Abstract
A mutation in the Arabidopsis gene ssi2/fab2, which encodes stearoyl–acyl carrier protein desaturase (S-ACP-DES), results in the reduction of oleic acid (18:1) levels in the mutant plants and also leads to the constitutive activation of NPR1-dependent and -independent defense responses. By contrast, ssi2 plants are compromised in the induction of the jasmonic acid (JA)–responsive gene PDF1.2 and in resistance to the necrotrophic pathogen Botrytis cinerea. Although S-ACP-DES catalyzes the initial desaturation step required for JA biosynthesis, a mutation in ssi2 does not alter the levels of the JA precursor linolenic acid (18:3), the perception of JA or ethylene, or the induced endogenous levels of JA. This finding led us to postulate that the S-ACP-DES–derived fatty acid (FA) 18:1 or its derivative is required for the activation of certain JA-mediated responses and the repression of the salicylic acid (SA) signaling pathway. Here, we report that alteration of the prokaryotic FA signaling pathway in plastids, leading to increased levels of 18:1, is required for the rescue of ssi2-triggered phenotypes. 18:1 levels in ssi2 plants were increased by performing epistatic analyses between ssi2 and several mutants in FA pathways that cause an increase in the levels of 18:1 in specific compartments of the cell. A loss-of-function mutation in the soluble chloroplastic enzyme glycerol-3-phosphate acyltransferase (ACT1) completely reverses SA- and JA-mediated phenotypes in ssi2. In contrast to the act1 mutation, a loss-of-function mutation in the endoplasmic reticulum–localized ω6 oleate desaturase (FAD2) does not alter SA- or JA-related phenotypes of ssi2. However, a mutation in the plastidial membrane–localized ω6 desaturase (FAD6) mediates a partial rescue of ssi2-mediated phenotypes. Although ssi2 fad6 plants are rescued in their morphological phenotypes, including larger size, absence of visible lesions, and straight leaves, these plants continue to exhibit microscopic cell death and express the PR-1 gene constitutively. In addition, these plants are unable to induce the expression of PDF1.2 in response to the exogenous application of JA. Because the act1 mutation rescues all of these phenotypes in ssi2 fad6 act1 triple-mutant plants, act1-mediated reversion may be mediated largely by an increase in the free 18:1 content within the chloroplasts. The reversion of JA responsiveness in ssi2 act1 plants is abolished in the ssi2 act1 coi1 triple-mutant background, suggesting that both JA- and act1-generated signals are required for the expression of the JA-inducible PDF1.2 gene. Our conclusion that FA signaling in plastids plays an essential role in the regulation of SSI2-mediated defense signaling is further substantiated by the fact that overexpression of the N-terminal–deleted SSI2, which lacks the putative plastid-localizing transit peptide, is unable to rescue ssi2-triggered phenotypes, as opposed to overexpression of the full-length protein.
INTRODUCTION
Plants resist pathogen infection by inducing a defense response that is targeted specifically to combat invasion by the pathogen (Keen, 1990; Van der Hoorn et al., 2002). In many cases, the induction of these responses is accompanied by localized cell death at the site of pathogen entry, which often is able to restrict the spread of pathogen to cells within and immediately surrounding the lesions. This phenomenon, known as the hypersensitive response, is one of the earliest visible manifestations of induced defense response and resembles programmed cell death in animals (Flor, 1971; Greenberg et al., 1994; Dangl et al., 1996; Hammond-Kosack and Jones, 1996; Jabs et al., 1996; Gray, 2002). Concurrent with hypersensitive response development, defense reactions are triggered locally and in parts distant from the site of primary infection. This phenomenon, known as systemic acquired resistance, is one of the most studied induced defense responses and is accompanied by a local and systemic increase in endogenous salicylic acid (SA) and a concomitant upregulation of a large set of defense genes, including genes that encode pathogenesis-related (PR) proteins (Ward et al., 1991; Gaffney et al., 1993; Uknes et al., 1993; Dong, 2001).
A substantial body of evidence has demonstrated that SA plays an important signaling role in the activation of PR gene expression and resistance to certain pathogens (Delaney et al., 1994; Dempsey et al., 1999; Feys and Parker, 2000; Dong, 2001; Thomma et al., 2001; Kunkel and Brooks, 2002; Shah, 2003). In addition to SA, jasmonic acid (JA) and ethylene are two other important signal molecules in plant defense against pathogens. The role of JA in defense signaling was proven conclusively by creating the fad3 fad7 fad8 triple mutant, which is unable to accumulate JA and highly susceptible to infection by Pythium spp (Vijayan et al., 1998). Similarly, the JA-insensitive coi1 and jar1 mutants show enhanced susceptibility to fungal pathogens (Penninckx et al., 1996; Staswick et al., 1998; Thomma et al., 1998; Kachroo et al., 2001) and suppress the expression of the JA-inducible defense genes PDF1.2 and THI2.1 (Epple et al., 1995; Penninckx et al., 1998; Kachroo et al., 2001). A defect in ethylene signaling also impairs the expression of PDF1.2 and renders plants susceptible to infection by Alternaria brassicicola (Thomma et al., 1999).
Several lines of evidence suggest the existence of an intricate signaling network involving SA, JA, and ethylene, which leads to fine tuning of defense responses. Although SA and JA activate distinct signaling pathways, there is a growing body of literature that shows that these pathways do not function entirely independently. Rather, they are involved in a complex signaling network that influences the magnitude or amplitude of various signals derived from these pathways (Kunkel and Brooks, 2002). One of the major roles postulated for JA is its antagonistic action on SA-dependent signaling pathways (Creelman and Mullet, 1997; Seo et al., 1997). Similarly, various pharmacological and genetic experiments have shown that SA is a potent suppressor of the JA signaling pathway (Pena-Cortes et al., 1993; Doares et al., 1995; Harms et al., 1995; Niki et al., 1998; Gupta et al., 2000; Spoel et al., 2003).
The mechanism of crosstalk between SA and JA signaling pathways in the plant defense response remains to be elucidated. Results from several laboratories suggest that NPR1, a positive regulator of systemic acquired resistance, also may play a role in crosstalk and modulate the expression of the JA-inducible gene PDF1.2 (Shah et al., 1999; Devadas et al., 2002; Spoel et al., 2003). Recently, Spoel et al. (2003) showed that the NPR1-interacting protein TGA2 also can bind to a TGACG motif in the PDF1.2 promoter. Although the TGACG motif does not appear to be required for NPR1-dependent crosstalk, these results indicate that SA- and JA-mediated pathways are likely to share common components that may have a regulatory role.
Although NPR1 is a key transducer of SA signal (Cao et al., 1997; Ryals et al., 1997), several NPR1-independent pathways also have been identified that regulate PR gene expression and/or resistance to certain bacterial and fungal pathogens (Bowling et al., 1997; Clarke et al., 1998; Rate et al., 1999; Shah et al., 1999, 2001; Kachroo et al., 2000; Shirano et al., 2002). The ssi2 mutant represents one such component of the NPR1-independent signaling pathway. The recessive ssi2 mutation confers constitutive PR gene expression, spontaneous lesion formation, and enhanced resistance to both bacteria and oomycete pathogens (Shah et al., 2001). By contrast, a subset of defense responses regulated by the JA signaling pathway, including expression of the defensin gene PDF1.2 and resistance to Botrytis cinerea, are impaired in ssi2 plants (Kachroo et al., 2001). SSI2 encodes a stearoyl–acyl carrier protein desaturase (S-ACP-DES), which is an archetype member of a family of soluble fatty acid (FA) desaturases; these enzymes play an important role in regulating the overall level of desaturated FAs in the cell. The activity of the mutant S-ACP-DES enzyme was reduced 10-fold, resulting in an increase of the stearic acid (18:0) content in ssi2 plants (Kachroo et al., 2001). Because reduced S-ACP-DES activity leads to the induction of certain defense responses and the inhibition of others, we proposed that a FA-derived signal modulates crosstalk between different defense signaling pathways.
In plants, FAs have started to emerge as important molecules that participate in diverse biological processes (Lee et al., 1997; Ryu and Wang, 1998; Shanklin and Cahoon, 1998; Piffanelli and Murphy, 1999; Kachroo et al., 2001, 2003; Laxalt and Munnik, 2002; Maldonado et al., 2002; Weber, 2002; Li et al., 2003). Interestingly, FA signaling in plants and animals show several intriguing parallels. For example, both JA and its structural equivalent in animals, prostaglandins, are derived from similar enzymatic reactions starting with the hydrolysis of polyunsaturated FAs (Straus and Glass, 2001). Furthermore, the stearoyl desaturase–catalyzed enzymatic step that leads to the synthesis of JA and prostaglandin precursors, linolenic acid and arachidonic acid, respectively, also is conserved between plants and animals (Shanklin and Somerville, 1991). In animals, altered stearoyl-CoA desaturase activity has been implicated in the regulation of cell growth, differentiation, and signal transduction (Kates et al., 1984; Gyorfy et al., 1997, Kim et al., 1999; Kumar et al., 1999). Additionally, altered activity of this enzyme is correlated with apoptosis (de Vries et al., 1997) and neoplasia (Li et al., 1994); these phenomena are similar to the cell death and altered defense signaling phenotypes associated with the ssi2 mutation. Thus, although the mechanism(s) by which a mutation in S-ACP-DES affects defense response activation in plants is unclear, the discovery that a defect in FA desaturation can modulate defense responses opens novel approaches in the study of pathways that lead to disease resistance. Here, we show that a loss-of-function mutation in the chloroplastic enzymes glycerol-3-phosphate acyltransferase and ω6 desaturase can reverse various ssi2-triggered phenotypes. We also show that alteration of the prokaryotic FA signaling pathway in plastids, leading to increased levels of 18:1, can rescue ssi2-triggered phenotypes. Our analysis suggests that FA signaling plays an important role in modulating the defense response against pathogens.
