Abstract
One of the significant hurdles toward safe and efficacious systemic treatment of cancer with oncolytic adenoviruses (Ads) is dose-limiting hepatotoxicity that prevents the increase of a therapeutic dose. In this study, we expanded the therapeutic window of oncolytic serotype 5 Ad (Ad5) by a genetic modification of hypervariable loop 5 (HVR5) in the capsid protein hexon that prevented infection of hepatocytes due to ablation of binding to blood factors. This oncolytic virus, Ad-GL-HB, had significantly reduced levels of hepatocyte transduction in immunocompetent and immunodeficient mice as compared to parental virus Ad-GL. The hepatocyte detargeting decreased liver damage and increased the maximum tolerated dose of Ad-GL-HB tenfold relative to that of Ad-GL. Intravenous (i.v.) injection of Ad-GL or Ad-GL-HB into tumor-bearing mice produced equally increased survival rates demonstrating that while Ad-GL-HB detargeted hepatocytes, it sustained tumor cell infection after systemic administration. The significantly improved safety of the virus allowed it to be used at increased doses for improved systemic antitumor efficacy. Our results suggest that hexon modifications provide valuable strategies for systemic oncolytic Ad therapy.
Introduction
Oncolytic virotherapy is a novel class of anticancer treatment with unique mechanism of action based on natural ability of viruses to replicate in and lyse infected cells.1 The first engineered oncolytic adenovirus (Ad) was recently approved for intratumoral treatment of head-and-neck cancer patients.2 Systemic administration of oncolytic Ads could allow for the treatment of inaccessible tumors and metastases; however, higher doses of systemically delivered Ad were required to achieve similar levels of efficacy relative to the doses delivered intratumorally.3 It was suggested that initial virus concentration at the tumor site was essential for the antitumor efficacy.3 Ad type 5 (Ad5) displays natural tropism to the liver with >90% of intravenously (i.v.) injected Ad dose found in the liver during first 24 hours after the injection;4 this tropism reduces the therapeutic efficacy of the systemic treatment with oncolytic Ads. Liver macrophages (Kupffer cells) were shown to take up and rapidly destroy the majority of Ad particles present in the blood.5,6 As Ad clearance by Kupffer cells results in death of these cells,7,8 sequential systemic delivery of Ads allows for a depletion of Kupffer cells by the first dose (“predosing” effect) and increased anticancer efficacy of the second Ad dose.9 However, depletion of Kupffer cells by Ad predosing increases transduction of hepatocytes with subsequently delivered Ad.5,9 Infection of hepatocytes with Ad5 and Ad5-based replication-competent vectors leads to a dose-dependent hepatotoxicity10,11,12 representing a significant safety concern for clinical application of oncolytic Ads.
Natural tropism of Ad to hepatocytes was shown to be mediated by blood coagulation factors.13,14 We have previously demonstrated that decreasing plasma concentration of blood factors in mice by pretreatment with anticoagulant drug warfarin detargeted oncolytic Ad from the hepatocytes and reduced liver toxicity.9 Combination of Kupffer cell depletion with warfarin pretreatment significantly expanded therapeutic window of systemic treatment with oncolytic Ad.9
Higher doses of oncolytic Ad applied systemically were shown to deliver more virions to the tumors relative to the lower doses.3 Transaminitis observed in cancer patients given high dose (HD) of oncolytic Ad11,12 prevented further increase of the therapeutic dose underscoring the need to reduce virus-mediated hepatotoxicity. Although warfarin is a widely used anticoagulant drug, it has a very narrow therapeutic window in humans with unpredictable pharmacokinetic profile and bleeding as a most common side effect.15 Therefore, the strategies of replacing the need of using warfarin with systemically administered oncolytic Ads are required. Hexon was recently identified as a major Ad capsomere mediating hepatocyte transduction via binding to blood factor X (FX).16,17,18
Previous work has demonstrated that insertion of a large 75 amino acid biotin acceptor peptide (BAP) into hypervariable loop 5 (HVR5) of the hexon of replication-defective Ad5 reduced the affinity of FX for the virus 10,000-fold.17 This change in blood factor binding markedly reduced liver hepatocyte transduction by the virus.17 These data demonstrated that such a modification could block uptake by hepatocytes, but did not demonstrate that this “detargeting” effect had a functional benefit in a therapeutic setting. In this work, we demonstrate the practical utility of detargeting liver infection by Ad5 in the context of systemic oncolytic treatment of distant tumor sites after i.v. injection. We show that oncolytic Ad5 bearing the BAP modification has reduced liver hepatocyte infection, reduced liver damage, and an expanded therapeutic window that allows higher doses of virus to be used for systemic therapy.
Results
Construction and in vitro characterization of hexon-modified oncolytic Ad
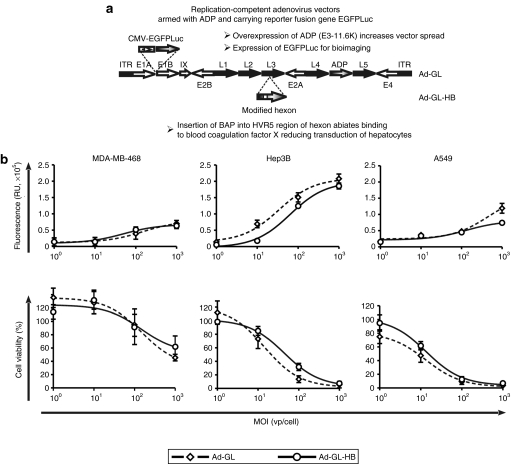
Ad capsid protein hexon was shown to bind FX and redirect the virus to hepatocytes.16,17 Modifications of hexon HVR5 of replication-defective Ad5 vectors reduced blood factor binding and liver gene transfer.17,18 To assess whether ablation of blood factor binding could reduce liver toxicity associated with local and systemic administration of oncolytic Ads, we modified previously described oncolytic Ad-GL9 by introducing BAP into the hexon HVR5 region to obtain Ad-GL-HB (Figure 1a). Ad-GL and Ad-GL-HB express the enhanced green fluorescent protein–firefly luciferase fusion protein (EGFPLuc) to allow bioluminescence imaging of the vector transduction and biodistribution profiles. Additionally, adenovirus death protein (E3-11.6K) is overexpressed by Ad-GL and Ad-GL-HB for increased spread of the viruses.19,20,21
Figure 1.
Genomes and anticancer activity of Ad-GL and Ad-GL-HB in vitro. (a) Open arrows, early Ad regions; closed arrows, late Ad regions; gray arrows, inserted or modified genes; closed boxes, ITRs. Ad-GL and Ad-GL-HB are replication-competent Ad vectors that overexpress ADP (E3-11.6K) for increased spread and express EGFP-firefly luciferase fusion protein for imaging virus transduction, spread, and persistence. Ad-GL-HB has BAP inserted into HVR5 loop of hexon to prevent binding to blood FX and reduce hepatocyte transduction. (b) MDA-MB-468, Hep3B, and A549 cells were infected with Ad-GL or Ad-GL-HB at the indicated multiplicities of infection (MOIs) (n = 4). Five days later EGFP expression (upper row) and cell viability (bottom row) were measured. Data are presented as mean ± SEM. Ad, adenovirus; ADP, adenovirus death protein; BAP, biotin acceptor peptide; EGFP, enhanced green fluorescent protein; FX, factor X; HVR5, hypervariable loop 5; ITR, inverted terminal repeat.
