Abstract
In songbirds, neurons that regulate learned song behavior undergo extensive seasonal plasticity in their number and size in relation to the bird's reproductive status. Seasonal plasticity of these brain regions is primarily regulated by changes in circulating concentrations of testosterone. Androgen receptors are present in all of the major song nuclei, but it is unknown whether levels of androgen receptor mRNA in the telencephalic song regions HVC, the robust nucleus of the arcopallium, and the lateral magnocellular nucleus of the anterior nidopallium change as a function of season in white-crowned sparrows. To determine whether seasonal changes in levels of androgen receptor mRNA are specific to the song control system, we also measured levels of androgen receptor mRNA in a limbic nucleus, the the lateral division of the bed nucleus of the stria terminalis (the lateral division of the bed nucleus of the stria terminalis). We found that levels of androgen receptor mRNA were higher in HVC and the lateral division of the bed nucleus of the stria terminalis of birds in the breeding-condition compared with the nonbreeding-condition; however, we observed no seasonal differences in levels of androgen receptor mRNA in either the the robust nucleus of the arcopallium or the lateral magnocellular nucleus of the anterior nidopallium. These results are consistent with previous observations that seasonal plasticity of the song nuclei results fromtestosteronacting directly on HVC, which then exerts transsynaptic trophic effects on its efferent targets. The seasonal change in the expression of androgen receptor in HVC may be one component of the cellular mechanisms underlying androgenic effects on seasonal plasticity of the song control nuclei.
Introduction
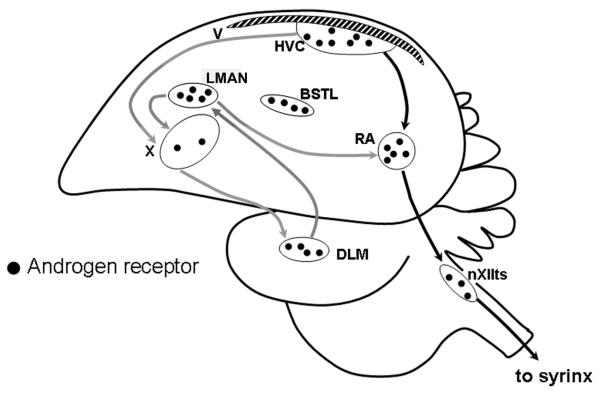
In songbirds, the development and production of song is regulated by discrete nuclei in the brain known as the song control system (Fig.1). Seasonal change in the morphology and electrical activity of this circuit serves as a useful model for understanding hormone-dependent neuroplasticity. Most song nuclei undergo seasonal changes in neuronal number and size, which are related to the bird's reproductive status. Seasonal plasticity of the song nuclei is primarily regulated by seasonal changes in circulating concentrations of testosterone and its androgenic and estrogenic metabolites (reviewed in Brenowitz, 2008). Neurons in all of the major song nuclei express the androgen receptor and its coding mRNA (Metzdorf et al., 1999; Perlman et al., 2003; Schlinger and Brenowitz, 2002; Smith et al., 1996). It is unclear, however, to what degree seasonal plasticity of the song nuclei is accompanied by steroid-induced changes in androgen receptor expression in these regions.
Figure 1.
A schematic sagittal drawing of the songbird brain showing the locations and projections of major nuclei in the song control system. The descending motor pathway (black arrows) controls the production of song by the syrinx. The grey arrows indicate the anterior forebrain pathway that is essential for song learning. It indirectly links HVC to the robust nucleus of the arcopallium, via area X in the parolfactory lobe, DLM in the thalamus, and the lateral magnocellular nucleus of the anterior nidopallium in the neostriatum. the lateral magnocellular nucleus of the anterior nidopallium also projects to area X. the lateral division of the bed nucleus of the stria terminalis is a limbic nucleus. The black dots indicate the presence of androgen receptors. Abbreviations: the lateral division of the bed nucleus of the stria terminalis, the lateral division of the bed nucleus of the stria terminalis; DLM, medial portion of the dorsolateral nucleus of the thalamus; the lateral magnocellular nucleus of the anterior nidopallium, lateral portion of the magnocellular nucleus of the anterior nidopallium; the robust nucleus of the arcopallium, the robust nucleus of the arcopallium; V, lateral ventricle; X, area X; nXIIts, tracheosyringeal part of the hypoglossal nucleus.
In the white-crowned sparrow (Zonotricihia leucophrys gambelli), the song nucleus HVC (acronym used as proper name) has a higher density, number, and percent of androgen receptor-like immunoreactive (ir) cells; moreover, these cells stain more darkly for androgen receptor-ir in breeding than nonbreeding-condition birds (Soma et al., 1998). The increase in the expression of androgen receptor protein in the breeding season may reflect amplified transcription of the androgen receptor gene. On the other hand, it is possible that seasonal regulation of androgen receptor immunoreactivity reflects post-transcriptional and/or post-translational modification of transcripts or protein (Brinkman et al., 1999; Chauchereau et al., 1992; Faus and Haendler, 2006; Mora and Mahesh, 1999). In this case we would not expect to observe a seasonal change in expression of the androgen receptor gene. Fusani et al. (2000) reported that the HVC within one strain of domesticated canary (Serinus canarius), the expression of androgen receptor mRNA does not apparently vary across seasons— despite seasonal variation in circulating levels of T. The goal of the present study was to determine whether the seasonal changes in androgen receptor-ir previously reported in the HVC of white-crowned sparrows reflect seasonal changes in the expression of androgen receptor mRNA. To determine whether any seasonal changes in androgen receptor mRNA levels were specific to the song nuclei, we also examined androgen receptor gene expression in an androgen-sensitive, non-song region— the the lateral division of the bed nucleus of the stria terminalis (the lateral division of the bed nucleus of the stria terminalis). We report that expression of androgen receptor mRNA is increased in both the HVC and the lateral division of the bed nucleus of the stria terminalis as animals transition to the breeding condition. These observations suggest that androgens have important effects on the neuronal circuitry that regulates song but may also target regions of the brain that control other androgen-dependent behaviors.
