Abstract
Context: Vitamin D status can influence insulin resistance.
Objective: The aim of the study was to determine the prevalence of vitamin D deficiency in obese African-American (AA) adolescent females in a southeastern latitude and to determine the relationship of 25-hydroxyvitamin D [25(OH)D] with insulin and glucose dynamics.
Design: We conducted a cross-sectional study in a University Children’s Hospital.
Methods: Serum 25(OH)D, fasting glucose, PTH, serum calcium, serum lipids, serum transaminases, and C-reactive protein were assessed. Indices of insulin sensitivity and resistance were determined from an oral glucose tolerance test. Subjects were classified as vitamin D deficient or sufficient, based on the traditional vitamin D deficiency definition [serum 25(OH)D <20 ng/ml] and also by a lower 25(OH)D cut-point of 15 ng/ml or less.
Results: A total of 51 AA adolescent females (body mass index, 43.3 ± 9.9 kg/m2; age, 14 ± 2 yr) were studied. Serum 25(OH)D concentrations were 20 ng/ml or less in 78.4% and 15 ng/ml or less in 60.8% of subjects. There were no significant group differences in the metabolic outcomes when subjects were classified using the traditional vitamin D deficiency definition. The Matsuda index of insulin sensitivity was significantly lower (P = 0.02), and insulin area under the curve was significantly higher (P = 0.04) in subjects with 25(OH)D concentrations of 15 ng/ml or less vs. those with higher concentrations.
Conclusions: Vitamin D deficiency is highly prevalent in obese, AA female adolescents and may promote insulin resistance. Our data suggest that a 25(OH)D concentration of 15 ng/ml or less may be the threshold by which vitamin D deficiency confers negative effects on insulin sensitivity.
The results suggest that 25(OH)D concentrations ≤15 ng/mL may be the threshold by which vitamin D deficiency confers negative effects on insulin sensitivity.
Adiposity is inversely related to circulating serum 25-hydroxyvitamin D [25(OH)D] (1). Accordingly, vitamin D deficiency is highly prevalent among obese children and adolescents (2,3,4). Darker skin pigmentation is also inversely associated with circulating 25(OH)D (5,6); thus it may be expected that obese African-Americans (AA) are especially at high risk of vitamin D deficiency compared with Caucasian-Americans (CA) (7). The prevalence of vitamin D deficiency [defined as serum 25(OH)D ≤20 ng/ml] among obese AA children and adolescents residing in northern latitudes was shown to be 57% in a 40° N latitude, Pennsylvania (3) and 48.7% in a 43° N latitude, Wisconsin (4). The prevalence of vitamin D deficiency among obese AA adolescents in southern latitudes, geographically a region in the United States with a high prevalence of type 2 diabetes, is unknown.
In addition to greater prevalence of vitamin D deficiency, AA have elevated insulin resistance (8,9) and, subsequently, greater risk for type 2 diabetes mellitus (10) than CA. Vitamin D has been postulated to play a role in glucose homeostasis, pancreatic β-cell function, and insulin sensitivity (11). In vitro, vitamin D enhances insulin-stimulated glucose transport and up-regulates transcription of the insulin receptor gene (12,13). Cross-sectional (4,14,15,16,17) and prospective (18) studies also support the role of vitamin D in type 2 diabetes mellitus risk prevention. The relationship between 25(OH)D and insulin action/glucose metabolism in a pediatric population has only been assessed in one previous study (4) that used surrogate indices of insulin sensitivity based on fasting measures of insulin and glucose (19).
Serum C-reactive protein (CRP) has a role in prognostic information to the National Cholesterol Education Program Adult Treatment Panel III definition of metabolic syndrome, and there are emerging data from experimental studies in animals and humans on the etiological role of subclinical inflammation in the pathogenesis of type 2 diabetes mellitus, primarily as a mediator of obesity-induced insulin resistance (20). It is not clear whether vitamin D has any effect on inflammation or on fatty liver, crucial components of metabolic syndrome.
The primary aim of the present study was to determine the prevalence of subclinical vitamin D deficiency in obese AA adolescents residing in the southeastern region of the United States. A secondary aim was to assess the relationship of serum 25(OH)D status to insulin sensitivity measures based on dynamic changes in glucose and insulin concentrations after an oral glucose tolerance test (OGTT). Furthermore, we examined both 15 and 20 ng/ml as potential 25(OH)D benchmarks for vitamin D deficiency as related to disturbances in glucose metabolism. We also examined the relationship of serum 25(OH)D to serum transaminases and CRP.
Subjects and Methods
Subjects
This study involved obese, AA adolescent females attending the weight management clinic at the Children’s Hospital, University of Alabama at Birmingham (UAB) (latitude 33° N). Race and ethnicity were reported by the subject’s parent. Because insulin resistance is affected by pubertal status, only subjects who were menarchal and in Tanner stage 4 or 5 for breast and pubic hair development (21) were included. Exclusion criteria were: diagnosis of overt diabetes, malabsorptive disorders, or metabolic rickets; use of diuretics, systemic glucocorticoids, or anticonvulsant medications; and body mass index (BMI; kilograms/meter2) below the 95th centile for age and sex according to the Centers for Disease Control and Prevention growth charts (22). This study was approved by the UAB Institutional Review Board for Human Use, and written parent consent and subject assent were obtained before all testing.