Infectivity and spread of Ad-GL and Ad-GL-HB were compared in vitro in three cancer cell lines infected with varying multiplicities of infection (MOIs) (Figure 1b). The data from all MOIs were analyzed by two-way analysis of variance (ANOVA) (factors: “MOI” and “virus”). Increased fluorescence and decreased cell viability were observed at higher MOIs for both viruses in all cell lines (P < 0.001). In MDA-MB-468 breast cancer cells, Ad-GL and Ad-GL-HB had similar levels of EGFP expression (P = 0.880) and cytotoxicity (P = 0.967) at all tested MOIs. EGFP expression was slightly but significantly higher in Hep3B hepatocellular carcinoma cells infected with Ad-GL compared to Ad-GL-HB (P < 0.001); however, this increase did not translate into higher cytotoxicity (P = 0.467). Infection of A549 lung cancer cells with Ad-GL produced higher EGFP expression levels relative to those of Ad-GL-HB (P = 0.005) that resulted in slightly higher cytotoxicity of Ad-GL in this cell line that, however, did not reach statistical significance (P = 0.056). These data demonstrated that modification of hexon did not significantly change the oncolytic properties of replicative Ad.
Ad-GL-HB is detargeted from hepatocytes but retains systemic anticancer activity in vivo
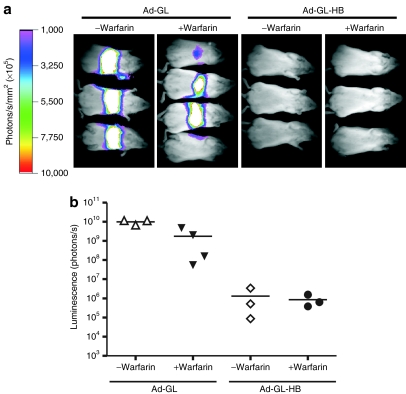
To characterize the effect of hexon modification on Ad infection profile in vivo, ICR mice were injected i.v. with 3 × 1010 virus particles (vp) of Ad-GL or Ad-GL-HB with or without pretreatment with warfarin to inhibit blood factor binding.9 Luciferase expression was imaged 24 hours after virus injections (Figure 2a) and was quantified (Figure 2b). All Ad-GL-injected mice demonstrated high luminescence signal in the livers that was significantly reduced by pretreatment with warfarin (P = 0.007). Injection of Ad-GL-HB produced significantly lower liver luminescence regardless of whether the mice were pretreated or not pretreated with warfarin (P = 0.003 for Ad-GL versus Ad-GL-HB, P = 0.295 for Ad-GL-HB versus Ad-GL-HB + warfarin). These results demonstrate that insertion of BAP into hexon HVR5 efficiently detargets oncolytic Ad from infecting hepatocytes and substitutes for the use of warfarin.
Figure 2.
Modification of the hexon reduces infection of hepatocytes with oncolytic Ad. (a) ICR mice were injected i.v. with 3 × 1010 vp of Ad-GL or Ad-GL-HB. Some mice were pretreated with warfarin. Luciferase expression in the liver was detected by whole-body imaging on day 1 after virus injection. (b) Quantification of the luminescence signal from a. Ad, adenovirus; ICR, Imperial Cancer Research; i.v., intravenous; vp, virus particles.
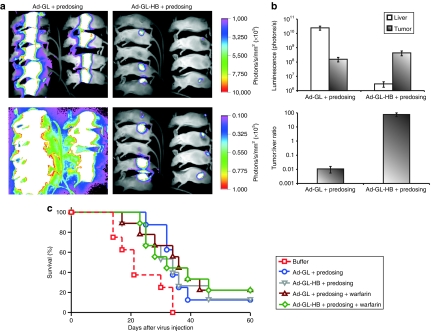
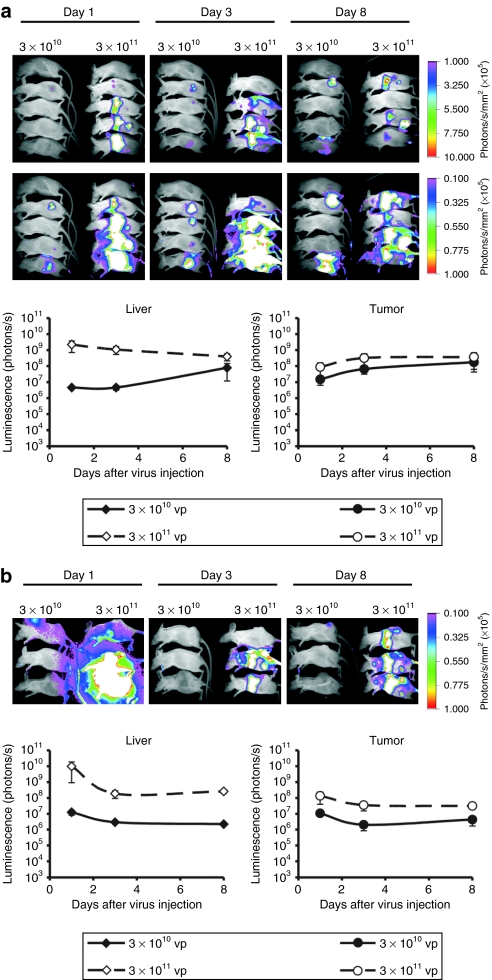
Next, we studied the anticancer activity of Ad-GL-HB after systemic administration into mice bearing subcutaneous (s.c.) hepatocellular carcinoma Hep3B tumors. Previous work showed that depleting liver Kupffer cells by predosing with an irrelevant Ad was crucial for achieving significant systemic anticancer efficacy with oncolytic Ad after i.v. injection in this tumor model.9 Mice were predosed and then injected once with 3 × 1010 vp of Ad-GL or Ad-GL-HB with or without pretreatment with warfarin. Whole-body imaging was performed on day 1 after virus injection (Figure 3a) and luminescence signals from livers and tumors were quantified (Figure 3b). Ad-GL-HB-injected mice had 8,800-fold lower level of luciferase expression in the liver as compared to the level in Ad-GL-injected mice (P = 0.006). The luminescence signal from the tumors was similar in both groups (P = 0.125) (Figure 3b, top). These results demonstrated that although Ad-GL-HB was detargeted from hepatocytes, it maintained its ability to infect distant tumors after systemic administration. Reduced transduction of hepatocytes by Ad-GL-HB resulted in 7,000-fold increased tumor:liver infection ratio relative to that in Ad-GL-injected mice (P = 0.004) (Figure 3b, bottom). Mice in all virus-injected groups had significantly increased survival rates as compared with the buffer-treated animals (P < 0.05) (Figure 3c). Median survival time for the groups was as follows: buffer, 21 days; Ad-GL+predosing, 34 days; Ad-GL-HB+predosing, 34 days; Ad-GL+predosing+warfarin, 36 days; Ad-GL-HB+predosing+warfarin, 32 days. There were no significant differences between any virus-injected groups (P > 0.05) demonstrating that hexon modification did not attenuate the in vivo oncolytic activity of Ad.
Figure 3.
Hexon modification does not attenuate Ad oncolytic activity in vivo. (a) Nude mice bearing s.c. Hep3B tumors (23 days after cell injection, average tumor volume 264 mm3) were predosed i.v. with 3 × 1010 vp of replication-defective Ad-DsRed-RD followed 4 hours later by i.v. injection of 3 × 1010 vp of Ad-GL or Ad-GL-HB. Luciferase expression was detected by whole-body imaging on day 1 after virus injection. High (upper row) and low (bottom row) scales are shown. (b) Luminescence signals from liver and tumor areas in mice from a were quantified (top) and tumor:liver ratios were calculated (bottom). (c) Kaplan–Meyer survival curves. Mice with established Hep3B tumors as described in a were treated with buffer (phosphate-buffered saline), 3 × 1010 vp of Ad-GL or Ad-GL-HB with predosing with 3 × 1010 vp of Ad-DsRed-RD with or without warfarin pretreatment. Log rank test, buffer (n = 8) versus Ad-GL + predosing (n = 9) P = 0.0155; versus Ad-GL + predosing + warfarin (n = 9) P = 0.0129; versus Ad-GL-HB + predosing (n = 9) P = 0.0436; versus Ad-GL-HB + predosing + warfarin (n = 9) P = 0.0335. Ad, adenovirus; i.v., intravenous; s.c., subcutaneous; vp, virus particles.
