Abstract
Gastrointestinal stromal tumors (GISTs) are the most common mesenchymal tumors in the gastrointestinal tract (GIT). Although interstitial cells of Cajal has been suggested as origin of this tumor, the cytological and ultrastructural features of GISTs are heterogeneous and unclear. A total 10 cases of normal gastrointestinal tissue (control), 13 GISTs of the stomach (8), small intestine (3), mesocolon (1) and liver (1), and 2 gastrointestinal autonomic nervous tumor (GANT) of small intestine were ultrastructurally studied. Normal interstitial cells of Cajal (ICC) were abundantly present around the myenteric plexuses or individually scattered through the wall of GIT. ICC was characterized by slender cytoplasmic processes, well-developed endoplasmic reticulum (ER), mitochondria, Golgi apparatus, caveolae and intermediate filaments. The GISTs and GANTs had overlapping ultrastructures. The most common and important ultrastructural features of GISTs were rich villous cytoplasmic processes, dispersed intermediate filaments and abundant SER, and those of GANTs were neurosecretory granules and skenoid fibers. Compared with ICC, the GISTs and GANTs had remarkably reduced caveolae and gap junctions. Our study suggested that ultrastructural analysis gives much information to investigate lineage differentiation of neoplastic cells and make a differential diagnosis of these tumors from other mesenchymal tumors and between GISTs and GANTs.
Keywords: Gastrointestinal Neoplasms; Stromal Tumors; Autonomic Pathways; Microscopy, Electron; Immunohistochemistry; Proto-Oncogene Protein c-Kit
INTRODUCTION
The mesenchymal tumors of the gastrointestinal (GI) tract have been called as various names including stromal tumor of unknown malignant potential (STUMP), gastrointestinal stromal tumors (GIST), gastrointestinal autonomic nervous tumors (GANT) and gastrointestinal pacemaker cell tumors (GIPACT) (1, 2). After stem cell kinase receptor CD117 (c-Kit) was introduced, the GISTs presumed to derive from stem cells differentiating to the GI pacemaker cells, known as interstitial cells of Cajal (ICC), because nearly all GISTs are implicated with c-Kit gene abnormality and CD117 (c-Kit) overexpression (1). Even though the new entity of GIST were initially introduced from the ultrastructural observation, the number of ultrastructural studies on GISTs (3-5) have been limited, compared to the quite a few ultrastructural reports of GANTs (6-8).
GISTs can occur primarily in every intraabdominal site including omentum, mesentery or peritoneum in addition to GI tract. CD117 is being demonstrated increasingly in a wide variety of tumors including inflammatory myofibroblastic tumors, intraabdominal desmoids (9), angiomyolipoma and perivascular epithelioid cell tumors (10), and malignant peripheral nerve sheath tumor (11), which give rise to quite confusion.
We investigated the GIST and GANT ultrastructurally to identify the specific ultrastructure of these tumors, any clue of their histogenesis and the comparison of the GISTs and GANTs.
MATERIALS AND METHODS
Fifteen cases of GISTs including 2 cases of GANTs were surgically resected at the Ilsan Paik Hospital, Inje University from February 2000 to May 2003. Among them, thirteen cases had been carried out the electron microscopic studies. The preservation and quality of processing of all cases were excellent or enough for evaluation. The pathologic diagnoses and grading of GISTs were made by 3 pathologists, based on morphology and immunohistochemistry with Miettinen et al.'s grading system of GISTs, which recommended the grading by tumor size and mitotic rate along with the location (12). We excluded c-Kit negative pure smooth muscle tumors, Schwannomas and other gastrointestinal mesenchymal tumors.
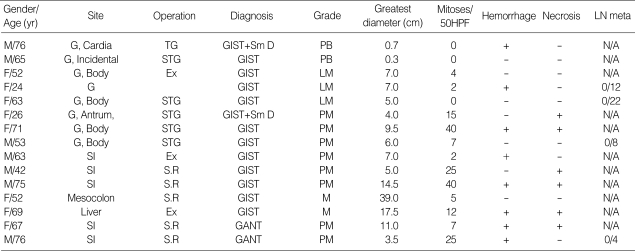
Clinical findings are summarized in Table 1. Males and females were equally represented. Age range was 24 to 76 yr old (mean: 58.4 yr old). Most patients presented with abdominal pain or intestinal bleeding. The size range was 0.3 cm to 39 cm in greatest diameter (mean: 9.1 cm). Eight cases were located in the stomach, 5 cases were in the small intestine, one in the mesocolon and one in the liver. One gastric GIST was incidentally found at operation for gastric adenocarcinoma. The last one was late recurrent GIST 10 yr after resection of primary gastric GIST.
Table 1.
