Abstract
A novel plasmid-borne resistance gene cluster comprising the genes erm(T) for macrolide-lincosamide-streptogramin B resistance, dfrK for trimethoprim resistance, and tet(L) for tetracycline resistance was identified in a porcine methicillin-resistant Staphylococcus aureus sequence type 398 (ST398) strain. This erm(T)-dfrK-tet(L) region was flanked by copies of the novel IS element ISSau10. The erm(T) region resembled that of Streptococcus pyogenes plasmid pRW35. The erm(T) gene of pKKS25 was expressed constitutively due to a 57-bp deletion in the erm(T) translational attenuator.
In recent years, methicillin (meticillin)-resistant Staphylococcus aureus (MRSA) strains of sequence type 398 (ST398) have gained particular attention as a new clonal lineage associated with livestock (7, 22, 23, 28, 30). MRSA ST398 strains have been mainly found in pigs (7, 22, 23) but are also able to colonize and cause infections in other animal species and humans (13, 21, 28, 30). Recent studies have shown that virtually all MRSA ST398 strains are resistant to β-lactam antibiotics and tetracyclines (7, 13, 21, 22, 28) but vary in their patterns of resistance to other antimicrobial agents, including macrolides, lincosamides, and/or streptogramins (MLS). Resistance to MLS antibiotics in staphylococci is mediated by a number of different resistance genes which specify either target site-modifying enzymes, efflux pumps, or drug-inactivating enzymes (16). The latest update of MLS resistance genes (http://faculty.washington.edu/marilynr) listed the rRNA methylase genes erm(A), erm(B), erm(C), erm(F), erm(G), erm(Q), erm(Y), and erm(33) as occurring in staphylococci. Among these, erm(A), erm(B), and erm(C) have been found most frequently among staphylococci of animal origin (5, 11, 12). In MRSA ST398, macrolide-lincosamide (ML) resistance has been detected at various levels (7, 17, 21, 22, 28). In the few cases in which the genes responsible for ML resistance have been determined, the rRNA methylase genes erm(A), erm(B), and/or erm(C) have been identified (7, 10, 17, 28). In the present study, we investigated a porcine MRSA ST398 strain for the presence of the ML resistance gene, its location on a plasmid, and its association with other resistance genes.
MRSA ST398 strain 25 was obtained in 2008 from the nasal swab of a young sow in the GERM-Vet monitoring program. MIC determination by broth microdilution following the recommendations and using the clinical breakpoints given in CLSI documents M31-A3 (1) and M100-S19 (2) confirmed that this strain was resistant to β-lactam antibiotics, tetracycline, macrolides, lincosamides, and trimethoprim (Table 1). PCR analysis (9, 14, 15, 18) confirmed the presence of the genes mecA and blaZ for β-lactam resistance, the tetracycline resistance genes tet(K), tet(L), and tet(M), and the novel trimethoprim resistance gene dfrK. PCR screening for the most predominant ML resistance genes in staphylococci (11, 12) did not yield positive results. To investigate whether ML resistance was plasmid borne, plasmids were prepared and transformed into S. aureus RN4220 with subsequent selection on medium containing erythromycin (15 μg/ml) (8-10). A plasmid of ca. 40 kb, designated pKKS25, was identified and shown to confer not only combined resistance to macrolides and lincosamides but also tetracycline resistance via tet(L) and trimethoprim resistance via dfrK. Plasmid pKKS25 was subjected to restriction analysis with EcoRI, HindIII, and BglII, and the resulting fragments were cloned into the plasmid vector pBlueScript II SK+. Recombinant plasmids were transformed into macrolide-susceptible Escherichia coli strain AS19, and transformants were selected on erythromycin-containing Luria-Bertani agar (15 μg/ml).
TABLE 1.
