Abstract
Cadherin-7 (Cad7) and cadherin-6B (Cad6B) are expressed in early and late phases of cranial motoneuron development, respectively. Cad7 is expressed by cranial motoneurons soon after they are generated, as well as in the environment through which their axons extend. By contrast, Cad6B is expressed by mature cranial motoneurons. We demonstrate in chick that these cadherins play distinct roles in cranial motor axon morphology, branching and projection. Using in vitro approaches, we show that Cad7 enhances motor axon outgrowth, suppresses the formation of multiple axons and restricts interstitial branching, thus promoting the development of a single unbranched axon characteristic of differentiating motoneurons. Conversely, Cad6B in vitro promotes motor axon branching, a characteristic of mature motoneurons. In vivo gain- and loss-of-function experiments for these cadherins yielded phenotypes consistent with this interpretation. In particular, a loss of cadherin-mediated interactions in vivo led to dysregulation of the cranial motoneuron normal branching programme and caused axon navigation defects. We also demonstrate that Cad6B functions via the phosphatidylinositol 3-kinase pathway. Together, these data show that Cad7 and Cad6B differentially regulate cranial motoneuron growth, branching and axon guidance.
Keywords: Cadherins, Cad7, Cad6B, Cranial motoneuron, Hindbrain, Branching, Axon guidance, Chick
INTRODUCTION
Developing axons navigate towards their targets by responding to diffusible or contact-mediated cues. Candidates for the latter include members of the cadherin family of calcium-dependent cell adhesion molecules, which are implicated in several aspects of neural development (reviewed by Redies, 2000; Takeichi, 2007). In particular, they are involved in axonogenesis, axon guidance, nucleus formation and synaptogenesis (Andrews and Mastick, 2003; Boscher and Mege, 2008; Iwai et al., 2002; Marthiens et al., 2005; Salinas and Price, 2005; Togashi et al., 2002).
Classic cadherins comprise two structurally similar families, the type I and type II cadherins, both of which possess an extracellular domain composed of five tandem cadherin repeats (EC1-EC5), a single transmembrane domain and a conserved cytoplasmic domain. Cadherin-dependent adhesion occurs via the extracellular domain, which interacts with a cadherin of the same type (Patel et al., 2006). The cytoplasmic domain interacts with the armadillo family catenins, β-catenin or γ-catenin, which in turn bind to α-catenin, an interaction crucial to cadherin function (reviewed by Hirano et al., 2003). These interactions with catenins are also believed to underpin cadherin-dependent regulation of the actin cytoskeleton and membrane dynamics (Nelson, 2008). This influence on actin dynamics underscores the roles that cadherins may play in axon guidance during development. Here, we investigate a role for differential cadherin expression in the growth and guidance of cranial motoneurons.
The branchiomotor (BM) subset of cranial motoneurons is born in columns adjacent to the floor plate at Hamburger and Hamilton stage 14 [embryonic day (E) 2.5] (Hamburger and Hamilton, 1992; Varela-Echavarria et al., 1996). Initial unbranched motor axon outgrowth occurs at stages 14-15 in a lateral direction through the neuroepithelium, with the majority of axons exiting the dorsal hindbrain by stage 17 (E3), and axon trajectories being well established by stages 19-25 (E4-5) (Guthrie and Lumsden, 1992). The first BM axons start to reach the branchial arch muscles at stage 19 (E4) (see Fig. 1B). Concomitant with axon extension into the periphery, BM neuronal somata migrate from their ventral origin and subsequently cluster to form nuclei adjacent to their dorsal exit points (Simon et al., 1994). BM axons initially innervate a single muscle mass within each branchial arch (Fig. 1B), which later splits into a number of muscles, accompanied by branch formation of the motor nerve and arborisation of nerve terminals (McClearn and Noden, 1988). Whereas early BM axon extension has been shown to depend on the action of repellent cues produced by the floor plate (Hammond et al., 2005; Varela-Echavarria et al., 1997), the molecular cues that mediate subsequent stages of BM neuron development remain largely unknown (reviewed by Guthrie, 2007).
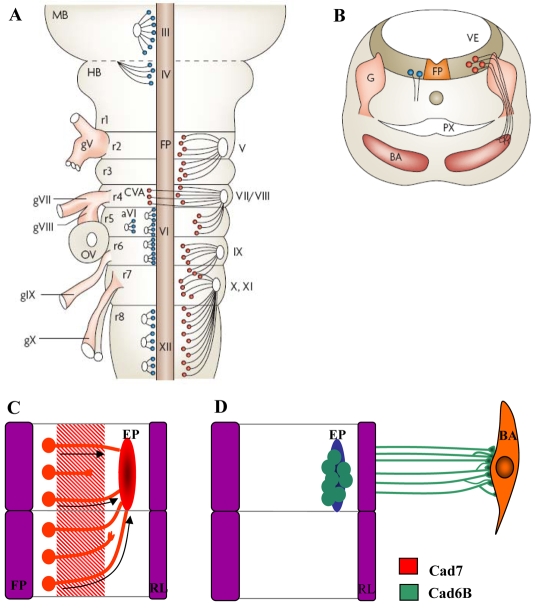
Fig. 1.
Motoneuron development and Cad7 and Cad6B expression in the chick hindbrain. (A) Diagrams of flat-mounted brainstem showing the positions of cranial nerves and motor nuclei. Dorsally projecting branchiomotor (BM) neurons are shown in red, ventrally projecting somatic motoneurons in blue. Roman numerals indicate cranial nerves (III, oculomotor; IV, trochlear; V, trigeminal; VI, abducens; aVI, accessory abducens; VII/VII, facial/vestibuloacoustic; IX, glossopharyngeal; X, vagus; XI, cranial accessory; XII, hypoglossal; gV-X indicate sensory ganglia and cranial nerves as above). Rhombomeres are numbered. (B) Transverse section through the branchial region, showing hindbrain neuroepithelium and branchial arches (BA), with dorsally projecting BM (red) and ventrally projecting somatic motor (blue) motor axons. Adapted with permission from Guthrie (Guthrie, 2007). (C,D) Diagram of flat-mounted even- and odd-numbered rhombomere pairs at early (C) and late (D) developmental stages, with respect to BM axon outgrowth. (C) Cad7 (red) is expressed from E2-5 by the hindbrain neuroepithelium, extending cranial motoneurons and the boundary cap cells at the nerve exit point. (D) Cad6B (green) is expressed by mature motoneurons and their axons between E5 and E9. Arrows indicate the direction of migration of BM neuron somata. Expression data derived from Ju et al. (Ju et al., 2004). MB, midbrain; HB, hindbrain; FP, floor plate; OV, otic vesicle; BA, branchial arch; VE, ventricle; G, ganglion; PX, pharynx; EP, exit point; RL, rhombic lip.
The type II cadherins, cadherin-7 (Cad7) and cadherin-6B (Cad6B), are expressed during early and late phases of chick BM neuron development, respectively (Fig. 1C,D) (Ju et al., 2004). Here, we demonstrate that Cad7 and Cad6B play distinct roles in BM neuron development, consistent with their different expression profiles. Using in vitro and in vivo approaches, we show that Cad7 promotes the growth of a single unbranched axon and regulates axon guidance, whereas Cad6B promotes axon branching, acting via the phosphatidylinositol 3-kinase (PI3K) pathway.
MATERIALS AND METHODS
Embryos
Fertilised chicken eggs (Henry Stewart Farm, Louth, Lincolnshire, UK) were incubated to appropriate stages (Hamburger and Hamilton, 1992) at 37°C in a humidified forced draft air incubator.
Plasmids
myristylated-GFP (control) (Hammond et al., 2005), truncated MNcadΔ-nlacZ (extracellular domain deletion), truncated Cad7 (cytoplasmic and transmembrane deletion; kind gift of Dr M. Vermeren, Cambridge University, UK), full-length Cad7-flag (kind gift of Dr C. Redies, University of Jena, Germany) and full-length Cad6B-nlacZ, myristylated-Akt-GFP (kind gift of Dr W. Snider, University of North Carolina, USA) were used, all utilising a chick β-actin promoter with a CMV enhancer (pCAGGS). shRNA plasmids were Cad7-shRNA-RFP, Cad6B-shRNA-RFP and their control (scrambled sequence) variants, all utilising a chick U6 promoter (Das et al., 2006). Full-length Cad6B-nlacZ, MNcadΔ-nlacZ and full-length Cad7-flag were co-electroporated with myristylated-GFP in order to label the electroporated axons.
