Abstract
Background:
Bisphosphanates are used primarily for the prevention and treatment of osteoporosis, and are also indicated for osseous complications of malignancy. In addition to their bone resorption properties, the most commonly used nitrogen-containing bisphosphonate compounds also inhibit protein prenylation, and thus may exert anti-tumour properties.
Methods:
To evaluate whether the use of these drugs may be associated with cancer, specifically breast cancer, we conducted a population-based case–control study in Wisconsin from 2003 to 2006. Participants included 2936 incident invasive breast cancer cases and 2975 population controls aged <70 years. Bisphosphonate use and potential confounders were assessed by interview.
Results:
Using multivariable logistic regression, the odds ratio for breast cancer in current bisphosphonate users compared with non-users was 0.67 (95% confidence interval 0.51–0.89). Increasing duration of use was associated with a greater reduction in risk (P-trend=0.01). Risk reduction was observed in women who were not obese (P-interaction=0.005).
Conclusion:
These results are suggestive of an additional benefit of the common use of bisphosphonates, in this instance, the reduction in breast cancer risk.
Keywords: bisphosphonates, osteoporosis, breast cancer, case–control
Bisphosphonates are commonly prescribed for the prevention and treatment of osteoporosis (Rosen, 2005). By inhibiting bone resorption, bisphosphonate therapy also reduces skeletal complications from bone involvement in breast, prostate, and other cancers, and from the bone-depleting consequences of some treatments (Berenson et al, 1996; Aapro et al, 2008; Saad et al, 2008). In addition to the reduction in osteoclastic bone resorption, the more potent aminobisphosphonates inhibit the mevalonate pathway, thereby affecting cell function and survival(Luckman et al, 1998; Dunford et al, 2001). As this pathway leads to the prenylation of key signalling proteins required by all cells, aminobisphosphonates may affect the viability of other cell types, such as tumours (Russell et al, 1999; Caraglia et al, 2006). Nitrogen-containing bisphosphonates have also been shown to directly induce tumour apoptosis, inhibit angiogenesis, and prevent tumour cell adhesion (Neville-Webbe et al, 2002; Wood et al, 2002). These anti-proliferative actions may be relevant to the development of a broad range of cancers, including breast neoplasms(Green, 2003; Winter et al, 2008). We examined the association between bisphosphonate use and breast cancer in a population-based case–control study.
Materials and methods
Women aged 20–69 years with a new diagnosis of invasive breast cancer from 2003 to 2006 were identified from Wisconsin's mandatory cancer registry (Sprague et al, 2008; Wernli et al, 2009). For comparison, similarly aged community women without a personal history of breast cancer were randomly selected from driver's licence lists. The study included 2988 breast cancer cases (74% of eligible) and 3004 controls (67% of eligible). Women were invited to participate in a structured interview to ascertain the use of bisphosphonates, type of drug, duration and recency of use, as well as physician diagnosis of osteoporosis, history of fractures, height change since the age of 18 years, and other risk factors for breast cancer. Exposure information was limited to events before a reference date that was defined as the date of diagnosis for cases and, for comparability, the date approximately 1 year before interview for control women. The study was approved by the University of Wisconsin-Madison institutional review board.
Multivariable logistic regression models were used to estimate odds ratios and 95% confidence intervals and tests for linear trend across ordinal values of categorical variables. Current use of bisphosphonates was defined as use in the year before the reference year, and former users had taken bisphosphonates at any time before this period. Duration of use was the sum of all periods of bisphosphonate use in the year before the reference period. Analyses were adjusted for potentially confounding variables selected a priori: age at reference date, year of interview, parity (0–1, 2, 3, 4+), age at first birth (nulliparous, <20, 20–24, 25–29, 30+), first-degree family history of breast cancer, body mass index 1 year before reference date (<24.9, 25–29.9, ⩾30 kg m–2), menopausal status, age at menopause (<45, 45–49, 50–54, 55+), screening mammograms over the past 5 years (0, 1–4, ⩾5), a physician diagnosis of osteoporosis, smoking status, and height change from the age of 18 years. Effect modification was evaluated by inclusion of cross-product interaction terms in logistic models. Owing to missing or incomplete information on osteoporosis medications or their indications, 26 breast cancer cases and 29 control women were excluded, leaving 2936 cases and 2975 controls for analysis.
