Abstract
The activities of total extracts and lupane triterpenes, isolated from the stem bark of Acacia mellifera, were evaluated against Plasmodium berghei strain ANKA in a female Swiss mouse model. Five isolated compounds and the crude extracts were evaluated for antimalarial activity and Quinine hydrochloride was used as a positive control. Only betulin and the methanolic extract produced considerable antimalarial activity in mice infected with P. berghei parasites. This study demonstrated the presence of bioactive agents in Acacia mellifera.
Keywords: Acacia mellifera, lupane triterpenes, Antimalarial activity, mice
Introduction
Malaria is a disease of immense public importance. Available data suggest that there are 200–300 million cases, with more than two million deaths each year (Krogstad et al, 1990) out of which one million is from the African continent alone [Daniel et al, 1988]. Chloroquine, a 4-aminoquinolone, has been the mainstay of malaria chemotherapy for almost 50 years now. This is because of its rapid onset of action, low toxicity and, almost low expense. It has remained the cheapest and most widely used antimalarial drug in parts of Africa (Daniel et al, 1988; Adubofour et al, 1992; WHO, 1984]. Chloroquine-resistant Plasmodium falciparum has been reported in parts of Africa (Le Bras et al, 1986; Sansonetti et al, 1985]. Resistance to chloroquine developed in Southeast Asia and South America at the end of the 1950s and in Africa by the late 1970s (Wharhurst, 2001]. Since the emergence of chloroquine resistant strains of P. falciparum, the rate of resistance has been increasing and limiting adequate treatment of malaria [Peters, 1998; Neequaye et al, 1986]. Consequently, there is an urgent global need to isolate from natural sources new classes of antimalarial compounds. A natural product artemisinin has previously shown excellent efficacy as non-alkaloidal, fast acting drug effective for chemotherapy against malaria (Ziffer et al, 1997]. Acacia mellifera has been reported to be used to treat malaria (Kokwaro, 1976] and used locally in Kenya traditional medicine.
As part of the search for new natural product for the treatment of various diseases (Mutai et al, 2004), we carried out a screening procedure to test A. mellifera total extract for the ability to treat malaria. On further purification of total extracts, pure metabolites were obtained and were also further investigated (Mutai et al, 2004].
Materials and Methods
Plant collection and Extraction
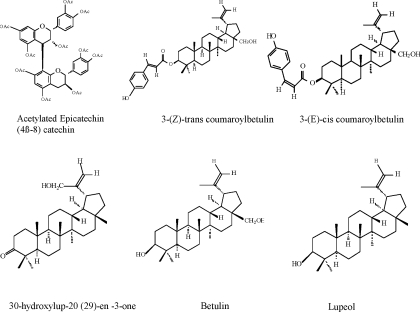
Stem bark of A. mellifera was collected in January 2000, from at Machakos Kenya. The plant was authenticated at the East Africa Herbarium, National Museum of Kenya and a voucher specimen deposited at the same Institute in Nairobi, (Voucher number ChM-I). Air-dried, powdered stem bark of A.. mellifera (2.25 kg) was extracted at room temperature, successively with dichloromethane (CH2Cl2) and methanol (MeOH) for 48 h each. The solvents were removed in vacuo to yield CH2Cl2 extract and MeOH extract. The extracts were each subjected separately to silica gel column chromatography eluted with a mixture of EtOAc and MeOH of increasing polarity. The purity of fractions was monitored using TLC. Identical fractions were pooled together. Further purification was done by rechromatography using gravity columns and then later subjected to HPLC. Six pure compounds namely: Acetylated epicatechin (4β-8) catechin from methanol extract and five lupan types (Figure 1): 3-(Z)-trans coumaroylbetulin, 3-(E)-cis coumaroylbetulin, 30-hydroxylup-20 (29)-en -3-one, Betulin and Lupeol from dichloromethane extract were isolated and their structures elucidated using spectroscopic methods.
Figure 1.
Isolated compounds
Parasites and Antimalarial Tests
The Plasmodium berghei strain ANKA used in the study was obtained from Kenya Medical Research Institute (KEMRI). The parasite strain was propagated in mice and maintained by serial passage of infected erythrocytes.
