Abstract
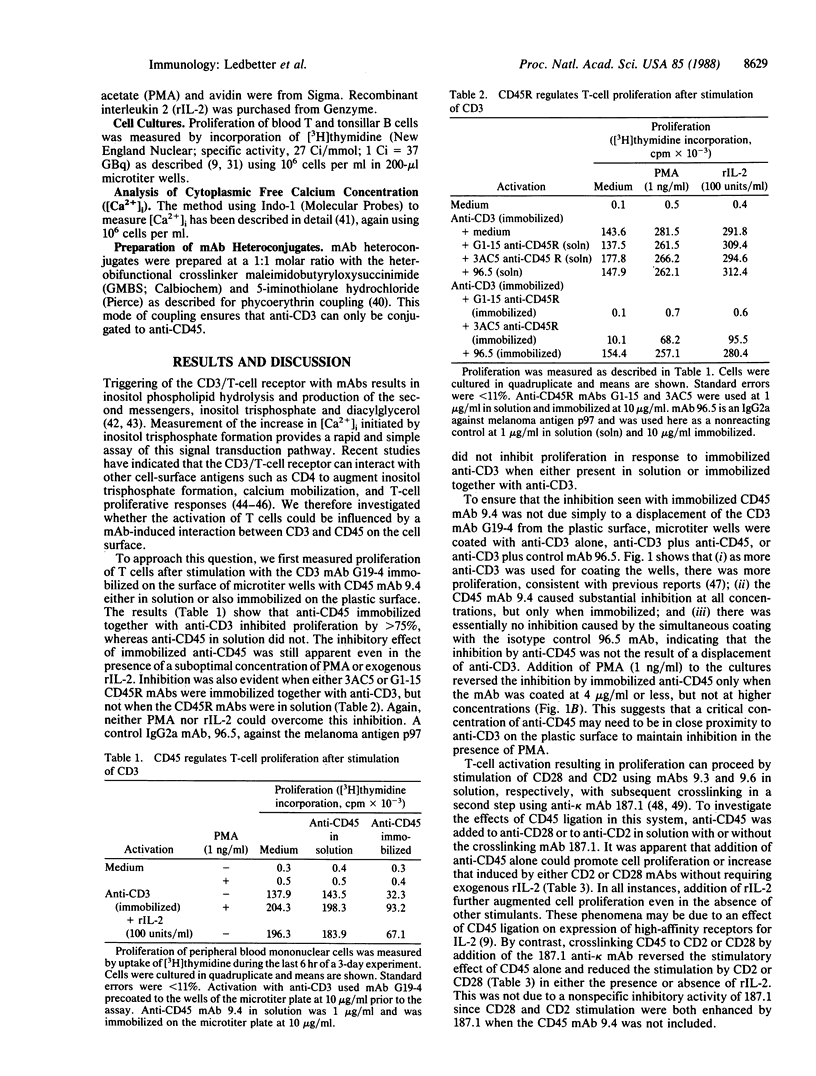
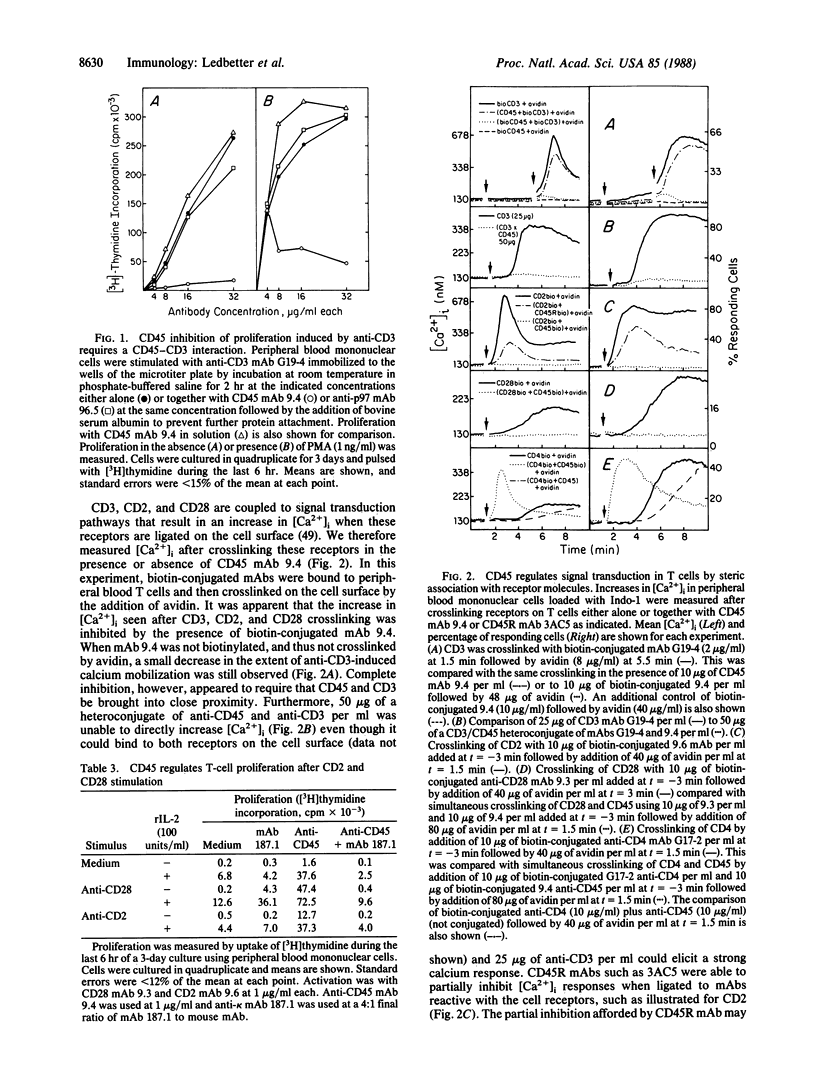
Evidence is presented that the leukocyte common antigen CD45 can regulate both signal transduction by lymphocyte receptor molecules and T- and B-cell proliferation in a manner dependent on specific interactions between these receptors on the cell surface. Formation of homoaggregates of CD3, CD2, or CD28 on the surface of T cells induced by crosslinking with monoclonal antibodies (mAbs) results in an increase in cytoplasmic free calcium concentration ([Ca2+]i). This increase in [Ca2+]i was abolished when these receptors were crosslinked to CD45 on the cell surface. In contrast, the increase in [Ca2+]i induced by formation of homoaggregates of CD4 was strongly amplified when CD4 was coupled to CD45. T-cell proliferation initiated by immobilized anti-CD3 was inhibited by anti-CD45 or anti-CD45R when immobilized on the same surface, but not when in solution. Similarly, proliferation after stimulation of the CD2 and CD28 receptors was inhibited when a CD45 mAb was crosslinked to either CD2 or CD28 mAbs, but not when a CD45-specific mAb was bound to the cell surface separately. In B cells, the increase in [Ca2+]i and resulting proliferation induced by crosslinking either the CD19 or Bgp95 receptors was inhibited by coupling these molecules to CD45. Thus, CD45 appears to modify other cellular receptors functionally when brought into close physical association with them. The homology of the CD45 conserved cytoplasmic domains with a major human placental protein tyrosine phosphatase suggests that the effects of CD45 described here result from alterations in the phosphorylation state of tyrosyl residues in membrane-associated proteins.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Autero M., Gahmberg C. G. Phorbol diesters increase the phosphorylation of the leukocyte common antigen CD45 in human T cells. Eur J Immunol. 1987 Oct;17(10):1503–1506. doi: 10.1002/eji.1830171018. [DOI] [PubMed] [Google Scholar]
- Barclay A. N., Jackson D. I., Willis A. C., Williams A. F. Lymphocyte specific heterogeneity in the rat leucocyte common antigen (T200) is due to differences in polypeptide sequences near the NH2-terminus. EMBO J. 1987 May;6(5):1259–1264. doi: 10.1002/j.1460-2075.1987.tb02362.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernabeu C., Carrera A. C., De Landázuri M. O., Sánchez-Madrid F. Interaction between the CD45 antigen and phytohemagglutinin. Inhibitory effect on the lectin-induced T cell proliferation by anti-CD45 monoclonal antibody. Eur J Immunol. 1987 Oct;17(10):1461–1466. doi: 10.1002/eji.1830171012. [DOI] [PubMed] [Google Scholar]
