Abstract
Susceptibility to mouse adenovirus type 1 (MAV-1) is associated with the major quantitative trait locus (QTL) Msq1. Msq1 was originally mapped to a 13 megabase (Mb) region of mouse Chromosome (Chr) 15 in crosses between SJL/J and BALB/cJ inbred mice. We have now narrowed Msq1 to a 0.75 Mb interval from 74.68 to 75.43 Mb defined by two anonymous markers, rs8259436 and D15Spn14, using data from 1396 backcross mice. The critical interval includes fourteen Ly6 or Ly6-related genes including Ly6a (encoding Sca-1/TAP), Ly6e (Sca-2/Tsa1), Ly6g (Gr-1) and gpihbp1 (GPI-anchored HDL-binding protein 1), as well as the gene encoding an aldosterone synthase (Cyp11b2). The Ly6 family members are attractive candidates for virus susceptibility genes because their products are GPI-anchored membrane proteins expressed on lymphoid and myeloid cells, with proposed functions in cell adhesion and cell signaling. To determine interstrain variation in susceptibility and produce additional resources for cloning Msq1, we assayed the susceptibility phenotype of four previously untested inbred mouse strains. Susceptibility of strain 129S6/SvEvTac was subsequently localized to the Ly6 complex region using polymorphic genetic markers on Chr 15 in a population of 271 (129S6/SvEvTac x BALB/cJ)F1 x BALB/cJ backcross mice. We identified a major 129S6/SvEvTac susceptibility allele, Msq1129S6, on Chr 15 in the same region as Msq1SJL. The results indicate that a major host factor in MAV-1 susceptibility is likely to be a member of the Ly6 gene family.
Introduction
The genetic background of an individual can significantly influence the outcome of viral infection. Identification of genes involved in susceptibility or resistance to viruses has increased our understanding of many viral disease processes (1). Unbiased genetic approaches have revealed specific host proteins involved in susceptibility to both RNA and DNA viruses, such as West Nile virus and mouse cytomegalovirus (2–5).
Inbred and outbred strains of mice vary significantly in their susceptibility to MAV-1 (6–8). MAV-1 has similarities to human adenoviruses in structure, genome organization, and some aspects of pathogenesis (9). Both MAV-1 and human adenoviruses cause acute and persistent infections with high morbidity and mortality in immunocompromised hosts. Respiratory infection by both mouse and human adenoviruses results in chemokine upregulation (reviewed in 10). MAV-1 primarily infects endothelial cells, which is only rarely seen for human adenoviruses. The endotheliotropism is particularly marked in the brain, spinal cord, and spleen (7, 11, 12), and MAV-1 causes hemorrhagic encephalomyelitis accompanied by breakdown of the blood-brain barrier and altered expression of tight junction proteins (6, 7, 13). In addition, MAV-1 targets monocytes and macrophages, which are also effectors of the host response to MAV-1 infection (14). We are investigating the mechanisms involved in MAV-1 pathogenesis using a variety of methods, and we have shown that both adaptive and innate immune responses play a role in the viral disease process (10, 14–16). We are using a positional cloning approach to identify the important host genes involved in susceptibility of mice to MAV-1.
Susceptibility to MAV-1 is a dominant quantitative trait, with a QTL, Msq1, mapping to Chr 15 (logarithm of odds (LOD) score = 21) (17). Msq1SJL accounts for 40% of the trait variance between the susceptible SJL/J strain and resistant BALB/cJ strain. Initially the 2 LOD confidence interval for Msq1SJL was 18 Mb and included ~300 predicted genes, including Hsf1 (the gene encoding heat shock factor 1 protein), Sla1 (Src-like adapter protein 1), and the Ly6 gene-family complex (lymphocyte antigen 6). The Ly6 genes are particularly attractive as candidate genes for susceptibility because they encode GPI-anchored cell-surface proteins (18). There are more than 20 predicted Ly6 or Ly6-related genes in this interval of Chr 15, and protein functions or expression patterns are known for some. LY6 proteins are variably expressed on different cell types including lymphoid and non-lymphoid cells, and during hematopoietic cell differentiation. Some LY6 proteins are involved in lymphocyte adhesion, T-cell responses, and the interferon response. The chicken Ly6E gene has been identified as a candidate gene for resistance to the avian herpesvirus Marek’s disease virus (19). Recently two different approaches have identified Ly6 or Ly6-related gene involvement in HIV-1 susceptibility. A whole-genome linkage analysis using cellular susceptibility to HIV in vitro identified the Ly6 locus on human Chr 8q24 (20). A small interfering RNA screen identified 250 HIV-dependency factors, 3 of which were members of the LY6 family: Gml, Ly6D, and Lypd4 (21). In a similar approach, LY6E was identified as one of 305 human proteins that affect West Nile virus infection (22).
To determine which of the candidate gene(s) in the Msq1SJL locus correlates with susceptibility to MAV-1, we used fine mapping and haplotype analysis to reduce the interval from 18 Mb to 0.75 Mb, a region which encompasses 14 Ly6 or Ly6-related genes. The Ly6 gene complex in mice has two major haplotypes, Ly6a and Ly6b, that code for two lymphocyte specificities with distinct inbred mouse strain distribution patterns (23–25). These strain distribution patterns correlate with known susceptibility or resistance to MAV-1 among inbred strains (8). We therefore identified additional strains of mice that are susceptible or resistant to MAV-1 to extend this correlation and develop additional mapping tools. We analyzed progeny of a backcross between BALB/cJ mice and one additional MAV-1-susceptible mouse strain, 129S6/SvEvTac. We identified a QTL allele, Msq1129S6, that maps to the same region of Chr 15 as Msq1SJL (LOD = 46). Taken together, the results provide strong evidence that susceptibility to MAV-1 is encoded by one or more members of the Ly6 gene complex.
Materials and methods
Viruses and mice
Wild-type MAV-1 (26) was propagated and titrated as described (27). All virus stocks used for phenotyping were aliquots from a single preparation that were thawed only one time, for preparation of inocula. All mice were obtained from The Jackson Laboratory except for 129S6/SvEvTac (129S6), which were obtained from Taconic. All animal work complied with relevant federal and institutional policies. Mice were maintained in microisolator housing with food and water provided ad libitum. A selective phenotyping scheme was used on 1140 backcross mice from an expanded backcross, as was used to initially identify the Msq1SJL locus (17), (BALB/cJ x SJL/J)F1 x BALB/cJ. In a different backcross, mice were obtained by mating both (129S6 x BALB/cJ)F1 and (BALB/cJ x 129S6)F1 mice as the female parent with BALB/cJ mice as the male parent. Breeding pairs were maintained on breeder chow (Purina 5008).
Phenotyping
Mice 3–5 weeks of age were injected intraperitoneally with 102 PFU of MAV-1 diluted in 0.1 ml PBS. Mice were euthanized at 8 days p.i., and organs and tissues were collected and stored at −70°C. Brain viral loads were determined by capture ELISA using brain homogenates as described (28); this method gives results that correlate with virus quantitation by plaque assay (17). An average ELISA value from homogenates of mock-infected mice was background-subtracted from the infected samples. To standardize assays performed at different times, a MAV-1 virus stock was included as a positive control in each assay performed; resulting experimental sample values were normalized to the MAV-1 control. PBS was used as a negative control in each assay. Susceptible mice were originally defined as those with an LD50 of <103 PFU, and resistant mice all had LD50 values >104.4 (8). These values correspond to OD450 ~0.04 and 0.31 in the capture ELISA (17).