Summary of the clinicopathologic features
G, gastric; SI, small intestine; PB, probably benign; LM, Uncertain or low malignant potential; PM, probably malignant; M, Malignant; Meta, metastasis; Hemo, hemorrhage; Necro, necrosis; LN, lymph nodes; Incidental, incidentally found when adenocarcinoma resected; Sm D, smooth muscle differentiation; -, absent; +, present; N/A, not available (lymph nodes was not dissected); TG, total gastrectomy; STG, subtotal gastrectomy; Ex, Excision only; S.R, Segmental resection of intestine; HPF, high power field.
After sampling for electron microscopic (EM) study, tumor tissue was fixed in 10% neutral formalin solution. For EM study the tissue fixed in 3.0% glutaraldehyde was washed in cold 0.1 M phosphate buffer (pH 7.4) and postfixed in 1% osmium tetroxide, buffered with 0.1 M phosphate. The tissue was embedded in epoxy resin. Sections were cut with an ultramicrotome, stained with uranyl acetate and lead citrate, and examined by EM (Jeol, Japan).
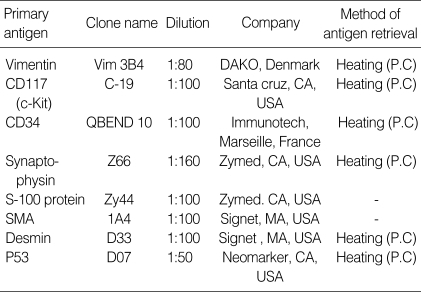
For immunohistochemical study, all representative formalin (cold 10% buffered) fixed and paraffin-embedded tumor tissues were cut to 2-3 µm thickness. All antigens used here including c-Kit, CD34, vimentin, smooth muscle actin (SMA), desmin, S100 protein, synaptophysin, CD56 and Ki67 (MIB-1) were summarized in Table 2. After deparaffinization and rehydration, the sections were subjected to high temperature antigen unmasking in a citrate buffer in the autoclave (121℃) for 20 min. After blocking in 2% skimmed milk, the sections were incubated with primary antibodies with ideal dilution for 60 min at room temperature. The sections were incubated with biotinylated secondary antibody, which is polyvalent and universal (prediluted, Immunotech, Marseille, France), and the expression was detected using the peroxidase labeled streptavidin biotin complex technique according to the manufacturer's recommendations. Hematoxylin counter staining was performed. Normal small intestine and resident mast cells were served as the positive tissue control for all used antibodies. For antibody control, primary antibodies were omitted.
Table 2.
Primary antigens used for immunohistochemical study
P.C, pressure cooker; SMA, smooth muscle actin.
RESULTS
Gross findings
Most tumors were large and ranged from 0.3 cm to 17.5 cm (mean: 9.1 cm). The cut surfaces of the tumors showed pinkish gray fish flesh to granular appearance with some hemorrhage and necrosis. The tumors were mainly located in the muscular wall of the intestine with or without involvement of the mucosa and serosa. One case of the small intestinal GISTs showed overt multiple mesenteric metastatic nodules.
Light microscopic findings
Among thirteen cases of GISTs, six were spindle cell type (Fig. 1), three epithelioid cell type (Fig. 2) and four mixed type. The two GANTs revealed mainly epithelioid feature (Fig. 3). However, the epithelioid cell type also had some spindle cells areas. The cytoplasm of most tumor cells was eosinophilic and abundant. Marked myxoid change was present in some cases. Focal cytoplasmic vacuolization was often seen. No signet ring-like or oncocytic cells or cells with a clear cytoplasm were noted. The spindle cell type tumors were arranged in interlacing fascicles or whorls. Generally mitotic counts were low but various in each case from 0/50 to 40/50 HPF. Hemorrhage and/or necrosis were present in 9/15 cases. Eleven cases were pure GIST, 2 cases were GIST with smooth muscle differentiation and 2 cases were GANT. According to the Miettinen et al. report (2002), two cases were malignant having metastasis, six cases were probably malignant, three cases were low malignant potential and two cases were categorized as probably benign. All GANTs were probably malignant. There was no case with lymph node metastasis.
Fig. 1.
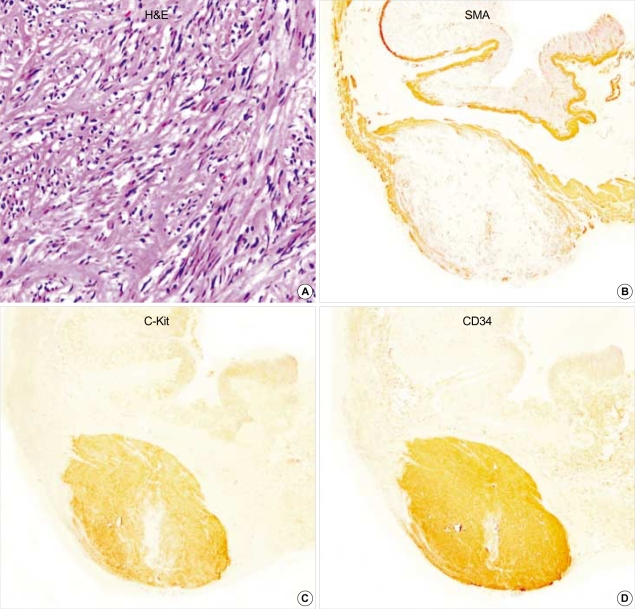
Gastric GIST (case 1). The tumor is composed of interlacing fascicles of spindle cells with eosinophilic cytoplasm. The tumor cells show patch immunopositivity for SMA, and express c-Kit and CD34, strongly and diffusely. (A: H&E,×200, B: SMA, ×40, C: c-Kit, ×40, D: CD34 immunostaining, ×40).
