved in ethanol at 30 mg/ml and diluted with an equal volume of Cremophor EL. Immediately before administration, it was diluted 5-fold with sterile saline to a final concentration of 3 mg/ml.
Paclitaxel was formulated in l-α-phosphatidylcholine/l-α-phosphatidylglycerol (9:1 mol/mol) liposomes at a drug/lipid ratio of 3 mol% as described previously (Sharma and Straubinger, 1994). In brief, the drug/lipid mixture in chloroform was dried, resuspended in t-butanol at a phospholipid concentration of 150 mM, shell-frozen in liquid nitrogen, and lyophilized for 24 h. The resulting powder was hydrated at room temperature with saline and extruded sequentially through dual stacked polycarbonate membranes of 2.0, 0.4, 0.1, and 0.08 μm, with three passes through each pore size. The particle size resulting from this extrusion procedure would be approximately 110 nm ± 25% (Mayer et al., 1986). Association of drug with liposomes was greater than 90% (Sharma and Straubinger, 1994). The final paclitaxel concentration was 3 to 4 mg/ml, and the lipid concentration was 100 to 130 mM. The physical stability of L-pac and absence of precipitated material was verified by microscopy (Sharma and Straubinger, 1994).
Dosing Regimens.
Treatment was initiated by intravenous administration on day 8 (d8), when the tumor was well established and vascularized. A pilot experiment provided the basis for the selection of three regimens: 1) 50 mg/kg administered on d8; 2) 40 mg/kg on d8 and d15 (cumulative dose, 80 mg/kg); and 3) 20 mg/kg on d8, d11, and d15 (cumulative dose, 60 mg/kg). The amount of lipid administered at these doses was 195, 156, and 75 μmol/kg per injection, respectively, and it would not be expected to contribute to toxicity in the animals.
Toxicity Evaluation.
Animals were weighed every other day. Body weight loss of >20% below the pretreatment value represents lethal toxicity, and the protocol required euthanasia. For neutrophil counts, 0.3 ml of blood was drawn from the tail vein. The red cells were lysed by 10-fold dilution in 0.01% (w/v) crystal violet/2% (v/v) acetic acid. The total leukocyte count was determined by hemocytometer. Blood smears were prepared in parallel and stained with Giemsa; differential leukocyte counts revealed the relative percentage of neutrophils (%neutrophils). The absolute neutrophil count (ANC) was calculated by:
The change in ANC during treatment was calculated as ANC = (ANC1 − ANC0) × 100/ANC0, where ANC1 is the ANC during treatment, and ANC0 is the pretreatment value.
Lifespan.
Animals were monitored for signs of drug toxicity, such as diarrhea (gastrointestinal tract damage), increased intracranial pressure due to tumor growth (seizures, paralysis in contralateral limbs), or pigmentation around the eyes, mouth, and nose. Death ensues within 24 h of these signs; therefore, moribund animals were euthanized. The death was recorded as occurring on the following day. Statistical significance of changes in the median lifespan of treatment groups was evaluated using the nonparametric Mann-Whitney test. A two-tailed Student's t test was used to compare tumor volumes of treatment and control groups.
Tumor Volume Estimation.
Rats were anesthetized with ketamine/xylazine (66.7/6.7 mg/kg i.m.), and their heads were immobilized in nonferrous stereotaxic frames. Transverse relaxation time (T2)-weighted proton spin echo images (repetition time/echo time = 2000/120 ms) were acquired on a 1.5 T whole body scanner (Signa Endoplus; General Electric, Milwaukee, WI) interfaced to a custom transceiver coil tuned at 63.87 MHz. The voxel size was 0.31 × 0.31 × 1 mm3. Tumor volume was calculated from image data as described previously (Zhou et al., 2002).
Pharmacodynamic Theory and Analysis.
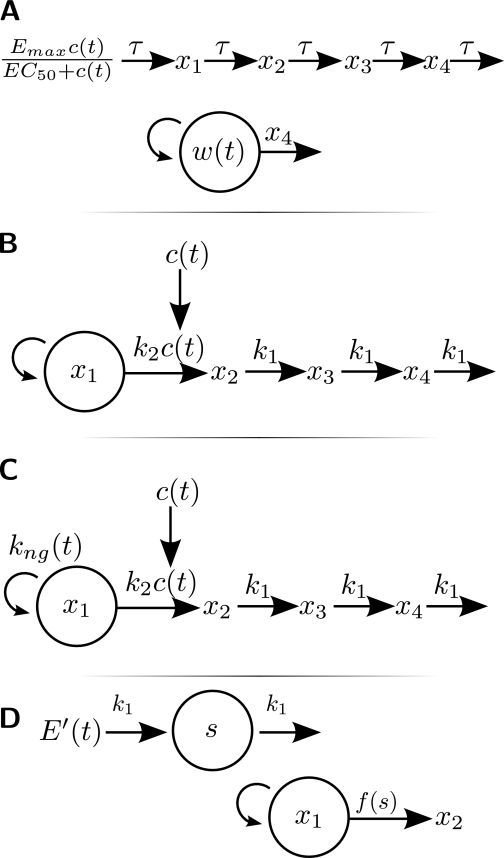
Two semimechanistic systems pharmacology models of drug-mediated tumor cell growth inhibition (Lobo and Balthasar, 2002; Simeoni et al., 2004) were used to investigate formulation- and schedule-dependent differences in paclitaxel efficacy. A key feature common to these models is the mathematical accommodation of the protracted delay between treatment and observable effects on tumor volume. Model A (Fig. 1A) hypothesizes a signal transduction cascade interposed between drug/receptor interaction and tumor growth delay (Lobo and Balthasar, 2002), whereas model B (Fig. 1B) hypothesizes immediate drug effects on the tumor that elicit a temporally protracted process of cell death (Simeoni et al., 2004). Despite the apparent similarity of these transit compartment models, they are noninterchangeable in terms of their behavior with different types of cancer drug response data (Yang et al., 2009). Additional refinements of model B were explored; model C (Fig. 1C) incorporates the concept that treatment induces an additional, temporally transient tumor component that is neither dividing nor committed to cell death. This fraction could correspond to drug treatment effects such as edema or creation of a population of quiescent cells. A fourth preliminary model (S1; not shown) explored the concept that treatment transiently increases tumor susceptibility to drug, either through increased vascular permeability or increased intratumor drug diffusion (Jang et al., 2001; Lu et al., 2007). This concept alone did not improve capture of the experimental data beyond that achieved with the other hypothesized mechanisms. Model D (Fig. 1D) represents a hybrid of models A and B, and includes the concept of transiently enhanced drug sensitivity from model S1.