Hexon modification significantly reduces hepatotoxicity of oncolytic Ad
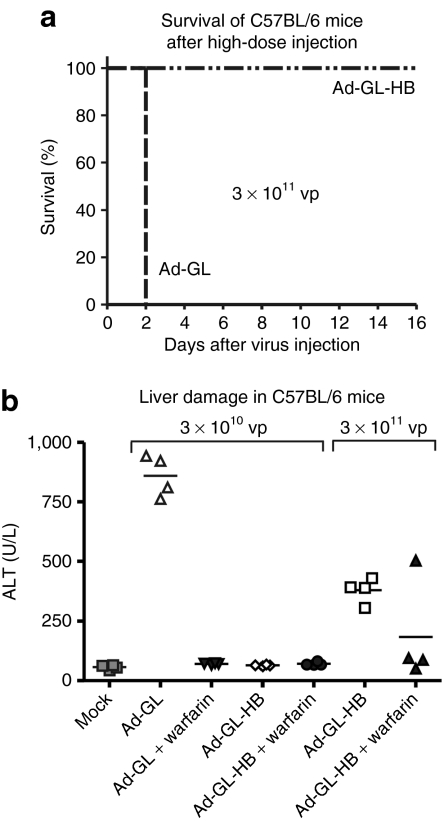
To assess the effect of hexon modification on liver toxicity mediated by oncolytic Ad after systemic administration, we injected C57BL/6 mice with 3 × 1010 or 3 × 1011 vp of Ad-GL or Ad-GL-HB. Warfarin pretreatment was used to evaluate its protective effect at these virus doses. All mice injected with 3 × 1010 vp dose survived the experiment in agreement with previous results.9 However, all mice injected with 3 × 1011 vp of Ad-GL died on day 2 after injection regardless of whether they were pretreated (data not shown) or not pretreated with warfarin (Figure 4a). Ad-GL-HB at the same HD, 3 × 1011 vp, was well tolerated and all animals survived until day 16 when the experiment was terminated (Figure 4a). Liver damage in the survived mice was assessed by measuring serum levels of liver enzyme alanine aminotransferase (ALT) at day 3 after virus injection (Figure 4b) and was analyzed by one-way ANOVA followed by Tukey's honestly significant difference post hoc test. Ad-GL at low dose (LD) produced increased ALT levels (P < 0.001 versus buffer) which were reduced by pretreatment with warfarin (P < 0.001). ALT levels in LD Ad-GL-HB-injected mice with or without warfarin pretreatment were not different from those in buffer-treated mice (P = 1.000). ALT levels in HD Ad-GL-HB-injected mice were increased relative to those in buffer-treated mice (P = 0.001), but were still significantly lower relative to those in LD Ad-GL-treated mice (P < 0.001). Pretreatment with warfarin further reduced ALT levels in HD Ad-GL-HB-injected mice (P = 0.068; P = 0.442 versus buffer) suggesting that either insertion of BAP does not completely prevent binding of blood factors to hexon, or additional sites on Ad capsid mediate blood factor–dependent hepatocyte transduction.
Figure 4.
Increased safety of Ad-GL-HB. (a) Survival of mice injected i.v. with high virus dose. C57BL/6 mice were injected i.v. with 3 × 1011 vp of Ad-GL or Ad-GL-HB (n = 4) on day 0. Mice were monitored till day 16. (b) Hepatotoxicity of Ad-GL and Ad-GL-HB after systemic administration. Blood was collected from survived mice from a and from additional C57BL/6 mice injected with 3 × 1010 vp of Ad-GL, pretreated or not pretreated with warfarin (n = 4), on day 3 after injection and was analyzed for serum ALT levels as a marker of liver damage. Ad, adenovirus; ALT, alanine aminotransferase; i.v., intravenous; vp, virus particles.
Infection of hepatocytes and tumor cells after HD i.v. injection of Ad-GL-HB
Higher doses were shown to deliver more Ad virions to the tumor cells after systemic injection as compared with lower doses.3 As hexon modification increased safety of oncolytic Ad and permitted the use of higher doses, we compared tumor and liver transduction levels after injecting i.v. 3 × 1010 or 3 × 1011 vp of Ad-GL-HB in two tumor models, hepatocellular carcinoma Hep3B and prostate carcinoma DU145 (Figure 5). Kupffer cells were depleted by injecting 3 × 1010 vp of Ad-DsRed-RD 4 hours before injecting Ad-GL-HB. The data from all time points were analyzed with repeated measures ANOVA. Injection of 3 × 1011 vp of Ad-GL-HB significantly increased hepatocyte transduction in Hep3B tumor-bearing mice (Figure 5a) relative to tenfold lower dose (P = 0.049), whereas the increase in liver transduction in DU145 tumor-bearing mice (Figure 5b) was not statistically significant (P = 0.320). We have not found significant differences between the tumor transduction levels produced by i.v. injection of 3 × 1010 or 3 × 1011 vp of Ad-GL-HB (P = 0.400 for Hep3B tumors, P = 0.144 for DU145 tumors); however, in both tumor models, we observed a trend toward increased tumor luminescence levels at higher virus dose (Figure 5).
Figure 5.
Infection of hepatocytes and tumor cells after low- and high-dose i.v. injections of Ad-GL-HB. (a) Mice with established s.c. Hep3B tumors (55 days after cell injection; average tumor volume 450 mm3) were predosed i.v. with 3 × 1010 vp of Ad-DsRed-RD followed 4 hours later by i.v. injection of 3 × 1010 or 3 × 1011 vp of Ad-GL-HB (n = 5). Luciferase expression was detected by whole-body imaging on days 1, 3, and 8 after virus injection. High (upper row) and low (bottom row) scales are shown (top). Luminescence signals from liver and tumor areas were quantified and plotted (bottom). Data are presented as mean ± SEM. (b) Mice with established s.c. DU145 tumors (34 days after cell injection, average tumor volume 736 mm3) were injected as in a (n = 3) and imaged on days 1, 3, and 8 after virus injection. Luminescence signals from liver and tumor areas were quantified and plotted. Data are presented as mean ± SEM. Ad, adenovirus; i.v., intravenous; s.c., subcutaneous; vp, virus particles.
Increased safety of Ad-GL-HB allows the use of higher doses during systemic anticancer treatment
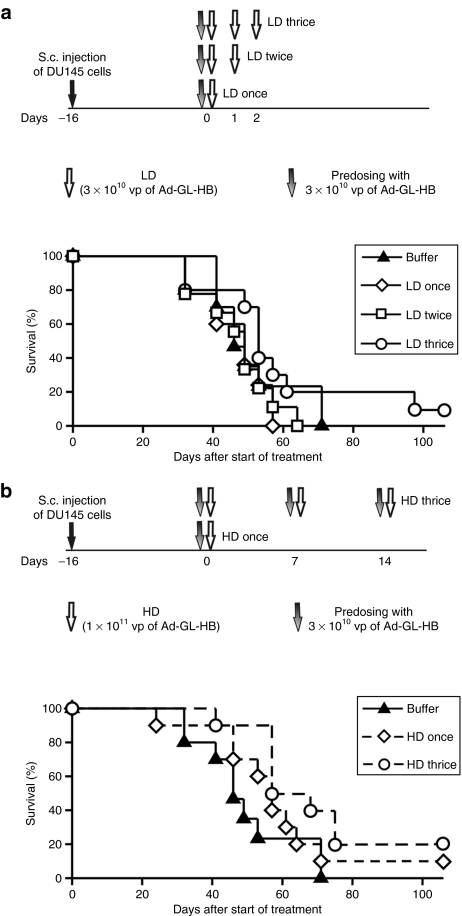
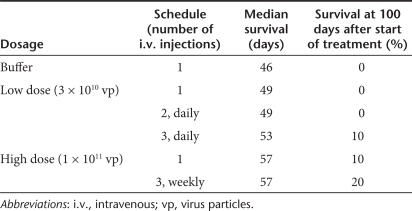
To examine the potential beneficial effect of higher Ad dose on therapeutic efficacy, we studied the anticancer activity of multiple i.v. doses of Ad-GL-HB at varying administration schedules in the DU145 prostate tumor model. Mice with established s.c. DU145 tumors were injected i.v. with one, two, or three doses of 3 × 1010 vp of Ad-GL-HB daily (LD groups) (Figure 6a). Kupffer cells were depleted by single predosing with 3 × 1010 vp of Ad-GL-HB 4 hours before injecting therapeutic dose. Predosing by replication-competent Ad-GL-HB was as effective as predosing with replication-defective Ad-DsRed-RD (Supplementary Figure S1) offering a more practical way of using one virus for a treatment course. In contrast to Hep3B tumor model (Figure 3), one dose of 3 × 1010 vp of Ad-GL-HB did not prolong survival time of DU145 tumor-bearing mice (P = 0.716 versus buffer; median survival time 49 days versus 46 days, respectively) (Figure 6a). Additional LD injections also did not increase survival rates (P > 0.260 for LD twice and LD thrice versus buffer; median survival time 49 and 53 days). However, 10% of the animals in the three LD group survived >100 days after start of treatment suggesting that increased numbers of i.v. doses had some benefit (Table 1). These data demonstrate that the DU145 model is more stringent for systemic virus delivery, as LDs of oncolytic Ad are relatively ineffective in this model after i.v. injection.
