Abstract
Nitric oxide (NO), superoxide and lipid peroxidation (LPO) produced under oxidative stress may contribute to development of postoperative adhesions. The objective of this study was to determine the effects of polychlorinated biphenyls (PCBs) on LPO, superoxide dismutase (SOD3), myeloperoxidase (MPO), and nitrite/nitrate in human normal peritoneal and adhesion fibroblasts. PCB treatment reduced iNOS expression as well as levels of nitrite/nitrate in both cell lines. Although there was no difference in iNOS expression between the two cell lines, adhesion fibroblasts manifested lower basal levels of MPO as compared to normal peritoneal fibroblasts. There was a reduction in MPO expression and its activity in response to PCB treatment in normal peritoneal fibroblasts; however this effect was minimal in adhesion fibroblasts. Moreover, adhesion fibroblasts manifested higher levels of LPO as compared to normal peritoneal fibroblasts, whereas PCB treatment increased LPO levels in both cell types. We conclude that PCBs promote the development of the adhesion phenotype by generating an oxidative stress environment. This is evident by lower iNOS, MPO, nitrite/nitrate and the simultaneous increase in LPO. Loss of MPO activity, possibly through a mechanism involving MPO heme depletion and free iron release, is yet another source of oxidative stress.
Keywords: PCB180, PCB077, organochlorine, adhesions, fibrosis, oxidative stress, myeloperoxidase
Introduction
Increased oxidative stress and lipid peroxidation (LPO) significantly contribute to development of postoperative adhesions [1, 2]. In mammalian cells, there is a highly regulated antioxidant defense system to maintain appropriate intracellular reactive oxygen species (ROS) levels to prevent oxidative damage [3]. This system includes antioxidant enzymes such as myeloperoxidase (MPO), superoxide dismutases (SODs), catalase, and various peroxidases that effectively remove these noxious ROS [4]. Oxidative stress is associated with increased free radicals, including the bioregulatory molecule nitric oxide (NO) and ROS such as superoxide (O2•−), and peroxynitrite (ONOO−). Reactive oxygen species participate in host defense by killing or damaging invading microbes [5]. Meanwhile, ROS can induce human cell death, apoptosis, and senescence in high levels [4].
Postoperative adhesion development and fibrosis are major complications following abdominopelvic operations [6–8]. They are major causes of infertility, abdominopelvic pain and small bowel obstruction [9–11]. Furthermore, when present in re-operative procedures, they can prolong operative time and increase the risk of intraoperative/postoperative complications [7, 10]. Many factors can affect and/or induce the development of adhesions and fibrosis. These include operative trauma, infection, foreign bodies, desiccation, irradiation, allergic reaction, or chemical injury [12, 13]. Additionally, polychlorinated biphenyls (PCBs) increase the expression of type I collagen in human normal peritoneal and adhesion fibroblasts and are also involved in tissue fibrosis in both men and women [1].
Nitric oxide is generated by enzymes collectively termed NO synthases (NOS) [14, 15]. Although there are three forms of NOS (endothelial NOS (eNOS); neuronal NOS (nNOS); and inducible NOS (iNOS)), only iNOS is responsible for NO synthesis during inflammation [16]. We have previously shown that lower NO levels are observed in adhesion fibroblasts and reduction in NO levels may significantly contribute to enhancement in ROS [2].
Superoxide is yet another free radical; SODs are the key enzymes required for the removal of O2•− by converting it to hydrogen peroxide (H2O2), which is utilized in combination with chloride ions by MPO, a highly cationic heme protein, to generate cytotoxic hypohalous acid and diffusible radical species [17–19]. Human Cu/Zn SOD (SOD3, extracellular-SOD), a member of the unique family of SODs, is found in the extracellular matrix of tissues and is ideally situated to prevent cell and tissue damage initiated by extracellularily produced ROS [20]. The loss of endogenous SOD3 activity exacerbates oxidative stress and pathologic damage [21].