Genotyping
All marker positions in this work correspond to NCBI Build 37 of the Mus musculus genome sequence. Simple sequence length polymorphism (SSLP) markers that differentiate between the strains used were identified from the Chr 15 region of interest (http://www.informatics.jax.org/). We confirmed that the SSLPs were polymorphic between BALB/cJ and SJL/J or 129S6 mice by performing PCR on genomic DNAs of each strain. To produce additional SSLP markers, we identified regions of tandem repeats in the mouse genome interval of interest using the Tandem Repeats Database (http://tandem.bu.edu/cgi-bin/trdb/trdb.exe) (29). Primers were designed to amplify regions of ~200 nt from template DNAs for the three mouse strains. Primer pairs that amplified newly identified polymorphic PCR products used in this work are listed in Table I.
Table 1.
New microsatellite loci used in this study
| Polymorphic between |
|||||
|---|---|---|---|---|---|
| Locus | Map positiona | Forward primer | Reverse primer | BALB/cJ and SJL/J | BALB/cJ and 129S6 |
| D15Spn101 | 72,556,431 | 5′TCAGGCAAGATCTGCTGAAG3′ | 5′ACAGTTGTCCCACGTTCTGG3′ | + | + |
| D15Spn106 | 72,849,859 | 5′ATCCCACACGGAGTGAAAAC3′ | 5′TTTATGCACCAGTGGACAGC3′ | + | + |
| D15Spn14 | 75,432,404 | 5′GGCTGGCCTTAAACTCTCAG3′ | 5′AAGGCACAGCTGGACATGG3′ | + | + |
| D15Spn54 | 75,850,197 | 5′CCTGCTGGGAGAGAGACTTCA3′ | 5′GGAATGAAAGGGGGATCATAC3′ | + | − |
Map position indicated is the first nt at the centromeric side of the repeat (based on NCBI Build 37, July 2007).
Single nucleotide polymorphisms (SNPs) potentially polymorphic for the mouse strains and interval of interest were initially identified using SNP databases extant in 2005–2006, GNF3 and Celera2 (30, 31). The SNPs are now compiled in the Mouse Phenome Database (http://www.jax.org/phenome/SNP) and are shown in Fig. 1 by numbers with an “rs” prefix. Further SNP haplotype analysis of additional resistant and susceptible strains was done using the Center for Genome Dynamics Mouse SNP Database v. 1.3 (http://cgd.jax.org/cgdsnpdb/).
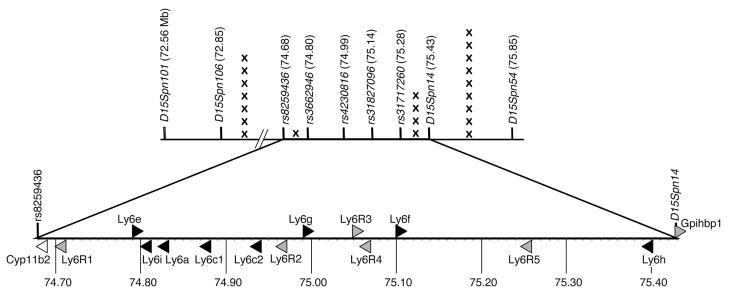
Figure 1.
Genetic mapping of Msq1. The top line shows SSLP and SNP markers used in fine mapping (BALB/cJ x SJL/J)F1 x BALB/cJ backcross progeny in the Chr 15 interval between 72.56 and 75.85 Mb; physical positions of each marker are indicated in parentheses. The X symbols indicate the number of mice with genotypic recombination within each subinterval. The lower line is an expansion of the deduced critical interval with predicted candidate genes. Ly6 genes are indicated by black triangles, Ly6-related genes by gray triangles, and the other gene by a white triangle. Genbank transcript designations for the Ly6-related genes (Ly6R1 – 5) are given in Table II. Genes transcribed from the top (+) strand point to the right and genes transcribed from the bottom (−) strand point to the left. Numbers below the line indicate the physical map position in Mb.
DNAs were isolated from tail snips and amplified by PCR in 20 μl reactions as described (32). Samples were electrophoresed on 7% polyacrylamide gels. Alternatively, DNA samples were analyzed by the University of Michigan Sequencing Core using fluorescently labeled primers. For SNP analysis, only an aliquot of the PCR reaction was electrophoresed on a gel. The remainder of each sample was purified using QIAquick PCR Purification columns (Qiagen), and the DNA was then sequenced by the University of Michigan Sequencing Core.
Statistical analysis and QTL mapping
QTL analysis was performed using R/qtl software. SSLP markers used in the QTL analysis were D15Mit13, D15Mit6, D15Spn101, D15Mit70, and D15Mit107. Individual markers were quality tested for difference from the expected 1:1 ratio using the chi-square test for Mendelian segregation. The D15Mit6 marker showed significant deviation from the 1:1 ratio (P = 7.87 × 10−19) with a surplus of BALB/cJ homozygote allele calls relative to heterozygotes. None of the other markers had significant deviation from expectation (P > 0.05). Mouse brain virus loads were entered as the background-subtracted average ELISA value of triplicate measurements from each mouse. QTL mapping was performed using the R/qtl scanone function with single marker regression and the imputation method, using 271 backcross mice genotyped at 5 markers. General linear modeling (ANOVA) of the allelic effect was performed using the R/qtl fitqtl function at the marker nearest to the QTL peak.
Results
Fine mapping of the Msq1 interval
The 2 LOD confidence interval previously identified for Msq1SJL on Chr 15 was estimated to be from 39 to 54 centimorgans, corresponding roughly to ~65 to 78 Mb on the physical map (17). We determined the MAV-1 susceptibility phenotypes for 256 (BALB/cJ x SJL/J)F1 x BALB/cJ backcross progeny and then genotyped for markers on all 20 mouse chromosomes. Forty backcross mice were recombinant across the Chr 15 2-LOD confidence interval defined by two markers used in the original analysis, D15Mit270 (63.3 Mb) and D15Mit70 (81.0 Mb). An additional microsatellite marker that had been previously described (D15Mit211, 66.6 Mb) was supplemented with two others that we designed, D15Spn101 and D15Spn54 (Table I), to further genotype the recombinant mice. Five of the 256 backcross mice were recombinant across the interval. To fine map the genetic location of Msq1SJL, we expanded the backcross, collecting 1140 additional mice. A selective phenotyping scheme was used: genotypes were determined for all progeny mice at D15Spn101 and D15Spn54; subsequently, only mice recombinant across this interval were infected with MAV-1 for susceptibility phenotype measurement. We identified 19 additional backcross mice that were recombinant across the interval (Fig. 1); 16 of these had phenotypes concordant with their genotypes. The three mice with discordant genotypes and phenotypes are discussed below. Taken together, the original and the expanded backcrosses yielded 21/1396 recombinants (1.50%) across the Msq1SJL interval from 72.56 – 75.85 Mb of Chr 15.
To further refine the candidate interval, we developed additional microsatellite markers and identified SNP markers that are polymorphic for BALB/cJ and SJL/J. The 21 recombinant mice were each genotyped using two or more of these 9 markers (Fig. 1); from these results we deduced a maximal 0.75 Mb interval for the Msq1SJL QTL. One individual had a crossover between rs8259436 and rs3662946, defining the left end of the interval at 74.68 Mb, and four individuals had crossovers between rs31717260 and D15Spn14, defining the right end of the interval at 75.43 Mb.