- Bourguignon L. Y., Suchard S. J., Nagpal M. L., Glenney J. R., Jr A T-lymphoma transmembrane glycoprotein (gp180) is linked to the cytoskeletal protein, fodrin. J Cell Biol. 1985 Aug;101(2):477–487. doi: 10.1083/jcb.101.2.477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Charbonneau H., Tonks N. K., Walsh K. A., Fischer E. H. The leukocyte common antigen (CD45): a putative receptor-linked protein tyrosine phosphatase. Proc Natl Acad Sci U S A. 1988 Oct;85(19):7182–7186. doi: 10.1073/pnas.85.19.7182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark E. A., Ledbetter J. A. Activation of human B cells mediated through two distinct cell surface differentiation antigens, Bp35 and Bp50. Proc Natl Acad Sci U S A. 1986 Jun;83(12):4494–4498. doi: 10.1073/pnas.83.12.4494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark E. A., Ledbetter J. A., Holly R. C., Dinndorf P. A., Shu G. Polypeptides on human B lymphocytes associated with cell activation. Hum Immunol. 1986 May;16(1):100–113. doi: 10.1016/0198-8859(86)90039-x. [DOI] [PubMed] [Google Scholar]
- Clark E. A., Shu G., Ledbetter J. A. Role of the Bp35 cell surface polypeptide in human B-cell activation. Proc Natl Acad Sci U S A. 1985 Mar;82(6):1766–1770. doi: 10.1073/pnas.82.6.1766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalchau R., Fabre J. W. Identification with a monoclonal antibody of a predominantly B lymphocyte-specific determinant of the human leukocyte common antigen. Evidence for structural and possible functional diversity of the human leukocyte common molecule. J Exp Med. 1981 Apr 1;153(4):753–765. doi: 10.1084/jem.153.4.753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalchau R., Kirkley J., Fabre J. W. Monoclonal antibody to a human leukocyte-specific membrane glycoprotein probably homologous to the leukocyte-common (L-C) antigen of the rat. Eur J Immunol. 1980 Oct;10(10):737–744. doi: 10.1002/eji.1830101003. [DOI] [PubMed] [Google Scholar]
- Emmrich F., Kanz L., Eichmann K. Cross-linking of the T cell receptor complex with the subset-specific differentiation antigen stimulates interleukin 2 receptor expression in human CD4 and CD8 T cells. Eur J Immunol. 1987 Apr;17(4):529–534. doi: 10.1002/eji.1830170415. [DOI] [PubMed] [Google Scholar]
- Gelfand E. W., Mills G. B., Cheung R. K., Lee J. W., Grinstein S. Transmembrane ion fluxes during activation of human T lymphocytes: role of Ca2+, Na+/H+ exchange and phospholipid turnover. Immunol Rev. 1987 Feb;95:59–87. doi: 10.1111/j.1600-065x.1987.tb00500.x. [DOI] [PubMed] [Google Scholar]
- Geppert T. D., Lipsky P. E. Accessory cell independent proliferation of human T4 cells stimulated by immobilized monoclonal antibodies to CD3. J Immunol. 1987 Mar 15;138(6):1660–1666. [PubMed] [Google Scholar]
- Harp J. A., Davis B. S., Ewald S. J. Inhibition of T cell responses to alloantigens and polyclonal mitogens by Ly-5 antisera. J Immunol. 1984 Jul;133(1):10–15. [PubMed] [Google Scholar]