The factors regulating NO and MPO function following peritoneal injury have yet to be elucidated. We have previously shown that fibroblasts isolated from adhesion tissues had significantly lower levels of NO and MPO as compared to normal peritoneal fibroblasts, although there was no difference in the expression levels of iNOS in both cell lines [2, 22]. Myeloperoxidase performs its functions in environments where NO and ONOO− are formed and uses H2O2 and chloride ion to produce hypohalous acid [5, 14, 15]. Recently, we have shown that the MPO/H2O2 system, like other NO scavengers, consumed NO released by iNOS during steady-state catalysis, thereby preventing the NO-induced inhibition of MPO attributed to the formation of the iNOS–nitrosyl complex in vitro [23]. Thus, removal of NO from the iNOS milieu by the MPO/H2O2 system during steady-state catalysis causes a significant increase in iNOS catalytic activity, production of citrulline, and presumably, production of NO [23]. Myeloperoxidase may play an important role in converting PCB metabolites very slowly into the corresponding semiquinone (SQ•−), but may function differently under oxidative stress [24].
In the present study, we hypothesized that human normal peritoneal and adhesion fibroblasts exposed to PCBs (PCB180 or PCB077), known to generate O2•−, promote MPO inactivation and LPO. To test this hypothesis, fibroblasts isolated from normal peritoneal and adhesion tissues from the same patient(s) were exposed to either PCB180 or PCB077 (20 ppb, 24 hours) and iNOS, NO, MPO, SOD3, and LPO levels were measured in cell lysates or cell culture medium. PCB180 was chosen because it is usually reported to be one of the most common PCBs measured from human serum and tissue samples, and is usually present in the greatest amounts, while PCB077 was chosen because it is considered to be dioxin-like in terms of its mechanism of action and toxicity [25]. Furthermore, we have recently found that both PCB180 and PCB077 can increase the expression of type I collagen in human normal peritoneal and adhesion fibroblasts [1].
Materials and Methods
Source and Culture of Human Fibroblasts
At initiation of the surgery, normal parietal peritoneal tissues from the anterior abdominal wall lateral to the midline incision and adherent tissues were excised from different patients undergoing laparotomy for pelvic pain. Normal peritoneum was ≥ 7.6 cm from any adhesions. Patients (n=3) did not have an active pelvic or abdominal infection and were not pregnant. The patients gave informed written consent to tissue collection, which was conducted under a protocol approved by the Wayne State University Institutional Review Board. Harvested tissue samples were immediately placed in standard media (Dulbecco’s Modified Eagle Medium (DMEM) containing 10% fetal bovine serum (FBS), and 2% penicillin and streptomycin). The tissues were cut into small pieces in a sterile culture dish and transferred into another fresh T-25 flask with 3 mL of dispase solution (2.4 U/mL; Invitrogen, Inc.). The flasks were incubated 4 hours at 37 °C. The samples were then centrifuged for 5 minutes at 1400g, transferred into a fresh T-25 flask with pre-warmed DMEM medium, and put in a 37 °C incubator (95% air and 5% CO2).
The outgrowth of fibroblasts generally took two weeks. When confluence was reached, the cells were transferred to 100-mm tissue culture dishes and cultured in standard media with 10% FBS. Thereafter, the confluent dishes were subcultured by trypsinization (1:5 split ratios). Studies were conducted using passage 3 to 5 cells to maintain comparability.
Confirming that the cells are indeed fibroblasts was ascertained by staining for vimentin using passages three to five, as we have previously described [26]. One of the strengths of the analysis is that the fibroblasts from normal peritoneum and the adhesions were from the same patients; thus each subject could serve as her own control.
Treatment of human normal peritoneal and adhesion fibroblasts
Fibroblasts isolated from normal peritoneal and adhesions tissues from the same patient(s) were treated with either PCB180 or PCB077 (20 ppb, 20% O2 24 hours). The PCBs were obtained from the Michigan Department of Community Health Analytic Chemistry Laboratory. The concentration was chosen based on the upper limit of human in vivo levels. All experiments were performed in triplicate.