Protocol
Anthropometric evaluation, including weight, height, and BMI, was performed on each patient, and pubertal development was assessed according to the method of Marshall and Tanner (21). All participants underwent an OGTT as follows: after a fasting sample for measurement of plasma glucose and insulin, a standard flavored glucose dose (1.75 g/kg of body weight up to a maximum of 75 g) was given orally over 5 min, and measurements of plasma glucose and insulin were drawn at 30-, 60-, and 120-min intervals. Patients also had serum 25(OH)D, intact PTH, serum calcium, hemoglobin A1c (HbA1c), serum CRP, and transaminase [aspartate aminotransferase (AST) and alanine aminotransferase (ALT)] measurements with the baseline sampling. All blood samples were obtained between 0800 and 1200 h.
Laboratory analyses
Plasma glucose concentration was determined using the glucose oxidase method, using the Vitros 5.1 (Ortho Clinical Diagnostics, Rochester, NY). The coefficients of variation of the assay are 1.5% at a glucose level of 83 mg/dl and 1.2% at a glucose level of 292 mg/dl. Serum insulin was measured with two-site immunoenzymometric assay (AIA-PACK IRI; Tosoh, Tokyo, Japan). The 25(OH)D assays were obtained commercially (Quest Diagnostics, Nichols Institute, San Juan Capistraho, CA), using liquid chromatography-tandem mass spectrometry (LC-MS/MS) methodology, which measures both 25(OH)D2 and 25(OH)D3. LC-MS/MS is considered the reference standard for 25(OH)D assay (23). Values are reported as total serum 25(OH)D [including 25(OH)D2 and 25(OH)D3]. Serum PTH was determined with a two-site immunoradiometric assay that detects intact PTH (1–84) and the amino-terminally truncated PTH (7–84) fragments (normal range, 10–65 pg/ml). CRP was measured with Vitros Chemistry Systems (Ortho Clinical Diagnostics) using a “MicroSlide” method through the hospital laboratory.
Assessment of insulin resistance/sensitivity
The whole body insulin sensitivity index (WBISI) was calculated during the OGTT, as proposed by Matsuda et al. (24), where WBISI = 10,000/√(fasting glucose × fasting insulin) × (mean glucose × mean insulin during OGTT). WBISI was validated in obese children and was found to correlate (r = 0.78; P < 0.0005) with insulin sensitivity derived from the hyperinsulinemic-euglycemic clamp (25,26). The homeostatic model assessment of basal insulin resistance (HOMA-IR) was calculated using the formula: HOMA-IR = [fasting insulin (μU/ml) × fasting glucose (mmol/liter)]/22.5 (26,27). Post-OGTT area under the curve (AUC) and incremental AUC for insulin and glucose were calculated using the trapezoidal method (28,29).
Statistical analysis
Descriptive statistical analysis was used for demographic and baseline information. Results are expressed as means ± sd. Two-group t tests were used to determine statistical differences in continuous variables between groups divided on the basis of a traditional 25(OH)D cutoff value of less than 20 ng/ml to define vitamin D deficiency (30,31). When the subjects were categorized using less than 20 ng/ml as the vitamin D deficiency cutoff, there were no statistical differences in any outcome variables. Some argue that vitamin D deficiency should be defined as serum 25(OH)D of 15 ng/ml or less (32). Therefore, we performed analyses on subjects divided into groups based on the lower vitamin D threshold (<15 ng/ml) as a post hoc amendment. Analysis of covariance was used to determine group differences after adjustment for BMI and PTH separately. Pearson correlation coefficient analysis assessed correlations between continuous variables, and partial correlation coefficient analysis was used to determine relationships after adjusting for relevant covariates. Fasting glucose and WBISI deviated from a normal distribution and were, therefore, log10-transformed before analyses. Statistical analyses were performed using the SAS software package (version 9.1; SAS Institute, Cary, NC). A two-sided P value ≤0.05 was considered statistically significant.
Results
A total of 51 AA adolescent females were studied during a period from March 2007 to October 2008. Descriptive characteristics of the subjects are presented in Table 1. Serum 25(OH)D concentration was less than 20 ng/ml in 40 subjects (78.4%), and 31 subjects (60.8%) had 25(OH)D concentrations of 15 ng/ml or less. A two-group t test showed no significant difference (P = 0.51) between summer and winter 25(OH)D concentrations (15.31 ± 6.5 vs. 13.67 ± 10.17 ng/ml) for summer and winter, respectively.
Table 1.