Results
The average age at the reference date for breast cancer cases was 54.2 years (s.d.=8.9 years) and for controls it was 54.8 years (8.9 years). As expected, women with breast cancer were more likely than controls to have a family history of breast cancer, a college degree, to be older at first birth or nulliparous, to have gained more weight since the age of 18 years, and to use postmenopausal hormones; controls were more likely to report a physician's diagnosis of osteoporosis (data not shown).
Bisphosphonates were more commonly used by controls than by cases; the multivariate-adjusted odds ratio for ever use was 0.70 (95% confidence interval 0.54–0.92; Table 1), which was similar to the age-adjusted odds ratio, suggesting that the influence of confounding was limited. The reduction in risk was significant only among current users of bisphosphonates, although few women were former users at the reference date. Increasing duration of use was associated with greater reductions in breast cancer risk (P-trend=0.01).
Table 1. Odds ratios (ORs) (95% confidence intervals, CIs) of breast cancer associated with bisphosphonate use.
| Cases (N=2936) | Controls (N=2975) | Age adjusted | Multivariable adjusted | |
|---|---|---|---|---|
| Bisphosphonate use | N (%) | N (%) | OR (95% CI)a | OR (95% CI)b |
| Never use | 2808 (95.6) | 2788 (93.7) | 1.00 (Referent) | 1.00 (Referent) |
| Ever use | 128 (4.4) | 187 (6.2) | 0.70 (0.56–0.89) | 0.70 (0.54–0.92) |
| Former | 22 (0.7) | 26 (0.9) | 0.85 (0.48–1.51) | 0.91 (0.50–1.64) |
| Current | 106 (3.6) | 161 (5.4) | 0.68 (0.53–0.88) | 0.67 (0.51–0.89) |
| Duration of use (months) | ||||
| 3–12 | 58 (2.0) | 76 (2.5) | 0.78 (0.55–1.11) | 0.78 (0.54–1.12) |
| 13–24 | 26 (0.9) | 37 (1.2) | 0.72 (0.44–1.20) | 0.69 (0.41–1.17) |
| ⩾25 | 44 (1.5) | 74 (2.5) | 0.61 (0.42–0.90) | 0.63 (0.42–0.95) |
| P-trend | 0.01 | 0.01 | ||
Adjusted for age at diagnosis.
Additionally adjusted for parity, age at first live birth, family history of breast cancer, body mass index, menopausal status, age at menopause, type of hormone use, mammography, osteoporosis, smoking history, and height change.
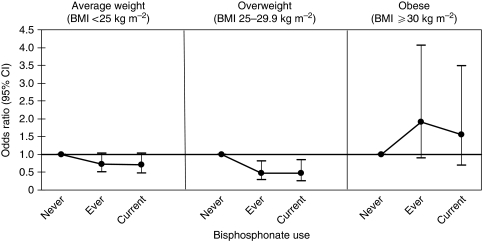
To evaluate whether the inverse effects of bisphosphonates were attributable to the medications themselves or to the conditions for which they were prescribed, we examined use according to fracture history (excluding women diagnosed with distant disease), height loss, and physician-diagnosed osteoporosis. There was a suggestion that use of bisphosphonates was associated with a reduced risk of breast cancer only among women reporting symptoms of bone loss (postmenopausal fractures, osteoporosis, and height loss), but the differences were not statistically significant (P-interaction=0.29). The inverse effect of bisphosphonates seemed limited to women who were not obese (P-interaction=0.005, Figure 1). There were no statistical interactions between bisphosphonates and age, smoking, or hormone therapy (data not shown).
Figure 1.