Fig. 2.
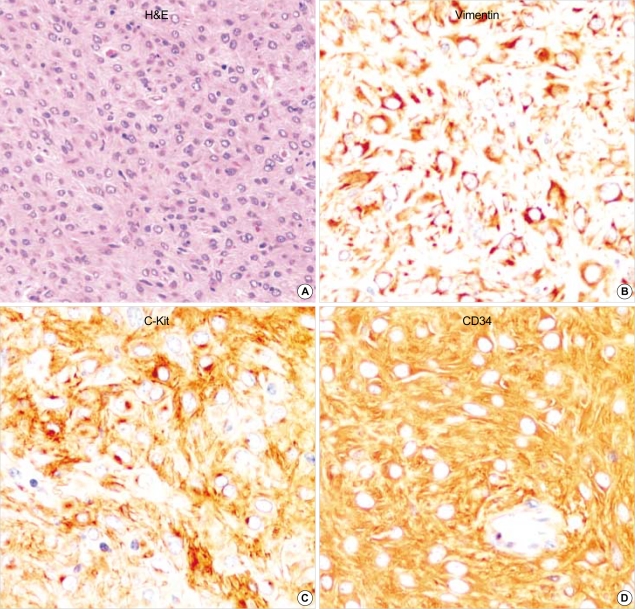
Epithelioid GIST shows sheets of polygonal neoplastic cells with eosinophilic cytoplasm. This tumor reveals diffuse and strong immunoreactivity for vimentin, c-Kit and CD34. (A: H&E, ×200, B: Vimentin, ×400, C: c-Kit, ×400, D: CD34 immunostaining, ×400).
Fig. 3.
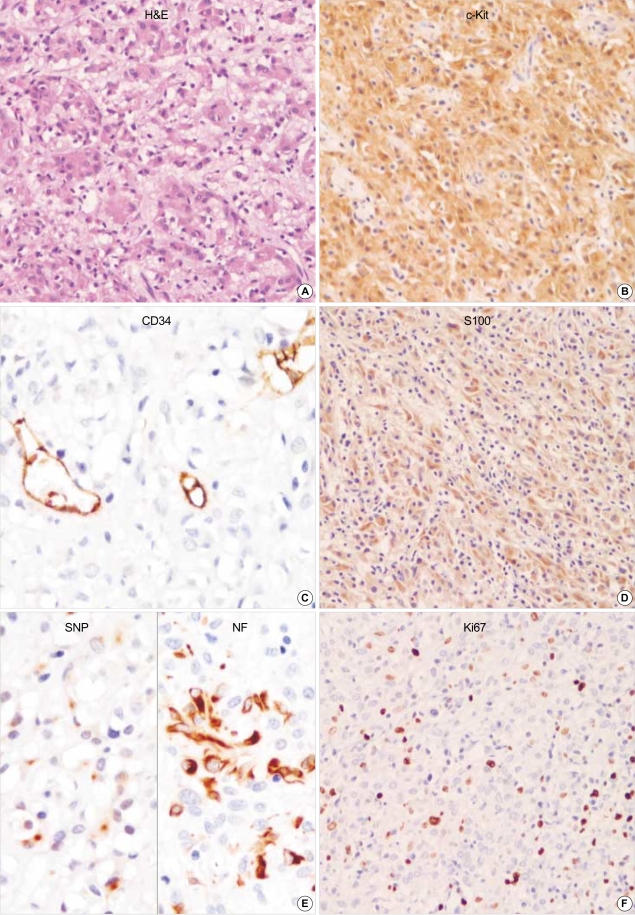
GANT reveals mainly epithelioid feature. Focal cytoplasmic vacuolization is seen. The tumor cells are robustly positive for CD117 (c-kit) but negative for CD34. S-100 protein, synaptophysin and NF are partially positive in neoplastic cells. Ki67 labelling index is high. (A: H&E, ×200, B: c-Kit, ×200, C: CD34, ×400, D: S100, ×200, E: left: Synaptophysin, right: Neurofilaments, ×400, F: Ki67 immunostaining, ×200).
Immunohistochemical findings
All of the fifteen GISTs including two GANTs were robustly positive for vimentin (100%) and CD117 (c-Kit) (100%) (Fig. 1-3). CD34 was patch or diffuse but strongly immunoreactive in 12 out of 15 cases. Two cases coexpressed SMA (Fig. 1), strongly but focally. GANTs revealed strong but focally positive for S-100 protein and neurofilament (NF) (Fig. 3). Synaptophysin was only focally and less intensely immunoreactive in one case of GANTs (Fig. 3). P53 was positive in 61.5% (8/13 cases) of GISTs and 100% (2/2 cases) of GANTs.