Fig. 1.
System pharmacological models of tumor volume progression. Schematic illustrations of pharmacodynamic models that were applied or developed to analyze formulation- and regimen-dependent differences in the therapeutic effects of paclitaxel on intracranial 9L brain tumors. All are based on the concept of signal or cell transduction initiated by exposure to drug. Detailed equations for the final model (D) are provided in Materials and Methods. Model A (Lobo and Balthasar, 2002): drug activates a signal transduction cascade that ultimately exerts lethality in the tumor compartment. The magnitude of drug effect is defined by a Hill-type relationship; Emax is the maximal drug effect, EC50 is the concentration inducing half-maximal response, and c(t) is the plasma concentration of drug at time t. The drug signal propagates through a series of compartments (x1–x4) with a mean time of signal propagation (τ). Tumor volume is w(t); the curved arrow represents a tumor growth function, and x4 denotes that the elimination rate of cells from the tumor is determined by the drug signal from compartment x4. Model B (Simeoni et al., 2004): drug induces a dose-proportional fraction of cells to progress through stages of commitment to cell death. Compartment x1 represents the tumor mass, which increases according to a growth function. The concentration-time profile of drug in plasma c(t) induces a fraction of tumor cells to commit to cell death according to second-order killing constant k2, which was defined in the original publication. Terminally committed cells progress through compartments x2 to x4 with a rate constant of k1. The observed tumor volume is the sum of cells in compartments x1 to x4. Model C is identical to model B, with the exception that the tumor growth function is modified to assume that drug induced a fraction of tumor [kng(t)] that is transiently nondividing. Model D: hybrid based on models A and B. This model is described in detail, with equations, in Materials and Methods. As in model A, the drug initiates a signal [E′(t)] according to a Hill-type relationship. Drug signal s governs the rate of elimination of cells from the tumor mass x1. The first-order rate constant (k1) defines the rate of signal turnover. The model also incorporates the concept that drug treatment causes a transient period of heightened sensitivity to subsequent treatments; f(s) represents the effect of the drug signal on the elimination rate of tumor cells from the tumor mass. It includes the second-order kill constant k2 of model B, but is modulated by drug signal s (see Materials and Methods). The transient sensitization signal is modeled as an increase in the quantity of tumor-associated drug, but could also represent treatment-induced changes in tumor interstitial density or vascular permeability (Jang et al., 2001; Lu et al., 2007).
Optimal parameter estimates were calculated for each model. Quantifiable metrics such as sum-of-squared-error (SSE) and Akaike's information criterion (AIC) were used to weigh the impact of each conceptual component on the fidelity with which the model captured the experimental data. Based on these considerations, model D seemed to be superior (see Results) and was accepted as the final model. The model consists of three main components, a drug signal, a tumor progression model, and a relationship between drug effect and tumor volume. Several expedients were used to simplify implementation of key conceptual points so that the models did not become overparameterized with assumptions that were not experimentally justifiable.
Model D (Fig. 1D) is driven by the pharmacokinetic driving function A(t) (amount of paclitaxel in the central compartment at time t), which was derived from published data for Cre-pac and L-pac in rats (Fetterly and Straubinger, 2003). As shown in eq. 1, the initiating signal, driven by the drug amount in plasma, is nonlinear and characterized by the Hill function parameters Emax (maximal effect) and A50 (amount of drug producing 50% of Emax). This assumption is defensible pharmacologically, given the numerous cases in which the Hill function accurately describes drug concentration-response data based on receptor occupancy, and was also found to be necessary to account for the greater efficacy observed for lower doses administered more frequently (see Results).
 |
 |
In eq. 2, the signal initiated by exposure to drug is implemented as a signaling transit compartment (s) (Mager and Jusko, 2001) (Fig. 1D), and includes the concept explored in model S1 that treatment transiently increases tumor susceptibility to drug (Jang et al., 2001; Lu et al., 2007). This signal might reflect increased cellular drug uptake/accumulation, tumor decompression resulting from paclitaxel-induced apoptosis, or other mathematically equivalent, mechanistically plausible concepts. The rate constant (k1) defines the rate of signal turnover in this compartment, and is proportional to 1/(mean transit time). Diminution of the drug signal is proportional to 1/w(t) (inverse of tumor size); this assumption is justified by the observation that larger tumors may contain poorly functional vascular networks (Trédan et al., 2007).
The tumor volume w(t) is composed of cells in proliferating (x1) and quiescent states (x2) (eq. 3). These states are characterized by eq. 4 and eq. 5.
 |
 |
Nominal tumor growth in the proliferating state x1 [A(t) = 0] is characterized by eq. 4; initially the tumor volume increases according to the first-order growth rate constant λ0, but eventually transitions to a zero-order growth rate (λ1). The rate of this transition is controlled by ψ (Simeoni et al., 2004). The term f(s) represents the effect of the drug signal s on the tumor cell elimination rate (Fig. 1D). Certain assumptions were developed, tested, and incorporated into the model to account for the observed schedule-dependent tumor responses to drug. First, an initial dose renders the tumor transiently more sensitive to a subsequent dose. This could arise from increased tumor vascular permeability and enhanced drug accumulation, tumor decompression, and increased intratumor drug diffusion, or other mechanisms. Second, the rate of cells transitioning from the proliferating (x1) to the quiescent state (x2) is a saturable function of the drug-induced signal (s). The parameter k2 is a second-order cell-kill constant representing the maximum effect, and the s50 term represents the signal level producing half-maximal effect. This functionality also serves to increase the efficacy of lower dose/higher frequency schedules, which was observed experimentally.
The model structure could not account for formulation-dependent differences in efficacy. To do so, it was necessary to estimate Emax and s50 independently for the two formulations, which improved capture of the data by the model predictions. For this reason, they are designated E*max and s*50 in eq. 1 and eq. 6. The value of ψ, the transition term for tumor growth rate, was fixed to 20 per the original publication (Simeoni et al., 2004). The remaining parameters were regressed simultaneously in MATLAB (version 7; The Mathworks, Natick, MA). For parameter estimation purposes, a weighted minimization of the residual sum of the squared error was used.
