Abstract
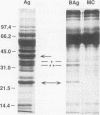
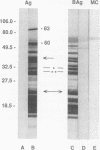
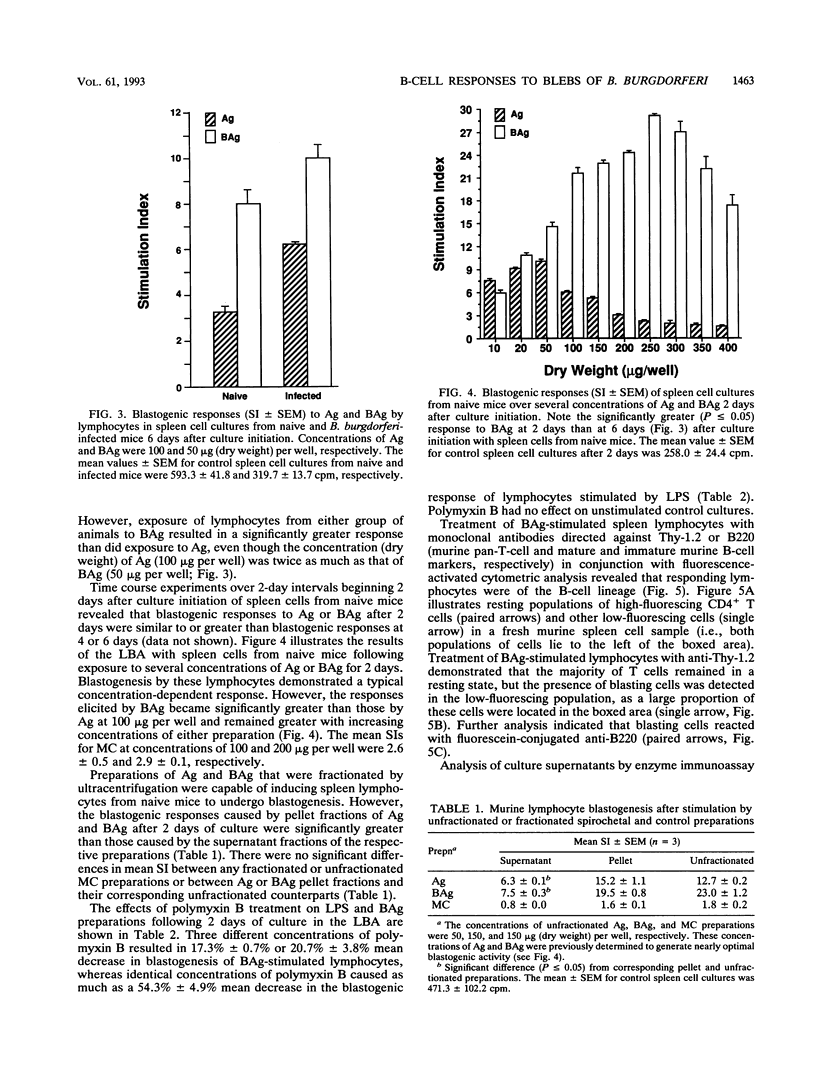
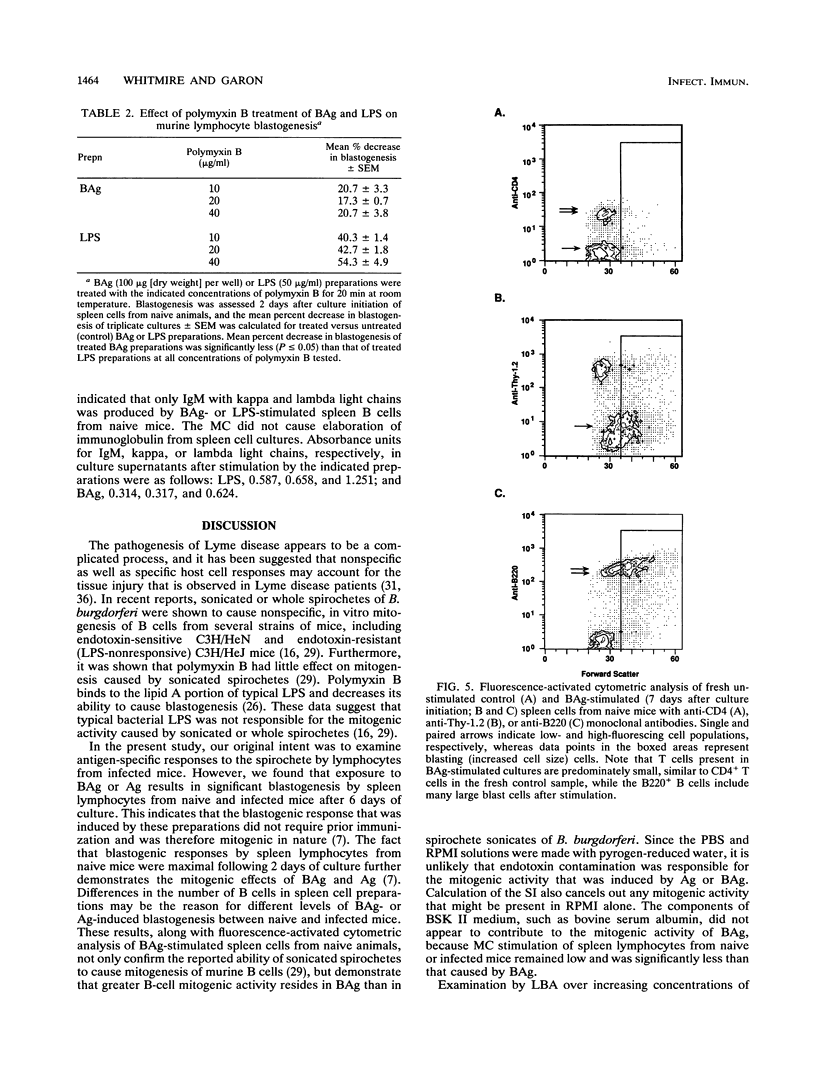
Lymphocyte blastogenesis assays and immunoblotting were used to investigate and compare murine B-cell responses to preparations of extracellular membrane blebs (BAg) and spirochetes (Ag) of Borrelia burgdorferi. Immunoblotting BAg, Ag, and medium control preparations with serum from naive and infected C57BL/10 mice revealed that BAg and Ag had similar specific reactivity profiles except that major antigens of 83, 60, and 41 kDa were detected in Ag but not in BAg. It was determined that 1 microgram (dry weight) of Ag contained 0.0051 and 0.0063 microgram of outer surface proteins A (OspA) and OspB, respectively, whereas 1 microgram (dry weight) of BAg contained 0.0024 microgram of OspA and 0.0015 microgram of OspB. Both BAg and Ag caused blastogenesis in cultures of spleen cells from both groups of mice, but BAg-stimulated lymphocytes exhibited significantly greater (P < or = 0.05) blastogenesis after 2 or 6 days of culture than did lymphocytes stimulated by Ag or medium control. Flow cytometry and antibody capture enzyme-linked immunosorbent assays identified responding lymphocytes as B cells which secreted polyclonal immunoglobulin M (IgM) but not IgG or IgA. Treatment of BAg and lipopolysaccharide controls with polymyxin B resulted in as much as 20.7 and 54.3% mean decreases in blastogenesis, respectively. Fractionation of BAg or Ag by ultracentrifugation before culture with spleen cells from naive mice indicated that B-cell blastogenesis was probably associated with spirochetal membranes. The results of this study demonstrate that specific humoral responses are directed towards extracellular membrane blebs which lack the 83-, 60-, and 41-kDa antigens of intact spirochetes and that blebs also possess significant nonspecific mitogenic activity for murine B cells. This activity was not due entirely to typical lipopolysaccharide or OspA and OspB lipoproteins.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barbour A. G., Hayes S. F., Heiland R. A., Schrumpf M. E., Tessier S. L. A Borrelia-specific monoclonal antibody binds to a flagellar epitope. Infect Immun. 1986 May;52(2):549–554. doi: 10.1128/iai.52.2.549-554.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbour A. G. Isolation and cultivation of Lyme disease spirochetes. Yale J Biol Med. 1984 Jul-Aug;57(4):521–525. [PMC free article] [PubMed] [Google Scholar]
- Beck G., Benach J. L., Habicht G. S. Isolation of interleukin 1 from joint fluids of patients with Lyme disease. J Rheumatol. 1989 Jun;16(6):800–806. [PubMed] [Google Scholar]
