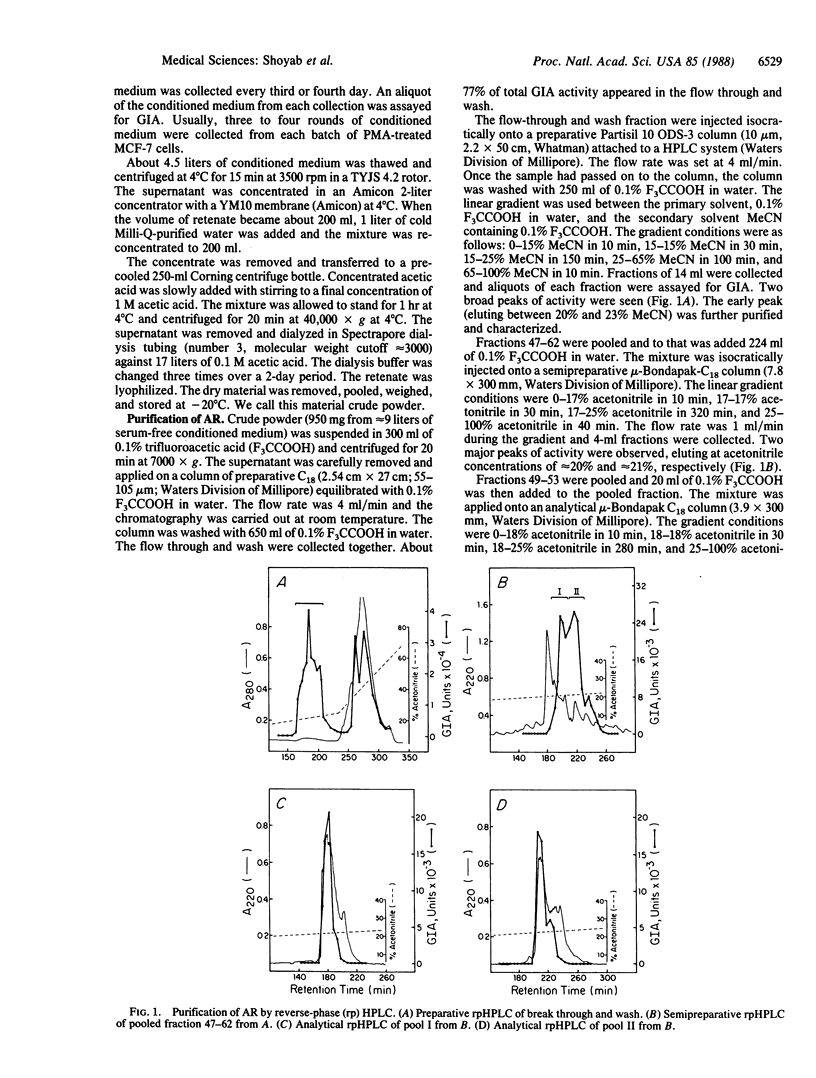
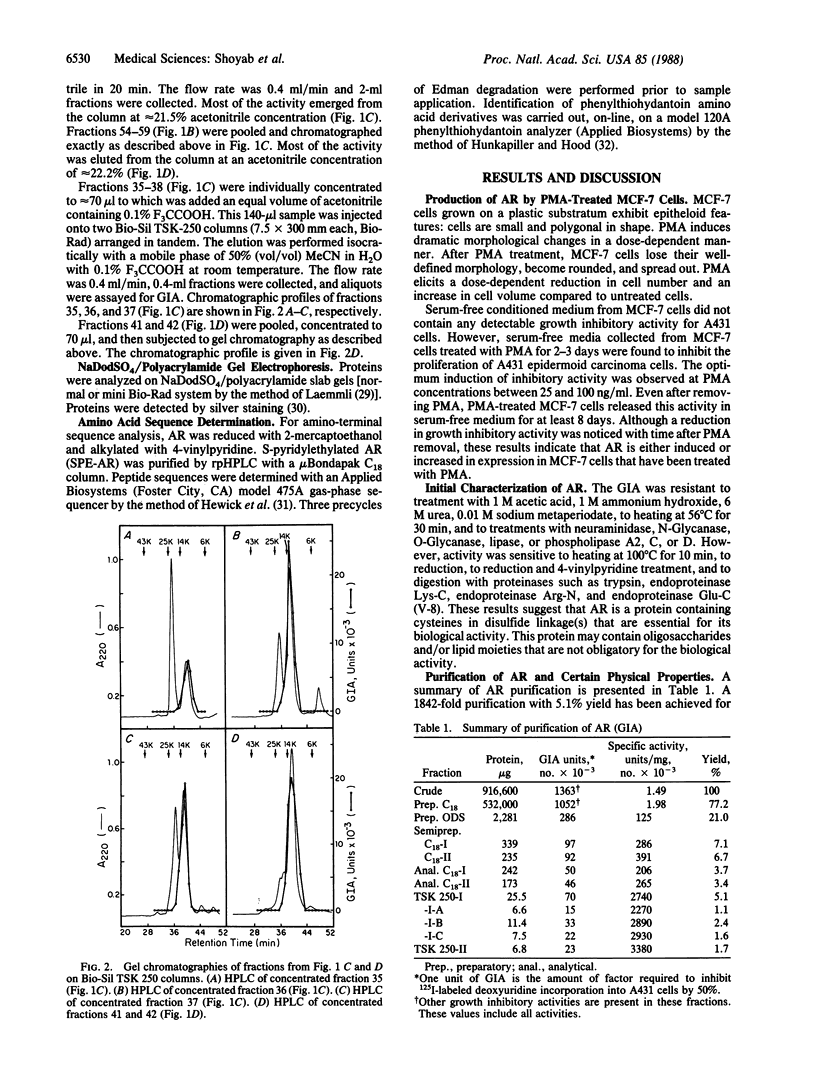
Abstract
A glycoprotein, termed amphiregulin (AR), inhibits growth of several human carcinoma cells in culture and stimulates proliferation of human fibroblasts and certain other tumor cells. It has been purified to apparent homogeneity from serum-free conditioned medium of MCF-7 human breast carcinoma cells that had been treated with phorbol 12-myristate 13-acetate. AR is a single-chain extremely hydrophilic glycoprotein containing cysteines in disulfide linkage(s) that are essential for biological activity; it is stable between pH 2 and pH 12 and after heating for 30 min at 56 degrees C but unstable at 100 degrees C. The apparent molecular weights of AR and N-Glycanase-treated AR are 14,000 and 15,000, respectively, as assessed by gel chromatography, and approximately 22,500 and approximately 14,000, respectively, as determined by polyacrylamide gel electrophoresis. Treatment of AR with N-Glycanase, O-Glycanase, or neuraminidase does not affect its activity. The pI of AR is approximately 7.8. The amino-terminal amino acid sequence of AR has been determined, and no significant sequence homology between AR and other proteins was found. The molecule thus appears to be a distinct growth regulatory protein.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barnes D. W. Epidermal growth factor inhibits growth of A431 human epidermoid carcinoma in serum-free cell culture. J Cell Biol. 1982 Apr;93(1):1–4. doi: 10.1083/jcb.93.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beutler B., Cerami A. Cachectin: more than a tumor necrosis factor. N Engl J Med. 1987 Feb 12;316(7):379–385. doi: 10.1056/NEJM198702123160705. [DOI] [PubMed] [Google Scholar]
- Degliantoni G., Murphy M., Kobayashi M., Francis M. K., Perussia B., Trinchieri G. Natural killer (NK) cell-derived hematopoietic colony-inhibiting activity and NK cytotoxic factor. Relationship with tumor necrosis factor and synergism with immune interferon. J Exp Med. 1985 Nov 1;162(5):1512–1530. doi: 10.1084/jem.162.5.1512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fryling C. M., Iwata K. K., Johnson P. A., Knott W. B., Todaro G. J. Two distinct tumor cell growth-inhibiting factors from a human rhabdomyosarcoma cell line. Cancer Res. 1985 Jun;45(6):2695–2699. [PubMed] [Google Scholar]
- Gill G. N., Lazar C. S. Increased phosphotyrosine content and inhibition of proliferation in EGF-treated A431 cells. Nature. 1981 Sep 24;293(5830):305–307. doi: 10.1038/293305a0. [DOI] [PubMed] [Google Scholar]