Results
The maximum tolerated dose (MTD) of both paclitaxel formulations was investigated to establish doses and treatment schedules for therapeutic experiments. A single dose of 85 mg/kg i.v. paclitaxel was reported to be lethal in 20 to 40% of Sprague-Dawley rats (Kadota et al., 1994). Incorporation of paclitaxel in liposomes increases the MTD of paclitaxel (Sharma et al., 1993), and so the above-reported dose was tested in rats bearing 14-day intracranial 9L tumors, as were doses that were approximately 20% higher and lower. Severe gastrointestinal tract effects or weight loss were observed for both formulations at ≥85 mg/kg (not shown). Onset of these symptoms was delayed approximately 24 h in animals treated with L-pac. From these studies, 50 mg/kg was chosen as the maximum single-administration dose. Two additional dosing regimens were selected: 80 mg/kg given as two injections of 40 mg/kg separated by 7 days, and 60 mg/kg, given as three injections of 20 mg/kg over 7 days (i.e., 3–4 days apart).
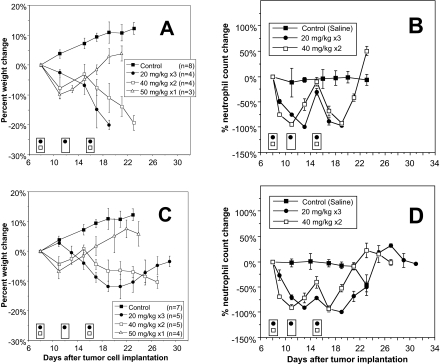
Therapeutic experiments were initiated on day 8 (d8) after tumor implantation. Body weight was monitored as a PD marker of overall toxicity (Fig. 2) and to verify that each dosing regimen approximated the MTD for that treatment protocol. Tumor-bearing control rats experienced a 10% weight gain. Body weight peaked approximately 19d after tumor implantation, at which time the animals became symptomatic from tumor growth and succumbed quickly. In groups treated with Cre-pac (Fig. 2A), a single dose of 50 mg/kg on d8 resulted in approximately 10% weight loss, which recovered within a week. L-pac at 50 mg/kg also mediated weight loss (Fig. 2C), but with a shallower nadir. The amount of phospholipid administered was approximately 39 μmol, which could result in mild, reversible blockade of the reticuloendothelial system, but should not contribute to toxicity.
Fig. 2.
Formulation-dependent toxicocodynamic effects of paclitaxel. Tumor-bearing rats were treated with Cre-pac or L-pac beginning on day 8 after implantation. Points represent the mean ± S.D. of each group; n indicates the number of animals per group. Symbols on abscissa indicate times of treatment for each group. A, C, change in body weight for animals treated with Cre-pac (A) and L-pac (C). B, D, change in neutrophil counts for animals treated with Cre-pac (B) and L-pac (D).
Cre-pac, given as 40 mg/kg on d8 and d15 (80 mg/kg cumulative), resulted in 8% weight loss after the first injection that recovered in part. However, weight loss was precipitous after the second treatment and did not recover. L-pac given at the same dose and schedule resulted in a similar pattern of weight loss, but the loss was less severe, and the nadir after dosing was shallower. The phospholipid dose was approximately 21 μmol, and would not be expected to contribute to toxicity.
The lowest cumulative dose of Cre-pac (60 mg/kg given in 7 days as 20 mg/kg doses on d8, d11, and d15) was more toxic than the 80 mg/kg cumulative-dose schedule, also given in 7 days (40 mg/kg doses on d8 and d15). Body weight declined with each administration and fell precipitously after the third injection. In striking contrast, animals treated with the equivalent regimen of L-pac continued to gain weight after the first dose. Weight declined after the second and third doses, but increased after the regimen was completed. Each phospholipid dose was approximately 21 μmol, and would not be expected to contribute to toxicity.
Bone marrow suppression was also evaluated to compare formulation toxicity. For animals receiving Cre-pac as 40 mg/kg ×2, neutrophil counts returned to baseline in the 7 days between treatments (Fig. 2B). Neutropenia was more sustained with L-pac at this dose level, and counts did not return to baseline before the second dose was given (Fig. 2D).
Progressive neutropenia was observed in animals receiving either formulation in the 20 mg/kg ×3 dose regimen. After the second dose, neutrophil counts fell to 10% of the pretreatment value, a level regarded clinically as severe neutropenia. By d23 after tumor implantation (8 days after the third dose), all animals treated with Cre-pac succumbed. It is noteworthy that d23 represents the median survival time for untreated tumor-bearing controls (below). Therefore, ineffective control of tumor progression could not be discounted as a cause of death. In striking contrast, animals treated with 20 mg/kg ×3 L-pac recovered to baseline neutrophil counts 9 days after the third dose (Fig. 2D).
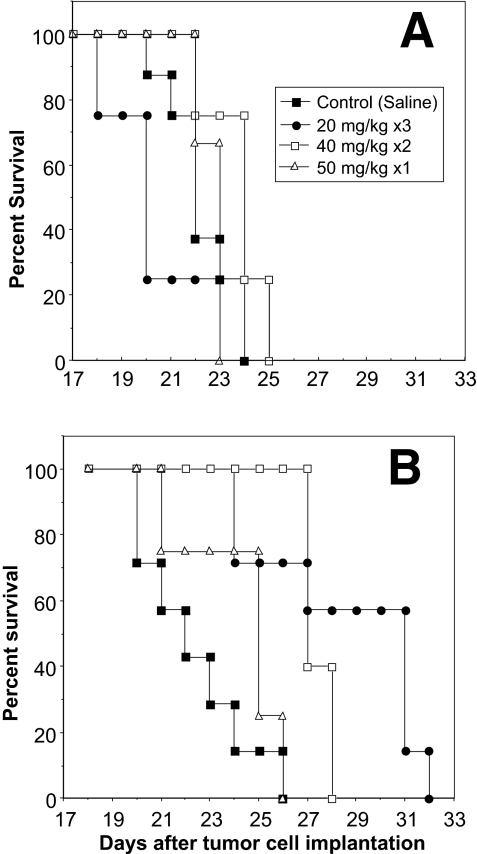
Increase in median lifespan was examined as a measure of overall therapeutic efficacy (Fig. 3) and for statistical comparison of the two formulations (Table 1). L-pac given as 20 mg/kg ×3 conferred the greatest ILS (40%, p < 0.0001). A higher cumulative L-pac dose (40 mg/kg ×2) mediated a significant but lesser ILS (27%, p < 0.005). A single dose of 50 mg/kg L-pac did not alter lifespan significantly (Table 1).