- Beck G., Benach J. L., Habicht G. S. Isolation, preliminary chemical characterization, and biological activity of Borrelia burgdorferi peptidoglycan. Biochem Biophys Res Commun. 1990 Feb 28;167(1):89–95. doi: 10.1016/0006-291x(90)91734-a. [DOI] [PubMed] [Google Scholar]
- Beck G., Habicht G. S., Benach J. L., Coleman J. L. Chemical and biologic characterization of a lipopolysaccharide extracted from the Lyme disease spirochete (Borrelia burgdorferi). J Infect Dis. 1985 Jul;152(1):108–117. doi: 10.1093/infdis/152.1.108. [DOI] [PubMed] [Google Scholar]
- Brandt M. E., Riley B. S., Radolf J. D., Norgard M. V. Immunogenic integral membrane proteins of Borrelia burgdorferi are lipoproteins. Infect Immun. 1990 Apr;58(4):983–991. doi: 10.1128/iai.58.4.983-991.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brusca J. S., McDowall A. W., Norgard M. V., Radolf J. D. Localization of outer surface proteins A and B in both the outer membrane and intracellular compartments of Borrelia burgdorferi. J Bacteriol. 1991 Dec;173(24):8004–8008. doi: 10.1128/jb.173.24.8004-8008.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burgdorfer W., Barbour A. G., Hayes S. F., Benach J. L., Grunwaldt E., Davis J. P. Lyme disease-a tick-borne spirochetosis? Science. 1982 Jun 18;216(4552):1317–1319. doi: 10.1126/science.7043737. [DOI] [PubMed] [Google Scholar]
- Craft J. E., Fischer D. K., Shimamoto G. T., Steere A. C. Antigens of Borrelia burgdorferi recognized during Lyme disease. Appearance of a new immunoglobulin M response and expansion of the immunoglobulin G response late in the illness. J Clin Invest. 1986 Oct;78(4):934–939. doi: 10.1172/JCI112683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Craft J. E., Grodzicki R. L., Steere A. C. Antibody response in Lyme disease: evaluation of diagnostic tests. J Infect Dis. 1984 May;149(5):789–795. doi: 10.1093/infdis/149.5.789. [DOI] [PubMed] [Google Scholar]
- Dattwyler R. J., Volkman D. J., Halperin J. J., Luft B. J., Thomas J., Golightly M. G. Specific immune responses in Lyme borreliosis. Characterization of T cell and B cell responses to Borrelia burgdorferi. Ann N Y Acad Sci. 1988;539:93–102. doi: 10.1111/j.1749-6632.1988.tb31842.x. [DOI] [PubMed] [Google Scholar]
- Dattwyler R. J., Volkman D. J., Luft B. J., Halperin J. J., Thomas J., Golightly M. G. Seronegative Lyme disease. Dissociation of specific T- and B-lymphocyte responses to Borrelia burgdorferi. N Engl J Med. 1988 Dec 1;319(22):1441–1446. doi: 10.1056/NEJM198812013192203. [DOI] [PubMed] [Google Scholar]