Odds ratios and 95% confidence intervals of breast cancer according to bisphosphonate use and body mass index (BMI). Odds ratios were adjusted for age at diagnosis, party, age at first live birth, family history of breast cancer, BMI menopausal status, age at menopause, type of hormone use, mammography, osteoporosis, smoking history, and height change. P-interaction for BMI and use of bisphosphonates=0.005.
Conclusion
In this case–control study, use of bisphosphonates was associated with approximately a 30% reduction in breast cancer risk, and this decrease in risk was greatest for longer durations of use, and among leaner women. This association was not attributed to the primary indication for use, bone density loss, or fractures, which we and others have previously shown as a risk factor for breast cancer (Cauley et al, 1996; Newcomb et al, 2001; Silverman et al, 2004). Our confidence in these results is strengthened by the large size of the study, the population-based sampling, our consideration of important confounders such as body mass index and postmenopausal hormone use, and the generally high reliability and validity of self-reports of medication use (Lipworth et al, 2001; Boudreau et al, 2004, 2005).
There are limited data that directly support our epidemiological findings. However, a number of observations may be relevant to a protective effect of these medications on breast cancer incidence. In animals bearing breast cancer xenografts, bisphosphonates block the growth of bone metastases, and also extraskeletal disease (Senaratne et al, 2000; Hiraga et al, 2001). Indeed, direct cytotoxicity seen in vitro (Fromigue et al, 2000) may account for some of the clinical benefit in patients taking bisphosphonates, specifically aminobisphosphonates (Neville-Webbe et al, 2002; Green, 2003; Caraglia et al, 2004). Although the molecular targets of bisphosphonates are not fully known, several phases of tumour growth and progression seem to be influenced (Neville-Webbe et al, 2002; Wood et al, 2002; Caraglia et al, 2006). In a recent clinical trial of early-stage breast cancer, nitrogen-containing bisphosphonates seemed to improve long-term outcomes (Gnant et al, 2009), and several large clinical trials are currently underway to clarify this issue (Bedard et al, 2009). Our finding that bisphosphonates were not associated with breast cancer risk reductions in obese women suggests that their inhibitory actions may be related to some threshold effect of hormonal or other growth factors; these are important aetiological exposures in breast cancer (Hankinson et al, 2004). Thus, the pluripotent actions of bisphosphonates are consistent with our observed findings.
Unmeasured or unknown confounding factors may have been present, for which adjustment was not possible. Although we inquired about clinical symptoms and reports of physician-diagnosed osteoporosis, bone mineral density was not available. Nearly all users had used aminobisphosphonates (97% of users); therefore, we could not evaluate specific types of preparations that have very different mechanisms of action (Russell and Rogers, 1999; Bedard et al, 2009). Sample size was also too modest to evaluate other breast cancer subgroups that may experience different responses to bisphosphonates, such as specific histological type or hormone receptor status. Finally, because the study population was limited to women under the age of 70 years, the prevalence of bisphosphonate use was lower than might be expected in older women with clinical osteoporosis.
In summary, this study provides new evidence that the use of bisphosphonates is associated with a potentially important reduction in breast cancer risk. Because we were able to account for important confounders, these findings may reflect real benefits due to the anti-tumour mechanisms of these medications.
Acknowledgments
This work was supported by the National Cancer Institute at the National Institutes of Health (R01 CA47147). The authors are grateful to Drs Henry Anderson, Patrick L Remington, Meir J Stampfer, Walter C Willett, John A Baron, Kathleen Egan, Linda Titus-Ernstoff, John A Baron, E Robert Greenberg, and Richard R Love for their contributions in planning and conduct at various stages of this study; Laura Stephenson and the Wisconsin Cancer Reporting System, and the Women's Health Study staff, especially Julie McGregor, for generous assistance with study management.