Fifteen candidate genes in the 0.75 Mb interval were identified using Ensembl and are depicted in Fig. 1 and listed in Table II. Eight are genes encoding Ly6 family proteins (Ly6e, Ly6i, Ly6a, Ly6c1, Ly6c2, Ly6g, Ly6f, and Ly6h). Six genes (gpihbp1 and five single transcripts of unknown association, 2010109I03Rik-201, I830127L07Rik, BC025446, AC116498.15, 9030619P08Rik) encode predicted proteins that have LY6/uPAR domains, and are thus termed Ly6-related. LY6/uPAR sequences are found in a large protein family including the LY6 proteins, urokinase-type plasminogen activator receptor (uPAR) and other proteins (44, 45). LY6/uPAR domains have 10 cysteine residues in a consensus pattern with the C-terminal cysteine followed by an asparagine.
Table II.
Predicted Genes in the Msq1 Interval
| Class | Symbol | Position (Mb), stranda | Alternate name, function, expression |
|---|---|---|---|
| Ly6 | |||
| Ly6e | 74.79, + | Thymic shared antigen-1 (TSA-1), stem cell antigen-2 (Sca-2), expressed on most intra-thymic precursor cells of the lymphoid lineageb | |
| Ly6i | 74.81, − | Expressed on bone marrow cells, monocytes, macrophages, granulocytes, and myeloid precursors; and in spleen, thymus, kidney and lungc | |
| Ly6a | 74.83, − | Stem cell antigen-1 (Sca-1), T cell-activating protein (TAP), expressed on bone marrow stem cells that are precursors to all thymic subsetsb,d | |
| Ly6c1 | 74.88, − | Expressed on inflammatory monocytes, some natural killer cells, and plasmacytoid dendritic cellse | |
| Ly6c2 | 74.94, − | Expressed on leukemia cells and on macrophages infiltrating rejected allograftsf | |
| Ly6g | 74.99, + | Granulocyte differentiation antigen (Gr-1), expressed on mature granulocytesb | |
| Ly6f | 75.10, + | mRNA expressed in non-lymphoid tissuesg | |
| Ly6h | 75.40, − | mRNA expressed in brainh | |
| Ly6-related | |||
| Ly6R1i | 74.71, − | 2010109I03Rik, intectinj | |
| Ly6R2i | 74.97, − | I830127L07Rik | |
| Ly6R3i | 75.05, + | BC025446 | |
| Ly6R4i | 75.07, − | AC116498.15–201 | |
| Ly6R5i | 75.26, − | 9030619P08Rik | |
| Gpihbp1 | 75.43, + | GPI-anchored HDL-binding protein 1, expressed in heart, lung, liverk | |
| Other | |||
| Cyp11b2 | 74.69, − | Cytochrome P450 11b2, aldosterone synthasel | |
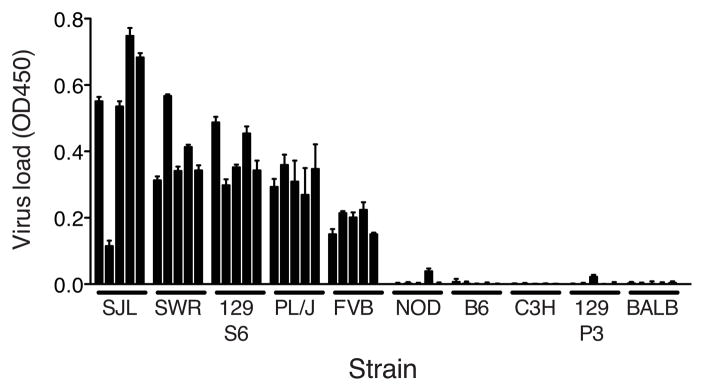
Susceptibility of additional inbred mouse strains to MAV-1
The Ly6 complex has long been known to have two haplotypes, Ly6a and Ly6b, that code for two lymphocyte specificities, Ly6.1 and Ly6.2, respectively, determined by alloantisera to cell-membrane antigens (23–25). The distribution pattern of these Ly6.1 and Ly6.2 specificities among inbred mouse strains correlates with susceptibility that we had previously identified for a number of inbred strains. For example, Ly6.1 strains include BALB/c, A/J, A.SW, C3H/HeJ (23), which are resistant to MAV-1 (8, 17). Ly6.2 strains include SJL and SWR, which are susceptible to MAV-1. To test this correlation further and develop additional resources for mapping Msq1, we screened additional inbred mouse strains for susceptibility to MAV-1. We infected mice and assayed for viral load in the brain using capture ELISA (17) (Fig. 2). The results supported findings in two strains previously characterized by LD50 assay as susceptible to MAV-1 (SJL/J and SWR/J), and four as resistant (129P3/J, BALB/cJ, C3H/HeJ, C57BL/6J) (8). We also identified three additional susceptible strains, PL/J, 129S6, and FVB/NJ; and one additional resistant strain, NOD/ShiLtJ. For two of those strains the Ly6 lymphocyte specificity has been reported, and it correlates with that reported for other susceptible strains: PL/J and129Sv are both Ly6.2 (23, 25).
Figure 2.
MAV-1 susceptibility of inbred strains of mice. Brain viral loads were determined by capture ELISA. Each brain homogenate was assayed in triplicate and each bar represents the average of the background-subtracted triplicate measurements per homogenate with standard deviation. Samples were standardized to a virus stock positive control included in each assay. OD450, optical density at 450 nm. Strain abbreviations: SJL, SJL/J; SWR, SWR/J; 129S6, 129S6/SvEvTac; FVB, FVB/NJ; NOD, NOD/ShiLtJ; B6, C57BL/6J; C3H, C3H/HeJ; 129P3, 129P3/J; BALB, BALB/cJ.
We examined the SNP haplotypes for susceptible and resistant strains across the interval from 72.9 to 75.3 Mb at 15 SNPs spaced 0.2 to 0.3 Mb apart (30). In the region from 73.4 to 74.5 Mb there was haplotype identity among strains, i.e. the susceptible strains SJL/J, SWR/J, FVB/NJ and PL/J had haplotypes in the region identical to the resistant strains BALB/cJ, A/J, and C3H/HeJ (data not shown). This is consistent with our inability to find known SNPs or design microsatellite markers that detect polymorphisms between BALB/cJ and SJL/J mice for the interval from 72.85 to 74.68 Mb. In contrast, in the region from 74.6 to 75.3 Mb, the 4 susceptible strains had identical haplotypes to each other that were different from the shared haplotypes of the 3 resistant strains (data not shown). With a denser SNP database (Center for Genome Dynamics Mouse SNP Database v. 1.3), the haplotype identity persisted from 73.4 to 74.5 Mb (data not shown). The haplotype analysis did not reduce the number of candidate genes in the interval.
Susceptibility of 129S6 mice maps to Chr 15
To determine whether susceptibility of 129S6 mice maps to the same region of Chr 15 as susceptibility of SJL/J mice, we crossed 129S6 and BALB/cJ mice. Progeny F1 animals were infected with MAV-1 and had susceptibility phenotypes (brain virus loads) intermediate between the parental phenotypes (data not shown). This result is similar to the case for (BALB/cJ x SJL/J)F1 mice (17) and indicates that susceptibility in 129S6 mice is a semidominant quantitative trait.
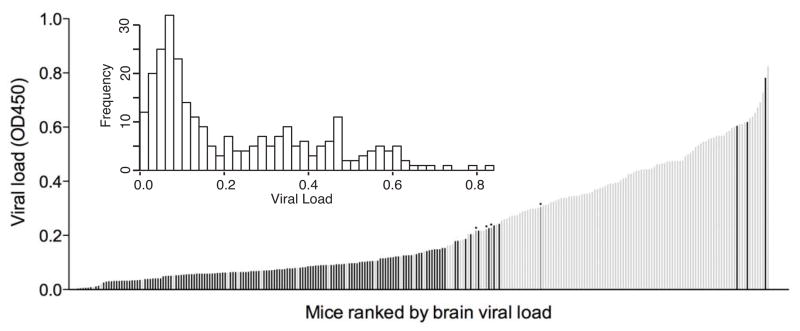
We obtained 271 backcross mice (consisting of both (129S6 x BALB/cJ)F1 x BALB/cJ and (BALB/cJ x 129S6)F1 x BALB/cJ progeny) and determined their susceptibility phenotypes. Figure 3 shows the distribution of phenotypes of the entire backcross population. The distribution was similar when phenotypes of backcross mice from two types of F1 parents were plotted separately (data not shown). The phenotype distribution for the backcross mice is consistent with susceptibility of 129S6 mice being a quantitative trait determined by a single major locus.