- Kamoun M., Martin P. J., Hansen J. A., Brown M. A., Siadak A. W., Nowinski R. C. Identification of a human T lymphocyte surface protein associated with the E-rosette receptor. J Exp Med. 1981 Jan 1;153(1):207–212. doi: 10.1084/jem.153.1.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klausner R. D., O'Shea J. J., Luong H., Ross P., Bluestone J. A., Samelson L. E. T cell receptor tyrosine phosphorylation. Variable coupling for different activating ligands. J Biol Chem. 1987 Sep 15;262(26):12654–12659. [PubMed] [Google Scholar]
- Kupfer A., Singer S. J., Janeway C. A., Jr, Swain S. L. Coclustering of CD4 (L3T4) molecule with the T-cell receptor is induced by specific direct interaction of helper T cells and antigen-presenting cells. Proc Natl Acad Sci U S A. 1987 Aug;84(16):5888–5892. doi: 10.1073/pnas.84.16.5888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ledbetter J. A., June C. H., Grosmaire L. S., Rabinovitch P. S. Crosslinking of surface antigens causes mobilization of intracellular ionized calcium in T lymphocytes. Proc Natl Acad Sci U S A. 1987 Mar;84(5):1384–1388. doi: 10.1073/pnas.84.5.1384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ledbetter J. A., June C. H., Rabinovitch P. S., Grossmann A., Tsu T. T., Imboden J. B. Signal transduction through CD4 receptors: stimulatory vs. inhibitory activity is regulated by CD4 proximity to the CD3/T cell receptor. Eur J Immunol. 1988 Apr;18(4):525–532. doi: 10.1002/eji.1830180406. [DOI] [PubMed] [Google Scholar]
- Ledbetter J. A., Rose L. M., Spooner C. E., Beatty P. G., Martin P. J., Clark E. A. Antibodies to common leukocyte antigen p220 influence human T cell proliferation by modifying IL 2 receptor expression. J Immunol. 1985 Sep;135(3):1819–1825. [PubMed] [Google Scholar]
- Lefrançois L., Bevan M. J. Functional modifications of cytotoxic T-lymphocyte T200 glycoprotein recognized by monoclonal antibodies. Nature. 1985 Apr 4;314(6010):449–452. doi: 10.1038/314449a0. [DOI] [PubMed] [Google Scholar]
- Martorell J., Vilella R., Borche L., Rojo I., Vives J. A second signal for T cell mitogenesis provided by monoclonal antibodies CD45 (T200). Eur J Immunol. 1987 Oct;17(10):1447–1451. doi: 10.1002/eji.1830171010. [DOI] [PubMed] [Google Scholar]
- Mittler R. S., Greenfield R. S., Schacter B. Z., Richard N. F., Hoffmann M. K. Antibodies to the common leukocyte antigen (T200) inhibit an early phase in the activation of resting human B cells. J Immunol. 1987 May 15;138(10):3159–3166. [PubMed] [Google Scholar]
- Morimoto C., Letvin N. L., Distaso J. A., Aldrich W. R., Schlossman S. F. The isolation and characterization of the human suppressor inducer T cell subset. J Immunol. 1985 Mar;134(3):1508–1515. [PubMed] [Google Scholar]
