Abstract
We have previously described a murein hydrolase activity for the surface layer (S-layer) of Lactobacillus acidophilus ATCC 4356. Here we show that, in combination with nisin, this S-layer acts synergistically to inhibit the growth of pathogenic Gram-negative Salmonella enterica and potential pathogenic Gram-positive bacteria, Staphylococcus aureus and Bacillus cereus. In addition, bacteriolytic effects were observed for the Gram-positive species tested. We postulate that the S-layer enhances the access of nisin into the cell membrane by enabling it to cross the cell wall, while nisin provides the sudden ion-nonspecific dissipation of the proton motive force required to enhance the S-layer murein hydrolase activity.
Natural preservatives active against Gram-positive and Gram-negative pathogens are highly desirable for the food industry and consumers. Nisin, a small peptide bacteriocin produced by Lactococcus lactis subsp. lactis, is the only bacteriocin that has approved GRAS (generally recognized as safe) status for certain applications (Generally Regarded as Safe; US 21CFR170.30-Food Additives) by the Food and Drug Administration (FDA) for use in food products (3, 19). The antimicrobial mechanism of nisin has been extensively studied and is well documented (1, 5, 8, 9, 20). Nisin is not active against Gram-negative bacteria compared to Gram-positive bacteria, due to its inability to penetrate the external membrane, which prevents its access to the inner membrane. However, the combination of nisin with the chelating agent lactoferrin acts synergistically to inhibit the growth of Escherichia coli O157:H7 (13). Nisin has been used as a food preservative for over 30 years, is active in the nanomolar range, and has no known toxicity to humans (5, 19), which has placed it in the unique position of worldwide acceptance as a powerful and safe food additive in control of food spoilage and certain food pathogens.
We have previously described a new enzymatic functionality for the surface layer (S-layer) of Lactobacillus acidophilus ATCC 4356, namely, an endopeptidase activity against cell wall preparations of Salmonella enterica serovar Newport (17); however, we failed to observe any effect on whole cells from Gram-positive bacteria such as Bacillus cereus.
We now wonder if the purified S-layer protein having this endopeptidase activity may act synergistically with nisin, allowing reduction of the levels of the bacteriocin and control of bacterial growth. For this purpose, the antibacterial activity of S-layer from Lactobacillus acidophilus ATCC 4356, either alone or in combination with nisin, was analyzed using three models of food-borne pathogenic bacteria: Salmonella enterica serovar Newport (10), Staphylococcus aureus ATCC 6538 (used as a representative of Gram-positive bacteria in standard medium tests; laboratory collection), and Bacillus cereus 6A1 (Bacillus Genetic Stock Center [BGSC]). The S-layer proteins were extracted as previously described (17) and conserved as a 1-mg/ml suspension in sterile distilled water at −20°C until use. Purity was determined by SDS-PAGE as a unique band and detected by Western blotting.
Nisin was used as its commercial product, 2.5% (wt/wt) (Sigma), and was dissolved in 0.02 N HCl to a concentration of 16,000 IU/ml, equivalent to 0.4 mg/ml pure nisin. The MIC that inhibited growth was determined twice by microtiter broth dilution, and we measured the optical density at 600 nm (OD600) reached after 16 h of growth. Bacillus cereus and Salmonella enterica were grown in LB (yeast extract, 5 g/liter; peptone, 10 g/liter; NaCl, 10 g/liter; pH 7.0), while Staphylococcus aureus was grown in BHI (brain heart infusion; Biokar, France) and incubated at 37°C. The MIC was 595 IU/ml for Bacillus cereus, 298 IU/ml for Staphylococcus aureus, and over 5,000 IU/ml for Salmonella enterica serovar Newport. For Salmonella enterica, nisin solution was prepared to a concentration of 160,000 IU/ml, equivalent to 4 mg/ml pure nisin, in order to avoid a pH modification once it was added to the growth medium.
In a first approach, growth curves of the Gram-negative Salmonella enterica serovar Newport in the presence of nisin, S-layer, or both were performed. Nisin at half of its MIC showed no inhibition. The S-layer alone was inhibitory, but the addition of both nisin and S-layer decreased growth (for growth rate and maximal OD reached, see Fig. 1). In view of these findings, we decided to evaluate the effects of the combination (nisin and S-layer) in Gram-positive bacteria. Bacillus cereus and Staphylococcus aureus, two food-borne pathogen models, were chosen. No effect was observed when the S-layer (10 μg/ml) was added alone, and nisin added alone, at a subinhibitory concentration of 250 IU/ml for B.cereus and 150 IU/ml for S. aureus, partially delayed growth. In contrast, the combination of both the S-layer (10 μg/ml) and nisin at a subinhibitory concentration inhibited growth of both B. cereus and S. aureus cultures (Fig. 1).
