Abstract
Vinclozolin is an endocrine-disrupting chemical (EDC) that binds with high affinity to the androgen receptor (AR) and blocks the action of gonadal hormones on male reproductive organs. An alternative mechanism of action of Vinclozolin involves transgenerational effects on the male reproductive tract. We previously reported in utero Vinclozolin exposure-induced prostatitis (prostate inflammation) in postpubertal rats concurrent with down-regulation of AR and increased nuclear factor-κB activation. We postulated the male reproductive abnormalities induced by in utero Vinclozolin exposure could be reversed by testosterone supplementation, in contrast to the permanent modifications involving DNA methyltransferases (Dnmts) described by others. To test this hypothesis, we administered high-dose testosterone at puberty to Vinclozolin-treated rats and determined the effect on anogenital distance (AGD); testicular germ cell apoptosis, concentration of elongated spermatids, and the onset of prostatitis. Concurrently we examined Dnmt1, −3A, −3B, and −3L mRNA expression. Consistent with previous reports, in utero exposure to Vinclozolin significantly reduced AGD, increased testicular germ cell apoptosis 3-fold, reduced elongated spermatid number by 40%, and induced postpubertal prostatitis in 100% of exposed males. Administration of high-dose testosterone (25 mg/kg) at puberty normalized AGD, reduced germ cell apoptosis, and restored elongated spermatid number. Testosterone restored AR and nuclear factor-κB expression in the prostate and abolished Vinclozolin-induced prostatitis. Altered Dnmt expression was evident with in utero Vinclozolin exposure and was not normalized after testosterone treatment. These data demonstrate in utero Vinclozolin-induced male reproductive tract abnormalities are AR mediated and reversible and involve a mechanism independent of Dnmt expression.
Vinclozolin is an endocrine disrupting chemical that causes inflammation in the prostate (prostatitis) and reduces sperm production; previously thought to be trans-generational and irreversible, data show both effects can be abrogated by high dose androgen supplementation.
Environmental endocrine disrupting chemicals (EDCs) are defined by the U.S. Environmental Protection Agency as “exogenous agents that interfere with the synthesis, secretion, binding, action, or elimination of natural hormones in the body that are responsible for the maintenance of homeostasis, reproduction, development, and/or behavior.” The first evidence for adverse effects of EDCs came from observational changes and reports on the effects on wildlife (1). In humans, trends in reproductive system abnormalities such as declining sperm counts, cryptorchidism, and hypospadias (2) and testicular cancer (3) have been postulated to be linked to EDC exposure.
Vinclozolin [3-(3,5-dichlorophenyl)-5-methyl-5-vinyl-oxazolidine-2,4-dione] is an antiandrogenic fungicide used in the control of Botrytis cinerea, Sclerotinia sclerotiorum, and Moniliniam spp on fruits, vegetables, ornamental plants, and vines. Vinclozolin is degraded to several metabolites: 2-(3,5-dichloropheniyl)-carbamoyl-oxy-2-methyl-3-butenoic acid and 3′,5′-dichloro-2-hydroxy-2-methylbut-3-enanilide, which are competitive antagonists of androgen receptor (AR) ligand binding. Vinclozolin also inhibits AR transactivation and androgen-dependent gene expression and increases androgen degradation (4,5,6).
The timing of Vinclozolin exposure alters the effect on male reproductive tract development. Exposure between gestational day (GD) 14 and postnatal day (PND) 3 in the rat, at concentrations as low as 3 mg/kg · d, permanently reduces weights of androgen-dependent tissues (7), emphasizing the sensitivity of the developing fetus to the activity of antiandrogenic chemicals. A window of sensitivity for male reproductive tract development occurs in rats when ARs are activated between GD14 and GD19, rather than during embryonic gonadal sex determination around GD8-14 (8). Although the exposure may be transient, the functional alterations and consequences are reported to be permanent and irreversible and may not be discovered until puberty or thereafter.
Recent studies from our laboratory demonstrated the induction of prostatitis in adult male rodent offspring after in utero exposure to the antiandrogenic EDC, Vinclozolin, during the period of male reproductive tract development (GD14-19), (9). Furthermore, induction of prostate inflammation was associated with a down-regulation of AR expression and an up-regulation of nuclear factor-κB (NF-κB) activity (9).
Vinclozolin has also been implicated in epigenetic modifications of male reproductive tract, specifically via changes in DNA methyltransferase expression. Reports by Anway and colleagues (10,11) demonstrated that administration of the antiandrogenic EDC Vinclozolin during sex determination (GD8-14), modifies the developing testis and prostate transcriptome, and specifically reduces gene expression of DNA methyltransferases (Dnmts) Dnmt1, Dnmt3A, and Dnmt3L. This altered epigenetic programming was speculated to be the mechanism involved in both the adult-onset disease and trans-generational phenotypes previously reported (12). The transgenerational effects of Vinclozolin reported by Anway et al. were novel and noteworthy, but subsequently two independent groups were unable to replicate the results (13,14). More recently retraction of a portion of the original data continues to stimulate debate (15,16).
Based on these studies and the previously characterized antiandrogenic actions of Vinclozolin, we postulated in utero exposure to Vinclozolin leads to adverse effects on male reproductive organs by a reversible hormone-induced mechanism, in contrast to the irreversible modifications mediated by Dnmts proposed by Anway and colleagues (10,11,12,17). To test our hypothesis, we measured anogenital distance (AGD), testicular germ cell apoptosis, and elongated spermatid number. We assessed the prostate for evidence of inflammation and AR and NF-κB expression. We also measured Dnmt mRNA expression in the testis and prostate of in rats exposed in utero to Vinclozolin followed by administration of high-dose testosterone at puberty. In addition, we characterized the prostate of mice lacking Dnmt3L (18).
Materials and Methods
Animals
All animal procedures were conducted in accordance with the National Health and Medical Research Council guidelines and Animal Experimentation Ethics Committee at Monash Medical Centre, Clayton, Australia (MMCA/2008/10). Animals were treated humanely and with regard for alleviation of suffering. Time-mated, female outbred Sprague Dawley rats were obtained on GD 8 and housed under controlled 12-h light, 12-h dark cycle and temperature conditions. Animals were fed ad libitum. GD0 was the day plugs were observed. Male offspring were euthanized humanely by CO2 asphyxiation at PND56. Male Dnmt3L wild-type and knockout mice were from The Walter and Eliza Hall Institute of Medical Research (Parkville, Victoria, Australia) and were generated as previously described (18). Analysis in Dnmt3L mice was limited to characterization of the prostate.
