Abstract
Alzheimer’s disease (AD) is the leading cause of dementia in elderly populations throughout the world and its incidence is on the rise. Current clinical diagnosis of AD requires intensive examination that includes neuropsychological testing and costly brain imaging techniques, and a definitive diagnosis can only be made upon postmortem neuropathological examination. Additionally, antemortem clinical AD diagnosis is typically administered following onset of cognitive and behavioral symptoms. As these symptoms emerge relatively late in disease progression, therapeutic intervention occurs after significant neurodegeneration, thereby limiting efficacy. The identification of noninvasive diagnostic biomarkers of AD is becoming increasingly important to make diagnosis more widely available to clinics with limited access to neuropsychological testing or state-of-the-art brain imaging, reduce the cost of clinical diagnosis, provide a biological measure to track the course of therapeutic intervention, and most importantly, allow for earlier diagnosis – possibly even during the prodromal phase – with hopes of therapeutic intervention prior to appreciable neurodegeneration. Circulating leukocytes are attractive candidate AD biomarkers as they can be obtained in a minimally invasive manner and are easily analyzed by widely available flow cytometry techniques. In this review, we critically analyze the potential utility of peripheral leukocytes as biological markers for AD.
Keywords: autoantibody, B lymphocyte, biological marker, cytokine, dementia, dendritic cell, granulocyte, immune, inflammation, macrophage, monocyte, natural killer cell, polymorphonuclear cell, T lymphocyte
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder pathologically characterized by deposition of amyloid-b (Ab) peptides as Ab plaques, intracellular neurofibrillary tangles, low-level chronic neuroinflammation and neuronal injury, resulting in a devastating loss of cognitive function [1]. AD has emerged as a national and international epidemic. It is the most common form of dementia, affecting an estimated 5.2 million Americans in 2008 alone, and that number is projected to increase to more than 13.2 million by 2050 [2,3]. While epidemiological studies have implicated genetic and environmental risk factors for AD, aging remains the most significant risk-incurring variable. Risk for developing AD is low before the age of 65 years, when prevalence for the disease is less than one person per 1000. Yet, AD prevalence is approximately 2.5–3% at 65 years of age, and it approximately doubles every 5 years until age 85–90 years, when it approaches 50% and appears to plateau [4–6]. However, AD prevalence is expected to increase as diagnosis of the disease improves and general medicine prolongs longevity.
While a ‘gold standard’ definitive diagnosis of AD can only be determined upon postmortem neuropathological examination, a battery of antemortem clinical tests may be employed to detect the disease with 85–90% accuracy. Neuropsychological assessment and neuroimaging procedures for AD, including mini-mental state examination (MMSE), AD assessment scale-cognitive subscale (ADAS-cog), behavioral pathology in AD rating scale (BEHAVE-AD), Consortium to Establish a Registry for AD (CERAD) neuropsychological battery, computed tomography (CT), MRI and PET, are noninvasive and sensitive diagnostic tools that are often relied upon to arrive at a clinical diagnosis of AD. Yet, AD clinical diagnoses are typically made only after the emergence of cognitive symptoms, when significant brain pathology and neuronal injury has already transpired. In fact, there is mounting evidence that pathogenic mechanisms manifest early within the particularly long AD prodromal phase (anywhere from 5 to 20 years), which includes the mild cognitive impairment (MCI) stage that is now generally regarded to precede dementia onset [5]. Thus, development of sensitive biological markers would ideally lead to an earlier clinical AD diagnosis that could be made during the extended prodromal stage of the disease, thereby allowing earlier and more effective therapeutic intervention.
As first proposed by Hardy and Allsop, the ‘amyloid cascade’ hypothesis purports that mismetabolism and deposition of Ab peptides as Ab plaques is the principal etiopathological event in AD, which triggers downstream events culminating in neuronal demise [7]. Ab peptides are normally generated in relatively small amounts by coordinated proteolytic cleavage of the amyloid precursor protein (APP), which is executed by enzymes known as secretases. However, at some point during the AD prodromal stage, APP metabolism and/or clearance goes awry, and Ab peptides begin accumulating on the inside and outside of neurons, forming aggregates of oligomers, protofibrils and fibrils. These aggregates combine with other cellular components to form a rigid, insoluble deposit known as an Ab plaque [8,9].
Innate immune responses mediated by activated microglia and astrocytes seem to occur in tight temporal and spatial proximity to these Ab deposits [10,11]. These immune/inflammatory cells respond by synthesizing and releasing a myriad of proinflammatory cytokines, chemokines, complement proteins, proteinases, cell surface immune activation molecules and reactive oxygen species (ROS) [12–19]. While this neuroinflammatory response may be beneficial for clearing infection and initiating tissue repair mechanisms, if left unresolved it exposes sensitive neurons to chronically elevated levels of potentially toxic molecules that can lead to bystander injury [20].
Although initially thought to be an epiphenomenon, multiple lines of evidence now show that neuroinflammation is more intimately involved in AD pathoetiology. This evidence includes data from over 25 epidemiologic studies that generally support an inverse risk relationship between the use of NSAIDs and AD [21–23]. These earlier reports were often case–control based [24]; however, additional support came from studies with more robust designs, including prospective studies with relatively long periods of follow-up [21,25–28]. Some of these cohort studies indicated increased risk reduction with longer duration of NSAID exposure. Two recent studies showed results that were contrary to the previous findings [29,30]. One possible explanation for the discordance is that both recent studies were conducted on older cohorts compared with the earlier work. The results of these observational reports and data from the animal literature also prompted clinical trials investigating whether NSAIDs were effective in treating already-established disease (treatment trials) [31–35], whether NSAIDs can prevent progression from MCI to AD (secondary prevention trial) [36], or whether NSAIDs can prevent incidence of AD in cognitively healthy people (primary prevention trial) [37]. In general, the results of the clinical trials have not been promising, with all but the earliest trial showing null (or even negative) findings. Interestingly, Breitner presented updated findings at the 2009 International Conference on Alzheimer’s Disease from the only primary prevention trial. These new results indicated a difference in treatment effect in early versus later follow-up periods. Specifically, in the early follow-up period, there was increased risk of AD in the NSAID group. However, in the later follow-up period (and after exclusion of individuals with baseline cognitive problems), the NSAID group showed a reduced risk of AD. Although the emerging picture is complex, it seems that timing and duration of exposure may be important determinants of NSAID effectiveness.
The above results and other evidence have led to the suggestion that age-associated low-grade chronic upregulation of certain types of immune/inflammatory responses that occurs with aging may be pathological during the prodromal stage of AD. Arguably, ‘inflammaging’ as it has been termed, is not restricted to the CNS, but is also apparent in circulating immune cells [38–40]. The key open questions are whether peripheral immune/inflammatory responses act as a driving force in AD pathoetiology, or are markers of aging-related neurodegeneration, including AD. As certain types of systemic leukocytes are long lived, it stands to reason that these cells may ‘sense’ AD-type CNS damage and respond by altering expression of immune molecules. If peripheral leukocytes could indicate AD-type CNS pathology, they would have considerable clinical utility as biological markers for the disease. Circulating leukocytes are particularly attractive as candidate AD biomarkers because they can be obtained in a minimally invasive manner and are easily analyzed in the clinic by widely available flow cytometry techniques. In this article, we critically review the potential for circulating leukocytes to serve as clinically relevant AD biomarkers.
The naive-to-memory T-lymphocyte switch
T-cell activation markers are perhaps the most widely studied peripheral leukocyte features that may serve as antecedent biomarkers for AD [40]. Alterations in peripheral immune cell phenotypes have previously been found to be associated with aging [41], which remains the greatest risk factor for AD. Interestingly, Monsonego and colleagues detected greater amounts Ab-reactive T cells in blood from healthy elderly individuals and individuals with AD compared with middle-aged adults, who demonstrated increased T-cell reactivity to self-antigen and shifts in T-cell characteristics as a function of age [42]. The ability for T cells to react to Ab is a prime demonstration of the adaptive immune response in the context of AD, and may occur via a shift from naive to memory T cells. Memory T cells express a distinct pattern of cell surface markers and respond in ways that are functionally different from naive cells. Specifically, upon a second encounter with a particular antigen (such as Ab) in the context of immunological costimulation, memory T cells can mount a faster and more vigorous immune response than naive cells.
Human memory T cells can be classified by relative expression levels of CD45 isoforms on the cell surface; they are CD45RA− and CD45RO + [43]. Utilizing flow cytometry, Tan and colleagues measured expression levels of CD45 isoforms on isolated CD4+ T lymphocytes from patients with clinical diagnoses of AD, cognitively abnormal individuals, normal elderly controls and patients with other (non-AD) forms of dementia [44]. These authors noted significantly lower levels of CD45RA and an increase in the CD45RO/CD45RA ratio on CD4 + T cells in patients diagnosed with probable AD and in cognitively abnormal individuals compared with age-matched normal participants or patients with other forms of dementia. In order to assess the validity of the CD45 isoform switch as a potential biomarker for AD, these authors genotyped their study subjects for the apolipoprotein E (APOE), the ε4 allele of which is the most robust genetic risk factor for AD and has been used as a diagnostic test [45–48]. Interestingly, a testing strategy combining APOE ε4 genotype with CD45RA or the CD45RO/CD45RA ratio increased sensitivity, specificity and predictive value over APOE ε4 alone and correctly classified AD approximately 75% of the time, suggesting that a combination of these biomarkers may be a useful AD diagnostic screen [44]. These results have been confirmed by Larbi and coworkers, who reported decreased percentages of naive cells and elevated numbers of circulating CD4 +, but not CD8 + T cells in patients with AD compared with controls [49]. Jago and colleagues further investigated expression of a negative T-cell costimulatory molecule, known as CTLA-4, on cultured human CD4 +/CD45RO + T cells, and found that CTLA-4 expression was enhanced in this subset. These data suggest that memory T cells may be undergoing anergy or tolerance, perhaps owing to persistent antigenic challenge [50]. Altered memory T cells do not seem to be restricted to the systemic compartment in the context of AD; rather, T lymphocytes, including CD45RO + T cells, can also be detected in AD patient brains, raising the possibility that altered T-cell profiles occur in response to AD-type neuropathology [51–53]. Although a shift from naive to memory T cells in the periphery seems to indicate altered T-cell responses in AD and may have utility as a biomarker for the disease, it is unclear whether such a test could predict MCI and, more importantly, which individuals would convert from MCI to AD.