In normal adults stomach and intestine, well-developed c-Kit immunoreactive ICC were present around the myenteric plexus and muscle layers (Fig. 4). They were also immunoreactive for CD34 (Fig. 4).
Fig. 4.
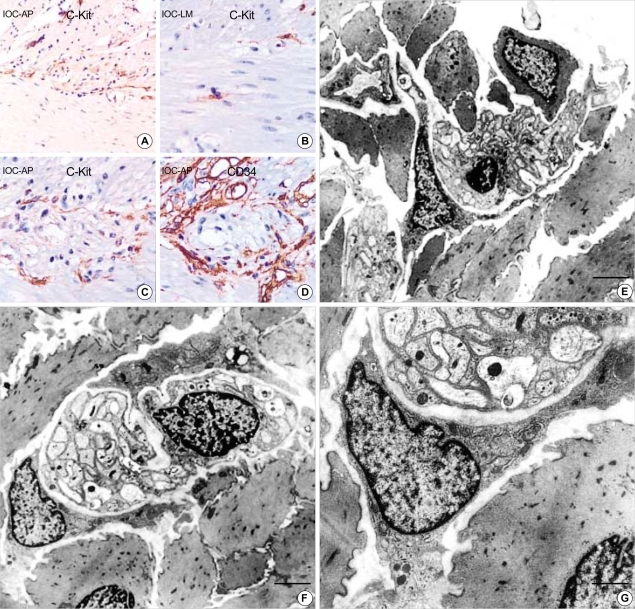
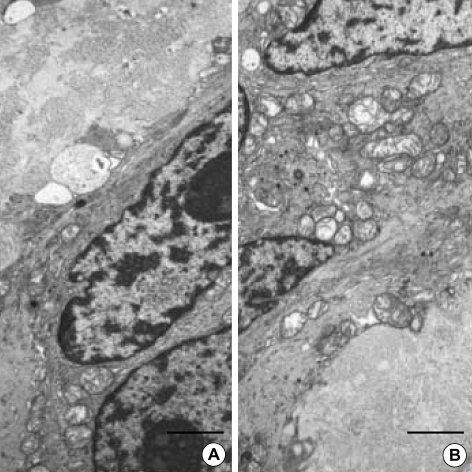
(A) Normal adult (M/45 yr) small intestine. (A-D) The immunohistochemical staining for c-Kit and CD34 shows immunoreactive interstitial cell of Cajal in myenteric plexus (A: c-Kit, ×200, B: ×400, C: ×400, D: CD34, ×400). (E-G) Ultrastructure of the interstitial cell of Cajal in normal stomach. It has slender cytoplasmic processes and the nucleus is heterochromatic and lobulated in apprearance. Two interstitial cell of Cajal hug the myenteric plexuses composed of many well-delineated peripheral nerves surrounded by Schwann cell cytoplasm and continuous external lamina (F). The cytoplasm of ICC has well-defined organelles such as rough endoplasmic reticulum, mitochondria, Golgi apparatus and caveolae. Gap junctions (arrows) are present but no external lamina is noted (G). (uranyl acetate and lead citrate stain E-F: bar: 2 µm, G: bar: 1 µm, E: ×2,500, F: ×3,000, G: ×8,000).
Ultrastructural findings
In normal controls, the myenteric ICC had heterochromatic and lobulated nuclei with slender cytoplasmic processes (Fig. 4). They hug the myenteric plexuses. ICC had well developed cytoplasmic organelles such as endoplasmic reticulum, mitochondria, Golgi apparatus and caveolae. Gap junctions are well seen, but no external lamina was present (Fig. 4).
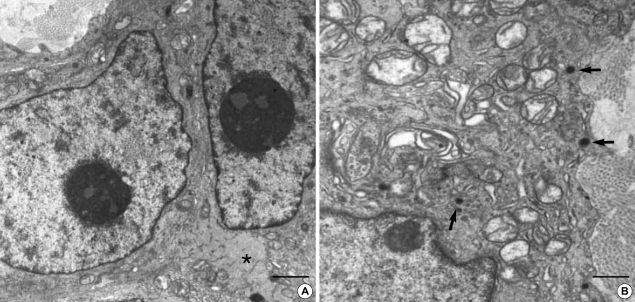
Ultrastructurally, the neoplastic cells were polygonal or elongated in appearance. The polygonal cells usually had rich interdigitating cytoplasmic processes and most of them were villous (filopodia-like) (Fig. 5). However spindle shaped neoplastic cells did not have well developed villous processes (Fig. 6). The nuclei were oval to irregularly elongated and generally heterochromatic, but some nuclei were exceptionally euchromatic (Fig. 5, 6). The tumor cells were closely apposed except the areas having intercellular collagenous stroma. There were also abundant wavy intermediate filaments, which were generally dispersed in the cytoplasm. The cytoplasmic organelles were abundant, smooth and rough endoplasmic reticulum, mitochondria, lysosomes and Golgi apparatus. There were desmosome or hemidesmosome-like junctions as well as a few gap junctions (Fig. 6) in 2 cases of gastric GISTs. A discrete external lamina was usually absent but incontinuous external lamina material was present in half of the GISTs (Fig. 6). Caveolae or pinocytotic vesicles were rarely seen. A case with focal immunohistochemical smooth muscle differentiation showed ultrastructual differentiation toward smooth muscle, such as cytoplasmic myofilaments (Fig. 6). Small round dense core neurosecretory granules were observed in GANTs, measuring 40-150 nm in diameter (Fig. 7), but GISTs also had electron dense granules, favor lysosomes, which could be confused with neurosecretory granules. No definite synaptic junction, synaptic vesicles or microtubles were found in GANTs. In all two cases of GANTs, skenoid fibers were characteristically present (Fig. 8).