Figure 3.
MAV-1 susceptibility phenotypes in backcross mice. Viral loads in 271 (129S6 x BALB/cJ)F1 x BALB/cJ and (BALB/cJ x 129S6)F1 x BALB/cJ backcross mice were determined by ELISA assay as in Fig. 2 and plotted in rank order. Each bar represents the mean value for an individual mouse brain homogenate assayed in triplicate; for clarity standard deviations are not shown but are comparable to those in Fig. 2. Genotypes at D15Spn101 are indicated by the bar colors: black, homozygous BALB/cJ; light gray, heterozygous, BALB/cJ/129S6; and four medium-gray bars marked with dots, undetermined. Inset, the frequency of phenotypic classes of the same population.
We determined genotypes of the 271 backcross mice for 5 markers on Chr 15 that differentiate between 129S6 and BALB/cJ mice. We performed QTL analysis on the 271 backcross mice using a QTL mapping package for the statistical software R, R/qtl, as previously described (17). We observed an association of susceptibility with markers on Chr 15. A one-way scan for QTLs using single marker regression and the imputation method (256 imputations using 5000 permutations and a 2cM step interval) of the mapping analysis showed that viral load is associated with a major effect QTL on Chr 15. The peak marker was D15Spn101 from single marker analysis, and imputation mapping showed the effect maxima on Chr 15 at approximately 69 Mb with a peak LOD of 46.2. The results from fitting the candidate QTL using ANOVA showed the percentage of phenotypic variation explained by the QTL at 69 Mb on Chr 15 analyzed alone was 54% (F[1, 269] = 331, P = 9.01 × 10−49, LOD = 47.3). This interval on Chr 15 for Msq1129S6 encompasses the interval we have previously identified for Msq1SJL.
Discussion
To understand the contribution of genetic variation in host defense pathways to adenoviral pathogenesis and to identify which host genes play a role in MAV-1 susceptibility, we have used a positional cloning approach. An 18 Mb region of mouse Chr 15 was initially identified as the region with the major QTL for MAV-1 susceptibility; this region encoded ~300 predicted genes (17). We used backcross mice, additional polymorphic markers, and SNP haplotype identity to reduce the size of the critical interval to 0.75 Mb. This region encodes fourteen Ly6 or Ly6-related genes and an aldosterone synthase gene. We cannot eliminate aldosterone synthase as a candidate gene, but we believe that Ly6 family members are more likely for reasons based on expression and function, discussed below. The fine mapping eliminated the candidate genes Hsf1 and Sla1, which were previously considered as candidates in the 18 Mb interval based on their biological function, as well as several Chr 15 Ly6 and Ly6-related genes, such as Ly6d, Ly6k, Lynx1, Slurp1, and Psca.
In the (BALB/cJ x SJL/J)F1 x BALB/cJ backcross analysis, 3 of 19 mice that were recombinant across the interval delineated by D15Spn101 and D15Spn54 had phenotypes for susceptibility to MAV-1 that were discordant with their genotypes for markers within the interval. The mice were resistant to MAV-1, as measured by brain viral load, and yet their genotype was heterozygous (BALB/cJ/SJL/J) at the four markers in the interval from 74.80 to 75.28 Mb. Because susceptibility to MAV-1 is dominant (17), mice with the heterozygous genotype should be susceptible. In each case, the phenotype-genotype discordance in these three individuals is in the direction of unexpected resistance. The virus did not replicate to high levels in their brains, despite being injected with virus. We have previously seen such “false negative” occurrences of low viral load in known genetically susceptible mice, at a frequency of 2–4% (Spindler et al., unpublished). An example of a single animal of this type can be seen for the susceptible SJL/J mice assayed in Fig. 2. These events may be due to experimental failures in the infection procedure. A second possibility is that one or more of these three mice does not represent a false negative phenotyping error, but was truly MAV-1-resistant, despite being genetically heterozygous for the interval. That is, cumulative resistant alleles at genes elsewhere in the genome may be sufficient to produce the resistant phenotype. Susceptibility is a quantitative trait, with multiple candidate loci identified in the initial QTL analysis (17). For example, a locus on Chr 5 has an additive effect on the phenotype. We favor false negative infection as the most likely explanation, since we did not obtain any mice with discordance as unexpected susceptibility. However the number of discordant individuals was small and further analysis would be needed to resolve the possible explanations.
We analyzed additional strains for their MAV-1 susceptibility phenotype by measuring brain virus loads by capture ELISA. For strains that had been previously assayed for susceptibility by LD50 rather than ELISA, the ELISA and LD50 results were in agreement (SWR/J, C3H/HeJ, and 129P3/J) (8). We also identified additional susceptible and resistant strains using the ELISA. We found that the strain distribution pattern of Ly6.1 and Ly6.2 specificities among inbred strains correlated with susceptibility to MAV-1. That is, Ly6.1 strains include BALB/c, A/J, A.SW, and C3H/HeJ (23); all of these are resistant to MAV-1 (8). Ly6.2 strains include SJL, SWR, PL/J, and 129Sv (23, 25), and these are susceptible to MAV-1.
Analysis of a backcross of a second susceptible strain (129S6) with the resistant BALB/cJ strain showed that susceptibility to MAV-1 mapped to the same interval on Chr 15 as SJL/J susceptibility. This Msq1129S6 QTL alone accounts for 54% of the variation in susceptibility phenotype. The result that susceptibility in an additional strain maps to the same locus as Msq1SJL enabled us to use SNP haplotype analysis in conjunction with recombinant mouse analysis to confirm the critical interval. The information also has enabled us to produce bacterial artificial chromosome-based transgenic mice from a 129S6 library (Hsu and Spindler, unpublished). Analysis of these mice for dominant MAV-1 susceptibility may reduce the candidate interval and confirm the identity of the susceptibility gene(s) within the 0.75 Mb interval.
The Ly6 complex on Chr 15 in mice is a multigene family with at least 20 related genes that likely arose by intra-chromosomal gene duplication (18, 46–48). LY6 antigens were first identified by polyvalent antisera and subsequently through the use of monoclonal antibodies raised against lymphocytes. This resulted in a complex nomenclature for the LY6 antigens. This was further complicated by the fact that it was not known at first that LY6 antigens were encoded by a multigene family, and the relationship between genes and alleles was not clear. For example, Ly6A.2 and Ly6E.1 were initially thought to be different genes, but they are actually alleles of Ly6A (24). For the Ly6c1 and Ly6c2 genes, the highly similar LY6C1 and LY6C2 proteins were originally thought to be two allelic forms encoded by one gene (18, 49–51). The most recent comprehensive Ly6 gene-family review was published in August 2004 and did not make extensive use of the emerging annotated mouse genome sequence (18). Furthermore, even in the current literature, references are made to populations of cells based on old designations that are ambiguous, such as LY6A/E and LY6G/C. The Gr-1 antigenic specificity of monoclonal antibody RB6-8C5 was first reported to be shared by the Ly6c and Ly6g gene products, with LY6C and LY6G being expressed on distinct cell types (52). However, a more recent study indicates that for this LY6G antibody there is no cross-reactivity with LY6C on multiple hematopoietic cell types (53). To assist in clarifying the Ly6 gene family nomenclature, in this discussion we use the nomenclature of genes in the Ly6 locus established by the International Committee on Standardized Genetic Nomenclature for Mice as found in the Mouse Genome Database (www.informatics.jax.org, August 2009) (43). In addition, five Ly6-related transcripts have been identified solely by genomic sequence annotation within the critical interval for MAV-1 susceptibility (Table II). These Ly6-related transcripts are discussed below using abbreviated names, Ly6R1 – Ly6R5, since they have predicted proteins that all have LY6/uPAR domains (data not shown).