Treatment
Vinclozolin ± testosterone treatment was administered to the rats described above. Vinclozolin was obtained from BASF AG (Research Triangle Park, NC) as Ronilan EG (a 50% Vinclozolin mixture) and Vinclozolin purified and recrystallized. Catalogued as lot no. 357-141A, it was certified as being of greater than 99% purity by ChemService Inc. (West Chester, PA). On GD14 dams were weighed and animal allocation to treatment groups done by body weight (bw) randomization to ensure unbiased weight distribution among groups. Dams were assigned to one of two treatment groups (0, 100 mg/kg · bw Vinclozolin) (n = 15 dams per group) and one of three pubertal (PND28) treatment groups (0, 5, or 25 mg testosterone; five litters per group). Dams were orally dosed daily at 1000 h from GD14 to GD19 with 100 mg/kg/bw Vinclozolin (BASF) or corn oil vehicle control (2.5 ml/kg · bw; Sigma, St. Louis, MO) and examined for clinical signs of toxicity. The window of sensitivity to Vinclozolin for male reproductive development was established by Wolf et al. in 2000 (8). The dose we used was chosen so that we would observe a definitive effect and was based on previous work (7) whereby the same dose was used and proven to be effective. Furthermore, because this current study sought to determine whether high-dose testosterone could reverse the Vinclozolin-mediated prostate pathology, we used the same dose and same treatment regimen as our earlier study (9). Because the pubertal period in the rodent is controversial, puberty was defined in relation to functional puberty, or the time at which sperm appear and serum testosterone rises, which occurs around PND43 in rats (19,20).
At PND 28 male rats were treated with sc testosterone esters (5 or 25 mg) supplied as Sustanon 100 or Sustanon 250 (Organon, Oss, The Netherlands) every third day for 4 wk (until PND56). Control rats received the equivalent volume of peanut oil (Sigma). The doses of testosterone were chosen because they had previously been identified to raise serum testosterone levels to 4.5- to 24-fold above control (21).
Necropsy of male littermates
Male offspring were collected at PND56, weighed and euthanized by CO2 asphyxiation. External genitalia, including scrotum, prepuce, and penis, were visually examined and AGD measured with a caliper.
Tissue collection
Ventral (VP), lateral, dorsal, and anterior prostate lobes were dissected from PND56 corn oil or Vinclozolin-treated male offspring and Dnmt3L knockout and wild-type mice aged 2–3 months (n = 3/group) and 9–14 months (n = 5/group). Wet weights were recorded and pair matched organs fixed in Bouin’s or immediately frozen. All lobes were hematoxylin and eosin (H&E) stained and examined for evidence of abnormalities.
Histology
Immunohistochemistry was performed using the Dako Autostainer universal staining system (Dako A/S, Glostrup, Denmark) (22). Antibodies were purchased from Santa Cruz Biotechnology Inc. (Santa Cruz, CA) to AR (N-20; Dako Corp.) and phospho-NF-κB p65 (Ser536; Cell Signaling Technology Inc., Beverly, MA) and were used as previously described (23,24,25). Apoptosis was analyzed by ApopTag in situ apoptosis detection kit (Intergen, Purchase, NY) as previously described (23)
RT-PCR
Total RNA was extracted and reverse transcription performed from prostate tissues (26). Semiquantitative PCR analysis was performed using primer sets previously published (10,26). The relative gene expression levels of Dnmt1, Dnmt3A, Dnmt3B, and Dnmt3L were determined from a minimum of four prostates per group in duplicate by semiquantitative RT-PCR. Results were presented relative to β-actin mRNA expression, which did not change with Vinclozolin or testosterone treatment, and analyzed with Scion Image (version 4.0.3.2; National Institutes of Health, Bethesda, MD).
SDS-PAGE and Western blot
Fifty milligrams of tissue were homogenized in Complete Lysis-M buffer containing protease inhibitors (Roche Applied Science, Indianapolis, IN) and the DC Protein Assay (Bio-Rad Laboratories, Hercules, CA) used to determine total protein concentration. Fifty micrograms of total protein were loaded onto SDS-PAGE gels and assessed for Dnmt protein expression (Dnmt1 clone C-17, Dnmt3A clone-15, Dnmt3B clone T16, and Dnmt3L clone N14; Santa Cruz Biotechnology). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH; ab9484; AbCam, Cambridge, UK) was assessed as a loading control and intensity of bands were assessed with Scion Image (version 4.0.3.2; NIH) (27).
Stereology
An unbiased assessment of apoptosis, AR localization, incidence of inflammation, and nuclear localization of NF-κB was obtained using stereological techniques, based on the Cavalieri principle (28) and as previously described (29,30,31). Counting was performed on a minimum of three tissues per group using a 3 × 3 counting frame on a minimum of five sections uniformly spaced throughout the tissue. Positively and negatively stained cells were identified and percentages determined. To determine the incidence of inflammatory lesions, random fields were designated as positive or negative for inflammation. Abnormal inflammatory regions were classified as areas that displayed chronic inflammation.
Sperm production
Elongated spermatid content per testis was determined from a minimum of five animals, selected at random, per group at PND56 using the method of Robb et al. (32). Briefly, testes were weighed and homogenized in buffer of 0.15 m NaCl, 0.1 mm NaN3, and 0.05% Triton X-100. The concentration of elongated spermatids (Triton resistant cells) per testis was determined using a hemocytometer and used to calculate the number of elongated spermatids per testis (32).
Dnmt3L knockout mice
Stereological analysis was performed on tissue sections from the wild-type and knockout 2- to 3-month age group to quantify relative compartmental volumes (epithelial, stromal, and lumen), proliferative cells, and AR-positive epithelial cells as previously described (29,30,31).
Statistical analysis
All pup data were analyzed individually and nested by dam to yield litter means. To test for significance of treatment effects, the litter was corrected for as a main effect variable using one-way analysis of covariance on SPSS (version 16; SPSS Inc., Chicago, IL) and data expressed as litter mean ± sem. AGD and organ weights were analyzed with body weight as a covariate, and all groups were compared using a one-way ANOVA with a post hoc Bonferoni analysis, with the significance threshold used at a level of 5% (P < 0.05). Gene expression is presented as mean ± sem and was compared using ANOVA with a post hoc Bonferroni analysis, with the significance threshold used at a level of 5% (P < 0.05). Statistical analysis was performed using Prism 4.0 software (GraphPad Software, Inc., San Diego, CA).