Fig. 5.
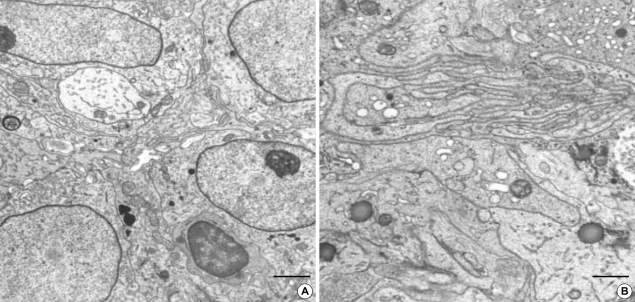
The Epithelioid GIST shows euchromatic nuclei and have prominent skein appearing nucleolus with rich filopodia-like cytoplasmic processes. The cytoplasmic processes are interdigitating each other. Electron dense cytoplasmic granules, favor lysosomes, are frequently seen, which may be confused with neurosecretory granules. (uranyl acetate and lead citrate stain, A: bar: 2 µm, B: bar: 1 µm, A: ×4,000, B: ×6,000).
Fig. 6.
(A) Spindle cell GIST also shows cytoplasmic processes but do not have much villous processes. The neoplastic cells have a nuclear pseudoinclusion and rich organelles including electron dense lysosomal granules. There are hemidesmosome-like junctions (arrowheads) as well as a few gap junctions (arrow). Incontinuous external lamina is seen. (B) Ultrastructual differentiation toward smooth muscle, such as cytoplasmic myofilaments are seen (arrows) (uranyl acetate and lead citrate stain, bar: 2 µm, A: ×5,000, B: ×6,000).
Fig. 7.
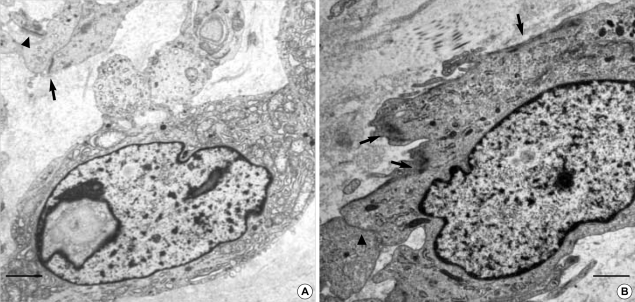
The GANT have oval to irregular generally heterochromatic nuclei with prominent nucleolus. There are also abundant and evenly dispersed wavy intermediate filaments (asteric mark). The tumor cells are closely apposed except some areas having intercellular collagenous tissue. Small round neurosecretory granules (arrows) are observed, measuring 40-150 nm (uranyl acetate and lead citrate stain, A: bar: 1 µm, B: bar: 500 nm, A: ×6,000, B: ×10,000).
Fig. 8.
Both cases of GANTs have characteristic extracellular skenoid fibers. (inlet: high power view of skenoid fibers). (uranyl acetate and lead citrate stain, A, B: bars: 1 µm, A, B: ×4,000).
DISCUSSION
In 1983, Mazur and Clark first introduced the vague term 'gastrointestinal stromal tumor' because many mesenchymal tumors of the gastrointestinal tract did not express the known markers of the smooth muscle or Schwann cell (13). Perineurial or mesenchymal nerve sheath cells of myenteric plexus were thought as a possible origin of these stromal tumors (13). After that, owing to lots of efforts to clarify these tumors, a new entity of GISTs and its pathogenesis have been established.
GISTs are most commonly occurred in the stomach (60-70%) and the small intestine (20-30%). In addition, the tumors arose in the esophagus, colon, rectum, omentum and mesentery have been reported less than 10%. For predicting prognosis, Ma et al. (14) graded the GISTs into benign (<5 cm in size and <5 mitosis/50 HPF), malignant (>5 mitosis/50 HPF), and borderline lesions (>5 cm in size but <5 mitosis/50 HPF) regardless of site. However, several reports suggested that stromal tumors should be analysed as a site-specific fashion because tumors from different location brought out different growth patterns and prognosis. According to suggestion of Miettinen et al. in the grading (12), 2 of our GISTs were malignant having metastasis, 6 were probably malignant, 3 were low malignant potential and 2 were probably benign. All 2 GANTs were probably malignant.
