Abstract
Purpose
Overexpression of the breast cancer oncogene HER2 correlates with poor survival. Current HER2-directed therapies confer limited clinical benefits and most patients experience progressive disease. Because refractory tumors remain strongly HER2+, vaccine approaches targeting HER2 have therapeutic potential, but wild type (wt) HER2 cannot safely be delivered in imunogenic viral vectors because it is a potent oncogene. We designed and tested several HER2 vaccines devoid of oncogenic activity to develop a safe vaccine for clinical use.
Experimental Design
We created recombinant adenoviral vectors expressing the extracellular domain of HER2 (Ad-HER2-ECD), ECD plus the transmembrane domain (Ad-HER2-ECD-TM) and full length HER2 inactivated for kinase function (Ad-HER2-ki) and determined their immunogenicity and anti-tumor effect in wild type (WT) and HER2 tolerant mice. To assess their safety, we compared their effect on the cellular transcriptome, cell proliferation, anchorage-dependent growth, and transformation potential in vivo.
Results
Ad-HER2-ki was the most immunogenic vector in WT animals, retained immunogenicity in HER2-transgenic tolerant animals, and showed strong therapeutic efficacy in treatment models. Despite being highly expressed, HER2-ki protein was not phosphorylated and did not produce an oncogenic gene signature in primary human cells. And, in contrast to HER2-wt, cells overexpressing HER2-ki were less proliferative, displayed less anchorage independent growth and were not transformed in vivo.
Conclusions
Vaccination with mutationally inactivated, non-oncogenic Ad-HER2-ki results in robust polyclonal immune responses to HER2 in tolerant models, which translates into strong and effective anti-tumor responses in vivo. Ad-HER2-ki is thus a safe and promising vaccine for evaluation in clinical trials.
Keywords: HER2, antitumor immunity, adenoviral vaccine, immunization, high-risk breast cancer
Introduction
Human epidermal growth factor receptor 2 (HER2), overexpressed in 25-30% of breast cancers, is a well-known oncogene which is strongly associated with more aggressive tumors and poorer overall patient survival[1]. A major contributor to tumorigenicity, HER2 is the target of multiple clinical therapies, such as the anti-HER2 antibody trastuzumab and HER2 kinase inhibitor lapatinib. While both have demonstrated some measure of efficacy in the clinic, their effects are limited and do not affect HER2 expression in treated patients[2, 3]. Thus, given the limitations of current treatments and their inability to affect HER2 expression, targeting HER2 with T cells and antibodies induced by cancer vaccines represents a promising and potentially effective strategy to treat patients refractory to lapatinib and trastuzumab[4, 5].
Numerous phase I and phase II breast cancer vaccine studies have been conducted6 which have used proteins, peptides, modified tumor cells, and dendritic cells loaded with breast tumor antigens to elicit anti-tumor immune responses to Tumor Associated Antigens (TAAs), such as HER2. While some of these studies have demonstrated beneficial clinical outcomes, by and large they have only achieved modest anti-HER2 immune responses. To elicit maximal therapeutic immunity, a vaccine must break immunologic tolerance to a self-expressed TAA as well as elicit strong polyclonal antibody and T-cell responses to multiple epitopes across the TAA of interest, yet possess a high safety profile. Of the many vaccine platforms utilized, recombinant Adenoviruses (Ad) show great promise of fulfilling these criteria in a cancer vaccine platform. Recombinant Ads are widely used in clinical gene therapy and vaccine applications with an extensive safety profile and well-documented history of eliciting strong transgene-specific adaptive immune responses[6-9]. Thus, to elicit maximal anti-tumor responses, we focused on using an adenoviral (Ad) platform to target the well-validated breast cancer oncogene, HER2.
Although we wished to incorporate the HER2 gene into Ad vectors for therapeutic vaccination against HER2, the use of a wild-type HER2 (HER2-wt) oncogene raised serious safety concerns regarding its tumorigenicity[10, 11]. HER2 is a well-documented member of the ErbB family of tyrosine-kinase receptors and functions in tandem with different binding partners to elicit multiple signaling pathways that enable tumorigenesis[12]. HER2 is also known to bind and elicit signaling with other non-ErbB family members as well as to directly activate transcription by nuclear translocation and binding[13, 14]. To potentially eliminate the oncogenic potential and maximize the immunologic potential encoded by full-length HER2, we constructed and tested the immunogenicity of several vectors potentially ablated for HER2 kinase function. One vector contained a mutation in the HER2 ATP binding site (HER2-ki) while two others were truncated either before or after the transmembrane domain to completely eliminate the HER2 intracellular signaling domain. We hypothesized that these functionally inactivated oncogenes could be used in the adenoviral platform to elicit strong adaptive immune responses that would show efficacy against metastatic HER2+ mammary tumors in both naïve and tolerant preclinical models. We further hypothesized that strong overexpression of the most promising inactivated HER2 gene would be non-oncogenic and thus safe for future clinical use.
Our results revealed that both Ad-HER2-ki and Ad-HER2-ECD-TM were highly effective in eliciting significant T-cell and antibody responses to HER2 in naïve mouse models compared to plasmid vaccination with HER2-ki constructs, thus validating the immunologic efficacy of the adenoviral platform. In contrast, we did not observe any induction of HER2 specific T-cell or antibody responses in HER2-ECD vaccinated animals. While the strong immune responses from both Ad-HER2-ki and Ad-HER2-ECD-TM vectors translated into significantly retarded tumor growth in naïve animals, our studies revealed that Ad-HER2ki vaccinated animals had the most significant HER2-specific T-cell responses as well as the most significant anti-tumor response. When HER2+ tolerant animal models were used, Ad-HER2-ki vaccination elicited only slightly diminished T-cell and antibody responses along with significant anti-tumor responses. Subsequent investigation into the oncogenicity associated with strong overexpression of HER2-ki revealed no evidence for its oncogenic functionality in terms of phosphorylation or transcriptional signature in primary human cells. We also found no evidence for its oncogenicity in enabling enhanced cellular proliferation, anchorage-independent growth, or transformation in vivo. Thus, our results strongly suggest that Ad-HER2-ki is an effective and safe vaccine which could show therapeutic efficacy in future clinical trials.
Materials and Methods
Cell lines
Tumor cell lines MCF-10a and 3T3 were obtained from the American Tissue Culture Collection (Manasas, VA), and maintained according to ATCC recommendations. The human-HER2 and control 4T1 cells were kindly provided by Dr. Michael Kershaw (Cancer Immunology Program, Peter MacCallum Cancer Centre, Victoria, Australia)[15].
Adenoviral vector and cell line construction
The LTR-2/erbB2 plasmid was provided by Dr. L. E. Samelson, (NCI, Bethesda, MD, USA) and HER2-ki (K753A) created by quick-change mutagenesis. Adenoviral vectors were generated using standard cloning techniques as previously described[16, 17]. Ad-Ras (H-Ras G12V) was kindly provided by Dr. Joseph Nevins, Duke University, Durham, NC. HER2+ cell lines were created through retroviral infection and/or plasmid transfection of 3T3 and MCF-10a cell lines and selection with Hygromycin (500ug/ml). Cell expression of HER2 was tested and confirmed in selected cells by FACS analysis using a HER2-PE labeled antibody (BD Biosciences, San Jose, CA, see Supplementary Table 3)
Microarray and quantative rt-PCR Assessments
Cellular RNA was extracted using TRI-Reagent (Molecular Reagents Center, Madison, WI) and further purified by using a RNeasy kit (QIAGEN). RNA quality was assessed using an Agilent Lab-on-a-Chip 2100 Bioanalyzer (Agilent Technologies, Palo Alto, CA) and samples were processed for HG-U133+ v2.0 Affymetrix microarray hybridization according to standard protocols at the Duke Microarray Facility. Datasets were deposited at NCBI's Gene Omnibus Express (GEO) in a MIAME-compliant form (along with complete details of all procedures) as accession number GSE13274. Datasets were analyzed using Genespring 7.2 and the Database for Annotation Visulaization and Integrated Discovery (DAVID) using standard methods[18].
