Abstract
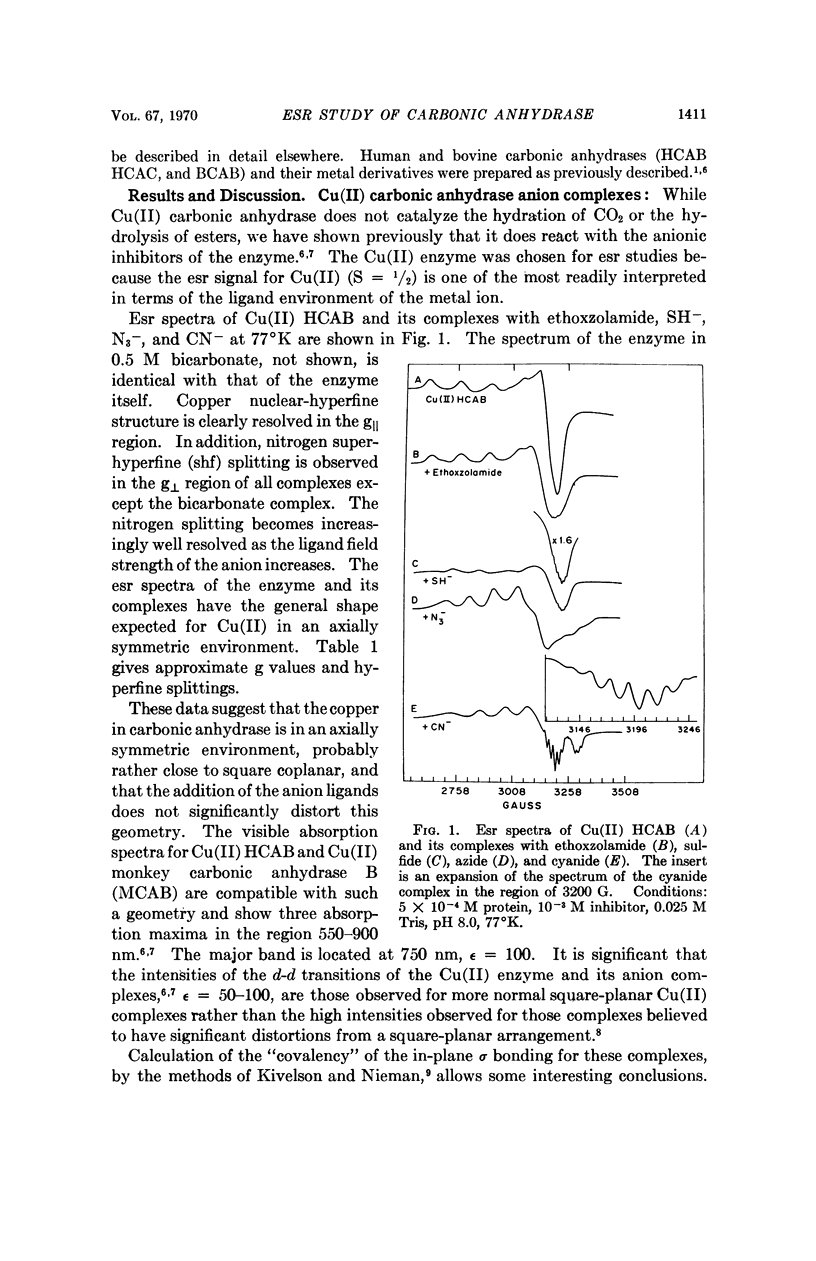
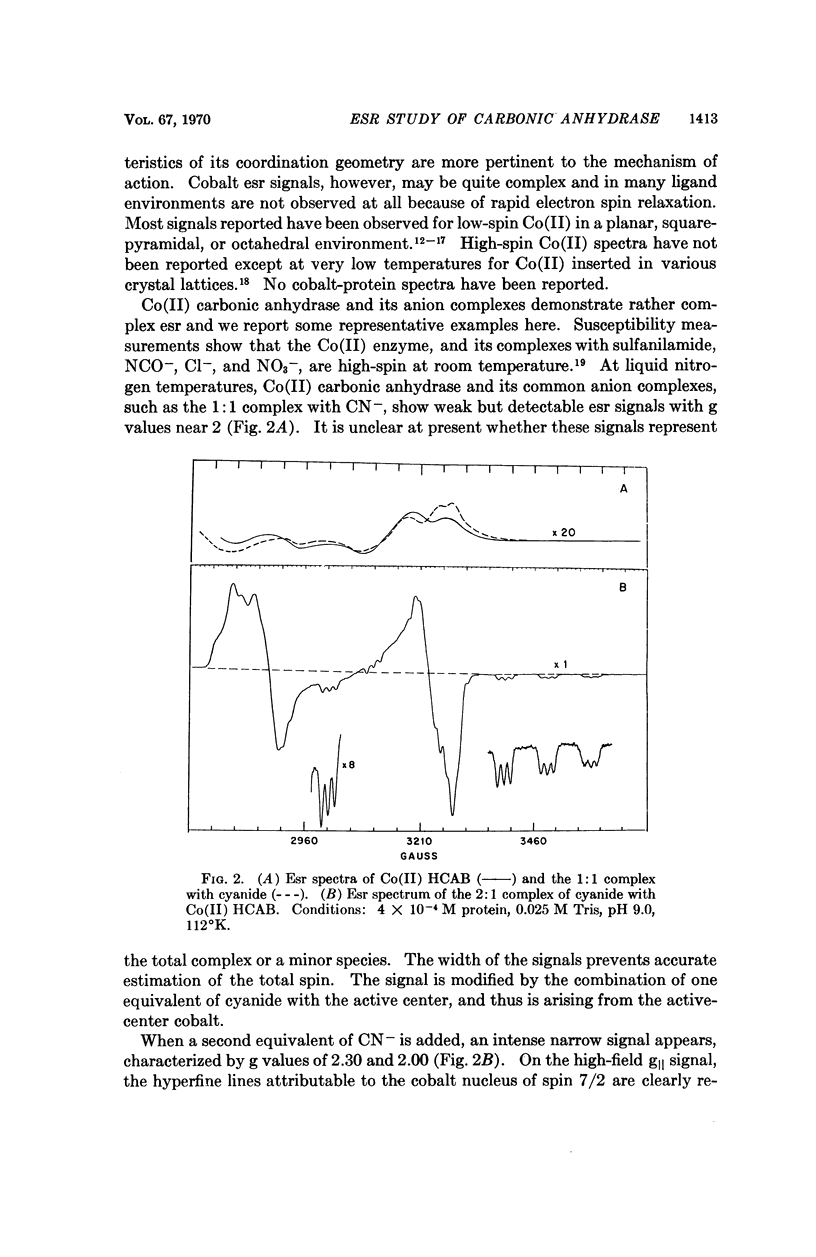
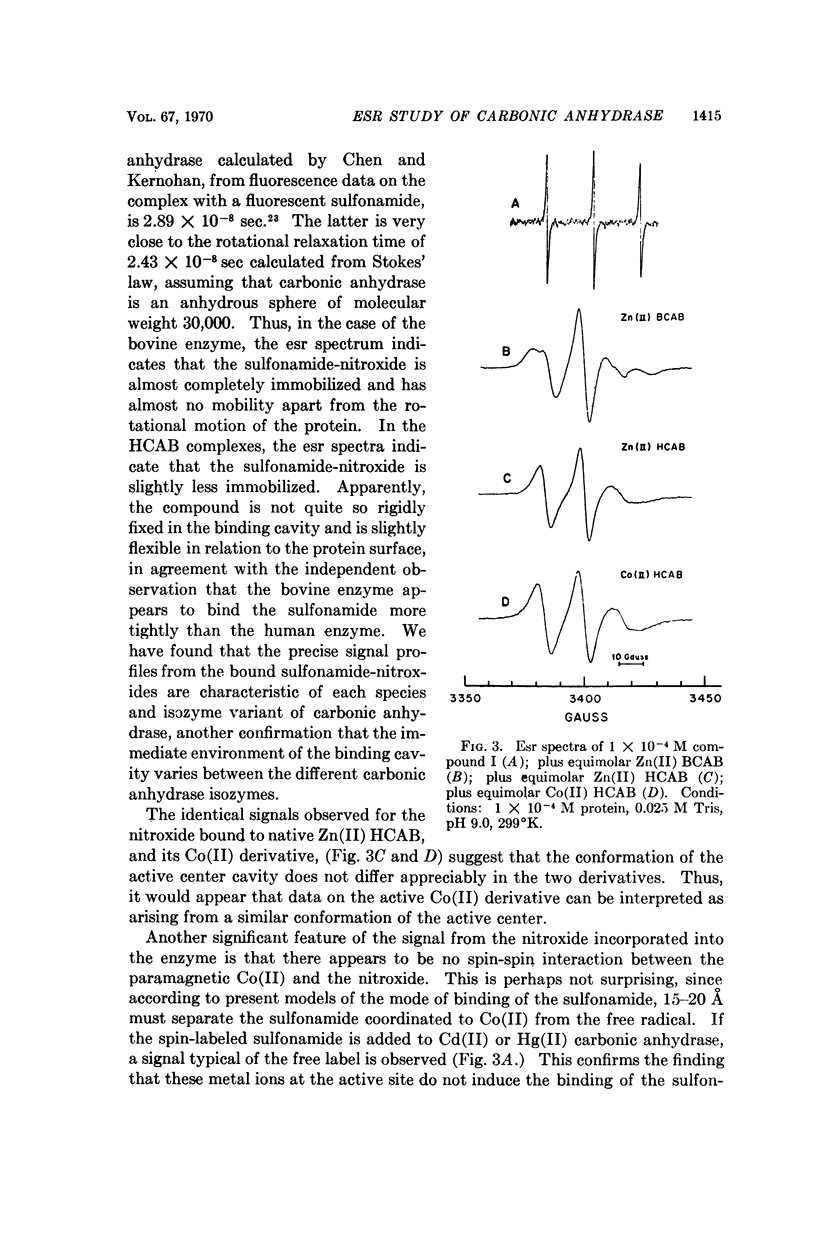
Electron spin resonance (esr) spectra of Cu(II) and Co(II) carbonic anhydrase, and a spin-labeled sulfonamide complex of the Zn(II) enzyme, are reported. The coordination geometry of Cu(II) bound in the enzyme appears to have approximately axial symmetry. Esr spectra of enzyme complexes with metal-binding anions also show axial symmetry and greater covalency, in the order ethoxzolamide < SH- < N3- ≤ CN-. Well-resolved superhyperfine structure in the spectrum of the cyanide complex suggests the presence of two, and probably three, equivalent nitrogen ligands from the protein. Esr spectra of the Co(II) enzyme and its complexes show two types of Co(II) environment, one typical of the native enzyme and the 1:1 CN- complex, and one typical of a 2:1 CN- complex. Co(II) in the 2:1 complex appears to be low-spin and probably has a coordination number of 5. Binding of a spin-labeled sulfonamide to the active center immobilizes the free radical. The similarity of the esr spectra of spin-labeled Zn(II) and Co(II) carbonic anhydrases suggests that the conformation at the active center is similar in the two metal derivatives.