Fig. 3.
Effects of formulation and treatment regimen on lifespan. Survival of animals bearing intracranial 9L tumors treated with Cre-pac (A) or L-pac (B). Table 2 shows the group sizes and statistical comparisons.
Table 1.
Survival of rats bearing advanced intracranial 9L tumors
| Treatment Groups | Rats per Group | MLSa (Range) | %ILSb | pc | Days to Volume Thresholdd |
|---|---|---|---|---|---|
| Control (saline) | 15 | 22 (20–24) | N/A | 21 | |
| Cre-PAC 50 mg/kg ×1 | 3 | 23 (22–23) | 5 | 0.65 | 21 |
| Cre-PAC 40 mg/kg ×2 | 4 | 24 (22–25) | 9 | 0.12 | 22.7 |
| Cre-PAC 20 mg/kg ×3 | 4 | 20 (18–24) | −9 | 0.15 | 26 |
| L-PAC 50 mg/kg ×1 | 4 | 25 (21–26) | 14 | 0.10 | 21 |
| L-PAC 40 mg/kg ×2 | 9e | 27.8 (27–28) | 26 | <0.0002 | 26 |
| L-PAC 20 mg/kg ×3 | 11e | 30.8 (24–32) | 40 | <0.0001 | 31.4 |
N/A, not applicable.
Median lifespan.
Percentage increase in lifespan: (mlstreated − MLScontrol)/MLScontrol.
Two-tailed p value from nonparametric Mann-Whitney test compared with control group.
Extrapolation of tumor growth rate to lethal volume of 400 mm3.
Average of two experiments. Median lifespan did not differ significantly in the two experiments.
In contrast, no dose or schedule of Cre-pac conferred a survival advantage relative to vehicle-treated controls. L-pac given as 20 mg/kg ×3 yielded the greatest ILS; Cre-pac given as 20 mg/kg ×3 was the most toxic dosing regimen. Lifespan was reduced approximately 10%, but this trend was not statistically significant (p = 0.15).
Further empirical optimization of L-pac dose and treatment schedule plausibly could improve survival. However, both drug toxicity and tumor growth can result in death, thus confounding the use of lifespan as a quantitative pharmacodynamic endpoint for optimizing therapeutic outcome. Therefore, MR imaging of tumor volume progression was used to provide an independent, continuous, quantitative measure of pharmacodynamic effects during therapy.
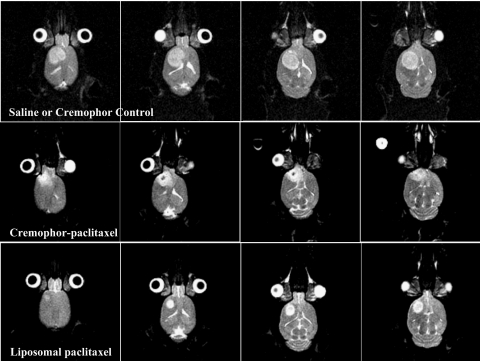
Figure 4 shows representative (T2)-weighted brain images of animals treated with saline (control), Cre-pac, and L-pac. Tumors appeared as hyperintense, well defined masses in the left-frontal quadrant. Tumor volumes were calculated from three-dimensional data sets for each animal, and volume progression was monitored repetitively during treatment (Fig. 5, A and B).
Fig. 4.
Representative serial MR images for treatment groups. Rows shows consecutive 1-mm-thick (T2)-weighted MR image slices from individual rats treated with saline (top row), or 20 mg/kg ×3 doses of Cre-pac (middle row) or L-pac (bottom row). Tumor appears as a hyperintense mass in the left frontal quadrant.
Fig. 5.
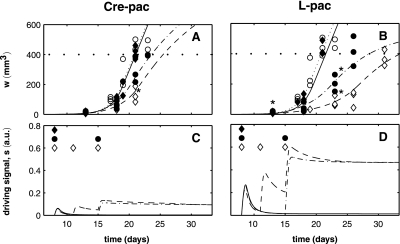
Tumor volume progression in treatment groups by repetitive MR images. Tumor volumes were measured during treatment of animals with Cre-pac (A) and L-pac (B). Symbols represent the measured tumor volume for each individual animal (n = 3/group), and lines represent the best fit of pharmacodynamic model D (Fig. 1D) to the data for each group. ○, dotted line, control group; ♦, solid line, 50 mg/kg ×1 dose; ●, dash-dot line, 40 mg/kg ×2 dose; ◊, dashed line, 20 mg/kg ×3 dose. Asterisks indicate time points at which treated versus control differ significantly at p < 0.002 or lower. C, D, magnitude of cytotoxicity “driving function” for Cre-pac (C) and L-pac (D) treatments, based on model D. Symbols on abscissa are the same as for A and B, and show the treatment times for each group. a.u., arbitrary units.
The rate of tumor expansion was reduced significantly for animals treated with L-pac (Fig. 5B). As early as d13, tumors in animals treated with 20 mg/kg ×3 L-pac were significantly smaller than controls (p < 0.002), and on d23, the last day on which volumes could be compared with controls, animals treated with 20 mg/kg ×3 and 40 mg/kg ×2 L-pac had smaller tumors (p < 0.0001 and p < 0.002, respectively). Consistent with the ILS data, the greatest reduction in tumor volume progression was observed in animals treated with L-pac at a lower cumulative dose but higher frequency (20 mg/kg ×3). The highest L-pac dose (50 mg/kg ×1) was ineffective.
For animals treated with Cre-pac, significant tumor growth retardation was observed only in animals treated with 20 mg/kg ×3 (Fig. 5A; p < 0.002 on d21). However, 100% of those animals succumbed by d23, which coincided with the median day of death for control animals. The 40 mg/kg ×2 Cre-pac regimen was ineffective.