Real-time reverse transcription polymerase chain reaction (RT-PCR) for iNOS, MPO and SOD3
RNA isolation
Total RNA was extracted from human normal peritoneal fibroblasts and adhesions using an RNeasy Mini Kit (QIAGEN Inc., Valencia, CA) according to the protocol provided by the manufacturer.
Reverse transcription
A 20 μL cDNA reaction volume was prepared with the use of QuantiTect Reverse Transcription Kit (QIAGEN Inc., Valencia, CA), as described by the manufacturer’s protocol.
Real-time RT-PCR Analysis
Real-time RT-PCR was performed with a QuantiTect SYBR Green RT-PCR kit (QIAGEN Inc., Valencia, CA) and a Cepheid 1.2f Detection System (Cepheid, Sunnyvale, CA). Each reaction was 25-μL including 12.5 μL of 2 X QuantiTect SYBR Green RT-PCR master mixes, 1 μL of cDNA template, and 0.2 μM each of target-specific primer that was designed to amplify a part of the gene of interest. Human oligonucleotide primers, which amplify variable portions of the protein coding regions, were used. Sequences of the oligonucleotides used for amplification of iNOS, MPO, SOD3, and β-actin mRNA are shown in Table 1. To quantify each target transcript, a standard curve was constructed with serial dilutions of standard plasmid (Invitrogen). The three-step polymerase chain reaction protocol applied for iNOS, MPO and SOD3 reaction consisted of 35 cycles of 95 °C for 15s, 54 °C (iNOS, MPO) or 60 °C (SOD3) for 30s, and 72 °C for 30s. Following real-time RT-PCR, a melting curve analysis was performed to demonstrate the specificity of the PCR product as a single peak. A control, which contained all the reaction components except for the template, was included in all experiments.
Table 1.
RT-PCR primer sequences.
| Locus | Sense (5?-3?) | Antisense (5?-3?) | bp |
|---|---|---|---|
| β-Actin | GCATTGTTACAGGAAGTC | TTACATAATTTACACGAAAGC | 126 |
| iNOS | GTTCTCAAGGCACAGGTCTC | GCAGGTCACTTATGTCACTTATC | 127 |
| MPO | GCTGAATGTGTTGTCCAAGTC | TGATGGTGCGGTATTTGTCC | 86 |
| SOD3 | GCCTCCATTTGTACCGAAAC | AGGGTCTGGGTGGAAAGG | 78 |
Detection of lipid peroxidation levels in normal peritoneal and adhesion fibroblasts
Lipid peroxidation, a well-established mechanism of cellular injury in animals and plants, was used as an indicator of oxidative stress in cells. Polyunsaturated fatty acid peroxides generate malondialdehyde (MDA) and 4-hydroxyalkenals (HAE) upon decomposition, and the measurement of MDA and HAE has been used as an indicator of LPO [27]. Normal peritoneal and adhesion fibroblasts were exposed to either PCB180 or PCB077 for (20ppb, 24 hours). Cells were homogenized and a LPO colorimetric microplate assay (Oxford Biomed Research, Oxford, MI) was utilized to measure the indicators of LPO, MDA and HAE, according to the protocol provided by the manufacturer. LPO was determined at 586 nm using a microplate reader, and the concentration was calculated using the MDA standards.
Detection of nitric oxide levels in normal peritoneal and adhesion fibroblasts
The nitrate/nitrite colorimetric assay (Griess assay) was used to measure the levels of stable NO byproducts in normal peritoneal and adhesion fibroblasts treated with either PCB180 or PCB077 (20 ppb, 24 hours). Nitrate is a stable oxidation product of NO, and its measurement serves as a convenient assay for NO production. DMEM media from cultured normal peritoneal and adhesion fibroblasts was analyzed for NO utilizing a nitrate/nitrite colorimetric assay kit (Cayman Chemical, Ann Arbor, MI) according to the manufacturer’s protocol. Nitrite was determined at 540 nm using a microplate reader, and the concentration was calculated using the nitrate standards.