Shifts in distribution of other T-cell subsets
In addition to a switch from naive to memory T lymphocytes, a number of studies suggest that the distribution of other types of peripheral T-cell subsets is altered in patients with AD, and CD4 + and CD8 + T cells have even been detected in the brain parenchyma of AD patients [51,52]. Yet, the notion that T-cell subset distributions are altered in AD remains controversial. In the aforementioned report by Larbi and colleagues, increased proportions of CD4 +, but not CD8 + cells were detected in AD patients. Additionally, these cells lacked the costimulatory receptor CD28, a B7 receptor that is constitutively active on naive T cells, further suggestive of a shift from naive to memory T cells in AD patients [49]. In an earlier study, Pirttilä and colleagues observed a selective decrease of CD8 + lymphocytes in patients with AD compared with age-matched controls [54], as have several other groups [55,56]. However, there are also conflicting reports. Recent work by Bonotis and collaborators failed to demonstrate differences between percentages of CD4 + or CD8 + T cells by flow cytometry in the peripheral blood of mild-to-moderate AD patients versus healthy controls. However, these researchers did find a positive correlation between CD4 + T-cell percentages and patients’ cognitive state as assessed by MMSE score [57]. In fact, several earlier reports were unable to detect statistically significant differences on peripheral CD4 + and CD8 + T-cell subset distributions [58–61] . Even if such alterations in peripheral T-cell subset distributions do occur, these changes may not be specific to dementia of the Alzheimer type. This has been suggested by an analysis of peripheral T-cell subsets in AD and other forms of dementia, where Hu and colleagues demonstrated a decreased CD8+ lymphocyte subpopulation in all-cause dementia, suggesting that such an analysis may not be able to specifically identify AD patients but is instead a universal dementia index [62]. Potential explanations for variable results regarding T-cell subsets may include sample size, sample population demographics and differences in methodologies between groups. Nonetheless, the aforementioned contradictory findings call into question the utility of CD4 + and CD8 + T-cell subset distributions as a robust diagnostic indicator of AD.
Soluble CD40 & soluble CD40 ligand as biomarkers
Two integral players in the pathogenic immune/inflammatory response that occurs in AD are the cell surface molecule CD40 (a type I integral membrane glycoprotein) and its cognate ligand, CD40 ligand (CD40L; a trimeric type II membrane glycoprotein). This immunoregulatory dyad serves to orchestrate proinflammatory adaptive and innate immune responses. Within the CNS, CD40 is expressed by several cell types, including vascular endothelial cells, smooth muscle cells, astrocytes, neurons and microglia, which are resident brain macrophages that are critical in mediating the innate immune proinflammatory response to CD40L [16,18,63–70]. In the CNS, CD40L is expressed by activated astrocytes [71]. In the periphery, CD40L is most typically expressed by activated T lymphocytes, and is capable of triggering a series of intracellular proinflammatory signaling events, culminating in secretion of potentially neurotoxic cytokines and chemokines [63,72–74]. Importantly, CD40L exists not only as a cell membrane-associated glycoprotein, but also occurs in a soluble, secreted form that retains biological activity to bind to and activate membrane-bound CD40 [75,76]. Furthermore, CD40 can also be secreted in a soluble form, which may serve to neutralize CD40L [77].
Previous reports have clearly established the pathogenic contribution of CD40–CD40L interaction to AD. In an early report, it was demonstrated that CD40L enabled microglial activation in response to low levels of soluble Ab peptides. This form of microglial activation was pathogenic, as it led to the release of TNF-a, which was neurotoxic [15]. Furthermore, when transgenic APPsw mice (line 2576; a mouse model of AD), which overproduce Ab1–40 and Ab1–42 and develop age-dependent cerebral amyloidosis, were crossed with mice deficient in CD40L, these TgAPPsw/CD40L-deficient mice manifested less neuroinflammation and gliosis, reduced hyperphosphorylation of tau (a key index of neuronal stress) and reduction in cerebral amyloidosis [15,17]. These authors undertook a pharmacological approach to elucidate the role of the CD40/CD40L interaction in Ab plaque pathology in an accelerated PSAPP mouse model of AD [78]. In this study, PSAPP mice were treated with a CD40L depleting antibody, and this resulted in marked attenuation of Ab plaque pathology as well as superior spatial memory performance in the Morris water maze task compared with PSAPP mice that received a nonrelevant isotype-matched control antibody [17,79].
Expression of both CD40 and CD40L is altered in AD patient brains and in the mouse models of AD [66,71]. In one study, levels of soluble CD40 receptor (sCD40; which may be derived from innate immune cells) in plasma were found to be higher in AD patients compared with controls (that consisted of healthy young and aged subjects), but no difference was detected between patients with other forms of dementia or neurodegenerative disease and these control groups [77]. Interestingly, the positive relationship between sCD40 and AD was independent of the APOE ε4 allele, suggesting that sCD40 was adding predictive value over and above APOE genotype. Furthermore, it has recently been suggested that soluble CD40 ligand (sCD40L; possibly produced by activated lymphocytes or platelets) could be a potential AD biomarker. Specifically, it was found that patients with plasma concentrations of 6.0 ng/ml and above incur a threefold greater risk of suffering cognitive decline in the following 2 years [80,81]. Importantly, the subjects in the Desideri and coworkers study did not use drugs that could potentially affect sCD40L levels, including statins, antioxidants or anti-inflammatory agents. In a recent study, the authors examined both sCD40 and sCD40L levels in plasma and found that they were increased in AD patients compared with nondemented controls that had similar demographics. When these authors combined plasma sCD40, sCD40L, Ab, and APOE ε4 genotype, they found a high sensitivity and specificity (>90%) for this panel as a predictor of clinical AD diagnosis. The consistent findings in the above studies make a compelling case for using both sCD40 and sCD40L as plasma biomarkers of AD; however, it should be noted that these measures would probably need to be combined with others in order to attain the requisite sensitivity, specificity and predictive value to be useful biomarkers.
Apoptosis markers in T lymphocytes
It has been hypothesized that with aging comes a decline in anti-oxidant mechanisms and, consequently, an increase in damage owing to production of ROS. This hypothesis, known as the ‘free radical theory of aging’ originated from Harman in the 1950s and continues to draw support even today [82,83]. Several reports have applied this theory to AD, suggesting that failure of anti-oxidant mechanisms and increased inflammation associated with aging leads to oxidative damage and promotes neuronal injury and demise [39,84,85]. Interestingly, Eckert and colleagues showed that similar oxidative stressors can induce comparable amounts of apoptosis in neurons and in T cells [86], raising the possibility that peripheral T cells, such as neurons, may be susceptible to oxidative stress as a function of age and perhaps more so in AD. Further studies showed that T cells derived from elderly individuals were more susceptible to apoptosis induced by the oxidative stressor 2-deoxyribose than those derived from young subjects [87]. Furthermore, T cells expressing a presenilin 1 mutation linked to familial early-onset AD demonstrate hypersensitivity to cell death as do peripheral lymphocytes derived from AD patients [88]. These findings are consistent with an earlier observation that peripheral leukocytes from AD patients have elevated levels of intracellular calcium, a prerequisite for apoptosis [89]. Building on these results, Richartz and colleagues investigated levels of soluble Fas/CD95, a key proapoptotic receptor implicated in a number of inflammatory diseases, in serum from AD patients and controls. These investigators found increased serum levels of Fas/CD95 in a subgroup of AD patients compared with controls, indicating a proapoptotic alteration in the peripheral milieu in AD [90]. These results are in concert with previous data showing increased expression of Fas/CD95 on CD4 + T cells in AD patients compared with healthy age-matched controls [91]. Selective vulnerability to programmed cell death appears to be altered in T cells derived from AD patients, particularly in the presence of ROS. Measuring the amount of ROS and Fas/CD95 in the blood has the potential to be a peripheral indicator of AD-type neurodegeneration. However, lymphocytes derived from elderly individuals without AD also show susceptibility to apoptosis induced by oxidative stress [87], and the molecular mechanism by which ROS induce cell death is not well characterized. While the above results were generally obtained in a relatively small number of subjects, they hint at the possibility of using apoptotic markers on T lymphocytes as AD biomarkers. However, it will be important to determine whether these proapoptotic changes are specific to AD lymphocytes or occur more generally as a result of the aging process or other forms of neurodegeneration.