FIG. 1.
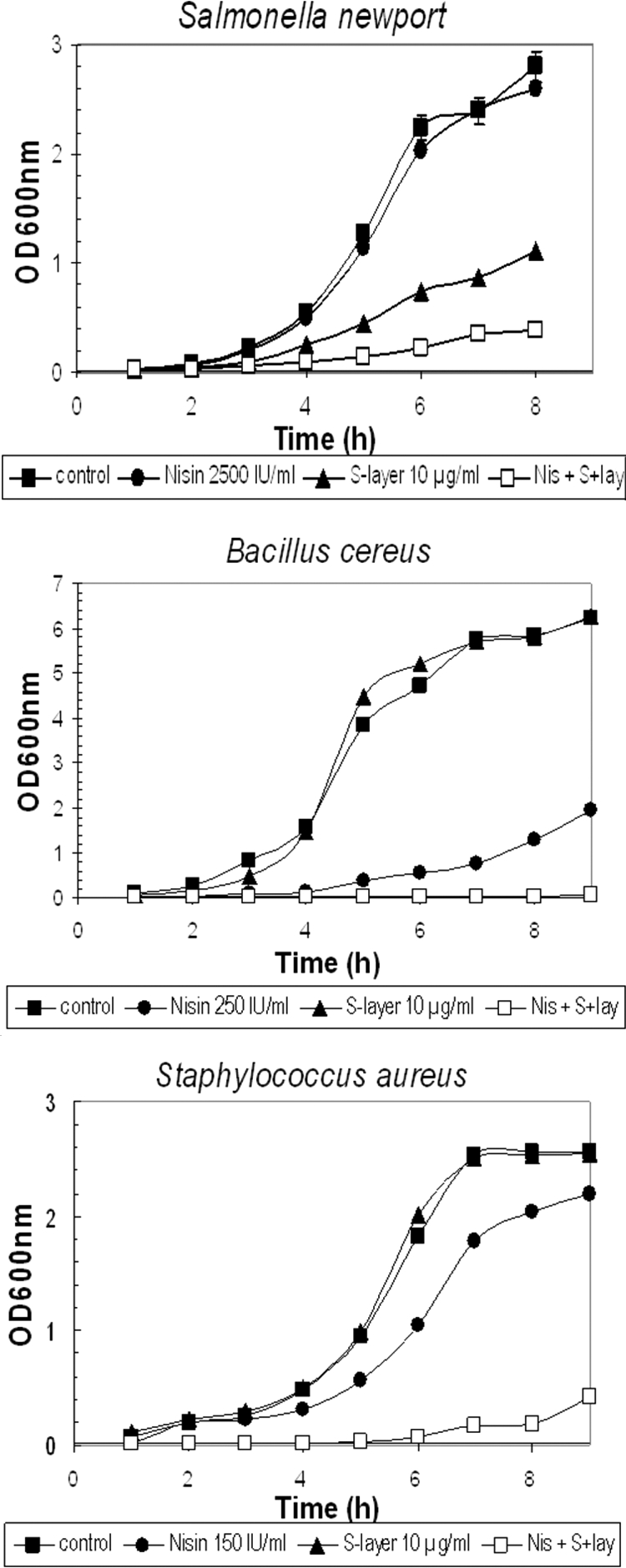
Effect of S-layer, nisin, or both on the growth of S. enterica, S. aureus, and B. cereus. To determine the effect of nisin and S-layer protein separately or in combination, bacterial cells were first cultured at 37°C for 16 h with agitation. These cultures were diluted in fresh medium (0.5 ml into 10 ml) and distributed in aliquots, each containing either the S-layer, nisin, or both, and OD600 was monitored every hour. Three or more independent experiments were performed for each bacterium. Nisin was added at the following concentrations: 2,500 IU/ml for Salmonella enterica, 250 IU/ml for Bacillus cereus, and 150 IU/ml for Staphylococcus aureus; the concentration of S-layer was 10 μg/ml.