The mRNA sequences of Ly6R1 – 5 are distinct from one another and from the characterized Ly6 genes. Predicted proteins of the Ly6R1, Ly6R2, and Ly6R3 transcripts are noted in the Mouse Genome Database (www.informatics.jax.org/, August 2009) (43) as having LY6/uPAR domains, i.e., sequences with 10 cysteine residues in a consensus pattern with the C-terminal cysteine followed by an asparagine (44, 45). Ly6R2 lacks a predicted initiator methionine, but we have preliminary evidence of a complete Ly6R2 cDNA with an initiator methionine from SJL/J mice (M. Stier and K. Spindler, unpublished). We analyzed the predicted protein sequences of the other two transcripts, Ly6R4 and Ly6R5, and found that the entire LY6/uPAR motif is found in the open reading frame for Ly6R4. The protein predicted from Ly6R5 terminates soon after the 5th cysteine residue; however, downstream in an alternate reading frame we found sequence corresponding to the remainder of the LY6 homology (data not shown). Thus it is possible that Ly6R5 is in the process of becoming a pseudogene. There is little expression or functional data available for the five Ly6-related transcripts in the Chr 15 interval. Ly6R1 encodes a protein that has been identified as intectin, an intestine-specific protein involved in apoptosis of intestinal epithelial cells (41).
Ly6 and Ly6-related genes are found in other vertebrates, including chickens, rats, and humans. In humans, 11 Ly6-related genes are found on Chr 8q24, syntenic to Chr 15 (54); in both mouse and humans, Ly6-related genes are also found on other chromosomes. The mouse Chr 15 interval that we have identified for susceptibility to MAV-1 has more genes and is longer than the corresponding human interval on Chr 8q24. That is, in the 0.61 Mb between mouse Ly6e and Ly6h there are 10 Ly6 and Ly6-related genes (Fig. 1), but there are no predicted genes with sequence similarity to Ly6 genes in the corresponding 0.14 Mb of human Chr 8 between Ly6e and Ly6h (55). Notably, LY6A and LY6C proteins do not have human orthologs.
LY6 proteins encoded in the critical interval for MAV-1 susceptibility are expressed on cells that are either targets of MAV-1 (monocytes/macrophages and endothelial cells) (14, 56) or are lymphoid or myeloid cells (or their precursors) that are effectors in innate and adaptive immunity (Table II), making them strong candidates for involvement in susceptibility. Differences in sequence or expression of these proteins between susceptible and resistant strains may underlie the susceptibility differences among mouse strains. LY6I, LY6C1, and LY6C2 are expressed on monocytes, macrophages, and dendritic cells (reviewed in 18); strain differences in expression patterns on Ly6.1 and Ly6.2 strains have been reported for LY6C (though it is not known whether these differences were for LY6C1 or LY6C2 expression) (51). As measured by immunohistochemistry, the LY6A protein is prominently expressed in brain vascular elements in Ly6.2 strains (C57BL/6 and AKR/J) (57, 58). These authors did not definitively show that the antibody staining was specific to endothelial cells, but it is consistent with endothelial expression of LY6A. In contrast, no expression is seen in vascular elements of Ly6.1 strains (BALB/c and C3H/HeJ) using the same antibody (58). It will be informative to determine whether similar LY6A expression differences are seen for additional Ly6.1 (MAV-1-resistant) and Ly6.2 (MAV-1-susceptible) strains, as this may be a cause or effect of differences in MAV-1 susceptibility.
Alterations in effector functionality of lymphoid or myeloid cells might be caused by Ly6 gene differences and lead to susceptibility to MAV-1. LY6E is expressed on intrathymic cells that give rise to all the thymic subsets; it is differentially expressed during T cell development and is found on immature T cells and peripheral B cells (59, 60). LY6A is expressed on embryonic and fetal aorta and liver, and on adult hematopoietic stem cells (reviewed in 55). LY6A is differentially expressed on hematopoietic stem cells in Ly6.1 and Ly6.2 strains, with ~100% of cells and 25% of cells being positive, respectively. This is reminiscent of the different levels of expression of LY6A in brain vascular elements described above. Both T and B lymphocytes are important for control of MAV-1 infection (15, 16). It is possible that functional differences in T and or B cell-mediated host response to MAV-1 infection result from differences in LY6E, LY6A, or less well characterized LY6 proteins in susceptible and resistant strains.
Little is known about the physiological function of the LY6 proteins, but they are thought to be involved in cell-cell adhesion and signaling (reviewed in 18, 55). Functional roles for LY6 proteins in disease pathogenesis that may relate to viral disease susceptibility are suggested by several findings. Expression of LY6 proteins is induced on many mouse tissues in the animal upon antigenic stimulation (reviewed in 18). During infection of mice by lymphocytic choriomeningitis virus, LY6A expression increases on CD4+ and CD8+ T cells and persists at a high level on memory T cells (61). “Inflammatory” monocytes express high levels of LY6C and are recruited to sites of inflamed tissue during infection by viruses, bacteria, and parasites, in contrast to “resident” monocytes, which are LY6Clo and rarely migrate to inflamed sites (36, 62–65). Both in vivo and in vitro stimulation of immune cells to interferons (IFNs) increases expression of Ly6 genes. For example, both type I and type II IFNs stimulate LY6 expression on T and B lymphocytes (66–68). Interferons also stimulate non-immune cells: IFN-γ stimulates LY6 expression in kidney (69) and on inflamed intestinal epithelial cells in murine models of colitis (70). Since interferon production is a major innate immune response to viral infection, it is reasonable to hypothesize that viral infection may result in altered expression of LY6 proteins. We suggest that this may underlie susceptibility to viral infections caused by Ly6 or Ly6-related genes.
In addition to our findings that Ly6 genes underlie the QTL for susceptibility to MAV-1, studies have implicated Ly6 genes for susceptibility to two other viruses. The chicken Ly6E locus is a major candidate for susceptibility to Marek’s disease virus, a herpesvirus, based on several experimental approaches (19, 71). The human Ly6 locus was identified in a whole genome analysis of lymphoblastoid cell susceptibility to HIV-1 (20), and 3 Ly6 or Ly6-related genes were identified in small interfering RNA screen for host factors required by HIV-1 (21). Ly6E was identified as a host susceptibility factor for West Nile virus in a similar small interfering RNA screen (22). Nothing has been reported about the mechanism by which Ly6 genes contribute to infection by these or other viruses. In fact, infection of Ly6a knockout mice by lymphocytic choriomeningitis virus demonstrated that LY6A is dispensable for normal T cell function in acute and memory responses (61). We found no difference in susceptibility to MAV-1 infection between Ly6a knockout mice and BALB/c controls at either high or low virus doses (105 or 102 PFU respectively) (Althaus and Spindler, unpublished observations). Given the number of Ly6-related proteins and the variety of cell and organ types in which these genes are expressed, the mechanisms may be quite different for various viruses. However the findings that susceptibility to viruses from three different families maps to Ly6 genes strongly suggest that the Ly6 gene products are key players in virus-host interactions.