RESULTS
ssi2 Plants Are Not Altered in the Perception or Biosynthesis of JA
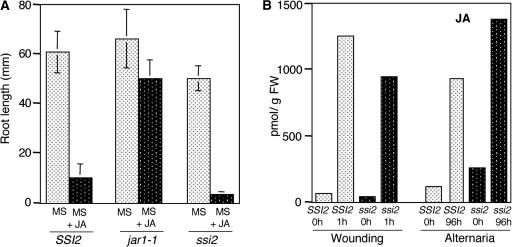
SSI2-encoded S-ACP-DES preferentially desaturates stearic acid (18:0) between carbons 9 and 10 to yield 18:1. Compared with wild-type S-ACP-DES, the activity of the mutant protein is reduced 10-fold, which causes an increase in 18:0 levels and a reduction in 18:1 levels in ssi2 plants (Table 1). The inability of the ssi2 mutant plants to induce PDF1.2 expression upon JA treatment could be attributable to any of the following causes: (1) ssi2 plants are insensitive to JA and therefore unable to respond to exogenous treatment; (2) ssi2 plants are impaired in the JA biosynthetic pathway and therefore unable to accumulate endogenous JA; or (3) ssi2 plants lack a JA-coactivating signal, which is derived from S-ACP-DES. To assess these possibilities, we first determined the ability of ssi2 plants to perceive JA by conducting a root-growth bioassay (Staswick et al., 1998). Approximately 30 to 40 seeds each of SSI2, jar1-1, and ssi2 were grown on plates containing either 25 μM JA or an equal proportion of alcohol and allowed to grow vertically. Mean root length was measured 14 days later. As expected, JA inhibited root growth of the wild-type plants, whereas root growth in the JA-insensitive mutant jar1-1 remained largely unaffected (Figure 1A). ssi2 plants behaved like the wild-type plants and showed inhibition of root growth on JA-containing medium. These results suggest that ssi2 plants perceive JA in a manner similar to that of wild-type plants.
Table 1.
Fatty Acid Composition of Total Leaf Lipids from SSI2, ssi2, act1, fad2, fad6, ssi2 act1, ssi2 fad6, and ssi2 fad2 Plants
| Genotype | 16:0 | 16:1 | 16:2 | 16:3 | 18:0 | 18:1 | 18:2 | 18:3 |
|---|---|---|---|---|---|---|---|---|
| SSI2 | 16.6 ± 0.9 | 3.1 ± 0.7 | 0.8 ± 0.1 | 14.5 ± 0.5 | 0.8 ± 0.2 | 2.0 ± 1.0 | 15.4 ± 0.6 | 46.8 ± 2.0 |
| ssi2 | 13.3 ± 1.0 | 2.7 ± 0.3 | 0.7 ± 0.0 | 10.1 ± 0.5 | 14.3 ± 1.0 | 0.7 ± 0.2 | 14.7 ± 0.4 | 43.5 ± 2.4 |
| act1 | 13.1 ± 0.3 | 2.5 ± 0.2 | 0.5 ± 0.1 | 1.5 ± 0.2 | 0.7 ± 0.0 | 5.8 ± 0.5 | 15.8 ± 1.0 | 60.1 ± 4.2 |
| ssi2 act1 | 13.1 ± 0.9 | 1.9 ± 0.3 | — | 1.7 ± 0.2 | 7.4 ± 0.5 | 11.1 ± 1.3 | 14.7 ± 0.8 | 50.1 ± 2.1 |
| fad6 | 12.9 ± 0.6 | 16.4 ± 0.5 | 0.8 ± 0.1 | 0.1 ± 0.0 | 0.9 ± 0.3 | 25.0 ± 1.3 | 15.0 ± 1.6 | 28.9 ± 1.5 |
| ssi2 fad6 | 13.8 ± 0.8 | 13.1 ± 0.4 | 0.5 ± 0.0 | — | 9.8 ± 1.2 | 17.3 ± 1.3 | 17.4 ± 1.1 | 28.1 ± 2.1 |
| fad2 | 16.0 ± 1.0 | 3.2 ± 0.5 | 0.6 ± 0.0 | 14.1 ± 0.9 | 0.7 ± 0.1 | 12.1 ± 1.1 | 5.5 ± 0.8 | 47.8 ± 2.3 |
| ssi2 fad2 | 14.2 ± 1.1 | 2.7 ± 0.4 | 0.7 ± 0.1 | 8.2 ± 1.1 | 8.6 ± 0.8 | 15.9 ± 1.3 | 5.5 ± 0.5 | 44.2 ± 1.1 |
All measurements were made on plants grown at 22°C, and data are described as mol% ± se calculated for a sample size of six.
Figure 1.
Root Growth Bioassay and JA Levels in the Leaves of SSI2 (Wild-Type) and ssi2 Plants after Wounding or Pathogen Infection.
(A) Approximately 30 to 40 seeds each of SSI2, jar1-1, and ssi2 plants were sown on Murashige and Skoog (1962) (MS) plates with or without 25 μM JA and allowed to grow vertically for 14 days. Mean root length (mm ± sd) was determined for 30 seeds from each line.
(B) Leaves were wounded with a razor blade or inoculated by spraying with A. brassicicola spores at a concentration of 106 spores/mL. Maximum JA levels were observed at 1 h after wounding and 96 h after inoculation and are shown as averages of two to three replicates. FW, fresh weight.
To assess if the inability of ssi2 plants to induce the expression of PDF1.2 was associated with a defect in JA biosynthesis, we determined the levels of JA in wild-type and ssi2 plants after pathogen infection and wounding. Maximum levels of JA induction in wild-type plants were seen at 1 h after wounding or 96 h after spraying of A. brassicicola spores (Figure 1B). At these time points, the induced levels of JA in ssi2 were comparable to or higher than the levels in wild-type plants. These results suggest that ssi2 plants are not defective in the pathway leading to JA biosynthesis. Because ssi2 plants do not appear to be impaired in the perception or biosynthesis of JA, our results strongly support the third possibility mentioned above, that the lack of JA responses in ssi2 may be caused by the absence or reduced levels of a JA-coactivating signal.
ssi2 Seedlings Are Not Affected in Their Response to Ethylene
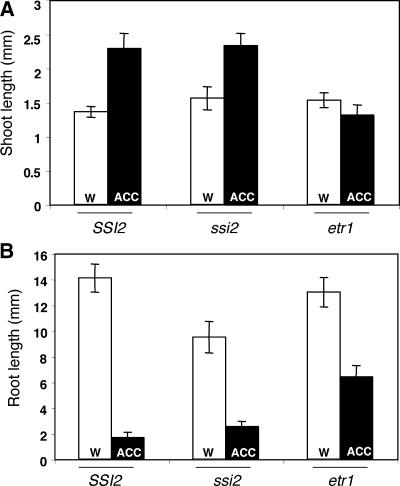
Because expression of the PDF1.2 gene requires the concomitant activation of both JA and ethylene signal transduction pathways (Penninckx et al., 1998), it also is possible that ssi2 plants do not induce PDF1.2 expression because they carry a defect in the ethylene signaling pathway. To test this possibility, we compared the seedling responses of ssi2, wild-type, and ethylene-insensitive etr1 plants to the ethylene precursor aminocyclopropane carboxylic acid (ACC) (Guzman and Ecker, 1990; Smalle et al., 1997). Arabidopsis plants grown under continuous light on water-agar plates containing ACC exhibit enhanced elongation of the hypocotyl (Smalle et al., 1997). Compared with etr1, both ssi2 and wild-type seedlings showed elongated hypocotyls on plates containing ACC as opposed to seedlings grown on plates without ACC (Figure 2A). In addition, both ssi2 and wild-type seedlings showed a drastic inhibition of root growth on ACC plates as opposed to only a partial inhibition in etr1 plants (Figure 2B). These data suggest that ssi2 plants are not impaired in their response to ethylene.
Figure 2.
Effect of ACC Treatments on Seedlings of SSI2, ssi2, and etr1 Plants.
(A) Comparison of hypocotyl lengths of SSI2, ssi2, and etr1 seedlings grown on water-agar plates (w) or water-agar plates containing 50 μM ACC (ACC). Approximately 30 to 40 seeds each of SSI2, ssi2, and etr1 were grown on water-agar plates with or without ACC, and the plates were incubated at 22°C in continuous light. Hypocotyl lengths were measured after 10 days of growth.
(B) Comparison of root lengths of 10-day-old seedlings from the same plants used in (A).
A Mutation in the act1 Gene Causes Reversion of the ssi2 Phenotypes
Because ssi2 plants contain highly reduced levels of 18:1 compared with 18:2 or 18:3, we proposed that the JA-coactivating signal might be derived from 18:1 or that the balance between 18:0 and 18:1 may be critical for normal signaling. This notion is supported by our previous work, in which we showed that the administration of 18:1 rescues the JA responsiveness of ssi2 plants (Kachroo et al., 2001). However, because a pharmacological approach is fairly limiting for determining the effects of increased levels of 18:1 on SA and JA signal transduction pathways, we chose a genetic approach to determine if increasing 18:1 levels in ssi2 plants can restore the normal signaling of SA and JA pathways.
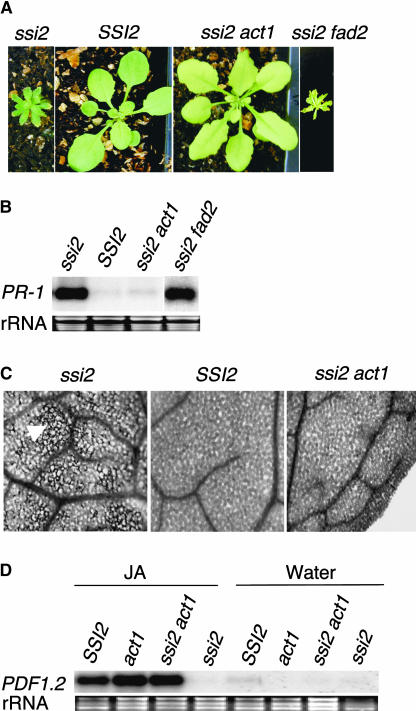
The Arabidopsis genes FAD2 (Miquel and Browse, 1992) and ACT1 (Kunst et al., 1989) encode enzymes that are localized in extrachloroplast or chloroplast, respectively, and mutations in these genes result in the upregulation of 18:1 levels in the cell. By contrast, the FAE1 gene product is expressed specifically in seeds, and a mutation in this gene causes an increase in 18:1 levels in the seeds (James et al., 1995). To determine if mutations in these genes can upregulate the levels of 18:1 in ssi2 and thereby complement defective crosstalk, we crossed ssi2 with fad2-1, act1-1, and fae1-1 mutants and screened the F2 populations for double-mutant plants. Interestingly, F2 analysis of the ssi2 × act1 cross revealed that a certain proportion of these plants, corresponding to recessive double mutants, had nearly wild-type-like leaf morphology but were slightly pale compared with either parent (Figure 3A). By contrast, the ssi2 fad2 (Figure 3A) and ssi2 fae1 (data not shown) plants showed ssi2-like phenotypes. Because ssi2 act1 plants contained the ssi2 mutation in the homozygous state, we concluded that the presence of the act1 mutant allele had resulted in the reversion of ssi2-conferred phenotypes. To confirm the genotype at the act1 locus, we sequenced the act1 gene and developed a cleaved amplified polymorphic sequence (CAPS) marker, which allowed us to score for the mutant allele. As expected, analysis of the double-mutant plants with the ACT1-CAPS marker showed that these contained the act1 mutation in the homozygous state. Unlike ssi2 plants, ssi2 act1 double-mutant plants did not show spontaneous cell death or constitutively increased levels of PR-1 gene expression (Figures 3B and 3C). To determine if these double mutant plants had regained sensitivity to the exogenous application of JA, we treated SSI2 (wild-type), ssi2, act1, and ssi2 act1 plants with JA or methyl jasmonate. As expected, ssi2 showed no induction of PDF1.2. By contrast, high levels of PDF1.2 expression were induced in wild-type, act1, and ssi2 act1 plants (Figure 3D). These results indicate that the act1 mutation was sufficient to revert ssi2 plants to wild-type-like plants.