Real-time PCR was performed using an ABI 7300 system using standard methods and intron spanning primers described in Supplementary Table 2. Expression differences were assessed using the comparative cycle threshold (CT) method against GAPDH and Beta-Actin control genes.
Assessment of HER2 mediated phosphorylation, proliferation, and anchorage-independent growth in vitro
Proliferation of stable cells was determined by 3-[4, 5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazoliumbromide (MTT) assay using 5,000 cells per well over the course of 3 days (against control counterparts) in 96-well plates. MTT growth assessments were performed using a Bio-Rad plate reader after cell solubilization in DMSO. Soft Agar Assays for stable and Ad-infected MCF-10A cells were performed as described in O'Hayer and Counter, 2006 with Ad infected MCF-10a cell infected at a MOI of 150[19]. Briefly, 50,000 cells per well were plated in .3% soft agar (on a base of .6% soft agar) and allowed to grow for a period of two weeks in DMEM with 10% FCS (3T3 cells) or DMEM:F12 with 5% Horse Serum and MEGM singlequot growth additives (Clonetics, Walkersville, MD) (MCF-10a cells). At the end of this time, colonies of more than 15 cells were counted and scored. Western blots of infected HMECs were performed using standard procedures with HER2 phosphorylation was being assessed using a HER2-phospho specific antibody[20].
Mouse Experiments
Experiments using BALB/c, NOD CB17-Prkdc SCID/J and SCID-B6.129S7-Rag1(tm1Mom) (obtained from Jackson Labs, Bar Harbor, MA) were performed in accordance with Duke IACUC-approved protocols. Human HER2-transgenic mice (kindly provided by Dr. Wei-Zen Wei, Wayne State University, Detroit, MI[21]) were crossed with BALB/c mice (Jackson Labs, Bar Harbor, ME) to permit implantation of 4T1-HER2 tumors and genotyped by PCR as previously described[15]. For HER2 measurement of 4T1-HER2 tumors, excised tumors were enzymatically digested as previously described[22, 23] and measured using a HER2-PE labeled antibody (BD Biosciences, San Jose, CA). Stable 3T3 cells were injected subcutaneously into the flank of NOD-SCID mice (1×106 or 1×105 cells per animal) as previously described and measured after 28 days. Tumor measurements were made using calipers and volumes calculated using the formula (v=width*width*(length/2)) while statistical differences were calculated using a mixed effects regression model using autoregressive covariance.
Assesment of vector immunogenicity in vivo
Immunogenicity experiments involved footpad injection of Ad-HER2-ki, Ad-HER2-ECD-TM, Ad-HER2-ECD, and Ad-LacZ vectors (2.6×1010 particles/mouse) in transgenic and naïve animals. Plasmid injection was performed by injecting 100ug of plasmid DNA in 50ul of PBS intramuscularly as previously described. Fourteen days post-injection, mice were euthanized and splenocytes and sera were collected for analysis. IFN-γ ELISPOT assays (Mabtech Inc., Cincinnati, OH) were performed according to manufacturer's instructions using overlapping HER2 peptide mixes (2.6 μg/ml: BD Bioscience, San Jose, CA) as stimulating antigens and HIV irrelevant overlapping peptide mixes as negative controls (BD Biosciences, San Jose, CA). PMA (50 ng/ml) and Ionomycin (1 μg/ml) served as a positive control for splenocyte responsiveness. Antibodies were assessed by a flow cytometry adapted methodology reported by Piechocki et al.[24]. Briefly, 3 × 105 cells (mouse 4T1-HER2, HER2+; mouse 4T1, HER2-) were incubated with diluted (1:100, 1:400, 1:1600) mouse serum for 1h at 4°C, washed with 1% BSA-PBS, stained with PE-conjugated anti-Mouse IgG (Dako, Cat # R0480) for 30 min. at 4°C, washed and then samples were analyzed on a BD LSRII or BDFACScalibur flow cytometer and results represented as histograms or mean fluorescence intensity (MFI). Epitope mapping was performed using spotted peptide arrays of 14 mer peptides overlapping by 4 amino acids representing the full length of the human HER2 protein or portions of the full length human HER2 protein on cellulose membranes using a Spot Robot ASP 222 (AbiMed, Langenfeld, Germany) and performed as described[25].
Results
Development of recombinant adenoviral vectors expressing inactivated forms of human HER2
To maximize adaptive immune responses against multiple epitopes of human HER2 while disabling its oncogenic potential, we developed three different adenoviral vectors, one encoding a full-length inactive form of human HER2 and two others encoding truncated forms of human HER2, either before or after the transmembrane region (and thus no intracellular signaling domain) (Fig. 1A). The truncated forms of HER2 were produced by deleting the HER2 intracellular domain coding region after amino acid 684 for Ad-HER2-ECD-TM (nucleotide position 2084) and after amino acid 653 for Ad-HER2-ECD (nucleotide position 1959). To mutationally inactive human HER2, we mutated a key residue in the ATP binding region (K753A) to render the tyrosine kinase inactive (Fig. 1A)[26]. The K753A mutation had been previously shown to reduce HER2 phosphorylation, however, its full impact on HER2 function and oncogenicity was undetermined at this time.
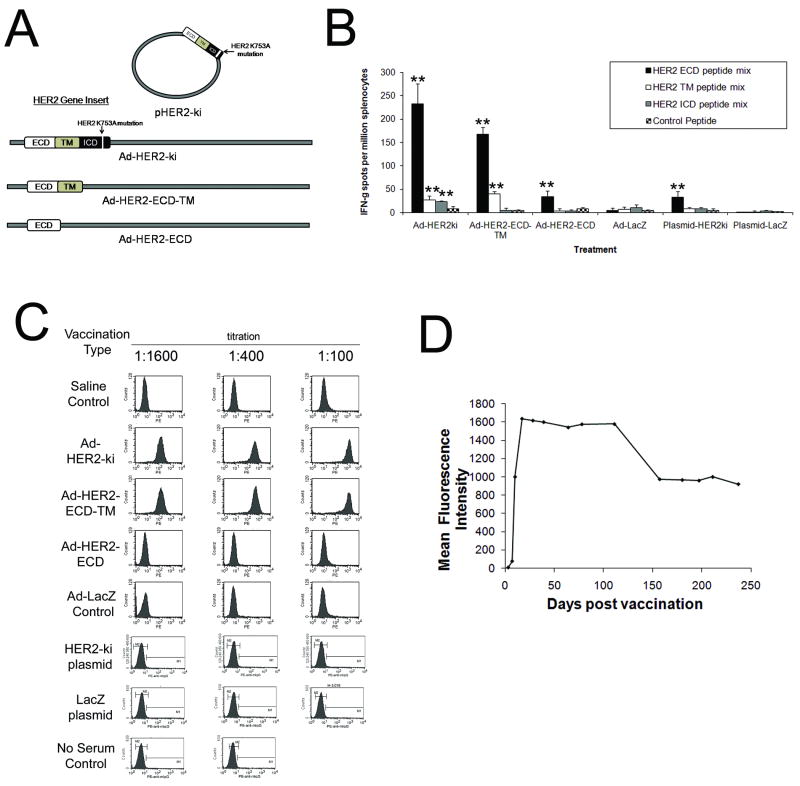
Figure 1. Ad-HER2-ki and Ad-HER2-ECD-TM vaccination elicit robust HER2-specific cellular and polytypic humoral responses in vivo, with longterm maintenance of antibody responses.
A, Diagram of Adenoviral and plasmid based HER2 vectors constructed and compared in this study. All vectors utilized an identical CMV immediate early promoter to drive transgene expression. B, Interferon-gamma ELISPOT responses at 2 weeks post immunization (wpi) in Ad or plasmid vaccinated animals (2.6×1010vp or 100ug of plasmid per mouse, n=5, * indicates p<.05 and ** indicates p<.01 from control peptide stimulation. Error bars represent S.D.) C, FACS analysis using 4T1-HER2 cells to detect HER2-specific IgG antibodies from Ad or plasmid vaccinated animals at 2wpi at different dilutions (n=5 mice, pooled composite results shown). D, Composite Mean Fluorescence Intensity (MFI) FACS assessment of HER2-specific IgG responses after single Ad-HER2-ki vaccination at day 0 (2.6×1010vp per mouse) of C57BL/6 mice (n=3, 1:100 dilution composite results shown).