Detection of iNOS activity in normal peritoneal and adhesion fibroblasts
The quantitative sandwich enzyme immunoassay technique (ELISA) was utilized to determine protein levels of iNOS in normal peritoneal and adhesion fibroblasts. Cell lysates were prepared after exposure to either PCB180 or PCB077 (20 ppb, for 24 hours). Levels of iNOS were assessed with the Human iNOS Quantikine ELISA Kit (R&D Systems, Inc., Minneapolis, MN) using a microplate reader at 450 nm according to the protocol provided by the manufacturer. The iNOS concentration was calculated by comparing the results to the standard.
Detection of MPO activity in normal peritoneal and adhesion fibroblasts
The NWLSS myeloperoxidase activity assay kit (Northwest Life Science Specialties, LLC., Vancouver, WA) was utilized for the study of MPO activity in normal peritoneal and adhesion fibroblasts exposed to either PCB180 or PCB077 (20 ppb, 24 hours). MPO activity was analyzed by measuring the hypochlorous acid (HOCL)-dependent chlorination of β-amino acid taurine. One unit of MPO activity is defined as the amount of enzyme that can catalyze sufficient HOCL production resulting in formation of 1 nM taurine chloramine at pH 6.5, 25 °C over 30 minutes in the presence of 100 mM chloride and 100 μM H2O2[19].
Detection of free iron in normal peritoneal and adhesion fibroblasts
To detect free iron (Fe), the VITROS Fe Slide method is performed using the VITROS Fe Slides and the VITROS Chemistry Products Calibrator Kit4 on VITROS Chemistry Systems. The VITROS Fe Slide is a multilayered, analytical element coated on a polyester support. A drop of patient sample is deposited on the slide and is evenly distributed by the spreading layer to the underlying layers. Iron (as ferric ion) is removed from transferrin at acidic pH and migrates to the reducing layer, where ascorbic acid reduces iron to the ferrous form. The ferrous ion then is bound to the dye and forms a colored complex in the reagent layer. Following addition of the sample, the slide is incubated and the reflection density measured after 1 and 5 minutes. The difference in reflection density is proportional to the iron concentration in the sample.
Statistical analysis
Data were analyzed using SPSS 15.0 for Windows. A mixed model repeated measures ANOVA was used with treatment as the within factor and cell type as the between factor. Paired comparisons with a Bonferroni correction were used to compare pairs of treatments. Significant interactions between treatment and cell type were analyzed with independent sample t-tests by cell type on each treatment. Statistical significance of P < 0.05 is considered significant for all analyses.
Results
We first confirmed that culturing of the human peritoneal and adhesion fibroblasts in the presence of PCB180 or PCB077 had no effect on mRNA levels of β-actin, the housekeeping gene used to normalize RNA levels for the real-time RT-PCR, nor did it affect cell viability as assessed by the trypan blue exclusion method.