Comparative analysis of the MICs of original pKKS25-carrying MRSA strain 25, S. aureus RN4220, and the S. aureus RN4220 transformant carrying plasmid pKKS25
| Bacterial strain | Resistance genes | MIC (μg/ml)a |
|||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PEN | AMP | OXA | ERY | TYL | CLI | TMP | TET | GEN | APR | SPE | CHL | ENR | TIA | VAL | LZD | VAN | Q-D | ||
| S. aureus RN4220 | ≤0.03 | 0.06 | 0.06 | 0.25 | 1 | ≤0.12 | 0.5 | 0.06 | 0.25 | 1 | 64 | 4 | 0.25 | 0.12 | 0.03 | 0.5 | 1 | 0.25 | |
| S. aureus RN4220/ pKKS25 | erm(T), dfrK, tet(L) | ≤0.03 | 0.06 | 0.06 | ≥128 | ≥64 | ≥64 | ≥128 | 32 | 0.25 | 1 | 64 | 4 | 0.25 | 0.12 | 0.03 | 0.5 | 1 | 0.5 |
| MRSA 25 | erm(T),bdfrK,btet(L),btet(K), tet(M), mecA, blaZ | 16 | 16 | 8 | ≥128 | ≥64 | ≥64 | ≥128 | 128 | 0.5 | 4 | 64 | 8 | 0.25 | 0.5 | 0.03 | 0.5 | 1 | 1 |
Abbreviations: PEN, penicillin G; AMP, ampicillin; OXA, oxacillin; ERY, erythromycin; TYL, tylosin tartrate; CLI, clindamycin; TMP, trimethoprim; TET, tetracycline; GEN, gentamicin; APR, apramycin; SPE, spectinomycin; CHL, chloramphenicol; ENR, enrofloxacin; TIA, tiamulin; VAL, valnemulin; LZD, linezolid; VAN, vancomycin; Q-D, quinupristin-dalfopristin.
Associated with plasmid pKKS25.
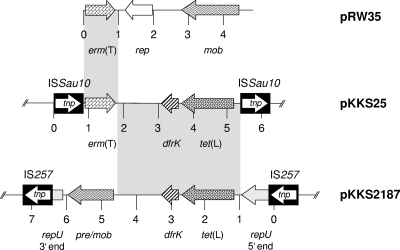
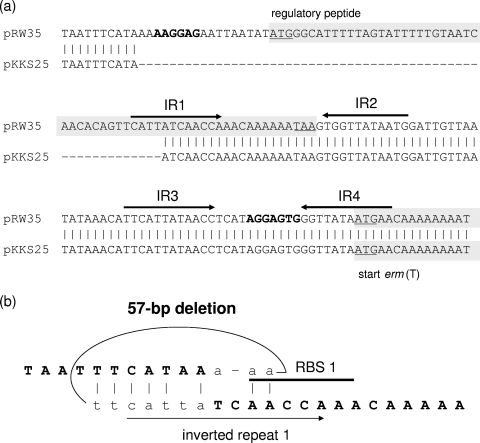
A 6,229-bp segment of pKKS25 was sequenced by primer walking on both strands from overlapping clones. A schematic representation of the genes found in this segment is shown in Fig. 1. ML resistance proved to be due to the rRNA methylase gene erm(T), which has previously been found in lactobacilli (6, 19, 27), streptococci (3, 4, 20, 29), and enterococci (4) but has not yet been identified in staphylococci. The erm(T) gene coded for a 244-amino-acid (aa) protein which was indistinguishable from the chromosomal Erm(T) protein of Streptococcus pasteurianus (20) but differed by 2 aa from the plasmid-encoded Erm(T) proteins of Lactobacillus sp. (27), Lactobacillus reuteri (19), and Streptococcus pyogenes (29). A detailed analysis of the erm(T) gene region of plasmid pKKS25 revealed identity to the erm(T) region of plasmid pRW35 from S. pyogenes in a stretch of 123 bp upstream and 139 bp downstream of erm(T) (Fig. 1). Previous studies identified a complete translational attenuator immediately upstream of the erm(T) gene which consisted of two pairs of inverted-repeat sequences of 12 bp each and a reading frame for a regulatory peptide of 19 aa (19, 20, 29). This translational attenuator closely resembled the erm(C)-associated translational attenuator in staphylococci. A complete translational attenuator is required for inducible erm gene expression, while constitutive erm gene expression is often based on deletions or duplications in this regulatory region (24). Strains carrying constitutively expressed erm genes are resistant to noninducers such as lincosamides and 16-membered macrolides (24, 26). A truncated attenuator had been identified upstream of the erm(T) gene of plasmid p121BS from a tylosin-resistant Lactobacillus sp. (27). Plasmid pKKS25 was isolated from an MRSA strain that exhibited a resistance phenotype indicative of constitutive erm gene expression, namely, resistance to clindamycin and tylosin. Analysis of the pKKS25-associated translational attenuator revealed a 57-bp deletion which comprised the initial 40 bp of the reading frame for the 19-aa regulatory peptide, including the upstream part with the ribosome binding site (Fig. 2a). Based on the knowledge of structural changes in the translational attenuators of erm(C) genes, limited areas of homology are sufficient to allow recombinational events that account for deletions (26). Hence, it is most likely that the observed 57-bp deletion was also due to such a recombination (Fig. 2b). The remaining part of the resistance gene region of plasmid pKKS25, including the dfrK and tet(L) genes, corresponded closely (>99% nucleotide sequence identity) to that of plasmid pKKS2187 (9).