Figure 3.
Morphological and Molecular Phenotypes of SSI2, ssi2, ssi2 act1, and ssi2 fad2 Plants.
(A) Comparison of the morphological phenotypes displayed by SSI2, ssi2, and ssi2 act1 plants. Plants were grown on soil and photographed after 3 (SSI2) or 4 (ssi2, ssi2 act1, and ssi2 fad2) weeks.
(B) Expression of the PR-1 gene in SSI2, ssi2, ssi2 act1, and ssi2 fad2 plants. RNA gel blot analysis was performed on 5 μg of total RNA extracted from 4-week-old soil-grown plants. Ethidium bromide staining of rRNA was used as a loading control.
(C) Microscopy of trypan blue–stained leaves from SSI2, ssi2, and ssi2 act1 plants. Leaves from ssi2 plants contain intensely stained areas of dead cells (arrowhead), whereas ssi2 act1 leaves show a wild-type-like phenotype.
(D) Expression of the PDF1.2 gene in SSI2, act1, ssi2 act1, and ssi2 plants in response to 50 μM JA or water treatment. Samples were harvested at 48 h after treatment and examined by RNA gel blot analysis performed on 5 μg of total RNA. Ethidium bromide staining of rRNA was used as a loading control.
The act1 Mutation Restores the Altered Pathogen Response of ssi2 Plants
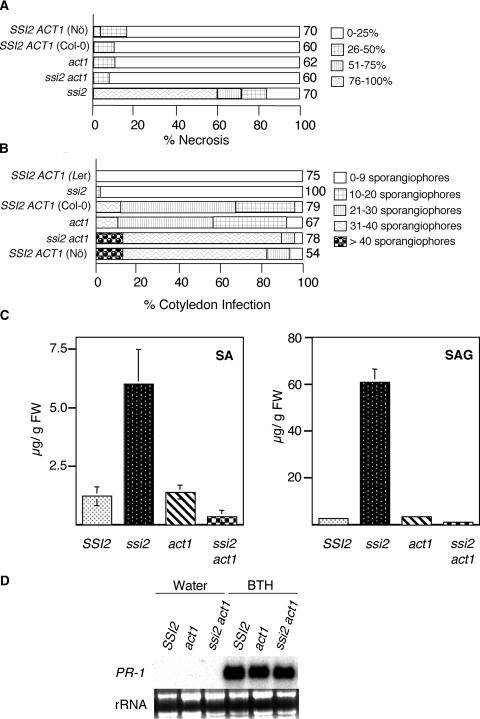
To determine if the act1 mutation also restored wild-type-like resistance to necrotrophic pathogens in ssi2 plants, we inoculated ssi2, act1, ssi2 act1, and wild-type plants with B. cinerea and assayed necrosis at 8 days after inoculation. Interestingly, ssi2 act1 plants were as resistant to infection by B. cinerea as wild-type plants (Figure 4A). By contrast, ssi2 plants either in the Nössen background or derived from a cross between Columbia (Col-0) and ssi2 were highly susceptible to B. cinerea, and >80% of these plants showed necrosis accompanied by extensive proliferation and sporulation of the fungal mycelia (Figure 4A).
Figure 4.
Resistance Response, Levels of SA and SA Glucoside, and SA Responsiveness.
(A) Comparison of necrosis caused by B. cinerea in SSI2 (Col-0 and Nössen [Nö] ecotypes), ssi2, ssi2 act1, and act1 genetic backgrounds. Inoculations were performed on leaves of 4-week-old plants pricked with a needle at two to three well-spaced spots per leaf. The shading of each box indicates the severity of necrosis, and the numbers at right of the sample boxes indicate the number of leaves assayed.
(B) Growth of P. parasitica ecotype Emco5. Cotyledons of 7-day-old seedlings from the various plant genotypes listed at left were inoculated by spraying conidiospores at a concentration of 106 spores/mL. There was no difference in the size of cotyledons between wild-type and mutant plants at the time of infection. Pathogen growth was assessed by counting the number of sporangiophores per cotyledon at 8 days after inoculation. The shading of each box indicates the severity of infection, based on the number of sporangiophores per cotyledon (see key at right). Numbers at right of the sample boxes indicate the number of cotyledons assayed. Ler, Landsberg erecta.
(C) Endogenous SA and SA glucoside levels in leaves of 4-week-old soil-grown SSI2, ssi2, act1, and ssi2 act1 plants. The values presented are averages of three replicates. FW, fresh weight.
(D) SA responsiveness of act1 and ssi2 act1 plants. Wild-type, act1, and ssi2 act1 plants were treated with water or 100 μM benzo(1,2,3)thiadiazole-7-carbothioic acid (BTH) and analyzed for PR-1 gene expression after 48 h. Ethidium bromide staining of rRNA was used as a loading control.
In contrast to a susceptible response toward B. cinerea, ssi2 plants show heightened resistance to biotrophic pathogens, and this resistance is partially compromised in a NahG background, which abolishes the accumulation of SA (Shah et al., 2001). To determine the effect of the act1 mutation on ssi2-conferred resistance, ssi2, act1, ssi2 act1, and wild-type plants were challenged with a virulent biotype of Peronospora parasitica. As anticipated, ssi2 plants and a resistant ecotype of Arabidopsis supported 5% or no growth of the pathogen, respectively, and act1, like its parental ecotype Col-0, showed partial susceptibility to this pathogen. By contrast, ssi2 act1 plants supported profuse growth of the pathogen, similar to that observed in the susceptible wild-type plants (Figure 4B).
The act1 Mutation Restores Wild-Type-Like SA Levels in ssi2 Plants and Does Not Impair SA Responsiveness
To test if reversion of the enhanced resistance phenotype conferred by the ssi2 mutation also influences the accumulation of increased levels of SA, we determined the levels of SA and SA glucoside (SAG) in wild-type, ssi2, act1, and ssi2 act1 plants (Figure 4C). The ssi2 act1 plants showed a dramatic decrease in the levels of SA/SAG compared with the ssi2 plants, suggesting that the act1 mutation suppresses all of the SA-related phenotypes of the ssi2 mutant. Interestingly, suppression of SA levels alone is not sufficient to abolish any of the ssi2 phenotypes, because ssi2 nahG plants continue to remain stunted, show spontaneous cell death, express PR genes, and show partial resistance to bacterial and oomycete pathogens (Shah et al., 2001).
To determine if the act1 mutation restores various ssi2 phenotypes by impairing SA signaling, we sprayed wild-type, act1, and ssi2 act1 plants with water or benzo(1,2,3)thiadiazole-7-carbothioic acid and analyzed these for the induction of PR-1 gene expression. Both act1 and ssi2 act1 plants showed high levels of PR-1 expression, which was comparable to the induced levels of PR-1 seen in the wild-type plants (Figure 4D). These results suggest that ssi2 act1 plants are SA responsive and that the act1 mutation does not impair SA signaling. Therefore, act1-mediated suppression of the SA signaling pathway in ssi2 act1 plants may involve the generation of a signal that acts as a repressor and shuts off the SA signaling pathway.
The act1 Mutation Causes the Upregulation of 18:1 Levels in ssi2 Plants
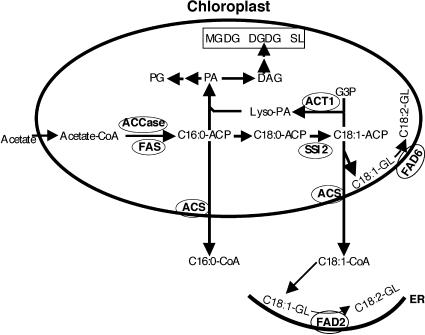
ACT1 encodes glycerol-3-phosphate acyltransferase, which functions in the first step of the prokaryotic pathway of glycerolipid biosynthesis (Kunst et al., 1988) (Figure 5). The FAs 16:0 and 18:1 synthesized de novo in the chloroplasts either enter into the “prokaryotic pathway” in the chloroplast envelope or are exported to the endoplasmic reticulum as CoA thioesters, where they are incorporated into lipids via the “eukaryotic pathway.” A mutation in act1 effectively converts a 16:3 plant such as Arabidopsis to an 18:3 plant, resulting in the accumulation of 18:1-ACP and the redirection of C-16 chains into the eukaryotic pathway (Kunst et al., 1988, 1989). Therefore, act1 mutant plants are deficient in their levels of 16:3 FA. To determine how act1 influences the FA content of ssi2 plants, levels of the various FAs were determined in leaf tissue of ssi2, act1, ssi2 act1, and wild-type plants (Table 1). As expected, the FA profile of ssi2 act1 plants overlapped with the profiles from single-mutant plants; ssi2 act1 plants contained lower levels of 16:3, similar to act1 plants, and high levels of 18:0, similar to ssi2 plants. However, the 18:1 content in ssi2 act1 plants was ∼15-fold higher compared with that in ssi2 plants, whereas the levels of all other FAs in general remained similar to those of control plants. These data suggest that the restoration of phenotypes in ssi2 act1 plants is likely to be mediated by increased levels of 18:1 in these plants. Because the act1-mediated increase in 18:1-ACP levels was initiated within chloroplasts, this result also suggests that reversion of the ssi2 phenotype may be linked to chloroplastic 18:1 content. This finding is supported by the fact that ssi2 fad2 plants accumulated an ∼17-fold increase in 18:1 content but continued to show ssi2-like phenotypes (Table 1).
Figure 5.
Condensed Scheme for Lipid Biosynthesis in the Chloroplasts of Arabidopsis Leaves.
De novo FA biosynthesis from acetyl-CoA occurs exclusively in the plastids of all cells (represented by the oval). Acetyl-CoA carboxylase (ACCase) and fatty acid synthase (FAS) complex are key enzymes involved in the biosynthesis of C16:0 FA. Upon elongation to C18:0, this FA undergoes desaturation to C18:1. This step is catalyzed by the SSI2-encoded S-ACP-DES. The product of this reaction (C18:1-ACP) either enters the prokaryotic pathway of lipid biosynthesis through the acylation of glycerol-3-phosphate (G3P) or is exported out of the plastids as a CoA thioester to enter the eukaryotic pathway. The acylation of G3P is catalyzed by an ACT1-encoded G3P acyltransferase. Desaturation of the C18:1 present on membrane glycerolipids (GL) is catalyzed by FAD2- or FAD6-encoded ω6 desaturases that are present on the endoplasmic reticulum (ER) or the plastid envelop, respectively. Esterification of the CoA group is mediated by acyl-CoA synthetase (ACS). Symbols for various components are as follows: ACP, acyl carrier protein; C, carbon; DAG, diacylglycerol; DGDG, digalactosyldiacylglycerol; Lyso-PA, 1-acyl-G3P; MGDG, monogalactosyldiacylglycerol; PA, phosphatidic acid; PG, phosphatidylglycerol; SL, sulfolipid.