- Defosse D. L., Johnson R. C. In vitro and in vivo induction of tumor necrosis factor alpha by Borrelia burgdorferi. Infect Immun. 1992 Mar;60(3):1109–1113. doi: 10.1128/iai.60.3.1109-1113.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorward D. W., Huguenel E. D., Davis G., Garon C. F. Interactions between extracellular Borrelia burgdorferi proteins and non-Borrelia-directed immunoglobulin M antibodies. Infect Immun. 1992 Mar;60(3):838–844. doi: 10.1128/iai.60.3.838-844.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorward D. W., Schwan T. G., Garon C. F. Immune capture and detection of Borrelia burgdorferi antigens in urine, blood, or tissues from infected ticks, mice, dogs, and humans. J Clin Microbiol. 1991 Jun;29(6):1162–1170. doi: 10.1128/jcm.29.6.1162-1170.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garon C. F., Dorward D. W., Corwin M. D. Structural features of Borrelia burgdorferi--the Lyme disease spirochete: silver staining for nucleic acids. Scanning Microsc Suppl. 1989;3:109–115. [PubMed] [Google Scholar]
- Habicht G. S., Beck G., Benach J. L., Coleman J. L., Leichtling K. D. Lyme disease spirochetes induce human and murine interleukin 1 production. J Immunol. 1985 May;134(5):3147–3154. [PubMed] [Google Scholar]
- Hansen K., Bangsborg J. M., Fjordvang H., Pedersen N. S., Hindersson P. Immunochemical characterization of and isolation of the gene for a Borrelia burgdorferi immunodominant 60-kilodalton antigen common to a wide range of bacteria. Infect Immun. 1988 Aug;56(8):2047–2053. doi: 10.1128/iai.56.8.2047-2053.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hawkes R., Niday E., Gordon J. A dot-immunobinding assay for monoclonal and other antibodies. Anal Biochem. 1982 Jan 1;119(1):142–147. doi: 10.1016/0003-2697(82)90677-7. [DOI] [PubMed] [Google Scholar]
- Kurashige S., Bissett M., Oshiro L. Characterization of a tick isolate of Borrelia burgdorferi that possesses a major low-molecular-weight surface protein. J Clin Microbiol. 1990 Jun;28(6):1362–1366. doi: 10.1128/jcm.28.6.1362-1366.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lefebvre R. B., Perng G. C., Johnson R. C. The 83-kilodalton antigen of Borrelia burgdorferi which stimulates immunoglobulin M (IgM) and IgG responses in infected hosts is expressed by a chromosomal gene. J Clin Microbiol. 1990 Jul;28(7):1673–1675. doi: 10.1128/jcm.28.7.1673-1675.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrison D. C., Jacobs D. M. Binding of polymyxin B to the lipid A portion of bacterial lipopolysaccharides. Immunochemistry. 1976 Oct;13(10):813–818. doi: 10.1016/0019-2791(76)90181-6. [DOI] [PubMed] [Google Scholar]