Results
Body weight and AGD
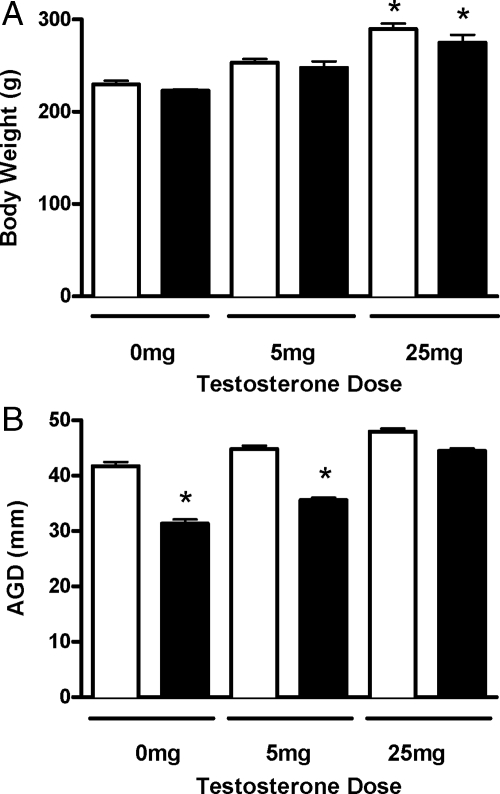
Consistent with our previous study (9), analysis of PND56 male offspring, exposed to 0 mg testosterone at puberty, revealed no significant differences in body weight, (Fig. 1A). After in utero control and Vinclozolin exposure, administration of 5 mg testosterone at puberty did not significantly change body weight, whereas 25 mg testosterone significantly increased body weight (P < 0.05). No significant difference was observed between animals exposed in utero to control or Vinclozolin at all testosterone doses (Fig. 2A). In utero exposure to Vinclozolin significantly reduced AGD. In rats exposed in utero to Vinclozolin, administration of 0 or 5 mg testosterone on PND28 did not restore AGD (P < 0.05). However, the AGD of rats exposed in utero to Vinclozolin and 25 mg testosterone was not significantly different from controls (Fig. 1B). Collectively these data show that in utero exposure to Vinclozolin decreases AGD, but this can be returned to control values by exposure to high concentrations of testosterone at puberty
Figure 1.
Gross analysis of male offspring exposed in utero to control (open bar) or Vinclozolin (solid bar) and exposed at puberty to 0, 5, or 25 mg testosterone. *, Body weight denotes significance vs. 0 mg testosterone-treated groups (A) and AGD (B). *, Significance vs. matched testosterone-treated control group (P < 0.05, mean ± sem).
Figure 2.
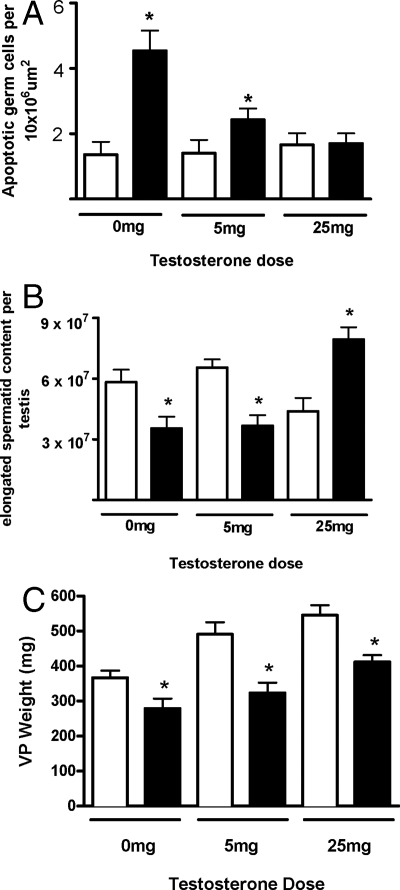
Germ cell apoptosis (A), elongated spermatid content (B) in the testis, and VP weight (C) of male offspring exposed in utero to control (open bar) or Vinclozolin (solid bar) and exposed at puberty to 0, 5, or 25 mg testosterone. Significant difference in the proportion of apoptotic cells between control (open bar) and Vinclozolin (solid bar)-treated testis per 10 × 106 μm2 were evident and testosterone treatment (25 mg) at puberty normalized germ cell apoptosis (A). A significant reduction in elongated spermatid content was observed in Vinclozolin-treated animals (solid bar) compared with control (open bar). VP weight in male offspring exposed in utero to control (open bar) or Vinclozolin (solid bar) (B) and exposed at puberty to 0, 5, or 25 mg testosterone (C) is shown. *, P = 0.05 vs. matched testosterone-treated control group. Mean ± sem
Characterization of the testis of in utero Vinclozolin-treated offspring
There was no evidence of inflammation or other overt pathology in the testis of Vinclozolin-treated animals (data not shown). No significant difference in testicular weight was observed between control and treated male offspring; however, an unbiased stereological analysis revealed a significant 3-fold increase (P < 0.05) in the number of apoptotic germ cells in testis of in utero Vinclozolin-treated males compared with controls. Treatment with 25 mg, but not 5 mg, of testosterone at puberty abolished Vinclozolin-induced germ cell apoptosis (Fig. 2A). A significant reduction (P < 0.05) in elongated spermatid content per testis was observed in Vinclozolin-treated offspring treated with 0 and 5 mg of testosterone. High concentrations of testosterone tended to decrease elongated spermatid content per testis in the control group (P = 0.058), whereas in utero exposure to Vinclozolin followed by 25 mg testosterone treatment increased elongated spermatid content per testis significantly (P < 0.01 vs. testosterone matched control and no testosterone control, Fig. 2B). Although not examined in any detail, the data presented in Fig. 3B are consistent with a testosterone-mediated spermiation (sperm release) failure similar to that observed in both rats and humans after administration of exogenous systemic testosterone (33,34).
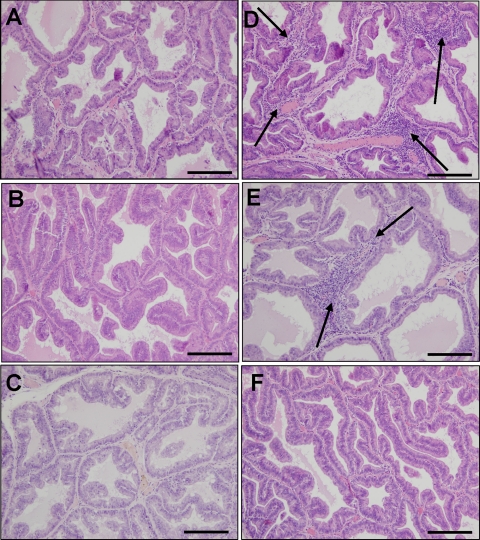
Figure 3.
H&E of prostate specimens exposed in utero to control (A and B) or Vinclozolin and 5 mg (C and D) or 25 mg testosterone at puberty (E and F). Prostate inflammation (←) was evident in prostates exposed in utero to Vinclozolin and 0 or 5 mg testosterone (B). Bar, 20 μm .