Immunization with Ad-HER2-ki and Ad-HER2-ECD-TM but not Ad-HER2-ECD elicit strong functional T-cell and antibody responses in naïve mice
As previous studies have reported widely differing efficiencies in the induction of HER2-specific immune responses, we first wanted to determine the strength of T-cell and antibody responses to the modified forms of HER2 using the adenoviral vaccine platform in comparison to the previously reported immunogenicity of HER2-ki plasmid based vaccination (Fig. 1A)[26]. Naïve mice were vaccinated with a single dose of the vaccines and after 2 weeks, T-cell and antibody responses were assessed by ELISPOT assays and FACS assays (to demonstrate antibody binding to HER2+ human breast tumors) respectively. We found that Ad-HER2-ki and Ad-HER2-ECD-TM elicited highly significant responses compared to control vaccinations, but that Ad-HER2-ECD and plasmid-based HER2-ki vaccination elicited far weaker responses (Fig 1B). ELISPOT assays demonstrated that T-cell responses were strongest to epitopes in the Extracellular Domain of HER2 (ECD) and weakest to the Intracellular and Transmembrane domains (ICD and TM) in both Ad-HER2-ki and Ad-HER2-ECD-TM adenoviral vaccinations. As expected, T-cell responses to ICD domain epitopes were only present in the Ad-HER2-ki vaccinated animals and were not observed in animals vaccinated with Ad-HER2-ECD-TM, which was ICD truncated. Differences in T-cell responses between Ad-HER2-ki and Ad-HER2-ECD-TM vaccinated animals were mostly minor, however T-cell responses to the ECD and TM regions were slightly more robust in Ad-HER2-ki vaccinated animals. In contrast, HER2-ki plasmid and Ad-HER2-ECD vaccination showed only weak responses to the ECD domain and ICD/TM were completely absent.
Our assessment of HER2-specific IgG responses mirrored our ELISPOT assessments, with both Ad-HER2-ki and Ad-HER2-ECD-TM vaccination eliciting highly significant levels of HER2-specific antibodies compared to control vector (Fig. 1C). Interestingly, plasmid-based vaccination with HER2-ki and adenoviral vaccination with HER2-ECD were both unable to elicit significant quantities of HER2-specific antibodies compared to control vaccinations.
Since antibody efficacy is a function of its targeted epitope, the antibody isotype, and the duration of the humoral response, we next investigated these aspects of our Ad-HER2-ki and Ad-HER2-ECD-TM vaccine induced antibodies (VIA). Isotype-specific ELISA indicated the presence of multiple antibody isotypes from both vaccinations, with all IgG subtypes predominanting, modest levels of IgM and with low concentrations of IgA and IgE isotypes (data not shown). Epitope mapping revealed that Ad-HER2-ki elicited IgG antibodies that specifically bound multiple epitopes in the intra- and extracellular regions of HER2 (Table 1), which Ad-LacZ did not elicit, thus demonstrating a polyclonal HER2-specific adaptive response against full-length HER2. Lastly, we investigated the longevity of the polyclonal antibody response by immunizing wild type C57BL/6 mice with Ad-HER2-ki and periodically assessing HER2-specific IgG antibody levels at different times post-injection by FACS (Fig. 1D). We found that peak antibody responses represent a titer of 1:25,000 based upon ELISA at day 78 (data not shown) and that while antibody responses gradually diminished, a single immunization resulted in considerable (roughly ∼50%) HER2-specific antibody levels seven months after injection. Thus, vaccination using Ad-HER2-ki is sufficient to elicit robust levels of polyclonal antibodies which are part of a long-lived response that is significant at 7 months post-injection.
Table 1. HER2-VIA antibody epitopes.
| Epitope(s) position | Protein region |
|---|---|
| 29-50 | ECD |
| 365-385 | ECD |
| 405-420 | ECD |
| 577-595 | ECD |
| 605-630 | ECD |
| 817-840 | ICD |
| 909-940 | ICD |
| 941-965 | ICD |
| 989-1040 | ICD |
| 1061-1085 | ICD |
| 1097-1160 | ICD |
| 1169-1190 | ICD |
| 1195-1211 | ICD |
| 1229-1245 | ICD |
In sum, Ad-HER2-ki and Ad-HER2-ECD-TM vectors elicited significantly greater T-cell and antibody responses in comparison to Ad-HER2-ECD and HER2-ki plasmid-based vaccination.
Ad-HER2-ki and Ad-HER2-ECD-TM vaccines are effective against HER2+ tumors in vivo
While both Ad-HER2-ki and Ad-HER2-ECD-TM were able to efficiently induce polyclonal T-cell and humoral immunity, it was unknown if these responses could effectively inhibit aggressive HER2+ tumor growth in vivo. To assess the anti-tumor effect of these vectors, an aggressive herceptin-resistant [15] (Supplementary Figure 1) HER2+ metastatic mouse mammary carcinoma line (4T1-HER2) was implanted in mice, which were subsequently vaccinated with either Ad-HER2-ki, Ad-HER2-ECD-TM, or with a control vector (Ad-LacZ) (Fig. 2A). This approach effectively mimicked conditions seen in patients, as 4T1-HER2 cells were not solely dependent on HER2 for growth, but grew aggressively and could initiate metastases. Furthermore, once the most promising vector was determined in the naïve model, that vector could be tested using the same approach in the more stringent HER2 transgenic mouse model that mimics the human HER2 tolerance seen in patients (Fig. 2A).
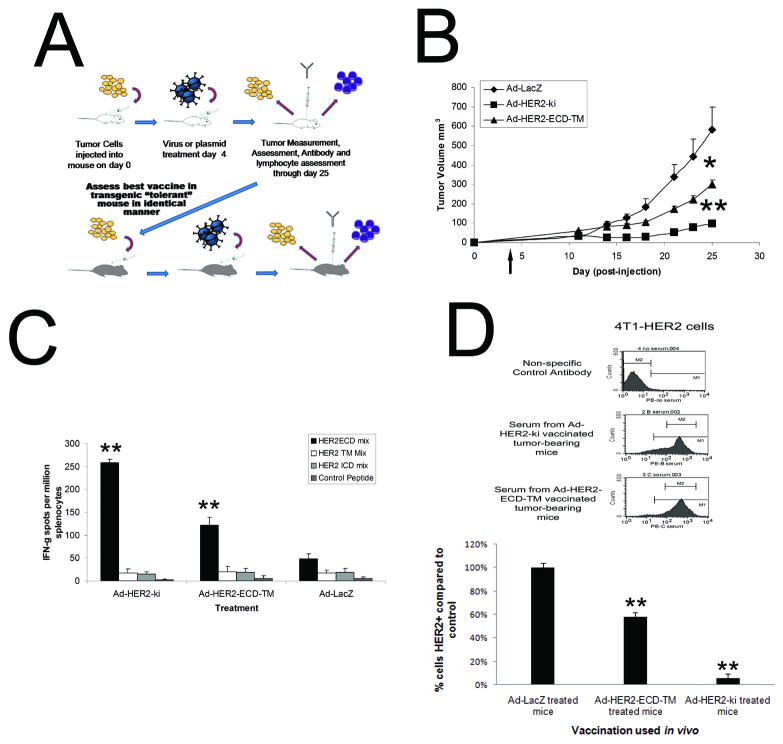
Figure 2. Vaccination using Ad-HER2-ECD-TM and Ad-HER2-ki significantly inhibits HER2+ tumor growth in naïve mice in vivo.