Figure 6.
Multiple high-dose injections of Ad-GL-HB demonstrate improved systemic anticancer activity. (a) Mice with established s.c. DU145 tumors (16 days after cell injection; average tumor volume 337 mm3) were predosed i.v. once with 3 × 1010 vp of Ad-GL-HB on day 0 followed by one, two, or three daily i.v. injections of 3 × 1010 vp of Ad-GL-HB (n = 9 or 10). Mice were euthanized when tumor volume reached 2,000 mm3. Kaplan–Meyer survival curves were plotted. (b) Additional mice with s.c. DU145 tumors established as described in a were injected i.v. with 1 × 1011 vp of Ad-GL-HB one or three times weekly with prior predosing with 3 × 1010 vp of Ad-GL-HB. Mice were euthanized when tumor volume reached 2,000 mm3. Kaplan–Meyer survival curves were plotted. Survival data for the same group of buffer-treated animals is shown in a and b. HD, high dose; i.v., intravenous; LD, low dose; s.c., subcutaneous; vp, virus particles.
Table 1.
Survival data after systemic administration of Ad-GL-HB in DU145 tumor model
Another set of mice was injected i.v. with one or three (once a week) HDs of 1 × 1011 vp of Ad-GL-HB (Figure 6b). As liver macrophages begin to repopulate the liver on day 5 after Kupffer cell depletion,22 we predosed the weekly injected animals with 3 × 1010 vp of Ad-GL-HB 4 hours before each therapeutic dose injection. Single HD i.v. injection of 1 × 1011 vp of Ad-GL-HB prolonged median survival time to 57 days (Figure 6b) but this was not statistically significant (P = 0.360 versus buffer). In contrast, the three weekly HD i.v. injections significantly increased survival rates (P = 0.023 versus buffer, P = 0.005 versus LD once and P = 0.004 versus LD twice). In single HD group, 10% of the animals survived through 100 days and 20% of the animals in three HDs group survived to this time point (Table 1). Together, these results suggest that hexon modification allows increasing therapeutic doses of oncolytic Ad leading to improved anticancer efficacy of systemic treatment.
Effect of predosing and multiple doses on toxicity profile of Ad-GL-HB after systemic administration
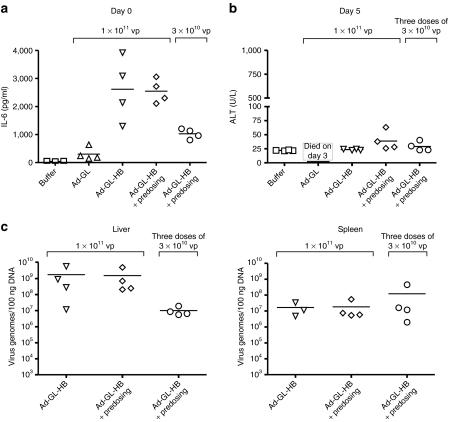
Predosing and administration of multiple doses can theoretically increase toxicity of oncolytic Ad. We studied effects of these treatment schedules and the presence of hexon modification on hepatotoxicity (as measured by serum ALT levels) and inflammatory response [as measured by serum interleukin-6 (IL-6) levels] of oncolytic Ad in C57BL/6 mice. Mice were injected with phosphate-buffered saline (PBS), 1 × 1011 vp of Ad-GL or Ad-GL-HB, or 3 × 1010 vp of Ad-GL-HB. Mice in one of the groups injected with 1 × 1011 vp of Ad-GL-HB and the mice injected with 3 × 1010 vp of Ad-GL-HB were predosed 4 hours earlier with 3 × 1010 vp of Ad-GL-HB. Serum IL-6 levels were measured 3 hours after virus injections (Figure 7a). Statistical analysis of the data by one-way ANOVA detected significant difference between the groups (P < 0.001). Pair-wise comparisons between the groups were performed with Tukey's honestly significant difference post hoc test. Injection of Ad-GL did not significantly increase IL-6 levels relative to buffer injection (P = 0.975). In contrast, Ad-GL-HB at 1 × 1011 vp dose both with and without predosing significantly increased IL-6 levels relative to buffer (P < 0.001), Ad-GL (P < 0.002), and Ad-GL-HB at lower (3 × 1010 vp) dose (P < 0.018). Predosing did not have significant effect on IL-6 levels in mice injected with 1 × 1011 vp of Ad-GL-HB (P = 1.000). IL-6 levels in LD Ad-GL-HB-injected mice were increased in comparison with those in buffer and Ad-GL-injected mice, but the increase was not statistically significant (P = 0.224 versus buffer, P = 0.442 versus Ad-GL).
Figure 7.
Effect of hexon modification, predosing, and multiple virus injections on IL-6 and ALT levels. (a) C57BL/6 mice were injected i.v. via tail vein with indicated doses of Ad-GL or Ad-GL-HB on day 0 (n = 4). For predosing, mice were injected with 3 × 1010 vp of Ad-GL-HB 4 hours before dosing. Three hours after the dosing blood was collected and serum levels of IL-6 were measured. (b) Mice from a injected with 3 × 1010 vp of Ad-GL-HB received additional two i.v. doses of 3 × 1010 vp of Ad-GL-HB on days 1 and 2. Three days after the last virus injection (day 5 after first injection) blood was collected and serum ALT levels were measured. Mice injected with Ad-GL died on day 3 after the injection. (c) Mice from b were euthanized after blood collection (day 5 after first virus injection) and livers and spleens were collected. Virus genome levels were quantified in the organs by real-time PCR. ALT, alanine aminotransferase; IL, interleukin; i.v., intravenous; vp, virus particles.
To assess whether multiple injections of Ad-GL-HB increase liver toxicity, predosed mice injected with 3 × 1010 vp of Ad-GL-HB on day 0 received additional two injections of 3 × 1010 vp of Ad-GL-HB on days 1 and 2. Three days after the last injection (day 5 after first injection) hepatotoxicity was assessed by measuring serum ALT levels (Figure 7b). All mice injected with Ad-GL died on day 3 after the injection. ALT levels in all Ad-GL-HB injected groups were not significantly different from buffer (P > 0.114) and from one another (P > 0.161). These data suggest that high and multiple doses of Ad-GL-HB can be applied systemically without significant hepatotoxicity.
