Abstract
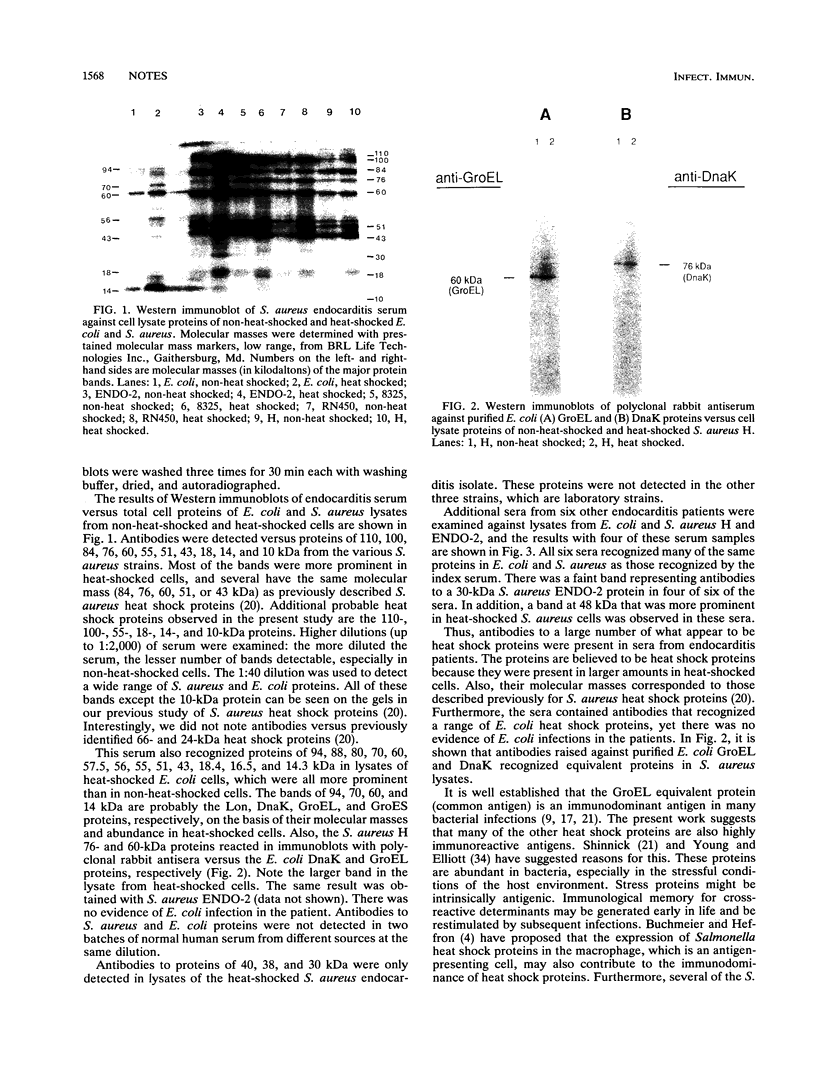
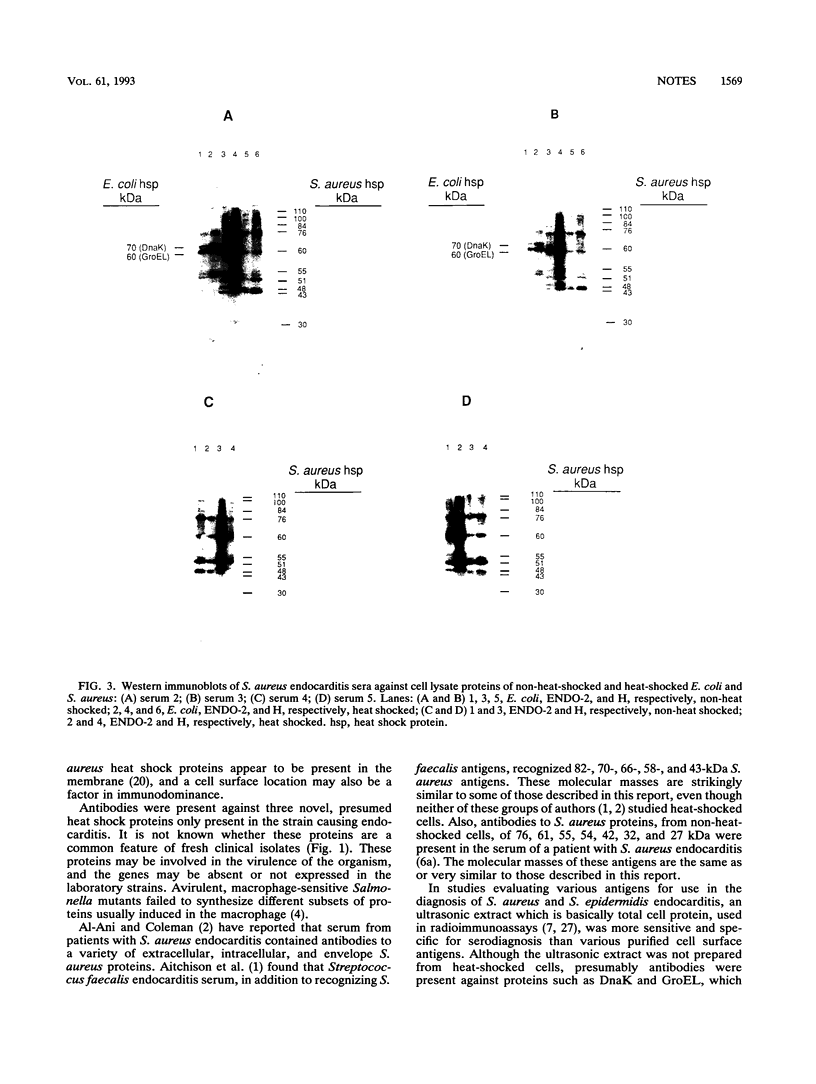
Antibodies to a range of Staphylococcus aureus and Escherichia coli heat shock proteins were present in sera from patients with S. aureus endocarditis. This suggests the highly immunoreactive nature of a range of heat shock proteins in addition to the GroEL equivalent (common antigen) protein. In one case, antibodies to three proteins unique to the infecting S. aureus strain, which were more prominent in heat-shocked cells, were also detected.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aitchison E. J., Lambert P. A., Smith E. G., Farrell I. D. Serodiagnosis of Streptococcus faecalis endocarditis by immunoblotting of surface protein antigens. J Clin Microbiol. 1987 Feb;25(2):211–215. doi: 10.1128/jcm.25.2.211-215.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allan B., Linseman M., MacDonald L. A., Lam J. S., Kropinski A. M. Heat shock response of Pseudomonas aeruginosa. J Bacteriol. 1988 Aug;170(8):3668–3674. doi: 10.1128/jb.170.8.3668-3674.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchmeier N. A., Heffron F. Induction of Salmonella stress proteins upon infection of macrophages. Science. 1990 May 11;248(4956):730–732. doi: 10.1126/science.1970672. [DOI] [PubMed] [Google Scholar]
- Cluss R. G., Boothby J. T. Thermoregulation of protein synthesis in Borrelia burgdorferi. Infect Immun. 1990 Apr;58(4):1038–1042. doi: 10.1128/iai.58.4.1038-1042.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danilition S. L., Maclean I. W., Peeling R., Winston S., Brunham R. C. The 75-kilodalton protein of Chlamydia trachomatis: a member of the heat shock protein 70 family? Infect Immun. 1990 Jan;58(1):189–196. doi: 10.1128/iai.58.1.189-196.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iandolo J. J. Genetic analysis of extracellular toxins of Staphylococcus aureus. Annu Rev Microbiol. 1989;43:375–402. doi: 10.1146/annurev.mi.43.100189.002111. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- NOVICK R. P. ANALYSIS BY TRANSDUCTION OF MUTATIONS AFFECTING PENICILLINASE FORMATION IN STAPHYLOCOCCUS AUREUS. J Gen Microbiol. 1963 Oct;33:121–136. doi: 10.1099/00221287-33-1-121. [DOI] [PubMed] [Google Scholar]