PCB treated fibroblasts reduced the mRNA levels of iNOS, MPO, and SOD3
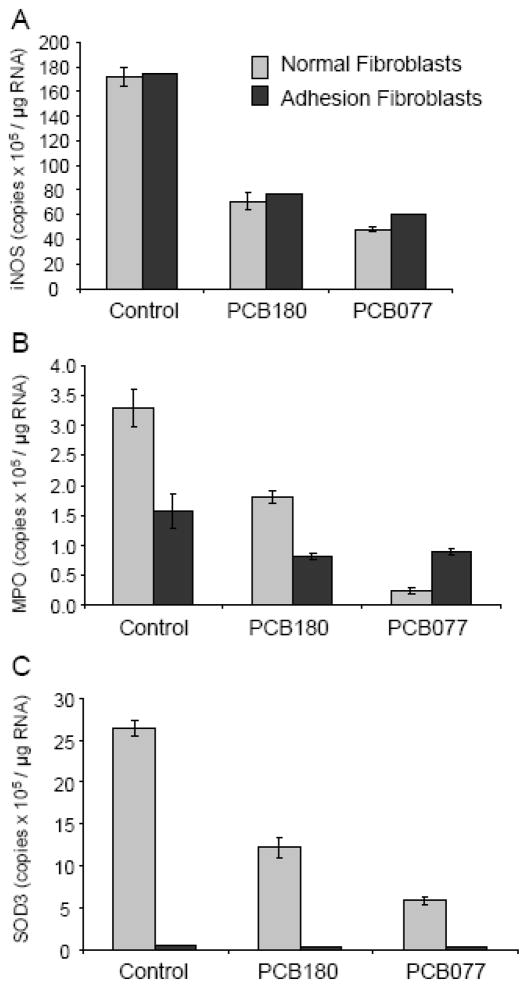
The results demonstrated that, as previously reported, iNOS mRNA levels were not significantly different in normal peritoneal fibroblasts (1.7 × 108 copies/μg RNA) as compared to adhesion fibroblasts (1.8 × 108 copies/μg RNA, p=0.846) (Figure 1A). Treatment of normal peritoneal fibroblasts with PCB180 resulted in a 2.4-fold decrease in iNOS mRNA levels (p<0.0001); a similar marked decrease (3.6-fold) was seen with PCB077 (p<0.0001, Figure 1A). Furthermore, treatment with PCB180 or PCB077 also resulted in a marked decrease in iNOS mRNA levels in adhesion fibroblasts (2.3 and 2.9 fold decrease, respectively, p<0.0001, Figure 1A). As we have previously reported, mRNA levels of MPO were lower in adhesion fibroblasts as compared to normal peritoneal fibroblasts (Figure 1B) [22]. Additionally, we found that mRNA levels of SOD3 were also lower in adhesion fibroblasts as compared to normal peritoneal fibroblasts (Figure 1C). In comparison with the effects of PCBs on iNOS expression, PCB180 and PCB077 had the same appreciable effects on mRNA levels of MPO or SOD3 from either the normal or adhesion fibroblasts (p<0.01 or p<0.0001, Figure 1B and C).
FIGURE 1.
Real-time RT-PCR for iNOS, MPO, and SOD3. Expression of iNOS, MPO, and SOD3 mRNA levels in normal peritoneal and adhesion fibroblast treated with various PCBs. Results are representative of the mean of three independent experiments.
PCB treated fibroblasts reduced the level of nitrate/nitrite
Nitrite is a stable oxidation product of NO, and its measurement serves as a convenient assay for NO production. We utilized the Greiss assay to measure levels of stable NO byproducts (nitrate/nitrite) in normal peritoneal and adhesion fibroblasts. As we have previously reported, adhesion fibroblasts exhibited significantly lower levels of nitrate/nitrite as compared to normal peritoneal fibroblasts [2]. Here, we confirmed our previous finding and further showed that treatment of normal peritoneal fibroblasts with PCBs significantly reduced nitrate/nitrite levels from 2.46 to 1.41 μM for PCB180 and to 1.28 μM for PCB077 (p < 0.01, Figure 2). Similarly, treatment of adhesion fibroblasts with PCBs markedly reduced nitrate/nitrite levels from 2.10 to 0.95 μM for PCB180 and to 1.23 μM for PCB077 (p < 0.001, Figure 2). Together, this indicates that PCBs significantly reduce the production of NO.
FIGURE 2.
Nitric oxide levels in human normal peritoneal and adhesion fibroblasts (n=3). Griess assay was performed in 24 hrs culture media collected from human normal peritoneal and adhesion fibroblasts before and after various PCB treatments. *p<0.0002, **p=0.0003, #p=0.0116, ##p<0.0001, and ###p<0.0001 compared to adhesion fibroblasts, respectively. Results are representative of the mean of three independent experiments.