A, Schematic representation of vaccination and antitumor assessment strategy in both naïve and tolerant models. B, Anti-tumor effect of vaccines in naïve model. BALB/c mice were implanted with 2×104 4T1-HER2+ cells on day zero and were vaccinated via footpad with 2.6×1010 particles of Ad-HER2-ki (square symbols), Ad-HER2-ECD-TM (triangles) or control Ad-LacZ (diamonds), on day 4 (denoted by the arrow). (n=8, * indicates p<.05 and ** indicates p<.01, Error bars represent S.D). C, Activation of HER2-specific T-cells correlates with the strength of tumor regression in vaccinated tumor-bearing animals. IFN-gamma ELISPOT assay employed different overlapping pools of HER2-specific epitopes for various HER2 domains at a concentration of 1ug/ml per million splenocytes (n=8 vaccinated animals, * indicates p<.05 and ** indicates p<.01 compared to control, Error bars represent S.D). D, upper panel depicts HER2-specific IgG antibody responses are equivalent between vaccinated tumor-bearing animals and HER2 is selected against in tumors of vaccinated animals. Pooled composite FACS analysis using 4T1-HER2 cells to detect HER2-specific IgG antibodies from Ad vaccinated animals at 25 dp implantation (dilution 1:100, n=8). D, lower panel depicts HER2 expression on tumor cells from treated animals show evidence of anti HER2-specific selection. Explanted 4T1-HER2 cells from vaccinated animals (n=8, cell explanted 25 days post-implantation) were assessed using HER2-specific FACS.
Using the naïve model, we found that mice receiving a single vaccination with either Ad-HER2-ki or Ad-HER2-ECD-TM demonstrated significant retardation in tumor growth compared to the control Ad-LacZ (Fig. 2B). Comparison of the two vectors revealed that Ad-HER2-ki gave a significant protective advantage in blunting tumor growth compared to Ad-HER2-ECD-TM and a more significant advantage when compared to Ad-LacZ control animals. Assessment of T-cell responses and antibody responses in vaccinated animals revealed that while HER2-specific IgG responses were not significantly different but T-cell responses showed significant differences (Fig 2C and 2D). In all tumor bearing animals, HER2 peptide stimulation of splenocytes produced significant responses compared to unstimulated controls, indicating that aggressive HER2+ tumor growth had elicited a baseline level of T-cell activation in naïve animals against HER2 epitopes, which had been unable to curtail tumor growth. The domain-specificity of activated T-cell epitopes mirrored that of vaccinated non-tumor bearing animals (Fig. 1B and 2C), in that ECD responses were dominant in all groups. Significantly though, we observed that the dominant ECD-specific responses of the Ad-HER2-ki and Ad-HER2-ECD-TM were significantly greater than control-vaccinated animals and directly correlated with the strength of the anti-tumor effect observed in vaccinated animals.
While Ad-HER2-ki and Ad-HER2-ECD-TM vaccinated animals had significantly repressed tumor growth, tumors in these animals were not fully eliminated. We hypothesized that if HER2-specific responses were attributable for the diminished growth, then the 4T1 tumor outgrowths in these animals would have a reduced level of HER2 expression as a result of immunoselection induced by vaccination. We further hypothesized that if the HER2-specific cytotoxic T-cells were the major selective pressure on tumors, then the level of HER2 expression in the remaining tumor cells would inversely correlate with the strength of anti-HER2 T-cell responses we observed in vaccinated animals. Tumors from vaccinated animals were excised and dissociated, and HER2-expression determined quantitatively by FACS. Supporting the hypothesis, we found that HER2 expression was significantly diminished in both Ad-HER2-ECD-TM and Ad-HER2ki vaccinated animals (compared to Ad-LacZ control), being lowest in Ad-HER2-ki vaccinated animals (about 5% displaying expression comparable to control vaccinated animals, Fig 2D). Thus, HER2 expression was indeed found to be inversely associated with HER2-specific T-cell responses, strongly suggesting that HER2-specific T-cells were predominantly responsible for the efficacy of Ad-HER2-ki and Ad-HER2-ECD-TM vaccinations.
Ad-HER2-ki vaccine effectiveness against HER2+ tumors in a tolerant in vivo model
As tolerance to self-antigen is a major impediment to the successful clinical use of vaccines, we sought to determine if our most promising vaccine (Ad-HER2-ki) could be as effective in its immune induction and anti-tumor effects in a human HER2+ tolerant mouse[27]. To first determine if immune responses elicited by Ad-HER2-ki vaccination were comparable in a tolerant setting, we vaccinated naïve and human HER2 transgenic animals with Ad-HER2-ki and assessed T-cell and antibody induction using ELISPOT and HER2 specific FACS. Our ELISPOT results revealed that HER2+ transgenic animals produced nearly equivalent HER2-specific T-cell responses compared to naive controls (Fig 3A). Similarly, FACS analysis of mouse serum for HER2-specific antibodies demonstrated similar levels of specific binding to HER2+ cells (but not parental HER2- cells) between vaccinated HER2-transgenic mice, non-HER2-transgenic littermates, and wildtype C57BL/6 mice (Fig. 3B). As expected, serum from Ad-LacZ vaccinated mice did not significantly bind to HER2+ cells, demonstrating that antibody responses were HER2-specific in Ad-HER2ki vaccinated mice.
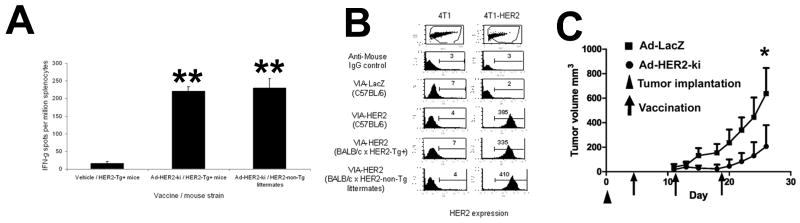
Figure 3. Ad-HER2-ki significantly inhibits HER2+ tumor growth in tolerant HER2+ transgenic mice in vivo.
A, Interferon-gamma ELISPOT responses at 2wpi in Ad-HER2-ki or Ad-LacZ injected human-HER2-transgenic (HER2-Tg+) and non-transgenic (HER2-Tg-) littermates. A pool of epitopes to full-length human HER2 was used to stimulate splenocytes. (n=5, * indicates p<.05 and ** indicates p<.01, Error bars represent S.D) B, FACS analysis using 4T1 or 4T1-HER2 cells to detect HER2-specific IgG antibodies from mice vaccinated with Ad-HER2-ki or Ad-LacZ (2wpi, n=5, * indicates p<.05 and ** indicates p<.01, Error bars represent S.D). C, Vaccination using Ad-HER2-ki inhibits the growth of established 4T1-HER2 tumors in human-HER2-transgenic mice. Human HER2 transgenic mice were implanted with 2×104 4T1-HER2+ cells on day zero (triangle) and were vaccinated with 2.6×10E10 particles of Ad-HER2-ki (circles) or Ad-LacZ (square symbols), on day 4, 11, and 18 (denoted by arrows). (n=5, regression analysis p<0.0001). Error bars represent S.D.
As immune responses in naïve and tolerant mice were nearly equivalent, we next wished to determine if the therapeutic effect elicited by the induction of HER2 immunity after Ad-HER2-ki vaccination was equivalent in tolerant animals. To determine therapeutic effect in tolerant animals, transgenic HER2+ tolerant animals were injected with the same dose of 4T1-HER2+ cells and vaccination with Ad-HER2-ki was given at 4 days post-implantation as previously done in naïve counterparts (Fig 3C). We found that as with immune responses, Ad-HER2-ki vaccination was again able to significantly blunt the growth of 4T1-HER2+ tumors with similar kinetics in tolerant animals. However, it should be noted that it required repeated dosing to achieve these effects in tolerant mice, while a single vaccination was sufficient in WT mice (Fig 2B). Indeed, tumors from Ad-HER2-ki vaccinated HER2+ transgenic mice outgrew slightly quicker after the initial tumor regression compared to naïve Ad-HER2-ki vaccinated mice (Fig 2B and 3C), despite three vaccinations. Thus, our results indicate that the Ad-HER2-ki is a potent anti-tumor vaccine against aggressive non HER2 dependent tumor growth in a tolerant preclinical model.