Changes in B-lymphocyte population distribution & phenotype
As a logical corollary of the hypothesis that the character and distribution of T-cell populations are altered in AD, changes in B-lymphocyte population distribution and characteristics have also been investigated in AD patients. A number of studies entertain the concept that lymphopenia accompanies dementia of the Alzheimer type, suggesting that perhaps general immunosenescence may be an indicator of AD pathological changes. Along these lines, Richartz-Salzburger and coworkers reported significantly diminished quantities of both CD3 + T cells and CD19+ B cells in patients with probable AD compared with age-matched controls [61]. These findings agree with earlier reports, where decreases in both T- and B-cell subsets were exhibited in AD patients compared with age-matched controls [92,93]. By contrast, other investigations have yielded no significant differences in the distribution of CD19+ B cells or total lymphocytes between individuals with AD and control subjects [60,94]. While Magaki and colleagues also found no difference in the distribution of CD19+ cells in patients with MCI, they did observe increased lymphocytic expression of APP in these individuals [95]. Previous studies have suggested that lymphocyte surface expression of APP occurs primarily on B cells [96–98]. Importantly, disturbances in the regulation of expression or processing of APP on these lymphocytes may be reflective of dysregulated APP metabolism in the brain [97,98]. Reduced surface expression of the early activation cell surface antigen CD69 on B cells following mitogenic challenge has also been demonstrated in AD patients [99]. These characteristic changes in APP and CD69 expression in the B-cell proteome seem to suggest that B lymphocytes may be useful cellular biomarkers of AD. Such findings also beg the question of whether a systemic failure of B cells to properly activate and process APP contributes more directly to AD pathoetiology. However, further work will be necessary to begin to address these important questions.
Changes in Ab autoantibody repertoire
If we accept that B lymphocytes are altered in AD, then the hypothesis arises that humoral (antibody) responses mediated by differentiated B cells (known as plasma cells) may be similarly perturbed in the circulation. Although a general decline in humoral immunity occurs with normal aging, it has been hypothesized that there are specific changes in both the quantity and quality of immunoglobulins secreted by plasma cells from AD patients. Given the placement of Ab peptides at the epicenter of AD pathoetiology, secreted autoantibodies directed against the Ab peptide have been the most well studied. Two of the four initial studies examining Ab autoantibodies in humans reported that they were significantly lower in AD patients compared with age-matched controls, while the remaining two studies reported that levels were unchanged [100–103]. Brettschneider and coworkers observed similar decreases in serum Ab1–42 peptide autoantibodies in AD patients, but ultimately concluded that these antibodies were poor diagnostic markers of AD owing to weak sensitivity [104]. While Baril and colleagues found no significant differences in serum concentrations of antibodies directed against both Ab1–40 and Ab1–42 in AD patients, they also concluded that these autoantibodies were unlikely to be useful AD biomarkers [105]. However, a number of recent studies have consistently suggested the contrary: that circulating Ab autoantibodies increase in AD patients and may be highly correlated with incidence of the disease. Multiple studies have found as much as four-fold increased levels of circulating Ab autoantibodies in AD patients relative to control (non-AD) subjects [106–109]. It has even been suggested that these autoantibodies may enhance the neurotoxic activity of the Ab peptide [106]. At least three reports to date have demonstrated increased Ab1–42 antibody titers in AD patients, and it has also been determined that antibody titers strongly correlated with global dementia scores [108–110]. Yet, conflicting reports illustrate the controversy surrounding the use of Ab autoantibodies as AD biomarkers.
However, a recent study from Britschgi and colleagues may shed some light on a cause for discordance in the literature: methodology. While a number of previous studies failed to assess the detailed molecular signature of Ab antibodies (i.e., whether these antibodies were free or bound to Ab, or exhibit avidity for specific Ab assemblies, such as mononomers, oligomers, or fibrils), the authors of this study carefully investigated the levels and molecular profile of Ab autoantibodies in the plasma of AD patients and age-matched controls. Interestingly, they found no overall differences in anti-Ab antibodies between a diverse group of AD patients and controls; however, patients with moderate-to-severe AD had significantly lower levels of antibody reactivity against oligomeric preparations of Ab1–42 than patients with mild disease. Britschgi and colleagues should be commended for setting a new standard for analysis of anti-Ab antibodies in AD [111]; however, the diagnostic utility of these specific immunoglobulins is yet to be determined. In contrast to their observation that autoantiboides against redox-modified cross-linked oligomeric species of Ab (anti-CAPS) were unchanged, a previous study by Moir and coworkers found significantly reduced levels of anti-CAPS antibodies in AD patients compared with nondemented controls. Furthermore, Moir et al. reported that the age at onset for AD correlated significantly with plasma immunoreactivity to CAPS [112]. This work, along with another study demonstrating elevation of autoantibodies specific to Ab25–35 oligomers in AD [107], would suggest that additional factors other than methodology may be affecting the reproducibility of Ab autoantibody studies. Whether or not a consensus can be reached regarding specific Ab autoantibody changes and their diagnostic capacity in AD remains an open question.
Natural killer cell activity
Natural killer (NK) cells are often regarded as a bridge between the innate and adaptive immune systems [113]. These intrinsically cytotoxic lymphocytes are critically involved in the elimination of both tumorigenic and virally infected cells. NK cells may initiate cytolytic or apoptotic processes in their cellular targets which lack MHC surface expression. As such, NK cell activity is tightly regulated in the periphery and in the CNS by way of the neuroendocrine system. Aside from their role in this neuroimmune interface, NK cells would seem to be far-removed from the neurodegenerative mechanisms involved in AD. In support of a null association, multiple studies failed to show any CNS infiltration or appreciable differences in NK populations (as defined by CD3−/CD16 +/CD56 + cells) in AD patients as compared with nondemented controls. However, differences in the responsiveness of NK cells to stimulatory or inhibitory cues in AD patients have been reported. An initial study by Araga and colleagues found that the amounts of NK activity induced by IL-2 or IFN-a in cells isolated from AD patients were significantly lower than normal control cells [114]. Conversely, there is evidence of enhanced cytotoxic responses by NK cells in subjects with AD [115]. More recent investigations demonstrated an overall increased sensitivity of these lymphocytes to physiological modifiers [116,117]. Moreover, a longitudinal study by Prolo and coworkers suggests that changes in the response of NK cells to negative (cortisol) or positive (IL-2) modifiers follow the progression of AD [117]. Despite a lack of additional studies in this area and some discrepancies in the data reported on NK cell phenotypes in AD, these lymphocytes may yet prove to be valuable diagnostic tools for the disease.
Altered immune responses in monocytes & macrophages
Monocytes/macrophages (M/M) represent obvious leukocytes to target as potential AD biomarkers, owing to their primary role as phagocytes that may serve to limit cerebral amyloidosis. Stimulated M/M synthesize the chemical neopterin, which can be used as an index of innate immune inflammatory status in the circulation. In a study by Leblhuber and colleagues, serum levels of neopterin were found to be higher in AD patients than age-matched controls [118]. These investigators also found an inverse correlation between MMSE scores and neopterin serum concentrations, suggesting that neopterin levels may signal severity of AD cognitive impairment. Furthermore, the abundance of neopterin synthesized by activated M/M correlates with their capacity to release ROS [119]. Although elevated neopterin levels may indicate a proinflammatory, proapoptotic immune response in AD, appreciable overlap between AD and control subjects limits the clinical relevance of neopterin as a biomarker for AD. Additionally, the inverse correlation between MMSE score and neopterin levels as reported by Leblhuber and coworkers was not reproduced in a similarly designed study by Hull and colleagues [120].
Defective phagocytosis of Ab by microglia, the resident macrophages of the brain, and by peripheral M/M has been suggested to be a pathogenic mechanism in AD [121–124]. Recent work by Avagyan and colleagues found that phagocytosis of Ab was significantly depressed in M/M isolated from AD patients compared with a control group of university professors. In this study, peripheral blood mononuclear cells (PBMCs) were isolated from individuals and treated with Ab overnight, and then analyzed by flow cytometry for the M/M cell surface marker CD14. Patients with sporadic AD were found to have two distinct blood monocyte biomarkers: defective Ab phagocytosis as defined by reduced mean fluorescence intensity of Ab in CD14 + monocytes, and transcriptional defects in these cells after stimulation with curcuminoid [125], a derivative of curcumin (a component of the curry spice tumeric) previously suggested to increase phagocytic clearance of Ab [126]. While PBMC Ab phagocytosis, as conducted by Avagyan and coworkers, reasonably distinguishes between patients with AD and highly functioning university professors (who can be classified as ‘supercontrols’), this marker demonstrated poor class prediction of AD patients when compared with MCI patients, nondemented AD patient caregivers or amyotrophic lateral sclerosis patients. The authors attribute the reduction in specificity to stress and lack of cognitive and physical exercise among these control groups; yet, a case for PBMC Ab phagocytosis as a biomarker for AD will be difficult to make unless these clear specificity and class prediction issues can be resolved.
Additional work characterizing altered M/M populations in peripheral blood by Kusdra and collaborators demonstrated elevated percentages of CD14 +/CD69 + monocytes in AD patients versus age-matched controls [127]. Interestingly, when conditioned media from M/M cultures was added to neural cells in vitro, the greatest amount of cell death was induced by supernatants from AD patients’ M/M that had a higher percentage of CD14 +/CD69 + expression. Therefore, the elevated percentage of CD14 +/CD69 + cells in AD patients versus age-matched controls may be an indication of the general heightened activation state of the innate immune system in these patients; however, this immune phenotype is probably not specific to AD (e.g., sepsis patients would be expected to demonstrate this immune activation phenotype and by the authors’ own admission they observed this immunophenotype in patients with HIV dementia), thus the issue of specificity would need to be addressed again.