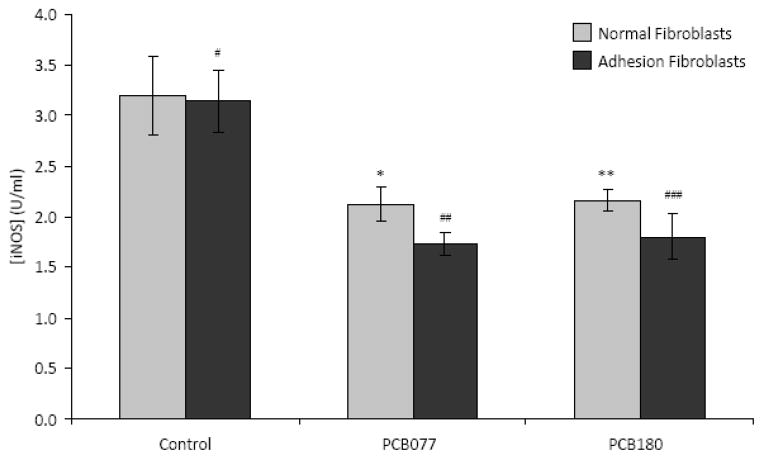
PCB treated fibroblasts decreased the activity of iNOS
Since the iNOS isoform is the major source of NO in these cells [28], we next assessed whether PCBs affect iNOS expression and activity. Although the iNOS activity did not change in either untreated cell lines (Figure 3), it was significantly inhibited by PCB180 (from 3.20 to 2.2 U/mL, p = 0.012) and PCB077 (from 3.20 to 2.1 U/mL, p = 0.010) in normal peritoneal fibroblasts, and even more markedly inhibited by the PCB180 (from 3.10 to 1.8 U/mL, p < 0.01) and PCB077 (from 3.10 to 1.7 U/mL, p < 0.01) in adhesion fibroblasts (Figure 3).
FIGURE 3.
iNOS activity in human normal peritoneal and adhesion fibroblasts (n=3). ELISA was performed in cell lysates from human normal peritoneal and adhesion fibroblasts before and after PCBs treatment for 24hrs. *p=0.0102, **p=0.0119, #p=0.7418, ##p<0.01, and ###p<0.01 compared to adhesion fibroblasts before PCBs treatment, respectively. Results are representative of the mean of three independent experiments.
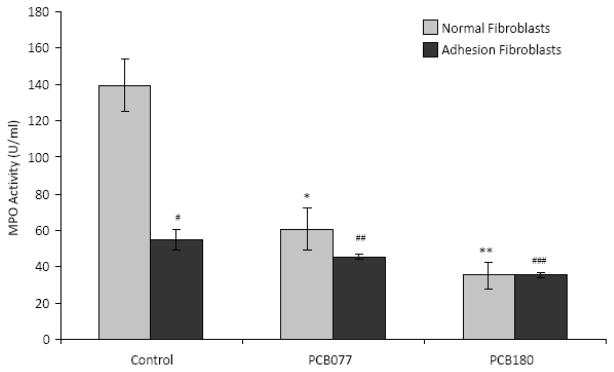
PCB treated fibroblasts decreased the activity of MPO
To confirm the effect of PCBs on MPO activity, we performed a MPO activity assay by measuring the HOCL-dependent chlorination of β-amino acid taurine (TauNH2). We confirmed our previous finding that adhesion fibroblasts exhibited significantly lower level of MPO activity as compared to normal peritoneal fibroblasts (P < 0.01, Figure 6) and further demonstrated that treatment of normal peritoneal fibroblasts with PCBs significantly reduced MPO activity from 139.47 to 35.37 U/mL for PCB180 and to 60.64 U/mL for PCB077 (p < 0.01, Figure 4)[28]. Similarly, treatment of adhesion fibroblasts with PCBs markedly reduced MPO levels from 54.57 to 35.37 U/mL for PCB180 and to 45.48 U/mL for PCB077, p < 0.05, Figure 4). Free iron levels were also measured as described in methods in normal peritoneal and adhesion fibroblasts and it was found to be significantly lower in adhesion fibroblasts (5 μg/dl) as compared to normal peritoneal fibroblasts (10 μg/dl). These data indicate that PCBs significantly affect the activity of MPO.