Partial Reversion of ssi2 Phenotypes by a Mutation in Plastidial ω6 Oleate Desaturase
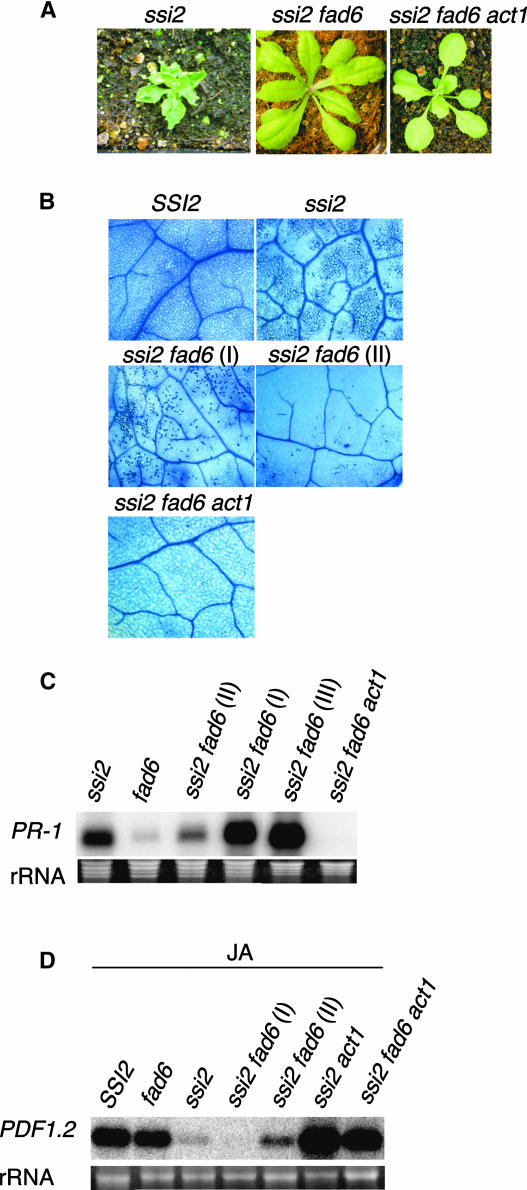
The 18:1 present in the chloroplasts can be found as either free 18:1-ACP or 18:1 bound to membrane lipids. To investigate the importance of free versus membrane-bound chloroplastic 18:1 in rescuing ssi2-related phenotypes, we crossed ssi2 to the fad6 mutant (Falcone et al., 1994). The fad6 mutant is defective in the activity of chloroplastic ω6 desaturase, which converts 16:1 and 18:1 to 16:2 and 18:2, respectively, in chloroplastic membrane lipids. Therefore, a mutation in fad6 results in increased 16:1 and 18:1 levels in the membrane lipids. To isolate double-mutant plants, we sequenced the fad6 gene and identified the mutant base that leads to a loss of function of fad6-encoded desaturase. A CAPS marker was developed for this mutant base, and double-mutant plants were identified based on CAPS primer analysis and subsequently confirmed by FA profiling. Analysis of the double-mutant plants revealed that they were much larger than ssi2 plants but smaller than wild-type plants (Figure 6A). Like ssi2 act1 plants, ssi2 fad6 plants showed no visible lesions on their leaves, although their leaves were not pale like those of ssi2 act1 plants. Trypan blue staining of ssi2 fad6 plants revealed that these plants showed microscopic cell death, although the dead cells were less dense than those in ssi2 plants (Figure 6B). Surprisingly, even though 90% of the randomly selected ssi2 fad6 leaves showed prominent cell death, 10% or fewer showed a marked reduction in the cell-death phenotype (Figure 6B, marked as I and II). This was observed particularly in one of the four F4 ssi2 fad6 homozygous lines and may result from the fact that ssi2 or fad6 segregates for another mutant loci that has a suppressive effect on cell death. Nonetheless, both types of leaves were analyzed for various ssi2-like phenotypes. As expected, the expression levels of PR-1 correlated well with the extent of dead cells present in the ssi2 fad6 leaves; leaves showing less density of dead cells showed reduced accumulation of PR-1 transcript compared with leaves showing prominent cell death (Figure 6C). RNA gel blot analysis of RNA extracted from one individual plant in which a few leaves showed no cell death revealed that ssi2 fad6 plants expressed high levels of PR-1 and that these levels were similar to those found in ssi2 plants (Figure 6B, marked as III). These results suggest that the fad6 mutation was unable to suppress cell death and PR-1 gene expression in ssi2 fad6 plants.
Figure 6.
Morphological and Molecular Phenotypes of ssi2, ssi2 fad6, and ssi2 fad6 act1 Plants.
(A) Comparison of the morphological phenotypes displayed by the ssi2, ssi2 fad6, and ssi2 fad6 act1 plants. Plants were grown on soil and photographed after 4 weeks.
(B) Microscopy of trypan blue–stained leaves from SSI2, ssi2, ssi2 fad6, and ssi2 fad6 act1 plants. More than 90% of the ssi2 fad6 leaf samples showed prominent cell death (shown as I), whereas the remaining samples showed a marked reduction in the cell-death phenotype (shown as II).
(C) Expression of PR-1 in ssi2, fad6, ssi2 fad6, and ssi2 fad6 act1 plants. For ssi2 fad6 plants, PR-1 gene expression was analyzed for three different sets of leaves. The lanes marked I and II represent RNA extracted from ssi2 fad6 leaves that showed high and low numbers of dead cells (shown in [B]), respectively. The lane marked III represents RNA extracted from leaves of one individual ssi2 fad6 plant in which a few leaves showed no cell death. RNA gel blot analysis was performed on 5 μg of total RNA extracted from 4-week-old soil-grown plants. Ethidium bromide staining of rRNA was used as a loading control.
(D) Expression of PDF1.2 in SSI2, fad6, ssi2 fad6, and ssi2 fad6 act1 plants in response to 50 μM JA. Lanes marked I and II represent RNA extracted from ssi2 fad6 plants that showed high and low numbers of dead cells, respectively. Samples were harvested at 48 h after treatment and examined by RNA gel blot analysis performed on 5 μg of total RNA. Ethidium bromide staining of rRNA was used as a loading control.
To determine if a partial rescue of the morphological phenotype in ssi2 fad6 plants also includes the rescue of JA responsiveness, we treated ssi2 fad6 and control plants with JA and analyzed them for the expression of PDF1.2. As expected wild-type, fad6, and ssi2 act1 plants induced high levels of PDF1.2 transcript, whereas ssi2 plants showed basal-level expression of this gene (Figure 6D). The ssi2 fad6 leaves displaying prominent cell death showed basal-level expression of PDF1.2, similar to that seen in ssi2 plants. The ssi2 fad6 leaves, which contained reduced numbers of dead cells, showed a slightly higher expression of PDF1.2, but these levels were appreciably lower than those seen in wild-type, fad6, or ssi2 act1 plants. These results suggest that a partial rescue of phenotypes by the fad6 mutation was insufficient to rescue JA responsiveness in ssi2 plants.
We next determined the FA profiles of fad6 and ssi2 fad6 plants and found that both fad6 and ssi2 fad6 plants contained reduced levels of 16:3 and 18:3 and increased levels of 16:1 and 18:1, compared with wild-type plants (Table 1). Compared with ssi2 plants, ssi2 fad6 plants accumulated ∼24-fold greater levels of 18:1; these levels were ∼10-fold higher than those in ssi2 act1 plants. This finding suggests that an increase in the 18:1 content of chloroplastic membrane lipids is not sufficient to completely revert the altered defense signaling in ssi2 plants.
The act1 Mutation Mediates a Complete Rescue of ssi2 fad6 Plants
A partial recovery of ssi2 phenotypes by the fad6 mutation suggests that a simple increase in membranous 18:1 levels may not be enough to restore defective signaling in ssi2 plants. On the other hand, a complete reversal of ssi2 phenotypes by the act1 mutation suggests that the act1 mutation may generate free 18:1 in addition to channelizing 18:1 into chloroplastic membrane lipids. If these assumptions were correct, then we would expect the act1 mutation to completely restore all ssi2-related phenotypes in ssi2 fad6 plants. We generated ssi2 fad6 act1 triple-mutant plants by crossing ssi2 act1 with fad6 and identified triple mutants plants by CAPS and derived CAPS analysis of act1, fad6, and ssi2. The triple-mutant plants were morphologically similar to ssi2 act1 plants and showed no visible or microscopic cell death in their leaves (Figures 6A and 6B). These plants showed basal levels of PR-1 gene expression (Figure 6C) and high levels of the JA-inducible PDF1.2 gene (Figure 6D). Together, these results suggest that the act1 mutation is epistatic to the fad6 mutation and can completely suppress the ssi2-related phenotypes in ssi2 fad6 plants.
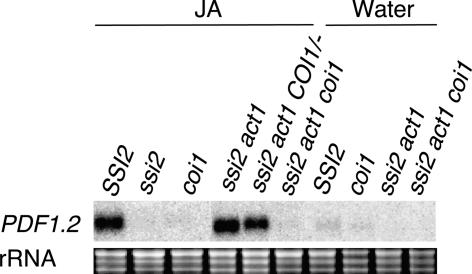
Induction of PDF1.2 in ssi2 act1 Plants Requires a Functional JA Pathway
Because the JA-induced expression of PDF1.2 requires the COI1 gene (Xie et al., 1998), we further assessed if the rescue of JA responsiveness in ssi2 act1 plants requires a fully functional JA pathway. To obtain triple-mutant plants, ssi2 act1 plants were crossed with coi1 plants and the F2 population was scored for plants homozygous for the ssi2 and act1 loci. Because tight linkage between the ssi2 and coi1 loci did not allow us to obtain ssi2 act1 plants that were homozygous for the coi1 locus, we isolated ssi2 act1 homozygous plants that were heterozygous at the coi1 locus. Three such plants were isolated after screening ∼460 F2 plants, and these were selfed to isolate ssi2 act1 coi1 triple-mutant plants. Genotypes at the coi1, act1, and ssi2 loci were determined by CAPS or derived CAPS marker analysis. The triple-mutant plants were morphologically similar to ssi2 act1 plants. Single-, double-, and triple-mutant plants between ssi2, act1, and coi1 were sprayed with water or JA, and the RNA isolated from these plants was analyzed for the expression of PDF1.2 after 48 h of treatment (Figure 7). As observed previously, ssi2 plants failed to induce any expression of PDF1.2, as opposed to the high levels seen in ssi2 act1 and ssi2 act1 COI1/− plants. By contrast, ssi2 act1 coi1 plants behaved like coi1 and ssi2 single-mutant plants and did not allow any JA-mediated induction of PDF1.2. These results suggest that COI1 is required for the induction of PDF1.2 in ssi2 act1 plants and that the act1-mediated rescue of JA responsiveness in ssi2 plants involves a step upstream of the COI1 gene in the JA pathway. However, it is likely that the act1 mutation regenerates a putative signal that in combination with JA is required for the induction of PDF1.2. In such a situation, blocking either the step leading to the generation of this putative signal or the JA signaling pathway will block the expression of the PDF1.2 gene. This notion is supported by our observation that exogenous application of both JA and 18:1 are required for the rescue of PDF1.2 expression in ssi2 plants (Kachroo et al., 2001).