Polymorphonuclear cell phenotype
Polymorphonuclear (PMN) leukocytes (also known as granulocytes) may be a largely overlooked peripheral immune cell in terms of AD biomarkers. Studies have suggested that these cells may be activated by Ab and are capable of trafficking to and infiltrating the CNS in instances of inflammation. At present, however, no definitive evidence has emerged linking PMNs with AD neuropathology. Nonetheless, studies examining changes in the pool of circulating PMNs in the context of AD have yielded curious results. While Song and coworkers found significant decreases in percentages of circulating basophils from AD patients [128], others have demonstrated an overall increase in the presence of CD14 + PMNs in MCI patients versus controls. For example, Licastro et al. found that the metabolic activity of circulating neutrophils from patients with AD was significantly elevated compared with nondemented controls [129]. In addition, increases in both oxidative stress and induction of CD11b expression on neutrophils have been shown in AD patients [130]. In another interesting report, Költringer et al. analyzed the rheological (physical) properties of PMNs from AD patients. Although they point out that statistical significance was not reached owing to the small number of AD patients used in their prospective study, the authors suggest that granulocytes appear to be more physically rigid in AD [131]. Altogether, these studies seem to suggest the existence of a systemic compartmentalized PMN response in AD. There does not seem to be any overt inconsistency in this literature; however, additional studies focused on dissecting out the status of individual sub-populations of granulocytes (i.e., eosinophils, basophils and neutrophils) in AD are required before any conclusions can be drawn regarding whether these peripheral immune cells that are often overlooked in the context of AD can provide diagnostic information relating to disease state.
Expert commentary
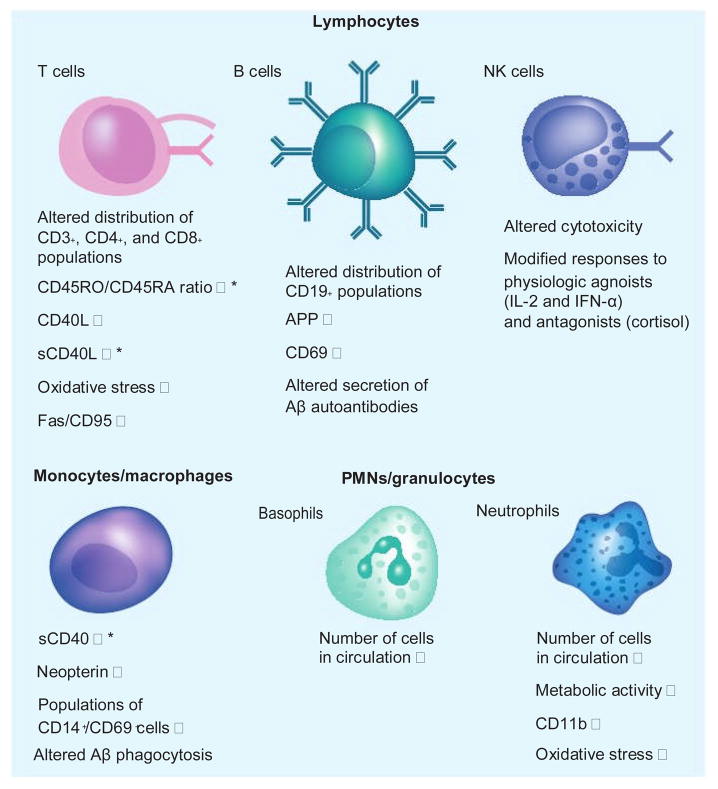
The Biomarkers and Surrogate Endpoint Working Group, under the auspices of the Office of the Director, National Institutes of Health defined the term ‘biomarker’ as: “a characteristic that is objectively measured and evaluated as an indicator of normal biological processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention” [132]. In this review, we have explored the potential of different subpopulations of circulating leukocytes to serve as biological markers for AD (Figure 1). It has been hypothesized that peripheral leukocytes may serve as ‘sensors’ for the neurodegeneration and/or neuroinflammation hallmarks of AD. Furthermore, some believe that changes in peripheral leukocytes may reflect these processes early in the prodromal phase of the disease, and perhaps allow for the early diagnosis of AD in a noninvasive manner. If this were the case, then a simple and relatively noninvasive blood collection procedure may feasibly provide all the necessary diagnostic information, as both serum proteins and groups of leukocytes could be isolated and subsequently analyzed for putative AD biomarkers. A subset of clinical and laboratory data support this notion, and we have critically examined many of these reports in this review. Furthermore, an interesting recent report demonstrated that 18 signaling proteins in plasma, many of which were immune/inflammatory molecules, could be used to correctly classify AD diagnosis with nearly 90% accuracy [133], further bolstering the idea that peripheral leukocyte-mediated immune/inflammatory responses may be useful AD biomarkers.
Figure 1. Leukocytic alterations in patients with Alzheimer’s disease and mild cognitive impairment.
Candidate biomarkers were selected based on criteria of high sensitivity, specificity, and consistency of association with disease in the literature.
*Candidate biomarkers.
However, studies have not yet led to the identification of a definitive leukocyte biomarker for AD. While the multiple leukocytic analytes discussed here are all potentially related to the immune/inflammatory responses present in AD, none of these biomarkers have unequivocally been associated with incidence or progression of the disease. While such definitive studies are difficult to conduct owing to the inclusion of an exhaustive list of leukocyte markers and the large sample sizes needed, such unbiased approaches will ultimately be necessary to determine the utility of peripheral leukocytes as AD biomarkers. It is likely that, in the case of a complex disease such as AD, a multimodal biomarker approach will be needed to attain acceptable sensitivity, specificity, predictive value and reliability. Furthermore, such an approach will probably need to be adopted to have any chance of diagnosing presymptomatic neurodegeneration and to assess the efficacy of AD therapeutics [134].
Five-year view
As the longevity of the human population increases and additional therapeutic approaches become available, so too does the importance of a reliable biomarker for AD. Although there are tantalizing reports demonstrating altered peripheral leukocyte phenotypes in AD, this field has been plagued by inconsistency and irreproducible results. Differences in findings may be owing to variations in methodology, small sample sizes and heterogeneity of sample populations. Generalized changes in immunity that accompany the aging process also confound the identification of suitable leukocytic biomarkers for AD. These, and other important questions, remain regarding the diagnostic potential of peripheral immune cell alterations for AD. Whether these immunophenotypic changes result from the highly developed dialog between the CNS and the peripheral immune compartment or are due to a generalized ‘inflammaging’ phenomenon remains to be elucidated in future years.
Even if we are able to uncover robust immune-based biomarkers for AD within the next 5 years, such findings would undoubtedly raise more questions than answers. Due to the inherently correlative nature of biomarker studies, an obvious query will be: what is the etiologic relevance of leukocyte biomarkers? A related question that can be raised is whether alterations in peripheral leukocytes are caused by AD-type neuropathological changes or perhaps even contribute to the disease. While it is still too early on in the field of leukocyte biomarkers for AD, the answers to these questions are tantalizing nonetheless. Some of these questions may be answered by utilizing genetically engineered mouse models of AD, which provide a more controllable system within which to address both the AD leukocyte biomarker hypothesis and whether these cells are pathoetiological contributors to disease. We look forward to results from these and related studies as the field of leukocyte AD biomarkers moves into the future.
Key issues
Alzheimer’s disease (AD) has significant immune and inflammatory components, and circulating leukocytes may represent valuable biomarkers for the disease.
Certain lymphocyte populations, such as ‘memory’ CD4+ T cells, are attractive candidate biological markers for AD.
Most of the studies suggesting that peripheral leukocytes can be biomarkers for AD are plagued by other reports showing null effects or contrary results.
Future studies designed with large subject numbers will be needed to definitively establish whether candidate leukocyte populations are indeed robust AD biomarkers.
Footnotes
For reprint orders, please contact reprints@expert-reviews.com
Financial & competing interests disclosure
Terrence Town is supported by a NIH/National Institute on Aging ‘Pathway to Independence’ grant (5R00AG029726–03) and by Cedars-Sinai Medical Center, Department of Neurosurgery and Department of Biomedical Sciences Faculty start-up funds. Terrence Town is the inaugural holder of the Ben Winters Endowed Chair in Regenerative Medicine.
The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
Contributor Information
Kavon Rezai-Zadeh, Email: rezaik@cshs.org, Department of Biomedical Sciences, Cedars-Sinai Medical Center, Los Angeles, CA 90048, USA, Tel.: +1 310 423 7611, Fax: +1 310 423 0302.
David Gate, Email: gated@cshs.org, Department of Neurosurgery, Maxine Dunitz Neurosurgical Institute, Los Angeles, CA 90048, USA, Tel.: +1 310 423 7611, Fax: +1 310 423 0302.
Christine A Szekely, Email: szekelyc@cshs.org, Department of Medicine, Samuel Oschin Comprehensive Cancer Institute, Cedars-Sinai Medical Center, Los Angeles, CA 90048, USA, Tel.: +1 310 423 6887, Fax: +1 310 423 8300.
Terrence Town, Email: terrence.town@cshs.org, Departments of Neurosurgery and Biomedical Sciences, Cedars-Sinai Medical Center, Department of Medicine, David Geffen School of Medicine at UCLA, 8700 Beverly Blvd, Davis Building, Room 2091, Los Angeles, CA 90048, USA, Tel.: +1 310 423 1202, Fax: +1 310 423 0302.