Prostate pathology
The VP is the most androgen-sensitive prostate lobe in rodents and as such was the only lobe that responded to Vinclozolin exposure. All prostate lobes were collected, weighed, and subject to H&E staining to assess morphology. In agreement with our previous study (9), the VP was the only lobe with observable pathology (inflammatory disease/lesions) after in utero Vinclozolin exposure and was therefore the only prostate lobe reported herein. The VP weights of animals exposed in utero to Vinclozolin followed by 0 mg testosterone were significantly reduced compared with in utero control (P < 0.05). The administration of 5 or 25 mg testosterone at puberty significantly increased VP weight in both in utero control and Vinclozolin-exposed groups; however, in utero Vinclozolin-exposed groups were still significantly reduced compared with in utero control counterparts (P < 0.05) (Fig. 2C). Collectively these data show that in utero exposure to Vinclozolin decreases VP weight, regardless of testosterone administration at puberty.
Consistent with our previous study (9), postpubertal, 56-d-old male offspring exposed to Vinclozolin in utero displayed prostate inflammation (prostatitis) (Fig. 3B). Vinclozolin treatment resulted in a significant increase (P < 0.05) in the percentage of prostatic inflammatory lesions from 1.55 ± 0.69 to 17.65 ± 1.41%.
Testosterone treatment reverses in utero EDC-induced prostatitis and restores AR expression and NFκB activation
After exposure to Vinclozolin in utero and 5 mg testosterone at puberty, prominent, but focal, regions of inflammation remained evident (Fig. 3D). Lesions were characterized by increased numbers of inflammatory cells, particularly leukocytes and macrophages, surrounding the ducts and infiltrating into the vessels. The incidence of inflammatory lesions after in utero Vinclozolin and 5 mg testosterone exposure was not significantly reduced compared with in utero Vinclozolin and 0 mg testosterone (15.85 ± 1.65 vs. 17.65 ± 1.41%, respectively). Animals exposed in utero to Vinclozolin and 25 mg testosterone showed no evidence of inflammatory pathology (Fig. 3F). The absence of any inflammatory lesions was confirmed using an unbiased stereological analysis. Inflammatory lesions were not detectable in corresponding in utero control prostates at all doses of testosterone (Fig. 3, A, C, and E).
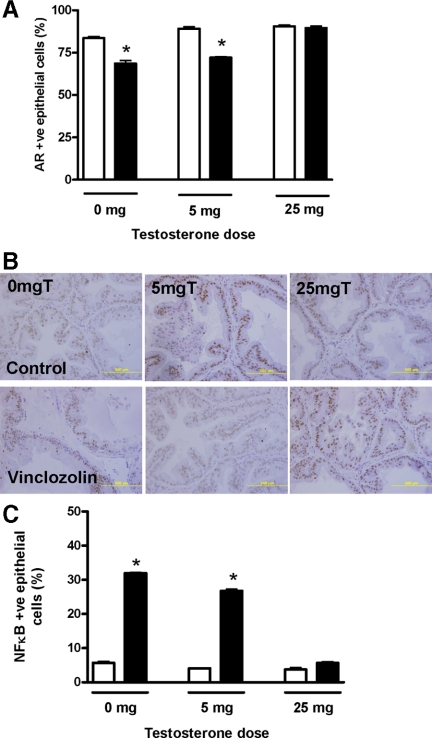
In utero Vinclozolin treatment plus 0 mg testosterone at puberty resulted in a significant reduction in the percent of prostate epithelial cells that are AR positive (P < 0.05) compared with in utero control treatment (Fig. 4A). After in utero control exposure and 5 or 25 mg testosterone treatment, a significant increase in AR expression was observed (P < 0.05) compared with exposure to 0 mg testosterone. Vinclozolin treatment significantly reduced AR levels in both 0 and 5 mg testosterone treatment groups (P < 0.05) compared with their in utero control counterparts. In utero Vinclozolin-treated animals that received 25 mg testosterone showed no significant difference in AR expression compared with controls (Fig. 4A). Representative examples of AR immunohistochemistry are shown for each treatment in Fig. 4B.
Figure 4.
Unbiased stereological analysis of the percent of epithelial cells that were AR (A) and nuclear NFκB localization (B) in tissues exposed in utero to control (open bar) or Vinclozolin (solid bar) and 0, 5, or 25 mg testosterone at puberty. A, Compared with control counterparts, epithelial expression of AR was significantly decreased in all tissues except after 25 mg pubertal testosterone treatment. B, Compared with control counterparts, nuclear NF-κB immunolocalization was increased in all specimens except after 25 mg pubertal testosterone treatment. *, Significance vs. matched testosterone-treated control groups (P < 0.05). Mean ± sem (n = 5)..
An unbiased stereological analysis of nuclear phospho-NF-κB p65 (Ser536) localization revealed a significant increase in nuclear NF-κB localization from 5.69 ± 0.39 to 31.99 ± 0.18% after Vinclozolin exposure (P < 0.05) (Fig. 4C). Pubertal testosterone treatment (5 mg) significantly reduced in utero Vinclozolin-induced nuclear localization of NF-κB (P < 0.05), but levels remained significantly above those seen in controls (Fig. 4C). Treatment of animals exposed in utero to Vinclozolin with 25 mg testosterone at puberty, however, prevented nuclear localization of phospho-NF-κB, with levels comparable with those seen controls (Fig. 4C).
Dnmt expression in the prostate and testis
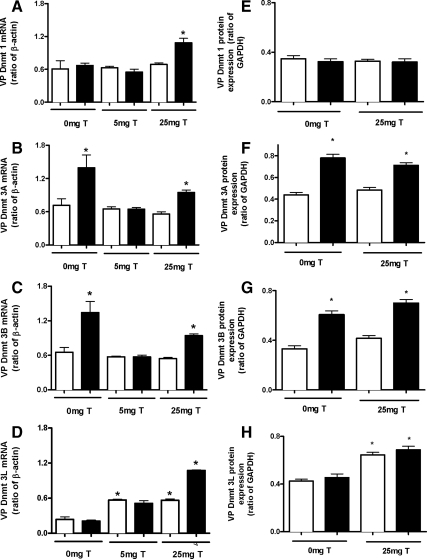
Whereas no change in Dnmt1 expression was observed in the prostate after in utero Vinclozolin treatment plus 0 or 5 mg testosterone, a significant increase (P < 0.05) was observed after in utero Vinclozolin plus 25 mg testosterone compared with control counterparts (Fig. 5A). Dnmt3A and Dnmt3B mRNA was significantly increased after in utero Vinclozolin exposure plus 0 and 25 mg testosterone (P < 0.05) (Fig. 5, B and C). Dnmt3L expression was unchanged after in utero Vinclozolin treatment plus 0 mg testosterone but significantly increased after in utero Vinclozolin plus 25 mg testosterone (P < 0.05) (Fig. 5D). With the exception of Dnmt1 (Fig. 5E), protein expression was indicative of Dnmt mRNA expression (Fig. 5, E–H).