Material and Methods
Animals
All procedures followed NIH animal use guidelines and were approved by the University of Washington Institutional Animal Care and Use Committee. We captured 12 male Gambel's white-crowned sparrows in eastern Washington during their post-breeding season migration. We housed the birds in indoor group aviaries for at least 10 weeks on short days (8 hrs light) before the start of the experiment to ensure that the song system was sufficiently regressed and sensitive to the stimulatory effects of testosterone and long-day (LD) photoperiods.
We shifted six birds overnight to LD (20 hrs light) and implanted them subcutaneously with a 12mm Silastic capsule (inner diameter 1.47 mm × outer diameter 1.96 mm) filled with crystalline T. The song control nuclei in wild male white-crowned sparrows increase in size in response to a gradual increase in circulating testosterone levels as day length increases and the testes grow. However, the timing of the increase in circulating testosterone levels varies considerably across individuals (Wingfield and Farner, 1978). To reduce individual variability, we exposed all birds to the same long-day photoperiod and administered exogenous T. This treatment rapidly elevates plasma concentrations of testosterone to those seen in breeding birds (4-12 ng/ml) (Wingfield and Farner, 1978). Despite the fact that the transition to breeding condition occurs more gradually in the wild, this laboratory manipulation recreates the two most important seasonal influences on white-crowned sparrows: elevated levels of circulating testosterone and a long-day photoperiod typical of their Alaskan breeding grounds. The other six birds remained on short days. All birds were killed by decapitation 28 days after the long day + testosterone group received testosterone implants. This time period is sufficient to induce full growth of the song control system under these conditions (Tramontin et al., 2000). Birds in both the short day and long day + testosterone groups were killed during the same 3 hour window after the lights came on at the birds' subjective dawn to control for possible circadian rhythms in androgen receptor gene expression.
Blood Collection and Hormone Assays
Blood samples (~ 400 μl) were collected from trunk blood at decapitation into heparinized microcentrifuge tubes. Each blood sample was immediately centrifuged and the plasma was removed and stored at 20°C until assay. We used the Coat-a-Count Total Testosterone kit (Diagnostic Products, Los Angeles, CA) to measure plasma concentrations of T. This assay has been validated for measurement of plasma testosterone levels in birds (Tramontin et al., 2001). The minimum detectable plasma testosterone concentration was 0.2 ng/ml. Interassay variability (CV) was 3.5 ± 0.8%.
Brain Collection and Preparation
We rapidly removed the brains, rinsed them in cold phosphate-buffered saline (PBS), coated them in M1 compound, froze them on dry ice, and stored them at −80°C until sectioning. We sectioned the brains coronally on a cryostat at 20 μm, thaw-mounted sections onto Superfrost Plus™ slides (VWR, Inc., Seattle, WA) in 4 sets, and stored the slides in air-tight boxes at −80°C until in situ hybridization. We analyzed androgen receptor mRNA expression in the song control nuclei HVC, the robust nucleus of the arcopallium, and the lateral magnocellular nucleus of the anterior nidopallium (Fig. 1). To determine whether any changes in expression were specific to the song system, we also analyzed androgen receptor mRNA in the telencephalic the lateral division of the bed nucleus of the stria terminalis, which is part of the extended amygdala complex and as such may be involved in autonomic regulation, neuroendocrine control, and/or sexual behavior (Aste et al., 1998; Atoji et al., 2006; Egli and Winder, 2003; Naftolin et al., 2001).
Androgen Receptor mRNA In Situ Hybridization
Plasmids containing a partial cDNA for songbird androgen receptor (46, generously provided by Dr. Arthur Arnold, UCLA) (Perlman et al., 2003) were cloned as previously described (Fraley, 2006; Fraley et al., 2004; Johnson and Fraley, 2008; Juréus et al., 2000). We linearized androgen receptor cDNA with Not I and transcribed for antisense riboprobes with T7 RNA polymerase (New England Biologicals, Beverly, MA) in the presence of 33P-UTP (Perkin-Elmer, Indianapolis, IN). Sense transcriptions were carried out with T3 RNA polymerase after linearization with EcoRI (Roche Biochemicals, Indianapolis, IN). We determined appropriate riboprobe length with formaldehyde gel electrophoresis. Following purification, we quantified labeling of riboprobes in a scintillation counter.
One out of every four sections containing HVC, the robust nucleus of the arcopallium, the lateral magnocellular nucleus of the anterior nidopallium, and the lateral division of the bed nucleus of the stria terminalis was removed from the freezer, fixed in 4% paraformaldehyde (pH 7.4), washed in phosphate-buffered saline, acetylated in acetic anhydride, delipidized in chloroform, and allowed to air-dry prior to the hybridization procedure. We calculated the volume of probe (0.2 pM/slide), allowed it to thaw on ice, and adjusted the final volume of probe mix plus hybridization buffer to equal to 100 μl/slide. We combined the probe with 1/20 volume yeast tRNA (Roche Biochemicals, Indianapolis, IN) in TE (0.1M tris/0.01M EDTA, pH = 8.0). Probe mix was denatured by placing into boiling water for 3 min and returned to ice for 5 min. We added the denatured probe mix to pre-warmed hybridization buffer at a ratio of 1:4. We added the hybridization mix to the slides. As a control, sense probe was placed over some sections containing the nuclei of interest. We covered the sections with glass coverslips,placed the slides in humidity chambers, and incubated them at 55°C for 16 h.