References
- 1.Selkoe DJ. Alzheimer’s disease: genes, proteins, and therapy. Physiol Rev. 2001;81(2):741–766. doi: 10.1152/physrev.2001.81.2.741. [DOI] [PubMed] [Google Scholar]
- 2.Hebert LE, Scherr PA, Bienias JL, Bennett DA, Evans DA. Alzheimer disease in the US population: prevalence estimates using the 2000 census. Arch Neurol. 2003;60(8):1119–1122. doi: 10.1001/archneur.60.8.1119. [DOI] [PubMed] [Google Scholar]
- 3.Plassman BL, Langa KM, Fisher GG, et al. Prevalence of dementia in the United States: the Aging, Demographics, and Memory study. Neuroepidemiology. 2007;29(1–2):125–132. doi: 10.1159/000109998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brookmeyer R, Gray S, Kawas C. Projections of Alzheimers-disease in the United States and the public health impact of delaying disease onset. Am J Public Health. 1998;88(9):1337–1342. doi: 10.2105/ajph.88.9.1337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Petersen RC. Challenges of epidemiological studies of mild cognitive impairment. Alzheimer Dis Assoc Disord. 2004;18(1):1–2. doi: 10.1097/00002093-200401000-00001. [DOI] [PubMed] [Google Scholar]
- 6.Tanzi RE. Alzheimer’s disease and related dementias: the road to intervention. Exp Gerontol. 2000;35(4):433–437. doi: 10.1016/s0531-5565(00)00109-1. [DOI] [PubMed] [Google Scholar]
- 7.Hardy J, Allsop D. Amyloid deposition as the central event in the aetiology of Alzheimer’s disease. Trends Pharmacol Sci. 1991;12(10):383–388. doi: 10.1016/0165-6147(91)90609-v. [DOI] [PubMed] [Google Scholar]
- 8.Sipe JD, Cohen AS. Review: history of the amyloid fibril. J Struct Biol. 2000;130(2–3):88–98. doi: 10.1006/jsbi.2000.4221. [DOI] [PubMed] [Google Scholar]
- 9.Walsh DM, Klyubin I, Fadeeva JV, Rowan MJ, Selkoe DJ. Amyloid-b oligomers: their production, toxicity and therapeutic inhibition. Biochem Soc Trans. 2002;30(4):552–557. doi: 10.1042/bst0300552. [DOI] [PubMed] [Google Scholar]
- 10.Benzing WC, Wujek JR, Ward EK, et al. Evidence for glial-mediated inflammation in aged APP(SW) transgenic mice. Neurobiol Aging. 1999;20(6):581–589. doi: 10.1016/s0197-4580(99)00065-2. [DOI] [PubMed] [Google Scholar]
- 11.McGowan E, Sanders S, Iwatsubo T, et al. Amyloid phenotype characterization of transgenic mice overexpressing both mutant amyloid precursor protein and mutant presenilin 1 transgenes. Neurobiol Dis. 1999;6(4):231–244. doi: 10.1006/nbdi.1999.0243. [DOI] [PubMed] [Google Scholar]
- 12.McGeer EG, McGeer PL. The importance of inflammatory mechanisms in Alzheimer disease. Exp Gerontol. 1998;33(5):371–378. doi: 10.1016/s0531-5565(98)00013-8. [DOI] [PubMed] [Google Scholar]
- 13.McGeer EG, McGeer PL. Inflammatory processes in Alzheimer’s disease. Prog Neuropsychopharmacol Biol Psychiatry. 2003;27(5):741–749. doi: 10.1016/S0278-5846(03)00124-6. [DOI] [PubMed] [Google Scholar]
- 14.Akiyama H, Barger S, Barnum S, et al. Inflammation and Alzheimer’s disease. Neurobiol Aging. 2000;21(3):383–421. doi: 10.1016/s0197-4580(00)00124-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tan J, Town T, Paris D, et al. Microglial activation resulting from CD40–CD40L interaction after β– αμΨλοιδ stimulation. Science. 1999;286(5448):2352–2355. doi: 10.1126/science.286.5448.2352. [DOI] [PubMed] [Google Scholar]
- 16.Tan J, Town T, Mullan M. CD40–CD40L interaction in Alzheimer’s disease. Curr Opin Pharmacol. 2002;2(4):445–451. doi: 10.1016/s1471-4892(02)00180-7. [DOI] [PubMed] [Google Scholar]
- 17.Tan J, Town T, Crawford F, et al. Role of CD40 ligand in amyloidosis in transgenic Alzheimer’s mice. Nat Neurosci. 2002;5(12):1288–1293. doi: 10.1038/nn968. [DOI] [PubMed] [Google Scholar]
- 18.Town T, Tan J, Mullan M. CD40 signaling and Alzheimer’s disease pathogenesis. Neurochem Int. 2001;39(5–6):371–380. doi: 10.1016/s0197-0186(01)00044-4. [DOI] [PubMed] [Google Scholar]
- 19.Town T, Nikolic V, Tan J. The microglial ‘activation’ continuum: from innate to adaptive responses. J Neuroinflammation. 2005;2:24. doi: 10.1186/1742-2094-2-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wyss-Coray T, Mucke L. Inflammation in neurodegenerative disease a double-edged sword. Neuron. 2002;35(3):419–432. doi: 10.1016/s0896-6273(02)00794-8. [DOI] [PubMed] [Google Scholar]
- 21.in t’ Veld BA, Ruitenberg A, Hofman A, et al. Nonsteroidal antiinflammatory drugs and the risk of Alzheimer’s disease. N Engl J Med. 2001;345(21):1515–1521. doi: 10.1056/NEJMoa010178. [DOI] [PubMed] [Google Scholar]
- 22.Szekely CA, Thorne JE, Zandi PP, et al. Nonsteroidal anti-inflammatory drugs for the prevention of Alzheimer’s disease: a systematic review. Neuroepidemiology. 2004;23(4):159–169. doi: 10.1159/000078501. [DOI] [PubMed] [Google Scholar]
- 23.Szekely CA, Town T, Zandi PP. NSAIDs for the chemoprevention of Alzheimer’s disease. Subcell Biochem. 2007;42:229–248. doi: 10.1007/1-4020-5688-5_11. [DOI] [PubMed] [Google Scholar]
- 24.McGeer PL, Schulzer M, McGeer EG. Arthritis and anti-inflammatory agents as possible protective factors for Alzheimer’s disease: a review of 17 epidemiologic studies. Neurology. 1996;47(2):425–432. doi: 10.1212/wnl.47.2.425. [DOI] [PubMed] [Google Scholar]
- 25.Zandi PP, Anthony JC, Hayden KM, Mehta K, Mayer L, Breitner JC. Reduced incidence of AD with NSAID but not H2 receptor antagonists: the Cache County study. Neurology. 2002;59(6):880–886. doi: 10.1212/wnl.59.6.880. [DOI] [PubMed] [Google Scholar]
- 26.Stewart WF, Kawas C, Corrada M, Metter EJ. Risk of Alzheimer’s disease and duration of NSAID use. Neurology. 1997;48(3):626–632. doi: 10.1212/wnl.48.3.626. [DOI] [PubMed] [Google Scholar]
- 27.Cornelius C, Fastbom J, Winblad B, Viitanen M. Aspirin, NSAIDs, risk of dementia, and influence of the apolipoprotein E epsilon 4 allele in an elderly population. Neuroepidemiology. 2004;23(3):135–143. doi: 10.1159/000075957. [DOI] [PubMed] [Google Scholar]
- 28.Szekely CA, Breitner JC, Fitzpatrick AL, et al. NSAID use and dementia risk in the Cardiovascular Health study: role of APOE and NSAID type. Neurology. 2008;70(1):17–24. doi: 10.1212/01.wnl.0000284596.95156.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Breitner JC, Haneuse SJ, Walker R, et al. Risk of dementia and AD with prior exposure to NSAIDs in an elderly community-based cohort. Neurology. 2009;72(22):1899–1905. doi: 10.1212/WNL.0b013e3181a18691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Arvanitakis Z, Grodstein F, Bienias JL, et al. Relation of NSAIDs to incident AD, change in cognitive function, and AD pathology. Neurology. 2008;70(23):2219–2225. doi: 10.1212/01.wnl.0000313813.48505.86. [DOI] [PubMed] [Google Scholar]
- 31.Reines SA, Block GA, Morris JC, et al. Rofecoxib: no effect on Alzheimer’s disease in a 1-year, randomized, blinded, controlled study. Neurology. 2004;62(1):66–71. doi: 10.1212/wnl.62.1.66. [DOI] [PubMed] [Google Scholar]
- 32.Rogers J, Kirby LC, Hempelman SR, et al. Clinical trial of indomethacin in Alzheimer’s disease. Neurology. 1993;43(8):1609–1611. doi: 10.1212/wnl.43.8.1609. [DOI] [PubMed] [Google Scholar]
- 33.Scharf S, Mander A, Ugoni A, Vajda F, Christophidis N. A double-blind, placebo-controlled trial of diclofenac/misoprostol in Alzheimer’s disease. Neurology. 1999;53(1):197–201. doi: 10.1212/wnl.53.1.197. [DOI] [PubMed] [Google Scholar]
- 34.Aisen PS, Schmeidler J, Pasinetti GM. Randomized pilot study of nimesulide treatment in Alzheimer’s disease. Neurology. 2002;58(7):1050–1054. doi: 10.1212/wnl.58.7.1050. [DOI] [PubMed] [Google Scholar]
- 35.Aisen PS, Schafer KA, Grundman M, et al. Effects of rofecoxib or naproxen vs placebo on Alzheimer disease progression: a randomized controlled trial. JAMA. 2003;289(21):2819–2826. doi: 10.1001/jama.289.21.2819. [DOI] [PubMed] [Google Scholar]