FIG. 1.
Comparison of plasmid pKKS25 (accession no. FN390947), identified in the present study, with the tet(L)-dfrK segment of plasmid pKKS2187 (accession no. FM207105) and erm(T)-carrying plasmid pRW35 (accession no. EU192194). The arrows indicate the extents and directions of transcription of the genes rep, repU (plasmid replication), tet(L) (tetracycline resistance), dfrK (trimethoprim resistance), pre/mob (plasmid recombination/mobilization), and erm(T) (combined resistance to macrolides and lincosamides). The IS257 elements in the map of pKKS2187 and the ISSau10 elements in the map of pKKS25 are shown as black boxes with the white arrow indicating the transposase gene tnp. The regions with >99% homology between pKKS25 and plasmids pKKS2187 and pRW35 are marked by gray shading.
FIG. 2.
(a) Comparison of the complete erm(T) regulatory region of pRW35 with the truncated region of pKKS25. The 57-bp deletion in pKKS25 is indicated by dashes. Vertical bars indicate bases that are identical in the two sequences. The two pairs of inverted-repeat sequences, IR1-IR2 and IR3-IR4, are marked by arrows. The reading frames for the 19-aa regulatory peptide and the 5′-terminal part of the erm(T) gene are indicated by gray shading; translational start and stop codons are underlined. Ribosomal binding sites RBS 1 and RBS 2 are displayed in boldface letters. (b) Possible site for recombination in the erm(T) translational attenuator of plasmid pKKS25 leading to the observed 57-bp deletion. The sequence determined in the pKKS25-associated translational attenuator is displayed in boldface capital letters. The lowercase letters represent the beginning and end of the deleted sequence. Important structural elements with respect to panel a, such as the ribosomal binding site RBS 1 and the IR1 sequence, are indicated. Vertical bars mark identical bases in the sequences involved in the recombinational events.
This erm(T)-dfrK-tet(L) resistance gene region was flanked by two copies of the novel insertion sequence ISSau10 oriented in the same direction (Fig. 1). This insertion sequence consisted of 793 bp and showed 87 to 88% identity to IS431 or IS257. The 224-aa transposase of ISSau10 exhibited 92 and 93% amino acid identity to the transposases of IS431 and IS257, respectively. The novel IS element carried 16-bp perfect inverted repeats (5′-GGTTCTGTTGCAAAGT-3′) at both termini. IS257 elements were shown to be involved in the integration of tet(L)-dfrK-carrying, as well as tet(K)-carrying, plasmids into larger plasmids (9, 25), and IS1216V-like elements were shown to be involved in the integration of the erm(T) gene into the chromosome of S. pasteurianus (20). It is likely that the novel ISSau10 insertion sequences played a role in the formation of the resistance gene cluster detected on plasmid pKKS25.
In conclusion, this is the first description of the gene erm(T) in staphylococci. Taking into account that tetracyclines, trimethoprim, and macrolides are among the most frequently sold antimicrobial agents in veterinary medicine worldwide, a gene cluster consisting of erm(T), dfrK, and tet(L) offers excellent options for coselection under the selective pressure imposed by the use of any of these antimicrobial agents. Moreover, the finding of erm(T) in an MRSA ST398 strain points toward the gene acquisition capacities of such strains and possible partners for gene exchange processes beyond the genus Staphylococcus.
Nucleotide sequence accession number.
The sequence of the resistance gene region of plasmid pKKS25 has been deposited in the EMBL database under accession number FN390947.
Acknowledgments
We thank Kerstin Meyer and Vera Nöding for excellent technical assistance and Heike Kaspar and Ulrike Steinacker for providing MRSA strain 25.
This study was financially supported by internal funding from the Friedrich-Loeffler-Institut.
Footnotes
Published ahead of print on 14 December 2009.
REFERENCES
- 1.Clinical and Laboratory Standards Institute. 2008. Performance standards for antimicrobial disk and dilution susceptibility tests for bacteria isolated from animals; approved standard—third edition. CLSI document M31-A3. Clinical and Laboratory Standards Institute, Wayne, PA.