Figure 7.
JA Responsiveness in ssi2 act1 coi1 Triple-Mutant Plants.
Expression of PDF1.2 in SSI2, ssi2, coi1, ssi2 act1, ssi2 act1 COI1/−, and ssi2 act1 coi1 plants in response to 50 μM JA or water treatment. Samples were harvested at 48 h after treatment and examined by RNA gel blot analysis performed on 5 μg of total RNA. Ethidium bromide staining of rRNA was used as a loading control.
SSI2 Lacking the N-Terminal Transit Peptide Does Not Complement the ssi2 Mutation
SSI2-encoded S-ACP-DES is a soluble chloroplastic desaturase that shows ∼83 to 93% identity with similar enzymes from other plant species (Lightner et al., 1994; Ohlrogge and Browse, 1995) (data not shown). Phylogenetic analysis of various S-ACP-DES revealed that SSI2 shares a high level of amino acid identity with S-ACP-DES from castor bean. Based on the aligned amino acid sequence and the presence of conserved domains between the castor bean and the SSI2-encoded S-ACP-DES, the predicted N-terminal transit peptide was removed from SSI2. Previously, we showed that SSI2 protein with an N-terminal deletion (SSI2 ΔN-t) had 10-fold higher activity compared with the mutant ssi2 protein (Kachroo et al., 2001). The SSI2 ΔN-t also showed the expected 18:16 substrate preference and Δ9 regiospecificity, suggesting that the N-terminal truncation had not caused any alteration in the predicted enzymatic activity of SSI2. Because removal of the N terminus from SSI2 was likely to abolish its targeting to the chloroplasts, we predicted that removal of the signal peptide would prevent its ability to complement the ssi2 mutant. We first overexpressed the full-length SSI2 cDNA in ssi2 plants and scored these plants for various ssi2 phenotypes. Transgenic plants overexpressing SSI2 (35S:SSI2) were similar to wild-type plants in morphology (Figure 8A) and showed the absence of spontaneous cell death (data not shown) and PR-1 gene expression (Figure 8B). By contrast, transgenic plants overexpressing SSI2 ΔN-t (35S:SSI2 ΔN-t) were similar to ssi2 plants in morphology and showed high levels of PR-1 expression (Figures 8A and 8B). Both groups of transgenic plants showed comparable levels of the SSI2 transcript, and as predicted, the size of the SSI2 ΔN-t transcript was slightly lower than that of the endogenous transcript (Figure 8B). These results suggest that targeting of SSI2 to the chloroplasts is important for the rescue of ssi2-associated phenotypes.
Figure 8.
Complementation of ssi2 Plants with Full-Length and N-Terminal Truncated SSI2 cDNAs.
(A) Morphological phenotypes displayed by the transgenic plants overexpressing SSI2 or SSI2 ΔN-t cDNA. Plants were grown on soil and photographed after 4 weeks.
(B) Expression of the SSI2 and PR-1 genes in transgenic plants overexpressing SSI2 or SSI2 ΔN-t cDNA. The band marked by the arrow represents endogenous SSI2 transcript. RNA gel blot analysis was performed on 5 μg of total RNA extracted from 4-week-old soil-grown plants. Ethidium bromide staining of rRNA was used as a loading control.
To ascertain if the ability to complement ssi2 phenotypes was associated with an increase in 18:1 levels, we conducted FA profiling of 35S:SSI2 and 35S:SSI2 ΔN-t plants (Table 2). Surprisingly, even though 35S:SSI2 plants showed complementation of all ssi2 phenotypes, they still continued to accumulate higher levels of 18:0. However, the levels of 18:0 in 35S; SSI2 plants were appreciably lower than those of 35S:SSI2 ΔN-t plants or ssi2-like F2 segregants from the 35S:SSI2 T2 population. The high levels of 18:0 in 35S:SSI2 may be the result of transcriptional or translational regulation of S-ACP-DES genes, which are under negative feedback control (Bené et al., 2001), or of low levels of S-ACP-DES protein transcribed during the initial stages of growth. Alternatively, it may represent the 18:0 levels channeled outside the chloroplasts (Lightner et al., 1994). As expected, 35S:SSI2 plants showed a fourfold increase in the levels of 18:1 compared with ssi2 and 35S:SSI2 ΔN-t plants. 35S:SSI2 plants resembled wild-type plants in their levels of all other FAs, including 16:3, which consistently showed lower levels in ssi2 plants. Together, these results reinforce the role of 18:1 in the restoration of altered defense signaling in ssi2 plants.
Table 2.
Fatty Acid Composition of Total Leaf Lipids from SSI2, ssi2, and Transgenic Plants Overexpressing SSI2 or the N-Terminal Truncated Form of SSI2
| Genotype | 16:0 | 16:1 | 16:2 | 16:3 | 18:0 | 18:1 | 18:2 | 18:3 |
|---|---|---|---|---|---|---|---|---|
| SSI2 | 16.6 ± 0.9 | 3.1 ± 0.7 | 0.8 ± 0.1 | 14.5 ± 0.5 | 0.8 ± 0.2 | 2.0 ± 1.0 | 15.4 ± 0.6 | 46.8 ± 2.0 |
| ssi2 | 13.3 ± 1.0 | 2.7 ± 0.3 | 0.7 ± 0.0 | 10.1 ± 0.5 | 14.3 ± 1.0 | 0.7 ± 0.2 | 14.7 ± 0.4 | 43.5 ± 2.4 |
| 35S-SSI2 F2 wild-type-like | 11.5 ± 1.0 | 3.0 ± 0.7 | 0.9 ± 0.1 | 14.8 ± 1.1 | 7.2 ± 0.8 | 4.4 ± 0.3 | 13.6 ± 1.0 | 44.6 ± 2.2 |
| 35S-SSI2 F2 ssi2-like | 14.2 ± 1.4 | 2.6 ± 0.3 | 0.8 ± 0.1 | 10.4 ± 0.9 | 16.8 ± 1.3 | 0.5 ± 0.0 | 12.3 ± 0.9 | 42.4 ± 2.1 |
| 35S-SSI2 ΔN-t | 12.6 ± 0.9 | 2.3 ± 0.7 | 0.7 ± 0.2 | 9.3 ± 1.1 | 14.0 ± 1..5 | 0.5 ± 0.1 | 20.6 ± 1.3 | 40.0 ± 1.8 |
All measurements were made on plants grown at 22°C, and data are described as mol% ± se calculated for a sample size of six.
DISCUSSION
Here, we have attempted to reveal an as yet unexplored role of FAs as signal molecules in the regulation of various plant defense signaling pathways. The ssi2 plants accumulate high levels of 18:0 FA, which could have a direct or indirect effect on these plants and cause stress-induced constitutive SA signaling. However, high levels of 18:0 are unlikely to induce SA signaling, because ssi2 act1 plants continue to accumulate high levels of 18:0 but have normal SA levels and PR gene expression. Moreover, we isolated several mutants in the ssi2 background that are restored in ssi2-triggered altered defense signaling but continue to accumulate high levels of 18:0 (Kachroo et al., 2003). In addition, we also studied shs1, a previously characterized mutant in the fab2 background, that shows nearly wild-type levels of 18:0 but continues to show spontaneous cell death and high levels of PR gene expression (Lightner et al., 1997; Kachroo et al., 2003). These results suggest that activation of the SA pathway in the ssi2 mutant most likely is caused by the absence of SSI2-mediated repression.
There are two possible ways that a mutation in act1 can rescue various ssi2-mediated phenotypes. First, because act1 suppresses SA/SAG levels in ssi2, it is likely that the act1 mutation renders plants defective in the SA signaling pathway, thereby shutting off constitutive SA signaling conferred by the ssi2 mutation. However, removal of SA by expressing the NahG transgene in ssi2 plants does not abolish any of its phenotypes (Shah et al., 2001). Furthermore, act1 plants are able to respond to the exogenous application of SA and are not altered in their resistance response to virulent or avirulent pathogens (Figures 4B and 4D). These results suggest that ACT1 may not participate in the SA signaling pathway and that reducing SA levels is not sufficient to cure the defect in JA-mediated responses in ssi2 plants. The second and most plausible explanation is that the act1 mutation acts indirectly by increasing the levels of 18:1 FA, and this increase leads to the restoration of ssi2-mediated signaling. This notion is supported by the observations that the injection of 18:1 rescues the responsiveness of ssi2 to JA (Kachroo et al., 2001) and that ssi2 act1 plants accumulate ∼15-fold higher levels of 18:1 compared with ssi2 plants, whereas levels of other FAs remain similar to those in control plants.
The mechanism underlying the wild-type-like reversion of ssi2 by a mutation in act1 is likely to involve FA signaling and to be mediated by 18:1 or its derivative. However, it also is possible that the signaling between various defense pathways is regulated by the ratios of saturated versus unsaturated FAs. This notion is supported by the observation that unsaturated FAs can stimulate or inhibit the activities of various regulatory enzymes (Klumpp et al., 1998; Baudouin et al., 1999). Furthermore, in animal systems, the ratio of saturated to unsaturated FAs is thought to control many cellular functions, including cell growth, differentiation, and apoptosis (Kates et al., 1984; Ntambi, 1995). Therefore it is likely that the act1 mutation mediates its effect by increasing the proportion of unsaturated 18:1 and balancing the ratio between 18:0 and 18:1. Interestingly, the ratios of 18:0 to 18:1 were more similar in wild-type and ssi2 act1 plants than in ssi2 plants, in which 18:0/18:1 ratios were 10-fold or more higher.