Oncogenic signaling deficiency after over expression of kinase inactivated HER2
As our preclinical studies of immunogenicity and therapeutic effectiveness had revealed Ad-HER2-ki to be the most promising candidate for a human HER2 vaccine, we next wanted to determine if HER2-ki was truly non-oncogenic. As strong over-expression of HER2 is known to be an early causative event in breast cancer it was of the utmost importance to understand if the single amino acid mutation could fully block phosphorylation and oncogenic signaling in the context of strong adenoviral overexpression.
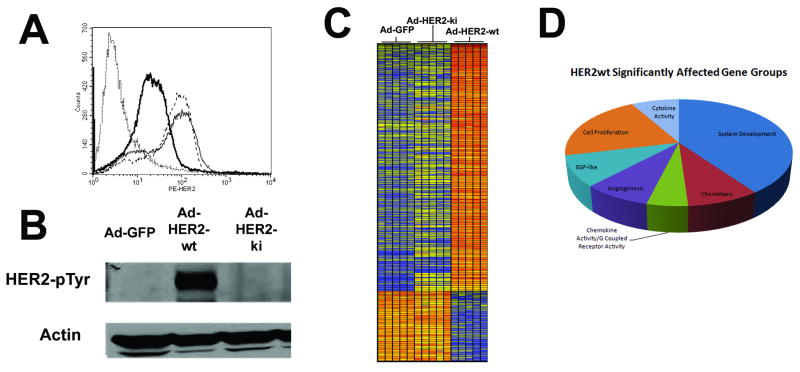
To initially determine if there were differences between HER2-ki (kinase inactive) and HER2-wt (wild-type) expression and phosphorylation, we infected primary human mammary epithelial cells (HMECs) with Ad-HER2-ki or Ad-HER2-wt. While infection with both vectors resulted in similar levels of high surface expression of HER2 as shown by FACS analysis and western blot analysis (Fig. 4A, Supplementary Figure 2), we could detect no evidence of HER2 phosphorylation in Ad-HER2-ki infected HMECs, in contrast to Ad-HER2-wt infected HMECs (Fig. 4B, Supplementary Figure 2). We next examined the signaling consequences of this phosphorylation defect through a comparison of transcriptome profiles from Ad-HER2-wt, Ad-HER2-ki, and Ad-GFP control infected HMECs. Using a 1-way ANOVA (p<.05 with Benjamini-Hochberg False Discovery Rate (BH FDR) Multiple Testing Correction) analysis additionally filtered for genes with a >3 fold difference, we found 423 genes were statistically different between the Ad-HER2-wt and Ad-GFP, while only 21 were different between Ad-HER2-ki and Ad-GFP (Fig. 4C). Subsequent investigation of 9 representative gene targets by quantitative real-time PCR (Supplementary Table 1 and 2) revealed a high degree of array concordance and confirmed our transcriptome findings. A functional analysis of HER2-wt revealed significant over-representation and clustering into seven Gene Ontology (GO) groups: systems development, EGF-like, cytokine activity, chemotaxis, chemokine activity/GPCR activity, proliferation, and angiogenesis (Fig. 4D). These findings suggest that much of the oncogenic potential attributed to HER2 kinase activity could be mediated through inflammatory and chemotaxis pathways (an area currently under investigation in our lab), as well as through more established angiogenic and EGF typical pathways. In sum, immuno-blot and transcriptome assessments indicate the vast majority of cellular pathway activation mediated by HER2-wt is critically dependent upon phosphorylation and kinase functionality, strongly suggesting a critical oncogenic signaling deficit in HER2-ki.
Figure 4. Equivalent expression of HER2-wt and HER2-ki but differential phosphorylation and oncogenic signaling capacity.
HMECs were serum starved for 36 hrs, infected with Ad-HER2-wt, Ad-HER2-ki, or Ad-GFP at a MOI of 150. A, FACS analysis at 18hpi using anti-HER2 PE labeled antibody revealed surface expression of human HER2 infected with the Ad-HER2-wt (thin dotted line), Ad-HER2-ki (thin solid line), or Ad-GFP (thick solid line) vectors. B, Western blots performed on infected HMECs at 18hpi using anti-HER2 pTyr705 and actin antibodies. C, A hierarchically clustered heatmap of significantly affected genes (p<.05 with BH MTC, filtered by >3 fold difference) where each gene probe is represented by a single row and each column denotes an individual infected sample (n=5 for all conditions). The more transcriptionally active a gene is, the greater the intensity of the shade of red, while less active genes are depicted by greater intensity of shades of blue. D, HER2-wt significantly dysregulated genes (p<.05 with Benjamini-Hochberg Multiple Testing Correction) with more than 3 fold difference from control (Ad-GFP) were interrogated for their functional classification using DAVID as described in Materials and Methods. The most significantly affected gene groups are displayed in proportional representation to the number found in those groups to be dysregulated by HER2-wt.
Diminished proliferation and anchorage-independent growth of kinase-inactive HER2
While immunoblots and transcriptome analysis revealed strong HER2-ki signaling deficiencies, it was unknown if these would diminish oncogenic function. To assess HER2 oncogenic function in vitro, we constructed several stable HER2-wt and HER2-ki 3T3 fibroblasts lines that had different levels of HER2 expression (retrovirally selected lines expressed high levels of HER2 and are denoted as ‘High expression’ while plasmid transfection selected lines expressed lower levels of HER2 and are denoted as ‘low expression’, Supplementary Table S3) and tested their proliferative and transformative capacity in vitro. We found that HER2-wt significantly augmented 3T3 proliferation in an expression sensitive manner (Fig 5A). In contrast, we found that strong HER2-ki expression significantly reduced cell proliferation, while weak HER2-ki expression had no effect on proliferation (Fig. 5A). Likewise, high expression stable HER-wt 3T3 formed colonies in soft agar, while high expression stable HER2-ki 3T3 cells formed colonies at a significantly reduced basal rate compared to control cells (Fig. 5A). Thus HER2-wt enhanced cell proliferation and transformation in vitro, while HER2-ki inhibited proliferation and transformation in vitro. Although statistically significant, these colony formation differences were modest and only apparent in highly expressed lines. Thus, to elicit a more sensitive measure of in vitro transformation, EGF (an ErbB family growth factor) was used to enhance HER2 soft agar colony formation[28]. While EGF addition enhanced colony formation of all cell types (Fig. 5B and 5A), only HER2-wt cell displayed significantly enhanced anchorage-independent growth over control cells (Fig. 5B and 5A). Thus, even in the presence of EGF, HER2-ki displays a significant transformation deficiency compared to HER2-wt.
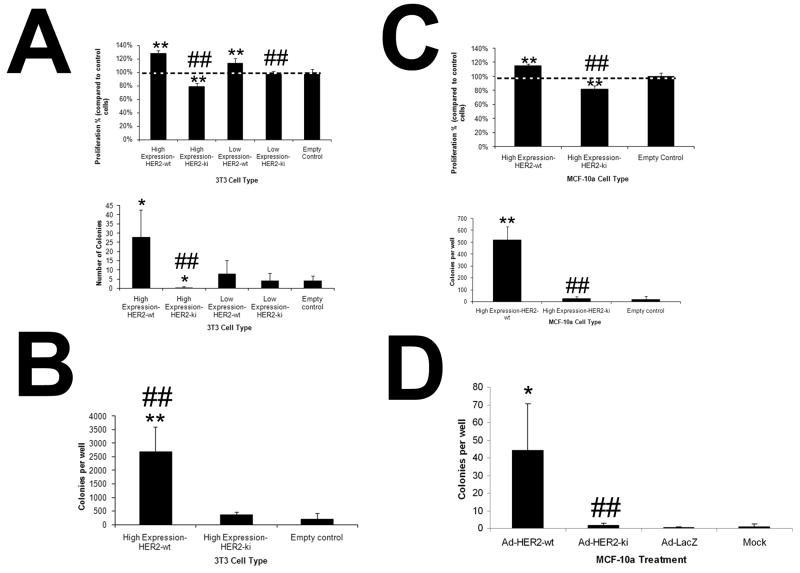
Figure 5. Diminished proliferation and anchorage-dependent growth of HER2-ki expressing 3T3 and MCF-10a cells.