Figure 5.
Dnmt mRNA and protein expression in the VP. Relative mRNA expression after in utero Vinclozolin exposure and exposed at puberty to 0, 5, or 25 mg testosterone, Dnmt1 (A), Dnmt3A (B), Dnmt3B (C), and Dnmt3L (D) normalized to β-actin. Relative protein expression after in utero Vinclozolin exposure and exposed at puberty to 0 or 25 mg testosterone, Dnmt1 (E), Dnmt3A (F), Dnmt3B (G), and Dnmt3L (H) was normalized to GAPDH. Control (open bar) and Vinclozolin (solid bar) (mean ± sem, n = 4–5 in duplicate). *, Significance vs. matched testosterone-treated control group (P < 0.05).
Testis
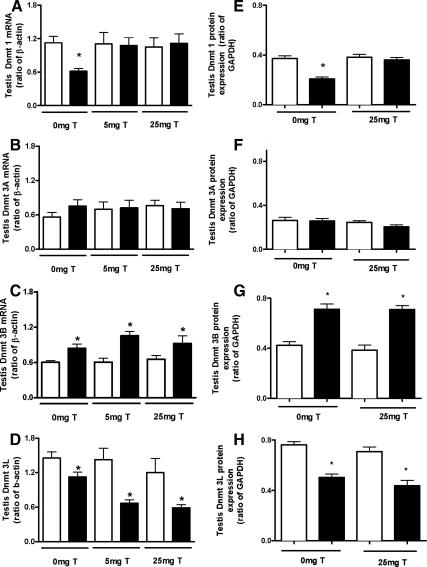
No significant difference in Dnmt3A (Fig. 6B) expression was observed in the testis of Vinclozolin-treated tissues compared with control, but similar to the prostate, a significant increase (P < 0.05) in Dnmt3B (Fig. 6C) expression was observed. In contrast, a significant reduction (P < 0.05) in the expression of both Dnmt1 (Fig. 6A) and Dnmt3L (Fig. 6D) was evident in the testis of Vinclozolin-treated offspring compared with vehicle control. Treatment of rats with 25 mg of testosterone at puberty normalized Dnmt1 expression to that of nontreated controls and did not alter Dnmt3A expression. Testosterone treatment did not restore testicular Dnmt3B and Dnmt3L mRNA expression. Protein expression was indicative of Dnmt mRNA expression (Fig. 6, E–H).
Figure 6.
DNA methyltransferase mRNA and protein expression in the testis. Relative mRNA expression following in utero Vinclozolin exposure and exposed at puberty to 0, 5, or 25 mg testosterone, Dnmt1 (A), Dnmt3A (B), Dnmt3B (C), and Dnmt3L (D) normalized to β-Actin. Relative protein expression following in utero Vinclozolin exposure and exposed at puberty to 0 or 25 mg testosterone, Dnmt1 (E), Dnmt3A (F), Dnmt3B (G), and Dnmt3L (H) normalized to GAPDH. Control (open bar) and Vinclozolin (solid bar). Mean ± SEM (n = 4-5 in duplicate). *, Significance vs. matched testosterone-treated control group (P < 0.05).
Dnmt3L knockout mice
Webster et al. (18) demonstrated that Dnmt3L knockout mice have a testicular phenotype with germ cell arrest and apoptosis of spermatocytes, confirming a requirement for Dnmt3L in germ cell development. The lack of altered Dnmt3L expression in the prostate after in utero Vinclozolin exposure suggests that this Dnmt is not required for normal adult prostate development, nor is it involved in the development of prostatitis. To further assess the role of DNTM3L in the VP, DNTM3L knockout mice were examined (18). Ventral prostates from adult (2–3 months, n = 3) and aged (9–14 months, n = 5) wild-type and knockout animals were examined. There was a significant increase in total body weight in the Dnmt3L knockout mice aged 2–3 months, whereas no differences in VP weight were apparent (supplemental Table S1, published as supplemental data on The Endocrine Society’s Journals Online web site at http://endo.endojournals. org). Histological analysis of VP specimens revealed no overt aberrations or any evidence of inflammatory lesions (supplemental Fig. S1). Unbiased stereological analysis revealed no significant differences in epithelial, stromal, or luminal volume (supplemental Fig. S2A) proliferative activity (supplemental Fig. S2B) or AR expression (supplemental Fig. S2C) in knockout prostates compared with wild-type controls at any age. These results confirm Dnmt3L is not a crucial regulator in the prostate.
Discussion
Altered androgen signaling before puberty has the potential to predispose the adult prostate to abnormal growth and disease. The antiandrogenic activity of the EDC Vinclozolin has been previously demonstrated as a causative factor in the etiology of early-onset prostatitis in the rat. This occurs concurrently with a down-regulation of AR and heightened activity of NF-κB (9). The current study sought to establish whether restoration of AR-mediated androgen signaling could be achieved by administration of high-dose exogenous testosterone consequently suppressing NF-κB and the development of postpubertal prostatitis. In addition, we examined other reproductive abnormalities previously associated with in utero exposure to Vinclozolin, AGD, and concentration of elongated spermatids.
The current investigation demonstrates that 4 wk treatment with supraphysiological levels of testosterone (25 mg) over the pubertal period prevents the onset of prostate inflammation coincident with a significant reduction in NF-κB activation. An increase in AR expression was observed, confirming current literature reports of an inverse correlation between AR and NF-κB activity (35,36,37). A majority of prostatitis cases, approximately 95%, are nonbacterial in origin, precluding the use of antibiotics as targeted therapy (38). Whereas our current study demonstrates that pubertal androgen treatment prevents the onset of in utero EDC-induced prostatitis, the exact mechanisms by which testosterone/AR activity inhibits NF-κB activation remain unclear. Whereas several inflammatory genes in the NF-κB signaling pathway have been identified to have androgen response elements (AREs) in their promoter, currently no AREs have been identified in the promoter of NF-κB. The identification of functional AREs in the promoter of NF-κB would demonstrate direct regulation by androgens.
Steroid hormones have long been recognized to be essential modulators of a variety of cellular processes in normal physiology. Androgens are critical for normal prostate differentiation and development, primarily mediating their effects in target cells by interacting with the AR. There is evidence to show cross talk between AR and NF-κB, yet the regulatory mechanisms remain unclear (39). Furthermore, there is increasing evidence to suggest that NF-κB activation plays an important role in the development and progression of human malignancies (reviewed in Refs. 40,41,42). Alterations in NF-κB regulated genes has been associated with leukemia and lymphoma, and there is increasing interest in the possible role of NF-κB in prostate cancer initiation and/or progression because NF-κB is highly up-regulated in prostate cancer, promoting proliferation and inhibiting apoptosis (43,44). It is interesting to note that in prostate cancer there is an inverse correlation between AR expression and NF-κB activity (45,46,47). Transient cotransfection studies indicate mutual transcriptional antagonism for AR and NF-κB, and it is speculated that compounds that activate NF-κB may interfere with normal androgen signaling, resulting in androgen insensitivity in tissues in which AR is normally expressed (36,48). It is therefore reasonable to postulate a hormonally regulated mechanism of EDC-induced prostatitis whereby persistent down-regulation of AR signaling induced by Vinclozolin results in activation of NF-κB and the emergence of prostatitis.