- 36.Thal LJ, Ferris SH, Kirby L, et al. A randomized, double-blind, study of rofecoxib in patients with mild cognitive impairment. Neuropsychopharmacology. 2005;30(6):1204–1215. doi: 10.1038/sj.npp.1300690. [DOI] [PubMed] [Google Scholar]
- 37.ADAPT Research Group. Lyketsos CG, Breitner JC, et al. Naproxen and celecoxib do not prevent AD in early results from a randomized controlled trial. Neurology. 2007;68(21):1800–1808. doi: 10.1212/01.wnl.0000260269.93245.d2. [DOI] [PubMed] [Google Scholar]
- 38.Franceschi C, Capri M, Monti D, et al. Inflammaging and anti-inflammaging: a systemic perspective on aging and longevity emerged from studies in humans. Mech Ageing Dev. 2007;128(1):92–105. doi: 10.1016/j.mad.2006.11.016. [DOI] [PubMed] [Google Scholar]
- 39.Giunta B, Fernandez F, Nikolic WV, et al. Inflammaging as a prodrome to Alzheimer’s disease. J Neuroinflammation. 2008;5:51. doi: 10.1186/1742-2094-5-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Town T, Tan J, Flavell RA, Mullan M. T-cells in Alzheimer’s disease. Neuromolecular Med. 2005;7(3):255–264. doi: 10.1385/NMM:7:3:255. [DOI] [PubMed] [Google Scholar]
- 41.Flurkey K, Stadecker M, Miller RA. Memory T lymphocyte hyporesponsiveness to non-cognate stimuli: a key factor in age-related immunodeficiency. Eur J Immunol. 1992;22(4):931–935. doi: 10.1002/eji.1830220408. [DOI] [PubMed] [Google Scholar]
- 42.Monsonego A, Zota V, Karni A, et al. Increased T cell reactivity to amyloid β protein in older humans and patients with Alzheimer disease. J Clin Invest. 2003;112(3):415–422. doi: 10.1172/JCI18104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Dutton RW, Bradley LM, Swain SL. T cell memory. Annu Rev Immunol. 1998;16:201–223. doi: 10.1146/annurev.immunol.16.1.201. [DOI] [PubMed] [Google Scholar]
- 44.Tan J, Town T, Abdullah L, et al. CD45 isoform alteration in CD4 + T cells as a potential diagnostic marker of Alzheimer’s disease. J Neuroimmunol. 2002;132(1–2):164–172. doi: 10.1016/s0165-5728(02)00309-0. [DOI] [PubMed] [Google Scholar]
- 45.Burke JR, Roses AD. Genetics of Alzheimer’s disease. Int J Neurol. 1991;25–26:41–51. [PubMed] [Google Scholar]
- 46.Mayeux R, Stern Y, Ottman R, et al. The apolipoprotein epsilon 4 allele in patients with Alzheimer’s disease. Ann Neurol. 1993;34(5):752–754. doi: 10.1002/ana.410340527. [DOI] [PubMed] [Google Scholar]
- 47.Roses AD. Apolipoprotein E genotyping in the differential diagnosis, not prediction, of Alzheimer’s disease. Ann Neurol. 1995;38(1):6–14. doi: 10.1002/ana.410380105. [DOI] [PubMed] [Google Scholar]
- 48.Roses AD. Genetic testing and Alzheimer disease: the promise. Alzheimer Dis Assoc Disord. 1998;12(Suppl 3):S3–S9. [PubMed] [Google Scholar]
- 49.Larbi A, Pawelec G, Witkowski JM, et al. Dramatic shifts in circulating CD4 but not CD8 T cell subsets in mild Alzheimer’s disease. J Alzheimers Dis. 2009;17(1):91–103. doi: 10.3233/JAD-2009-1015. [DOI] [PubMed] [Google Scholar]
- 50.Jago CB, Yates J, Camara NO, Lechler RI, Lombardi G. Differential expression of CTLA-4 among T cell subsets. Clin Exp Immunol. 2004;136(3):463–471. doi: 10.1111/j.1365-2249.2004.02478.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Itagaki S, McGeer PL, Akiyama H. Presence of T-cytotoxic suppressor and leucocyte common antigen positive cells in Alzheimer’s disease brain tissue. Neurosci Lett. 1988;91(3):259–264. doi: 10.1016/0304-3940(88)90690-8. [DOI] [PubMed] [Google Scholar]
- 52.Rogers J, Luber-Narod J, Styren SD, Civin WH. Expression of immune system-associated antigens by cells of the human central nervous system: relationship to the pathology of Alzheimer’s disease. Neurobiol Aging. 1988;9(4):339–349. doi: 10.1016/s0197-4580(88)80079-4. [DOI] [PubMed] [Google Scholar]
- 53.Togo T, Akiyama H, Iseki E, et al. Occurrence of T cells in the brain of Alzheimer’s disease and other neurological diseases. J Neuroimmunol. 2002;124(1–2):83–92. doi: 10.1016/s0165-5728(01)00496-9. [DOI] [PubMed] [Google Scholar]
- 54.Pirttila T, Mattinen S, Frey H. The decrease of CD8-positive lymphocytes in Alzheimer’s disease. J Neurol Sci. 1992;107(2):160–165. doi: 10.1016/0022-510x(92)90284-r. [DOI] [PubMed] [Google Scholar]
- 55.Skias D, Bania M, Reder AT, Luchins D, Antel JP. Senile dementia of Alzheimer’s type (SDAT): reduced T8+-cell-mediated suppressor activity. Neurology. 1985;35(11):1635–1638. doi: 10.1212/wnl.35.11.1635. [DOI] [PubMed] [Google Scholar]
- 56.Shalit F, Sredni B, Brodie C, Kott E, Huberman M. T lymphocyte subpopulations and activation markers correlate with severity of Alzheimer’s disease. Clin Immunol Immunopathol. 1995;75(3):246–250. doi: 10.1006/clin.1995.1078. [DOI] [PubMed] [Google Scholar]
- 57.Bonotis K, Krikki E, Holeva V, Aggouridaki C, Costa V, Baloyannis S. Systemic immune aberrations in Alzheimer’s disease patients. J Neuroimmunol. 2008;193(1–2):183–187. doi: 10.1016/j.jneuroim.2007.10.020. [DOI] [PubMed] [Google Scholar]
- 58.Leffell MS, Lumsden L, Steiger WA. An analysis of T lymphocyte subpopulations in patients with Alzheimer’s disease. J Am Geriatr Soc. 1985;33(1):4–8. doi: 10.1111/j.1532-5415.1985.tb02851.x. [DOI] [PubMed] [Google Scholar]
- 59.Singh VK. Neuroimmune axis as a basis of therapy in Alzheimer’s disease. Prog Drug Res. 1990;34:383–393. doi: 10.1007/978-3-0348-7128-0_12. [DOI] [PubMed] [Google Scholar]
- 60.Ikeda T, Yamamoto K, Takahashi K, Yamada M. Immune system-associated antigens on the surface of peripheral blood lymphocytes in patients with Alzheimer’s disease. Acta Psychiatr Scand. 1991;83(6):444–448. doi: 10.1111/j.1600-0447.1991.tb05573.x. [DOI] [PubMed] [Google Scholar]
- 61.Richartz-Salzburger E, Batra A, Stransky E, et al. Altered lymphocyte distribution in Alzheimer’s disease. J Psychiatr Res. 2007;41(1–2):174–178. doi: 10.1016/j.jpsychires.2006.01.010. [DOI] [PubMed] [Google Scholar]
- 62.Hu GR, Walls RS, Creasey H, McCusker E, Broe GA. Peripheral blood lymphocyte subset distribution and function in patients with Alzheimer’s disease and other dementias. Aust NZ J Med. 1995;25(3):212–217. doi: 10.1111/j.1445-5994.1995.tb01525.x. [DOI] [PubMed] [Google Scholar]
- 63.Chen K, Huang J, Gong W, Zhang L, Yu P, Wang JM. CD40/CD40L dyad in the inflammatory and immune responses in the central nervous system. Cell Mol Immunol. 2006;3(3):163–169. [PubMed] [Google Scholar]
- 64.Abdel-Haq N, Hao HN, Lyman WD. Cytokine regulation of CD40 expression in fetal human astrocyte cultures. J Neuroimmunol. 1999;101(1):7–14. doi: 10.1016/s0165-5728(99)00124-1. [DOI] [PubMed] [Google Scholar]
- 65.Tan J, Town T, Mori T, et al. CD40 is expressed and functional on neuronal cells. EMBO J. 2002;21(4):643–652. doi: 10.1093/emboj/21.4.643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Togo T, Akiyama H, Kondo H, et al. Expression of CD40 in the brain of Alzheimer’s disease and other neurological diseases. Brain Res. 2000;885(1):117–121. doi: 10.1016/s0006-8993(00)02984-x. [DOI] [PubMed] [Google Scholar]
- 67.Townsend KP, Town T, Mori T, et al. CD40 signaling regulates innate and adaptive activation of microglia in response to amyloid β–πεπτιδε. Eur J Immunol. 2005;35(3):901–910. doi: 10.1002/eji.200425585. [DOI] [PubMed] [Google Scholar]
- 68.Tan J, Town T, Suo Z, et al. Induction of CD40 on human endothelial cells by Alzheimer’s β– αμΨλοιδ peptides. Brain Res Bull. 1999;50(2):143–148. doi: 10.1016/s0361-9230(99)00122-7. [DOI] [PubMed] [Google Scholar]
- 69.Tan J, Town T, Paris D, et al. Activation of microglial cells by the CD40 pathway: relevance to multiple sclerosis. J Neuroimmunol. 1999;97(1–2):77–85. doi: 10.1016/s0165-5728(99)00053-3. [DOI] [PubMed] [Google Scholar]
- 70.Aloisi F, Ria F, Penna G, Adorini L. Microglia are more efficient than astrocytes in antigen processing and in Th1 but not Th2 cell activation. J Immunol. 1998;160(10):4671–4680. [PubMed] [Google Scholar]