Descriptive characteristics of entire sample
| Mean ± sd (n = 51) | |
|---|---|
| Age (yr) | 14 ± 2 |
| Weight (kg) | 114.3 ± 26.1 |
| Height (cm) | 161.5 ± 12.6 |
| BMI (kg/m2) | 43.3 ± 9.9 |
| Fasting insulin (μU/ml) | 34.3 ± 14.2 |
| Fasting glucose (mg/dl) | 88.6 ± 11.7 |
| 2-h insulin (μU/ml) | 135.4 ± 93.3 |
| HbA1c (%)a | 5.5 ± 0.4 |
| PTH (pg/ml) | 33.5 ± 15.3 |
| Serum calcium (mg/dl) | 9.40 ± 0.41 |
| 25(OH)D (ng/ml) | 14.92 ± 7.44 |
| Total cholesterol (mg/dl)a | 158.96 ± 23.20 |
| HDL (mg/dl)b | 43.94 ± 8.79 |
| LDL (mg/dl)b | 96.84 ± 24.00 |
| Triglycerides (mg/dl)a | 85.7 ± 44.7 |
| CRP (mg/dl)c | 1.02 ± 0.65 |
| ALT (u/liter) | 25.96 ± 14.80 |
| AST (u/liter) | 29.67 ± 10.55 |
| WBISI (Matsuda index) | 1.89 ± 1.83 |
| HOMA-IR | 1.10 ± 0.55 |
| Glucose AUC (mg/dl × 120 min) | 14,492.94 ± 3,246.17 |
| Incremental glucose AUC (mg/dl × 120 min) | 3,864.71 ± 2,679.92 |
| Insulin AUC (μU/ml × 120 min) | 20,800.53 ± 10,668.84 |
| Incremental insulin AUC (μU/ml × 120 min) | 16,680.84 ± 10,022.77 |
n = 50.
n = 49.
n = 44.
Pearson correlation coefficient analyses revealed a trend toward a significant relationship between 25(OH)D and BMI (r = −0.24; P = 0.087), PTH (r = − 0.24; P = 0.088), serum calcium (r = 0.24; P = 0.087), and fasting glucose (r = −0.24; P = 0.085) (Table 2). After adjustment for BMI, a trend remained for a significant association between 25(OH)D and fasting glucose (r = −0.23; P = 0.10). Serum 25(OH) D was not significantly associated with fasting insulin, 2-h insulin, HbA1c, total cholesterol, high-density lipoprotein (HDL), low-density lipoprotein (LDL), triglycerides, CRP, ALT, AST, WBISI (Matsuda index), or HOMA-IR (Table 2). The serum concentration of PTH was significantly associated with BMI (r = 0.37; P = 0.007). PTH was not significantly associated with other variables.
Table 2.
Pearson correlations of 25(OH)D and PTH with metabolic variables
| 25 (OH)D
|
PTH
|
|||
|---|---|---|---|---|
| r | P value | r | P value | |
| BMI | −0.242 | 0.09 | 0.376 | 0.01 |
| Fasting insulin | −0.074 | 0.61 | 0.118 | 0.41 |
| Fasting glucosea | −0.243 | 0.09 | 0.156 | 0.27 |
| 2-h insulin | −0.114 | 0.43 | 0.062 | 0.67 |
| HbA1cb | −0.022 | 0.88 | −0.138 | 0.34 |
| PTH | −0.241 | 0.09 | ||
| Serum calcium | 0.242 | 0.09 | −0.118 | 0.41 |
| 25(OH)D | −0.241 | 0.09 | ||
| Total cholesterolb | −0.011 | 0.94 | 0.026 | 0.86 |
| HDLc | 0.012 | 0.94 | 0.111 | 0.45 |
| LDLc | 0.037 | 0.80 | 0.061 | 0.68 |
| Triglyceridesb | 0.067 | 0.64 | −0.219 | 0.13 |
| CRPd | 0.032 | 0.84 | −0.150 | 0.33 |
| ALT | −0.046 | 0.75 | −0.044 | 0.76 |
| AST | −0.059 | 0.68 | −0.115 | 0.42 |
| Glucose AUC | −0.158 | 0.27 | −0.090 | 0.53 |
| Incremental glucose AUC | −0.066 | 0.64 | −0.177 | 0.22 |
| Insulin AUC | −0.081 | 0.57 | 0.052 | 0.72 |
| Incremental insulin AUC | −0.074 | 0.61 | 0.035 | 0.81 |
| WBISI (Matsuda index)a | 0.150 | 0.29 | −0.102 | 0.48 |
| HOMA-IR | −0.119 | 0.41 | 0.139 | 0.33 |
Boldface indicates statistical significance or trend towards significance.
Log10-transformed for analyses.
n = 50.
n = 49.
n = 44.
We categorized the subjects using the frequently referenced vitamin D deficiency cutoff (<20 ng/ml). The two-group t test showed no statistical differences in any outcome variables (data not shown). Subsequently, we categorized vitamin D deficiency status as 25(OH)D below 15 ng/ml. Table 3 shows characteristics of subjects based on this cutoff value. The vitamin D-deficient group had significantly higher PTH (P = 0.045). WBISI was significantly lower (P = 0.018), and insulin AUC was significantly higher (P = 0.039) in the vitamin D-deficient group. There was a trend toward greater fasting glucose (P = 0.078), 2-h insulin (P = 0.05), and incremental insulin AUC (P = 0.05) in the vitamin D-deficient group.
