Abstract
In this Letter the authors introduce a wide-field transmission ultrasound approach to breast imaging based on the use of a large area acousto-optic (AO) sensor. Accompanied by a suitable acoustic source, such a detector could be mounted on a traditional mammography system and provide a mammographylike ultrasound projection image of the compressed breast in registration with the x-ray mammogram. The authors call the approach acoustography. The hope is that this additional information could improve the sensitivity and specificity of screening mammography. The AO sensor converts ultrasound directly into a visual image by virtue of the acousto-optic effect of the liquid crystal layer contained in the AO sensor. The image is captured with a digital video camera for processing, analysis, and storage. In this Letter, the authors perform a geometrical resolution analysis and also present images of a multimodality breast phantom imaged with both mammography and acoustography to demonstrate the feasibility of the approach. The geometric resolution analysis suggests that the technique could readily detect tumors of diameter of 3 mm using 8.5 MHz ultrasound, with smaller tumors detectable with higher frequency ultrasound, though depth penetration might then become a limiting factor. The preliminary phantom images show high contrast and compare favorably to digital mammograms of the same phantom. The authors have introduced and established, through phantom imaging, the feasibility of a full-field transmission ultrasound detector for breast imaging based on the use of a large area AO sensor. Of course variations in attenuation of connective, glandular, and fatty tissues will lead to images with more cluttered anatomical background than those of the phantom imaged here. Acoustic coupling to the mammographically compressed breast, particularly at the margins, will also have to be addressed.
Keywords: breast imaging, acoustography, ultrasound, mammography
INTRODUCTION
Breast cancer is the second leading cause of cancer-related death in American women.1 It is now generally accepted that screening mammography reduces the rate of death from breast cancer among women who are 40 years of age or older.2 However, as many as 50% of breast cancers are not detected on film mammograms at the time of screening.3 In addition, 90% of the “suspicious” indications on mammograms turn out to be noncancerous after callback.4
Recently, attempts have been made to integrate ultrasound breast imaging with mammography equipment to improve mammography’s performance.5, 6, 7 However, conventional ultrasound imaging is based on the well-known pulse-echo principle that generates a single cross-sectional B-scan image at the position of the ultrasound probe. Therefore, accurate mechanical scanning of the ultrasound probe with fine indexing is required to generate data of the entire compressed breast, which may make the approach impractical for routine clinical use. Although ultrasound data can be digitally manipulated to achieve spatial correlation with the mammogram, interpreting the large number of B-scan images can be time consuming and fatiguing for the radiologist.
Alternatively, Lasser et al. explored the possibility of integrating a 2D ultrasound detector array with mammography to provide projection images of the breast.8 The detector array used in this system was a modified CMOS infrared chip consisting of 128×128 elements over a 1×1 cm2 area, which was used with a complex acoustic lens system to provide a 1 in. diameter field of view projection image. To achieve a large field of view to provide an 8×10 in.2 mammographylike ultrasound projection image, a very large acoustic lens would be required. Increasing the acoustic lens size is possible but the resulting extended water path9 would make this system dramatically large, heavy, and impractical for integration with mammography equipment. Moreover, the lateral resolution of the ultrasound breast image would be very poor with a 128×128 element CMOS-type ultrasound detector array. The acoustic detection technology we are introducing addresses the resolution issue because it is based on a continuous sensing layer, not an array of discrete sensors. It can also be incorporated into a compact design that is compatible with existing mammography systems.
MATERIALS AND METHODS
The acousto-optic sensor
An ideal ultrasound detector for generating full-field projected breast images would be similar in size to a film or x-ray detector used in mammography and would offer comparably high resolution. One of us (J.S.) developed such a detector, called an acousto-optic (AO) sensor (Santec Systems, Inc., Wheeling, IL), and it has been used successfully to produce radiographylike ultrasound projection images of industrial materials for detection of hidden flaws.10, 11 The AO sensor lends itself naturally to the possibility of producing mammographylike ultrasound projection images of the compressed breast, as will be shown in this Letter.
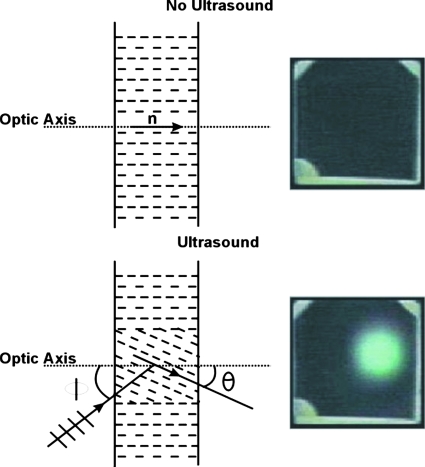
The AO sensor converts ultrasound directly into a visual image by virtue of the acousto-optic effect of a liquid crystal (LC) layer10, 11 (Fig. 1). The LC molecules are initially arranged to be parallel to each other but perpendicular to the confining substrates. The two confining substrates are both acoustically transparent; the rear substrate is also optically transparent to allow for visualization of the LC by a still or video camera. In this configuration of the molecules, the LC material behaves like a slab of uniaxial positive birefringent crystal with optic axis parallel to the LC molecular alignment direction.