- 71.Calingasan NY, Erdely HA, Altar CA. Identification of CD40 ligand in Alzheimer’s disease and in animal models of Alzheimer’s disease and brain injury. Neurobiol Aging. 2002;23(1):31–39. doi: 10.1016/s0197-4580(01)00246-9. [DOI] [PubMed] [Google Scholar]
- 72.van Kooten C, Banchereau J. CD40–CD40 ligand. J Leukoc Biol. 2000;67(1):2–17. doi: 10.1002/jlb.67.1.2. [DOI] [PubMed] [Google Scholar]
- 73.Grewal IS, Flavell RA. CD40 and CD154 in cell-mediated immunity. Annu Rev Immunol. 1998;16:111–135. doi: 10.1146/annurev.immunol.16.1.111. [DOI] [PubMed] [Google Scholar]
- 74.Grewal IS, Foellmer HG, Grewal KD, et al. Requirement for CD40 ligand in costimulation induction, T cell activation, and experimental allergic encephalomyelitis. Science. 1996;273(5283):1864–1867. doi: 10.1126/science.273.5283.1864. [DOI] [PubMed] [Google Scholar]
- 75.Obradovic SD, Antovic JP, Antonijevic NM, et al. Elevations in soluble CD40 ligand in patients with high platelet aggregability undergoing percutaneous coronary intervention. Blood Coagul Fibrinolysis. 2009;20(4):283–289. doi: 10.1097/MBC.0b013e328329f28c. [DOI] [PubMed] [Google Scholar]
- 76.Lessiani G, Dragani A, Falco A, Fioritoni F, Santilli F, Davi G. Soluble CD40 ligand and endothelial dysfunction in aspirin-treated polycythaemia vera patients. Br J Haematol. 2009;145(4):538–540. doi: 10.1111/j.1365-2141.2009.07636.x. [DOI] [PubMed] [Google Scholar]
- 77.Mocali A, Cedrola S, Della Malva N, et al. Increased plasma levels of soluble CD40, together with the decrease of TGFβ1, as possible differential markers of Alzheimer disease. Exp Gerontol. 2004;39(10):1555–1561. doi: 10.1016/j.exger.2004.07.007. [DOI] [PubMed] [Google Scholar]
- 78.Holcomb L, Gordon MN, McGowan E, et al. Accelerated alzheimer-type phenotype in transgenic mice carrying both mutant amyloid precursor protein and presenilin 1 transgenes. Nat Med. 1998;4(1):97–100. doi: 10.1038/nm0198-097. [DOI] [PubMed] [Google Scholar]
- 79.Todd Roach J, Volmar CH, Dwivedi S, et al. Behavioral effects of CD40–CD40L pathway disruption in aged PSAPP mice. Brain Res. 2004;1015(1–2):161–168. doi: 10.1016/j.brainres.2004.05.004. [DOI] [PubMed] [Google Scholar]
- 80.Desideri G, Cipollone F, Necozione S, et al. Enhanced soluble CD40 ligand and Alzheimer’s disease: evidence of a possible pathogenetic role. Neurobiol Aging. 2008;29(3):348–356. doi: 10.1016/j.neurobiolaging.2006.10.019. [DOI] [PubMed] [Google Scholar]
- 81.Giunta B, Figueroa KP, Town T, Tan J. Soluble CD40 ligand in dementia. Drugs Future. 2009;34(4):333–339. doi: 10.1358/dof.2009.034.04.1358595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Harman D. Aging: a theory based on free radical and radiation chemistry. J Gerontol. 1956;11(3):298–300. doi: 10.1093/geronj/11.3.298. [DOI] [PubMed] [Google Scholar]
- 83.Harman D. Origin and evolution of the free radical theory of aging: a brief personal history, 1954–2009. Biogerontology. 2009 doi: 10.1007/s10522-009-9253-z. [DOI] [PubMed] [Google Scholar]
- 84.Smith MA, Kutty RK, Richey PL, et al. Heme oxygenase-1 is associated with the neurofibrillary pathology of Alzheimer’s disease. Am J Pathol. 1994;145(1):42–47. [PMC free article] [PubMed] [Google Scholar]
- 85.Smith MA, Tabaton M, Perry G. Early contribution of oxidative glycation in Alzheimer disease. Neurosci Lett. 1996;217(2–3):210–211. [PubMed] [Google Scholar]
- 86.Eckert A, Cotman CW, Zerfass R, Hennerici M, Muller WE. Lymphocytes as cell model to study apoptosis in Alzheimer’s disease: vulnerability to programmed cell death appears to be altered. J Neural Transm Suppl. 1998;54:259–267. doi: 10.1007/978-3-7091-7508-8_25. [DOI] [PubMed] [Google Scholar]
- 87.Schindowski K, Leutner S, Muller WE, Eckert A. Age-related changes of apoptotic cell death in human lymphocytes. Neurobiol Aging. 2000;21(5):661–670. doi: 10.1016/s0197-4580(00)00171-8. [DOI] [PubMed] [Google Scholar]
- 88.Eckert A, Schindowski K, Leutner S, et al. Alzheimer’s disease-like alterations in peripheral cells from presenilin-1 transgenic mice. Neurobiol Dis. 2001;8(2):331–342. doi: 10.1006/nbdi.2000.0378. [DOI] [PubMed] [Google Scholar]
- 89.Sulger J, Dumais-Huber C, Zerfass R, Henn FA, Aldenhoff JB. The calcium response of human T lymphocytes is decreased in aging but increased in Alzheimer’s dementia. Biol Psychiatry. 1999;45(6):737–742. doi: 10.1016/s0006-3223(98)00218-2. [DOI] [PubMed] [Google Scholar]
- 90.Richartz E, Noda S, Schott K, Gunthner A, Lewczuk P, Bartels M. Increased serum levels of CD95 in Alzheimer’s disease. Dement Geriatr Cogn Disord. 2002;13(3):178–182. doi: 10.1159/000048650. [DOI] [PubMed] [Google Scholar]
- 91.Lombardi VR, Garcia M, Rey L, Cacabelos R. Characterization of cytokine production, screening of lymphocyte subset patterns and in vitro apoptosis in healthy and Alzheimer’s disease (AD) individuals. J Neuroimmunol. 1999;97(1–2):163–171. doi: 10.1016/s0165-5728(99)00046-6. [DOI] [PubMed] [Google Scholar]
- 92.Tavolato B, Argentiero V. Immunological indices in presenile Alzheimer’s disease. J Neurol Sci. 1980;46(3):325–331. doi: 10.1016/0022-510x(80)90057-x. [DOI] [PubMed] [Google Scholar]
- 93.Tollefson GD, Godes M, Warren JB, Haus E, Luxenberg M, Garvey M. Lymphopenia in primary degenerative dementia. J Psychiatr Res. 1989;23(3–4):191–199. doi: 10.1016/0022-3956(89)90024-1. [DOI] [PubMed] [Google Scholar]
- 94.Dysken MW, Minichiello MD, Hill JL, et al. Distribution of peripheral lymphocytes in Alzheimer patients and controls. J Psychiatr Res. 1992;26(3):213–218. doi: 10.1016/0022-3956(92)90024-i. [DOI] [PubMed] [Google Scholar]
- 95.Magaki S, Yellon SM, Mueller C, Kirsch WM. Immunophenotypes in the circulation of patients with mild cognitive impairment. J Psychiatr Res. 2008;42(3):240–246. doi: 10.1016/j.jpsychires.2007.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Bullido MJ, Munoz-Fernandez MA, Recuero M, Fresno M, Valdivieso F. Alzheimer’s amyloid precursor protein is expressed on the surface of hematopoietic cells upon activation. Biochim Biophys Acta. 1996;1313(1):54–62. doi: 10.1016/0167-4889(96)00015-8. [DOI] [PubMed] [Google Scholar]
- 97.Pallister C, Jung SS, Shaw I, Nalbantoglu J, Gauthier S, Cashman NR. Lymphocyte content of amyloid precursor protein is increased in Down’s syndrome and aging. Neurobiol Aging. 1997;18(1):97–103. doi: 10.1016/s0197-4580(96)00207-2. [DOI] [PubMed] [Google Scholar]
- 98.Jung SS, Gauthier S, Cashman NR. β– αμΨλοιδ precursor protein is detectable on monocytes and is increased in Alzheimer’s disease. Neurobiol Aging. 1999;20(3):249–257. doi: 10.1016/s0197-4580(99)00051-2. [DOI] [PubMed] [Google Scholar]
- 99.Stieler JT, Lederer C, Bruckner MK, et al. Impairment of mitogenic activation of peripheral blood lymphocytes in Alzheimer’s disease. Neuroreport. 2001;12(18):3969–3972. doi: 10.1097/00001756-200112210-00023. [DOI] [PubMed] [Google Scholar]
- 100.Lopez OL, Rabin BS, Huff FJ. Serum auto-antibodies in Alzheimer’s disease. Acta Neurol Scand. 1991;84(5):441–444. doi: 10.1111/j.1600-0404.1991.tb04985.x. [DOI] [PubMed] [Google Scholar]
- 101.Du Y, Dodel R, Hampel H, et al. Reduced levels of amyloid β– πεπτιδ ε antibody in Alzheimer disease. Neurology. 2001;57(5):801–805. doi: 10.1212/wnl.57.5.801. [DOI] [PubMed] [Google Scholar]
- 102.Hyman BT, Smith C, Buldyrev I, et al. Autoantibodies to amyloid-b and Alzheimer’s disease. Ann Neurol. 2001;49(6):808–810. doi: 10.1002/ana.1061. [DOI] [PubMed] [Google Scholar]
- 103.Weksler ME, Relkin N, Turkenich R, LaRusse S, Zhou L, Szabo P. Patients with Alzheimer disease have lower levels of serum anti-amyloid peptide antibodies than healthy elderly individuals. Exp Gerontol. 2002;37(7):943–948. doi: 10.1016/s0531-5565(02)00029-3. [DOI] [PubMed] [Google Scholar]