The antimalarial activity of the extracts or pure compounds was evaluated by the 4-day suppressive test described by Peters et al (1970, 1982). Adult female Swiss mice weighing 18–21 g were inoculated through i.p. route with 1 × 107 P. berghei-infected erythrocytes. The mice were randomly divided into groups of five animals per cage and treated during 4 consecutive days with daily doses of the extracts, by oral route. Two control groups were used in each experiment; one treated with quinine at low non-curative dose (10mg/ kg/ day, orally) and the other group was kept untreated. Each day from day 0 to 4, parasites were determined in coded blood smears by randomly counting 2000–6000 erythrocytes in the case of low parasitaemia (≤10%) or up to 1000 erythrocytes in the case of higher parasitaemia. Overall mortality was monitored daily in all groups during a period of four weeks following inoculation. The inhibition of parasite growth in the drug-treated group was calculated. The extracts were considered partially active when parasitaemia was reduced by > 30%.
Statistical analysis
The statistical significance of differences in mean parasitemias between control and test groups were assessed using the Student t-test. P-values of 0.05 or less were considered statistically significant.
Results and Discussion
The results of the 4-day suppressive antimalarial screening of the total extracts/pure compounds (and Quinine Hydrochroride) at different doses in mice parasitized with P. berghei berghei are summarised in Tables 1 and 2. The suppression of parasitemia expressed as percentage (S.P.; %) and standard deviations (n = 5) are given for each extract/drug.
Table 1.
Mean chemosuppression of mice treated with total extracts
| Treatment | Dose mg/kg/day | Mean (S.D) value % | |
| Parasitemia | Chemosuppression | ||
| Control | saline | 31.97 (5.83) | 0.0 (0.0) |
| A | 200 | 25.00 (0.57) | 21.80 (1.77) |
| A2 | 200 | 27.30 (2.31) | 14.61 (7.21) |
| M200 | 200 | 29.10 (2.69) | 22.32 (1.79) |
| M800 | 800 | 18.07 (2.80) | 43.49 (8.77) |
| Quinine Hydrochroride | 10 | 3.03 (3.52) | 90.51 (11.0) |
Key: A : Dichloromethane Extract; A2 : 25% dichloromethane in methanol Extract M200: Methanol Extract 200mg/kg/day; M800: Methanol Extract 800mg/kg/day
Table 2.
Mean chemosuppression of mice treated with pure compounds.
| Treatment | Dose mg/kg/day | Mean (S.D) value % | |
| Parasitemia | Chemosuppression | ||
| Control | Water | 36.70 (6.22) | 0.0 (0.0) |
| ML 4 | 20 | 35.20 (7.41) | 15.67 (3.27) |
| ML 9 | 20 | 30.60 (2.77) | 16.50 (7.55) |
| ML 13 | 20 | 30.67 (2.77) | 16.44 (7.56) |
| ML 11 | 20 | 37.03 (2.55) | 00.82 (8.86) |
| ML 12 | 20 | 33.37 (10.49) | 24.80 (12.33) |
| ML 14 | 20 | 33.87 (5.91) | 16.62 (6.55) |
| Quinine Hydrochroride |
10 | 04.85 (0.64) | 86.79 (1.73) |
Key: ML 4: Acetylated epicatechin (4β-8) catechin; ML 9: 3-(Z)-trans coumaroylbetulin ML13: 3-(E)-cis coumaroylbetulin ; ML 11: 30-hydroxyl lupan-20 (29)-en -3-on ML 12 : Betulin; ML 14: Lupeol
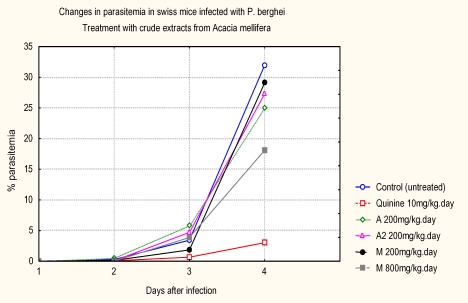
At 200 mg/kg the suppression of parasites was low: 21.80 % for dichloromethane extract, 14.6 % for 25 % dichloromethane in methanol extract, and 22.3 % for methanol extract. However, high dose for methanol extract (800 mg/kg), the percent of parasitemia suppression improved to 43.5 %. The three extracts had low antimalarial activities when compared to Quinine hydrochroride. The mean percentage parasitaemia in Quinine hydrochroride control group was 3.03 ± 3.52 %, while mean percentage parasitaemia in the untreated control was 31.97 ± 5.83%. The progression of parasitemia is shown in Figure 2.