Following hybridization, we removed the humidity chambers from the ovens and allowed them to return to room temperature. We removed the coverslips and washed sections twice in 4 X SSC for 15 min each at room temperature. We placed the slides in 37 mg/ml RNAse (Roche Biochemicals, Indianapolis, IN) in 0.15 M sodium chloride, 10 mM Tris, 1 mM EDTA, pH 8.0) for 30 min at 37°C, then in RNAse buffer without RNAse at 37°C for another 30 min. After a 3 min wash in 2X SSC at room temperature we washed the slides twice with agitation in 0.1X SSC at 62°C for 30 min each. After a 3 min room temperature wash in 0.1X SSC, sections were dehydrated in graded ethanols (3 min each) and allowed to air-dry. The slides are then dipped in Kodak NTB photographic emulsion and air-dried. Slides were allowed to expose the NTB3 for approximately one week. The emulsion was developed, dehydrated in graded ethanols, cleared in xylene, stained lightly with thionin, and coverslipped with DPX (Sigma, St. Louis, MO). A second set of sections for each nucleus were stained with thionin to facilitate identification of the nuclear boundaries.
Image Processing and Analysis
We analyzed all the sections containing HVC, the robust nucleus of the arcopallium, the lateral magnocellular nucleus of the anterior nidopallium, or the lateral division of the bed nucleus of the stria terminalis from each bird. For each section, we determined the borders of the nuclei based on the thionin stain. We counted the silver grains in three equally sized areas: within the nucleus, outside the nucleus over adjacent tissue, and over glass with no tissue. The within-nucleus, outside nucleus, and no-tissue background grain density (grains/area) measurements for each section were collected under the same light intensity and threshold parameters and the same sized sampling areas were used for both breeding and nonbreeding condition birds. For each nucleus we subtracted the average no-tissue background grain density both from the average within-nucleus density and from the average outside-nucleus density, and then expressed the adjusted data as a ratio of the grain density in the song nucleus over the grain density in the surrounding tissue. We observed some clustering of grains over heavily-labeled cells, but the increased cellular density in the HVC and the lateral division of the bed nucleus of the stria terminalis of breeding males did not allow us to do rigorous analyses of silver grain number per individual cell in each nucleus.
Statistical Analysis
Data for each nucleus were analyzed with a Student's t-test (Sigma Stat) . An alpha level of 0.05 was considered significant for all statistical tests.
Results
Hormone concentrations
Plasma testosterone levels were significantly higher for long day + testosterone birds (10.1 ± 8.7 ng/mL) than for short day males (0.05 ± 0.01 ng/mL, P < 0.02).
Androgen Receptor mRNA In Situ Hybridization
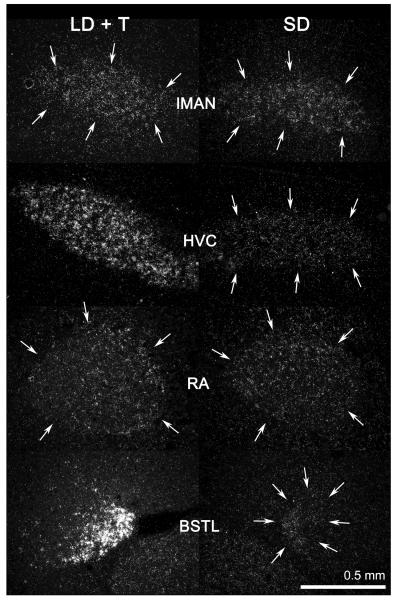
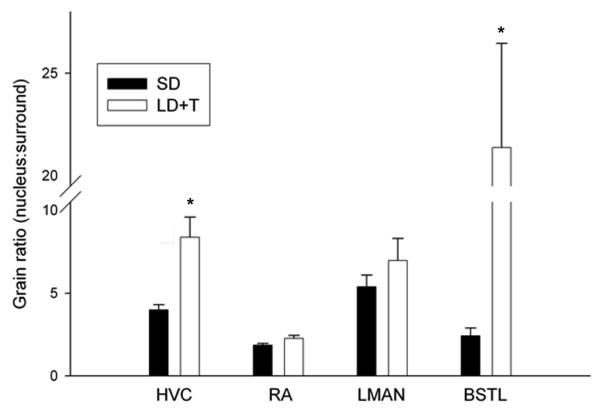
We observed labeling of androgen receptor mRNA in all four nuclei in both treatment groups (Fig. 2). androgen receptor mRNA levels were significantly (P < 0.05) increased by LD + testosterone treatment in HVC and the lateral division of the bed nucleus of the stria terminalis, but did not differ between the long day + testosterone and short day groups in the robust nucleus of the arcopallium or the lateral magnocellular nucleus of the anterior nidopallium (Fig. 3). The seasonal-like difference in androgen receptor mRNA levels was especially pronounced in the the lateral division of the bed nucleus of the stria terminalis. The mean grain ratio for the lateral division of the bed nucleus of the stria terminalis in long day + testosterone birds was nearly 9 fold higher than in short day birds. In the HVC the mean grain ratio was 2.1 fold higher in long day + testosterone birds than in the short day males. Sections covered with sense probe showed no labeling above background level.