Two mechanistically distinct types of pharmacological system models were used to analyze the observed formulation- and regimen-dependent therapeutic effects quantitatively. Neither base model A nor B (Lobo and Balthasar, 2002; Simeoni et al., 2004) fit the data well, based on objective criteria (Table 2) and visual inspection. Model D (Fig. 1) is a hybrid derived from the two base models; it hypothesizes that a signal transduction process conveys a “kill signal” that is related in magnitude to the drug pharmacokinetic profile, and that the effect of this signal is to shunt a proportion of tumor cells to a commitment to cell death. It also incorporates the concept of schedule-dependent tumor priming (Lu et al., 2007) that was tested in model S1 (see Materials and Methods).
Table 2.
Performance criteria for evaluated pharmacodynamic models
| Structurea | SSE | AIC |
|---|---|---|
| A | 2.58 × 106 | 1.25 × 103 |
| B | 1.19 × 106 | 1.18 × 103 |
| C | 1.91 × 107 | 1.44 × 103 |
| D | 3.29 × 105 | 1.06 × 103 |
Structure corresponds to models shown in Fig.1.
Objective criteria such as SSE and AIC values supported the selection of model D as the final system model (Table 2). When fit simultaneously to all data, it best captured the time course of tumor volume progression for control and treated animals (Fig. 5, A and B). Table 3 shows the estimated model parameters.
Table 3.
Parameter estimates for pharmacodynamic analysis
| Parameter (units)a | Estimate (CV%) |
|---|---|
| w0 (mm3) | 5.96 × 10−3 (1.95) |
| λ0 (h−1) | 2.30 × 10−2 (0.63) |
| λ1 (mm3 h−1) | 4.23 (7.59) |
| k1 (mm3 h−1) | 4.59 × 10−2 (0.90) |
| k2 (h−1) | 1.60 × 10−2 (1.04) |
| A50 (mg/kg) | 8.42 × 10−2 (0.43) |
| Emax,L (a.u. · mm−3) | 0.794 (1.97) |
| Emax,C (a.u. ·mm−3) | 0.227 (1.55) |
| s50,L (a.u.) | 0.18 (16.9) |
| s50,C (a.u.) | 9.45 × 10−2 (0.82) |
CV, coefficient of variation; a.u., arbitrary units.
Subscripts L and C (Emax, s50) refer to liposome- and Cremophor-based formulations, respectively.
Model D represents the tumor priming effect as a driving state (s) (shown in Fig. 5, C and D). Treatment transiently increased the magnitude of the driving state, and repetitive treatments of the appropriate frequency were observed to cause s to accumulate or increase in magnitude. This model feature was required to accommodate the greater observed efficacy of the higher frequency/lower dose schedules, which was confirmed by statistical analysis (Table 1) and is supported by empirical data from the literature (Jang et al., 2001; Lu et al., 2007).
Although the structure of model D could account for schedule-dependent differences in efficacy, it could not account for the formulation-dependent differences observed with identical doses/schedules. Formulation-specific effects were explored within the models, and capture of the data was improved by fitting specific parameters independently. It was found that the model predictions were best able to capture the higher efficacy of L-pac by fitting the Emax (the initiating signal capacity) independently for the two formulations. Figure 5, C and D, show the mechanistic predictions of the model: for the same dose level and schedule of administration, the susceptibility factor (driving force s) accumulates to higher levels for L-pac. This is a direct result of the fact that the estimated value of the Emax term is 3.5-fold higher for L-pac than for Cre-pac (0.794 versus 0.227), and this higher driving force serves to magnify L-pac PD effects. Interpreted literally, increased L-pac efficacy cannot be explained simply by the elevation of dose that is feasible because of reduced toxicity; rather, a higher maximal efficacy of L-pac must be invoked as well.
Time-to-progression also was examined. A volume of 400 ± 20 mm3 represented a threshold of morbidity, in that death generally ensued within 48 h. As shown in Table 1, growth delay seemed to be consistent with the observed change in median lifespan for all treatment groups except for the group treated with 20 mg/kg ×3 Cre-pac; MR imaging showed tumor growth delay for that regimen, but lifespan was not extended.
Discussion
The persistently poor prognosis of brain tumors drives the search for new therapeutic approaches. A growing realization from clinical trials is that “repackaging” clinically effective drugs in more efficacious carrier-based formulations (Sparreboom et al., 2005a) can provide important clinical advantages and, from an economic perspective, address what many regard as an unsustainable attrition rate for new oncology drugs.
Four particulate formulations of paclitaxel that have progressed to clinical trial or approval display important pharmacodynamic differences. The first-in-class Cremophor-based clinical standard (Cre-pac) circulates initially as a microemulsion in blood (ten Tije et al., 2003) from which drug partitions into the plasma. The Cremophor vehicle thus constitutes a circulating pharmacokinetic “compartment,” and the role of the vehicle as an additional PK compartment seems to be common to all four formulations. The albumin nanoparticulate formulation A-pac (Ibrahim et al., 2002; Desai et al., 2006) has an increased volume of distribution and peripheral penetration compared with Cre-pac (Sparreboom et al., 2005b; Desai et al., 2006). The enhanced biodistribution of A-pac may arise from transport of the carrier via the gp60 endothelial cell receptor for albumin and interactions with the albumin-binding secreted protein acid rich in cysteine (SPARC) (Nyman et al., 2005; Desai et al., 2006). The drug release rate from the albumin nanoparticle is unknown. The L-pac formulation has a larger central volume of distribution compared with Cre-pac, but much slower distributional clearance (Fetterly et al., 2001). Based on the data, L-pac is more effective than Cre-pac at confining the drug to the circulating reservoir, which restricts the exposure of critical normal tissues. The rate of drug release from L-pac also is unknown, but based on PK analysis, seems to be fairly rapid (Fetterly and Straubinger, 2003). Finally, the tocopherol-based T-pac nanoemulsion has a volume of distribution intermediate between L-pac and Cre-pac, and the release rate of drug from the circulating carrier to the plasma was estimated to be lower than from Cre-pac (Bulitta et al., 2009a).
The paradox presented by these formulations is that two are very different in physicochemical properties, but are similar pharmacodynamically: A-pac and L-pac both possess markedly lower toxicity in animal (Riondel et al., 1992; Sharma et al., 1993; Sharma and Straubinger, 1994; Desai et al., 2006) and human studies (Gradishar et al., 2005; Fetterly et al., 2008). In contrast, the T-pac formulation, which is compositionally more similar to L-pac, seems to be more toxic than Cre-pac clinically (Bulitta et al., 2009b). Thus, whereas the observed improvement in paclitaxel efficacy by incorporation into particulate carriers represents an important clinical advance, the physicochemical basis of this beneficial effect is unclear.