Table 3.
Variable characteristics by vitamin D status
| Vitamin D deficient [25(OH)D ≤15 ng/ml] | Vitamin D sufficient [25(OH)D >15 ng/ml] | Pa | |
|---|---|---|---|
| n | 31 | 20 | |
| Age (yr) | 14.5 ± 2.0 | 13.5 ± 2.5 | 0.125 |
| Weight (kg) | 119.3 ± 27.7 | 106.7 ± 21.8 | 0.091 |
| Height (cm) | 163.47 ± 8.46 | 158.37 ± 17.03 | 0.161 |
| BMI (kg/m2) | 45.0 ± 10.5 | 40.5 ± 8.3 | 0.112 |
| Fasting insulin (μU/ml) | 36.5 ± 12.7 | 30.9 ± 16.1 | 0.172 |
| Fasting glucose (mg/dl) | 90.8 ± 12.6 | 85.1 ± 9.2 | 0.078 |
| 2-h insulin (μU/ml) | 155.8 ± 107.4 | 103.7 ± 54.4 | 0.051 |
| HbA1c (%)b | 5.55 ± 0.46 | 5.54 ± 0.33 | 0.959 |
| PTH (pg/ml) | 36.90 ± 16.28 | 28.15 ± 12.17 | 0.045 |
| Serum calcium (mg/dl) | 9.38 ± 0.42 | 9.42 ± 0.40 | 0.743 |
| 25(OH)D (ng/ml) | 10.06 ± 3.44 | 22.45 ± 5.37 | <0.001 |
| Total cholesterol (mg/dl)c | 158.57 ± 26.00 | 159.55 ± 18.85 | 0.885 |
| HDL (mg/dl)d | 44.34 ± 8.56 | 43.35 ± 9.31 | 0.701 |
| LDL (mg/dl)d | 95.69 ± 26.17 | 98.50 ± 21.02 | 0.692 |
| Triglycerides (mg/dl)b | 83.63 ± 41.49 | 88.80 ± 50.10 | 0.693 |
| CRP (mg/dl)e | 1.02 ± 0.64 | 1.01 ± 0.70 | 0.982 |
| ALT (u/liter) | 26.39 ± 17.71 | 25.30 ± 8.96 | 0.838 |
| AST (u/liter) | 30.23 ± 12.75 | 28.80 ± 5.93 | 0.975 |
| Glucose AUC (mg/dl × 120 min) | 15,015.48 ± 3,790.73 | 13,683.00 ± 1,976.257 | 0.202 |
| Incremental glucose AUC (mg/dl × 120 min) | 41,18.71 ± 2,937.30 | 3,471.00 ± 2,238.01 | 0.405 |
| Insulin AUC (μU/ml × 120) | 23,258.72 ± 11,654.71 | 16,990.34 ± 7,741.09 | 0.039 |
| Incremental insulin AUC (μU/ml × 120) | 18,875.04 ± 10,953.84 | 13,279.82 ± 7,407.72 | 0.051 |
| WBISI | 1.50 ± 0.79 | 2.48 ± 2.69 | 0.018 |
| HOMA-IR | 1.19 ± 0.49 | 0.96 ± 0.61 | 0.148 |
Two-group t test.
n = 19 in nondeficient group.
n = 30 in deficient group.
n = 29 in deficient group.
n = 27 and 17 in deficient and nondeficient groups, respectively.
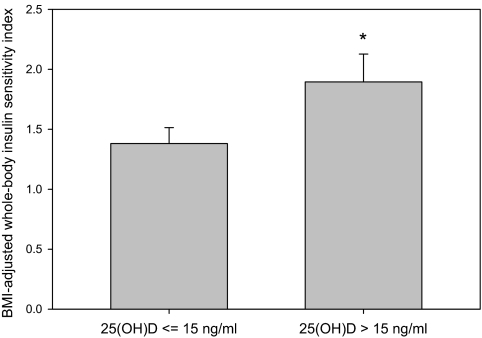
The group difference in WBISI remained after adjustment for BMI (analysis of covariance, P = 0.047) (Fig. 1). Trends toward greater insulin AUC (P = 0.055), incremental insulin AUC (P = 0.062), 2-h insulin (P = 0.08), and fasting glucose (P = 0.093) in the vitamin D-deficient group also remained after adjustment for BMI. Similarly, the group differences in WBISI and insulin AUC remained after adjustment for PTH (P = 0.026 and 0.043, respectively). The trends for group differences in 2-h insulin and incremental insulin AUC remained after adjustment for PTH (P = 0.058 and 0.051, respectively).
Figure 1.
BMI-adjusted WBISI by vitamin D-deficiency status. *, P for difference between groups = 0.047.
Discussion
In this study, we aimed to investigate the prevalence of subclinical vitamin D deficiency in obese AA adolescents and assess the relationship of 25(OH)D to insulin sensitivity and other metabolic outcomes. The prevalence of vitamin D deficiency was 78 and 60% as defined by serum levels below 20 ng/ml or below 15 ng/ml, respectively. Subjects with vitamin D deficiency had significantly lower insulin sensitivity, as assessed with WBISI, than subjects with 25(OH)D concentrations above 15 ng/ml. In addition, there was no relationship between vitamin D status and markers of inflammation, as determined by CRP, or ALT and AST (markers of fatty liver).