Figure 2.
Dark-field photomicrographs showing the expression of androgen receptor mRNA in the song nuclei the lateral magnocellular nucleus of the anterior nidopallium, HVC, and the robust nucleus of the arcopallium, and in the limbic nucleus the lateral division of the bed nucleus of the stria terminalis, in breeding (long day + testosterone) and nonbreeeding (short day) condition white-crowned sparrows. Sections are in the coronal plane
Figure 3.
Quantitative analysis of androgen receptor mRNA expression in the song nuclei HVC, the robust nucleus of the arcopallium, and the lateral magnocellular nucleus of the anterior nidopallium, and the limbic nucleus the lateral division of the bed nucleus of the stria terminalis in breeding (long day + testosterone) and nonbreeding (short day) condition birds. Data are expressed as a ratio of the grain density within the nucleus versus the surrounding tissue. Bars are group means ± SEM. * indicates that the differences between long day + testosterone and short day groups are significant (p < 0.05, t test).
Discussion
We observed an increase in the expression of androgen receptor mRNA in the HVC and the lateral division of the bed nucleus of the stria terminalis across the transition between the non-breeding and breeding condition of the white-crowned sparrows. The 2-fold increase in levels of androgen receptor mRNA in the HVC of breeding birds is comparable in magnitude to the increase in the number of androgen receptor-like immunoreactive cells in HVC of wild breeding white-crowned sparrows (Soma et al., 1998). Together, these observations suggest that the increase in levels of androgen receptor protein in the HVC of breeding birds results primarily from an increase in transcription instead of post-transcriptional and/or post-translational modification of transcripts or protein. These observations are critical to understand the molecular mechanisms of plasticity within song nuclei that are associated with seasonal gonadal recrudescence and increase in circulating testosterone levels.
We manipulated both photoperiod and plasma testosterone levels in the birds. Thus, the increase in levels of androgen receptor mRNA in HVC and the lateral division of the bed nucleus of the stria terminalis could be attributed to changes in either or both of these factors. Prins et al. (1990) reported a T-independent effect of photoperiod on nuclear androgen receptor protein levels in the pituitary (but not hypothalamus) of golden hamsters. However, we would argue that the observed changes in levels of androgen receptor mRNA primarily reflect changes in circulating levels of T. In previous studies we have found that photoperiod alone has only minor effects on the plasticity of the song nuclei compared with the isolated effects of testosterone (Singh et al., 2003; Smith et al., 1997; Wissman and Brenowitz, 2009).
The increase in levels of androgen receptor mRNA in the HVC of breeding birds and the lack of seasonal change in the robust nucleus of the arcopallium and the lateral magnocellular nucleus of the anterior nidopallium are consistent with the effects of testosterone on seasonal plasticity of the song nuclei. testosterone acts directly on HVC neurons, which then have transsynaptic trophic effects on area X and the robust nucleus of the arcopallium, including growth of these nuclei and increased spontaneous firing of action potentials by the robust nucleus of the arcopallium neurons (Brenowitz, 2008). Unilateral lesions of HVC block the growth of the ipsilateral the robust nucleus of the arcopallium and area X in response to LD plus systemic testosterone implant (Brenowitz and Lent, 2001). Intracerebral implants of testosterone near HVC— but not the robust nucleus of the arcopallium— induce the growth of the ipsilateral HVC, the robust nucleus of the arcopallium, and area X (Brenowitz and Lent, 2002). Infusions of 5-α dihydrotestosterone plus 17-β estradiol near HVC, but not the robust nucleus of the arcopallium, increase both neuronal size and spontaneous activity in the ipsilateral the robust nucleus of the arcopallium of short day birds, and infusions of androgen receptor and estrogen receptor antagonists near HVC but not the the robust nucleus of the arcopallium blocks these cellular changes in the robust nucleus of the arcopallium in response to LD plus systemic testosterone (Meitzen et al., 2007). These observations suggest that the effect of testosterone on the brain circuitry involved with song are limited to the HVC.
HVC is the primary site of action for T's action on growth of the song circuits. The seasonal change in the expression of androgen receptor in HVC may be one component of the cellular mechanisms underlying these androgenic effects. Implanting testosterone or infusing androgens and estrogens directly adjacent to the robust nucleus of the arcopallium is insufficient to stimulate growth or increased electrical activity of neurons in this nucleus. The lack of seasonal changes in levels of androgen receptor mRNA in the robust nucleus of the arcopallium may reflect its lack of direct hormone responsiveness of the robust nucleus of the arcopallium (Brenowitz and Lent, 2002; Meitzen et al., 2007). Like the robust nucleus of the arcopallium, the lateral magnocellular nucleus of the anterior nidopallium showed no seasonal changes in the expression of androgen receptor; moreover, the volume of the lateral magnocellular nucleus of the anterior nidopallium does not change across seasons in response to variations in circulating testosterone levels, implying that testosterone has little or no effect the lateral magnocellular nucleus of the anterior nidopallium (Tramontin et al., 2000).