- Neidhardt F. C., VanBogelen R. A., Vaughn V. The genetics and regulation of heat-shock proteins. Annu Rev Genet. 1984;18:295–329. doi: 10.1146/annurev.ge.18.120184.001455. [DOI] [PubMed] [Google Scholar]
- Novick R. Properties of a cryptic high-frequency transducing phage in Staphylococcus aureus. Virology. 1967 Sep;33(1):155–166. doi: 10.1016/0042-6822(67)90105-5. [DOI] [PubMed] [Google Scholar]
- Peterson P. K., Wilkinson B. J., Kim Y., Schmeling D., Douglas S. D., Quie P. G., Verhoef J. The key role of peptidoglycan in the opsonization of Staphylococcus aureus. J Clin Invest. 1978 Mar;61(3):597–609. doi: 10.1172/JCI108971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plikaytis B. B., Carlone G. M., Pau C. P., Wilkinson H. W. Purified 60-kilodalton Legionella protein antigen with Legionella-specific and nonspecific epitopes. J Clin Microbiol. 1987 Nov;25(11):2080–2084. doi: 10.1128/jcm.25.11.2080-2084.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qoronfleh M. W., Streips U. N., Wilkinson B. J. Basic features of the staphylococcal heat shock response. Antonie Van Leeuwenhoek. 1990 Aug;58(2):79–86. doi: 10.1007/BF00422721. [DOI] [PubMed] [Google Scholar]
- Shinnick T. M. Heat shock proteins as antigens of bacterial and parasitic pathogens. Curr Top Microbiol Immunol. 1991;167:145–160. doi: 10.1007/978-3-642-75875-1_9. [DOI] [PubMed] [Google Scholar]
- Smith P. K., Krohn R. I., Hermanson G. T., Mallia A. K., Gartner F. H., Provenzano M. D., Fujimoto E. K., Goeke N. M., Olson B. J., Klenk D. C. Measurement of protein using bicinchoninic acid. Anal Biochem. 1985 Oct;150(1):76–85. doi: 10.1016/0003-2697(85)90442-7. [DOI] [PubMed] [Google Scholar]
- Thole J. E., Hindersson P., de Bruyn J., Cremers F., van der Zee J., de Cock H., Tommassen J., van Eden W., van Embden J. D. Antigenic relatedness of a strongly immunogenic 65 kDA mycobacterial protein antigen with a similarly sized ubiquitous bacterial common antigen. Microb Pathog. 1988 Jan;4(1):71–83. doi: 10.1016/0882-4010(88)90049-6. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verbrugh H. A., Peters R., Rozenberg-Arska M., Peterson P. K., Verhoef J. Antibodies to cell wall peptidoglycan of Staphylococcus aureus in patients with serious staphylococcal infections. J Infect Dis. 1981 Jul;144(1):1–9. doi: 10.1093/infdis/144.1.1. [DOI] [PubMed] [Google Scholar]
- Wheat J., Kohler R. B., White A., Garten M., Wilkinson B. J. IgM and IgG antibody response to teichoic acid in infections due to Staphylococcus aureus. J Infect Dis. 1983 Jun;147(6):1101–1101. doi: 10.1093/infdis/147.6.1101. [DOI] [PubMed] [Google Scholar]
- Wheat L. J., Kohler R. B., White A. Solid-phase radioimmunoassay for immunoglobulin G Staphylococcus aureus antibody in serious staphylococcal infection. Ann Intern Med. 1978 Oct;89(4):467–472. doi: 10.7326/0003-4819-89-4-467. [DOI] [PubMed] [Google Scholar]
- Wheat L. J., Wilkinson B. J., Kohler R. B., White A. C. Antibody response to peptidoglycan during staphylococcal infections. J Infect Dis. 1983 Jan;147(1):16–22. doi: 10.1093/infdis/147.1.16. [DOI] [PubMed] [Google Scholar]
- Wilson W. R., Geraci J. E. Antibiotic treatment of infective endocarditis. Annu Rev Med. 1983;34:413–427. doi: 10.1146/annurev.me.34.020183.002213. [DOI] [PubMed] [Google Scholar]
- Woods M. L., 2nd, Bonfiglioli R., McGee Z. A., Georgopoulos C. Synthesis of a select group of proteins by Neisseria gonorrhoeae in response to thermal stress. Infect Immun. 1990 Mar;58(3):719–725. doi: 10.1128/iai.58.3.719-725.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young D., Lathigra R., Hendrix R., Sweetser D., Young R. A. Stress proteins are immune targets in leprosy and tuberculosis. Proc Natl Acad Sci U S A. 1988 Jun;85(12):4267–4270. doi: 10.1073/pnas.85.12.4267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young R. A., Elliott T. J. Stress proteins, infection, and immune surveillance. Cell. 1989 Oct 6;59(1):5–8. doi: 10.1016/0092-8674(89)90861-1. [DOI] [PubMed] [Google Scholar]
- Young R. A. Stress proteins and immunology. Annu Rev Immunol. 1990;8:401–420. doi: 10.1146/annurev.iy.08.040190.002153. [DOI] [PubMed] [Google Scholar]