It is interesting that ssi2 plants continue to make low levels of 18:1 even though they are defective in the step leading to the formation of 18:1 (Figure 5). Similarly, fab2, which contains a null mutation in the SSI2 gene, also accumulates low levels of 18:1 (Lightner et al., 1994), indicating that trace amounts of 18:1 in ssi2 plants are not contributed by the residual activity of the mutant enzyme. This finding indicates that in addition to SSI2, several other isozymes of S-ACP-DES contribute to the generation of 18:1, although SSI2 appears to be the major enzyme regulating 18:1 levels in the chloroplast. Chloroplasts have been shown to harbor signaling components for both the SA and JA pathways, and a close proximity of various intermediates from these pathways would not only favor crosstalk but provide tighter control over their regulation (Schmid and Amrhein, 1995; Verberne et al., 2000; Weber, 2002). This also would explain why other mutations, including fad2, which upregulates 18:1 levels in the extrachloroplast membranes, are unable to complement a defect in ssi2 plants.
One important difference between the cellular locations of ACT1 and FAD2 is that although the former is a soluble chloroplastic enzyme, the later is a membrane-bound extrachloroplastic enzyme. These membrane-bound desaturases increase the unsaturated levels of membrane lipids and are less likely to cause an increase in the free FA pool in the cell. By contrast, a mutation in act1 is more likely to increase the free 18:1-ACP levels in the cell (Figure 5). This leads to the possibility that the reversion of phenotypes in ssi2 act1 plants may be attributable to the free chloroplastic 18:1-ACP pool rather than to the increased 18:1 content in the chloroplastic membrane lipids. However, the partial reversion of the morphological phenotype seen in ssi2 fad6 plants suggests that increases in both the free pool of 18:1-ACP and the 18:1 content of chloroplastic membrane lipids may be required for the restoration of ssi2 phenotypes. These results also suggest that act1 may generate certain factor(s) in addition to channelizing 18:1 into chloroplastic membrane lipids. This appears to be the case, because the act1 mutation is epistatic to the fad6 mutation and restores all of the ssi2-triggered phenotypes in ssi2 fad6 plants. Because SSI2 must be targeted to the chloroplast to be able to complement the ssi2 phenotypes (Figure 8), signaling within chloroplasts appears to account for the altered defense signaling in ssi2. Thus, it may be reasonable to assume that free 18:1 levels within the chloroplasts are able to balance the high levels of 18:0 generated as a result of the ssi2 mutation. Alternatively, the free 18:1 generated within the chloroplasts may generate a signal that participates in the modulation of defense signaling between the SA and JA pathways.
Several studies in both plants and animals have suggested a role for 18:1 in various cellular processes. In parsley cells, 18:1 levels have been shown to be upregulated in a biphasic manner at 2 h after treatment with an elicitor from Phytophthora sojae (Kirsch et al., 1997). By contrast, the levels of 18:2 declined and the levels of 18:3 increased steadily and doubled at 12 h after treatment. These findings indicate that the pathway from 18:1 to 18:3 is not linear and that 18:1 could play other roles besides serving as a precursor for 18:2. Studies in mammalian systems have shown that 18:1 can induce the translocation of protein kinase C from the cytosol toward the membranes via a direct or indirect process (Diaz-Guerra et al., 1991). Interestingly, in a recent study by Spoel et al. (2003), nucleocytoplasmic localization of NPR1 was suggested to regulate the crosstalk between the SA and JA pathways. Therefore, it is conceivable that 18:1 levels in plants regulate crosstalk by inducing the translocation of regulators of defense gene expression.
In addition to increasing the levels of 18:1 in ssi2 plants, the act1 and fad6 mutations also decrease 16:3 levels in the double-mutant plants. Thus, it is possible that the lower levels of 16:3 may play a role in restoring various phenotypes in ssi2 act1 and ssi2 fad6 plants. It is equally probable that the reduction in 16:3 levels is merely coincidental, because both act1 and fad6 affect the prokaryotic pathway of glycerolipid biosynthesis. Several results support this view. First, we have isolated several suppressor mutants in the ssi2 background that restore all of the ssi2-triggered phenotypes but contain normal levels of 16:3 (Kachroo et al., 2003). Second, ssi2 plants themselves show slight reductions in 16:3 levels compared with wild-type plants. Third, 35S:SSI2 transgenic plants show increased levels of 16:3 similar to wild-type plants. Fourth, although both ssi2 act1 and ssi2 fad6 plants have reduced 16:3 levels, only ssi2 act1 plants are completely rescued for all the ssi2-triggered phenotypes. Fifth, a mutation in fad5, which also decreased the levels of 16:3, did not restore wild-type-like phenotypes in ssi2 plants (A. Kachroo and P. Kachroo, unpublished results).
Our results also show that in addition to JA and ethylene, expression of the PDF1.2 gene is dependent on normal levels of FAs. The lack of induction of PDF1.2 in ssi2 plants does not appear to involve the JA or ethylene pathway, because ssi2 plants are responsive to both of these hormones (Figures 1 and 2). Furthermore, the restoration of JA responsiveness in ssi2 act1 plants can be abolished by the coi1 mutation, which indicates that functional FA and JA signaling pathways are required for the expression of PDF1.2.
In conclusion, our results clearly demonstrate the importance of FAs in modulating signaling between SA- and JA-dependent defense pathways. Further analysis of the FA signaling pathway should provide useful insights into the complex networking of these pathways and their regulation.
METHODS
Plant Growth Conditions and Genetic Analysis
Plants were grown in MTPS 144 Conviron walk-in-chambers (Winnipeg, Manitoba, Canada) at 22°C with 65% RH and 14-h photoperiods. Crosses were performed by pollinating flowers of ssi2 or wild-type Nössen plants with pollen from fad2, act1, fad6, or fae1 plants. The ssi2 act1 coi1 triple-mutant plants were obtained by pollinating flowers from coi1 with pollen from ssi2 act1. The ssi2 fad6 act1 triple-mutant plants were obtained by pollinating ssi2 act1 flowers with pollen from fad6.
The genotype at the ssi2 locus was determined by derived cleaved amplified polymorphic sequence (CAPS) analysis (Kachroo et al., 2001). The genotype at the act1-1 locus was determined by identifying the mutant base, amplifying a 0.5-kb region flanking the mutant base, and digesting the amplified product with BsmFI. Primers used to amplify act1-specific CAPS were 5′-GCCATCAAGTGTTCATCTACT-3′ and 5′-GGAAGTCATACAAGGTTGCTA-3′. The genotype at the coi1 locus was determined as described previously (Xie et al., 1998). The genotype at the fad6 locus was identified by CAPS analysis. Sequence analysis of the fad6-1 gene revealed a mutation from G to A at bp 478 in fad6 cDNA, which abolishes the restriction enzyme site for AlwNI in the fad6 amplified DNA. Primers used for FAD6-CAPS were 5′-GGATACACTTCCCAAAGAGGTG-3′ and 5′-AGTTCACCCAGTGAGCTATGG-3′. The genotype at the fad2 locus was determined by sequence analysis. The fad2-1 mutation was determined by sequence analysis of the mutant gene and found to contain A instead of G at 310 bp in the cDNA. The genotype at the fae1 locus was determined indirectly by scoring for plants that segregated like ecotype Columbia for markers on the lower arm of chromosome 4.
RNA Extraction and RNA Gel Blot Analysis
Small-scale extraction of RNA from one or two leaves was performed with the TRIzol reagent (Gibco BRL, Gaithersburg, MD) according to the manufacturer's instructions. RNA gel blot analysis and the synthesis of randomly primed probes for PR-1 and PDF1.2 were performed as described (Kachroo et al., 2001).
Generation of Transgenic Plants Overexpressing SSI2 and SSI2 ΔN-t cDNAs
The putative transit peptide region of SSI2 was predicted by aligning it with the protein sequence from castor bean S-ACP-DES. SSI2 ΔN-t cDNA was amplified such that it lacked 34 amino acids of the putative signal peptide and the 35th amino acid was converted to Met. Both full-length and N-terminal truncated cDNAs were amplified as NcoI-XbaI–linked PCR products and cloned downstream of the double 35S promoter in pRTL2.GUS (β-glucuronidase) vector. For transformation of Arabidopsis thaliana, the fragment containing promoter, SSI2 cDNA, and terminator was removed from the pRTL2-SSI2 or pRTL2-SSI2 ΔN-t vectors and cloned into the HindIII site of the binary vectors pBAR1 and pVK18. Transgenic seeds containing pBAR1-derived vector were selected on soil sprayed with BASTA, and transgenic seeds containing pVK18 were selected on Murashige and Skoog (1962) medium containing hygromycin.
Trypan Blue Staining
Leaf samples were taken from 2-week-old plants grown on soil. Trypan blue staining was performed as described previously (Bowling et al., 1997).
Salicylic Acid, Jasmonic Acid, and Fatty Acid Analysis
Salicylic acid and salicylic acid glucoside were extracted and measured from 0.4 g (fresh weight) of leaf tissue as described previously (Bowling et al., 1994). Jasmonic acid and fatty acid analysis were performed as described previously (Dahmer et al., 1989; He et al., 2002). Endogenous levels of jasmonic acid were induced by spray inoculating Alternaria brassicicola spores at a concentration of 106 spores/mL or by wounding leaf tissue with a razor blade.
Pathogen Infection
Infection with Peronospora parasitica Emco5 was performed by spraying or applying a single drop of asexual inoculum suspension as described previously (Kachroo et al., 2001). Infection with Botrytis cinerea was performed by spraying the spores at a concentration of 107/mL on plants that were wounded by needle pricks. Plants were covered with a transparent plastic dome and kept in a plexiglass container to create maximum humidity conditions. Necrosis and fungal growth were scored at 8 to 10 days after inoculation. Necrosis was quantified by estimating the percentage of leaf tissue showing decay and was confirmed microscopically by determining fungal spread and the extent of sporulation.
Upon request, materials integral to the findings presented in this publication will be made available in a timely manner to all investigators on similar terms for noncommercial research purposes. To obtain materials, please contact Pradeep Kachroo, pk62@uky.edu.
Acknowledgments
We thank Maelor Davies for allowing us to use the research facilities at the Kentucky Tobacco Research and Development Center and Baochun Li and David Zaitlin for sharing their laboratory space. We also thank John Shanklin and John Browse for useful discussions and John Browse for providing act1 fab2 seeds. We thank David Smith for useful suggestions. We thank William Broekaert and Bart Thomma for providing a culture of Botrytis, and we thank the ABRC for providing seeds for act1, fad6, fad2, and fae1 mutants. This work was supported by University of Kentucky research support funds to P.K., by Grants MCB 9723952 and MCB 0110404 from the National Science Foundation to D.K., and by U.S. Department of Agriculture National Research Initiative Grant 2002-01661 and Kentucky Science and Engineering Foundation Grant 200110151401 to D.H. and H.F. This study is publication 03-12-133 of the Kentucky Agricultural Experiment Station.
Article, publication date, and citation information can be found at www.plantcell.org/cgi/doi/10.1105/tpc.017301.