Our results differ from some studies of canaries. Fusani et al. (2000) reported that the expression of androgen receptor mRNA expression in the HVC does not differ between domesticated canaries studied in April compared to birds studied in November, despite there being differences in levels of testosterone between these seasons. Notwithstanding, it is interesting to note that this particular strain of domesticated canaries continues to sing relatively well-structured song outside of the breeding season (Voigt and Leitner, 2008). This is in contrast to white-crowned sparrows, whose song becomes shorter and less stereotyped after the breeding season (Smith et al., 1995). Maintaining a constant level of androgen receptor expression when circulating testosterone concentrations decrease may permit continued androgenic action on HVC neurons, which in turn may allow continued production of well-structured song in nonbreeding canaries. However in a different study of this same strain of canary, Gahr and Metzdorf (1997) reported that levels of androgen receptor mRNA expression in HVC were higher in breeding males compared to nonbreeding males, an observation similar to our current result in white-crowned sparrows. Nastiuk and Clayton (1995) reported in yet another strain of domesticated canary (i.e., Waterslagers) that short-term treatment of ovariectomized females with testosterone decreased levels of androgen receptor mRNA expression in HVC. In general, domesticated canaries show reduced seasonal responsiveness in their physiology and song behavior compared with white-crowned sparrows (e.g., Bentley et al., 2003). The inconsistent reports relating to the effects of changes in circulating plasma levels of testosterone levels on androgen receptor gene transcription in HVC may reflect this reduced seasonality and the long history of domestication of canaries.
Comparison of our results with those of other studies indicates that there can be species differences in the regulation of androgen receptor mRNA levels in the brain by seasonal differences in circulating testosterone levels. In a tropical species, the spotted antbird (Hylophylax naevioides), the expression of androgen receptor mRNA is elevated in nucleus taeniae of the amygdala in the nonbreeding season, when circulating testosterone levels are low (Canoine et al., 2007). Antbirds aggressively defend territories year-round, and it may be that the elevation of androgen receptor expression in nonbreeding birds increases the brain's sensitivity to androgens when testosterone levels are reduced. Nucleus taeniae of the amygdala is analogous to the amygdala and is thought to play a role in aggression (Soma et al., 2003). Nucleus taeniae of the amygdala is located in the arcopallium, adjacent to where the robust nucleus of the arcopallium is located in songbirds. Taken together, these and the current observations indicate that even in adjacent brain regions the influence of circulating testosterone levels on androgen receptor expression is dynamic and varies in functional context across species.
Increased expression of androgen receptor mRNA in HVC and the lateral division of the bed nucleus of the stria terminalis in LD + T-treated birds shows that androgen receptor mRNA levels are up-regulated by testosterone in these regions. However, the lack of change in androgen receptor mRNA in the robust nucleus of the arcopallium and the lateral magnocellular nucleus of the anterior nidopallium suggests that at low levels the androgen receptor gene is constitutively expressed in these regions, independent of changes in circulating levels of T. Regional differences in the regulation of steroid receptor expression have been observed in other model systems. For example, Kerr et al. (1995) reported that androgen receptor mRNA decreases concomitantly with age-related declines in circulating testosterone levels in the rat hippocampus, but not in the cortex or hypothalamus. Also in the rat, the expression of estrogen receptor mRNA in the uterus is negatively regulated by circulating levels of estradiol, but positively regulated in the liver and pituitary (Shupnik et al., 1989). In ring doves, progestin receptor levels are positively regulated by testosterone in the hypothalamus, but not regulated in either the hyperpallium or midbrain (Balthazart et al., 1980). Thus, our observations of regional variability in T's influence on androgen receptor mRNA corroborate those earlier studies.
The increased expression of androgen receptor mRNA that we observed in HVC and the the lateral division of the bed nucleus of the stria terminalis associated with increased circulating testosterone levels differs from other studies that showed a negative regulation of androgen receptor expression by testosterone in other brain regions and peripheral organs, such as the prostate and testes (Blok et al., 1992; Quarmby et al., 1990; Takane et al., 1990). The positive regulation of androgen receptor mRNA that we observed in the HVC and the the lateral division of the bed nucleus of the stria terminalis, however, has also been reported in many other studies (Fraley and Ulibarri, 2002; Gonzalez-Cadavid et al., 1993; Jordan et al., 1997; Resko et al., 1993; Tsang et al., 2008; Yang and Arnold, 2000). In goldfish, total brain androgen receptor protein levels were reported to be 5- fold higher in breeding compared with nonbreeding fish (Pasmanik and Callard, 1988). The regulation of androgen receptor mRNA levels may involve highly dynamic mechanisms that are dependant upon the cellular and tissue localization of the androgen receptor transcript.
The most pronounced photoperiod/T-induced changes in levels of androgen receptor mRNA occurred in the the lateral division of the bed nucleus of the stria terminalis. In short day birds there was sparse expression of androgen receptor mRNA, whereas the LD+ T-treated birds showed a the robust and significant increase in the expression of androgen receptor mRNA levels. The specific function(s) of the avian the lateral division of the bed nucleus of the stria terminalis are unclear. Copulation and/or appetitive sexual behavior increases the number of fos-immunoreactive cells in the lateral division of the bed nucleus of the stria terminalis of male Japanese quail (Tlemcani et al., 2000), suggesting the the lateral division of the bed nucleus of the stria terminalis is involved in reproductive behaviors. The change in androgen receptor gene expression we observed could be related to seasonal changes in hormone-regulated breeding behavior in white-crowned sparrows. Given the extensive connectivity of the the lateral division of the bed nucleus of the stria terminalis with other limbic structures of the brain (Atoji et al., 2006), this nucleus represents an interesting focus for future studies of hormone-mediated seasonal plasticity in the songbird brain.