Although, Ad-GL-HB did not induce hepatotoxicity at the studied doses, we found high levels of the virus genomes in the livers of the injected mice on day 5 after the first injection as detected by the real-time PCR (Figure 7c). Mice injected with HD of Ad-GL-HB had lower levels of the virus DNA in the spleen relative to those in the liver (Figure 7c). We did not find significant differences between the levels of virus genomes in the spleens (one-way ANOVA, P = 0.514) (Figure 7c). Interestingly, we detected less virus genomes in the livers of the mice injected with three doses of 3 × 1010 vp of Ad-GL in comparison with the mice injected with 1 × 1011 vp dose with or without predosing (Figure 7c); however this difference was not statistically significant (one-way ANOVA, P = 0.462). Together, these data suggest that detargeting from the hepatocytes (by ablating blood factor binding) and Kupffer cells (by predosing or HD injection) does not prevent Ad sequestration by the liver. Simultaneous increase in IL-6 production and the absence of hepatotoxicity suggest retargeting of the virus to other cells in the liver.
Discussion
Intravascular administration of oncolytic Ads has been tested in clinical trials and have not shown significant antitumor efficacy.12 It was estimated that higher doses of systemically delivered Ads would be required for delivering sufficient numbers of viral particles to the tumor site(s) relative to the local virus administration.3 We have previously demonstrated that depletion of hepatic Kupffer cells and detargeting from hepatocytes by pretreatment with warfarin significantly increased therapeutic efficacy and safety of systemically delivered oncolytic Ad.9 In this report, we studied the feasibility of increasing systemic therapeutic doses of oncolytic Ad and replacing warfarin pretreatment with genetically engineered hepatocyte-detargeted feature to expand therapeutic window of the treatment.
We constructed replication-competent Ad-GL-HB that has a peptide BAP inserted into hexon HVR5 that was previously shown to ablate binding to blood FX and to detarget replication-defective Ad from the hepatocytes.17 Ad-GL-HB demonstrated oncolytic activity levels similar to those of the parental virus Ad-GL in vitro, in cell culture experiments, and in vivo, in Hep3B tumor-bearing mice after single i.v. injection. Hexon modification increased tumor:liver infection ratio 7,000-fold relative to that of Ad-GL due to dramatically reduced transduction of hepatocytes. Dose-dependent hepatotoxicity of oncolytic Ads due to viral replication was demonstrated in preclinical animal models10,23,24,25,26 and in clinical trials.11,27 Detargeting from hepatocytes significantly reduced liver toxicity of Ad-GL-HB relative to Ad-GL in C57BL/6 mice and allowed administering tenfold higher doses systemically without causing lethality that was observed with the same dose of Ad-GL. These results demonstrated that hexon-modified oncolytic Ad had significantly improved safety profile relative to the virus with wild-type hexon.
At higher doses, Ad-GL-HB demonstrated increased transduction of hepatocytes as compared with lower dose. The increased level of transduction with Ad-GL-HB was still lower relative to that of Ad-GL applied at tenfold lower dose. It was suggested that FX-mediated Ad5 transduction occurs through heparan sulfate proteoglycans.14 It is possible that administration of HD of Ad-GL-HB compensates for the absence of FX binding and allows for transduction via coxsackie and Ad receptor. Additionally, blood factors FVII, FIX, and protein C were also implicated in hepatocyte transduction with Ad5.13,14 These factors might contribute significantly to transduction when HDs of Ad-GL-HB are applied.
HD of Ad-GL-HB increased tumor transduction in two s.c. tumor models after systemic administration, although the increase did not reach statistical significance at the time points that were assessed. Multiple HD injections of Ad-GL-HB significantly improved survival of DU145 tumor-bearing mice, whereas LDs and single HD were ineffective in this model. As oncolytic Ad-GL cannot be used at HD due to severe hepatotoxicity, the results of this study demonstrate that hexon modification represents a valuable strategy for achieving increased therapeutic efficacy of systemic treatment with oncolytic Ads. However, the HD treatment with Ad-GL-HB resulted in only 10–20% survival rates. The low efficacy of the treatment might be explained by insufficient tumor transduction levels achieved after HD injection of Ad-GL-HB (Figure 5). Detargeting from the hepatocytes and Kupffer cells did not prevent sequestration of Ad-GL-HB by the liver as high levels of the virus genomes were detected by real-time PCR in the livers of C57BL/6 mice injected with Ad-GL-HB. Ad localization to liver sinusoidal endothelial cells after i.v. virus injection in Kupffer cell depleted mice was previously demonstrated.28 This distribution was dependent on Ad capsid RGD motif interaction with cellular integrins. Interaction with liver endothelial cells was recently reported as the mechanism of Ad sequestration by the mouse liver when Kupffer cell uptake and hepatocyte transduction are ablated or saturated.29 High i.v. dose of replication-defective Ad vector was associated with transduction and severe systemic injury to the vascular endothelium in baboons.30 In agreement with these reports, we observed significant decrease in vascular density in Hep3B tumors 3 days after i.v. injection of Ad-GL-HB (data not shown) suggesting that endothelial cells in the tumors might also sequester significant quantity of the therapeutic virus. Further studies are needed to delineate whether ablation of RGD-integrin binding might increase the dose of oncolytic Ad delivered to tumor after i.v. injection and achieve increased therapeutic efficacy.
HD injection of Ad-GL-HB induced production of IL-6 that was detected in the serum 3 hours after virus injection. In contrast, mice injected with the same dose of Ad-GL had IL-6 levels not different from those detected in buffer-treated mice. These data also suggest that detargeting from the hepatocytes retargets Ad to other cell type(s). Increased production of IL-6 was demonstrated in baboons that were injected i.v. with HD of replication-defective Ad vector and developed a severe injury to the vascular endothelium.30 We have recently reported that covalent coating of oncolytic Ad with high-molecular weight polyethylene glycol (PEG) efficiently detargeted the virus from hepatocytes and Kupffer cells.31 Interestingly, PEGylated Ad also induced production of IL-6 after systemic administration whereas unPEGylated Ad did not (unpublished observation). Cumulatively, these results support the observation that detargeting Ad from hepatocytes (by capsid modification or warfarin) and Kupffer cells (by predosing or using HDs) retargets Ad to the endothelial cells.29 This mechanism appears to be dose-dependent as HD of Ad-GL-HB induced higher IL-6 production relative to lower dose of the virus.
Increased production of IL-6 also suggests induction of inflammatory immune response by the Ad capsid.32,33 Development of inflammation represents a significant safety concern for the application of replication-defective Ad vectors. Interestingly, in our experiment, Ad-GL did not induce IL-6 production; however all injected mice died 3 days after the virus injection in accordance with the kinetics of virus replication in the hepatocytes.23 In contrast, Ad-GL-HB-injected mice appeared healthy for the duration of the experiment despite the increased serum IL-6 levels. Our results suggest that hepatotoxicity represents more significant concern for the replicative oncolytic Ads as compared to Ad-induced inflammation. Ad early region proteins present in the genomes of replication-competent Ads counteract inflammatory immune responses.34,35 Wild-type E1A was shown to downregulate IL-6 expression.36 Further studies are required to examine the kinetics and dose-dependence of inflammatory immune responses induced by replication-competent and replication-defective Ads carrying wild-type and modified hexons to define the safety of HD treatment with oncolytic Ads.