Figure 2.
Progression of parasitemia with time after treatment with total extracts
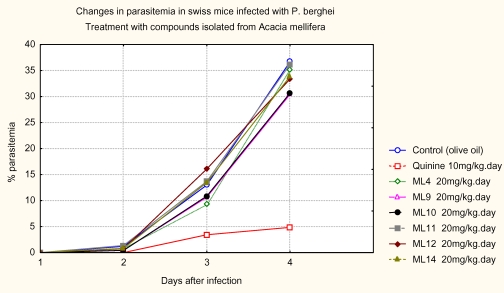
The progression of parasitemia in animals treated with the pure compounds is shown in Figure 3. The antimalarial activity of total and pure compounds from A. mellifera in P. berghei-infected mice is shown in Tables 1 and 2 respectiviely. The in vivo test showed that the dichloromethane (CH2Cl2) in 50% methanol (MeOH) extract at a dose of 200mg/kg was inactive because there was no significant reduction in the chemosuppression of parasitaemia on the fourth day. However, the dichloromethane (CH2Cl2) and MeOH extracts at the same dose, produced chemosuppression of parasitemia (> 21.8% and 22.3 % respectively) when administered orally. At a dose of 800mg/kg, methanol extract produced the highest chemosuppression of parasitemia (> 43.5%) when administered orally. The profound chemosuppression of parasitemia of the methanol extract of this plant in our study underscores the need to study all parts of this plant such as roots and the leaves, which had been reported to be used also but not yet, documented.
Figure 3.
Progression of parasitemia with time after treatment with pure compounds
The MeOH extract showed slight mortality rate at the dose of 800mg/kg weight of mice as compared to non-treated mice. This could be clear case of toxicity. The 3-(Z)-tran-coumaroylbetulin, lupeol and 3-(E)-cis-coumaroylbetulin were found to have close values of chemosuppression of parasitemia which was not significant. The parasitemia inhibition percent of betulin was the highest (24.8 %). This observation that betulin is not effective in vivo is consistent with earlier findings where it demonstrated in vitro inactivity at 500 mg/ml for both chloroquine resistant and sensitive P. falciparum [Steele et al, 1999]. However, the in vivo activity of betulin related triterpene; betulinic acid was ineffective at reducing parasitemia and also exhibited some toxicity [Steele et al, 1999]. On the other hand, lupeol has been described to exhibit inhibitory activity on Plasmodium falciparum growth in vitro but lacks in vivo activity in mice infected with P. berghei (Alves et al., 1997). This result clearly indicated peculiarities in the metabolic disposition of the chemical constituents. Therefore, plants and pure compounds found to be active in vitro must be tested in vivo before a definite statement can be made on their antimalarial potential.
Acknowledgement
The authors gratefully acknowledge financial support from State Scholarships Foundation of Greece and study leave granted by Director KEMRI. This work is published with the approval of the Director KEMRI.