The purpose of these experiments was to determine the effects of seasonal-like changes in photoperiod and plasma testosterone levels on androgen receptor mRNA expression in different brain nuclei involved with song and reproductive behaviors in songbirds. We observed that increased testosterone levels increased androgen receptor mRNA in the HVC and the the lateral division of the bed nucleus of the stria terminalis, but not in other nuclei involved with song behavior. These observations suggest that the influence of testosterone on seasonal plasticity of HVC and perhaps the the lateral division of the bed nucleus of the stria terminalis is mediated by changes in androgen receptor transcription and that non-transcriptional regulatory mechanisms may be more important in the robust nucleus of the arcopallium and the lateral magnocellular nucleus of the anterior nidopallium. These regional differences in the effect of changes in circulating testosterone on androgen receptor mRNA expression present opportunities for future investigation.
Acknowledgements
We thank Dr. Arthur Arnold for his generous gift of the androgen receptor cDNA probe. This work was supported by NIH R01 MH53032 and K02 MH66939 to EAB and the Eunice Kennedy Shriver National Institutes of Child Health and Human Development (NICHD) cooperative agreement U54 HD-12629 as part of the Specialized Cooperative Centers Program in Reproduction and Infertility Research and R01 HD-027142 to the robust nucleus of the arcopalliumS . The contents of this manuscript are solely the responsibility of the authors and do not necessarily represent the official views of the NICHD or the NIMH.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Aste N, Balthazart J, Absil P, Grossmann R, Mulhbauer E, Viglietti-Panzica C, Panzica GC. Anatomical and neurochemical definition of the nucleus of the stria terminalis in Japanese quail (Coturnix japonica) J Comp Neurol. 1998;396:141–57. [PubMed] [Google Scholar]
- Atoji Y, Saito S, Wild JM. Fiber connections of the compact division of the posterior pallial amygdala and lateral part of the bed nucleus of the stria terminalis in the pigeon (Columba livia) J Comp Neurol. 2006;499:161–82. doi: 10.1002/cne.21042. [DOI] [PubMed] [Google Scholar]
- Balthazart J, Blaustein JD, Cheng MF, Feder HH. Hormones modulate the concentration of cytoplasmic progestin receptors in the brain of male ring doves (Streptopelia risoria) J Endocrinol. 1980;86:251–61. doi: 10.1677/joe.0.0860251. [DOI] [PubMed] [Google Scholar]
- Bentley GE, Audage NC, Hanspal EK, Ball GF, Hahn TP. Photoperiodic response of the hypothalamo-pituitary-gonad axis in male and female canaries, Serinus canaria. J Exp Zoolog Part A Comp Exp Biol. 2003;296:143–51. doi: 10.1002/jez.a.10245. [DOI] [PubMed] [Google Scholar]
- Blok LJ, Hoogerbrugge JW, Themmen AP, Baarends WM, Post M, Grootegoed JA. Transient down-regulation of androgen receptor messenger ribonucleic acid (mRNA) expression in Sertoli cells by follicle-stimulating hormone is followed by up-regulation of androgen receptor mRNA and protein. Endocrinology. 1992;131:1343–9. doi: 10.1210/endo.131.3.1324158. [DOI] [PubMed] [Google Scholar]
- Brenowitz EA. Plasticity of the song control system in adult birds. In: Zeigler HP, Marler P, editors. Neuroscience of birdsong. Cambridge University Press; Cambridge: 2008. pp. 332–349. [Google Scholar]
- Brenowitz EA, Lent K. Afferent input is necessary for seasonal growth and maintenance of adult avian song control circuits. J Neurosci. 2001;21:2320–9. doi: 10.1523/JNEUROSCI.21-07-02320.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenowitz EA, Lent K. Act locally and think globally: intracerebral testosterone implants induce seasonal-like growth of adult avian song control circuits. Proc Natl Acad Sci U S A. 2002;99:12421–6. doi: 10.1073/pnas.192308799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brinkman AO, Blok LJ, de Ruiter PE, Doesburg P, Steketee K, Berrevoets CA, Trapman J. Mechanisms of androgen receptor activation and function. J. Steroid Biochem Mol Biol. 1999;69:307–313. doi: 10.1016/s0960-0760(99)00049-7. [DOI] [PubMed] [Google Scholar]
- Canoine V, Fusani L, Schlinger B, Hau M. Low sex steroids, high steroid receptors: Increasing the sensitivity of the nonreproductive brain. Dev Neurobiol. 2007;67:57–67. doi: 10.1002/dneu.20296. [DOI] [PubMed] [Google Scholar]
- Chauchereau A, Savouret JF, Milgrom E. Control of biosynthesis and post-transcriptional modification of the progesterone receptor. Biol Reprod. 1992;46:174–177. doi: 10.1095/biolreprod46.2.174. [DOI] [PubMed] [Google Scholar]
- Egli RE, Winder DG. Dorsal and ventral distribution of excitable and synaptic properties of neurons of the bed nucleus of the stria terminalis. J Neurophysiol. 2003;90:405–14. doi: 10.1152/jn.00228.2003. [DOI] [PubMed] [Google Scholar]