For making a diagnosis of GISTs, immunohistochemical stainings of the CD117 and ultrastructural examination are required, because morphological heterogeneity of the GISTs gives rise to quite confusion. Except rare cases, the diagnosis of the GIST should apply to the neoplasm displaying c-Kit immunoreactivity (15). The exceptions were very clearly described in the paper of Christopher et al.; including fixation artifact, technical error of immunostaining, sampling error, cessation of Kit expression perhaps following STI-571 therapy and very rare cases (2%) with real lack of Kit mutation and/or Kit expression (15).
GANTs are also GISTs based on c-Kit immunoreactivity. GISTs can show focal differentiation to autonomic nerve or smooth muscle, but pure smooth muscle or Schwann cell derived tumors, such as leiomyoma, leiomyosarcoma and Schwannoma are not GISTs.
Morphological heterogeneity of GISTs such as extreme epithelioid or spindle shape of the tumor cells or marked myxoid or cystic changes are well known. Rare oncocytic or signet ring cell type GANTs were reported (16). Tumors of our cases also showed variable histology, but immunohistochemically, our cases were compatible with GISTs or GANTs based on expression of vimentin (100%), CD117 (100%), and CD34 (66.7%). Two cases with focal immunoreactivity for SMA, were diagnosed as GISTs with smooth muscle differentiation. GANTs showed variable immunoreactivity for S-100 protein, NF, synaptophysin in addition to CD117. Besides a tumor size over 5 cm, mitotic rate still seems to be the best predictor for prognosis of GIST. Also MIB-1 proliferation index above 4% and p53 positivity indicate a more aggressive course (17, 18). However, Cunningham et al. found no relationship between p53 immunoreactivity and survival (19). In our series, p53 was positive in high percentage (69.2%, 9/13 cases) of GISTs and 100% (2/2 cases) of GANTs, reflecting the high percentage of high-grade tumors.
Until now, the ultrastructural reports of GISTs (3-5) were rare than those of GANTs (6-8). GISTs may originate from the pluripotent precursor cells of the ICC (5). According to the electron microscopic study on KitW-lacz/+ transgenic mice, Kit-expressing cells in the outer parts of the musculature had scattered caveolae, inconspicuous basal lamina and numerous mitochondria, whereas those in the submuscular region they had more pronounced myoid features. The ICCs are located in most parts of the digestive system, especially in myenteric plexus, submucosal Meissner's plexus and third plexus. ICCs demonstrate species-, region-, and location-dependent variations in the ultrastructures (20). The common ultrastructures of ICCs are exceptionally elongated shape having several lateral branches. They are communicated each other or with smooth muscle by peg and socket or gap junctions. Its cytoplasm characteristically possesses cisternae of smooth endoplasmic reticulum and intermediate filaments (21). In present study, normal ICCs present in the myenteric plexus had the similar ultrastructures as the previously reported ones. In the GISTs, the polygonal shaped neoplastic cells usually had rich interdigitating or villous cytoplasmic processes, but spindle shaped cells rarely showed villous processes. Polygonal shaped neoplastic cells have more numerous organelles than spindle shaped neoplastic cells. The nuclei were oval to irregularly elongated and generally heterochromatic. The tumor cells were closely apposed each other in all cases but spindle cells had more interstitium, which was fibrous. In our cases, small round neurosecretory granules were definitely found in GANTs, measuring 40-150 nm in diameter. Interestingly, GISTs also had many small electron dense granules, which were compatible with lysosomes. The prominent cytoplasmic organelles were quite a few smooth and rough endoplasmic reticulum, and some mitochondria, lysosomes and Golgi apparatus. Abundant intermediate filaments were dispersed in the cytoplasm and cytoplasmic processes. In 2 cases, desmosome-like or hemidesmosome-like junctions as well as gap junctions were found. Incontinuous external lamina material was noted in half of the cases. One of our cases was a recurrent GIST occurred in the liver 10 yr lag period after resection of gastric GIST, which was previously diagnosed as leiomyosarcoma. In all 15 cases, the caveolae were rarely seen except one case, which showed smooth muscle differentiation. All of these features were comparable with ultrastructure of ICC cells except reduced caveolae and gap junctions (20-22). Yantiss et al. described that ultrastructural site-specific similarities and differences of the GISTs (4), however, our cases showed different ultrastructures according to the cell shape, i.e., whether it is epithelioid or spindle, irrespective of location.