Figure 1.
The acousto-optic sensor converts ultrasound directly into a visual image. The liquid crystal molecules are initially parallel to the optic axis (labeled n). When exposed to ultrasound they twist through an angle θ, altering the reflectivity of the sensor. Areas exposed to ultrasound then appear bright when exposed to light.
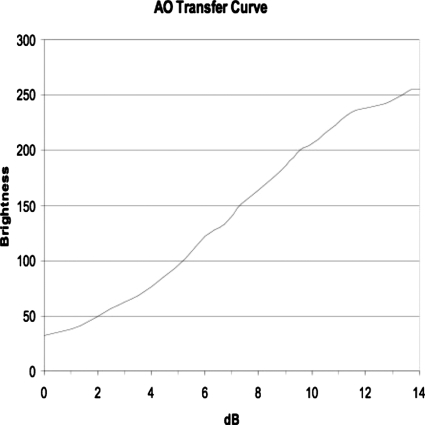
In the absence of ultrasound, the molecular tilt angle is zero and the AO sensor exhibits a uniform dark field of view (see Fig. 1). However, when an area of the AO sensor is exposed to ultrasound, the LC molecules in the exposed region experience a torque that tilts them. The titled molecules are optically reflective in proportion to the degree of tilt. As a result, the ultrasonically exposed area of the AO sensor appears bright when illuminated with an external source of light (see Fig. 1). The ultrasonically induced brightness change in the LC material is referred to as the acousto-optic effect. The AO sensor brightness change is related to ultrasonic intensity through a characteristic curve, as shown in Fig. 2. Therefore, the ultrasonic image, which is essentially a transmitted ultrasonic intensity distribution map, is converted directly into a visual image by the AO sensor.
Figure 2.
The experimentally measured transfer curve from acoustic intensity (in dB) incident on the AO sensor to optical brightness.
The AO sensor offers a high lateral resolution because the ultrasound is sensed by a continuous layer of the LC material, where the sensing molecules are on the order of 20×10−10 m in size. This high resolution has allowed a similar AO sensor to visualize ultrasound fields of a 40 MHz transducer; a focal zone with a cross-sectional area of less than 200 μm was easily visualized.12
The AO sensor circumvents the need for a large number of interconnects, analog-to-digital conversion∕multiplexing circuitry, high conversion rates, electronic cross-talk, etc., which are the limiting factors for 2D ultrasonic arrays. Therefore, the AO sensor can be extended to have a large area without compromising its lateral resolution.
The ultrasound detection threshold of the AO sensor has been shown to be around 10−6 W∕cm2 or better.13 This detection threshold will be adequate to image the compressed breast with the 100 mW∕cm−2 ultrasound exposure level typically used for diagnostic ultrasound; the total ultrasound attenuation of a 5 cm thick average compressed breast is 15 dB assuming 3 dB∕cm attenuation coefficient at 5 MHz. The acoustic dynamic range of current AO sensors is up to 50 dB and the optical dynamic range of the system can be tailored by varying the illumination and camera hardware.
Acoustomammography image formation
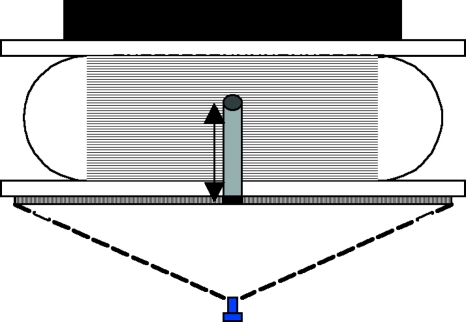
In the prototype system, the compressed breast is ultrasonically illuminated using plane waves (Fig. 3). As they propagate through the compressed breast, the waves are differentially attenuated and an ultrasonic shadow image of the breast anatomy is created on the AO sensor for instant conversion into a visual image. A high-resolution digital video camera is used to acquire the ultrasonic image on the AO sensor for computer storage, further image processing, and image enhancement.
Figure 3.
Mammographylike ultrasound breast image formation concept. An acoustic source at the base of the figure produces a plane wave illumination of the compressed breast. The ultrasonic transmission is then detected by the AO sensor depicted schematically as the black rectangle at the top of the figure.