To determine the mode of action of the combination of S-layer and nisin, lysis was monitored by OD600 decrease of exponential cultures of B. cereus and S. aureus when exposed to nisin or nisin plus S-layer and viable counts were determined by serial decimal dilutions on LB agar plates. A bacteriolytic effect was observed. As shown in Fig. 2A, B. cereus cultures lysed instantly in the presence of both compounds as observed from the decrease in OD and confirmed by the viable count determinations. However, S. aureus showed a 2-h lag period before lysis was observed (Fig. 2B). This delay may be attributed to the typical cell aggregation profile characteristic of Staphylococcus cultures, which might hide the target for the S-layer and/or nisin. In fact, after this period cells disaggregated as visualized by microscopic observation (data not shown) and both compounds led to cell lysis. In addition, when viable counts were performed, a rapid decline in viability was observed even before the OD decrease. The reduction in viability after a 6-h treatment with both nisin and S-layer was 5 and 4 logs for B. cereus and S. aureus, respectively.
FIG. 2.
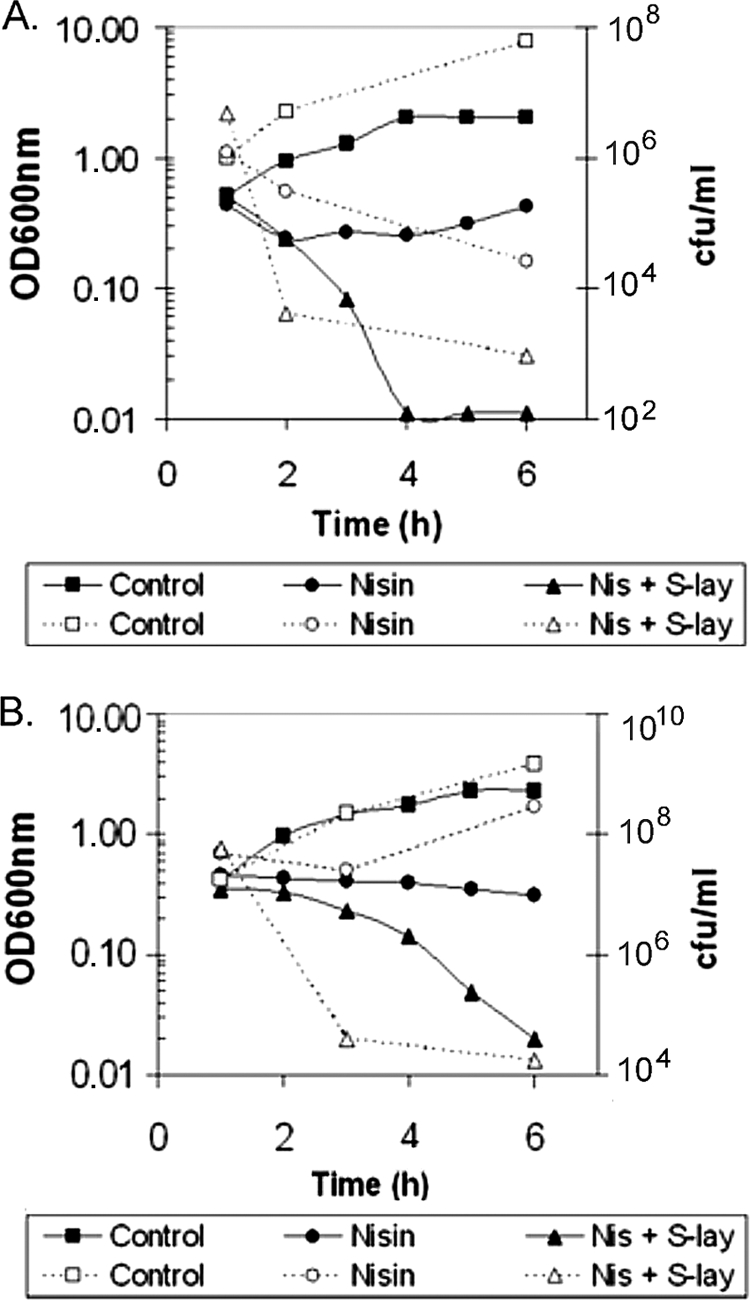
Type of effect of the combination of nisin and S-layer. Exponentially growing bacteria were incubated in the presence of either nisin or nisin and S-layer. (A) Effect on Bacillus cereus with nisin (250 IU/ml) or nisin and S-layer (250 IU/ml and 10 μg/ml, respectively). (B) Effect on Staphylococcus aureus with nisin (150 IU/ml) or nisin and S-layer (150 IU/ml and 10 μg/ml, respectively). Dotted line, cfu/ml; unbroken line, OD at 600 nm.