Intravascular administration of replication-selective oncolytic Ads have been evaluated in clinical trials.12 Single injection of 2 × 1013 vp of E1B-55kd gene deleted Ad (ONYX-015) was found to have acceptable safety. In another trial, dose escalation of prostate replication-selective Ad was halted at 6 × 1012 vp due to asymptomatic grade 1 to 2 transaminitis and/or isolated D-dimer elevations.11 It remains to be investigated whether additional detargeting approaches could allow using higher doses of cancer-selective oncolytic Ads in patients and provide increased anticancer efficacy. Modification of hexon provides transductional detargeting of Ad from hepatocytes. Alternate approaches involve transcriptional detargeting of Ad by preventing virus replication in normal cells37,38,39 or by retargeting viruses with new ligands.1,40,41 These approaches are complementary and it is likely that their combination might produce additive or synergistic improvement of oncolytic Ad efficacy/toxicity profile. Arming oncolytic Ads with therapeutic transgenes was shown to increase their anticancer activity.10,42,43,44,45,46,47 Combination of therapeutic transgene expression with genetic and/or chemical modification of Ad capsid allowing for use of high systemic doses might further increase anticancer activity of oncolytic Ads. These approaches will likely not only be important for systemic therapy, but also for intratumoral injections, as virus has been shown to leak into the blood and cause liver damage in animal models and in human patients.25,48,49
In summary, we have constructed and characterized in vitro and in vivo hexon-modified oncolytic Ad. To our knowledge, this is the first demonstration of applying a genetically hepatocyte-detargeted capsid-modified Ad vector in a therapeutic application. Our data indicate that modification of hexon allows for increased systemic anticancer efficacy and decreased hepatotoxicity of oncolytic Ad resulting in expanded therapeutic window of the treatment.
Materials and Methods
Cell lines. Human cancer cell lines MDA-MB-468 (breast carcinoma), Hep3B (hepatocellular carcinoma), DU145 (prostate carcinoma), and A549 (lung adenocarcinoma) were obtained from American Type Culture Collection (ATCC; Manassas, VA). Human embryonic kidney 293 cells and suspension N3S cells were obtained from Microbix (Toronto, Ontario, Canada). All cell lines were maintained in RPMI 1640 medium supplemented with 10% fetal bovine serum (HyClone, Logan, UT).
Ads. Ad-DsRed-RD is E1− and E3− replication-defective Ad5 that expresses reporter gene for DsRed under the control of human cytomegalovirus immediate early promoter. Ad-GL, previously described as Ad-EGFPLuc,9 is a derivative of Ad5 that overexpresses the adenovirus death protein (E3-11.6K) for improved virus spread.20,21 Adenovirus death protein in Ad-GL is located in its natural position in E3 region. Overexpression of adenovirus death protein is a compensatory splicing effect due to deletions in E3A and E3B regions.20 Ad-GL expresses EGFP-firefly luciferase fusion gene (EGFPLuc) under the control of human cytomegalovirus immediate early promoter early and late in infection cycle. The CMV-EGFPLuc cassette is inserted into HpaI site in the E1 region downstream of E1A open reading frames and upstream of E1A polyadenylation signal.9 Ad-GL-HB is a derivative of Ad-GL that has 75 amino acid BAP inserted into HVR5 loop of hexon. To obtain Ad-GL-HB, genomic DNA of Ad-GL was digested with SpeI and PmeI and cotransfected with shuttle pAdHexBAP plasmid50 into 293 cells. Resulting virus was plaque-purified in A549 cells and genomic structure was confirmed by sequencing. Viruses were purified by double CsCl banding. Vp concentration was determined by A260 measurements. Infectious virus titers of Ad-GL and Ad-GL-HB were determined by the limiting dilution assay using A549 cells. Particle to infectious particle ratios for both viruses were <10.
In vitro vector characterization. MDA-MB-468, Hep3B, and A549 cells were infected with Ad-GL or Ad-GL-HB at various MOIs (n = 4). Five days later EGFP expression was measured using Beckman Coulter Microplate Reader DTX800 Multimode Detector (Beckman Coulter, Fullerton, CA); cell viability was quantified using MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide) (Sigma-Aldrich, St Louis, MO) assay.
Animals. All animal studies have been approved by Mayo Clinic Institutional Animal Care and Use Committee. Four- to six-week-old ICR mice were purchased from National Cancer Institute, Rockville, MD; nude mice were purchased from Harlan Sprague Dawley, Indianapolis, IN; C57BL/6 mice were purchased from Jackson Laboratory, Bar Harbor, ME.
Ad predosing and pretreatment with warfarin. To deplete liver macrophages, mice were injected i.v. via tail vein with 3 × 1010 vp of Ad-DsRed-RD or Ad-GL-HB in 100 µl of PBS 4 hours before oncolytic Ad injection. Pretreatment with warfarin was done as follows: mice were injected s.c. with 133 µg of warfarin (Sigma-Aldrich) in 100 µl of peanut oil 3 and 1 days before Ad injection.
Bioluminescence imaging. Animals were anesthetized with ketamine and xylazine and injected intraperitoneally with 100 µl of D-luciferin (20 mg/ml) (Molecular Imaging Products, Bend, OR). Mice were imaged on Kodak In-Vivo F system (Carestream Health, Rochester, NY) for 5 minutes. Images were processed and analyzed using Kodak Imaging software (Carestream Health).
Toxicity studies. C57BL/6 mice were injected i.v. via the tail vein with varying doses of Ad-GL or Ad-GL-HB on day 0. Blood was collected at day 3 or day 5 and analyzed for serum levels of ALT by colorimetric endpoint reaction method according to manufacturer's instructions (Biotron Diagnostics, Hemet, CA). Serum IL-6 levels were measured by enzyme-linked immunosorbent assay (BD Biosciences, San Diego, CA) using blood samples collected 3 hours after virus injection.
Real-time PCR. C57BL/6 mice injected with various doses of Ad-GL-HB were euthanized on day 5 after first virus injection and livers and spleens were collected. DNA from the tissue samples was extracted using Qiagen DNeasy Blood and Tissue kit (Qiagen, Valencia, CA). Real-time PCR was performed with QuantiTest SYBR green PCR (Qiagen) master mix with primers specific for firefly luciferase gene as described previously.9,31
Animal tumor studies. Hep3B tumors were established s.c. in the hind flank of nude mice by injecting 1 × 106 Hep3B cells in 100 µl of medium containing 50% Matrigel (BD Biosciences, San Jose, CA). Twenty-three days after cell injection mice with Hep3B tumors were randomized based on tumor volumes and assigned into groups (average tumor volume 264 mm3; n = 8 or 9). The animals received single i.v. injection via the tail vein of PBS, 3 × 1010 vp of Ad-GL or Ad-GL-HB in 100 µl of PBS. Some animals were pretreated with warfarin and/or predosed with 3 × 1010 vp of Ad-DsRed-RD. DU145 tumors were established s.c. in the hind flank of nude mice by injecting 1 × 106 DU145 cells in 100 µl of medium containing 50% Matrigel (BD Biosciences). Treatment began 16 days later when average tumor volume reached 337 mm3. Mice (n = 9 or 10 per group) were injected i.v. via tail vein with PBS, 3 × 1010 or 1 × 1011 vp of Ad-GL-HB in 100 µl volume. Predosing was done 4 hours earlier with 3 × 1010 vp of Ad-GL-HB. Injections were repeated daily or weekly up to three times. Tumor dimensions were measured twice a week with calipers and tumor volumes were calculated as width2 × length × 1/2. Mice were euthanized when the tumor volume reached 2,000 mm3.
For comparison of Ad-GL-HB transduction after LD and HD injections, s.c. Hep3B tumor-bearing mice (day 55 after cell injection; average tumor volume 450 mm3) and s.c. DU145 tumor-bearing mice (day 34 after cell injection, average tumor volume 736 mm3) were predosed i.v. with 3 × 1010 vp of Ad-DsRed-RD followed 4 hours later by i.v. injection of 3 × 1010 or 3 × 1011 vp of Ad-GL-HB (n = 5 for Hep3B tumor model, n = 3 for DU145 tumor model). Mice were imaged for luciferase expression on days 1, 3, and 8 after virus injection.
Statistical analyses. Data are presented as mean ± SEM. Statistical analyses were performed using SPSS software (SPSS, Chicago, IL). The statistical significance was assessed using General Linear Model followed by Tukey's honestly significant difference test for pair-wise comparisons between groups. Survival data were analyzed with the log-rank test. P < 0.05 was considered significant.