Vitamin D deficiency is common in obese patients, and it is possibly a result of decreased vitamin D bioavailability due to sequestration in fat tissue (33), low dietary vitamin D intake due to poor nutritional habits, and minimal sun exposure due to sedentary indoor lifestyle (34). No difference was found in BMI between vitamin D-deficient and -sufficient groups, most likely because most of our subjects are morbidly obese and, hence had limited variability. Having darker skin pigmentation places an obese individual at particularly high risk for vitamin D deficiency due to reduced endogenous vitamin D synthesis. Previous reports of obese AA children and adolescents have not shown a prevalence of vitamin D insufficiency/deficiency to be as striking as our findings. In a cohort of obese (mean BMI, 25.5 ± 4.8 kg/m2) AA preadolescents in the Pittsburgh, Pennsylvania, region (latitude 40° N), the prevalence of serum 25(OH)D of 20 ng/ml or less was 57% (3). In a cohort of obese (mean BMI, 37.1 ± 8.5 kg/m2) children and adolescents in the Milwaukee, Wisconsin, region (latitude 43° N), 48.7% of AA children had vitamin D below 20 ng/ml (70% in winter/fall, 26% in summer/spring). Despite residing in an area that receives relatively more sun year-round (latitude 33° N), our subjects had a vitamin D insufficiency/deficiency prevalence of 78%. Serum 25(OH)D did not differ by season in our study. The greater adiposity of our children (mean BMI, 43.3 ± 93.9 kg/m2) may explain the especially high prevalence of vitamin D deficiency/insufficiency. This highlights the importance of screening for vitamin D even in moderate climate states.
The traditional concept of vitamin D solely as a skeletal vitamin has evolved in recent years. Vitamin D receptors are now recognized to be in numerous extraskeletal peripheral tissues, such as those of the pancreas and muscle (35). In addition, vitamin D deficiency has been implicated in metabolic disease risk, such as type 2 diabetes mellitus (11,36). Previous studies investigating the relationship of insulin sensitivity/resistance, a risk factor for type 2 diabetes mellitus, with 25(OH)D have not found significant associations in AA subjects (4,37). These studies have used indices of insulin sensitivity/resistance derived from fasting values of insulin and glucose, which more accurately reflect hepatic insulin sensitivity as opposed to whole-body insulin sensitivity (19). In our cohort of AA subjects, although we did not find a significant correlation between OGTT-derived WBISI (Matsuda index) and 25(OH)D, subjects with 25(OH)D less than 15 ng/ml had significantly lower WBISI than subjects with higher 25(OH)D concentrations, even after adjusting for BMI. Similar results were found using fasting glucose, 2-h insulin, insulin AUC, and incremental insulin AUC as indices of insulin resistance. In contrast, we did not find this to be the case using HOMA-IR, suggesting that postchallenge indices of insulin sensitivity are more sensitive in determining the detrimental effects of vitamin D deficiency. Direct measures of insulin sensitivity, such as a euglycemic clamp or frequently sampled iv glucose tolerance test, will be needed to confirm results.
Indices for separating hepatic vs. skeletal muscle insulin resistance based on an OGTT have recently become available (38), with hepatic insulin resistance index calculated as the product of the glucose AUC and the insulin AUC for the first 30 min of the OGTT (glucose0–30 AUC × insulin0–30 AUC), and muscle insulin resistance index calculated as the rate of the decline in peak to nadir glucose concentrations divided by mean insulin concentration (dG/dt ÷ mean insulin). Application of the hepatic insulin resistance index yielded similar results as HOMA-IR, such that there were no significant differences between our groups (P = 0.10). This was expected because HOMA-IR has been suggested to be a surrogate for hepatic insulin resistance. Using the equation for muscle insulin sensitivity (dG/dt ÷ mean insulin), subjects with 25(OH)D concentrations below 15 ng/ml had less muscle insulin sensitivity than those with greater 25(OH)D concentrations (P = 0.08). These findings are similar to those obtained with the whole-body index and suggest that vitamin D has more influence on peripheral, rather than hepatic, insulin sensitivity.
Before the appearance of overt symptoms of vitamin D deficiency (rickets in children and osteomalacia in adults), subclinical vitamin D deficiency causes an elevated serum PTH (39). Serum concentrations of 25(OH)D and PTH are typically shown to have an inverse relationship (32). However, we did not find significant Pearson correlations between 25(OH)D and PTH in our subjects. A lack of relationship between serum 25(OH)D concentrations and PTH in AA girls has been reported previously (40). As with our nonskeletal, insulin metabolism outcomes, although correlation analyses provided null results, serum PTH levels were significantly different when our subjects were dichotomized based on a 25(OH)D cutoff of 15 ng/ml.