In the present study, Cre-pac and L-pac were compared in terms of toxicity and antitumor efficacy in a drug-resistant model of intracranial brain cancer. Based on the balance of neutropenia and body weight loss, each of the treatment regimens evaluated efficacy at the MTD. No dose or treatment schedule of Cre-pac conferred a survival advantage relative to vehicle-treated controls, and in some cases, drug toxicity seemed to hasten death. In contrast, L-pac was less toxic and more efficacious: animals responded to therapy and lived up to 40% longer than controls. This extension in lifespan exceeded the results obtained previously in this advanced intracranial 9L tumor model for a long-circulating, doxorubicin-containing formulation dosed weekly at its MTD (Sharma et al., 1997). The resolution of observed toxicities at the end of treatment suggests that further extension of lifespan may be possible with the continuation of treatment.
L-pac administered at a higher cumulative dose (80 mg/kg), but with longer between-dose intervals (7 days) mediated a significant increase in median lifespan (29%; p < 0.0002). However, this regimen was inferior to the lower cumulative dose (60 mg/kg) given as smaller, more frequent doses.
Neutropenia was similar for Cre-pac and L-pac, but seemed to be more sustained with L-pac at the lower dose/higher frequency. Nonetheless, neutrophil counts returned to baseline values after cessation of treatment with L-pac, and the animals survived. In contrast, animals treated with Cre-pac did not survive. Recent clinical trials comparing a similar L-pac formulation with Cre-pac at more conservative dose intensities showed that neutropenia was milder for L-pac (Fetterly et al., 2008).
Body weight change clearly discriminated between formulations. Cre-pac caused a substantial reduction in body weight, and toxicity was severe with some dosing regimens. L-pac was better tolerated, and for some schedules, it would be feasible to administer higher dose intensities.
The superior tumor growth delay observed for L-pac may arise from a number of mechanisms. Liposomal incorporation retards drug distribution to the peripheral tissues, and may increase deposition in tumors. Although liposomes do not cross an intact blood-brain barrier, they can extravasate through defects in the tumor vasculature (Sharma et al., 1997; Arnold et al., 2005). For the L-pac formulation used here, our PK model estimated that drug release rates are rapid but delayed compared with Cre-pac. The PK of total paclitaxel in blood is similar for L-pac and Cre-pac (Fetterly and Straubinger, 2003). Understanding the role of the drug release rate in efficacy versus toxicity is not only elusive but perhaps key in understanding the differential toxicity and efficacy of these alternative formulations (Gardner et al., 2008). Because of the relatively rapid clearance of the type of liposomes used here, tumor drug deposition may be lower than achieved with long-circulating liposome formulations in this model (Arnold et al., 2005). Nonetheless, some enhancement of drug deposition in hyperpermeable regions of the tumor might occur with L-pac, in particular, given the previous observations that paclitaxel at appropriately spaced intervals can increase deposition of drug or drug-loaded nanoparticles (Jang et al., 2001; Lu et al., 2007). Observations of paclitaxel pharmacological activity at ultralow concentrations and protracted exposure times (Bocci et al., 2002; Wang et al., 2003; Ling et al., 2004) suggest that persistent low concentrations of paclitaxel in the blood, or a small intratumor depot of paclitaxel, could exert sustained pharmacodynamic effects.
Repetitive, noninvasive MR imaging enabled the disentanglement of ineffective tumor growth control versus life-shortening toxicity, and enabled the application of semimechanistic, quantitative pharmacodynamic analysis to explore hypotheses underlying the pharmacological differences between the formulations. Five model structures, derived from two distinct types of transit compartment models (Sun and Jusko, 1998; Mager and Jusko, 2001), were evaluated quantitatively for their ability to capture formulation- and treatment-dependent effects on tumor volume progression. One model type (Fig. 1A) represents the effect of drug as initiating a cellular signal transduction process that ultimately impinges on a “cell kill” mechanism (Lobo and Balthasar, 2002), whereas the second (Fig. 1B) represents the effect of drug as shunting a proportion of dividing cells of the tumor mass into a commitment to cell death (Simeoni et al., 2004). Both mechanisms have been observed for oncology drugs.
Although neither base model fit the data well, they represented the starting point for development of a novel hybrid model (model D, Fig. 1) that provided excellent fits to the data. Conceptually, a signal transduction process is initiated in this model by the administration of drug, and cell death is noninstantaneous. Both features resemble reported taxane mechanisms of action (Blagosklonny and Fojo, 1999; Ling et al., 2004). The addition of a hypothesized mechanism of transient tumor priming effect of closely spaced doses (Jang et al., 2001; Lu et al., 2007) provided the best simultaneous fitting of all data. It still has not been determined whether this priming effect arises from effects on tumor vasculature, tumor cells, or both. Nonetheless, it was observed that each of these mechanistic assumptions was necessary to explain the observation that a lower cumulative dose, given in smaller, more frequent administrations, was more effective than larger doses administered less frequently. It is noteworthy that the tumor priming effect was only observed with L-pac in this advanced, drug-resistant tumor model because therapeutically effective doses could not be achieved with Cre-pac.
The structure of model D could not account for the greater activity of L-pac over Cre-pac when both were given at identical doses and schedules. Fitting of the Emax model parameter independently for the two formulations produced the best objective capture of the data. At face value, this finding suggests that, although both formulations are similar in potency, the liposomal formulation possesses a greater maximal effect and a more persistent tumor priming signal. Regardless of the literal interpretation of these model components, the requirement to estimate them in a formulation-dependent manner suggests that reduction in dose-limiting toxicity may not be the only mechanism underlying the superiority of L-pac over Cre-pac in therapeutic efficacy.
In summary, paclitaxel incorporation in microparticulate carriers may alter pharmacodynamics by several mechanisms. Elevation of the MTD permits higher doses and increased therapeutic responses. In addition, hypotheses developed by mechanistic pharmacological system analysis suggest novel, schedule-dependent effects that were not observable with the Cremophor-based clinical standard. These alterations in pharmacodynamic effects may be shared by other particulate taxane formulations, and suggest that further improvement of clinical outcomes are possible for this important class of drug.
This work was supported by the National Institutes of Health National Cancer Institute [Grants CA55251, CA107570] (to R.M.S.).