SUPPLEMENTARY MATERIALFigure S1. Predosing with Ad-GL-HB increases hepatocyte transduction with consequently delivered Ad as efficiently as predosing with Ad-DsRed-RD.
Supplementary Material
Predosing with Ad-GL-HB increases hepatocyte transduction with consequently delivered Ad as efficiently as predosing with Ad-DsRed-RD.
Acknowledgments
We thank Mary Barry and DeAnn Frederixon for helpful technical assistance. This work was supported by the NIH P50 CA91956 Prostate Cancer SPORE grant to Mayo Clinic.
REFERENCES
- Cattaneo R, Miest T, Shashkova EV., and , Barry MA. Reprogrammed viruses as cancer therapeutics: targeted, armed and shielded. Nat Rev Microbiol. 2008;6:529–540. doi: 10.1038/nrmicro1927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garber K. China approves world's first oncolytic virus therapy for cancer treatment. J Natl Cancer Inst. 2006;98:298–300. doi: 10.1093/jnci/djj111. [DOI] [PubMed] [Google Scholar]
- Demers GW, Johnson DE, Tsai V, Wen SF, Quijano E, Machemer T, et al. Pharmacologic indicators of antitumor efficacy for oncolytic virotherapy. Cancer Res. 2003;63:4003–4008. [PubMed] [Google Scholar]
- Worgall S, Wolff G, Falck-Pedersen E., and , Crystal RG. Innate immune mechanisms dominate elimination of adenoviral vectors following in vivo administration. Hum Gene Ther. 1997;8:37–44. doi: 10.1089/hum.1997.8.1-37. [DOI] [PubMed] [Google Scholar]
- Tao N, Gao GP, Parr M, Johnston J, Baradet T, Wilson JM, et al. Sequestration of adenoviral vector by Kupffer cells leads to a nonlinear dose response of transduction in liver. Mol Ther. 2001;3:28–35. doi: 10.1006/mthe.2000.0227. [DOI] [PubMed] [Google Scholar]
- Xu Z, Tian J, Smith JS., and , Byrnes AP. Clearance of adenovirus by Kupffer cells is mediated by scavenger receptors, natural antibodies, and complement. J Virol. 2008;82:11705–11713. doi: 10.1128/JVI.01320-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schiedner G, Bloch W, Hertel S, Johnston M, Molojavyi A, Dries V, et al. A hemodynamic response to intravenous adenovirus vector particles is caused by systemic Kupffer cell-mediated activation of endothelial cells. Hum Gene Ther. 2003;14:1631–1641. doi: 10.1089/104303403322542275. [DOI] [PubMed] [Google Scholar]
- Manickan E, Smith JS, Tian J, Eggerman TL, Lozier JN, Muller J, et al. Rapid Kupffer cell death after intravenous injection of adenovirus vectors. Mol Ther. 2006;13:108–117. doi: 10.1016/j.ymthe.2005.08.007. [DOI] [PubMed] [Google Scholar]
- Shashkova EV, Doronin K, Senac JS., and , Barry MA. Macrophage depletion combined with anticoagulant therapy increases therapeutic window of systemic treatment with oncolytic adenovirus. Cancer Res. 2008;68:5896–5904. doi: 10.1158/0008-5472.CAN-08-0488. [DOI] [PubMed] [Google Scholar]
- Shashkova EV, Spencer JF, Wold WS., and , Doronin K. Targeting interferon-alpha increases antitumor efficacy and reduces hepatotoxicity of E1A-mutated spread-enhanced oncolytic adenovirus. Mol Ther. 2007;15:598–607. doi: 10.1038/sj.mt.6300064. [DOI] [PubMed] [Google Scholar]
- Small EJ, Carducci MA, Burke JM, Rodriguez R, Fong L, van Ummersen L, et al. A phase I trial of intravenous CG7870, a replication-selective, prostate-specific antigen-targeted oncolytic adenovirus, for the treatment of hormone-refractory, metastatic prostate cancer. Mol Ther. 2006;14:107–117. doi: 10.1016/j.ymthe.2006.02.011. [DOI] [PubMed] [Google Scholar]
- Liu TC, Galanis E., and , Kirn D. Clinical trial results with oncolytic virotherapy: a century of promise, a decade of progress. Nat Clin Pract Oncol. 2007;4:101–117. doi: 10.1038/ncponc0736. [DOI] [PubMed] [Google Scholar]
- Shayakhmetov DM, Gaggar A, Ni S, Li ZY., and , Lieber A. Adenovirus binding to blood factors results in liver cell infection and hepatotoxicity. J Virol. 2005;79:7478–7491. doi: 10.1128/JVI.79.12.7478-7491.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker AL, Waddington SN, Nicol CG, Shayakhmetov DM, Buckley SM, Denby L, et al. Multiple vitamin K-dependent coagulation zymogens promote adenovirus-mediated gene delivery to hepatocytes. Blood. 2006;108:2554–2561. doi: 10.1182/blood-2006-04-008532. [DOI] [PubMed] [Google Scholar]
- Lesko LJ. The critical path of warfarin dosing: finding an optimal dosing strategy using pharmacogenetics. Clin Pharmacol Ther. 2008;84:301–303. doi: 10.1038/clpt.2008.133. [DOI] [PubMed] [Google Scholar]
- Waddington SN, McVey JH, Bhella D, Parker AL, Barker K, Atoda H, et al. Adenovirus serotype 5 hexon mediates liver gene transfer. Cell. 2008;132:397–409. doi: 10.1016/j.cell.2008.01.016. [DOI] [PubMed] [Google Scholar]
- Kalyuzhniy O, Di Paolo NC, Silvestry M, Hofherr SE, Barry MA, Stewart PL, et al. Adenovirus serotype 5 hexon is critical for virus infection of hepatocytes in vivo. Proc Natl Acad Sci USA. 2008;105:5483–5488. doi: 10.1073/pnas.0711757105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vigant F, Descamps D, Jullienne B, Esselin S, Connault E, Opolon P, et al. Substitution of hexon hypervariable region 5 of adenovirus serotype 5 abrogates blood factor binding and limits gene transfer to liver. Mol Ther. 2008;16:1474–1480. doi: 10.1038/mt.2008.132. [DOI] [PubMed] [Google Scholar]
- Tollefson AE, Scaria A, Hermiston TW, Ryerse JS, Wold LJ., and , Wold WS. The adenovirus death protein (E3-11.6K) is required at very late stages of infection for efficient cell lysis and release of adenovirus from infected cells. J Virol. 1996;70:2296–2306. doi: 10.1128/jvi.70.4.2296-2306.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doronin K, Toth K, Kuppuswamy M, Ward P, Tollefson AE., and , Wold WS. Tumor-specific, replication-competent adenovirus vectors overexpressing the adenovirus death protein. J Virol. 2000;74:6147–6155. doi: 10.1128/jvi.74.13.6147-6155.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doronin K, Toth K, Kuppuswamy M, Krajcsi P, Tollefson AE., and , Wold WS. Overexpression of the ADP (E3-11.6K) protein increases cell lysis and spread of adenovirus. Virology. 2003;305:378–387. doi: 10.1006/viro.2002.1772. [DOI] [PubMed] [Google Scholar]
- Yamamoto T, Naito M, Moriyama H, Umezu H, Matsuo H, Kiwada H, et al. Repopulation of murine Kupffer cells after intravenous administration of liposome-encapsulated dichloromethylene diphosphonate. Am J Pathol. 1996;149:1271–1286. [PMC free article] [PubMed] [Google Scholar]
- Duncan SJ, Gordon FC, Gregory DW, McPhie JL, Postlethwaite R, White R, et al. Infection of mouse liver by human adenovirus type 5. J Gen Virol. 1978;40:45–61. doi: 10.1099/0022-1317-40-1-45. [DOI] [PubMed] [Google Scholar]
- Paielli DL, Wing MS, Rogulski KR, Gilbert JD, Kolozsvary A, Kim JH, et al. Evaluation of the biodistribution, persistence, toxicity, and potential of germ-line transmission of a replication-competent human adenovirus following intraprostatic administration in the mouse. Mol Ther. 2000;1:263–274. doi: 10.1006/mthe.2000.0037. [DOI] [PubMed] [Google Scholar]