Vitamin D insufficiency is typically defined as a serum 25(OH)D concentration below 20 ng/ml (<50 nmol/liter) (30,31). Nonetheless, there is no consensus as to the normal limit of serum 25(OH)D, although it is proposed that it is the serum concentration of 25(OH)D at which PTH concentrations are minimized (41). Thus, elevated PTH is considered a surrogate marker of vitamin D deficiency (41). Previous studies have reported elevated PTH concentrations with a serum 25(OH)D level of 15 ng/ml or less (39,42). Hence, some argue that vitamin D deficiency should be defined as a serum 25(OH)D concentration of 15 ng/ml or less (≤37.5 mmol/liter) (32). In our cohort of obese AA adolescents, using 20 ng/ml as the cutoff did not reveal any differences in glucose-insulin dynamics. On the other hand, when we used 15 ng/ml as the cutoff value, subjects with vitamin D deficiency had significantly less whole-body insulin sensitivity (via WBISI by Matsuda Index), greater insulin AUC, and greater PTH. This suggests that 15 ng/ml may be a more appropriate cut point for metabolic outcomes in AA female adolescents. Because so few of the study subjects had 25(OH)D concentrations above 20 ng/ml, we cannot conclude what the true cutoff for vitamin D deficiency is as it relates to either PTH or insulin resistance. However, the lower concentration of 25(OH)D appears to be associated with a higher PTH level and increased insulin resistance. More research is needed to determine whether a 25(OH)D level of less than 15 ng/ml is important for nonskeletal effects of vitamin D deficiency in other ethnicities and ages and in males. A further question needing to be answered is whether there are ethnic differences in vitamin D receptors in AA subjects compared with CA subjects that could result in imbalances in glucose metabolism at different 25(OH)D thresholds.
Strengths of the study include the fact that this is the first comprehensive project in an at-risk adolescent population to assess the prevalence of vitamin D deficiency and also introduce a concept of separate cutoff points for the definition of subclinical vitamin D deficiency. This study also shows that there are extraskeletal consequences of vitamin D deficiency beyond the relationships between adiposity and biochemical vitamin D deficiency. Limitations were that we have not studied CA children. The extreme obesity of our subjects may have been masking any relevant associations between 25(OH)D and metabolic outcomes; thus future research should incorporate lean subjects. Another limitation is the lack of a direct measure of insulin sensitivity, as well as the lack of a direct measure of adiposity, such as dual-energy X-ray absorptiometry. Furthermore, it has to be noted that different 25(OH)D assays yield markedly disparate results (43). Our results are based on 25(OH)D measured using LC-MS/MS.
Conclusion
Vitamin D deficiency is highly prevalent in obese, AA female adolescents residing in the southeastern United States, regardless of the definition used to define vitamin D deficiency. We propose that a serum 25(OH)D concentration of 15 ng/ml may be the more accurate cutoff below which glucose metabolism is dysregulated consequent to vitamin D deficiency. Early recognition and management of vitamin D deficiency may ameliorate the health consequences of obese AA adolescents.
Footnotes
This study is conducted from funds from Children’s Center for Research and Innovation (CCRI grants), Children’s Hospital, Birmingham, Alabama. J.A. is supported by the Alabama Louis Stokes Alliance for Minority Participation and the National Center for Research Resources (National Institutes of Health Grant TL1RR025775).
Disclosure Summary: The authors have nothing to declare.
First Published Online June 23, 2009
Abbreviations: AA, African-Americans; ALT, alanine aminotransferase; AST, aspartate aminotransferase; AUC, area under the curve; BMI, body mass index; CA, Caucasian-American(s); CRP, C-reactive protein; HbA1c, hemoglobin A1c; HDL, high-density lipoprotein; HOMA-IR, homeostatic model assessment of basal insulin resistance; LC-MS/MS, liquid chromatography-tandem mass spectrometry; LDL, low-density lipoprotein; OGTT, oral glucose tolerance test; 25(OH)D, 25-hydroxyvitamin D; WBISI, whole body insulin sensitivity index.