- 2.Clinical and Laboratory Standards Institute. 2009. Performance standards for antimicrobial susceptibility testing; nineteenth informational supplement. CLSI document M100-S19. Clinical and Laboratory Standards Institute, Wayne, PA.
- 3.DiPersio, L. P., and J. R. DiPersio. 2007. Identification of an erm(T) gene in strains of inducibly clindamycin-resistant group B Streptococcus. Diagn. Microbiol. Infect. Dis. 57:189-193. [DOI] [PubMed] [Google Scholar]
- 4.DiPersio, L. P., J. R. DiPersio, K. C. Frey, and J. A. Beach. 2008. Prevalence of the erm(T) gene in clinical isolates of erythromycin-resistant group D Streptococcus and Enterococcus. Antimicrob. Agents Chemother. 52:1567-1569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Eady, E. A., J. I. Ross, J. L. Tipper, C. E. Walters, J. H. Cove, and W. C. Noble. 1993. Distribution of genes encoding erythromycin ribosomal methylases and an erythromycin efflux pump in epidemiologically distinct groups of staphylococci. J. Antimicrob. Chemother. 31:211-217. [DOI] [PubMed] [Google Scholar]
- 6.Egervärn, M., S. Roos, and H. Lindmark. 2009. Identification and characterization of antibiotic resistance genes in Lactobacillus reuteri and Lactobacillus plantarum. J. Appl. Microbiol. 107:1658-1668. [DOI] [PubMed] [Google Scholar]
- 7.Kadlec, K., R. Ehricht, S. Monecke, U. Steinacker, H. Kaspar, J. Mankertz, and S. Schwarz. 2009. Diversity of antimicrobial resistance pheno- and genotypes of methicillin-resistant Staphylococcus aureus ST398 from diseased swine. J. Antimicrob. Chemother. 64:1156-1164. [DOI] [PubMed] [Google Scholar]
- 8.Kadlec, K., and S. Schwarz. 2009. Identification of a novel ABC transporter gene, vga(C), located on a multiresistance plasmid from a porcine methicillin-resistant Staphylococcus aureus ST398 strain. Antimicrob. Agents Chemother. 53:3589-3591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kadlec, K., and S. Schwarz. 2009. Identification of a novel trimethoprim resistance gene, dfrK, in a methicillin-resistant Staphylococcus aureus ST398 strain and its physical linkage to the tetracycline resistance gene tet(L). Antimicrob. Agents Chemother. 53:776-778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kehrenberg, C., C. Cuny, B. Strommenger, S. Schwarz, and W. Witte. 2009. Methicillin-resistant and -susceptible Staphylococcus aureus strains of clonal lineages ST398 and ST9 from swine carry the multidrug resistance gene cfr. Antimicrob. Agents Chemother. 53:779-781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lüthje, P., and S. Schwarz. 2007. Molecular basis of resistance to macrolides and lincosamides among staphylococci and streptococci from various animal sources collected in the resistance monitoring program BfT-GermVet. Int. J. Antimicrob. Agents 29:528-535. [DOI] [PubMed] [Google Scholar]
- 12.Lüthje, P., and S. Schwarz. 2006. Antimicrobial resistance of coagulase-negative staphylococci from bovine subclinical mastitis with particular reference to macrolide-lincosamide resistance phenotypes and genotypes. J. Antimicrob. Chemother. 57:966-969. [DOI] [PubMed] [Google Scholar]
- 13.Nienhoff, U., K. Kadlec, I. F. Chaberny, J. Verspohl, G.-F. Gerlach, S. Schwarz, D. Simon, and I. Nolte. 2009. Transmission of methicillin-resistant Staphylococcus aureus strains between humans and dogs: two case reports. J. Antimicrob. Chemother. 64:660-662. [DOI] [PubMed] [Google Scholar]
- 14.Pang, Y., T. Bosch, and M. C. Roberts. 1994. Single polymerase chain reaction for the detection of tetracycline-resistant determinants Tet K and Tet L. Mol. Cell. Probes 8:417-422. [DOI] [PubMed] [Google Scholar]
- 15.Roberts, M. C., Y. Pang, D. E. Riley, S. L. Hillier, R. C. Berger, and J. N. Krieger. 1993. Detection of Tet M and Tet O tetracycline resistance genes by polymerase chain reaction. Mol. Cell. Probes 7:387-393. [DOI] [PubMed] [Google Scholar]