Chronic inflammation is associated with approximately 20% of all human cancers (49,50) and NF-κB has been shown to be constitutively active in various human malignancies and has been identified as an important endogenous tumor promoter (36,37,51). Furthermore, prostate cancer cells have been reported to have constitutive NF-κB activity with an inverse correlation with AR status. Whereas no pre- or malignant lesions were observed in EDC-exposed offspring, the disruption of normal AR activity and the close relationship between chronic inflammation and cancer suggests that in utero EDC exposure can cause disruption of the homeostatic interactions, which disrupt stromal-epithelial signaling and may eventually lead to malignant transformation on aging. Recent studies demonstrated a connection between the sex steroid hormones, inflammation, and cancer, providing the first link between these two classic tumor-promoting pathways (52). Further investigation into the link between inflammation and cancer and its implications for hormonal therapy resistance could have huge implications for therapy, given the widespread use of selective AR modulators in the treatment of prostate cancer.
Normalization of AGD and concentration of elongated spermatids in in utero Vinclozolin-treated rats was also observed after pubertal androgen treatment. Previous work has assumed permanent and irreversible effects of EDCs, but the current study shows high-dose testosterone abolished Vinclozolin-induced postpubertal prostatitis, normalized AGD, and restored the concentration of elongated spermatids, although prostate weight overall was unaltered. These observations support the hypothesis that Vinclozolin induced effects on AGD, elongated spermatids, and prostatitis are reversible.
In addition to hormonal alterations, EDCs have the potential to induce disease through altering normal epigenetic mechanisms of gene regulation, such as DNA methylation, histone modification, and chromatin remodeling. Four mammalian Dnmts are responsible for DNA methylation pattern acquisition during gametogenesis, embryogenesis, and somatic tissue development. The current data demonstrate that exposure to Vinclozolin in utero, during a specific period of male reproductive tract development, results in adult-onset disease in the prostate and testis, which are associated with changes in Dnmt expression. Our study extends previous observations by Anway et al. (10,11) that demonstrated EDC exposure during gonadal sex determination (GD8-15) was associated with alterations in Dnmt expression in the testis and prostate leading to transgeneration effects. Changes in Dnmt3A and Dnmt3B but not Dnmt1 and Dnmt3L in the prostate of rats exposed in utero to Vinclozolin suggest alterations in Dnmt3A and Dnmt3B may contribute to the initiation and progression of Vinclozolin-induced postpubertal prostatitis. Alterations in Dnmt1 expression have been shown to contribute to both prostate cancer initiation and promotion (53,54). The absence of an alteration in Dnmt1 suggests that the prostatic inflammation observed after in utero Vinclozolin exposure is not likely to be a premalignant lesion. Furthermore, this suggests that alterations in Dnmt3A and Dnmt3B may contribute to the initiation and progression of antiandrogen-induced adult-onset prostatitis. However, supraphysiological doses of testosterone, prevented prostatitis, whereas Dnmt3A and Dnmt3B expression remained unchanged.
In contrast to the prostate, in utero Vinclozolin exposure is associated with significant reductions in Dnmt1 and Dnmt3L expression and a significant increase in Dnmt3B expression in the adult testis. The increased germ cell apoptosis coinciding with reductions in Dnmt3L and Elongated spermatid number is similar to the phenotype observed in Dnmt3L knockout mice (18). Thus, our testis data support previous observations by Webster et al. (18) and Anway et al. (10), suggesting that lack of Dnmt3L impacts on male fertility. However, administration of high-dose testosterone abolished germ cell apoptosis and restored Elongated sperm number despite alterations in Dnmt3B and Dnmt3L expression. Therefore, alterations in Dnmt expression in the prostate and testis occur concurrently with hormone-regulated changes but are irreversible and may therefore to contribute to the transgenerational phenotypes reported previously (10,11,12,17).
Our study was limited to the assessment of the effects of in utero exposure to Vinclozolin and is therefore not a transgenerational study. However, two recent independent studies were conducted to ascertain whether Vinclozolin causes transgenerational effects on the male reproductive system (13,14). In contrast to previous studies (10,11,12), no transgenerational effects were observed and both papers concluded that administration of Vinclozolin in utero during GD8–15 did not cause transgenerational male reproductive abnormalities or change DNA methylation status in those rats exposed in utero to Vinclozolin or subsequent generations (13,14).
Our data demonstrate for the first time that in utero Vinclozolin induced reduced AGD, increased germ cell apoptosis, reduced concentration of elongated spermatids and postpubertal prostatitis are hormonally regulated and reversible by administration of high-dose testosterone at puberty. Despite altered Dnmt expression indicative of epigenetic effects of Vinclozolin exposure, these results show this is not the primary mechanism by which Vinclozolin induces the male reproductive tract abnormalities described herein. Our results therefore support the hypothesis that the primary mechanism of action of Vinclozolin is as an AR antagonist.
Acknowledgments
We thank Ms. S. Hedwards for skilled technical assistance and Dr. S. McPherson (Monash University) for insightful discussions.
Footnotes
This work was supported by the U.S. Army Department of Defense Prostate Cancer Research Program Exploration-Hypothesis Development Award W81XWH-07-1-0126 (to G.P.R.) and supported in part by the Intramural Research Program of the National Institutes of Health, National Institute of Environmental Health Sciences (to P.M.D.F.) and National Health and Medical Research Council (NHMRC) fellowships (171601 and 461204), NHMRC program grant (to H.S.S.).
Current address for P.A.C.: Peter MacCallum Cancer Centre, Centre for Cancer Genomics and Predictive Medicine, East Melbourne, Victoria, Australia.
Disclosure Summary: The authors have nothing to declare.
First Published Online January 7, 2010
Abbreviations: AGD, Anogenital distance; AR, androgen receptor; ARE, androgen response element; bw, body weight; Dnmt, DNA methyltransferase; EDC, endocrine-disrupting chemical; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; GD, gestational day; H&E, hematoxylin and eosin; NF-κB, nuclear factor-κB; PND, postnatal day; VP, ventral prostate.