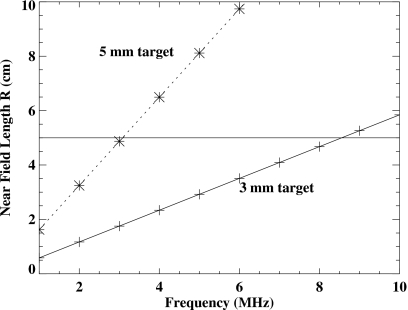
The geometrical resolution capability of the proposed ultrasonic image formation approach can be assessed using classical diffraction theory. According to the theory, an opaque target is recognizable as long as the distance L between the target (e.g., tumor) and the plane of observation (i.e., AO sensor) is less than R, the Fresnel or near field diffraction length, given by R=D2∕λ where D is the target diameter and λ is the ultrasound wavelength in the breast tissue. Since λ=c∕f, where c is the ultrasound velocity in breast tissue and f is the ultrasound frequency of the illuminating ultrasound plane waves, R can be written asR=fD2∕c.
Figure 4 shows R as a function of f for 3 and 5 mm target sizes, where the velocity c in breast tissue was assumed to be 1540 m∕s. To image a target size with good geometrical resolution, L must be less than R. For 3 and 5 mm size lesions located anywhere in a 5 cm thick compressed breast, the L<R condition can be achieved with ultrasound frequencies of 8.5 and 3 MHz, respectively. Smaller targets can be seen at locations closer to the detector. Of course, the use of higher frequencies such as 10 or 15 MHz could allow geometrical resolution of much smaller targets, though depth penetration might them become a limiting factor.
Figure 4.
To image a target of specified size with good geometrical resolution, the distance L between the target and the sensor must be less than the target size-dependent near field length R. The horizontal line represents the 5 cm thickness of a typical compressed breast. This illustrates that for 3 and 5 mm size lesions located in a 5 cm thick compressed breast, the L<R condition can be achieved anywhere in the breast with ultrasound frequencies of 3 and 8.5 MHz, respectively.
Phantom testing
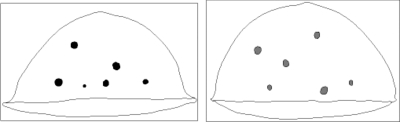
We tested the concept using two multimodality breast phantoms custom-built for us by CIRS, Inc. The phantoms used Zerdine, a water-based polymer developed by CIRS that mimics the ultrasonic properties of tissue.14 The attenuation coefficient was 0.42 dB∕cm∕MHz and the speed of sound was 1454 m∕s, according to CIRS, Inc. Schematics of the phantoms are shown in Fig. 5. Phantom 1 contained six cystic masses ranging in size from 3 to 10 mm diameter. The attenuation coefficient of the cystic masses was 0 dB∕cm∕MHz and speed of sound was 1490 m∕s. Phantom 2 contains six dense masses, also ranging in size from 3 to 10 mm diameter. The attenuation coefficient and speed of sound for the dense masses were 1.25 dB∕cm∕MHz and 1548 m∕s, respectively.
Figure 5.
Schematics of the solid mass (left) and cystic (right) phantoms used in the imaging studies.
To establish the feasibility of acoustomammography prior to building a full-field sensor, we imaged the phantom using the AcousticScope 200 system (Santec Systems, Inc., Wheeling, IL), which offers a 2×2 in.2 field of view and uses 3.3 MHz ultrasound to produce projection images. The system allowed 2×2 in.2 contiguous areas of the phantom, compressed between two Plexiglas plates, to be imaged and stitched together to present a projection image of the entire phantom. As mentioned above, there are no technical limitations to fabricating a larger AO sensor.
For comparison, we performed digital mammography with a GE2000 Premium View system. We employed a molybdenum target with molybdenum filtration, a tube potential of 25 kVp, and a 45.0 mAs tube current. The phantom was compressed to 4.5 cm thickness.
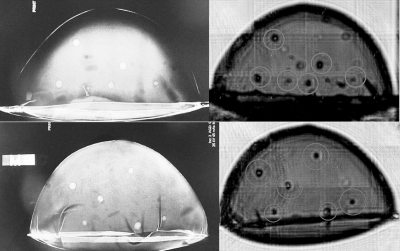
Results of the imaging are shown in Fig. 6. All of the masses and cysts were readily detected by acoustography. The other signals seen in the acoustography images are due to trapped air pockets in the phantom, which produce near-total acoustic reflection but which would not be an issue in real tissue. Many of these air pockets can be visualized in the digital mammogram as well.
Figure 6.
Each figure shows a digital mammogram (left) and corresponding acoustography image (right) for the dense mass (top) and cystic (bottom) phantoms.
These images establish the feasibility of the technique and its ability to produce high-contrast projected ultrasound images of the compressed breast. Of course variations in attenuation of connective, glandular, and fatty tissues will lead to images with more cluttered anatomical background compared with the phantom imaged here, which was homogeneous except for the nonoverlapping lesions. It remains to be seen whether more complex imaging strategies will be required to detect malignant masses in the human breast. Acoustic coupling to the mammographically compressed breast, particularly at the margins, will also have to be addressed. Work to characterize the resolution, noise, and contrast properties of the acoustography approach is ongoing, and patient imaging with a large area sensor is planned in the near future.