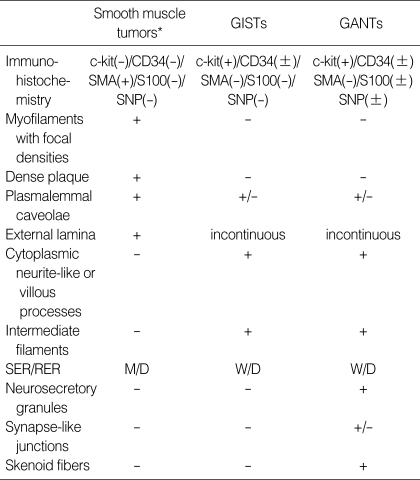
GANTs showed characteristic skenoid fibers. Lee et al. described that the clinicopathologic, histologic, immunohistologic, and molecular features of GANTs were similar to GISTs, indicating that GANTs merely represents a phenotypic variant of GISTs (23). In the ultrastructural level, although GISTs and GANTs have overlapping features, the ultrastructural characteristics of GANTs were dense core neurosecretory granules and skenoid fibers. Neurosecretory granules and skenoid fibers are not specific for GANT because the latter can be seen in smooth muscle tumors and the former are present in various neuroendocrine tumors and small cell carcinomas. However, when we differentiate GANTs from the GISTs, identification of these two features may be very helpful. In our case no definite synapses-like junctions or microtubules were found.
In conclusion, the GISTs and GANTs had overlapping ultrastructures. From exclusion diagnosis of smooth muscle or Schwannian tumors on the base of absence of hallmarks of these tumors and inclusion diagnosis of GISTs and GANTs can be easily made from electron microscopic examination. The most common and important ultrastructural features of GISTs were rich villous cytoplasmic processes and dispersed intermediate filaments and those of GANTs were neurosecretory granules and skenoid fibers. Ultrastructural analysis gives much benefit for identification of the lineage differentiation of neoplastic cells and differential diagnosis from other mesenchymal tumors and between GIST and GANT.
Table 3.
The comparison of the Immunohistochemical and ultrastructural features among pure smooth muscle tumors, GISTs, and GANTs
Smooth muscle tumors, leiomyomas/leiomyosarcoma; M/D, Moderately differentiated; W/D, well differentiated.
ACKNOWLEDGEMENTS
This work was supported by Grant from Inje University, 2002. Jea-Ok So assisted with excellent immunohistochemical stainings. Mee-Kyung Gong (Asan Mecidal Center, Seoul, Korea) kindly helped the ultrastructural examination.
References
- 1.Chan JK. Mesenchymal tumors of the gastrointestinal tract: a paradise for acronyms (STUMP, GIST, GANT, and now GIPACT), implication of c-kit in genesis, and yet another of the many emerging roles of the interstitial cell of Cajal in the pathogenesis of gastrointestinal diseases? Adv Anat Pathol. 1999;6:19–40. doi: 10.1097/00125480-199901000-00003. [DOI] [PubMed] [Google Scholar]
- 2.Kindblom LG, Remotti HE, Aldenborg F, Meis-Kindblom JM. Gastrointestinal pacemaker cell tumor (GIPACT): gastrointestinal stromal tumors show phenotypic characteristics of the interstitial cells of Cajal. Am J Pathol. 1998;152:1259–1269. [PMC free article] [PubMed] [Google Scholar]
- 3.Eyden B, Chorneyko KA, Shanks JH, Menasce LP, Banerjee SS. Contribution of electron microscopy to understanding cellular differentiation in mesenchymal tumors of the gastrointestinal tract: a study of 82 tumors. Ultrastruct Pathol. 2002;26:269–285. doi: 10.1080/01913120290104548. [DOI] [PubMed] [Google Scholar]
- 4.Yantiss RK, Rosenberg AE, Selig MK, Nielsen GP. Gastrointestinal stromal tumors: an ultrastructural study. Int J Surg Pathol. 2002;10:101–113. doi: 10.1177/106689690201000203. [DOI] [PubMed] [Google Scholar]
- 5.Liu P, Na J, Wang Y, He Q, Zhang Y, Tang X, Zou W. Study of gastrointestinal stromal tumors by light microscopy, electron microscopy and immunohistochemistry. Zhonghua Bing Li Xue Za Zhi. 2002;31:199–203. [PubMed] [Google Scholar]
- 6.Dhimes P, Lopez-Carreira M, Ortega-Serrano MP, Garcia-Munoz H, Martinez-Gonzalez MA, Ballestin C. Gastrointestinal autonomic nerve tumours and their separation from other gastrointestinal stromal tumours: an ultrastructural and immunohistochemical study of seven cases. Virchows Arch. 1995;426:27–35. doi: 10.1007/BF00194695. [DOI] [PubMed] [Google Scholar]