References
- Colborn T, Dumanoski D, Myers JP 1996 Our stolen future. Are we threatening our fertility, intelligence and survival? A scientific detective story. Boston: Little, Brown and Co. [Google Scholar]
- Carlsen E, Giwercman A, Keiding N, Skakkebaek NE 1992 Evidence for decreasing quality of semen during past 50 years. Br Med J 305:609–613 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weir HK, Thun MJ, Hankey BF, Ries LA, Howe HL, Wingo PA, Jemal A, Ward E, Anderson RN, Edwards BK 2003 Annual report to the nation on the status of cancer, 1975–2000, featuring the uses of surveillance data for cancer prevention and control. J Natl Cancer Inst 95:1276–1299 [DOI] [PubMed] [Google Scholar]
- Kelce WR, Gray L, Wilson EM 1998 Antiandrogens as environmental endocrine disruptors. Reprod Fertil Dev 10:105–111 [DOI] [PubMed] [Google Scholar]
- Kelce WR, Monosson E, Gamcsik MP, Laws SC, Gray Jr LE 1994 Environmental hormone disruptors: evidence that vinclozolin developmental toxicity is mediated by antiandrogenic metabolites. Toxicol Appl Pharmacol 126:276–285 [DOI] [PubMed] [Google Scholar]
- Kelce WR, Wilson EM 1997 Environmental antiandrogens: developmental effects, molecular mechanisms, and clinical implications. J Mol Med 75:198–207 [DOI] [PubMed] [Google Scholar]
- Gray Jr LE, Wolf C, Lambright C, Mann P, Price M, Cooper RL, Ostby J 1999 Administration of potentially antiandrogenic pesticides (procymidone, linuron, iprodione, chlozolinate, p,p′-DDE, and ketoconazole) and toxic substances (dibutyl- and diethylhexyl phthalate, PCB 169, and ethane dimethane sulphonate) during sexual differentiation produces diverse profiles of reproductive malformations in the male rat. Toxicol Ind Health 15:94–118 [DOI] [PubMed] [Google Scholar]
- Wolf CJ, LeBlanc GA, Ostby JS, Gray Jr LE 2000 Characterization of the period of sensitivity of fetal male sexual development to vinclozolin. Toxicol Sci 55:152–161 [DOI] [PubMed] [Google Scholar]
- Cowin PA, Foster P, Pedersen J, Hedwards S, McPherson SJ, Risbridger GP 2008 Early onset endocrine disruptor induced prostatitis in the rat. Environ Health Perspect 116:923–929 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anway MD, Rekow SS, Skinner MK 2008 Transgenerational epigenetic programming of the embryonic testis transcriptome. Genomics 91:30–40 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anway MD, Skinner MK 2008 Transgenerational effects of the endocrine disruptor vinclozolin on the prostate transcriptome and adult onset disease. Prostate 68:517–529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anway MD, Cupp AS, Uzumcu M, Skinner MK 2005 Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 308:1466–1469 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider S, Kaufmann W, Buesen R, van Ravenzwaay B 2008 Vinclozolin—the lack of a transgenerational effect after oral maternal exposure during organogenesis. Reprod Toxicol 25:352–360 [DOI] [PubMed] [Google Scholar]
- Inawaka K, Kawabe M, Takahashi S, Doi Y, Tomigahara Y, Tarui H, Abe J, Kawamura S, Shirai T 2009 Maternal exposure to anti-androgenic compounds, vinclozolin, flutamide and procymidone, has no effects on spermatogenesis and DNA methylation in male rats of subsequent generations. Toxicol Appl Pharmacol 237:178–187 [DOI] [PubMed] [Google Scholar]
- Chang HS, Anway MD, Rekow SS, Skinner MK 2006 Transgenerational epigenetic imprinting of the male germline by endocrine disruptor exposure during gonadal sex determination. Endocrinology 147:5524–5541 [DOI] [PubMed] [Google Scholar]
- 2009 Retraction. Transgenerational epigenetic imprinting of the male germline by endocrine disruptor exposure during gonadal sex determination. Endocrinology 150:2976 [DOI] [PubMed] [Google Scholar]
- Anway MD, Leathers C, Skinner MK 2006 Endocrine disruptor vinclozolin induced epigenetic transgenerational adult-onset disease. Endocrinology 147:5515–5523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webster KE, O'Bryan MK, Fletcher S, Crewther PE, Aapola U, Craig J, Harrison DK, Aung H, Phutikanit N, Lyle R, Meachem SJ, Antonarakis SE, de Kretser DM, Hedger MP, Peterson P, Carroll BJ, Scott HS 2005 Meiotic and epigenetic defects in Dnmt3L-knockout mouse spermatogenesis. Proc Natl Acad Sci USA 102:4068–4073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korenbrot CC, Huhtaniemi IT, Weiner RI 1977 Preputial separation as an external sign of pubertal development in the male rat. Biol Reprod 17:298–303 [DOI] [PubMed] [Google Scholar]
- Stoker TE, Laws SC, Guidici DL, Cooper RL 2000 The effect of atrazine on puberty in male Wistar rats: an evaluation in the protocol for the assessment of pubertal development and thyroid function. Toxicol Sci 58:50–59 [DOI] [PubMed] [Google Scholar]
- Meachem SJ, Wreford NG, Robertson DM, McLachlan RI 1997 Androgen action on the restoration of spermatogenesis in adult rats: effects of human chorionic gonadotrophin, testosterone and flutamide administration on germ cell number. Int J Androl 20:70–79 [DOI] [PubMed] [Google Scholar]
- Balanathan P, Ball EM, Wang H, Harris SE, Shelling AN, Risbridger GP 2004 Epigenetic regulation of inhibin α-subunit gene in prostate cancer cell lines. J Mol Endocrinol 32:55–67 [DOI] [PubMed] [Google Scholar]
- Bianco JJ, Handelsman DJ, Pedersen JS, Risbridger GP 2002 Direct response of the murine prostate gland and seminal vesicles to estradiol. Endocrinology 143:4922–4933 [DOI] [PubMed] [Google Scholar]
- Laczkó I, Hudson DL, Freeman A, Feneley MR, Masters JR 2005 Comparison of the zones of the human prostate with the seminal vesicle: morphology, immunohistochemistry, and cell kinetics. Prostate 62:260–266 [DOI] [PubMed] [Google Scholar]
- Wang Y, Hayward S, Cao M, Thayer K, Cunha G 2001 Cell differentiation lineage in the prostate. Differentiation 68:270–279 [DOI] [PubMed] [Google Scholar]