- Faus H, Haendler B. Post-translational modifications of steroid receptors. Biomedecine & Pharmacotherapy. 2006;60:520–528. doi: 10.1016/j.biopha.2006.07.082. [DOI] [PubMed] [Google Scholar]
- Fraley GS. Immunolesions of glucoresponsive projections to the arcuate nucleus alter glucoprivic-induced alterations in food intake, luteinizing hormone secretion, and GALP mRNA, but not sex behavior in adult male rats. Neuroendocrinology. 2006;83:97–105. doi: 10.1159/000094375. [DOI] [PubMed] [Google Scholar]
- Fraley GS, Scarlett JM, Shimada I, Teklemichael DN, Acohido BV, Clifton DK, Steiner RA. Effects of diabetes and insulin on the expression of galanin-like peptide in the hypothalamus of the rat. Diabetes. 2004;53:1237–42. doi: 10.2337/diabetes.53.5.1237. [DOI] [PubMed] [Google Scholar]
- Fraley GS, Ulibarri C. Development of androgen receptor and p75(NTR) mRNAs and peptides in the lumbar spinal cord of the gerbil. Brain Res Dev Brain Res. 2002;137:101–14. doi: 10.1016/s0165-3806(02)00412-1. [DOI] [PubMed] [Google Scholar]
- Fusani L, Van't Hof T, Hutchison JB, Gahr M. Seasonal expression of androgen receptors, estrogen receptors, and aromatase in the canary brain in relation to circulating androgens and estrogens. Journal of neurobiology. 2000;43:254–68. [PubMed] [Google Scholar]
- Gahr M, Metzdorf R. Distribution and dynamics in the expression of androgen and estrogen receptors in vocal control systems of songbirds. Brain research bulletin. 1997;44:509–17. doi: 10.1016/s0361-9230(97)00233-5. [DOI] [PubMed] [Google Scholar]
- Gonzalez-Cadavid N, Vernet D, Fuentes Navarro A, Rodriguez JA, Swerdloff RS, Rajfer J. Up-regulation of the levels of androgen receptor and its mRNA by androgens in smooth-muscle cells from rat penis. Mol Cell Endocrinol. 1993;90:219–29. doi: 10.1016/0303-7207(93)90155-d. [DOI] [PubMed] [Google Scholar]
- Johnson MA, Fraley GS. Rat RFRP-3 stimulates GHRH mRNA expression but not puberty or KiSS-1 gene expression in male rats. J. Neuroendocrinol. 2008 doi: 10.1159/000145718. in press. [DOI] [PubMed] [Google Scholar]
- Jordan CL, Padgett B, Hershey J, Prins G, Arnold A. Ontogeny of androgen receptor immunoreactivity in lumbar motoneurons and in the sexually dimorphic levator ani muscle of male rats. The Journal of comparative neurology. 1997;379:88–98. [PubMed] [Google Scholar]
- Juréus A, Cunningham MJ, McClain ME, Clifton DK, Steiner RA. Galanin-like peptide (GALP) is a target for regulation by leptin in the hypothalamus of the rat. Endocrinology. 2000;141:2703–6. doi: 10.1210/endo.141.7.7669. [DOI] [PubMed] [Google Scholar]
- Kerr JE, Allore RJ, Beck SG, Handa RJ. Distribution and hormonal regulation of androgen receptor (AR) and AR messenger ribonucleic acid in the rat hippocampus. Endocrinology. 1995;136:3213–21. doi: 10.1210/endo.136.8.7628354. [DOI] [PubMed] [Google Scholar]
- Meitzen J, Moore IT, Lent K, Brenowitz EA, Perkel DJ. Steroid hormones act transsynaptically within the forebrain to regulate neuronal phenotype and song stereotypy. J Neurosci. 2007;27:12045–57. doi: 10.1523/JNEUROSCI.3289-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Metzdorf R, Gahr M, Fusani L. Distribution of aromatase, estrogen receptor, and androgen receptor mRNA in the forebrain of songbirds and nonsongbirds. J. Comp. Neurol. 1999;407:115–29. [PubMed] [Google Scholar]
- Mora GR, Mahesh VB. Autoregulation of the androgen receptor at the translational level:: Testosterone induces accumulation of androgen receptor mrna in the rat ventral prostate polyribosomes. Steroids. 1999;64:587–591. doi: 10.1016/s0039-128x(99)00037-9. [DOI] [PubMed] [Google Scholar]
- Naftolin F, Horvath TL, Balthazart J. Estrogen Synthetase (Aromatase) Immunohistochemistry Reveals Concordance Between Avian and Rodent Limbic Systems and Hypothalami. Experimental Biology and Medicine. 2001;226:717–725. doi: 10.1177/153537020222600802. [DOI] [PubMed] [Google Scholar]
- Nastiuk KL, Clayton DF. The canary androgen receptor mRNA is localized in the song control nuclei of the brain and is rapidly regulated by testosterone. J Neurobiol. 1995;26:213–24. doi: 10.1002/neu.480260206. [DOI] [PubMed] [Google Scholar]
- Pasmanik M, Callard GV. A high abundance androgen receptor in goldfish brain: characteristics and seasonal changes. Endocrinology. 1988;123:1162–71. doi: 10.1210/endo-123-2-1162. [DOI] [PubMed] [Google Scholar]
- Perlman WR, Ramachandran B, Arnold AP. Expression of androgen receptor mRNA in the late embryonic and early posthatch zebra finch brain. J Comp Neurol. 2003;455:513–30. doi: 10.1002/cne.10510. [DOI] [PubMed] [Google Scholar]
- Prins GS, Bartke A, Steger R. Influence of photoinhibition, photostimulation and prolactin on pituitary and hypothalamic nuclear androgen receptors in the male hamster. Neuroendocrinology. 1990;52:511–516. doi: 10.1159/000125636. [DOI] [PubMed] [Google Scholar]