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BLUMBERG W. E., PEISACH J. AN ELECTRON SPIN RESONANCE STUDY OF COPPER UROPORPHYRIN 3 AND OTHER TOURACO FEATHER COMPONENTS. J Biol Chem. 1965 Feb;240:870–876. [PubMed] [Google Scholar]
- Chen R. F., Kernohan J. C. Combination of bovine carbonic anhydrase with a fluorescent sulfonamide. J Biol Chem. 1967 Dec 25;242(24):5813–5823. [PubMed] [Google Scholar]
- Coleman J. E. Human carbonic anhydrase. Protein conformation and metal ion binding. Biochemistry. 1965 Dec;4(12):2644–2655. doi: 10.1021/bi00888a014. [DOI] [PubMed] [Google Scholar]
- Coleman J. E. Mechanism of action of carbonic anhydrase. Subtrate, sulfonamide, and anion binding. J Biol Chem. 1967 Nov 25;242(22):5212–5219. [PubMed] [Google Scholar]
- Coleman J. E. Metal ion dependent binding of sulphonamide to carbonic anhydrase. Nature. 1967 Apr 8;214(5084):193–194. doi: 10.1038/214193a0. [DOI] [PubMed] [Google Scholar]
- Coleman J. E. Metallocarbonic anhydrases: optical rotatory dispersion and circular dichroism. Proc Natl Acad Sci U S A. 1968 Jan;59(1):123–130. doi: 10.1073/pnas.59.1.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fridborg K., Kannan K. K., Liljas A., Lundin J., Strandberg B., Strandberg R., Tilander B., Wirén G. Crystal structure of human erythrocyte carbonic anhydrase C. 3. Molecular structure of the enzyme and of one enzyme-inhibitor complex at 5-5 A resolution. J Mol Biol. 1967 May 14;25(3):505–516. doi: 10.1016/0022-2836(67)90202-1. [DOI] [PubMed] [Google Scholar]
- LINDSKOG S. Effects of pH and inhibitors on some properties related to metal binding in bovine carbonic anhydrase. J Biol Chem. 1963 Mar;238:945–951. [PubMed] [Google Scholar]
- LINDSKOG S., NYMAN P. O. METAL-BINDING PROPERTIES OF HUMAN ERYTHROCYTE CARBONIC ANHYDRASES. Biochim Biophys Acta. 1964 Jun 1;85:462–474. doi: 10.1016/0926-6569(64)90310-4. [DOI] [PubMed] [Google Scholar]
- Schrauzer G. N., Lian-Pin L. The molecular and electronic structure of vitamin B12r, cobaloximes(II), and related compounds. J Am Chem Soc. 1968 Nov 6;90(23):6541–6543. doi: 10.1021/ja01025a068. [DOI] [PubMed] [Google Scholar]