- Dhar D, Spencer JF, Toth K., and , Wold WS. Effect of preexisting immunity on oncolytic adenovirus vector INGN 007 antitumor efficacy in immunocompetent and immunosuppressed Syrian hamsters. J Virol. 2009;83:2130–2139. doi: 10.1128/JVI.02127-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toth K, Spencer JF, Dhar D, Sagartz JE, Buller RM, Painter GR, et al. Hexadecyloxypropyl-cidofovir, CMX001, prevents adenovirus-induced mortality in a permissive, immunosuppressed animal model. Proc Natl Acad Sci USA. 2008;105:7293–7297. doi: 10.1073/pnas.0800200105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reid T, Warren R., and , Kirn D. Intravascular adenoviral agents in cancer patients: lessons from clinical trials. Cancer Gene Ther. 2002;9:979–986. doi: 10.1038/sj.cgt.7700539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Q, Zaiss AK, Colarusso P, Patel K, Haljan G, Wickham TJ, et al. The role of capsid-endothelial interactions in the innate immune response to adenovirus vectors. Hum Gene Ther. 2003;14:627–643. doi: 10.1089/104303403321618146. [DOI] [PubMed] [Google Scholar]
- Di Paolo NC, van Rooijen N., and , Shayakhmetov DM. Redundant and synergistic mechanisms control the sequestration of blood-born adenovirus in the liver. Mol Ther. 2009;17:675–684. doi: 10.1038/mt.2008.307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morral N, O'Neal WK, Rice K, Leland MM, Piedra PA, Aguilar-Córdova E, et al. Lethal toxicity, severe endothelial injury, and a threshold effect with high doses of an adenoviral vector in baboons. Hum Gene Ther. 2002;13:143–154. doi: 10.1089/10430340152712692. [DOI] [PubMed] [Google Scholar]
- Doronin K, Shashkova EV, May SM, Hofherr SE., and , Barry MA.2009Chemical modification with high molecular weight polyethylene glycol reduces transduction of hepatocytes and increases efficacy of intravenously delivered oncolytic adenovirus Hum Gene Therepub ahead of print). [DOI] [PMC free article] [PubMed]
- Muruve DA. The innate immune response to adenovirus vectors. Hum Gene Ther. 2004;15:1157–1166. doi: 10.1089/hum.2004.15.1157. [DOI] [PubMed] [Google Scholar]
- Hartman ZC, Appledorn DM., and , Amalfitano A. Adenovirus vector induced innate immune responses: impact upon efficacy and toxicity in gene therapy and vaccine applications. Virus Res. 2008;132:1–14. doi: 10.1016/j.virusres.2007.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wold WS, Doronin K, Toth K, Kuppuswamy M, Lichtenstein DL., and , Tollefson AE. Immune responses to adenoviruses: viral evasion mechanisms and their implications for the clinic. Curr Opin Immunol. 1999;11:380–386. doi: 10.1016/S0952-7915(99)80064-8. [DOI] [PubMed] [Google Scholar]
- Schaack J. Induction and inhibition of innate inflammatory responses by adenovirus early region proteins. Viral Immunol. 2005;18:79–88. doi: 10.1089/vim.2005.18.79. [DOI] [PubMed] [Google Scholar]
- Janaswami PM, Kalvakolanu DV, Zhang Y., and , Sen GC. Transcriptional repression of interleukin-6 gene by adenoviral E1A proteins. J Biol Chem. 1992;267:24886–24891. [PubMed] [Google Scholar]
- Doronin K, Kuppuswamy M, Toth K, Tollefson AE, Krajcsi P, Krougliak V, et al. Tissue-specific, tumor-selective, replication-competent adenovirus vector for cancer gene therapy. J Virol. 2001;75:3314–3324. doi: 10.1128/JVI.75.7.3314-3324.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuppuswamy M, Spencer JF, Doronin K, Tollefson AE, Wold WS., and , Toth K. Oncolytic adenovirus that overproduces ADP and replicates selectively in tumors due to hTERT promoter-regulated E4 gene expression. Gene Ther. 2005;12:1608–1617. doi: 10.1038/sj.gt.3302581. [DOI] [PubMed] [Google Scholar]
- Ulasov IV, Rivera AA, Nettelbeck DM, Rivera LB, Mathis JM, Sonabend AM, et al. An oncolytic adenoviral vector carrying the tyrosinase promoter for glioma gene therapy. Int J Oncol. 2007;31:1177–1185. [PubMed] [Google Scholar]
- Campos SK., and , Barry MA. Current advances and future challenges in Adenoviral vector biology and targeting. Curr Gene Ther. 2007;7:189–204. doi: 10.2174/156652307780859062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waehler R, Russell SJ., and , Curiel DT. Engineering targeted viral vectors for gene therapy. Nat Rev Genet. 2007;8:573–587. doi: 10.1038/nrg2141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shashkova EV, Kuppuswamy MN, Wold WS., and , Doronin K. Anticancer activity of oncolytic adenovirus vector armed with IFN-alpha and ADP is enhanced by pharmacologically controlled expression of TRAIL. Cancer Gene Ther. 2008;15:61–72. doi: 10.1038/sj.cgt.7701107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Post DE, Sandberg EM, Kyle MM, Devi NS, Brat DJ, Xu Z, et al. Targeted cancer gene therapy using a hypoxia inducible factor dependent oncolytic adenovirus armed with interleukin-4. Cancer Res. 2007;67:6872–6881. doi: 10.1158/0008-5472.CAN-06-3244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarkar D, Lebedeva IV, Su ZZ, Park ES, Chatman L, Vozhilla N, et al. Eradication of therapy-resistant human prostate tumors using a cancer terminator virus. Cancer Res. 2007;67:5434–5442. doi: 10.1158/0008-5472.CAN-07-0195. [DOI] [PubMed] [Google Scholar]
- Barton KN, Paielli D, Zhang Y, Koul S, Brown SL, Lu M, et al. Second-generation replication-competent oncolytic adenovirus armed with improved suicide genes and ADP gene demonstrates greater efficacy without increased toxicity. Mol Ther. 2006;13:347–356. doi: 10.1016/j.ymthe.2005.10.005. [DOI] [PubMed] [Google Scholar]
- Hermiston T. A demand for next-generation oncolytic adenoviruses. Curr Opin Mol Ther. 2006;8:322–330. [PubMed] [Google Scholar]
- Cody JJ., and , Douglas JT. Armed replicating adenoviruses for cancer virotherapy. Cancer Gene Ther. 2009;16:473–488. doi: 10.1038/cgt.2009.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lohr F, Huang Q, Hu K, Dewhirst MW., and , Li CY. Systemic vector leakage and transgene expression by intratumorally injected recombinant adenovirus vectors. Clin Cancer Res. 2001;7:3625–3628. [PubMed] [Google Scholar]
- Sauthoff H, Hu J, Maca C, Goldman M, Heitner S, Yee H, et al. Intratumoral spread of wild-type adenovirus is limited after local injection of human xenograft tumors: virus persists and spreads systemically at late time points. Hum Gene Ther. 2003;14:425–433. doi: 10.1089/104303403321467199. [DOI] [PubMed] [Google Scholar]
- Campos SK., and , Barry MA. Rapid construction of capsid-modified adenoviral vectors through bacteriophage lambda Red recombination. Hum Gene Ther. 2004;15:1125–1130. doi: 10.1089/hum.2004.15.1125. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Predosing with Ad-GL-HB increases hepatocyte transduction with consequently delivered Ad as efficiently as predosing with Ad-DsRed-RD.