References
- Arunabh S, Pollack S, Yeh J, Aloia JF 2003 Body fat content and 25-hydroxyvitamin D levels in healthy women. J Clin Endocrinol Metab 88:157–161 [DOI] [PubMed] [Google Scholar]
- Smotkin-Tangorra M, Purushothaman R, Gupta A, Nejati G, Anhalt H, Ten S 2007 Prevalence of vitamin D insufficiency in obese children and adolescents. J Pediatr Endocrinol Metab 20:817–823 [DOI] [PubMed] [Google Scholar]
- Rajakumar K, Fernstrom JD, Holick MF, Janosky JE, Greenspan SL 2008 Vitamin D status and response to vitamin D(3) in obese vs. non-obese African American children. Obesity (Silver Spring) 16:90–95 [DOI] [PubMed] [Google Scholar]
- Alemzadeh R, Kichler J, Babar G, Calhoun M 2008 Hypovitaminosis D in obese children and adolescents: relationship with adiposity, insulin sensitivity, ethnicity, and season. Metabolism 57:183–191 [DOI] [PubMed] [Google Scholar]
- Armas LA, Dowell S, Akhter M, Duthuluru S, Huerter C, Hollis BW, Lund R, Heaney RP 2007 Ultraviolet-B radiation increases serum 25-hydroxyvitamin D levels: the effect of UVB dose and skin color. J Am Acad Dermatol 57:588–593 [DOI] [PubMed] [Google Scholar]
- Holick MF, MacLaughlin JA, Doppelt SH 1981 Regulation of cutaneous previtamin D3 photosynthesis in man: skin pigment is not an essential regulator. Science 211:590–593 [DOI] [PubMed] [Google Scholar]
- Yanoff LB, Parikh SJ, Spitalnik A, Denkinger B, Sebring NG, Slaughter P, McHugh T, Remaley AT, Yanovski JA 2006 The prevalence of hypovitaminosis D and secondary hyperparathyroidism in obese Black Americans. Clin Endocrinol (Oxf) 64:523–529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gower BA, Nagy TR, Goran MI 1999 Visceral fat, insulin sensitivity, and lipids in prepubertal children. Diabetes 48:1515–1521 [DOI] [PubMed] [Google Scholar]
- Haffner SM, D'Agostino R, Saad MF, Rewers M, Mykkänen L, Selby J, Howard G, Savage PJ, Hamman RF, Wagenknecht LE 1996 Increased insulin resistance and insulin secretion in nondiabetic African-Americans and Hispanics compared with non-Hispanic whites. The Insulin Resistance Atherosclerosis Study. Diabetes 45:742–748 [DOI] [PubMed] [Google Scholar]
- Cowie CC, Rust KF, Byrd-Holt DD, Eberhardt MS, Flegal KM, Engelgau MM, Saydah SH, Williams DE, Geiss LS, Gregg EW 2006 Prevalence of diabetes and impaired fasting glucose in adults in the U.S. population: National Health And Nutrition Examination Survey 1999–2002. Diabetes Care 29:1263–1268 [DOI] [PubMed] [Google Scholar]
- Peechakara SV, Pittas AG 2008 Vitamin D as a potential modifier of diabetes risk. Nat Clin Pract Endocrinol Metab 4:182–183 [DOI] [PubMed] [Google Scholar]
- Maestro B, Campión J, Dávila N, Calle C 2000 Stimulation by 1,25-dihydroxyvitamin D3 of insulin receptor expression and insulin responsiveness for glucose transport in U-937 human promonocytic cells. Endocr J 47:383–391 [DOI] [PubMed] [Google Scholar]
- Maestro B, Molero S, Bajo S, Dávila N, Calle C 2002 Transcriptional activation of the human insulin receptor gene by 1,25-dihydroxyvitamin D(3). Cell Biochem Funct 20:227–232 [DOI] [PubMed] [Google Scholar]
- Pittas AG, Dawson-Hughes B, Li T, Van Dam RM, Willett WC, Manson JE, Hu FB 2006 Vitamin D and calcium intake in relation to type 2 diabetes in women. Diabetes Care 29:650–656 [DOI] [PubMed] [Google Scholar]
- Chiu KC, Chu A, Go VL, Saad MF 2004 Hypovitaminosis D is associated with insulin resistance and β-cell dysfunction. Am J Clin Nutr 79:820–825 [DOI] [PubMed] [Google Scholar]
- Hyppönen E, Power C 2006 Vitamin D status and glucose homeostasis in the 1958 British birth cohort: the role of obesity. Diabetes Care 29:2244–2246 [DOI] [PubMed] [Google Scholar]
- Scragg R, Holdaway I, Singh V, Metcalf P, Baker J, Dryson E 1995 Serum 25-hydroxyvitamin D3 levels decreased in impaired glucose tolerance and diabetes mellitus. Diabetes Res Clin Pract 27:181–188 [DOI] [PubMed] [Google Scholar]
- Forouhi NG, Luan J, Cooper A, Boucher BJ, Wareham NJ 2008 Baseline serum 25-hydroxy vitamin D is predictive of future glycemic status and insulin resistance: the Medical Research Council Ely Prospective Study 1990–2000. Diabetes 57:2619–2625 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffman RP 2008 Indices of insulin action calculated from fasting glucose and insulin reflect hepatic, not peripheral, insulin sensitivity in African-American and Caucasian adolescents. Pediatr Diabetes 9:57–61 [DOI] [PubMed] [Google Scholar]
- Tataranni PA, Ortega E 2005 A burning question: does an adipokine-induced activation of the immune system mediate the effect of overnutrition on type 2 diabetes? Diabetes 54:917–927 [DOI] [PubMed] [Google Scholar]