Concerning the Gram-negative Salmonella enterica, a peculiar behavior was observed: while whole cells suspended in buffer were lysed by the sole presence of S-layer (17), no lytic effect was observed when exponentially growing cultures were treated even with both S-layer and nisin (data not shown). However, the S-layer protein remains intact after incubation with whole cells as observed by Western blotting after 8 h of incubation (Fig. 3). We suspect that actively dividing cells of this Gram-negative pathogen may release an inhibitor either of the S-layer activity or of nisin. Further experiments should be performed in order to evaluate these hypotheses.
FIG. 3.
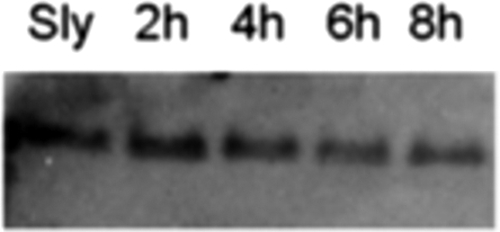
S-layer integrity during incubation with Salmonella. Viable cells from Salmonella enterica were washed once with phosphate-buffered saline buffer, resuspended at an optical density at 600 nm of 1 mixed with S-layer protein (10 μg/ml), and incubated at 37°C with constant rotation to keep the contents in suspension at the indicated times. At different times S-layer was analyzed by Western blotting. Electrotransfer to parablot polyvinylidene difluoride was according to the manufacturer's instructions (Macherey-Nagel, Germany). Polyclonal antibody anti-S-layer was used at 1:1,000.
To check if the synergic effect of S-layer on nisin is indeed due to its murein hydrolase activity, a mild condition of heat inactivation (60°C for 2 h) was assayed with the S-layer. This treatment inactivates the endopeptidase activity as seen by zymogram analysis (Fig. 4, zymogram, lane 3) but conserves the protein without dramatic changes, since no differences in migration or antibody detection were observed (compare lanes 3 and 1 in Fig. 4). The addition of mild-heat-inactivated S-layer protein and nisin to S. aureus cultures was unable to inhibit growth (Table 1). In contrast, while nisin and S-layer separately inhibited growth to 22% or 0%, respectively, the addition of both inhibited dramatically the growth (89%) and growth rate (90%), representing an 8-log reduction in viable cell counts, pointing to the benefit of this synergetic effect (Table 1).
FIG. 4.
S-layer treatments. S-layer preparations were submitted to different treatments and analyzed by PAGE, zymography, and Western immunoblotting as previously described (16). Lane 1, S-layer; lane 2, pretreatment with nisin of L. acidophilus cultures before S-layer preparation; lane 3, heat-inactivated S-layer (2 h at 60°C); lane 4, S-layer preparation incubated with nisin.
TABLE 1.
Nature of the synergic combinationa
| Growth condition | Final OD600 | Growth rate (μ; h−1) | % Inhibitionc |
|---|---|---|---|
| Control | 2.6 | 0.48 ± 0.02 | 0 |
| Nisin (150 IU/ml) | 2.2 | 0.37 ± 0.01 | 22 |
| S-layer (10 μg/ml) | 2.6 | 0.49 ± 0.02 | 0 |
| Nisin (150 IU/ml) + S-layer (10 μg/ml) | 0.7 | 0.05 ± 0.01 | 89 |
| S-layer (heat inactivated)b | 2.8 | 0.53 ± 0.02 | ≪0 |
| S-layer (heat inactivated) + nisin (150 IU/ml) | 2.5 | 0.42 ± 0.02 | 12 |
| CCCP (50 μM) | 0.9 | 0.11 ± 0.01 | 77 |
| CCCP (50 μM) + S-layer (10 μg/ml) | 0.4 | 0.04 ± 0.01 | 92 |
| CCCP (25 μM) | 1.2 | 0.21 ± 0.01 | 56 |
| CCCP (25 μM) + S-layer (10 μg/ml) | 0.6 | 0.11 ± 0.01 | 77 |
Staphylococcus aureus cultures were assayed as described for Fig. 1, and the different compounds were added at the indicated concentrations. Growth curves were performed, and the growth rate, μ, was determined from the resulting plots. Final OD600s after 8 h of growth were also indicated.