References
- Aapro M, Abrahamsson PA, Body JJ, Coleman RE, Colomer R, Costa L, Crino L, Dirix L, Gnant M, Gralow J, Hadji P, Hortobagyi GN, Jonat W, Lipton A, Monnier A, Paterson AH, Rizzoli R, Saad F, Thurlimann B (2008) Guidance on the use of bisphosphonates in solid tumours: recommendations of an international expert panel. Ann Oncol 19: 420–432 [DOI] [PubMed] [Google Scholar]
- Bedard PL, Body JJ, Piccart-Gebhart MJ (2009) Sowing the soil for cure? Results of the ABCSG-12 trial open a new chapter in the evolving adjuvant bisphosphonate story in early breast cancer. J Clin Oncol 27: 4043–4046 [DOI] [PubMed] [Google Scholar]
- Berenson JR, Lichtenstein A, Porter L, Dimopoulos MA, Bordoni R, George S, Lipton A, Keller A, Ballester O, Kovacs MJ, Blacklock HA, Bell R, Simeone J, Reitsma DJ, Heffernan M, Seaman J, Knight RD (1996) Efficacy of pamidronate in reducing skeletal events in patients with advanced multiple myeloma. Myeloma Aredia Study Group. N Engl J Med 334: 488–493 [DOI] [PubMed] [Google Scholar]
- Boudreau DM, Daling JR, Malone KE, Gardner JS, Blough DK, Heckbert SR (2004) A validation study of patient interview data and pharmacy records for antihypertensive, statin, and antidepressant medication use among older women. Am J Epidemiol 159: 308–317 [DOI] [PubMed] [Google Scholar]
- Boudreau DM, Doescher MP, Saver BG, Jackson JE, Fishman PA (2005) Reliability of Group Health Cooperative automated pharmacy data by drug benefit status. Pharmacoepidemiol Drug Saf 14: 877–884 [DOI] [PubMed] [Google Scholar]
- Caraglia M, D’Alessandro AM, Marra M, Giuberti G, Vitale G, Viscomi C, Colao A, Prete SD, Tagliaferri P, Tassone P, Budillon A, Venuta S, Abbruzzese A (2004) The farnesyl transferase inhibitor R115777 (Zarnestra) synergistically enhances growth inhibition and apoptosis induced on epidermoid cancer cells by Zoledronic acid (Zometa) and Pamidronate. Oncogene 23: 6900–6913 [DOI] [PubMed] [Google Scholar]
- Caraglia M, Santini D, Marra M, Vincenzi B, Tonini G, Budillon A (2006) Emerging anti-cancer molecular mechanisms of aminobisphosphonates. Endocr Relat Cancer 13: 7–26 [DOI] [PubMed] [Google Scholar]
- Cauley JA, Lucas FL, Kuller LH, Vogt MT, Browner WS, Cummings SR (1996) Bone mineral density and risk of breast cancer in older women: the study of osteoporotic fractures. Study of Osteoporotic Fractures Research Group. JAMA 276: 1404–1408 [PubMed] [Google Scholar]
- Dunford JE, Thompson K, Coxon FP, Luckman SP, Hahn FM, Poulter CD, Ebetino FH, Rogers MJ (2001) Structure-activity relationships for inhibition of farnesyl diphosphate synthase in vitro and inhibition of bone resorption in vivo by nitrogen-containing bisphosphonates. J Pharmacol Exp Ther 296: 235–242 [PubMed] [Google Scholar]
- Fromigue O, Lagneaux L, Body JJ (2000) Bisphosphonates induce breast cancer cell death in vitro. J Bone Miner Res 15: 2211–2221 [DOI] [PubMed] [Google Scholar]
- Gnant M, Mlineritsch B, Schippinger W, Luschin-Ebengreuth G, Postlberger S, Menzel C, Jakesz R, Seifert M, Hubalek M, Bjelic-Radisic V, Samonigg H, Tausch C, Eidtmann H, Steger G, Kwasny W, Dubsky P, Fridrik M, Fitzal F, Stierer M, Rucklinger E, Greil R, Marth C (2009) Endocrine therapy plus zoledronic acid in premenopausal breast cancer. N Engl J Med 360: 679–691 [DOI] [PubMed] [Google Scholar]
- Green JR (2003) Antitumor effects of bisphosphonates. Cancer 97: 840–847 [DOI] [PubMed] [Google Scholar]