- 16.Roberts, M. C., J. Sutcliffe, P. Courvalin, L. B. Jensen, J. Rood, and H. Seppala. 1999. Nomenclature for macrolide and macrolide-lincosamide-streptogramin B resistance determinants. Antimicrob. Agents Chemother. 43:2823-2830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Schwarz, S., K. Kadlec, and B. Strommenger. 2008. Methicillin-resistant Staphylococcus aureus and Staphylococcus pseudintermedius detected in the BfT-GermVet monitoring programme 2004-2006 in Germany. J. Antimicrob. Chemother. 61:282-285. [DOI] [PubMed] [Google Scholar]
- 18.Strommenger, B., C. Kehrenberg, C. Kettlitz, C. Cuny, J. Verspohl, W. Witte, and S. Schwarz. 2006. Molecular characterization of methicillin-resistant Staphylococcus aureus strains from pet animals and their relationship to human isolates. J. Antimicrob. Chemother. 57:461-465. [DOI] [PubMed] [Google Scholar]
- 19.Tannock, G. W., J. B. Luchansky, L. Miller, H. Connell, S. Thode-Andersen, A. A. Mercer, and T. R. Klaenhammer. 1994. Molecular characterization of a plasmid-borne (pGT633) erythromycin resistance determinant (ermGT) from Lactobacillus reuteri 100-63. Plasmid 31:60-71. [DOI] [PubMed] [Google Scholar]
- 20.Tsai, J. C., P. R. Hsueh, H. J. Chen, S. P. Tseng, P. Y. Chen, and L. J. Teng. 2005. The erm(T) gene is flanked by IS1216V in inducible erythromycin-resistant Streptococcus gallolyticus subsp. pasteurianus. Antimicrob. Agents Chemother. 49:4347-4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.van den Eede, A., A. Martens, U. Lipinska, M. Struelens, A. Deplano, O. Denis, F. Haesebrouck, F. Gasthuys, and K. Hermans. 2009. High occurrence of methicillin-resistant Staphylococcus aureus ST398 in equine nasal samples. Vet. Microbiol. 133:138-144. [DOI] [PubMed] [Google Scholar]
- 22.van Duijkeren, E., M. D. Jansen, S. C. Flemming, H. de Neeling, J. A. Wagenaar, A. H. W. Schoormans, A. van Nes, and A. C. Fluit. 2007. Methicillin-resistant Staphylococcus aureus in pigs with exudative epidermitis. Emerg. Infect. Dis. 13:1408-1410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Voss, A., F. Loeffen, J. Bakker, C. Klaassen, and M. Wulf. 2005. Methicillin-resistant Staphylococcus aureus in pig farming. Emerg. Infect. Dis. 11:1965-1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Weisblum, B. 1995. Insights into erythromycin action from studies of its activity as inducer of resistance. Antimicrob. Agents Chemother. 39:797-805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Werckenthin, C., S. Schwarz, and M. C. Roberts. 1996. Integration of pT181-like tetracycline resistance plasmids into large staphylococcal plasmids involves IS257. Antimicrob. Agents Chemother. 40:2542-2544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Werckenthin, C., S. Schwarz, and H. Westh. 1999. Structural alterations in the translational attenuator of constitutively expressed ermC genes. Antimicrob. Agents Chemother. 43:1681-1685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Whitehead, T. R., and M. A. Cotta. 2001. Sequence analyses of a broad host-range plasmid containing ermT from a tylosin-resistant Lactobacillus sp. isolated from swine feces. Curr. Microbiol. 43:17-20. [DOI] [PubMed] [Google Scholar]
- 28.Witte, W., B. Strommenger, C. Stanek, and C. Cuny. 2007. Methicillin-resistant Staphylococcus aureus ST398 in humans and animals, Central Europe. Emerg. Infect. Dis. 13:255-258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Woodbury, R. L., K. A. Klammer, Y. Xiong, T. Bailiff, A. Glennen, J. M. Bartkus, R. Lynfield, C. Van Beneden, and B. W. Beall for the Active Bacterial Core Surveillance Team. 2008. Plasmid-borne erm(T) from invasive, macrolide-resistant Streptococcus pyogenes strains. Antimicrob. Agents Chemother. 52:1140-1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wulf, M. W., A. Markestein, F. T. van der Linden, A. Voss, C. Klaassen, and C. M. Verduin. 2008. First outbreak of methicillin-resistant Staphylococcus aureus ST398 in a Dutch hospital, June 2007. Euro Surveill. 13:8051. [PubMed] [Google Scholar]