- Nakayama E., Shiku H., Stockert E., Oettgen H. F., Old L. J. Cytotoxic T cells: Lyt phenotype and blocking of killing activity by Lyt antisera. Proc Natl Acad Sci U S A. 1979 Apr;76(4):1977–1981. doi: 10.1073/pnas.76.4.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman W. Selective blockade of human natural killer cells by a monoclonal antibody. Proc Natl Acad Sci U S A. 1982 Jun;79(12):3858–3862. doi: 10.1073/pnas.79.12.3858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Omary M. B., Trowbridge I. S., Battifora H. A. Human homologue of murine T200 glycoprotein. J Exp Med. 1980 Oct 1;152(4):842–852. doi: 10.1084/jem.152.4.842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patarroyo M., Gahmberg C. G. Phorbol 12,13-dibutyrate enhances lateral redistribution of membrane glycoproteins in human blood lymphocytes. Eur J Immunol. 1984 Sep;14(9):781–787. doi: 10.1002/eji.1830140904. [DOI] [PubMed] [Google Scholar]
- Pezzutto A., Dörken B., Rabinovitch P. S., Ledbetter J. A., Moldenhauer G., Clark E. A. CD19 monoclonal antibody HD37 inhibits anti-immunoglobulin-induced B cell activation and proliferation. J Immunol. 1987 May 1;138(9):2793–2799. [PubMed] [Google Scholar]
- Rabinovitch P. S., June C. H., Grossmann A., Ledbetter J. A. Heterogeneity among T cells in intracellular free calcium responses after mitogen stimulation with PHA or anti-CD3. Simultaneous use of indo-1 and immunofluorescence with flow cytometry. J Immunol. 1986 Aug 1;137(3):952–961. [PubMed] [Google Scholar]
- Ralph S. J., Thomas M. L., Morton C. C., Trowbridge I. S. Structural variants of human T200 glycoprotein (leukocyte-common antigen). EMBO J. 1987 May;6(5):1251–1257. doi: 10.1002/j.1460-2075.1987.tb02361.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raschke W. C. Cloned murine T200 (Ly-5) cDNA reveals multiple transcripts within B- and T-lymphocyte lineages. Proc Natl Acad Sci U S A. 1987 Jan;84(1):161–165. doi: 10.1073/pnas.84.1.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudd C. E., Trevillyan J. M., Dasgupta J. D., Wong L. L., Schlossman S. F. The CD4 receptor is complexed in detergent lysates to a protein-tyrosine kinase (pp58) from human T lymphocytes. Proc Natl Acad Sci U S A. 1988 Jul;85(14):5190–5194. doi: 10.1073/pnas.85.14.5190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saga Y., Tung J. S., Shen F. W., Boyse E. A. Alternative use of 5' exons in the specification of Ly-5 isoforms distinguishing hematopoietic cell lineages. Proc Natl Acad Sci U S A. 1987 Aug;84(15):5364–5368. doi: 10.1073/pnas.84.15.5364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saga Y., Tung J. S., Shen F. W., Boyse E. A. Sequences of Ly-5 cDNA: isoform-related diversity of Ly-5 mRNA. Proc Natl Acad Sci U S A. 1986 Sep;83(18):6940–6944. doi: 10.1073/pnas.83.18.6940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seaman W. E., Talal N., Herzenberg L. A., Herzenberg L. A., Ledbetter J. A. Surface antigens on mouse natural killer cells: use of monoclonal antibodies to inhibit or to enrich cytotoxic activity. J Immunol. 1981 Sep;127(3):982–986. [PubMed] [Google Scholar]
- Smith S. H., Brown M. H., Rowe D., Callard R. E., Beverley P. C. Functional subsets of human helper-inducer cells defined by a new monoclonal antibody, UCHL1. Immunology. 1986 May;58(1):63–70. [PMC free article] [PubMed] [Google Scholar]