A, Stable 3T3 cells were assessed for proliferation by MTT assay 72 hrs. after plating (n=6) or for anchorage-independent growth by soft agar assay 2 weeks after plating (5×104 cells per well in 2mls, 2 week assessment, n=3). B, Soft agar assay was performed as described in (A) but in the presence of EGF (10ng/ml) with cells cultured for 3 weeks (5×104 cells per well in 2mls, 2 week assessment, n = 3). C, Stable MCF-10a cells were assessed for proliferation by MTT assay 72 hrs. after plating (n=5) or for anchorage-independent growth by soft agar assay 2 weeks after plating (n=3). D, MCF-10a cells were assessed for anchorage independent growth after Ad vector infection (MOI=150, n=3). In all samples, * and ** denotes conditions that showed p<0.05 and p<0.01 respectively, compared to control cells. Additionally, # and ## denote conditions that showed p<0.05 and p<0.01 respectively to HER2-wt stable or infected cells. Error Bars represent SD.
Having determined a proliferative and transformative defect in HER2-ki expressing mouse fibroblasts, we extended our investigation to a more relevant human cell type using engineered immortalized (but non-transformed) human breast epithelial cells (MCF-10a) to express either HER2-wt or HER2-ki. As before, we found that HER2-wt cells grew at a significantly enhanced rate while HER2-ki cells displayed a significant repression of proliferation compared to control cells (Figure 5C). Additionally, we determined that HER2-wt cells formed significantly more colonies in soft agar compared to HER2-ki cells and control cells (Fig. 5C).
Finally, we wished to explore if Ad mediated delivery and oncogene over-expression impacted human cell transformation in vitro. While unlikely, it remained possible that in the context of an inflammatory response and heavy over-expression of HER2-ki, the minor dysregulation observed in our microarray could impact transformative capacity. We infected MCF-10a cells with HER2-wt, HER2-ki, and control GFP Ad vectors and assessed anchorage independent growth in soft agar. While Ad-mediated overexpression of HER2-wt elicited anchorage-independent growth in those cells, Ad-HER2-ki infected cells were unable to form more colonies than Ad-GFP infected cells or uninfected controls (Fig 5D) Thus, Ad-mediated delivery did not significantly alter transformative capacity of HER2-ki.
Collectively these in vitro data show that the signaling defects conferred by the mutation of HER2-ki ablate the enhanced proliferation and anchorage-independent growth elicited by the HER2 oncogene. These defects were observed across different expression contexts and different cell types and thus strongly suggest that HER2-ki is a functionally ablated oncogene.
Lack of tumorigencity in immunodeficient xenografts
Since HER2-ki had demonstrated significant signaling and functional defects in vitro, we hypothesized that it would have a reduced oncogenic transformation potential in vivo. To assess this, stable HER2-ki, HER-wt, and control 3T3 cells were implanted in immunodeficient mice and tumor formation assessed. Data from five independent experiments are summarized in Table 2, which shows that while all mice injected with 3T3 cells expressing the wild type human HER2 gene (HER2-wt) developed tumors, mice injected with 3T3 cells expressing the kinase inactive gene (HER2-ki) did not develop tumors. Even at the highest concentrations of injected cells, the basal rate of spontaneous 3T3 transformation in control 3T3 cells proved higher than HER2-ki expressing cells. Thus, the slightly greater transformation potential observed in control 3T3 cells suggests that HER2-ki expression has a negative effect on 3T3 transformation in vivo. These findings demonstrate that HER2 kinase function and downstream signaling are critical to its transformative capacity in vivo and further demonstrate that the transformative capacity of HER2-ki is ablated and its expression is safe in vivo.
Table 2. Tumorgenicity assays demonstrate lack of oncogenic potential of the HER2-ki gene.
| 3T3 clone | Tumor formation at 28 days (1×106 cells) | Tumor formation at 28 days (1×105 cells) |
|---|---|---|
| 3T3-High Expression/HER2 wild type | 10/10 mice | 50/50 mice |
| 3T3-High Expression/HER2-ki | 0/10 mice | 0/30 mice |
| 3T3-Empty vector | 1/10 mice | 0/30 mice |
| 3T3 cells | 0/10 mice | 0/30 mice |
Discussion
More than 20 years after its discovery, therapeutic strategies to target the human oncogene HER2 are achieving measures of efficacy in the clinic, through use of the HER2-specific monoclonal antibody trastuzamab and the HER2/EGFR selective kinase inhibitor lapatinib. However, many tumors have de novo resistance to these therapies and the majority of tumors that initially respond to them eventually become refractory to treatment. In spite of this, these tumors retain HER2 expression, thus permitting the use of alternative anti-HER2 approaches as potential adjunct therapies. Along these lines, several HER2 vaccines have been developed that utilize protein or peptide fragments of either the intracellular and/or extracellular domain. In our study, we compare the use of several possible inactivated HER2 vaccines and report the strong efficacy of an adenoviral vector that encodes a kinase-inactive full-length HER2 gene in an attempt to maximize immunotherapeutic anti-HER2 efficacy and concomitantly minimize HER2 oncogenic potential.
While the oncogenic potential of HER2 is well-documented, there have been few in-depth studies investigating the oncogenic effect of kinase-inactive HER2. HER2 is well-known to signal in cooperation with the ErbB family of tyrosine kinase receptors, but has also been documented to translocate to the nucleus to activate genes such as Cox-2[13]. Other reports have also documented HER2 to be involved with other signaling receptor families[29], thus broadening the potential mechanisms whereby HER2 promotes oncogenesis. Our study demonstrates that overexpression of a kinase inactive HER2 (HER2-ki) results in a complete block of HER2 phosphorylation and has a dramatic impact on cellular signaling as measured by transcriptome dysregulation, in spite of high levels of HER2 expression. We also found the HER2-ki does not confer any proliferative or transformative potential to cells, in the context of different expression capacities, different cell lines, as well as in in vitro and in vivo settings. While some genes were dysregulated by expression of HER2-ki and reversion mutations could potentially occur with the single HER2-ki mutation, our investigation strongly suggests that HER2-ki is a functionally inactivated oncogene and thus safe for use in humans.
Beyond safety, our study also demonstrates that Ad-HER2-ki is able to induce strong adaptive immune responses against HER2 that translate into effective anti-tumor immunity in vivo. While previous studies have used a variety of different vaccination strategies that target ErbB2/HER2, none have targeted the human full-length HER2 using an adenoviral vector and extended these findings to a HER2 tolerant model. Indeed, studies that have used similar vaccine strategies to target ErbB2/HER2 have all utilized the rat neu gene (HER2 homolog) in mice, which is recognized as a foreign transgene (neu) and thus more immunogenic than a self-antigen[30-33]. A number of studies using the rat neu have also looked at prevention of spontaneous tumors in various rat neu-transgenic mouse models[33-37]. While the data demonstrate that these vaccine strategies can be effective in reducing or preventing the development of mammary tumors, the physiologic relevance of these tumor models has been questioned[38]. Additionally, the neuT model has been shown to be more responsive to DNA vaccination than the human HER2-transgenic model used in the present study, likely because the spontaneous neu tumors appear to be completely dependent upon neu for growth and are more sensitive to anti-neu antibodies[39]. For these reasons, we chose to use the more rigorous tolerance model provided by the human HER2-transgenic mice using 4T1-HER2+ cells, an aggressive and metastatic mouse tumor that is not completely dependent on HER2 for growth. Vaccination with Ad-HER2-ki in this model elicited the generation of strong T-cell and antibody responses comparable to those elicited in naïve mice. Ad-HER2-ki vaccination was able to strongly limit HER2+ cancer growth and elicited strong selection against HER2 expression in residual tumor mass. Since 4T1 cells are not dependent on HER2, it is possible that HER2-dependent models would offer even stronger evidence of Ad-HER2-ki efficacy. Our results indicate that the induction of HER2-specific T-cells highly correlates with anti-tumor efficacy and anti-HER2 tumor selection in multiple vaccines, strongly suggesting that vaccine efficacy is dependent upon the strong induction of HER2-specific T-cells. Of relevance for future clinical studies, we were also able to demonstrate Ad-HER2-ki anti-tumor effects against an aggressive HER2+ breast cancer in a human HER2-tolerant mouse model that most closely resembles the state of immunologic tolerance to human HER2 that exists in cancer patients.