Acknowledgments
We thank Steffi Ameres, Shanna Ashley, Marina Gerbin, and the University of Michigan Genotyping Core for technical assistance in genotyping. We are grateful to Dave Burke for numerous discussions on mapping strategy and comments on the manuscript. We thank Tien-Huei Hsu and Matt Stier for discussion about Ly6 genes and the manuscript. We thank William Stanford for the gift of Ly6a−/− mice and BALB controls. We thank Tim Wiltshire for generously providing SNP database files prior to publication and Janan Eppig for nomenclature advice. We are grateful to Phil King and Mike Imperiale for comments on the manuscript.
Abbreviations used in this paper
- Chr
chromosome
- LOD
logarithm of odds
- MAV-1
mouse adenovirus type 1
- Mb
megabases
- QTL
quantitative trait locus
- SNP
single nucleotide polymorphism
- SSLP
simple sequence length polymorphism
- uPAR
urokinase-type plasminogen activator receptor.
Footnotes
This work was supported by National Institutes of Health grant AI068645 to K.R.S.
References
- 1.Brinton MA. Host susceptibility to viral disease. In: Nathanson N, Ahmed R, Gonzalez-Scarano R, Griffin DE, Holmes KV, Murphy FA, Robinson HL, editors. Viral Pathogenesis. Lippincott-Raven; Philadelphia: 1997. pp. 303–328. [Google Scholar]
- 2.Perelygin AA, Scherbik SV, Zhulin IB, Stockman BM, Li Y, Brinton MA. Positional cloning of the murine flavivirus resistance gene. Proc Natl Acad Sci USA. 2002;99:9322–9327. doi: 10.1073/pnas.142287799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mashimo T, Lucas M, Simon-Chazottes D, Frenkiel MP, Montagutelli X, Ceccaldi PE, Deubel V, Guénet JL, Desprès P. A nonsense mutation in the gene encoding 2′-5′-oligoadenylate synthetase/L1 isoform is associated with West Nile virus susceptibility in laboratory mice. Proc Natl Acad Sci USA. 2002;99:11311–11316. doi: 10.1073/pnas.172195399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brown MG, Dokun AO, Heusel JW, Smith HRC, Beckman DL, Blattenberger EA, Dubbelde CE, Stone LR, Scalzo AA, Yokoyama WM. Vital involvement of a natural killer cell activation receptor in resistance to viral infection. Science. 2001;292:934–937. doi: 10.1126/science.1060042. [DOI] [PubMed] [Google Scholar]
- 5.Lee SH, Girard S, Macina D, Busa M, Zafer A, Belouchi A, Gros P, Vidal SM. Susceptibility to mouse cytomegalovirus is associated with deletion of an activating natural killer cell receptor of the C-type lectin superfamily. Nat Genet. 2001;28:42–45. doi: 10.1038/ng0501-42. [DOI] [PubMed] [Google Scholar]
- 6.Guida JD, Fejer G, Pirofski LA, Brosnan CF, Horwitz MS. Mouse adenovirus type 1 causes a fatal hemorrhagic encephalomyelitis in adult C57BL/6 but not BALB/c mice. J Virol. 1995;69:7674–7681. doi: 10.1128/jvi.69.12.7674-7681.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kring SC, King CS, Spindler KR. Susceptibility and signs associated with mouse adenovirus type 1 infection of adult outbred Swiss mice. J Virol. 1995;69:8084–8088. doi: 10.1128/jvi.69.12.8084-8088.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Spindler KR, Fang L, Moore ML, Brown CC, Hirsch GN, Kajon AK. SJL/J mice are highly susceptible to infection by mouse adenovirus type 1. J Virol. 2001;75:12039–12046. doi: 10.1128/JVI.75.24.12039-12046.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Spindler KR, Moore ML, Cauthen AN. The mouse in biomedical research. 2. Academic Press; New York: 2007. Mouse adenoviruses; pp. 49–65. [Google Scholar]
- 10.Weinberg JB, Stempfle GS, Wilkinson JE, Younger JG, Spindler KR. Acute respiratory infection with mouse adenovirus type 1. Virology. 2005;340:245–254. doi: 10.1016/j.virol.2005.06.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kajon AE, Brown CC, Spindler KR. Distribution of mouse adenovirus type 1 in intraperitoneally and intranasally infected adult outbred mice. J Virol. 1998;72:1219–1223. doi: 10.1128/jvi.72.2.1219-1223.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Smith K, Brown CC, Spindler KR. The role of mouse adenovirus type 1 early region 1A in acute and persistent infections in mice. J Virol. 1998;72:5699–5706. doi: 10.1128/jvi.72.7.5699-5706.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gralinski LE, Ashley SL, Dixon SD, Spindler KR. Mouse adenovirus type 1-induced breakdown of the blood-brain barrier. J Virol. 2009;83:9398–9410. doi: 10.1128/JVI.00954-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ashley SL, Welton AR, Harwood KM, Van Rooijen N, Spindler KR. Mouse adenovirus type 1 infection of macrophages. Virology. 2009;390:307–314. doi: 10.1016/j.virol.2009.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Moore ML, Brown CC, Spindler KR. T cells cause acute immunopathology and are required for long-term survival in mouse adenovirus type 1-induced encephalomyelitis. J Virol. 2003;77:10060–10070. doi: 10.1128/JVI.77.18.10060-10070.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Moore ML, McKissic EL, Brown CC, Wilkinson JE, Spindler KR. Fatal disseminated mouse adenovirus type 1 infection in mice lacking B cells or Bruton’s tyrosine kinase. J Virol. 2004;78:5584–5590. doi: 10.1128/JVI.78.11.5584-5590.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Welton AR, Chesler EJ, Sturkie C, Jackson AU, Hirsch GN, Spindler KR. Identification of quantitative trait loci for susceptibility to mouse adenovirus type 1. J Virol. 2005;79:11517–11522. doi: 10.1128/JVI.79.17.11517-11522.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bamezai A. Mouse Ly-6 proteins and their extended family: markers of cell differentiation and regulators of cell signaling. Arch Immunol Ther Exp (Warsz) 2004;52:255–266. [PubMed] [Google Scholar]
- 19.Liu HC, Niikura M, Fulton JE, Cheng HH. Identification of chicken lymphocyte antigen 6 complex, locus E (LY6E, alias SCA2) as a putative Marek’s disease resistance gene via a virus-host protein interaction screen. Cytogenet Genome Res. 2003;102:304–308. doi: 10.1159/000075767. [DOI] [PubMed] [Google Scholar]
- 20.Loeuillet C, Deutsch S, Ciuffi A, Robyr D, Taffe P, Munoz M, Beckmann JS, Antonarakis SE, Telenti A. In vitro whole-genome analysis identifies a susceptibility locus for HIV-1. PLoS Biol. 2008;6:e32. doi: 10.1371/journal.pbio.0060032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Brass AL, Dykxhoorn DM, Benita Y, Yan N, Engelman A, Xavier RJ, Lieberman J, Elledge SJ. Identification of host proteins required for HIV infection through a functional genomic screen. Science. 2008;319:921–926. doi: 10.1126/science.1152725. [DOI] [PubMed] [Google Scholar]