Characterisation of shRNA constructs
To verify knockdown of cadherin expression, shRNA constructs for Cad7 or Cad6B (or scrambled controls) were transfected into HEK293 cells together with Cad7-flag or Cad6B-nlslacZ using Fugene reagent (Roche), and immunostained after 48 hours using rabbit anti-flag (1:1000; Sigma) or chicken anti-β-galactosidase (1:1000; Abcam) antibodies (secondary antibodies as below); as controls, cells were transfected with tagged cadherin constructs only. Transfection of shRNAs, but not control shRNAs, reduced the expression of the tagged cadherins by 90-95% relative to controls (see Fig. S1 in the supplementary material). Western blots confirmed a similar reduction in cadherin levels by shRNAs (data not shown).
Dissociated neuron cultures and NIH3T3 cell co-cultures
Cranial motoneurons were grown on laminin-coated coverslips or on monolayers of transfected NIH3T3 cells. Glass coverslips were coated with poly-d-ornithine and laminin (15 μg/ml and 1 mg/ml, respectively; Sigma). NIH3T3 cells were transfected with Cad7-flag or Cad6B-nlacZ [`nucleofection' with cell line kit (Lonza Biosystems, Germany) or mock transfection without DNA as a control] using an Amaxa machine (Lonza Biosystems, Germany) and plated in chamber slides (Lab-Tek). Expression of Cad7 and Cad6B by 50-60% of cells in monolayers was confirmed by immunostaining using mouse anti-flag or chicken anti-β-galactosidase antibodies (both 1:1000; Abcam; data not shown).
For preparation of cranial motoneurons, we used a protocol adapted from Henderson et al. (Henderson et al., 1995), using the ventral portion of the hindbrain, which is enriched in cranial motoneurons at E3 (stage 16-17) or E5 (stage 25-26). Dissociated neurons were plated on laminin-coated coverslips or on NIH3T3 cell monolayers in chamber slides, in Neurobasal medium with 2% (v/v) B27 supplement, 2% (v/v) horse serum, 0.1% (v/v) β-mercaptoethanol, 0.35% (v/v) Glutamax, 1% (v/v) penicillin/streptomycin (all from Gibco), 1% (v/v) chick embryo extract and 50 ng/ml CNTF (R&D Systems). In some experiments, neurons were transfected with full-length Cad7 or full-length Cad6B constructs and myr-GFP, or with the myr-GFP construct alone as a control, using the chick nucleofection kit and an Amaxa machine. Co-expression of myr-GFP with β-galactosidase-tagged Cad6B or flag-tagged Cad7 in neurons was verified by immunostaining (data not shown).
In some experiments, dissociated BM neurons grown on laminin were treated with soluble Cad7 protein, which was generated from medium conditioned by HEK293 cells transfected by a construct encoding Cad7 lacking its cytoplasmic and transmembrane domains (Hammond et al., 2005). Controls for these experiments were treated with medium collected from mock-transfected HEK293 cells.
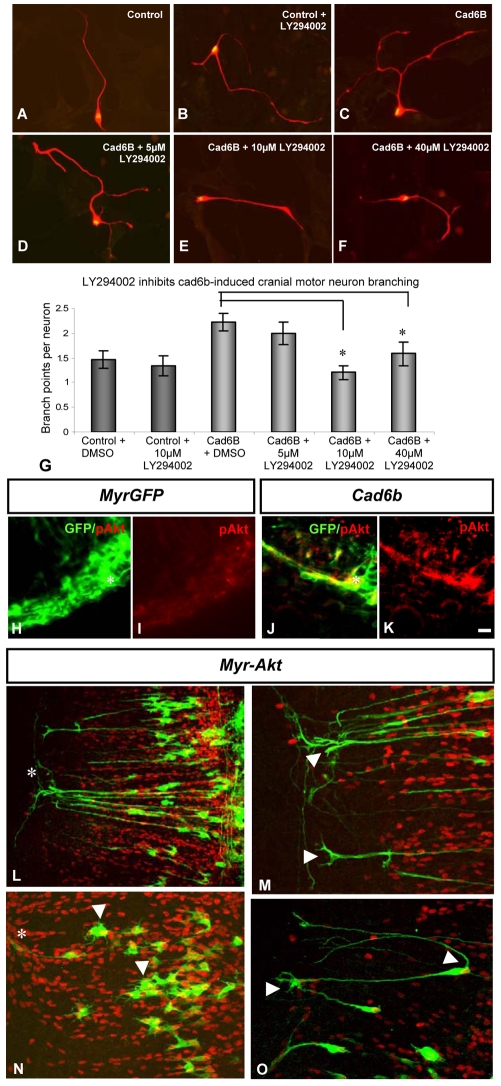
To investigate PI3K signalling, E5 neurons cultured on a Cad6B-expressing monolayer were treated with the PI3K inhibitor LY294002 (Cell Signaling Technologies) 24 hours after plating, at 5 μM, 10 μM and 40 μM. The inhibitor was diluted in pre-warmed culture medium. Controls were treated with dimethyl sulphoxide (DMSO) vehicle (Sigma).
Immunohistochemistry on cultures
Coverslips/chamber slides were fixed for 20 minutes in warmed 4% paraformaldehyde, rinsed in PBS and blocked using PBS containing 1% (v/v) sheep serum (Gibco) and 0.5% (v/v) Triton X-100. For quantitation, cultures were immunostained using mouse anti-Islet1/2 [1:200; 4D5, Developmental Studies Hybridoma Bank (DSHB), Iowa, USA] and rabbit anti-neurofilament H (1:1000; AB1991, Chemicon) antibodies diluted in blocking solution. Secondary antibodies were Alexa Fluor 568 anti-rabbit and Alexa Fluor 488 anti-mouse (1:500; Invitrogen). At least 30 Islet1/2-positive neurons per condition were imaged in three separate experiments. Total axon length was quantitated using SimplePCI software; the number of branch points per neuron and the number of axons extending from the cell body (`polarity') were counted manually. Statistical analysis utilised Student's t-test (two-tailed) or the Mann-Whitney U-test. Some neurons were immunostained using mouse anti-Cad7 or anti-Cad6B antibodies (1:100; DSHB) together with rabbit anti-Islet1/2 antibody (321; kind gift from Dr T. Jessell, Columbia University, USA).
Electroporation of chick embryos in ovo
Electroporation was performed at stage 10-11 (E2) as previously described (Hammond et al., 2005; Momose et al., 1999) and embryos were incubated to E4, E5 or E6. Immunohistochemistry was performed on whole-mount or cryosections as described previously (Hammond et al., 2005), using rabbit or chicken anti-GFP (1:1000; Invitrogen), mouse anti-Islet1/2 (4D5; 1:500), mouse anti-SC1 (1:100; DSHB), chicken anti-β-galactosidase, anti-sarcomeric myosin antibody MF20 (1:250; DSHB) and rabbit anti-phospho-Akt (1:500; Ab 9271, Cell Signaling Technologies). Visualisation of motor axon trajectories in hindbrains electroporated with cadherin gain- and loss-of-function constructs was achieved by co-electroporation of a myristylated-GFP (myr-GFP) construct. To verify co-expression of the constructs in the vast majority of cells, and to confirm expression by motoneurons, hindbrains were immunostained for the cadherin tag (β-galactosidase or flag), GFP and Islet1/2, and/or SC1 (see Fig. S2 in the supplementary material).
RESULTS
The expression patterns of Cad7 and Cad6B in the chick hindbrain were surveyed by Ju et al. (Ju et al., 2004) (Fig. 1C,D). These data show the early expression of Cad7 by radial glia in the hindbrain basal plate from stage 11 (E2) onwards. During stages of axon extension, BM neurons express Cad7 and show high levels of expression at stage 15 (E3) onwards. Boundary cap cells, which lie outside the dorsal exit points and are contacted by BM axons, also express Cad7. Cad7 is expressed throughout successive waves of BM neuron differentiation, starts to be downregulated at ∼stage 25, but persists until stage 30 (E8), when the majority of BM neurons have formed nuclei adjacent to the exit points (Simon et al., 1994). By contrast, Cad6B expression is upregulated during the phase of Cad7 downregulation (stages 25-30; E5-8) (Ju et al., 2004). We therefore tested the hypothesis that Cad7-mediated interactions play a role during the early unbranched outgrowth of cranial motoneurons, whereas Cad6B plays a role during later maturation and branching.