Heat-inactivated S-layer (10 μg/ml) was obtained by heating at 60°C for 2 h.
Percentage of inhibition was calculated from the growth rates and related to the control.
To verify that nisin does not act by activating the S-layer murein hydrolase activity, experiments were performed where this activity was evaluated from preparations incubated with nisin before S-layer extraction from L. acidophilus cultures or after its purification. In any case the SDS-PAGE and zymogram analysis revealed no difference in electrophoretic mobility or murein hydrolase activity (Fig. 4, compare lane 1 with lanes 2 and 4). In addition, the S-layer protein preparation is very stable since the incubation in the presence of target bacteria failed to reveal any proteolysis (Fig. 3).
A similar synergistic antibacterial activity between the peptide nisin and a cell wall hydrolase such as lysozyme (N-acetylmuramide glycanhydrolase) has been reported by various researchers (2, 7, 15). Nisin was involved in the dissipation of the membrane potential (18). To make sure that the synergic effect of nisin on S-layer is due to its proton motive force (PMF) dissipation, nisin was replaced by the PMF uncoupler carbonyl cyanide m-chlorophenylhydrazone (CCCP) and the effects compared with and without the addition of S-layer. Table 1 shows that nisin has an effect similar to that of CCCP.
The emergence of bacterial resistance to antibiotics following the widespread use of clinical, veterinary, and animal agricultural antibiotics has reduced their efficacy and made necessary new antibacterial alternatives. In this view bacteriophages, bacterial cell wall hydrolases, and antimicrobial peptides are among the most promising candidates (6, 11, 16). Most of them provide enzymes that degrade peptidoglycan, the main component of the bacterial cell wall, and cause bacteriolysis. The absence of lysis when the S-layer is alone may indicate the need of PMF dissipation for its full action. This effect has also been reported in the case of the holin-lysin system from bacteriophages for their delivery (14). The holin creates holes from inside into the cell-lipid bilayer, allowing the phage endolysin (peptidoglycan hydrolase) to break out and degrade the cell wall, allowing the release of phages. For it to be active, the endolysin requires the sudden ion-nonspecific dissipation of the proton motive force, an event undertaken by holin but also triggered by nisin from the outside (14). Therefore, cytoplasmic membrane electrochemical gradient dissipation is necessary but not sufficient for the full sensitization.
One important fact in our findings is that the combined effect of nisin and the S-layer resulted not only in the inhibition of growth of cultures when they are present in the initial inoculum but also in the ability to produce lysis in pregrown cultures, thus killing Gram-positive pathogen cells.
The S-layer-nisin synergetic duo seems to be a promising new antibacterial agent, which needs further investigation for its application in the food industry or food preservation as described previously (4, 12).
Acknowledgments
We thank the anonymous reviewers for comments that allowed us to improve this work.
This work was supported by grants from the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) from Argentina. M.P.-A., M.C.A., and M.M.P. are fellows of CONICET. C.S.R. and S.M.R. are career investigators of CONICET.
Footnotes
Published ahead of print on 30 November 2009.