- Marshall WA, Tanner JM 1969 Variations in pattern of pubertal changes in girls. Arch Dis Child 44:291–303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuczmarski RJ, Ogden CL, Grummer-Strawn LM, Flegal KM, Guo SS, Wei R, Mei Z, Curtin LR, Roche AF, Johnson CL 2000 CDC growth charts: United States. Adv Data:1–27 [PubMed] [Google Scholar]
- Roth HJ, Schmidt-Gayk H, Weber H, Niederau C 2008 Accuracy and clinical implications of seven 25-hydroxyvitamin D methods compared with liquid chromatography-tandem mass spectrometry as a reference. Ann Clin Biochem 45:153–159 [DOI] [PubMed] [Google Scholar]
- Matsuda M, DeFronzo RA 1999 Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care 22:1462–1470 [DOI] [PubMed] [Google Scholar]
- Weiss R, Dziura J, Burgert TS, Tamborlane WV, Taksali SE, Yeckel CW, Allen K, Lopes M, Savoye M, Morrison J, Sherwin RS, Caprio S 2004 Obesity and the metabolic syndrome in children and adolescents. N Engl J Med 350:2362–2374 [DOI] [PubMed] [Google Scholar]
- Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC 1985 Homeostasis model assessment: insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28:412–419 [DOI] [PubMed] [Google Scholar]
- Hosker JP, Matthews DR, Rudenski AS, Burnett MA, Darling P, Bown EG, Turner RC 1985 Continuous infusion of glucose with model assessment: measurement of insulin resistance and β-cell function in man. Diabetologia 28:401–411 [DOI] [PubMed] [Google Scholar]
- Le Floch JP, Escuyer P, Baudin E, Baudon D, Perlemuter L 1990 Blood glucose area under the curve. Methodological aspects. Diabetes Care 13:172–175 [DOI] [PubMed] [Google Scholar]
- Matthews JN, Altman DG, Campbell MJ, Royston P 1990 Analysis of serial measurements in medical research. BMJ 300:230–235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holick MF 2007 Vitamin D deficiency. N Engl J Med 357:266–281 [DOI] [PubMed] [Google Scholar]
- Greer FR 2003 Vitamin D deficiency—it’s more than rickets. J Pediatr 143:422–423 [DOI] [PubMed] [Google Scholar]
- Gordon CM, DePeter KC, Feldman HA, Grace E, Emans SJ 2004 Prevalence of vitamin D deficiency among healthy adolescents. Arch Pediatr Adolesc Med 158:531–537 [DOI] [PubMed] [Google Scholar]
- Wortsman J, Matsuoka LY, Chen TC, Lu Z, Holick MF 2000 Decreased bioavailability of vitamin D in obesity. Am J Clin Nutr 72:690–693 [DOI] [PubMed] [Google Scholar]
- Buffington C, Walker B, Cowan Jr GS, Scruggs D 1993 Vitamin D Deficiency in the Morbidly Obese. Obes Surg 3:421–424 [DOI] [PubMed] [Google Scholar]
- Johnson JA, Grande JP, Roche PC, Kumar R 1994 Immunohistochemical localization of the 1,25(OH)2D3 receptor and calbindin D28k in human and rat pancreas. Am J Physiol 267:E356–E360 [DOI] [PubMed] [Google Scholar]
- Palomer X, González-Clemente JM, Blanco-Vaca F, Mauricio D 2008 Role of vitamin D in the pathogenesis of type 2 diabetes mellitus. Diabetes Obes Metab 10:185–197 [DOI] [PubMed] [Google Scholar]
- Scragg R, Sowers M, Bell C 2004 Serum 25-hydroxyvitamin D, diabetes, and ethnicity in the Third National Health and Nutrition Examination Survey. Diabetes Care 27:2813–2818 [DOI] [PubMed] [Google Scholar]
- Abdul-Ghani MA, Matsuda M, Balas B, DeFronzo RA 2007 Muscle and liver insulin resistance indexes derived from the oral glucose tolerance test. Diabetes Care 30:89–94 [DOI] [PubMed] [Google Scholar]
- Webb AR, Pilbeam C, Hanafin N, Holick MF 1990 An evaluation of the relative contributions of exposure to sunlight and of diet to the circulating concentrations of 25-hydroxyvitamin D in an elderly nursing home population in Boston. Am J Clin Nutr 51:1075–1081 [DOI] [PubMed] [Google Scholar]
- Weaver CM, McCabe LD, McCabe GP, Braun M, Martin BR, Dimeglio LA, Peacock M 2008 Vitamin D status and calcium metabolism in adolescent black and white girls on a range of controlled calcium intakes. J Clin Endocrinol Metab 93:3907–3914 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heaney RP 2000 Vitamin D: how much do we need, and how much is too much? Osteoporos Int 11:553–555 [DOI] [PubMed] [Google Scholar]
- Lips P, Wiersinga A, van Ginkel FC, Jongen MJ, Netelenbos JC, Hackeng WH, Delmas PD, van der Vijgh WJ 1988 The effect of vitamin D supplementation on vitamin D status and parathyroid function in elderly subjects. J Clin Endocrinol Metab 67:644–650 [DOI] [PubMed] [Google Scholar]
- Binkley N, Krueger D, Cowgill CS, Plum L, Lake E, Hansen KE, DeLuca HF, Drezner MK 2004 Assay variation confounds the diagnosis of hypovitaminosis D: a call for standardization. J Clin Endocrinol Metab 89:3152–3157 [DOI] [PubMed] [Google Scholar]