Cad7 and Cad6B are expressed by dissociated cranial motoneurons
We confirmed cadherin protein expression in cultures of dissociated hindbrain neurons, which were used in subsequent experiments to test the effects of cadherins on motor axon growth and branching. Stage 25 (E5) neurons were dissociated and cultured for 48 hours and labelled with anti-Cad7 or anti-Cad6B, and anti-Islet1/2 antibodies. Both cadherins were expressed at this stage, but whereas Cad7 expression appeared in a proximal-to-distal gradient from the cell body (Fig. 2A,C), Cad6B was expressed uniformly (i.e. on cell bodies and axons) and was strongly expressed within the growth cone (Fig. 2B,D).
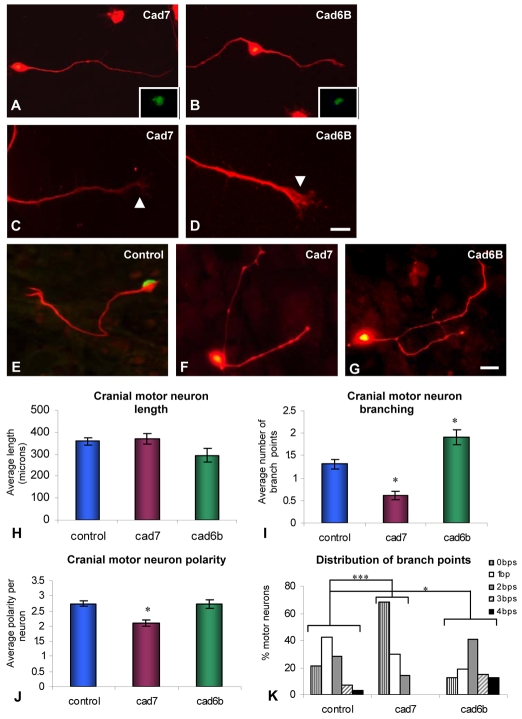
Fig. 2.
Expression of cadherins and responses of cranial motoneurons to culture on cadherin-expressing substrata. (A-D) Immunohistochemistry using anti-Cad7 and anti-Cad6B antibodies in chick E5 cultured cranial motoneurons (red). Insets in A and B show Islet1/2 expression (green). (E-G) E5 cranial motoneurons cultured on control, Cad7- or Cad6B-expressing NIH3T3 cell monolayers. (H-K)The effects of cadherin-mediated interactions on cranial motoneuron morphology (length, polarity and branching). Significant differences from controls are indicated by asterisks (*, P<0.05; ***, P<0.001; Student's t-test in all cases, Xxxxxxx? test); n=90 neurons per condition. bps, branch points. Scale bars: in D, 20 μm for A,B and 10 μm for C,D; in G, 20 μm for E-G.
Cad7 and Cad6B have contrasting effects on cranial motoneuron growth and branching in vitro
We sought to mimic in vitro the different cadherin environments that cranial motoneurons are exposed to in vivo, by challenging dissociated cranial BM neurons to grow on substrata expressing either Cad7 or Cad6B. NIH3T3 cells were mock transfected (control), or transfected with full-length Cad7 or Cad6B constructs and allowed to form a monolayer, on to which dissociated E5 cranial motoneurons were seeded, allowing direct cadherin-mediated interactions to occur (Fig. 2E-K). Cranial motoneurons were visualised by their expression of neurofilament H and Islet1/2 antigens (as above). Three aspects of axonogenesis were quantitated per condition: total neuron length, the total number of branch points per neuron, and the number of axons extending from the cell body, a measure of neuronal `polarity'. We quantitated both the mean number of branch points under a particular condition, and the percentage of neurons with a particular number of branch points (range of 0-4).
Dissociated E5 cranial motoneurons cultured on control or on Cad7-expressing NIH3T3 cells did not show a significant difference in axon length (Fig. 2H), but cranial motoneurons cultured on Cad7-expressing cells showed a significant reduction in the number of branch points compared with the control (an average of 0.6 compared with 1.36 branch points per neuron, respectively; P=3.5×10–5, Student's t-test; P=1×10–5, Mann-Whitney test; Fig. 2I,K). The number of processes extending from the cell body (polarity) was also significantly reduced in the presence of a Cad7-expressing monolayer (an average of 2.1 on Cad7-expressing cells compared with 2.5 on control cells; P=4.7×10–3, Mann-Whitney test; Fig. 2J). This is consistent with Cad7 interactions suppressing cranial motor axon branching and multipolar morphology in E5 neurons and promoting unbranched outgrowth without influencing total axon length.
We next examined the role of Cad6B-mediated interactions and found that, as for Cad7, they had no effect on E5 cranial motor axon length compared with controls (Fig. 2H). However, Cad6B-mediated interactions significantly increased branching (an average of 1.9 branch points on Cad6B-expressing cells compared with 1.31 on control cells; P=0.04, Mann-Whitney test; Fig. 2I,K). Therefore, interactions with Cad6B have the opposite effect to those with Cad7. This is underscored by the fact that the modal number of branch points for control neurons and those grown on Cad7- or Cad6B-expressing cells was 1, 0 or 2, respectively (Fig. 2K). However, culture on Cad6B cells had no significant effect on the number of axons extending from the cell body, indicating that it promotes interstitial branching rather than the development of multiple axons (Fig. 2J).
As Cad7 is expressed before Cad6B in the hindbrain (from E2 onwards), we investigated the effects of Cad7 on the initial growth of cranial motoneurons at E3. Preliminary experiments showed that soluble Cad7 had the same effect on the growth and morphology of E5 neurons as culture on cell monolayers (data not shown). Soluble Cad7 (which contains the extracellular portion only) was applied to cranial motoneurons plated on laminin-coated coverslips. This resulted in significantly longer axons than for control neurons treated with mock-conditioned medium (average total axon length of 344 μm and 201 μm, respectively; P=1.3×10–7, Student's t-test; Fig. 3A-C). However, cranial motoneuron branching and polarity were not affected (Fig. 3D,E).
Fig. 3.
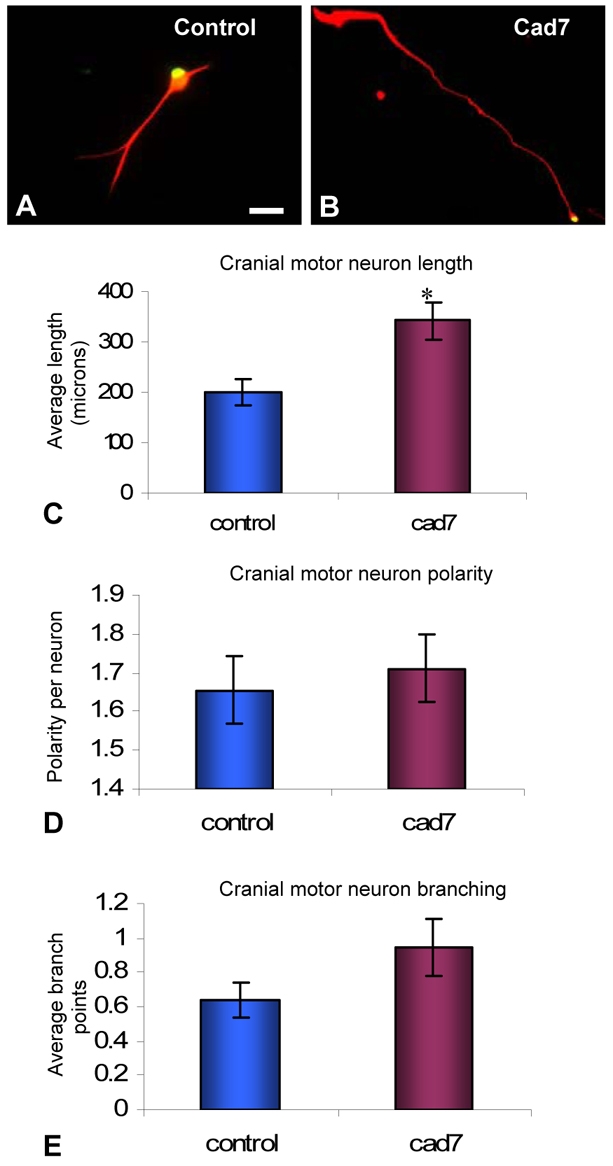
Effects of Cad7-mediated interactions on E3 cultured cranial motoneurons. (A,B) Chick E3 cranial motoneurons cultured on laminin and treated with control medium (A) or soluble Cad7 protein-containing medium (B). Immunostaining is with anti-Islet1/2 (green) and anti-neurofilament H (red) antibodies. (C-E) Effects of soluble Cad7 protein on E3 cranial motoneuron length, polarity and branching. Significant differences from controls are indicated by an asterisk (P≤0.05, Student's t-test); n=90 neurons per condition. Scale bar: in A, 40 μm for A and 50 μm for B.