- Hankinson SE, Colditz GA, Willett WC (2004) Towards an integrated model for breast cancer etiology: the lifelong interplay of genes, lifestyle, and hormones. Breast Cancer Res 6: 213–218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hiraga T, Williams PJ, Mundy GR, Yoneda T (2001) The bisphosphonate ibandronate promotes apoptosis in MDA-MB-231 human breast cancer cells in bone metastases. Cancer Res 61: 4418–4424 [PubMed] [Google Scholar]
- Lipworth L, Fryzek J, Fored C, Blot W, McLaughlin J (2001) Comparison of surrogate with self-respondents regarding medical history and prior medication use. Int J Epidemiol 30: 303–308 [DOI] [PubMed] [Google Scholar]
- Luckman SP, Hughes DE, Coxon FP, Graham R, Russell G, Rogers MJ (1998) Nitrogen-containing bisphosphonates inhibit the mevalonate pathway and prevent post-translational prenylation of GTP-binding proteins, including Ras. J Bone Miner Res 13: 581–589 [DOI] [PubMed] [Google Scholar]
- Neville-Webbe HL, Holen I, Coleman RE (2002) The anti-tumour activity of bisphosphonates. Cancer Treat Rev 28: 305–319 [DOI] [PubMed] [Google Scholar]
- Newcomb PA, Trentham-Dietz A, Egan KM, Titus-Ernstoff L, Baron JA, Storer BE, Willett WC, Stampfer MJ (2001) Fracture history and risk of breast and endometrial cancer. Am J Epidemiol 153: 1071–1078 [DOI] [PubMed] [Google Scholar]
- Rosen CJ (2005) Clinical practice. Postmenopausal osteoporosis. N Engl J Med 353: 595–603 [DOI] [PubMed] [Google Scholar]
- Russell RG, Rogers MJ (1999) Bisphosphonates: from the laboratory to the clinic and back again. Bone 25: 97–106 [DOI] [PubMed] [Google Scholar]
- Russell RG, Rogers MJ, Frith JC, Luckman SP, Coxon FP, Benford HL, Croucher PI, Shipman C, Fleisch HA (1999) The pharmacology of bisphosphonates and new insights into their mechanisms of action. J Bone Miner Res 14(Suppl 2): 53–65 [DOI] [PubMed] [Google Scholar]
- Saad F, Adachi JD, Brown JP, Canning LA, Gelmon KA, Josse RG, Pritchard KI (2008) Cancer treatment-induced bone loss in breast and prostate cancer. J Clin Oncol 26: 5465–5476 [DOI] [PubMed] [Google Scholar]
- Senaratne SG, Pirianov G, Mansi JL, Arnett TR, Colston KW (2000) Bisphosphonates induce apoptosis in human breast cancer cell lines. Br J Cancer 82: 1459–1468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverman SL, Delmas PD, Kulkarni PM, Stock JL, Wong M, Plouffe Jr L (2004) Comparison of fracture, cardiovascular event, and breast cancer rates at 3 years in postmenopausal women with osteoporosis. J Am Geriatr Soc 52: 1543–1548 [DOI] [PubMed] [Google Scholar]
- Sprague BL, Trentham-Dietz A, Egan KM, Titus-Ernstoff L, Hampton JM, Newcomb PA (2008) Proportion of invasive breast cancer attributable to risk factors modifiable after menopause. Am J Epidemiol 168: 404–411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wernli KJ, Hampton JM, Trentham-Dietz A, Newcomb PA (2009) Antidepressant medication use and breast cancer risk. Pharmacoepidemiol Drug Saf 18: 284–290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winter MC, Holen I, Coleman RE (2008) Exploring the anti-tumour activity of bisphosphonates in early breast cancer. Cancer Treat Rev 34: 453–475 [DOI] [PubMed] [Google Scholar]
- Wood J, Bonjean K, Ruetz S, Bellahcene A, Devy L, Foidart JM, Castronovo V, Green JR (2002) Novel antiangiogenic effects of the bisphosphonate compound zoledronic acid. J Pharmacol Exp Ther 302: 1055–1061 [DOI] [PubMed] [Google Scholar]