Article, publication date, and citation information can be found at http://jpet.aspetjournals.org.
doi:10.1124/jpet.109.160044
- Cre-pac
- paclitaxel formulated in Cremophor-EL/ethanol
- A-pac
- paclitaxel incorporated in albumin nanoparticles
- L-pac
- paclitaxel incorporated in liposome microparticles
- T-pac
- paclitaxel incorporated in tocopherol nanoemulsion
- d8
- 8th day after implantation of tumor
- d11
- 11th day after implantation of tumor
- d15
- 15th day after implantation of tumor
- AIC
- Akaike's information criterion
- ANC
- absolute neutrophil count
- ILS
- increase in lifespan
- MTD
- maximum tolerated dose
- MR
- magnetic resonance
- PD
- pharmacodynamics
- PK
- pharmacokinetics
- SSE
- sum-of-squared-error
- T2
- transverse relaxation time.
References
- Arnold RD, Mager DE, Slack JE, Straubinger RM. (2005) Effect of repetitive administration of doxorubicin-containing liposomes on plasma pharmacokinetics and drug biodistribution in a rat brain tumor model. Clin Cancer Res 11:8856–8865 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belotti D, Vergani V, Drudis T, Borsotti P, Pitelli MR, Viale G, Giavazzi R, Taraboletti G. (1996) The microtubule-affecting drug paclitaxel has antiangiogenic activity. Clin Cancer Res 2:1843–1849 [PubMed] [Google Scholar]
- Blagosklonny MV, Fojo T. (1999) Molecular effects of paclitaxel: myths and reality (a critical review). Int J Cancer 83:151–156 [DOI] [PubMed] [Google Scholar]
- Bocci G, Nicolaou KC, Kerbel RS. (2002) Protracted low-dose effects on human endothelial cell proliferation and survival in vitro reveal a selective antiangiogenic window for various chemotherapeutic drugs. Cancer Res 62:6938–6943 [PubMed] [Google Scholar]
- Bulitta JB, Zhao P, Arnold RD, Kessler DR, Daifuku R, Pratt J, Luciano G, Hanauske AR, Gelderblom H, Awada A, et al. (2009a) Mechanistic population pharmacokinetics of total and unbound paclitaxel for a new nanodroplet formulation versus Taxol in cancer patients. Cancer Chemother Pharmacol 63:1049–1063 [DOI] [PubMed] [Google Scholar]
- Bulitta JB, Zhao P, Arnold RD, Kessler DR, Daifuku R, Pratt J, Luciano G, Hanauske AR, Gelderblom H, Awada A, et al. (2009b) Multiple-pool cell lifespan models for neutropenia to assess the population pharmacodynamics of unbound paclitaxel from two formulations in cancer patients. Cancer Chemother Pharmacol 63:1035–1048 [DOI] [PubMed] [Google Scholar]
- Chang SM, Kuhn JG, Robins HI, Schold SC, Jr, Spence AM, Berger MS, Mehta M, Pollack IF, Rankin C, Prados MD. (2001) A Phase II study of paclitaxel in patients with recurrent malignant glioma using different doses depending upon the concomitant use of anticonvulsants: a North American Brain Tumor Consortium report. Cancer 91:417–422 [DOI] [PubMed] [Google Scholar]
- Constantinides PP, Han J, Davis SS. (2006) Advances in the use of tocols as drug delivery vehicles. Pharmacol Res 23:243–255 [DOI] [PubMed] [Google Scholar]
- Desai N, Trieu V, Yao Z, Louie L, Ci S, Yang A, Tao C, De T, Beals B, Dykes D, et al. (2006) Increased antitumor activity, intratumor paclitaxel concentrations, and endothelial cell transport of cremophor-free, albumin-bound paclitaxel, ABI-007, compared with cremophor-based paclitaxel. Clin Cancer Res 12:1317–1324 [DOI] [PubMed] [Google Scholar]
- Drummond DC, Meyer O, Hong K, Kirpotin DB, Papahadjopoulos D. (1999) Optimizing liposomes for delivery of chemotherapeutic agents to solid tumors. Pharmacol Rev 51:691–743 [PubMed] [Google Scholar]
- Fetterly GJ, Grasela TH, Sherman JW, Dul JL, Grahn A, Lecomte D, Fiedler-Kelly J, Damjanov N, Fishman M, Kane MP, et al. (2008) Pharmacokinetic/pharmacodynamic modeling and simulation of neutropenia during Phase I development of liposome entrapped paclitaxel. Clin Cancer Res 14:5856–5863 [DOI] [PubMed] [Google Scholar]
- Fetterly GJ, Straubinger RM. (2003) Pharmacokinetics of paclitaxel-containing liposomes in rats. AAPS PharmSci 5:E32 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fetterly GJ, Tamburlin JM, Straubinger RM. (2001) Paclitaxel pharmacodynamics: application of a mechanism-based neutropenia model. Biopharm Drug Dispos 22:251–261 [DOI] [PubMed] [Google Scholar]
- Gardner ER, Dahut WL, Scripture CD, Jones J, Aragon-Ching JB, Desai N, Hawkins MJ, Sparreboom A, Figg WD. (2008) Randomized crossover pharmacokinetic study of solvent-based paclitaxel and nab-paclitaxel. Clin Cancer Res 14:4200–4205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gradishar WJ, Tjulandin S, Davidson N, Shaw H, Desai N, Bhar P, Hawkins M, O'Shaughnessy J. (2005) Phase III trial of nanoparticle albumin-bound paclitaxel compared with polyethylated castor oil-based paclitaxel in women with breast cancer. J Clin Oncol 23:7794–7803 [DOI] [PubMed] [Google Scholar]
- Ibrahim NK, Desai N, Legha S, Soon-Shiong P, Theriault RL, Rivera E, Esmaeli B, Ring SE, Bedikian A, Hortobagyi GN, et al. (2002) Phase I and pharmacokinetic study of ABI-007, a Cremophor-free, protein-stabilized, nanoparticle formulation of paclitaxel. Clin Cancer Res 8:1038–1044 [PubMed] [Google Scholar]
- Jang SH, Wientjes MG, Au JL. (2001) Enhancement of paclitaxel delivery to solid tumors by apoptosis-inducing pretreatment: effect of treatment schedule. J Pharmacol Exp Ther 296:1035–1042 [PubMed] [Google Scholar]