Together, these data suggest that Cad7- and Cad6B-mediated interactions are required for distinct events during cranial motor axon outgrowth. Cad7 promotes outgrowth of E3 cranial motoneurons but suppresses branching, without affecting overall outgrowth at E5. Cad6B promotes branching of E5 motoneurons, thus enhancing a more mature motoneuron phenotype.
Do heterophilic interactions modulate the effects of Cad7 or Cad6B?
Cranial motoneurons at E5 express both Cad7 and Cad6B. Growth on Cad7-expressing substrata suppresses branching, whereas growth on Cad6B enhances branch formation. We asked whether the additional expression of Cad6B or Cad7 within the neurons would enhance or attenuate the effects of growth on Cad7 or Cad6B substrata, respectively. Dissociated E5 neurons were transfected with myr-GFP (`GFP') alone, myr-GFP and Cad7, or myr-GFP and Cad6B constructs, and were cultured on NIH3T3 cell monolayers. Motor axons were visualised by their expression of GFP and Islet1/2.
We found that the axonal outgrowth and branching of GFP neurons on Cad7 or Cad6B monolayers was very similar to that of untransfected neurons on the same monolayers, indicating that the transfection procedure does not affect cranial motoneuron behaviour (compare Fig. 4A with Fig. 2I). Overexpression of Cad7 by the neurons did not further reduce branching on a Cad7 monolayer in comparison to GFP neurons with endogenous levels of Cad7 (Fig. 4A-C). Overexpression of Cad6b in neurons cultured on a Cad6B monolayer did not cause a significant elevation of branching compared with GFP neurons with endogenous levels of Cad6B cultured on a Cad6B monolayer. These data indicate that expressing higher than endogenous levels of Cad7 or Cad6b on E5 cranial motoneurons does not lead to additional promotion/suppression of branching on a substratum expressing the same cadherin.
Fig. 4.
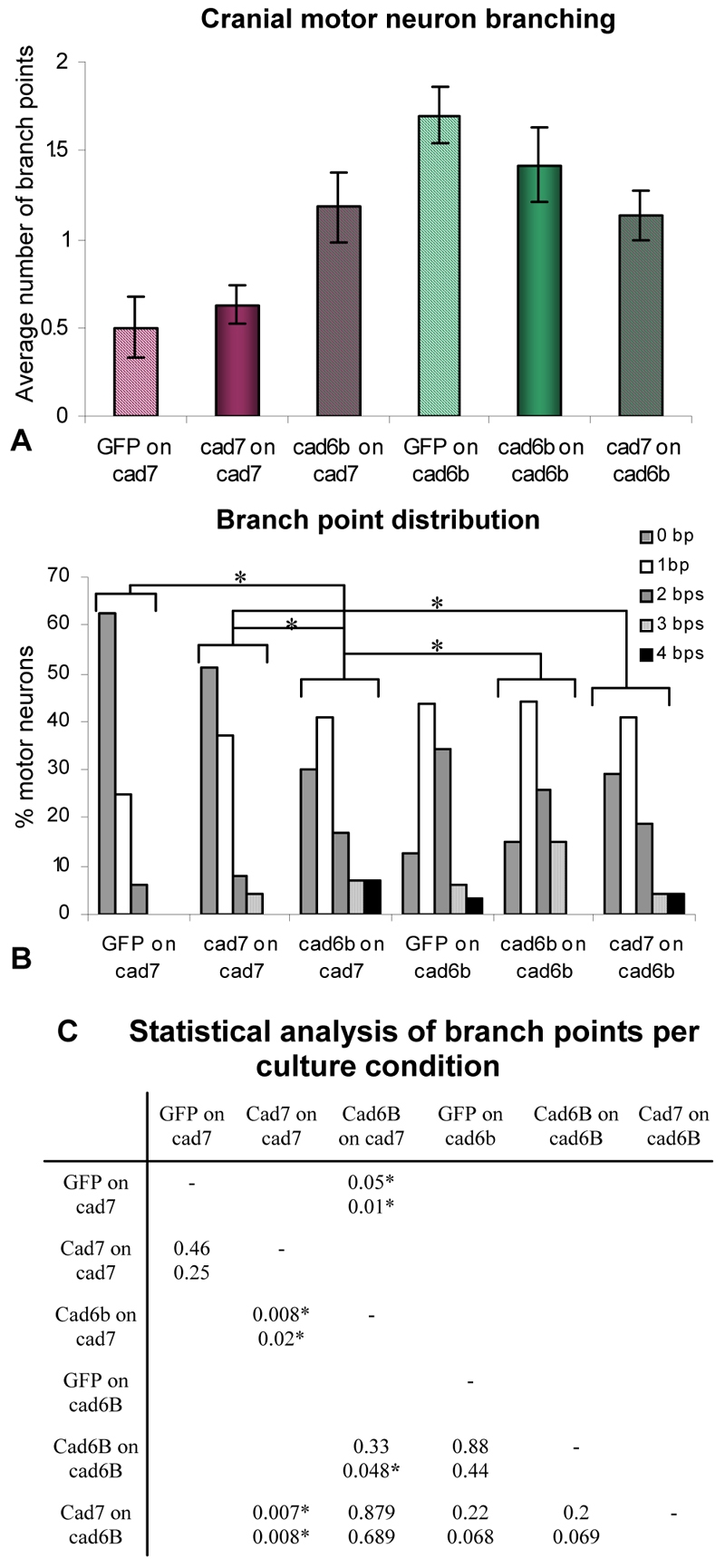
The effects on chick E5 cranial motor axon branching of overexpression of Cad6B or Cad7 and culture on Cad7- or Cad6B-expressing substrata. (A) The effects of Cad7 or Cad6B overexpression by the motoneurons or by the culture environment (NIH3T3 cell monolayers) on the average number of axon branch points. (B) The percentage of motoneurons showing a particular number of branch points per culture condition. Significant differences from controls are indicated by an asterisk (P≤0.05; Mann-Whitney test). (C) Statistical analysis of motoneuron branching based on Student's t-test analysis (upper) of data in A and Mann-Whitney test analysis (lower) of data in B. Statistical differences are indicated by an asterisk (P≤0.05).
We next tested the effect of culturing neurons transfected with additional Cad6B on a Cad7 monolayer. This resulted in a significant increase in branching relative to GFP- or Cad7-expressing neurons grown on a Cad7 monolayer (P=0.05, Student's t-test; Fig. 4A-C). The distribution of branch points between the data sets is also significantly different (P=0.01, Mann-Whitney test, Fig. 4B,C). Thus, whereas control neurons grown in a Cad7 environment show reduced axon branching, the expression of Cad6B on the cranial motoneurons increases branching to levels similar to those of neurons grown on control NIH3T3 cells (Fig. 2). Thus, additional Cad6B expression in the cranial motoneuron exerts an opposite effect to growth on a Cad7-expressing substratum, further indicating that Cad6B and Cad7 exert opposing effects on cranial motor axon growth in vitro. This suggests that heterophilic interactions between Cad6B on the neuron and Cad7 in the environment can mediate an increase in branching.
The converse experiment examined the effect of culturing neurons overexpressing Cad7 on a Cad6B monolayer. This did not result in a significant reduction in branching when compared with GFP neurons or Cad6B-expressing neurons on a Cad6B monolayer (P=0.22 and 0.2, respectively, Student's t-test; P=0.068 and 0.069, respectively, Mann-Whitney test; Fig. 4B). Therefore, Cad7 expression by the neuron cannot abolish the branching effects induced by the Cad6B monolayer, but does attenuate branching induced by the Cad6B monolayer to a level similar to that seen in Cad6B-transfected neurons grown on a Cad7 substrate (P=0.879, Student's t-test). Thus, with upregulation of Cad7 or of Cad6B within the cranial motoneuron, the effect of growth on the NIH3T3 monolayer expressing the other cadherin was diminished.
Furthermore, the branching observed for Cad7-transfected neurons cultured on a Cad6B monolayer was significantly elevated relative to Cad7-transfected neurons on a Cad7 monolayer (P=0.007, Student's t-test; P=0.008, Mann-Whitney test; Fig. 4B), indicating that exogenous Cad6B can elevate branching even when neurons express Cad7. These data strongly suggest that expression of cadherin within the environment and cadherin expression within the cranial motoneuron promote different axon behaviour.