- 7.Matsumoto K, Min W, Yamada N, Asano G. Gastrointestinal autonomic nerve tumors: immunohistochemical and ultrastructural studies in cases of gastrointestinal stromal tumor. Pathol Int. 1997;47:308–314. doi: 10.1111/j.1440-1827.1997.tb04498.x. [DOI] [PubMed] [Google Scholar]
- 8.Erlandson RA, Klimstra DS, Woodruff JM. Subclassification of gastrointestinal stromal tumors based on evaluation by electron microscopy and immunohistochemistry. Ultrastruct Pathol. 1996;20:373–393. doi: 10.3109/01913129609016340. [DOI] [PubMed] [Google Scholar]
- 9.Montgomery E, Torbenson MS, Kaushal M, Fisher C, Abraham SC. Beta-catenin immunohistochemistry separates mesenteric fibromatosis from gastrointestinal stromal tumor and sclerosing mesenteritis. Am J Surg Pathol. 2002;26:1296–1301. doi: 10.1097/00000478-200210000-00006. [DOI] [PubMed] [Google Scholar]
- 10.Makhlouf HR, Remotti HE, Ishak KG. Expression of KIT (CD117) in angiomyolipoma. Am J Surg Pathol. 2002;26:493–497. doi: 10.1097/00000478-200204000-00012. [DOI] [PubMed] [Google Scholar]
- 11.Leroy X, Aubert S, Leteurtre E, Gosselin B. Expression of CD117 in a malignant peripheral nerve sheath tumour arising in a patient with type 1 neurofibromatosis. Histopathology. 2003;42:511–513. doi: 10.1046/j.1365-2559.2003.01558.x. [DOI] [PubMed] [Google Scholar]
- 12.Miettinen M, El-Rifai W, Sobin HL, Lasota J. Evaluation of malignancy and prognosis of gastrointestinal stromal tumors: a review. Hum Pathol. 2002;33:478–483. doi: 10.1053/hupa.2002.124123. [DOI] [PubMed] [Google Scholar]
- 13.Mazur MT, Clark HB. Gastric stromal tumors: reappraisal of histogenesis. Am J Surg Pathol. 1983;7:507–519. doi: 10.1097/00000478-198309000-00001. [DOI] [PubMed] [Google Scholar]
- 14.Ma CK, Amin MB, Kintanar E, Linden MD, Zarbo RJ. Immunohistologic characterization of gastrointestinal stromal tumors: a study of 82 cases compared with 11 cases of leiomyomas. Mod Pathol. 1993;6:139–144. [PubMed] [Google Scholar]
- 15.Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, Miettinen M, O'Leary TJ, Remotti H, Rubin BP, Shmookler B, Sobin LH, Weiss SW. Diagnosis of gastrointestinal stromal tumors: A consensus approach. Hum Pathol. 2002;33:459–465. doi: 10.1053/hupa.2002.123545. [DOI] [PubMed] [Google Scholar]
- 16.Damiani S, Pasquinelli G, Eusebi V. GANT-like gastrointestinal pacemaker cell tumours with oncocytic features. Virchows Arch. 1999;435:143–150. doi: 10.1007/s004280050412. [DOI] [PubMed] [Google Scholar]
- 17.Al-Bosom IA. p53 expression in gastrointestinal stromal tumors. Pathol Int. 2001;51:519–523. doi: 10.1046/j.1440-1827.2001.01233.x. [DOI] [PubMed] [Google Scholar]
- 18.Seidal T, Edvardsson H. Expression of c-kit (CD117) and Ki67 provides information about the possible cell of origin and clinical course of gastrointestinal stromal tumours. Histopathology. 1999;34:416–424. doi: 10.1046/j.1365-2559.1999.00643.x. [DOI] [PubMed] [Google Scholar]
- 19.Cunningham RE, Abbondanzo SL, Chu WS, Emory TS, Sobin LH, O'Leary TJ. Apoptosis, bcl-2 expression, and p53 expression in gastrointestinal stromal/smooth muscle tumors. Appl Immunohistochem Mol Morphol. 2001;9:19–23. [PubMed] [Google Scholar]
- 20.Faussone-Pellegrini MS, Thuneberg L. Guide to the identification of interstitial cells of Cajal. Microsc Res Tech. 1999;47:248–266. doi: 10.1002/(SICI)1097-0029(19991115)47:4<248::AID-JEMT4>3.0.CO;2-W. [DOI] [PubMed] [Google Scholar]
- 21.Faussone-Pellegrini MS, Pantalone D, Cortesini C. An ultrastructural study of the interstitial cells of Cajal of the human stomach. J Submicrosc Cytol Pathol. 1989;21:439–460. [PubMed] [Google Scholar]
- 22.Min KW, Seo IS. Intestitial cells of Cajal in the human small intestine: immunochemical and ultrastructural study. Ultrastruct Pathol. 2003;27:67–78. doi: 10.1080/01913120309927. [DOI] [PubMed] [Google Scholar]
- 23.Lee JR, Joshi V, Griffin JW, Jr, Lasota J, Miettinen M. Gastrointestinal autonomic nerve tumor: immunohistochemical and molecular identity with gastrointestinal stromal tumor. Am J Surg Pathol. 2001;25:979–987. doi: 10.1097/00000478-200108000-00001. [DOI] [PubMed] [Google Scholar]