REFERENCES
- 1.Breukink, E., C. van Kraaij, A. van Dalen, R. A. Demel, R. J. Siezen, B. de Kruijff, and O. P. Kuipers. 1998. The orientation of nisin in membranes. Biochemistry 37:8153-8162. [DOI] [PubMed] [Google Scholar]
- 2.Chung, W., and R. E. W. Hancock. 2000. Action of lysozyme and nisin mixtures against lactic acid bacteria. Int. J. Food Microbiol. 60:25-32. [DOI] [PubMed] [Google Scholar]
- 3.Cleveland, J., T. J. Montville, I. F. Nes, and M. L. Chikindas. 2001. Bacteriocins: safe, natural antimicrobials for food preservation. Int. J. Food Microbiol. 71:1-20. [DOI] [PubMed] [Google Scholar]
- 4.Datta, S., M. E. Janes, Q. G. Xue, J. Losso, and J. F. La Peyre. 2008. Control of Listeria monocytogenes and Salmonella anatum on the surface of smoked salmon coated with calcium alginate coating containing oyster lysozyme and nisin. J. Food Sci. 73:67-71. [DOI] [PubMed] [Google Scholar]
- 5.Delves-Broughton, J., P. Blackburn, R. J. Evans, and J. Hugenholtz. 1996. Applications of the bacteriocin, nisin. Antonie Van Leeuwenhoek 69:193-202. [DOI] [PubMed] [Google Scholar]
- 6.Fischetti, V. A. 2008. Bacteriophage lysins as effective antibacterials. Curr. Opin. Microbiol. 11:393-400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gill, C. O., and R. A. Holley. 2000. Surface application of lysozyme, nisin, and EDTA to inhibit spoilage and pathogenic bacteria on ham and bologna. J. Food Prot. 63:1338-1346. [DOI] [PubMed] [Google Scholar]
- 8.Hasper, H. E., N. E. Kramer, J. L. Smith, J. D. Hillman, C. Zachariah, O. P. Kuipers, B. de Kruijff, and E. Breukink. 2006. An alternative bactericidal mechanism of action for lantibiotic peptides that target lipid II. Science 313:1636-1637. [DOI] [PubMed] [Google Scholar]
- 9.Hyde, A. J., J. Parisot, A. McNichol, and B. B. Bonev. 2006. Nisin-induced changes in Bacillus morphology suggest a paradigm of antibiotic action. Proc. Natl. Acad. Sci. U. S. A. 103:19896-19901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ibar, M. P., G. Vigo, P. Piñeyro, M. I. Caffer, P. Quiroga, and C. Perfumo. 2009. Serovariedades de Salmonella enterica subespecie enterica en porcinos de faena y su resistencia a los antimicrobianos. Rev. Argent. Microbiol. 41:156-162. [PubMed] [Google Scholar]
- 11.Loeffler, J. M., and V. A. Fischetti. 2003. Synergistic lethal effect of a combination of phage lytic enzymes with different activities on penicillin-sensitive and resistant Streptococcus pneumoniae strains. Antimicrob. Agents Chemother. 47:375-377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mangalassary, S., I. Han, J. Rieck, J. Acton, X. Jiang, B. Sheldon, and P. Dawson. 2007. Effect of combining nisin and/or lysozyme with in-package pasteurization on thermal inactivation of Listeria monocytogenes in ready-to-eat turkey bologna. J. Food Prot. 70:2503-2511. [DOI] [PubMed] [Google Scholar]
- 13.Murdock, C. A., J. Cleveland, K. R. Matthews, and M. L. Chikindas. 2007. The synergistic effect of nisin and lactoferrin on the inhibition of Listeria monocytogenes and Escherichia coli O157:H7. Lett. Appl. Microbiol. 44:255-261. [DOI] [PubMed] [Google Scholar]
- 14.Nascimento, J. G., M. C. Guerreiro-Pereira, S. F. Costa, C. São-José, and M. A. Santos. 2008. Nisin-triggered activity of Lys44, the secreted endolysin from Oenococcus oeni phage fOg44. J. Bacteriol. 190:457-461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nattress, F. M., and L. P. Baker. 2003. Effects of treatment with lysozyme and nisin on the microflora and sensory properties of commercial pork. Int. J. Food Microbiol. 85:259-267. [DOI] [PubMed] [Google Scholar]
- 16.Parisien, A., B. Allain, J. Zhang, R. Mandeville, and C. Q. Lan. 2008. Novel alternatives to antibiotics: bacteriophages, bacterial cell wall hydrolases, and antimicrobial peptides. J. Appl. Microbiol. 104:1-13. [DOI] [PubMed] [Google Scholar]
- 17.Prado Acosta, M., M. M. Palomino, M. C. Allievi, C. Sanchez Rivas, and S. M. Ruzal. 2008. Murein hydrolase activity in the surface layer of Lactobacillus acidophilus ATCC 4356. Appl. Environ. Microbiol. 74:7824-7827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ruhr, E., and H. G. Sahl. 1985. Mode of action of the peptide antibiotic nisin and influence on the membrane potential of whole cells and on cytoplasmic and artificial membrane vesicles. Antimicrob. Agents Chemother. 27:841-845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.U.S. Food and Drug Administration. 1988. Nisin preparation: affirmation of GRAS status as a direct human food ingredient. Fed. Regist. 53:11247. [Google Scholar]
- 20.Winkowski, K., R. T. Ludescher, and T. J. Montville. 1996. Physiochemical characterization of the nisin-membrane interaction with liposomes derived from Listeria monocytogenes. Appl. Environ. Microbiol. 62:323-327. [DOI] [PMC free article] [PubMed] [Google Scholar]