- 104.Brettschneider S, Morgenthaler NG, Teipel SJ, et al. Decreased serum amyloid β(1–42) autoantibody levels in Alzheimer’s disease, determined by a newly developed immuno-precipitation assay with radiolabeled amyloid β (1–42) peptide. Biol Psychiatry. 2005;57(7):813–816. doi: 10.1016/j.biopsych.2004.12.008. [DOI] [PubMed] [Google Scholar]
- 105.Baril L, Nicolas L, Croisile B, et al. Immune response to Ab-peptides in peripheral blood from patients with Alzheimer’s disease and control subjects. Neurosci Lett. 2004;355(3):226–230. doi: 10.1016/j.neulet.2003.10.071. [DOI] [PubMed] [Google Scholar]
- 106.Nath A, Hall E, Tuzova M, et al. Autoantibodies to amyloid β– πεπτιδ ε (Ab) are increased in Alzheimer’s disease patients and Ab antibodies can enhance Ab neurotoxicity: implications for disease pathogenesis and vaccine development. Neuromolecular Med. 2003;3(1):29–39. doi: 10.1385/nmm:3:1:29. [DOI] [PubMed] [Google Scholar]
- 107.Gruden MA, Davudova TB, Malisauskas M, et al. Autoimmune responses to amyloid structures of Ab(25–35) peptide and human lysozyme in the serum of patients with progressive Alzheimer’s disease. Dement Geriatr Cogn Disord. 2004;18(2):165–171. doi: 10.1159/000079197. [DOI] [PubMed] [Google Scholar]
- 108.Mruthinti S, Buccafusco JJ, Hill WD, et al. Autoimmunity in Alzheimer’s disease: increased levels of circulating IgGs binding Ab and RAGE peptides. Neurobiol Aging. 2004;25(8):1023–1032. doi: 10.1016/j.neurobiolaging.2003.11.001. [DOI] [PubMed] [Google Scholar]
- 109.Wilson JS, Mruthinti S, Buccafusco JJ, et al. Anti-RAGE and Ab immunoglobulin levels are related to dementia level and cognitive performance. J Gerontol A Biol Sci Med Sci. 2009;64(2):264–271. doi: 10.1093/gerona/gln002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Myagkova MA, Gavrilova SI, Lermontova NN, et al. Content of autoantibodies to bradykinin and β– αμΨλοιδ (1–42) as a criterion for biochemical differences between Alzheimer’s dementias. Bull Exp Biol Med. 2003;136(1):49–52. doi: 10.1023/a:1026036829237. [DOI] [PubMed] [Google Scholar]
- 111.Britschgi M, Olin CE, Johns HT, et al. Neuroprotective natural antibodies to assemblies of amyloidogenic peptides decrease with normal aging and advancing Alzheimer’s disease. Proc Natl Acad Sci USA. 2009;106(29):12145–12150. doi: 10.1073/pnas.0904866106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Moir RD, Tseitlin KA, Soscia S, Hyman BT, Irizarry MC, Tanzi RE. Autoantibodies to redox-modified oligomeric Ab are attenuated in the plasma of Alzheimer’s disease patients. J Biol Chem. 2005;280(17):17458–17463. doi: 10.1074/jbc.M414176200. [DOI] [PubMed] [Google Scholar]
- 113.Sun JC, Lanier LL. Natural killer cells remember: an evolutionary bridge between innate and adaptive immunity? Eur J Immunol. 2009;39(8):2059–2064. doi: 10.1002/eji.200939435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Araga S, Kagimoto H, Funamoto K, Takahashi K. Reduced natural killer cell activity in patients with dementia of the Alzheimer type. Acta Neurol Scand. 1991;84(3):259–263. doi: 10.1111/j.1600-0404.1991.tb04948.x. [DOI] [PubMed] [Google Scholar]
- 115.Solerte SB, Fioravanti M, Severgnini S, et al. Enhanced cytotoxic response of natural killer cells to interleukin-2 in Alzheimer’s disease. Dementia. 1996;7(6):343–348. doi: 10.1159/000106901. [DOI] [PubMed] [Google Scholar]
- 116.Masera RG, Prolo P, Sartori ML, et al. Mental deterioration correlates with response of natural killer (NK) cell activity to physiological modifiers in patients with short history of Alzheimer’s disease. Psychoneuroendocrinology. 2002;27(4):447–461. doi: 10.1016/s0306-4530(01)00062-2. [DOI] [PubMed] [Google Scholar]
- 117.Prolo P, Chiappelli F, Angeli A, et al. Physiologic modulation of natural killer cell activity as an index of Alzheimer’s disease progression. Bioinformation. 2007;1(9):363–366. doi: 10.6026/97320630001363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Leblhuber F, Walli J, Demel U, Tilz GP, Widner B, Fuchs D. Increased serum neopterin concentrations in patients with Alzheimer’s disease. Clin Chem Lab Med. 1999;37(4):429–431. doi: 10.1515/CCLM.1999.070. [DOI] [PubMed] [Google Scholar]
- 119.Murr C, Fuith LC, Widner B, Wirleitner B, Baier-Bitterlich G, Fuchs D. Increased neopterin concentrations in patients with cancer: indicator of oxidative stress? Anticancer Res. 1999;19(3A):1721–1728. [PubMed] [Google Scholar]
- 120.Hull M, Pasinetti GM, Aisen PS. Elevated plasma neopterin levels in Alzheimer disease. Alzheimer Dis Assoc Disord. 2000;14(4):228–230. doi: 10.1097/00002093-200010000-00007. [DOI] [PubMed] [Google Scholar]
- 121.Zhang L, Fiala M, Cashman J, et al. Curcuminoids enhance amyloid-b uptake by macrophages of Alzheimer’s disease patients. J Alzheimers Dis. 2006;10(1):1–7. doi: 10.3233/jad-2006-10101. [DOI] [PubMed] [Google Scholar]
- 122.Town T, Laouar Y, Pittenger C, et al. Blocking TGF-b-Smad2/3 innate immune signaling mitigates Alzheimer-like pathology. Nat Med. 2008;14(6):681–687. doi: 10.1038/nm1781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Town T. Alternative Ab immunotherapy approaches for Alzheimer’s disease. CNS Neurol Disord Drug Targets. 2009;8(2):114–127. doi: 10.2174/187152709787847306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Rezai-Zadeh K, Gate D, Town T. CNS infiltration of peripheral macrophages: D-day for neurodegenerative disease? J Neuroimmune Pharmacol. 2009 doi: 10.1007/s11481-009-9166-2. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Avagyan H, Goldenson B, Tse E, et al. Immune blood biomarkers of Alzheimer disease patients. J Neuroimmunol. 2009;210(1–2):67–72. doi: 10.1016/j.jneuroim.2009.02.015. [DOI] [PubMed] [Google Scholar]
- 126.Fiala M, Liu PT, Espinosa-Jeffrey A, et al. Innate immunity and transcription of MGAT-III and Toll-like receptors in Alzheimer’s disease patients are improved by bisdemethoxycurcumin. Proc Natl Acad Sci USA. 2007;104(31):12849–12854. doi: 10.1073/pnas.0701267104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Kusdra L, Rempel H, Yaffe K, Pulliam L. Elevation of CD69+ monocyte/macrophages in patients with Alzheimer’s disease. Immunobiology. 2000;202(1):26–33. doi: 10.1016/S0171-2985(00)80049-2. [DOI] [PubMed] [Google Scholar]
- 128.Song C, Vandewoude M, Stevens W, et al. Alterations in immune functions during normal aging and Alzheimer’s disease. Psychiatry Res. 1999;85(1):71–80. doi: 10.1016/s0165-1781(98)00130-9. [DOI] [PubMed] [Google Scholar]
- 129.Licastro F, Morini MC, Davis LJ, et al. Increased chemiluminescence response of neutrophils from the peripheral blood of patients with senile dementia of the Alzheimer’s type. J Neuroimmunol. 1994;51(1):21–26. doi: 10.1016/0165-5728(94)90124-4. [DOI] [PubMed] [Google Scholar]
- 130.Scali C, Prosperi C, Bracco L, et al. Neutrophils CD11b and fibroblasts PGE(2) are elevated in Alzheimer’s disease. Neurobiol Aging. 2002;23(4):523–530. doi: 10.1016/s0197-4580(01)00346-3. [DOI] [PubMed] [Google Scholar]
- 131.Koltringer P, Reiseeker F, Eber O, Langsteger W, Lind P. The rheology of erythrocytes and granulocytes in Alzheimer’s disease and multi-infarction-dementia: a prospective study. J Neural Trasm. 1989;1:91. [Google Scholar]
- 132.Biomarkers and surrogate endpoints: preferred definitions and conceptual framework. ClinPharmacol Ther. 2001;69(3):89–95. doi: 10.1067/mcp.2001.113989. [DOI] [PubMed] [Google Scholar]
- 133.Ray S, Britschgi M, Herbert C, et al. Classification and prediction of clinical Alzheimer’s diagnosis based on plasma signaling proteins. Nat Med. 2007;13(11):1359–1362. doi: 10.1038/nm1653. [DOI] [PubMed] [Google Scholar]
- 134.Frank R, Hargreaves R. Clinical biomarkers in drug discovery and development. Nat Rev Drug Discov. 2003;2(7):566–580. doi: 10.1038/nrd1130. [DOI] [PubMed] [Google Scholar]