- Quarmby VE, Yarbrough WG, Lubahn DB, French FS, Wilson EM. Autologous down-regulation of androgen receptor messenger ribonucleic acid. Mol Endocrinol. 1990;4:22–8. doi: 10.1210/mend-4-1-22. [DOI] [PubMed] [Google Scholar]
- Resko JA, Connolly PB, Roselli CE, Abdelgadir SE, Choate JV. Selective activation of androgen receptors in the subcortical brain of male cynomolgus macaques by physiological hormone levels and its relationship to androgen-dependent aromatase activity. J Clin Endocrinol Metab. 1993;76:1588–93. doi: 10.1210/jcem.76.6.8501167. [DOI] [PubMed] [Google Scholar]
- Schlinger BA, Brenowitz EA. Neural and hormonal control of birdsong. In: Pfaff DW, et al., editors. Hormones, brain and behavior. Academic Press; New York: 2002. pp. 799–839. [Google Scholar]
- Shupnik MA, Gordon MS, Chin WW. Tissue-specific regulation of rat estrogen receptor mRNAs. Mol Endocrinol. 1989;3:660–5. doi: 10.1210/mend-3-4-660. [DOI] [PubMed] [Google Scholar]
- Singh TD, Heinrich JE, Wissman AM, Brenowitz EA, Nordeen EJ, Nordeen KW. Seasonal regulation of NMDA receptor NR2B mRNA in the adult canary song system. J Neurobiol. 2003;54:593–603. doi: 10.1002/neu.10178. [DOI] [PubMed] [Google Scholar]
- Smith GT, Brenowitz EA, Prins GS. Use of PG-21 immunocytochemistry to detect androgen receptors in the songbird brain. J Histochem Cytochem. 1996;44:1075–80. doi: 10.1177/44.9.8773574. [DOI] [PubMed] [Google Scholar]
- Smith GT, Brenowitz EA, Wingfield JC. Roles of photoperiod and testosterone in seasonal plasticity of the avian song control system. J Neurobiol. 1997;32:426–42. doi: 10.1002/(sici)1097-4695(199704)32:4<426::aid-neu6>3.0.co;2-8. [DOI] [PubMed] [Google Scholar]
- Smith GT, Brenowitz EA, Wingfield JC, Baptista LF. Seasonal changes in song nuclei and song behavior in Gambel's white- crowned sparrows. J Neurobiol. 1995;28:114–25. doi: 10.1002/neu.480280110. [DOI] [PubMed] [Google Scholar]
- Soma KK, Hartman VN, Wingfield JC, Brenowitz EA. Seasonal changes in androgen receptor immunoreactivity in the song nucleus HVc of a wild bird. J. Comp. Neurol. 1998;409:224–236. [PubMed] [Google Scholar]
- Soma KK, Schlinger BA, Wingfield JC, Saldanha CJ. Brain aromatase, 5 alpha-reductase, and 5 beta-reductase change seasonally in wild male song sparrows: relationship to aggressive and sexual behavior. J Neurobiol. 2003;56:209–21. doi: 10.1002/neu.10225. [DOI] [PubMed] [Google Scholar]
- Takane KK, George FW, Wilson JD. Androgen receptor of rat penis is down-regulated by androgen. Am J Physiol. 1990;258:E46–50. doi: 10.1152/ajpendo.1990.258.1.E46. [DOI] [PubMed] [Google Scholar]
- Tlemcani O, Ball GF, D'Hondt E, Vandesande F, Sharp PJ, Balthazart J. Fos induction in the Japanese quail brain after expression of appetitive and consummatory aspects of male sexual behavior. Brain Res Bull. 2000;52:249–62. doi: 10.1016/s0361-9230(00)00233-1. [DOI] [PubMed] [Google Scholar]
- Tramontin AD, Hartman VN, Brenowitz EA. Breeding conditions induce rapid and sequential growth in adult avian song control circuits: a model of seasonal plasticity in the brain. J Neurosci. 2000;20:854–61. doi: 10.1523/JNEUROSCI.20-02-00854.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tramontin AD, Perfito N, Wingfield JC, Brenowitz EA. Seasonal growth of song control nuclei precedes seasonal reproductive development in wild adult song sparrows. Gen. Comp. Endocrinology. 2001;122:1–9. doi: 10.1006/gcen.2000.7597. [DOI] [PubMed] [Google Scholar]
- Tsang S, Wu S, Liu J, Wong TM. Testosterone protects rat hearts against ischaemic insults by enhancing the effects of alpha(1)-adrenoceptor stimulation. Br J Pharmacol. 2008;153:693–709. doi: 10.1038/sj.bjp.0707624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voigt C, Leitner S. Seasonality in song behaviour revisited: seasonal and annual variants and invariants in the song of the domesticated canary (Serinus canaria) Horm Behav. 2008;54:373–8. doi: 10.1016/j.yhbeh.2008.05.001. [DOI] [PubMed] [Google Scholar]
- Wingfield JC, Farner DS. The annual cycle of plasma irLH and steroid hormones in feral populations of the white-crowned sparrow, Zonotrichia leucophrys gambelii. Biol Reprod. 1978;19:1046–56. doi: 10.1095/biolreprod19.5.1046. [DOI] [PubMed] [Google Scholar]
- Wissman AM, Brenowitz EA. The role of neurotrophins in the seasonal-like growth of the avian song control system. J Neurosci. 2009 doi: 10.1523/JNEUROSCI.0638-09.2009. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang LY, Arnold AP. BDNF regulation of androgen receptor expression in axotomized SNB motoneurons of adult male rats. Brain Res. 2000;852:127–39. doi: 10.1016/s0006-8993(99)02225-8. [DOI] [PubMed] [Google Scholar]