The advantages of the acoustography based on an AO sensor include potential compatibility with existing mammography systems to allow for rapid sequential coregistered multimodality imaging. This is in contrast to dedicated whole breast ultrasound systems, such as the ultrasound tomography system of Duric et al., which would be performed as a follow-up to traditional mammography.15 Of course, the tomographic system would provide three-dimensional information unavailable from the projection-mode acoustography system envisioned.
CONCLUSIONS
We have introduced and established the feasibility of a full-field transmission ultrasound detector for breast imaging based on the use of a large area AO sensor. Such a system could be mounted on a traditional mammography system and provide a mammographylike ultrasound projection image of the compressed breast in registration with the x-ray mammogram. The hope is that this additional information could improve the sensitivity and specificity of screening mammography.
ACKNOWLEDGMENTS
This work is supported in part by NIH SBIR Grant No. 1R43 CA137925-01 to Santec Systems, Inc. Patrick J. La Riviere is supported in part by an American Cancer Society research scholar grant.
References
- American Cancer Society, Cancer Facts and Figures (American Cancer Society, Atlanta, 2008). [Google Scholar]
- Humphrey L. L., Helfand M., Chan B. K., and Woolf S. H., “Breast cancer screening: A summary of the evidence for the U.S. Preventive Services Task Force,” Ann. Intern. Med. 137, 347–360 (2002). [DOI] [PubMed] [Google Scholar]
- Pisano E. D., Gatsonis C., Hendrick E., Yaffe M., Baum J. K., Acharyya S., Conant E. F., Fajardo L. L., Bassett L., D’Orsi C., Jong R., and Rebner M., “Diagnostic performance of digital versus film mammography for breast-cancer screening,” N. Engl. J. Med. 353, 1773–1783 (2005). 10.1056/NEJMoa052911 [DOI] [PubMed] [Google Scholar]
- Rosenberg R. D., Yankaskas B. C., Abraham L. A., Sickles E. A., Lehman C. D., Geller B. M., Carney P. A., Kerlikowske K., Buist D. S. M., Weaver D. L., Barlow W. E., and Ballard-Barbash R., “Performance benchmarks for screening mammography,” Radiology 241, 55–66 (2006). 10.1148/radiol.2411051504 [DOI] [PubMed] [Google Scholar]
- Dines K. A., Kelly-Fry E., and Romilly A. P., “Mammography method and apparatus,” U.S. Patent No. 6,876,879 B2 (April 5, 2005).
- Shmulewitz A., “Method and apparatus for performing sonomammography and enhanced x-ray imaging,” U.S. Patent No. 5,664,573 (September 9, 1997).
- Sinha S. P. et al. , “Automated ultrasound scanning on a dual-modality breast imaging system,” J. Ultrasound Med. 26, 645–655 (2007). [DOI] [PubMed] [Google Scholar]
- Lasser R. S., Lasser M. E., and Gurney J. W., “Apparatus for multimodal plane wave ultrasound imaging,” U.S. Patent 6,971,991 B2 (December 6, 2005).
- Freeman M. T., “High-resolution speckle-free ultrasound imaging system-A potential solution for detecting missed breast cancer,” U.S. Army Medical Research and Material Command annual report, 2003. (unpublished).
- Sandhu J. S., “Acoustography: A new imaging technique and its applications to nondestructive evaluation,” Mater. Eval. 46, 608–613 (1988). [Google Scholar]
- Bond-Thorley A., Wang H., and Sandhu J. S., “Application of acoustography for the ultrasonic NDE of aerospace composites,” Proc. SPIE 3993, 23–34 (2000). 10.1117/12.385503 [DOI] [Google Scholar]
- Sandhu J. S., Pergantis C., and Popek W., “Fast and simple ultrasonic field mapping of transducers using acoustography,” Proceedings of the SAMPE Conference, 2007. (unpublished).
- Sandhu J. S., Wang H., and Popek W., “Liquid crystal based acoustic imaging,” Proc. SPIE 3955, 94–108 (2000). 10.1117/12.379970 [DOI] [Google Scholar]
- Zerhouni M. B. and Rachedine M., “Ultrasonic calibration material and method,” U.S. Patent No. 5196343 (March 23, 1993).
- Duric N., Littrup P., Poulo L., Babkin A., Pevzner R., Holsapple E., Rama O., and Glide C., “Detection of breast cancer with ultrasound tomography: First results with the computed ultrasound risk evaluation (CURE) prototype,” Med. Phys. 34, 773 (2007). 10.1118/1.2432161 [DOI] [PubMed] [Google Scholar]