- Goustin A. S., Leof E. B., Shipley G. D., Moses H. L. Growth factors and cancer. Cancer Res. 1986 Mar;46(3):1015–1029. [PubMed] [Google Scholar]
- Hewick R. M., Hunkapiller M. W., Hood L. E., Dreyer W. J. A gas-liquid solid phase peptide and protein sequenator. J Biol Chem. 1981 Aug 10;256(15):7990–7997. [PubMed] [Google Scholar]
- Hunkapiller M. W., Hood L. E. Protein sequence analysis: automated microsequencing. Science. 1983 Feb 11;219(4585):650–659. doi: 10.1126/science.6687410. [DOI] [PubMed] [Google Scholar]
- Imai Y., Leung C. K., Friesen H. G., Shiu R. P. Epidermal growth factor receptors and effect of epidermal growth factor on growth of human breast cancer cells in long-term tissue culture. Cancer Res. 1982 Nov;42(11):4394–4398. [PubMed] [Google Scholar]
- Iwata K. K., Fryling C. M., Knott W. B., Todaro G. J. Isolation of tumor cell growth-inhibiting factors from a human rhabdomyosarcoma cell line. Cancer Res. 1985 Jun;45(6):2689–2694. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Melchers F., Andersson J. B cell activation: three steps and their variations. Cell. 1984 Jul;37(3):713–720. doi: 10.1016/0092-8674(84)90407-0. [DOI] [PubMed] [Google Scholar]
- Merril C. R., Goldman D., Sedman S. A., Ebert M. H. Ultrasensitive stain for proteins in polyacrylamide gels shows regional variation in cerebrospinal fluid proteins. Science. 1981 Mar 27;211(4489):1437–1438. doi: 10.1126/science.6162199. [DOI] [PubMed] [Google Scholar]
- Nathan C. F. Secretory products of macrophages. J Clin Invest. 1987 Feb;79(2):319–326. doi: 10.1172/JCI112815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Old L. J. Tumour necrosis factor. Polypeptide mediator network. 1987 Mar 26-Apr 1Nature. 326(6111):330–331. doi: 10.1038/326330a0. [DOI] [PubMed] [Google Scholar]
- Osborne C. K., Hamilton B., Nover M., Ziegler J. Antagonism between epidermal growth factor and phorbol ester tumor promoters in human breast cancer cells. J Clin Invest. 1981 Apr;67(4):943–951. doi: 10.1172/JCI110144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardee A. B. Molecules involved in proliferation of normal and cancer cells: presidential address. Cancer Res. 1987 Mar 15;47(6):1488–1491. [PubMed] [Google Scholar]
- Podolsky D. K., Pleskow D. K., Jafari H. Latent transformed growth-inhibiting factor in human malignant effusions. Cancer Res. 1988 Jan 15;48(2):418–424. [PubMed] [Google Scholar]
- Ransom J. H., Evans C. H., McCabe R. P., Pomato N., Heinbaugh J. A., Chin M., Hanna M. G., Jr Leukoregulin, a direct-acting anticancer immunological hormone that is distinct from lymphotoxin and interferon. Cancer Res. 1985 Feb;45(2):851–862. [PubMed] [Google Scholar]