- 22.Krishnan MN, Ng A, Sukumaran B, Gilfoy FD, Uchil PD, Sultana H, Brass AL, Adametz R, Tsui M, Qian F, Montgomery RR, Lev S, Mason PW, Koski RA, Elledge SJ, et al. RNA interference screen for human genes associated with West Nile virus infection. Nature. 2008;455:242–245. doi: 10.1038/nature07207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Potter TA, I, McKenzie F, Morgan GM, Cherry M. Murine lymphocyte alloantigens. I. The Ly-6 locus. J Immunol. 1980;125:541–545. [PubMed] [Google Scholar]
- 24.Ortega G, Korty PE, Shevach EM, Malek TR. Role of Ly-6 in lymphocyte activation. I. Characterization of a monoclonal antibody to a nonpolymorphic Ly-6 specificity. J Immunol. 1986;137:3240–3246. [PubMed] [Google Scholar]
- 25.Horton MA, Sachs JA. Identity of murine lymphocyte alloantigens DAG, ALA-1, Ly-8, and Ly-6? Immunogenetics. 1979;9:273–280. [Google Scholar]
- 26.Ball AO, Beard CW, Villegas P, Spindler KR. Early region 4 sequence and biological comparison of two isolates of mouse adenovirus type 1. Virology. 1991;180:257–265. doi: 10.1016/0042-6822(91)90030-f. [DOI] [PubMed] [Google Scholar]
- 27.Cauthen AN, Welton AR, Spindler KR. Construction of mouse adenovirus type 1 mutants. Methods Mol Med. 2007;130:41–59. doi: 10.1385/1-59745-166-5:41. [DOI] [PubMed] [Google Scholar]
- 28.Welton AR, Spindler KR. Capture ELISA quantitation of mouse adenovirus type 1 in infected organs. Methods Mol Biol. 2007;130:215–221. doi: 10.1385/1-59745-166-5:215. [DOI] [PubMed] [Google Scholar]
- 29.Benson G. Tandem repeats finder: A program to analyze DNA sequences. Nuc Acids Res. 1999;27:573–580. doi: 10.1093/nar/27.2.573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pletcher MT, McClurg P, Batalov S, Su AI, Barnes SW, Lagler E, Korstanje R, Wang X, Nusskern D, Bogue MA, Mural RJ, Paigen B, Wiltshire T. Use of a dense single nucleotide polymorphism map for in silico mapping in the mouse. PLoS Biol. 2004;2:e393. doi: 10.1371/journal.pbio.0020393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mural RJ, Adams MD, Myers EW, Smith HO, Miklos GL, Wides R, Halpern A, Li PW, Sutton GG, Nadeau J, Salzberg SL, Holt RA, Kodira CD, Lu F, Chen L, et al. A comparison of whole-genome shotgun-derived mouse chromosome 16 and the human genome. Science. 2002;296:1661–1671. doi: 10.1126/science.1069193. [DOI] [PubMed] [Google Scholar]
- 32.Schauwecker PE, Williams RW, Santos JB. Genetic control of sensitivity to hippocampal cell death induced by kainic acid: A quantitative trait loci analysis. J Comp Neurol. 2004;477:96–107. doi: 10.1002/cne.20245. [DOI] [PubMed] [Google Scholar]
- 33.Pflugh DL, Maher SE, Bothwell ALM. Ly-6I, a new member of the murine Ly-6 superfamily with a distinct pattern of expression. J Immunol. 2000;165:313–321. doi: 10.4049/jimmunol.165.1.313. [DOI] [PubMed] [Google Scholar]
- 34.Patterson JM, Johnson MH, Zimonjic DB, Graubert TA. Characterization of Ly-6M, a novel member of the Ly-6 family of hematopoietic proteins. Blood. 2000;95:3125–3132. [PubMed] [Google Scholar]
- 35.Uchida N, Aguila HL, Fleming WH, Jerabek L, Weissman IL. Rapid and sustained hematopoietic recovery in lethally irradiated mice transplanted with purified Thy-1.1lo Lin-Sca-1+ hematopoietic stem cells. Blood. 1994;83:3758–3779. [PubMed] [Google Scholar]
- 36.Geissmann F, Auffray C, Palframan R, Wirrig C, Ciocca A, Campisi L, Narni-Mancinelli E, Lauvau G. Blood monocytes: Distinct subsets, how they relate to dendritic cells, and their possible roles in the regulation of T-cell responses. Immunol Cell Biol. 2008;86:398–408. doi: 10.1038/icb.2008.19. [DOI] [PubMed] [Google Scholar]
- 37.Takikawa O, Oku T, Ito N, Ushio Y, Yamamoto N, Yoneda Y, Tsuji J, Sanchez-Bueno A, Verkhusha V, Yoshida R. Multiple expression of Ly-6C and accumulation of a Ly-6C pre-mRNA in activated macrophages involved in rejection of an allografted tumor. Biochem Biophys Res Commun. 1996;226:247–253. doi: 10.1006/bbrc.1996.1341. [DOI] [PubMed] [Google Scholar]
- 38.Palfree RG, Sirlin S, Dumont FJ, Hammerling U. N-terminal and cDNA characterization of murine lymphocyte antigen Ly-6C.2. J Immunol. 1988;140:305–310. [PubMed] [Google Scholar]
- 39.Fleming TJ, O’HUigin C, Malek TR. Characterization of two novel Ly-6 genes. Protein sequence and potential structural similarity to alpha-bungarotoxin and other neurotoxins. J Immunol. 1993;150:5379–5390. [PubMed] [Google Scholar]
- 40.Apostolopoulos J, Chisholm LJ, Sandrin MS. Identification of mouse Ly6H and its expression in normal tissue. Immunogenetics. 1999;49:987–990. doi: 10.1007/s002510050583. [DOI] [PubMed] [Google Scholar]
- 41.Kitazawa H, Nishihara T, Nambu T, Nishizawa H, Iwaki M, Fukuhara A, Kitamura T, Matsuda M, Shimomura I. Intectin, a novel small intestine-specific glycosylphosphatidylinositol-anchored protein, accelerates apoptosis of intestinal epithelial cells. J Biol Chem. 2004;279:42867–42874. doi: 10.1074/jbc.M408047200. [DOI] [PubMed] [Google Scholar]
- 42.Ioka RX, Kang MJ, Kamiyama S, Kim DH, Magoori K, Kamataki A, Ito Y, Takei YA, Sasaki M, Suzuki T, Sasano H, Takahashi S, Sakai J, Fujino T, Yamamoto TT. Expression cloning and characterization of a novel glycosylphosphatidylinositol-anchored high density lipoprotein-binding protein, GPI-HBP1. J Biol Chem. 2003;278:7344–7349. doi: 10.1074/jbc.M211932200. [DOI] [PubMed] [Google Scholar]
- 43.Bult CJ, Eppig JT, Kadin JA, Richardson JE, Blake JA, Group MGD. The mouse genome database (MGD): Mouse biology and model systems. Nuc Acids Res. 2008;36(Database issue):D724–D728. doi: 10.1093/nar/gkm961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ploug M, Ellis V. Structure-function relationships in the receptor for urokinase-type plasminogen activator. Comparison to other members of the Ly-6 family and snake venom alpha-neurotoxins. FEBS Lett. 1994;349:163–168. doi: 10.1016/0014-5793(94)00674-1. [DOI] [PubMed] [Google Scholar]
- 45.Levitin F, Weiss M, Hahn Y, Stern O, Papke RL, Matusik R, Nandana SR, Ziv R, Pichinuk E, Salame S, Bera T, Vincent J, Lee B, Pastan I, Wreschner DH. PATE gene clusters code for multiple, secreted TFP/Ly-6/uPAR proteins that are expressed in reproductive and neuron-rich tissues and possess neuromodulatory activity. J Biol Chem. 2008;283:16928–16939. doi: 10.1074/jbc.M801454200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Rock KL, Reiser H, Bamezai A, McGrew J, Benacerraf B. The LY-6 locus: A multigene family encoding phosphatidylinositol-anchored membrane proteins concerned with T-cell activation. Immunol Rev. 1989;111:195–224. doi: 10.1111/j.1600-065x.1989.tb00547.x. [DOI] [PubMed] [Google Scholar]