Overexpression of Cad7 or Cad6B in vivo influences BM axon branching and morphology
We next asked whether in vivo manipulation of Cad7 or Cad6B affects cranial motoneuron development. We electroporated full-length Cad7 or Cad6B plasmids into the chick embryo ventral hindbrain at E2, with the myr-GFP construct as a control (Hammond et al., 2005). Cad7 or Cad6B plasmids were co-electroporated with myr-GFP (prior experiments confirmed co-expression of both plasmids; see Fig. S2 in the supplementary material). Embryos were incubated until E4 and then immunostained using anti-GFP and anti-Islet1/2 antibodies to visualise electroporated motoneurons. Electroporation of the myr-GFP construct alone resulted in widespread expression of myr-GFP by BM neurons, which showed normal morphology and axon pathways (n=15; Fig. 5A,B). The axons showed no evidence of branching during their lateral projection (Fig. 5B).
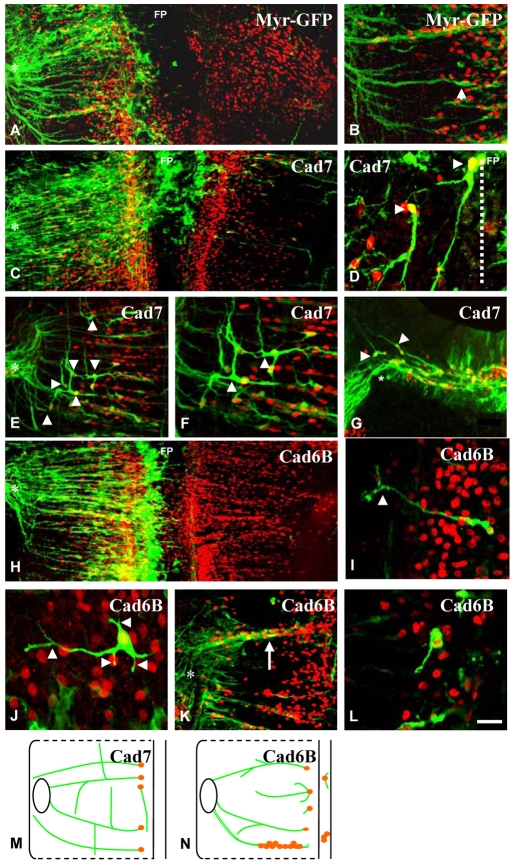
Fig. 5.
Effects of expression of full-length Cad7 and full-length Cad6b constructs in the chick hindbrain. (A-L) E4 hindbrains immunostained with anti-Islet1/2 (red) and anti-GFP (green) antibodies to detect electroporated neurons. Hindbrains were electroporated with myr-GFP control, Cad7 and Cad6B constructs as labelled. All panels show flat-mounts except G, which is a transverse section. Cranial motor axons project in a ventral-to-dorsal direction in controls (arrowhead in B). Medial is to the right and lateral to the left in all panels except A, C and H, which show both sides of the hindbrain. In Cad7-expressing hindbrains, axons project longitudinally (arrowheads in D-F). (F) A high-magnification image of E. In Cad6B-expressing hindbrains, cranial motor axons ectopically branch (I, arrowhead), extend multiple axons (J, arrowheads), aggregate at rhombomere boundaries (white arrow in K), and stall adjacent to the motor column (L). Asterisks indicate exit points. FP, floor plate. (M,N) Diagram of the behaviour of cranial motoneurons electroporated with full-length Cad7 (M) or full-length Cad6B (N). Scale bar: in L, 50 μm for A,C,E,H,K, 75 μm for B,F, 30 μm for G, and 15 μm for D,I,J,L.
Following electroporation with Cad7, the gross pattern of axon projection towards the exit points was normal (Fig. 5C), but some axon guidance defects were observed. Some GFP+ motor axons failed to project away from the floor plate (11/15 embryos), and instead projected longitudinally (Fig. 5D). Other GFP+ axons initially grew laterally but produced a longitudinal branch, often at 90° to the main axon shaft (Fig. 5E,F). This suggests a dysregulation of the normal branching programme and/or a disruption of responses to axon guidance information. Axons sometimes appeared disorganised in the vicinity of the exit point (asterisk in Fig. 5E), and transverse sectioning of Cad7-overexpressing embryos showed that axons overshot their dorsal exit point (6/15 embryos; Fig. 5G). Our interpretation of these data is that Cad7 expression by the radial glia of the neuroepithelium and boundary cap cells of the exit point act as a guidance cue via interactions with Cad7-expressing BM axons. Global expression of Cad7 disrupts these interactions and leads to branching and axon guidance defects, producing longitudinal rather than circumferential axon growth.
Next, we misexpressed full-length Cad6B at E2 (representing a premature onset of Cad6B expression, which normally occurs at ∼E5). This produced no gross disruptions to cranial motoneuron organisation (Fig. 5H). However, abnormal axon branching was observed, with neurons bifurcating even though general lateral extension was correct (16/20 embryos; Fig. 5I,J). In some cases, electroporated motoneurons extended multiple processes (10/20 embryos; Fig. 5J). Electroporation with Cad6B also induced the aggregation of BM axons at rhombomere boundaries (6/20 embryos; Fig. 5K). This suggests that BM axons expressing Cad6B prefer to grow with other Cad6B-positive axons rather than with the Cad7-expressing neuroepithelium. At low frequency, we also observed that Cad6B-expressing axons stalled and failed to exit the hindbrain (2/20 embryos; Fig. 5L).
Thus, premature Cad6B overexpression resulted in phenotypes distinct from those produced by Cad7 overexpression (see summary schematic in Fig. 5M,N) and similar to those observed upon manipulation of Cad6B function in vitro.
Effects of cadherin loss of function
In order to obtain further in vivo data on the role of cadherin-mediated interactions, a dominant-negative type II cadherin plasmid, or a Cad7 or Cad6B short hairpin RNA (shRNA) plasmid (Das et al., 2006), was electroporated into the chick hindbrain at E2, with analysis as described above.
A truncated form of MN-cadherin (MNcadΔ), which contains the cytoplasmic and transmembrane domains and an nlacZ tag, was co-electroporated with the myr-GFP construct and showed co-expression in the same cells (data not shown). Cadherins lacking their extracellular domain fail to form cadherin-dependent adhesions, and such constructs have been shown to act in a dominant-negative fashion to perturb cadherin function (Fujimori and Takeichi, 1993; Kintner, 1992). Transfection of this construct into BM neurons can abrogate the effects on axon branching of growth on Cad7- and Cad6B-expressing monolayers (data not shown). As Cad7 is expressed from E2 to E5, whereas Cad6B is expressed later, it is likely that Cad7-dependent interactions are disrupted by this manipulation in vivo.
In E4 hindbrains, electroporated motoneurons were identified by immunostaining with anti-GFP and anti-Islet1/2 antibodies as above. As with type II cadherin overexpression, the gross morphology of BM axon tracts was normal (Fig. 6A). However, hindbrains electroporated with MNcadΔ showed defects in BM axon morphology (20/20 electroporated embryos). MNcadΔ-GFP-expressing motor axons branched as they projected away from the floor plate (Fig. 6B) or extended multiple axons (Fig. 6C), suggesting a loss of polarisation. These data are in agreement with our in vitro data demonstrating that Cad7-mediated interactions restrict cranial motor axon branching and polarity. MNcadΔ-GFP-expressing motor axons were found ectopically located within, or entering, the floor plate and extending longitudinally (10/20 embryos) (Fig. 6D), suggesting that Cad7-mediated interactions are required for lateral growth from the midline.
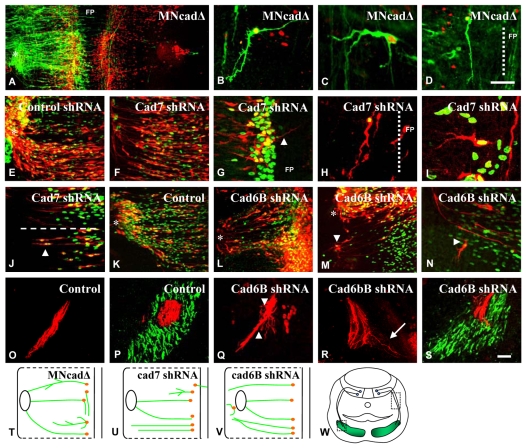
Fig. 6.