- Kadota T, Chikazawa H, Kondoh H, Ishikawa K, Kawano S, Kuroyanagi K, Hattori N, Sakakura K, Koizumi S, Hiraiwa E, et al. (1994) Toxicity studies of paclitaxel (I): single dose intravenous toxicity in rats. J Toxicol Sci 19 (Suppl 1):1–9 [DOI] [PubMed] [Google Scholar]
- Ling X, Bernacki RJ, Brattain MG, Li F. (2004) Induction of survivin expression by taxol (paclitaxel) is an early event, which is independent of taxol-mediated G2/M arrest. J Biol Chem 279:15196–15203 [DOI] [PubMed] [Google Scholar]
- Lobo ED, Balthasar JP. (2002) Pharmacodynamic modeling of chemotherapeutic effects: application of a transit compartment model to characterize methotrexate effects in vitro. AAPS PharmSci 4:E42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu D, Wientjes MG, Lu Z, Au JL. (2007) Tumor priming enhances delivery and efficacy of nanomedicines. J Pharmacol Exp Ther 322:80–88 [DOI] [PubMed] [Google Scholar]
- Mager DE, Jusko WJ. (2001) Pharmacodynamic modeling of time-dependent transduction systems. Clin Pharmacol Ther 70:210–216 [DOI] [PubMed] [Google Scholar]
- Mayer LD, Hope MJ, Cullis PR. (1986) Vesicles of variable sizes produced by a rapid extrusion procedure. Biochim Biophys Acta 858:161–168 [DOI] [PubMed] [Google Scholar]
- Nyman DW, Campbell KJ, Hersh E, Long K, Richardson K, Trieu V, Desai N, Hawkins MJ, Von Hoff DD. (2005) Phase I and pharmacokinetics trial of ABI-007, a novel nanoparticle formulation of paclitaxel in patients with advanced nonhematologic malignancies. J Clin Oncol 23:7785–7793 [DOI] [PubMed] [Google Scholar]
- Pasquier E, Honore S, Pourroy B, Jordan MA, Lehmann M, Briand C, Braguer D. (2005) Antiangiogenic concentrations of paclitaxel induce an increase in microtubule dynamics in endothelial cells but not in cancer cells. Cancer Res 65:2433–2440 [DOI] [PubMed] [Google Scholar]
- Postma TJ, Heimans JJ, Luykx SA, van Groeningen CJ, Beenen LF, Hoekstra OS, Taphoorn MJ, Zonnenberg BA, Klein M, Vermorken JB. (2000) A phase II study of paclitaxel in chemonaive patients with recurrent high-grade glioma. Ann Oncol 11:409–413 [DOI] [PubMed] [Google Scholar]
- Riondel J, Jacrot M, Fessi H, Puisieux F, Potier (1992) Effects of free and liposome-encapsulated taxol on two brain tumors xenografted into nude mice. In Vivo 6:23–27 [PubMed] [Google Scholar]
- Sharma A, Mayhew E, Straubinger RM. (1993) Antitumor effect of taxol-containing liposomes in a taxol-resistant murine tumor model. Cancer Res 53:5877–5881 [PubMed] [Google Scholar]
- Sharma A, Straubinger RM. (1994) Novel taxol formulations: preparation and characterization of taxol-containing liposomes. Pharm Res 11:889–896 [DOI] [PubMed] [Google Scholar]
- Sharma US, Sharma A, Chau RI, Straubinger RM. (1997) Liposome-mediated therapy of intracranial brain tumors in a rat model. Pharm Res 14:992–998 [DOI] [PubMed] [Google Scholar]
- Simeoni M, Magni P, Cammia C, De Nicolao G, Croci V, Pesenti E, Germani M, Poggesi I, Rocchetti M. (2004) Predictive pharmacokinetic-pharmacodynamic modeling of tumor growth kinetics in xenograft models after administration of anticancer agents. Cancer Res 64:1094–1101 [DOI] [PubMed] [Google Scholar]
- Sparreboom A, Baker SD, Verweij J. (2005a) Paclitaxel repackaged in an albumin-stabilized nanoparticle: handy or just a dandy? J Clin Oncol 23:7765–7767 [DOI] [PubMed] [Google Scholar]
- Sparreboom A, Scripture CD, Trieu V, Williams PJ, De T, Yang A, Beals B, Figg WD, Hawkins M, Desai N. (2005b) Comparative preclinical and clinical pharmacokinetics of a cremophor-free, nanoparticle albumin-bound paclitaxel (ABI-007) and paclitaxel formulated in Cremophor (Taxol). Clin Cancer Res 11:4136–4143 [DOI] [PubMed] [Google Scholar]
- Sun YN, Jusko WJ. (1998) Transit compartments versus gamma distribution function to model signal transduction processes in pharmacodynamics. J Pharm Sci 87:732–737 [DOI] [PubMed] [Google Scholar]
- ten Tije AJ, Verweij J, Loos WJ, Sparreboom A. (2003) Pharmacological effects of formulation vehicles: implications for cancer chemotherapy. Clin Pharmacokinet 42:665–685 [DOI] [PubMed] [Google Scholar]
- Trédan O, Galmarini CM, Patel K, Tannock IF. (2007) Drug resistance and the solid tumor microenvironment. J Natl Cancer Inst 99:1441–1454 [DOI] [PubMed] [Google Scholar]
- Wang J, Lou P, Lesniewski R, Henkin J. (2003) Paclitaxel at ultra low concentrations inhibits angiogenesis without affecting cellular microtubule assembly. Anticancer Drugs 14:13–19 [DOI] [PubMed] [Google Scholar]
- Weiss RB, Donehower RC, Wiernik PH, Ohnuma T, Gralla RJ, Trump DL, Baker JR, Jr, Van Echo DA, Von Hoff DD, Leyland-Jones B. (1990) Hypersensitivity reactions from taxol. J Clin Oncol 8:1263–1268 [DOI] [PubMed] [Google Scholar]
- Yang J, Mager DE, Straubinger RM. (2009) Comparison of two pharmacodynamic transduction models for the analysis of tumor therapeutic responses in model systems. AAPS J doi: 10.1208/s12248-009-9155-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou R, Mazurchuk R, Straubinger RM. (2002) Antivasculature effects of doxorubicin-containing liposomes in an intracranial rat brain tumor model. Cancer Res 62:2561–2566 [PubMed] [Google Scholar]