References
- Baudouin, E., Meskiene, I., and Hirt, H. (1999). Unsaturated fatty acids inhibit MP2C, a protein phosphatase 2C involved in the wound-induced MAP kinase pathway regulation. Plant J. 20, 343–348. [DOI] [PubMed] [Google Scholar]
- Bené, H., Lasky, D., and Ntambi, J.M. (2001). Cloning and characterization of the human stearoyl-CoA desaturase gene promoter: Transcriptional activation by sterol regulatory element binding protein and repression by polyunsaturated fatty acids and cholesterol. Biochem. Biophys. Res. Commun. 284, 1194–1198. [DOI] [PubMed] [Google Scholar]
- Bowling, S.A., Clarke, J.D., Liu, Y., Klessig, D.F., and Dong, X. (1997). The cpr5 mutant of Arabidopsis expresses both NPR1-dependent and NPR1-independent resistance. Plant Cell 9, 1573–1584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowling, S.A., Guo, A., Cao, H., Gordon, A.S., Klessig, D.F., and Dong, X. (1994). A mutation in Arabidopsis that leads to constitutive expression of systemic acquired resistance. Plant Cell 6, 1845–1857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao, H., Glazebrook, J., Clarke, J.D., Volko, S., and Dong, X. (1997). The Arabidopsis NPR1 gene that controls systemic acquired resistance encodes a novel protein containing ankyrin repeats. Cell 88, 57–63. [DOI] [PubMed] [Google Scholar]
- Clarke, J.D., Liu, Y., Klessig, D.F., and Dong, X. (1998). Uncoupling PR gene expression from NPR1 and bacterial resistance: Characterization of the dominant Arabidopsis cpr6-1 mutant. Plant Cell 10, 557–569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Creelman, R.A., and Mullet, J.E. (1997). Biosynthesis and action of jasmonates in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 48, 355–381. [DOI] [PubMed] [Google Scholar]
- Dahmer, M.L., Fleming, P.D., Collins, G.B., and Hildebrand, D.F. (1989). A rapid screening for determining the lipid composition of soybean seeds. J. Am. Oil Chem. Soc. 66, 534–538. [Google Scholar]
- Dangl, J.L., Dietrich, R.A., and Richberg, M.H. (1996). Death don't have no mercy: Cell death programs in plant–microbe interactions. Plant Cell 8, 1793–1807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delaney, T.P., Uknes, S., Vernooij, B., Friedrich, L., Weymann, K., Negrotto, D., Gaffney, T., Gut-Rella, M., Kessmann, H., Ward, E., and Ryals, J.A. (1994). A central role of salicylic acid in plant disease resistance. Science 266, 1247–1250. [DOI] [PubMed] [Google Scholar]
- Dempsey, D., Shah, J., and Klessig, D.F. (1999). Salicylic acid and disease resistance in plants. Crit. Rev. Plant Sci. 18, 547–575. [Google Scholar]
- Devadas, S.K., Enyedi, A., and Raina, R. (2002). The Arabidopsis hrl1 mutation reveals novel overlapping roles for salicylic acid, jasmonic acid and ethylene signalling in cell death and defence against pathogens. Plant J. 30, 467–480. [DOI] [PubMed] [Google Scholar]
- de Vries, J.E., Vork, M.M., Roemen, T.H., de Jong, Y.F., Cleutjens, J.P., van der Vusse, G.J., and van Bilsen, M. (1997). Saturated but not mono-unsaturated fatty acids induce apoptotic cell death in neonatal rat ventricular myocytes. J. Lipid Res. 38, 1384–1394. [PubMed] [Google Scholar]
- Diaz-Guerra, M.J., Junco, M., and Bosca, L. (1991). Oleic acid promotes changes in the subcellular distribution of protein kinase C in isolated hepatocytes. J. Biol. Chem. 266, 23568–23576. [PubMed] [Google Scholar]
- Doares, S.H., Narvaez-Vasquez, J., Conconi, A., and Ryan, C.A. (1995). Salicylic acid inhibits synthesis of proteinase inhibitors in tomato leaves induced by systemin and jasmonic acid. Plant Physiol. 108, 1741–1746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong, X. (2001). Genetic dissection of systemic acquired resistance. Curr. Opin. Plant Biol. 4, 309–314. [DOI] [PubMed] [Google Scholar]
- Epple, P., Apel, K., and Bohlmann, H. (1995). An Arabidopsis thaliana thionin gene is inducible via a signal transduction pathway different from that for pathogenesis-related proteins. Plant Physiol. 109, 813–820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falcone, D.L., Gibson, S., Lemieux, B., and Somerville, C. (1994). Identification of a gene that complements an Arabidopsis mutant deficient in chloroplast omega 6 desaturase activity. Plant Physiol. 106, 1453–1459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feys, B.J., and Parker, J.E. (2000). Interplay of signaling pathways in plant disease resistance. Trends Genet. 16, 449–455. [DOI] [PubMed] [Google Scholar]
- Flor, H. (1971). Current status of gene-for-gene concept. Annu. Rev. Phytopathol. 9, 275–296. [Google Scholar]
- Gaffney, T., Friedrich, L., Vernooij, B., Negrotto, D., Nye, G., Uknes, S., Ward, E., Kessmann, H., and Ryals, J.A. (1993). Requirement of salicylic acid for the induction of systemic acquired resistance. Science 261, 754–756. [DOI] [PubMed] [Google Scholar]
- Gray, W.M. (2002). Plant defence: A new weapon in the arsenal. Curr. Biol. 12, R352–R354. [DOI] [PubMed] [Google Scholar]
- Greenberg, J.T., Guo, A., Klessig, D.F., and Ausubel, F.M. (1994). Programmed cell death in plants: A pathogen-triggered response activated coordinately with multiple defense functions. Cell 77, 551–563. [DOI] [PubMed] [Google Scholar]
- Gupta, V., Willits, M.G., and Glazebrook, J. (2000). Arabidopsis thaliana EDS4 contributes to salicylic acid (SA)-dependent expression of defense responses: Evidence for inhibition of jasmonic acid signaling by SA. Mol. Plant-Microbe Interact. 13, 503–511. [DOI] [PubMed] [Google Scholar]
- Guzman, P., and Ecker, J.R. (1990). Exploiting the triple response of Arabidopsis to identify ethylene-related mutants. Plant Cell 2, 513–523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gyorfy, Z., Benko, S., Kusz, E., Maresca, B., Vigh, L., and Duda, E. (1997). Highly increased TNF sensitivity of tumor cells expressing the yeast delta 9-desaturase gene. Biochem. Biophys. Res. Commun. 241, 465–470. [DOI] [PubMed] [Google Scholar]
- Hammond-Kosack, K.E., and Jones, J.D.J. (1996). Resistance gene–dependent plant defense responses. Plant Cell 8, 1773–1791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harms, K., Atzorn, R., Brash, A., Kuhn, H., Wasternack, C., Willmitzer, L., and Pena-Cortes, H. (1995). Expression of a flax allene oxide synthase cDNA leads to increased endogenous jasmonic acid (JA) levels in transgenic potato plants but not to a corresponding activation of JA-responding genes. Plant Cell 7, 1645–1654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He, Y., Fukushige, H., Hildebrand, D.F., and Gan, S. (2002). Evidence supporting a role of jasmonic acid in Arabidopsis leaf senescence. Plant Physiol. 128, 876–884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jabs, T., Dietrich, R.A., and Dangl, J.L. (1996). Initiation of runaway cell death in an Arabidopsis mutant by extracellular superoxide. Science 273, 1853–1856. [DOI] [PubMed] [Google Scholar]
- James, D.W., Jr., Lim, E., Keller, J., Plooy, I., Ralston, E., and Dooner, H.K. (1995). Directed tagging of the Arabidopsis FATTY ACID ELONGATION1 (FAE1) gene with the maize transposon activator. Plant Cell 7, 309–319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kachroo, P., Kachroo, A., Lapchyk, L., Hildebrand, D., and Klessig, D. (2003). Restoration of defective cross talk in ssi2 mutants: Role of salicylic acid, jasmonic acid and fatty acids in SSI2-mediated signaling. Mol. Plant-Microbe Interact. 11, 1022–1029. [DOI] [PubMed] [Google Scholar]
- Kachroo, P., Shanklin, J., Shah, J., Whittle, E.J., and Klessig, D.F. (2001). A fatty acid desaturase modulates the activation of defense signaling pathways in plants. Proc. Natl. Acad. Sci. USA 98, 9448–9453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kachroo, P., Yoshioka, K., Shah, J., Dooner, H.K., and Klessig, D.F. (2000). Resistance to turnip crinkle virus in Arabidopsis is regulated by two host genes, is salicylic acid dependent but NPR1, ethylene and jasmonate independent. Plant Cell 12, 677–690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kates, M., Pugh, E.L., and Ferrante, G. (1984). Regulation of membrane fluidity by lipid desaturases. Biomembranes 12, 379–395. [Google Scholar]
- Keen, N.T. (1990). Gene-for-gene complementarity in plant-pathogen interactions. Annu. Rev. Genet. 24, 447–463. [DOI] [PubMed] [Google Scholar]
- Kim, J.H., Kim, Y., Lee, S.D., Lopez, I., Arnold, R.S., Lambeth, J.D., Suh, P.G., and Ryu, S.H. (1999). Selective activation of phospholipase D2 by unsaturated fatty acid. FEBS Lett. 454, 42–46. [DOI] [PubMed] [Google Scholar]
- Kirsch, C., Takamiya-Wik, M., Reinold, S., Hahlbrock, K., and Somssich, I.E. (1997). Rapid, transient, and highly localized induction of plastidial omega-3 fatty acid desaturase mRNA at fungal infection sites in Petroselinum crispum. Proc. Natl. Acad. Sci. USA 94, 2079–2084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klumpp, S., Selkem, D., and Hermesmeier, J. (1998). Protein phosphatase type 2C active at physiological Mg2+: Stimulation by unsaturated fatty acids. FEBS Lett. 437, 229–232. [DOI] [PubMed] [Google Scholar]
- Kumar, V.B., Vyas, K., Buddhiraju, M., Alshaher, M., Flood, J.F., and Morley, J.E. (1999). Changes in membrane fatty acids and delta-9 desaturase in senescence accelerated (SAMP8) mouse hippocampus with aging. Life Sci. 65, 1657–1662. [DOI] [PubMed] [Google Scholar]
- Kunkel, B.N., and Brooks, D.M. (2002). Cross talk between signaling pathways in pathogen defense. Curr. Opin. Plant Biol. 5, 325–331. [DOI] [PubMed] [Google Scholar]
- Kunst, L., Browse, J., and Somerville, C. (1988). Altered regulation of lipid biosynthesis in a mutant of Arabidopsis deficient in chloroplast glycerol-3-phosphate acyltransferase activity. Proc. Natl. Acad. Sci. USA 85, 4134–4147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kunst, L., Browse, J., and Somerville, C. (1989). Altered chloroplast structure and function in a mutant of Arabidopsis deficient in plastid glycerol-3-phosphate acyltransferase activity. Plant Physiol. 90, 846–853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laxalt, A.M., and Munnik, T. (2002). Phospholipid signalling in plant defence. Curr. Opin. Plant Biol. 5, 332–338. [DOI] [PubMed] [Google Scholar]