In sum, our study demonstrates the importance of HER2 kinase function in oncogenesis as well as the safety and efficacy of Ad-HER2-ki and Ad-HER2-ECD-TM for therapeutic vaccination against HER2+ breast cancer. Based on these findings, we are actively pursuing the use of Ad-HER2-ki in clinical studies to determine the safety and efficacy of this approach in patients with HER2+ breast cancer. And while adenoviral vaccines in humans with pre-existing anti-Ad5 immunity have lacked efficacy, we recently described a modified Ad5 platform that overcomes this barrier[40]. If successful, our strategy of targeting active oncogenes with inactive oncogene vaccines could become a significant therapeutic option in the treatment of different oncogene-dependent cancers.
Supplementary Material
4T1-HER2 cells (5,000 per well in 96-well plate) were plated with various concentrations of herceptin and assessed for proliferation by MTT assay 24 hrs. after plating (n=5). Error Bars represent SD.
HMECs were serum starved for 36 hrs, infected with Ad-HER2-wt, Ad-HER2-ki, or Ad-GFP at a MOI of 150. Western blots were performed at 18hpi using HER2-specific or Beta-Actin specific antibodies.
Acknowledgments
This study was supported by grants from the National Cancer Institute (NCI P50 CA89496-01 (H.K.L.), NCI R01 CA95447 (T.M.C.)), Department of Defense Breast Cancer Research Program Clinical Translational Research Award (BC050221) (T.M.C.), and a Susan G. Komen Foundation Postdoctoral Fellowship Award (KG080627) (Z.C.H.).
Footnotes
The work was performed at: Duke University Medical Center, Durham, NC 27710.
Conflict of Interest: None of the authors has any conflicts of interest to disclose.
Statement of Translational Relevance: De novo and acquired resistance to existing HER2-targeted therapies means that most patients will ultimately experience progressive disease. Since HER2 continues to be overexpressed on progressing tumors, we reasoned that a cancer vaccine eliciting both cellular and humoral immunity against HER2 might have efficacy in this setting. Additionally, while there have been numerous studies of HER2 peptide and protein vaccines, we sought to improve upon vaccine immunogenicity and epitope targeting by using an adenoviral vector to express a modified (non-oncogenic) full-length human HER2. This study clearly demonstrates the vaccine is non-oncogenic, highly immunogenic, and induces strong anti-tumor activity in human HER2-tolerant mice. This vaccine will shortly be tested in human clinical trials and if successful, the vaccine could complement existing HER2-targeted therapies. This study also demonstrates the principle of targeting tumor oncogenes by adenovirus-mediated expression of modified, non-oncogenic genes,which might similarly be applied to other cancer targets.
Reference List
- 1.Paik S, Hazan R, Fisher ER, et al. Pathologic findings from the National Surgical Adjuvant Breast and Bowel Project: prognostic significance of erbB-2 protein overexpression in primary breast cancer. J Clin Oncol. 1990;8:103–12. doi: 10.1200/JCO.1990.8.1.103. [DOI] [PubMed] [Google Scholar]
- 2.Konecny GE, Pegram MD, Venkatesan N, et al. Activity of the dual kinase inhibitor lapatinib ( GW572016) against HER-2-overexpressing and trastuzumab-treated breast cancer cells. Cancer Res. 2006;66:1630–9. doi: 10.1158/0008-5472.CAN-05-1182. [DOI] [PubMed] [Google Scholar]
- 3.Tuma RS. Lapatinib moves forward in inflammatory and early HER2-positive breast cancer trials. J Natl Cancer Inst. 2007;99:348–9. doi: 10.1093/jnci/djk122. [DOI] [PubMed] [Google Scholar]
- 4.Ritter CA, Perez-Torres M, Rinehart C, et al. Human breast cancer cells selected for resistance to trastuzumab in vivo overexpress epidermal growth factor receptor and ErbB ligands and remain dependent on the ErbB receptor network. Clin Cancer Res. 2007;13:4909–19. doi: 10.1158/1078-0432.CCR-07-0701. [DOI] [PubMed] [Google Scholar]
- 5.Martin AP, Miller A, Emad L, et al. Lapatinib resistance in HCT116 cells is mediated by elevated MCL-1 expression and decreased BAK activation and not by ERBB receptor kinase mutation. Mol Pharmacol. 2008;74:807–22. doi: 10.1124/mol.108.047365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wilson JM. Adenoviruses as gene-delivery vehicles. N Engl J Med. 1996;334:1185–7. doi: 10.1056/NEJM199605023341809. [DOI] [PubMed] [Google Scholar]
- 7.Barouch DH, Nabel GJ. Adenovirus vector-based vaccines for human immunodeficiency virus type 1. Hum Gene Ther. 2005;16:149–56. doi: 10.1089/hum.2005.16.149. [DOI] [PubMed] [Google Scholar]
- 8.Tatsis N, Ertl HC. Adenoviruses as vaccine vectors. Mol Ther. 2004;10:616–29. doi: 10.1016/j.ymthe.2004.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kozarsky KF, Wilson JM. Gene therapy: adenovirus vectors. Curr Opin Genet Dev. 1993;3:499–503. doi: 10.1016/0959-437x(93)90126-a. [DOI] [PubMed] [Google Scholar]
- 10.Di Fiore PP, Pierce JH, Kraus MH, Segatto O, King CR, Aaronson SA. erbB-2 is a potent oncogene when overexpressed in NIH/3T3 cells 21. Science. 1987;237:178–82. doi: 10.1126/science.2885917. [DOI] [PubMed] [Google Scholar]
- 11.Hudziak RM, Schlessinger J, Ullrich A. Increased expression of the putative growth factor receptor p185HER2 causes transformation and tumorigenesis of NIH 3T3 cells. Proc Natl Acad Sci U S A. 1987;84:7159–63. doi: 10.1073/pnas.84.20.7159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Moasser MM. Targeting the function of the HER2 oncogene in human cancer therapeutics. Oncogene. 2007;26:6577–92. doi: 10.1038/sj.onc.1210478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang SC, Lien HC, Xia W, et al. Binding at and transactivation of the COX-2 promoter by nuclear tyrosine kinase receptor ErbB-2. Cancer Cell. 2004;6:251–61. doi: 10.1016/j.ccr.2004.07.012. [DOI] [PubMed] [Google Scholar]
- 14.Wang SE, Narasanna A, Perez-Torres M, et al. HER2 kinase domain mutation results in constitutive phosphorylation and activation of HER2 and EGFR and resistance to EGFR tyrosine kinase inhibitors. Cancer Cell. 2006;10:25–38. doi: 10.1016/j.ccr.2006.05.023. [DOI] [PubMed] [Google Scholar]
- 15.Kershaw MH, Jackson JT, Haynes NM, et al. Gene-engineered T cells as a superior adjuvant therapy for metastatic cancer 1. J Immunol. 2004;173:2143–50. doi: 10.4049/jimmunol.173.3.2143. [DOI] [PubMed] [Google Scholar]
- 16.Amalfitano A, Hauser MA, Hu H, Serra D, Begy CR, Chamberlain JS. Production and characterization of improved adenovirus vectors with the E1, E2b, and E3 genes deleted. J Virol JID - 0113724. 1998;72:926–33. doi: 10.1128/jvi.72.2.926-933.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hartman ZC, Black EP, Amalfitano A. Adenoviral infection induces a multi-faceted innate cellular immune response that is mediated by the toll-like receptor pathway in A549 cells. Virology. 2006 doi: 10.1016/j.virol.2006.08.041. [DOI] [PubMed] [Google Scholar]