- Streuli M., Hall L. R., Saga Y., Schlossman S. F., Saito H. Differential usage of three exons generates at least five different mRNAs encoding human leukocyte common antigens. J Exp Med. 1987 Nov 1;166(5):1548–1566. doi: 10.1084/jem.166.5.1548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas M. L., Barclay A. N., Gagnon J., Williams A. F. Evidence from cDNA clones that the rat leukocyte-common antigen (T200) spans the lipid bilayer and contains a cytoplasmic domain of 80,000 Mr. Cell. 1985 May;41(1):83–93. doi: 10.1016/0092-8674(85)90063-7. [DOI] [PubMed] [Google Scholar]
- Thomas M. L., Reynolds P. J., Chain A., Ben-Neriah Y., Trowbridge I. S. B-cell variant of mouse T200 (Ly-5): evidence for alternative mRNA splicing. Proc Natl Acad Sci U S A. 1987 Aug;84(15):5360–5363. doi: 10.1073/pnas.84.15.5360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tonks N. K., Diltz C. D., Fischer E. H. Characterization of the major protein-tyrosine-phosphatases of human placenta. J Biol Chem. 1988 May 15;263(14):6731–6737. [PubMed] [Google Scholar]
- Tonks N. K., Diltz C. D., Fischer E. H. Purification of the major protein-tyrosine-phosphatases of human placenta. J Biol Chem. 1988 May 15;263(14):6722–6730. [PubMed] [Google Scholar]
- Trowbridge I. S., Ralph P., Bevan M. J. Differences in the surface proteins of mouse B and T cells. Proc Natl Acad Sci U S A. 1975 Jan;72(1):157–161. doi: 10.1073/pnas.72.1.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valentine M. A., Clark E. A., Shu G. L., Norris N. A., Ledbetter J. A. Antibody to a novel 95-kDa surface glycoprotein on human B cells induces calcium mobilization and B cell activation. J Immunol. 1988 Jun 15;140(12):4071–4078. [PubMed] [Google Scholar]
- Van Lier R. A., Brouwer M., Aarden L. A. Signals involved in T cell activation. T cell proliferation induced through the synergistic action of anti-CD28 and anti-CD2 monoclonal antibodies. Eur J Immunol. 1988 Jan;18(1):167–172. doi: 10.1002/eji.1830180125. [DOI] [PubMed] [Google Scholar]
- Ware C. F., Reade J. L., Der L. C. A rat anti-mouse kappa chain specific monoclonal antibody, 187.1.10: purification, immunochemical properties and its utility as a general second-antibody reagent. J Immunol Methods. 1984 Nov 16;74(1):93–104. doi: 10.1016/0022-1759(84)90371-5. [DOI] [PubMed] [Google Scholar]
- Weiss A., Imboden J., Hardy K., Manger B., Terhorst C., Stobo J. The role of the T3/antigen receptor complex in T-cell activation. Annu Rev Immunol. 1986;4:593–619. doi: 10.1146/annurev.iy.04.040186.003113. [DOI] [PubMed] [Google Scholar]
- Yakura H., Shen F. W., Bourcet E., Boyse E. A. On the function of Ly-5 in the regulation of antigen-driven B cell differentiation. Comparison and contrast with Lyb-2. J Exp Med. 1983 Apr 1;157(4):1077–1088. doi: 10.1084/jem.157.4.1077. [DOI] [PMC free article] [PubMed] [Google Scholar]