- Bai S, Ghoshal K, Datta J, Majumder S, Yoon SO, Jacob ST 2005 DNA methyltransferase 3b regulates nerve growth factor-induced differentiation of PC12 cells by recruiting histone deacetylase 2. Mol Cell Biol 25:751–766 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Gold E, Jetly N, O'Bryan MK, Meachem S, Srinivasan D, Behuria S, Sanchez-Partida LG, Woodruff T, Hedwards S, Wang H, McDougall H, Casey V, Niranjan B, Patella S, Risbridger G 2009 Activin C antagonizes activin A in vitro and overexpression leads to pathologies in vivo. Am J Pathol 174:184–195 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bertram JF 1995 Analyzing renal glomeruli with the new stereology. Int Rev Cytol 161:111–172 [DOI] [PubMed] [Google Scholar]
- McPherson SJ, Wang H, Jones ME, Pedersen J, Iismaa TP, Wreford N, Simpson ER, Risbridger GP 2001 Elevated androgens and prolactin in aromatase deficient (ArKO) mice cause enlargement but not malignancy of the prostate gland. Endocrinology 142:2458–2467 [DOI] [PubMed] [Google Scholar]
- Meachem SJ, McLachlan RI, de Kretser DM, Robertson DM, Wreford NG 1996 Neonatal exposure of rats to recombinant follicle stimulating hormone increases adult Sertoli and spermatogenic cell numbers. Biol Reprod 54:36–44 [DOI] [PubMed] [Google Scholar]
- Singh J, Handelsman DJ 1999 Morphometric studies of neonatal estrogen imprinting in the mature mouse prostate. J Endocrinol 162:39–48 [DOI] [PubMed] [Google Scholar]
- Robb GW, Amann RP, Killian GJ 1978 Daily sperm production and epididymal sperm reserves of pubertal and adult rats. J Reprod Fertil 54:103–107 [DOI] [PubMed] [Google Scholar]
- McLachlan RI, O'Donnell L, Meachem SJ, Stanton PG, de K, Pratis K, Robertson DM 2002 Hormonal regulation of spermatogenesis in primates and man: insights for development of the male hormonal contraceptive. J Androl 23:149–162 [PubMed] [Google Scholar]
- Matthiesson KL, McLachlan RI, O'Donnell L, Frydenberg M, Robertson DM, Stanton PG, Meachem SJ 2006 The relative roles of follicle-stimulating hormone and luteinizing hormone in maintaining spermatogonial maturation and spermiation in normal men. J Clin Endocrinol Metab 91:3962–3969 [DOI] [PubMed] [Google Scholar]
- Itoh Y, Hayashi H, Xu J, Takii T, Miyazawa K, Ariga H, Akahoshi T, Waguri-Nagaya Y, Otsuka T, Okamoto T, Onozaki K 2007 Dihydrotestosterone inhibits tumor necrosis factor α induced interleukin-1α mRNA expression in rheumatoid fibroblast-like synovial cells. Biol Pharm Bull 30:1140–1143 [DOI] [PubMed] [Google Scholar]
- McKay LI, Cidlowski JA 1999 Molecular control of immune/inflammatory responses: interactions between nuclear factor-κB and steroid receptor-signaling pathways. Endocr Rev 20:435–459 [DOI] [PubMed] [Google Scholar]
- Suh J, Rabson AB 2004 NF-κB activation in human prostate cancer: important mediator or epiphenomenon? J Cell Biochem 91:100–117 [DOI] [PubMed] [Google Scholar]
- Litwin M, Saigal C 2007 Urologic diseases in America. Bethesda, MD: National Institutes of Health [Google Scholar]
- De Bosscher K, Vanden Berghe W, Haegeman G 2006 Cross-talk between nuclear receptors and nuclear factor κB. Oncogene 25:6868–6886 [DOI] [PubMed] [Google Scholar]
- Baldwin AS 2001 Control of oncogenesis and cancer therapy resistance by the transcription factor NF-κB. J Clin Invest 107:241–246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karin M, Cao Y, Greten FR, Li ZW 2002 NF-κB in cancer: from innocent bystander to major culprit. Nat Rev Cancer 2:301–310 [DOI] [PubMed] [Google Scholar]
- Rayet B, Gélinas C 1999 Aberrant rel/nfkb genes and activity in human cancer. Oncogene 18:6938–6947 [DOI] [PubMed] [Google Scholar]
- Gasparian AV, Yao YJ, Kowalczyk D, Lyakh LA, Karseladze A, Slaga TJ, Budunova IV 2002 The role of IKK in constitutive activation of NF-κB transcription factor in prostate carcinoma cells. J Cell Sci 115:141–151 [DOI] [PubMed] [Google Scholar]
- Ghosh S, Karin M 2002 Missing pieces in the NF-κB puzzle. Cell 109(Suppl):S81–S96 [DOI] [PubMed] [Google Scholar]
- Altuwaijri S, Lin HK, Chuang KH, Lin WJ, Yeh S, Hanchett LA, Rahman MM, Kang HY, Tsai MY, Zhang Y, Yang L, Chang C 2003 Interruption of nuclear factor κB signaling by the androgen receptor facilitates 12-O-tetradecanoylphorbolacetate-induced apoptosis in androgen-sensitive prostate cancer LNCaP cells. Cancer Res 63:7106–7112 [PubMed] [Google Scholar]
- Shukla S, Maclennan GT, Marengo SR, Resnick MI, Gupta S 2005 Constitutive activation of PI3K-Akt and NF-κB during prostate cancer progression in autochthonous transgenic mouse model. Prostate 64:224–239 [DOI] [PubMed] [Google Scholar]
- Supakar PC, Jung MH, Song CS, Chatterjee B, Roy AK 1995 Nuclear factor-κB functions as a negative regulator for the rat androgen receptor gene and NF-κB activity increases during the age-dependent desensitization of the liver. J Biol Chem 270:837–842 [DOI] [PubMed] [Google Scholar]
- McKay LI, Cidlowski JA 1998 Cross-talk between nuclear factor-κB and the steroid hormone receptors: mechanisms of mutual antagonism. Mol Endocrinol 12:45–56 [DOI] [PubMed] [Google Scholar]
- Ames BN, Gold LS, Willett WC 1995 The causes and prevention of cancer. Proc Natl Acad Sci USA 92:5258–5265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coussens LM, Werb Z 2002 Inflammation and cancer. Nature 420:860–867 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mantovani A, Allavena P, Sica A, Balkwill F 2008 Cancer-related inflammation. Nature 454:436–444 [DOI] [PubMed] [Google Scholar]
- Mantovani A 2007 Cancer: an infernal triangle. Nature 448:547–548 [DOI] [PubMed] [Google Scholar]
- Esteller M 2005 Aberrant DNA methylation as a cancer-inducing mechanism. Annu Rev Pharmacol Toxicol 45:629–656 [DOI] [PubMed] [Google Scholar]
- Feinberg AP, Ohlsson R, Henikoff S 2006 The epigenetic progenitor origin of human cancer. Nat Rev Genet 7:21–33 [DOI] [PubMed] [Google Scholar]