References
- 1.Adubofour K O. Drug resistance in malaria: A review of West African situation. J Natl Med Assoc. 1992;84:1025–1029. [PMC free article] [PubMed] [Google Scholar]
- 2.Alves T M A, Nagem T J, Carvalho L H, Krettli AU. Antiplasmodial triterpene from Vernonia brasiliana. Planta Med. 1997;63:554–555. doi: 10.1055/s-2006-957764. [DOI] [PubMed] [Google Scholar]
- 3.Bisset B C, Nwaiwu M K. Quaternary alkaloids of Tinospora spp. Planta Medica. 1994;48:275–279. doi: 10.1055/s-2007-969933. [DOI] [PubMed] [Google Scholar]
- 4.Daniel H I, Molta N B. Efficacy of chloroquine in the treatment of malaria in children under the age of 5 years in Baissa (Gongola state), Nigeria. Annals Trop Med Parasitol. 1989;83:331–338. doi: 10.1080/00034983.1989.11812353. 1989. [DOI] [PubMed] [Google Scholar]
- 5.Eltord B C. L-glutamine influx in malaria; infected erythrocytes; a target for antimalarials. Parasitology Today. 1986;2:309–312. doi: 10.1016/0169-4758(86)90126-2. [DOI] [PubMed] [Google Scholar]
- 6.Gessler M C, Msuya D E, Nkunya M H H. Traditional helars in Tanzania: The Treatment of malaria with plant remedies. J Enthnopharmacol. 1995;48:131–144. doi: 10.1016/0378-8741(95)01293-m. [DOI] [PubMed] [Google Scholar]
- 7.Kokwaro O. Medicinal plant of East Africa. Kampala, Nairobi, Dar es Salaam: East African Literature Bureau; 1976. [Google Scholar]
- 8.Krogstad D J, Schlesinger P L, Herwaldt B L. Antimalarial agents: Mechanism of chloroquine resistance. Antimicrob Agents Chemother. 1990;32:799–801. doi: 10.1128/aac.32.6.799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Le Bras J, Haitin I, Bouree P, Coco-cianci O, Garin J P, Rey M, Charmont G, Roue R. Chloroquine-resistant falciparum in Benin. Lancet. 1986;11:1043–1044. doi: 10.1016/s0140-6736(86)92657-7. [DOI] [PubMed] [Google Scholar]
- 10.Mahato S B, Nandy AK, Roy G. Triterpenoids. Phytochemistry. 1992;31:2199–2249. doi: 10.1016/0031-9422(92)83257-y. 1992. [DOI] [PubMed] [Google Scholar]
- 11.Malan E. Derivatives of (+)-catechin-5-gallate from the bark of Acacia nilotica. Phytochemistr. 1991;30(8):2737–2739. [Google Scholar]
- 12.Mutai C, Abatis D, Vagias C, Moreau D, Roussakis C, Roussis V. Cytotoxic lupane-type triterpenoids from Acacia mellifera. Phytochemistry. 2004;65:1159–1164. doi: 10.1016/j.phytochem.2004.03.002. [DOI] [PubMed] [Google Scholar]
- 13.Neequaye J, Coe-Ene J, Taelman H. In vivo chloroquine resistant falciparum malaria in West Africa. Lancet. 1986;1:153–154. [PubMed] [Google Scholar]
- 14.Peters W. chemotherapy and drug resistance in malaria. London: Academic press; 1970. Techniques of Drug evaluation 1; primary screening. 1970. [Google Scholar]
- 15.Peter W. Antimalarial drug resistance. British Medical Bulletin. 1982;32:187–192. doi: 10.1093/oxfordjournals.bmb.a071757. [DOI] [PubMed] [Google Scholar]
- 16.Peters W. Drug resistance in malaria parasites of animals and man. Adv Parasitol. 1998;41:1–62. doi: 10.1016/s0065-308x(08)60421-2. [DOI] [PubMed] [Google Scholar]
- 17.Sansonetti P J, Le Bras C, Vedier F, Charmont G, Dupont B, Lepresle C. Chloroquine-resistant Plasmodium falciparum in Cameroon. Lancet. 1985;1:1154–1155. doi: 10.1016/s0140-6736(85)92453-5. 1985. [DOI] [PubMed] [Google Scholar]
- 18.Steele J C, Warhurst D C, Kirby GC, Simmonds M S. In vitro and In vivo evaluation of betulinic acid as an antimalaria. Phytother Res. 1999;13(2):115–119. doi: 10.1002/(SICI)1099-1573(199903)13:2<115::AID-PTR404>3.0.CO;2-1. [DOI] [PubMed] [Google Scholar]
- 19.Wharhurst D C. A molecular marker for chloroquine-resistant falciparum malaria (editorial) New Engl J Med. 2001;344:299–302. doi: 10.1056/NEJM200101253440411. [DOI] [PubMed] [Google Scholar]
- 20.World Health Organisation, author. WHO Advances in Malaria Chemotherapy. 1984. (Technical Report Series No. 711). [PubMed] [Google Scholar]
- 21.Ziffer H, Highet R J, Klayman D L. Artemisinin; An endoperoxidic antimalarial from Artemisia annua L. Prog Chem Org Nat Prod. 1997;72:123–214. doi: 10.1007/978-3-7091-6527-0_2. [DOI] [PubMed] [Google Scholar]