- Perng G. C., LeFebvre R. B., Johnson R. C. Further characterization of a potent immunogen and the chromosomal gene encoding it in the Lyme disease agent, Borrelia burgdorferi. Infect Immun. 1991 Jun;59(6):2070–2074. doi: 10.1128/iai.59.6.2070-2074.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaible U. E., Kramer M. D., Justus C. W., Museteanu C., Simon M. M. Demonstration of antigen-specific T cells and histopathological alterations in mice experimentally inoculated with Borrelia burgdorferi. Infect Immun. 1989 Jan;57(1):41–47. doi: 10.1128/iai.57.1.41-47.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoenfeld R., Araneo B., Ma Y., Yang L. M., Weis J. J. Demonstration of a B-lymphocyte mitogen produced by the Lyme disease pathogen, Borrelia burgdorferi. Infect Immun. 1992 Feb;60(2):455–464. doi: 10.1128/iai.60.2.455-464.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shanafelt M. C., Hindersson P., Soderberg C., Mensi N., Turck C. W., Webb D., Yssel H., Peltz G. T cell and antibody reactivity with the Borrelia burgdorferi 60-kDa heat shock protein in Lyme arthritis. J Immunol. 1991 Jun 1;146(11):3985–3992. [PubMed] [Google Scholar]
- Sigal L. H. Immunology of Lyme disease. N J Med. 1990 Jul;87(7):567–571. [PubMed] [Google Scholar]
- Sigal L. H., Steere A. C., Dwyer J. M. In vivo and in vitro evidence of B cell hyperactivity during Lyme disease. J Rheumatol. 1988 Apr;15(4):648–654. [PubMed] [Google Scholar]
- Steere A. C., Hardin J. A., Ruddy S., Mummaw J. G., Malawista S. E. Lyme arthritis: correlation of serum and cryoglobulin IgM with activity, and serum IgG with remission. Arthritis Rheum. 1979 May;22(5):471–483. doi: 10.1002/art.1780220506. [DOI] [PubMed] [Google Scholar]
- Steere A. C. Lyme disease. N Engl J Med. 1989 Aug 31;321(9):586–596. doi: 10.1056/NEJM198908313210906. [DOI] [PubMed] [Google Scholar]
- Szczepanski A., Benach J. L. Lyme borreliosis: host responses to Borrelia burgdorferi. Microbiol Rev. 1991 Mar;55(1):21–34. doi: 10.1128/mr.55.1.21-34.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takayama K., Rothenberg R. J., Barbour A. G. Absence of lipopolysaccharide in the Lyme disease spirochete, Borrelia burgdorferi. Infect Immun. 1987 Sep;55(9):2311–2313. doi: 10.1128/iai.55.9.2311-2313.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilske B., Preac-Mursic V., Schierz G., Kühbeck R., Barbour A. G., Kramer M. Antigenic variability of Borrelia burgdorferi. Ann N Y Acad Sci. 1988;539:126–143. doi: 10.1111/j.1749-6632.1988.tb31846.x. [DOI] [PubMed] [Google Scholar]
- Yssel H., Shanafelt M. C., Soderberg C., Schneider P. V., Anzola J., Peltz G. Borrelia burgdorferi activates a T helper type 1-like T cell subset in Lyme arthritis. J Exp Med. 1991 Sep 1;174(3):593–601. doi: 10.1084/jem.174.3.593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Souza M. S., Fikrig E., Smith A. L., Flavell R. A., Barthold S. W. Nonspecific proliferative responses of murine lymphocytes to Borrelia burgdorferi antigens. J Infect Dis. 1992 Mar;165(3):471–478. doi: 10.1093/infdis/165.3.471. [DOI] [PubMed] [Google Scholar]