Effects of expression of dominant-negative MNcadΔ, Cad7-shRNA and Cad6b-shRNA constructs in the chick hindbrain. (A-S) Hindbrains were fixed at E4 (A-J) or E5-6 (K-S) and immunostained with an anti-Islet1/2 antibody (red in A-D and green in E-N) to detect motoneurons, and with an anti-GFP antibody (green in A-D) or showing RFP natural fluorescence (red in E-S) to detect electroporated neurons (electroporation constructs as labelled). (A-N) MNcadΔ-expressing cranial motoneurons display morphological defects, such as an increase in branching (B) and ectopic axons from the cell body (C) and guidance defects, i.e. they project longitudinally (D). Dashed lines indicate the border of floor plate (FP). Cad7-shRNA-expressing axons project into floor plate (arrowhead in G), project longitudinally (H) or branch ectopically (I). Cad6B-shRNA-expressing axons display defects in the vicinity of the exit points, such as failing to turn rostrally (M,N; arrowheads). Asterisks indicate the exit point. (O-S) Transverse cryosections of embryos electroporated with Cad6B-shRNA or control (scrambled) Cad6B-shRNA, as labelled. (O,Q,R) Regions of the peripheral nerve are shown. Arrowheads in Q indicate defasciculation of the nerve and the arrow in R indicates an axon departing from the main axon bundle. (P,S) The muscle plate (boxed region in W) immunostained with MF20 antibody to label muscle (green). (T-V) Diagram of the behaviour of cranial motoneurons electroporated with MNcadΔ, Cad7-shRNA and Cad6B-shRNA plasmids. (W) Diagram of a transverse section through the hindbrain. Scale bars: 100 μm for A, in D, 60 μm for B-D,G-J, 20 μm for E,F; in S, 50 μm for K-S.
We next electroporated shRNA plasmids that were identifiable through RFP fluorescence. Misexpression of a control Cad7-shRNA plasmid containing scrambled sequence resulted in no observable effects on cranial motor axon extension or growth (Fig. 6E). However, electroporation of a Cad7-shRNA plasmid resulted in similar axon growth and pathfinding errors to those described with the dominant-negative MNcadΔ plasmid, further indicating that the effect of MNcadΔ misexpression is likely to be due to disruption of Cad7 function. Following Cad7-shRNA expression, cranial motor axons were observed projecting within or parallel to the floor plate (6/8 embryos; Fig. 6G,H), or with multiple branch points (6/8 embryos; Fig. 6I), similar to the phenotypes exhibited by the MNcadΔ constructs (Fig. 6T,U). We also observed that in some Cad7-shRNA-expressing embryos, BM axons grew laterally but failed to navigate to their exit points (Fig. 6J).
We next electroporated a Cad6B-shRNA plasmid and analysed axon growth at E5-6, which is after the normal onset of Cad6B expression. A control Cad6B-shRNA plasmid containing scrambled sequence did not adversely affect BM axon growth (Fig. 6K). In Cad6B-shRNA-expressing embryos, defects in the early projection of BM neurons were not observed, consistent with Cad6B not playing a role in early axon projection. However, in 8/16 embryos, BM axons were consistently observed overshooting their dorsal exit points (Fig. 6M), or failing to turn towards this region (Fig. 6N,V).
We therefore examined peripheral axon growth in transverse cryosections of electroporated embryos. Scrambled Cad6B-shRNA-RFP+ axons navigated towards the branchial muscle plate (Fig. 6O) and entered the branchial arch muscle in a tightly fasciculated manner (Fig. 6P,W) (Prin et al., 2005). By contrast, electroporation with Cad6B-shRNA resulted in BM axon defasciculation from the nerve bundle en route to the branchial arch (3/3 embryos; Fig. 6Q,R) and also within the branchial arch muscle (Fig. 6S,W). This suggests that Cad6B mediates fasciculation of BM axons en route to their targets.
Cad6b activates the PI3K/Akt pathway
The formation of an E-cadherin–catenin complex has been shown to recruit PI3K to the cell membrane (Xie and Bikle, 2007), and blocking E-cadherin function inhibits activation of the PI3K effector Akt (Calautti et al., 2005). Additionally, in sensory neurons, Akt enhances axon branching (Markus et al., 2002), a phenotype closely related to the effects of Cad6B expression that we have demonstrated in cranial motoneurons. We thus asked whether the PI3K signalling pathway might mediate Cad6B-induced cranial motor axon branching.
We cultured cranial motoneurons on mock-transfected (control) or Cad6B-expressing monolayers in conjunction with the potent PI3K inhibitor LY294002 (Vlahos et al., 1994). On control monolayers, there was no significant difference in branching between cranial motoneurons treated with DMSO (vehicle control) or with 10 μM LY294002 (P=0.66, Student's t-test; Fig. 7A,B,G). Cranial motoneurons cultured on a Cad6B-expressing monolayer in the presence of DMSO showed a significant elevation in branching relative to these controls (2.2 branch points compared with 1.5; Fig. 7C), consistent with our previous results (Fig. 2I). Application of LY294002 at 10 μM and 40 μM attenuated branching to levels equivalent to those for neurons on control monolayers (1.2 and 1.6 branch points, respectively; P=2.7×10–5 and P=0.03, respectively, Student's t-test; Fig. 7 D-G). Therefore, abrogation of PI3K signalling can block Cad6B-dependent BM neuron branching, suggesting that this pathway mediates the Cad6B effects.
Fig. 7.
Evidence that Cad6B signals via the PI3K pathway. (A-F) Chick cranial motoneurons treated with DMSO (control) or with various concentrations of the PI3K inhibitor LY294002 grown on a Cad6B-expressing NIH3T3 cell monolayer. (G) The average number of branch points per neuron per condition. Significant differences from the control are indicated by an asterisk (P≤0.05, Student t-test); n=90 neurons per condition. (H-K) Cryosectioned embryos that have been electroporated with myr-GFP or with myr-GFP and full-length Cad6B, grown to E5-6 and immunostained with anti-pAkt (red) and anti-GFP (green) antibodies. pAkt is upregulated in Cad6B-overexpressing motoneurons (K). Asterisks indicate the ventral neural tube. (L-O) Effects of expression of a constitutively active myr-Akt construct in the hindbrain. Hindbrains were fixed at E5 and immunostained with anti-Islet1/2 (red) and anti-GFP (green) antibodies to detect electroporated motoneurons. Motor axons branch (L,M,O, arrowheads) and fail to extend a single axon, and in some cases project multiple short processes (arrowheads in N). Asterisks indicate ventral neural tube. Scale bar: in K, 20 μm for A-F,H-K, and 10 μm for L-O.
Cad6B upregulates phospho-Akt in cranial motoneurons
We first determined whether the overexpression of Cad6B in vivo enhances phosphorylation of the PI3K effector Akt. Constructs for full-length Cad6B and myr-GFP, or myr-GFP alone (control), were electroporated into the hindbrain at E2 and transverse sections through the hindbrain examined at E5 using immunohistochemistry for GFP and phospho-Akt (pAkt; anti-phospho Ser473). Control, GFP-positive axons extending towards the dorsal BM exit points exhibited a low level of pAkt (Fig. 7H,I). By contrast, in axon bundles electroporated with full-length Cad6B and myr-GFP, pAkt was abundant and colocalised with GFP (Fig. 7J,K).
We next overexpressed a constitutively active form of Akt [myr-Akt (Markus et al., 2002)] in chick hindbrains at E2, and analysed electroporated motoneurons at E5-6. Myr-Akt expression produced striking phenotypes of ectopic branching and multipolar neuronal morphology (14/14 embryos). Many axons showed ectopic interstitial branching, often bifurcating (Fig. 7L,M) in a manner reminiscent of Cad6B-overexpressing neurons (compare with Fig. 5I,J). Many neurons also failed to extend a single axon and displayed multiple processes (Fig. 7N), or extended two axons from the cell body (Fig. 7O). This appears to be a similar, but more extreme, phenotype to that seen in vivo with Cad6B overexpression (e.g. Fig. 5J,L). This is consistent with PI3K mediating the effects of multiple signalling pathways in BM neuron axon specification (polarisation) and branching, including Cad6B effects. Taken together, the effects of PI3K inhibition in vitro and of Cad6B and myr-Akt expression in vivo suggest that PI3K signalling is a likely mediator of Cad6B signalling in BM neurons.