- 18.Huang dW, Sherman BT, Tan Q, et al. DAVID Bioinformatics Resources: expanded annotation database and novel algorithms to better extract biology from large gene lists. Nucleic Acids Res. 2007;35:W169–W175. doi: 10.1093/nar/gkm415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.O'Hayer KM, Counter CM. A genetically defined normal human somatic cell system to study ras oncogenesis in vivo and in vitro. Methods Enzymol. 2006;407:637–47. doi: 10.1016/S0076-6879(05)07050-3. [DOI] [PubMed] [Google Scholar]
- 20.Xia W, Gerard CM, Liu L, Baudson NM, Ory TL, Spector NL. Combining lapatinib ( GW572016), a small molecule inhibitor of ErbB1 and ErbB2 tyrosine kinases, with therapeutic anti-ErbB2 antibodies enhances apoptosis of ErbB2-overexpressing breast cancer cells. Oncogene. 2005;24:6213–21. doi: 10.1038/sj.onc.1208774. [DOI] [PubMed] [Google Scholar]
- 21.Piechocki MP, Ho YS, Pilon S, Wei WZ. Human ErbB-2 (Her-2) transgenic mice: a model system for testing Her-2 based vaccines 1. J Immunol. 2003;171:5787–94. doi: 10.4049/jimmunol.171.11.5787. [DOI] [PubMed] [Google Scholar]
- 22.duPre' SA, Redelman D, Hunter KW., Jr Microenvironment of the murine mammary carcinoma 4T1: endogenous IFN-gamma affects tumor phenotype, growth, and metastasis. Exp Mol Pathol. 2008;85:174–88. doi: 10.1016/j.yexmp.2008.05.002. [DOI] [PubMed] [Google Scholar]
- 23.DuPre SA, Redelman D, Hunter KW., Jr The mouse mammary carcinoma 4T1: characterization of the cellular landscape of primary tumours and metastatic tumour foci. Int J Exp Pathol. 2007;88:351–60. doi: 10.1111/j.1365-2613.2007.00539.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Piechocki MP, Pilon SA, Wei WZ. Quantitative measurement of anti-ErbB-2 antibody by flow cytometry and ELISA. J Immunol Methods. 2002;259:33–42. doi: 10.1016/s0022-1759(01)00487-2. [DOI] [PubMed] [Google Scholar]
- 25.Frank R, Overwin H. SPOT synthesis. Epitope analysis with arrays of synthetic peptides prepared on cellulose membranes. Methods Mol Biol. 1996;66:149–69. doi: 10.1385/0-89603-375-9:149. [DOI] [PubMed] [Google Scholar]
- 26.Wei WZ, Shi WP, Galy A, et al. Protection against mammary tumor growth by vaccination with full-length, modified human ErbB-2 DNA 3. Int J Cancer. 1999;81:748–54. doi: 10.1002/(sici)1097-0215(19990531)81:5<748::aid-ijc14>3.0.co;2-6. [DOI] [PubMed] [Google Scholar]
- 27.Piechocki MP, Ho YS, Pilon S, Wei WZ. Human ErbB-2 (Her-2) transgenic mice: a model system for testing Her-2 based vaccines. J Immunol. 2003;171:5787–94. doi: 10.4049/jimmunol.171.11.5787. [DOI] [PubMed] [Google Scholar]
- 28.Riedel H, Massoglia S, Schlessinger J, Ullrich A. Ligand activation of overexpressed epidermal growth factor receptors transforms NIH 3T3 mouse fibroblasts. Proc Natl Acad Sci U S A. 1988;85:1477–81. doi: 10.1073/pnas.85.5.1477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Qiu Y, Ravi L, Kung HJ. Requirement of ErbB2 for signalling by interleukin-6 in prostate carcinoma cells. Nature. 1998;393:83–5. doi: 10.1038/30012. [DOI] [PubMed] [Google Scholar]
- 30.Park JM, Terabe M, Steel JC, et al. Therapy of advanced established murine breast cancer with a recombinant adenoviral ErbB-2/neu vaccine. Cancer Res. 2008;68:1979–87. doi: 10.1158/0008-5472.CAN-07-5688. [DOI] [PubMed] [Google Scholar]
- 31.Lachman LB, Rao XM, Kremer RH, Ozpolat B, Kiriakova G, Price JE. DNA vaccination against neu reduces breast cancer incidence and metastasis in mice. Cancer Gene Ther. 2001;8:259–68. doi: 10.1038/sj.cgt.7700300. [DOI] [PubMed] [Google Scholar]
- 32.Wang X, Wang JP, Rao XM, Price JE, Zhou HS, Lachman LB. Prime-boost vaccination with plasmid and adenovirus gene vaccines control HER2/neu+ metastatic breast cancer in mice. Breast Cancer Res. 2005;7:R580–R588. doi: 10.1186/bcr1199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tegerstedt K, Lindencrona JA, Curcio C, et al. A single vaccination with polyomavirus VP1/VP2Her2 virus-like particles prevents outgrowth of HER-2/neu-expressing tumors. Cancer Res. 2005;65:5953–7. doi: 10.1158/0008-5472.CAN-05-0335. [DOI] [PubMed] [Google Scholar]
- 34.Gallo P, Dharmapuri S, Nuzzo M, et al. Adenovirus vaccination against neu oncogene exerts long-term protection from tumorigenesis in BALB/neuT transgenic mice. Int J Cancer. 2006 doi: 10.1002/ijc.22274. [DOI] [PubMed] [Google Scholar]
- 35.Renard V, Sonderbye L, Ebbehoj K, et al. HER-2 DNA and protein vaccines containing potent Th cell epitopes induce distinct protective and therapeutic antitumor responses in HER-2 transgenic mice. J Immunol. 2003;171:1588–95. doi: 10.4049/jimmunol.171.3.1588. [DOI] [PubMed] [Google Scholar]
- 36.Cipriani B, Fridman A, Bendtsen C, et al. Therapeutic vaccination halts disease progression in BALB-neuT mice: the amplitude of elicited immune response is predictive of vaccine efficacy. Hum Gene Ther. 2008;19:670–80. doi: 10.1089/hum.2007.127. [DOI] [PubMed] [Google Scholar]
- 37.Park JM, Terabe M, Sakai Y, et al. Early role of CD4+ Th1 cells and antibodies in HER-2 adenovirus vaccine protection against autochthonous mammary carcinomas. J Immunol. 2005;174:4228–36. doi: 10.4049/jimmunol.174.7.4228. [DOI] [PubMed] [Google Scholar]
- 38.Andrechek ER, Hardy WR, Siegel PM, Rudnicki MA, Cardiff RD, Muller WJ. Amplification of the neu/erbB-2 oncogene in a mouse model of mammary tumorigenesis. Proc Natl Acad Sci U S A. 2000;97:3444–9. doi: 10.1073/pnas.050408497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jacob J, Radkevich O, Forni G, et al. Activity of DNA vaccines encoding self or heterologous Her-2/neu in Her-2 or neu transgenic mice. Cell Immunol. 2006;240:96–106. doi: 10.1016/j.cellimm.2006.07.002. [DOI] [PubMed] [Google Scholar]
- 40.Osada T, Yang XY, Hartman ZC, et al. Optimization of vaccine responses with an E1, E2b and E3-deleted Ad5 vector circumvents pre-existing anti-vector immunity. Cancer Gene Ther. 2009;16:673–82. doi: 10.1038/cgt.2009.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
4T1-HER2 cells (5,000 per well in 96-well plate) were plated with various concentrations of herceptin and assessed for proliferation by MTT assay 24 hrs. after plating (n=5). Error Bars represent SD.
HMECs were serum starved for 36 hrs, infected with Ad-HER2-wt, Ad-HER2-ki, or Ad-GFP at a MOI of 150. Western blots were performed at 18hpi using HER2-specific or Beta-Actin specific antibodies.