- 47.McGrew JT, Rock KL. Isolation, expression, and sequence of the TAP/Ly-6A.2 chromosomal gene. J Immunol. 1991;146:3633–3638. [PubMed] [Google Scholar]
- 48.Gumley TP, I, McKenzie FC, Sandrin MS. Tissue expression, structure and function of the murine Ly-6 family of molecules. Immunol Cell Biol. 1995;73:277–296. doi: 10.1038/icb.1995.45. [DOI] [PubMed] [Google Scholar]
- 49.Kimura S, Tada N, Liu-Lam Y, Hammerling U. Studies of the mouse Ly-6 alloantigen system. II. Complexities of the Ly-6 region. Immunogenetics. 1984;20:47–56. doi: 10.1007/BF00373446. [DOI] [PubMed] [Google Scholar]
- 50.Bothwell A, Pace PE, LeClair KP. Isolation and expression of an IFN-responsive Ly-6C chromosomal gene. J Immunol. 1988;140:2815–2820. [PubMed] [Google Scholar]
- 51.Schlueter AJ, Malek TR, Hostetler CN, Smith PA, deVries P, Waldschmidt TJ. Distribution of Ly-6C on lymphocyte subsets: I. Influence of allotype on T lymphocyte expression. J Immunol. 1997;158:4211–4222. [PubMed] [Google Scholar]
- 52.Fleming TJ, Fleming ML, Malek TR. Selective expression of Ly-6G on myeloid lineage cells in mouse bone marrow. RB6-8C5 mAb to granulocyte-differentiation antigen (Gr-1) detects members of the Ly-6 family. J Immunol. 1993;151:2399–2408. [PubMed] [Google Scholar]
- 53.Nagendra S, Schlueter AJ. Absence of cross-reactivity between murine Ly-6C and Ly-6G. Cytometry A. 2004;58:195–200. doi: 10.1002/cyto.a.20007. [DOI] [PubMed] [Google Scholar]
- 54.Galat A. The three-fingered protein domain of the human genome. Cell Mol Life Sci. 2008;65:3481–3493. doi: 10.1007/s00018-008-8473-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Holmes C, Stanford WL. Concise review: Stem cell antigen-1: Expression, function, and enigma. Stem Cells. 2007;25:1339–1347. doi: 10.1634/stemcells.2006-0644. [DOI] [PubMed] [Google Scholar]
- 56.Alliot F, Rutin J, Pessac B. Ly-6C is expressed in brain vessels endothelial cells but not in microglia of the mouse. Neurosci Lett. 1998;251:37–40. doi: 10.1016/s0304-3940(98)00473-x. [DOI] [PubMed] [Google Scholar]
- 57.van de Rijn M, Heimfeld S, Spangrude GJ, Weissman IL. Mouse hematopoietic stem-cell antigen Sca-1 is a member of the Ly-6 antigen family. Proc Natl Acad Sci USA. 1989;86:4634–4638. doi: 10.1073/pnas.86.12.4634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cray C, Keane RW, Malek TR, Levy RB. Regulation and selective expression of Ly-6A/E, a lymphocyte activation molecule, in the central nervous system. Brain Res Mol Brain Res. 1990;8:9–15. doi: 10.1016/0169-328x(90)90003-v. [DOI] [PubMed] [Google Scholar]
- 59.Classon BJ, Coverdale L. Mouse stem cell antigen Sca-2 is a member of the Ly-6 family of cell surface proteins. Proc Natl Acad Sci USA. 1994;91:5296–5300. doi: 10.1073/pnas.91.12.5296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.MacNeil I, Kennedy J, Godfrey DI, Jenkins NA, Masciantonio M, Mineo C, Gilbert DJ, Copeland NG, Boyd RL, Zlotnik A. Isolation of a cDNA encoding thymic shared antigen-1. A new member of the Ly6 family with a possible role in T cell development. J Immunol. 1993;151:6913–6923. [PubMed] [Google Scholar]
- 61.Whitmire JK, Eam B, Whitton JL. Mice deficient in stem cell antigen-1 (Sca1, Ly-6A/E) develop normal primary and memory CD4+ and CD8+ T-cell responses to virus infection. Eur J Immunol. 2009;39:1494–1504. doi: 10.1002/eji.200838959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Drevets DA, Schawang JE, Dillon MJ, Lerner MR, Bronze MS, Brackett DJ. Innate responses to systemic infection by intracellular bacteria trigger recruitment of Ly-6Chigh monocytes to the brain. J Immunol. 2008;181:529–536. doi: 10.4049/jimmunol.181.1.529. [DOI] [PubMed] [Google Scholar]
- 63.Getts DR, Terry RL, Getts MT, Muller M, Rana S, Shrestha B, Radford J, Van Rooijen N, Campbell IL, King NJ. Ly6c+ “inflammatory monocytes” are microglial precursors recruited in a pathogenic manner in West Nile virus encephalitis. J Exp Med. 2008;205:2319–2337. doi: 10.1084/jem.20080421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Dunay IR, Damatta RA, Fux B, Presti R, Greco S, Colonna M, Sibley LD. Gr1+ inflammatory monocytes are required for mucosal resistance to the pathogen Toxoplasma gondii. Immunity. 2008;29:306–317. doi: 10.1016/j.immuni.2008.05.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Tacke F, Ginhoux F, Jakubzick C, van Rooijen N, Merad M, Randolph GJ. Immature monocytes acquire antigens from other cells in the bone marrow and present them to T cells after maturing in the periphery. J Exp Med. 2006;203:583–597. doi: 10.1084/jem.20052119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Khodadoust MM, Khan KD, Bothwell AL. Complex regulation of Ly-6E gene transcription in T cells by IFNs. J Immunol. 1999;163:811–819. [PubMed] [Google Scholar]
- 67.Dumont FJ, Dijkmans R, Palfree RG, Boltz RD, Coker L. Selective up-regulation by interferon-γ of surface molecules of the Ly-6 complex in resting T cells: the Ly-6A/E and TAP antigens are preferentially enhanced. Eur J Immunol. 1987;17:1183–1191. doi: 10.1002/eji.1830170816. [DOI] [PubMed] [Google Scholar]
- 68.Dumont FJ, Palfree RG, Coker LZ. Phenotypic changes induced by interferon in resting T cells: major enhancement of Ly-6 antigen expression. J Immunol. 1986;137:201–210. [PubMed] [Google Scholar]
- 69.Blake PG, Madrenas J, Halloran PF. Ly-6 in kidney is widely expressed on tubular epithelium and vascular endothelium and is up-regulated by interferon gamma. J Am Soc Nephrol. 1993;4:1140–1150. doi: 10.1681/ASN.V451140. [DOI] [PubMed] [Google Scholar]
- 70.Flanagan K, Modrusan Z, Cornelius J, Chavali A, Kasman I, Komuves L, Mo L, Diehl L. Intestinal epithelial cell up-regulation of LY6 molecules during colitis results in enhanced chemokine secretion. J Immunol. 2008;180:3874–3881. doi: 10.4049/jimmunol.180.6.3874. [DOI] [PubMed] [Google Scholar]
- 71.Morgan RW, Sofer L, Anderson AS, Bernberg EL, Cui J, Burnside J. Induction of host gene expression following infection of chicken embryo fibroblasts with oncogenic Marek’s disease virus. J Virol. 2001;75:533–539. doi: 10.1128/JVI.75.1.533-539.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]