- Roberts A. B., Anzano M. A., Lamb L. C., Smith J. M., Sporn M. B. New class of transforming growth factors potentiated by epidermal growth factor: isolation from non-neoplastic tissues. Proc Natl Acad Sci U S A. 1981 Sep;78(9):5339–5343. doi: 10.1073/pnas.78.9.5339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rozengurt E. Early signals in the mitogenic response. Science. 1986 Oct 10;234(4773):161–166. doi: 10.1126/science.3018928. [DOI] [PubMed] [Google Scholar]
- Sachs L. Growth, differentiation and the reversal of malignancy. Sci Am. 1986 Jan;254(1):40–47. doi: 10.1038/scientificamerican0186-40. [DOI] [PubMed] [Google Scholar]
- Schonbrunn A., Krasnoff M., Westendorf J. M., Tashjian A. H., Jr Epidermal growth factor and thyrotropin-releasing hormone act similarly on a clonal pituitary cell strain. Modulation of hormone production and inhbition of cell proliferation. J Cell Biol. 1980 Jun;85(3):786–797. doi: 10.1083/jcb.85.3.786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sone S., Mutsuura S., Ishii K., Shirahama T., Tsubura E. In vitro cytotoxicity to various human tumor cell lines of a tumor cytotoxic factor(s) produced by human alveolar macrophages. Gan. 1984 Oct;75(10):920–928. [PubMed] [Google Scholar]
- Sporn M. B., Roberts A. B. Autocrine growth factors and cancer. 1985 Feb 28-Mar 6Nature. 313(6005):745–747. doi: 10.1038/313745a0. [DOI] [PubMed] [Google Scholar]
- Sporn M. B., Roberts A. B. Peptide growth factors and inflammation, tissue repair, and cancer. J Clin Invest. 1986 Aug;78(2):329–332. doi: 10.1172/JCI112580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sporn M. B., Roberts A. B. Peptide growth factors are multifunctional. Nature. 1988 Mar 17;332(6161):217–219. doi: 10.1038/332217a0. [DOI] [PubMed] [Google Scholar]
- Valette A., Gas N., Jozan S., Roubinet F., Dupont M. A., Bayard F. Influence of 12-O-tetradecanoylphorbol-13-acetate on proliferation and maturation of human breast carcinoma cells (MCF-7): relationship to cell cycle events. Cancer Res. 1987 Mar 15;47(6):1615–1620. [PubMed] [Google Scholar]
- Weinstein I. B. Growth factors, oncogenes, and multistage carcinogenesis. J Cell Biochem. 1987 Mar;33(3):213–224. doi: 10.1002/jcb.240330308. [DOI] [PubMed] [Google Scholar]
- Zarling J. M., Shoyab M., Marquardt H., Hanson M. B., Lioubin M. N., Todaro G. J. Oncostatin M: a growth regulator produced by differentiated histiocytic lymphoma cells. Proc Natl Acad Sci U S A. 1986 Dec;83(24):9739–9743. doi: 10.1073/pnas.83.24.9739. [DOI] [PMC free article] [PubMed] [Google Scholar]