DISCUSSION
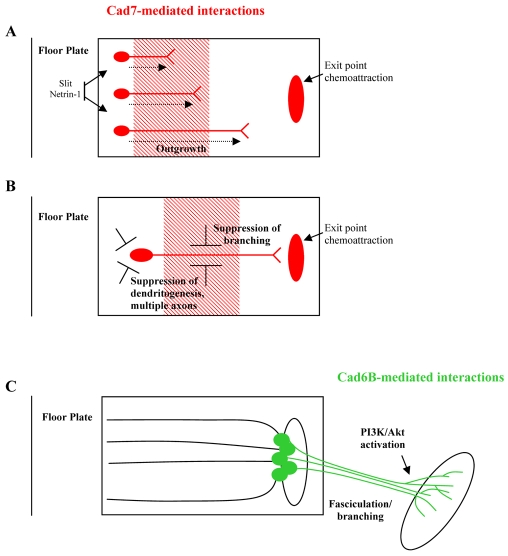
Our findings suggest that Cad7 and Cad6B play crucial roles in distinct aspects of cranial motoneuron development. In vitro, Cad7 interactions promote unbranched outgrowth and guidance, whereas Cad6B promotes branching. Furthermore, additional expression of either Cad6B or Cad7 in cranial motoneurons in vitro is sufficient to reverse the effects on axon branching of culture on substrates expressing the other cadherin. In vivo overexpression of cadherins or attenuation of cadherin-dependent interactions dysregulated the orderly projection of cranial motor axons away from the midline and produced ectopic branching within the hindbrain. Attenuation of Cad6B function also impaired peripheral axon fasciculation, en route to the branchial muscles. Results from in vitro and in vivo experiments suggested that the PI3K signalling pathway mediates Cad6B-dependent axon branching. These data point to an important function of type II cadherins in directing patterns of cranial motor axon extension, branching and guidance. We discuss these findings in the context of a model in which early Cad7 interactions between motor axons and radial glia drives early aspects of axon guidance, whereas later Cad6B interactions between cranial motor axons promotes peripheral cranial motor axon outgrowth (Fig. 8).
Fig. 8.
Model for the role of Cad7- and Cad6B-mediated interactions during BM neuron development in chick. (A-C) Flat-mount hindbrains showing the outgrowth of cranial BM neurons towards their exit points at ∼E3 (A) and ∼E4 (B) and their extension into the periphery at ∼E5 (C). (A) Cad7-mediated interactions (red) promote the early motor axon outgrowth. (B) Cad7-mediated interactions promote axon polarisation and suppress axon branching. (C) Cad6B-mediated interactions (green) promote branchiomotor/visceralmotor axon branching as they approach the branchial arch muscle plate, thus ensuring individual muscle masses are innervated. Cad6B branching is dependent upon the PI3K/Akt pathway.
Cad7 functions as a contact-mediated attractant to promote growth and to suppress multiple axons and axon branching
Soon after their differentiation, post-mitotic cranial motoneurons project a single axon laterally away from the floor plate, with few, if any, interstitial branches (Guthrie and Lumsden, 1992; Hammond et al., 2005; and the present study). We have demonstrated in vitro that in the first phase of this outgrowth at E3, Cad7-mediated interactions promote axon extension (Fig. 8A). This observation is consistent with previous data showing that members of the cadherin family enhance neuron initiation and outgrowth (Andrews and Mastick, 2003; Boscher and Mege, 2008; Marthiens et al., 2005; Piper et al., 2008; Uemura et al., 2007).
Cad7 is expressed by the neuroepithelial cells (`radial glia') of the basal plate, which lie immediately lateral to the site of motoneuron differentiation, and so could act as a guide for Cad7-positive motor axons. As Cad7-mediated interactions in vitro prevent the formation of multiple axons and branches, it is therefore plausible that they promote the establishment of a primary axon facing away from the floor plate (Fig. 8A). The phenotypes produced by gain- and loss-of-function of Cad7 in vivo are broadly consistent with these proposed roles for Cad7. Global overexpression of Cad7 led to a loss of axon projections to exit points and resulted in misorientation of axon projections longitudinally. Our interpretation of these phenotypes is that global, rather than patterned, expression of Cad7 impairs the mediolateral directionality of outgrowth. Phenotypes caused by dominant-negative cadherin expression and Cad7-shRNA caused an abnormal motoneuron morphology, with ectopic branching, misorientation and some axon guidance defects (e.g. longitudinal projection), consistent with a role for Cad7 in unbranched circumferential axon growth.
Additionally, Cad7 is expressed by the neural crest-derived boundary cap cells of the exit point which BM axons traverse (Niederlander and Lumsden, 1996), and might act as an exit point-derived chemoattractant (Guthrie and Lumsden, 1992).
Taken together, these data suggest that Cad7-mediated interactions are required to ensure BM axon projection away from the midline and to suppress inappropriate multiple axon extension and interstitial branching.
Cad6B functions to promote branching and peripheral axon growth
Cad6b is expressed by BM neurons and their axons (but not by their environment) at a stage when most motoneurons have been generated and have migrated dorsally towards their exit points (Ju et al., 2004). We therefore propose that Cad6B does not act as a directional guidance cue, but functions homophilically between adjacent BM neurons/axons (Fig. 8C). Global and premature overexpression of Cad6B in vivo led to the production of multiple axons, ectopic interstitial branching and fasciculation of BM axons in the hindbrain, whereas loss of function led to axonal defasciculation peripherally. In vitro assays have suggested that Cad6B-mediated interactions are stronger than those mediated by Cad7 (Nakagawa and Takeichi, 1998). Cad6B might therefore be required for the coalescence of cell bodies within nuclei and for the integrity of peripheral nerves, perhaps to guide `late'-extending axons along pre-existing tracts, consistent with the observation that axons selectively fasciculate with axons expressing the same cadherins (Redies et al., 1992; Treubert-Zimmermann et al., 2002).
Molecular mechanisms of cadherin-mediated interactions
Here we have demonstrated that Cad6B-mediated interactions activate the PI3K pathway, as has previously been shown for other cadherins (Calautti et al., 2005; Xie and Bikle, 2007). Overexpression of Cad6B caused an increase in Akt phosphorylated at Ser473. Our in vivo results also show that myr-Akt overexpression causes a loss of neuronal polarisation, probably owing to activation of PI3K signalling all around neurons, rather than in the leading process destined to become the axon. In C. elegans, PI3K signalling is required for the colocalisation of various signalling components and to create a stable leading process during neuronal polarisation (Adler et al., 2006).
Although PI3K is often required for axon extension per se, e.g. in cadherin-11-dependent axon growth (Boscher and Mege, 2008), we found that PI3K inhibition did not block BM axon outgrowth. Rather, both the in vitro data using PI3K inhibitors and the in vivo effect of myr-Akt suggest that the PI3K pathway regulates BM neuron interstitial branching. This is in accordance with a previous study that demonstrated that active Akt increases branching by sensory neurons (Markus et al., 2002). Cad6B-PI3K interaction presumably occurs following Cad6B recruitment of β-catenin, which in turn binds to PI3K, resulting in Akt recruitment to the plasma membrane (Xie and Bikle, 2007) and the initiation of new branch points along the axon shaft.
Cad7 and Cad6B collaborate with other cues to guide cranial motor axons
It is likely that Cad7 and Cad6B collaborate with the repertoire of both repulsive and attractive guidance cues that are required for BM axon guidance (reviewed by Guthrie, 2007). We have shown that Netrin-1, Slit1/2 and possibly Sema3A signalling repel BM axons away from the floor plate (Varela-Echavarria et al., 1997; Hammond et al., 2005). Cad7-mediated attraction from the basal plate could assist this repulsion to guide axons dorsally. Hepatocyte growth factor derived from branchial muscles and possibly other peripherally derived neurotrophic factors promote the growth of cranial motor axons and guide them to muscle targets (Caton et al., 2000; Naeem et al., 2002). Cad6B-mediated contact attraction might be an adjunct to these mechanisms in peripheral axon guidance.
Supplementary Material
Acknowledgments
We are indebted to the Wellcome Trust (grants to S.C.G. and S.R.P.) and the Medical Research Council (PhD studentship to S.H.B.) for funding this work, and to Dr U. Drescher for helpful discussions on the manuscript. Deposited in PMC for release after 6 months.
Competing interests statement
The authors declare no competing financial interests.