- Lee, S., Suh, S., Kim, S., Crain, R.C., Kwak, J.M., Nam, H.-G., and Lee, Y. (1997). Systemic elevation of phosphatidic acid and lysophospholipid levels in wounded plants. Plant J. 12, 547–556. [Google Scholar]
- Li, C., Liu, G., Xu, C., Lee, G.I., Bauer, P., Ling, H.Q., Ganal, M.W., and Howe, G.A. (2003). The tomato Suppressor of prosystemin-mediated responses2 gene encodes a fatty acid desaturase required for the biosynthesis of jasmonic acid and the production of a systemic wound signal for defense gene expression. Plant Cell 15, 1646–1661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, J., Ding, S.F., Habib, N.A., Fermor, B.F., Wood, C.B., and Gilmour, R.S. (1994). Partial characterization of a cDNA for human stearoyl-CoA desaturase and changes in its mRNA expression in some normal and malignant tissues. Int. J. Cancer 57, 348–352. [DOI] [PubMed] [Google Scholar]
- Lightner, J., James, D., Lark, E., and Browse, J. (1997). Novel mutations affecting leaf stearate content and plant size in Arabidopsis. Theor. Appl. Genet. 94, 975–981. [Google Scholar]
- Lightner, J., Wu, J., and Browse, J. (1994). A mutant of Arabidopsis with increased levels of stearic acid. Plant Physiol. 106, 1443–1451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maldonado, A.M., Doerner, P., Dixon, R.A., Lamb, C.J., and Cameron, R.K. (2002). A putative lipid transfer protein involved in systemic resistance signalling in Arabidopsis. Nature 419, 399–403. [DOI] [PubMed] [Google Scholar]
- Miquel, M., and Browse, J. (1992). Arabidopsis mutants deficient in polyunsaturated fatty acid synthesis: Biochemical and genetic characterization of a plant oleoyl-phosphatidylcholine desaturase. J. Biol. Chem. 267, 1502–1509. [PubMed] [Google Scholar]
- Murashige, T., and Skoog, F. (1962). A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant. 15, 473.–497. [Google Scholar]
- Niki, T., Mitsuhara, I., Seo, S., Ohtsubo, N., and Ohashi, Y. (1998). Antagonistic effect of salicylic acid and jasmonic acid on the expression of pathogenesis-related (PR) protein genes in wounded mature tobacco leaves. Plant Cell Physiol. 39, 500–507. [Google Scholar]
- Ntambi, J.M. (1995). The regulation of stearoyl-CoA desaturase (SCD). Prog. Lipid Res. 34, 139–150. [DOI] [PubMed] [Google Scholar]
- Ohlrogge, J., and Browse, J. (1995). Lipid biosynthesis. Plant Cell 7, 957–970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pena-Cortes, H., Albrecht, T., Prat, S., Weiler, E.W., and Willmitzer, L. (1993). Aspirin prevents wound-induced gene expression in tomato leaves by blocking jasmonic acid biosynthesis. Planta 191, 123–128. [Google Scholar]
- Penninckx, I.A.M.A., Eggermont, K., Terras, F.R.G., Thomma, B.P.H.J., De Samblanz, G.W., Buchala, A., Métraux, J.-P., Manners, J.M., and Broekaert, W.F. (1996). Pathogen-induced systemic activation of a plant defensin gene in Arabidopsis follows a salicylic acid-independent pathway. Plant Cell 8, 2309–2323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penninckx, I.A.M.A., Thomma, B.P.H.J., Buchala, A., Métraux, J.-P., and Broekaert, W.F. (1998). Concomitant activation of jasmonate and ethylene response pathways is required for induction of a plant defensin gene in Arabidopsis. Plant Cell 10, 2103–2113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piffanelli, P., and Murphy, D.J. (1999). Lipid accumulation and related gene expression in gametophytic and sporophytic anther tissues. In Fertilization in Higher Plants: Molecular and Cytological Aspects, M. Cresti, G. Cai, and A. Moscatelli, eds (Berlin: Springer), pp. 23–45.
- Rate, D.N., Cuenca, J.V., Bowman, G.R., Guttman, D.S., and Greenberg, J.T. (1999). The gain-of-function Arabidopsis acd6 mutant reveals novel regulation and function of the salicylic acid signaling pathway in controlling cell death, defenses, and cell growth. Plant Cell 11, 1695–1708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryals, J.A., Weymann, K., Lawton, K., Friedrich, L., Ellis, D., Steiner, H.Y., Johnson, J., Delaney, T.P., Jesse, T., Vos, P., and Uknes, S. (1997). The Arabidopsis NIM1 protein shows homology to the mammalian transcription factor inhibitor IκB. Plant Cell 9, 425–439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryu, S.B., and Wang, X. (1998). Increase in free linolenic and linoleic acids associated with phospholipase D-mediated hydrolysis of phospholipids in wounded castor bean leaves. Biochim. Biophys. Acta 1393, 193–202. [DOI] [PubMed] [Google Scholar]
- Schmid, J., and Amrhein, N. (1995). Molecular organization of the shikimate pathway in higher plants. Phytochemistry 39, 737–749. [Google Scholar]
- Seo, S., Sano, H., and Ohashi, Y. (1997). Jasmonic acid in wound signal transduction pathways. Physiol. Plant. 101, 740–745. [Google Scholar]
- Shah, J. (2003). The salicylic acid loop in plant defense. Curr. Opin. Plant Biol. 6, 365–371. [DOI] [PubMed] [Google Scholar]
- Shah, J., Kachroo, P., and Klessig, D.F. (1999). The Arabidopsis ssi1 mutation restores pathogenesis-related gene expression in npr1 plants and renders defensin gene expression salicylic acid dependent. Plant Cell 11, 191–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah, J., Kachroo, P., Nandi, A., and Klessig, D.F. (2001). A loss-of-function mutation in the Arabidopsis SSI2 gene confers SA- and NPR1-independent expression of PR genes and resistance against bacterial and oomycete pathogens. Plant J. 25, 563–574. [DOI] [PubMed] [Google Scholar]
- Shanklin, J., and Cahoon, E.B. (1998). Desaturation and related modifications of fatty acids. Annu. Rev. Plant Physiol. Plant Mol. Biol. 49, 611–641. [DOI] [PubMed] [Google Scholar]
- Shanklin, J., and Somerville, C. (1991). Stearoyl-acyl-carrier-protein desaturase from higher plants is structurally unrelated to the animal and fungal homologs. Proc. Natl. Acad. Sci. USA 88, 2510–2514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirano, Y., Kachroo, P., Shah, J., and Klessig, D.F. (2002). A gain-of-function mutation in an Arabidopsis Toll Interleukin1 Receptor–Nucleotide Binding Site–Leucine-Rich Repeat type R gene triggers defense responses and results in enhanced disease resistance. Plant Cell 14, 3149–3162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smalle, J., Haegman, M., Kurepa, J., Montagu, M.V., and Straeten, D.V.D. (1997). Ethylene can stimulate Arabidopsis hypocotyl elongation in the light. Proc. Natl. Acad. Sci. USA 94, 2756–2761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spoel, S.H., et al. (2003). NPR1 modulates cross-talk between salicylate- and jasmonate-dependent defense pathways through a novel function in the cytosol. Plant Cell 15, 760–770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staswick, P.E., Yuen, G.Y., and Lehman, C.C. (1998). Jasmonate signalling mutants of Arabidopsis are susceptible to the soil fungus Pythium irregulare. Plant J. 15, 747–754. [DOI] [PubMed] [Google Scholar]
- Straus, D.S., and Glass, C.K. (2001). Cyclopentenone prostaglandins: New insights on biological activities and cellular targets. Med. Res. Rev. 21, 185–210. [DOI] [PubMed] [Google Scholar]
- Thomma, B.P., Penninckx, I.A., Broekaert, W.F., and Cammue, B.P. (2001). The complexity of disease signaling in Arabidopsis. Curr. Opin. Immunol. 13, 63–68. [DOI] [PubMed] [Google Scholar]
- Thomma, B.P.H.J., Eggermont, K., Penninckx, I.A.M.A., Mauch-Mani, B., Vogelsang, R., Cammue, B.P.A., and Broekaert, W.F. (1998). Separate jasmonate-dependent and salicylate-dependent defense response pathways in Arabidopsis are essential for resistance to distinct microbial pathogens. Proc. Natl. Acad. Sci. USA 95, 15107–15111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomma, B.P.H.J., Eggermont, K., Tierens, K.F.M.-J., and Broekaert, W.F. (1999). Requirement of functional ethylene-insensitive 2 gene for efficient resistance of Arabidopsis to infection by Botrytis cinerea. Plant Physiol. 121, 1093–1101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uknes, S., Winter, A.M., Delaney, T., Vernooij, B., Morse, A., Friedrich, L., Nye, G., Potter, S., Ward, E., and Ryals, J.A. (1993). Biological induction of systemic acquired resistance in Arabidopsis. Mol. Plant-Microbe Interact. 6, 692–698. [Google Scholar]
- Van der Hoorn, R.A., De Wit, P.J., and Joosten, M.H. (2002). Balancing selection favors guarding resistance proteins. Trends Plant Sci. 7, 67–71. [DOI] [PubMed] [Google Scholar]
- Verberne, M.C., Verpoorte, R., Bol, J.F., Mercado-Blanco, J., and Linthorst, H.J. (2000). Overproduction of salicylic acid in plants by bacterial transgenes enhances pathogen resistance. Nat. Biotechnol. 18, 779–783. [DOI] [PubMed] [Google Scholar]
- Vijayan, P., Shockey, J., Levesque, C.A., Cook, R.J., and Browse, J. (1998). A role for jasmonate in pathogen defence of Arabidopsis. Proc. Natl. Acad. Sci. USA 95, 7209–7214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward, E.R., Uknes, S.J., Williams, S.C., Dincher, S.S., Wiederhold, D.L., Alexander, D.C., Al-Goy, P., Métraux, J.P., and Ryals, J.A. (1991). Coordinate gene activity in response to agents that induce systemic acquired resistance. Plant Cell 3, 1085–1094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber, H. (2002). Fatty acid derived signals in plants. Trends Plant Sci. 7, 217–224. [DOI] [PubMed] [Google Scholar]
- Xie, D.X., Feys, B.F., James, S., Nieto-Rostro, M., and Turner, J.G. (1998). COI1: An Arabidopsis gene required for jasmonate-regulated defense and fertility. Science 280, 1091–1094. [DOI] [PubMed] [Google Scholar]