Supplementary material
Supplementary material for this article is available at http://dev.biologists.org/lookup/suppl/doi:10.1242/dev.042457/-/DC1
References
- Adler C. E., Fetter R. D., Bargmann C. I. (2006). UNC-6/Netrin induces neuronal asymmetry and defines the site of axon formation. Nat. Neurosci. 9, 511-518 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews G. L., Mastick G. S. (2003). R-cadherin is a Pax6-regulated, growth-promoting cue for pioneer axons. J. Neurosci. 23, 9873-9880 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boscher C., Mege R. M. (2008). Cadherin-11 interacts with the FGF receptor and induces neurite outgrowth through associated downstream signalling. Cell Signal. 20, 1061-1072 [DOI] [PubMed] [Google Scholar]
- Calautti E., Li J., Saoncella S., Brissette J. L., Goetinck P. F. (2005). Phosphoinositide 3-kinase signaling to Akt promotes keratinocyte differentiation versus death. J. Biol. Chem. 280, 32856-32865 [DOI] [PubMed] [Google Scholar]
- Caton A., Hacker A., Naeem A., Livet J., Maina F., Bladt F., Klein R., Birchmeier C., Guthrie S. (2000). The branchial arches and HGF are growth-promoting and chemoattractant for cranial motor axons. Development 127, 1751-1766 [DOI] [PubMed] [Google Scholar]
- Das R. M., Van Hateren N. J., Howell G. R., Farrell E. R., Bangs F. K., Porteous V. C., Manning E. M., McGrew M. J., Ohyama K., Sacco M. A., et al. (2006). A robust system for RNA interference in the chicken using a modified microRNA operon. Dev. Biol. 294, 554-563 [DOI] [PubMed] [Google Scholar]
- Fujimori T., Takeichi M. (1993). Disruption of epithelial cell-cell adhesion by exogenous expression of a mutated nonfunctional N-cadherin. Mol. Biol. Cell 4, 37-47 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guthrie S. (2007). Patterning and axon guidance of cranial motor neurons. Nat. Rev. Neurosci. 8, 859-871 [DOI] [PubMed] [Google Scholar]
- Guthrie S., Lumsden A. (1992). Motor neuron pathfinding following rhombomere reversals in the chick embryo hindbrain. Development 114, 663-673 [DOI] [PubMed] [Google Scholar]
- Hamburger V., Hamilton H. L. (1992). A series of normal stages in the development of the chick embryo. Dev. Dyn. 195, 231-272 [DOI] [PubMed] [Google Scholar]
- Hammond R., Vivancos V., Naeem A., Chilton J., Mambetisaeva E., Andrews W., Sundaresan V., Guthrie S. (2005). Slit-mediated repulsion is a key regulator of motor axon pathfinding in the hindbrain. Development 132, 4483-4495 [DOI] [PubMed] [Google Scholar]
- Hirano S., Suzuki S. T., Redies C. (2003). The cadherin superfamily in neural development: diversity, function and interaction with other molecules. Front Biosci. 8, d306-d355 [DOI] [PubMed] [Google Scholar]
- Iwai Y., Hirota Y., Ozaki K., Okano H., Takeichi M., Uemura T. (2002). DN-cadherin is required for spatial arrangement of nerve terminals and ultrastructural organization of synapses. Mol. Cell. Neurosci. 19, 375-388 [DOI] [PubMed] [Google Scholar]
- Ju M. J., Aroca P., Luo J., Puelles L., Redies C. (2004). Molecular profiling indicates avian branchiomotor nuclei invade the hindbrain alar plate. Neuroscience 128, 785-796 [DOI] [PubMed] [Google Scholar]
- Kintner C. (1992). Regulation of embryonic cell adhesion by the cadherin cytoplasmic domain. Cell 69, 225-236 [DOI] [PubMed] [Google Scholar]
- Markus A., Zhong J., Snider W. D. (2002). Raf and akt mediate distinct aspects of sensory axon growth. Neuron 35, 65-76 [DOI] [PubMed] [Google Scholar]
- Marthiens V., Gavard J., Padilla F., Monnet C., Castellani V., Lambert M., Mege R. M. (2005). A novel function for cadherin-11 in the regulation of motor axon elongation and fasciculation. Mol. Cell. Neurosci. 28, 715-726 [DOI] [PubMed] [Google Scholar]
- McClearn D., Noden D. M. (1988). Ontogeny of architectural complexity in embryonic quail visceral arch muscles. Am. J. Anat. 183, 277-293 [DOI] [PubMed] [Google Scholar]
- Momose T., Tonegawa A., Takeuchi J., Ogawa H., Umesono K., Yasuda K. (1999). Efficient targeting of gene expression in chick embryos by microelectroporation. Dev. Growth Differ. 41, 335-344 [DOI] [PubMed] [Google Scholar]
- Naeem A., Abbas L., Guthrie S. (2002). Comparison of the effects of HGF, BDNF, CT-1, CNTF, and the branchial arches on the growth of embryonic cranial motor neurons. J. Neurobiol. 51, 101-114 [DOI] [PubMed] [Google Scholar]
- Nakagawa S., Takeichi M. (1998). Neural crest emigration from the neural tube depends on regulated cadherin expression. Development 125, 2963-2971 [DOI] [PubMed] [Google Scholar]
- Nelson W. J. (2008). Regulation of cell-cell adhesion by the cadherin-catenin complex. Biochem. Soc. Trans. 36, 149-155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niederlander C., Lumsden A. (1996). Late emigrating neural crest cells migrate specifically to the exit points of cranial branchiomotor nerves. Development 122, 2367-2374 [DOI] [PubMed] [Google Scholar]
- Patel S. D., Ciatto C., Chen C. P., Bahna F., Rajebhosale M., Arkus N., Schieren I., Jessell T. M., Honig B., Price S. R., et al. (2006). Type II cadherin ectodomain structures: implications for classical cadherin specificity. Cell 124, 1255-1268 [DOI] [PubMed] [Google Scholar]
- Piper M., Dwivedy A., Leung L., Bradley R. S., Holt C. E. (2008). NF-protocadherin and TAF1 regulate retinal axon initiation and elongation in vivo. J. Neurosci. 28, 100-105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prin F., Ng K. E., Thaker U., Drescher U., Guthrie S. (2005). Ephrin-As play a rhombomere-specific role in trigeminal motor axon projections in the chick embryo. Dev. Biol. 279, 402-419 [DOI] [PubMed] [Google Scholar]
- Redies C. (2000). Cadherins in the central nervous system. Prog. Neurobiol. 61, 611-648 [DOI] [PubMed] [Google Scholar]
- Redies C., Inuzuka H., Takeichi M. (1992). Restricted expression of N- and R-cadherin on neurites of the developing chicken CNS. J. Neurosci. 12, 3525-3534 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salinas P. C., Price S. R. (2005). Cadherins and catenins in synapse development. Curr. Opin. Neurobiol. 15, 73-80 [DOI] [PubMed] [Google Scholar]
- Simon H., Guthrie S., Lumsden A. (1994). Regulation of SC1/DM-GRASP during the migration of motor neurons in the chick embryo brain stem. J. Neurobiol. 25, 1129-1143 [DOI] [PubMed] [Google Scholar]
- Takeichi M. (2007). The cadherin superfamily in neuronal connections and interactions. Nat. Rev. Neurosci. 8, 11-20 [DOI] [PubMed] [Google Scholar]
- Togashi H., Abe K., Mizoguchi A., Takaoka K., Chisaka O., Takeichi M. (2002). Cadherin regulates dendritic spine morphogenesis. Neuron 35, 77-89 [DOI] [PubMed] [Google Scholar]
- Treubert-Zimmermann U., Heyers D., Redies C. (2002). Targeting axons to specific fiber tracts in vivo by altering cadherin expression. J. Neurosci. 22, 7617-7626 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uemura M., Nakao S., Suzuki S. T., Takeichi M., Hirano S. (2007). OL-Protocadherin is essential for growth of striatal axons and thalamocortical projections. Nat. Neurosci. 10, 1151-1159 [DOI] [PubMed] [Google Scholar]
- Varela-Echavarria A., Pfaff S. L., Guthrie S. (1996). Differential expression of LIM homeobox genes among motor neuron subpopulations in the developing chick brain stem. Mol. Cell. Neurosci. 8, 242-257 [DOI] [PubMed] [Google Scholar]
- Varela-Echavarria A., Tucker A., Puschel A. W., Guthrie S. (1997). Motor axon subpopulations respond differentially to the chemorepellents netrin-1 and semaphorin D. Neuron 18, 193-207 [DOI] [PubMed] [Google Scholar]
- Vlahos C. J., Matter W. F., Hui K. Y., Brown R. F. (1994). A specific inhibitor of phosphatidylinositol 3-kinase, 2-(4-morpholinyl)-8-phenyl-4H-1-benzopyran-4-one (LY294002). J. Biol. Chem. 269, 5241-5248 [PubMed] [Google Scholar]
- Xie Z., Bikle D. D. (2007). The recruitment of phosphatidylinositol 3-kinase to the E-cadherin-catenin complex at the plasma membrane is required for calcium-induced phospholipase C-gamma1 activation and human keratinocyte differentiation. J. Biol. Chem. 282, 8695-8703 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.