Figure 6.
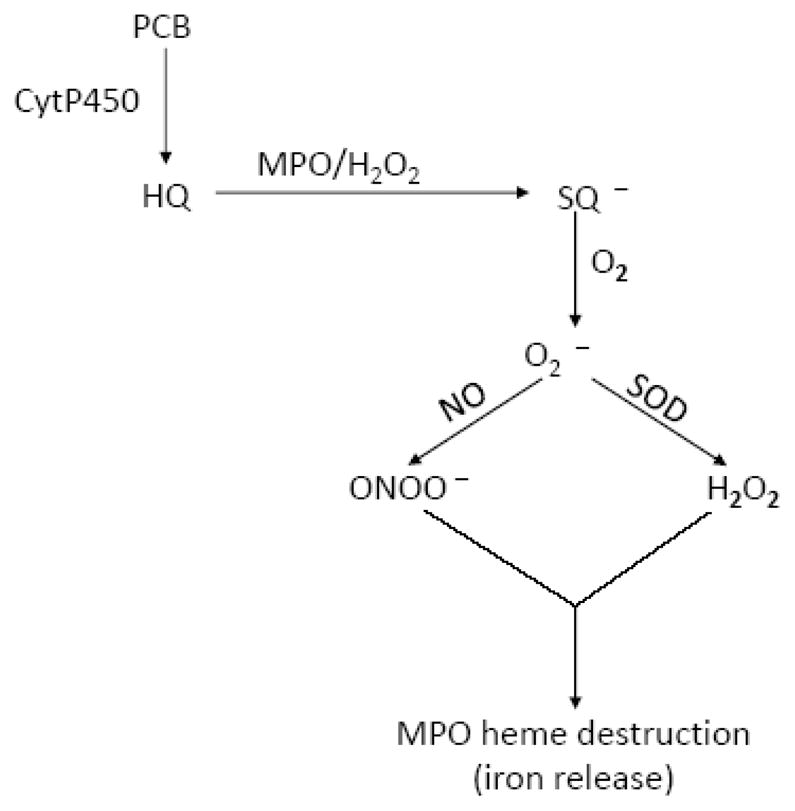
A theoretical model has been created that shows the link between PCBs, MPO, SOD3, iNOS, and O2•− production.
FIGURE 4.
MPO activity in human normal peritoneal and adhesion fibroblasts (n=3). MPO activity was analyzed in cell lysates from human normal peritoneal and adhesion fibroblasts before and after various PCB treatments for 24 hours. *p=0.0017, **p=0.0004, #p=0.0007, ##p=0.0003, and ###p=0.0002 compared to adhesion fibroblasts before PCBs treatment, respectively. Results are representative of the mean of three independent experiments.
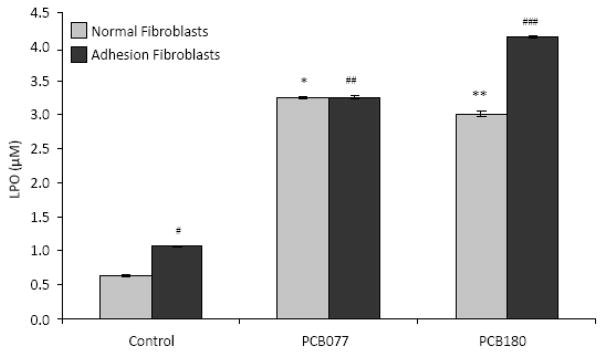
PCB treated fibroblasts exhibited higher levels of LPO
The level of LPO was determined by measuring LPO indicators; MAD and HAE (see materials and methods). There was a marked difference in basal LPO levels between normal peritoneal and adhesion fibroblasts, with adhesion fibroblasts manifesting significantly higher LPO (p < 0.01, Figure 5). Both PCB180 and PCB077 significantly increased LPO in normal peritoneal fibroblasts (from 0.63 μM to 3.01 μM and to 3.26 μM, p < 0.01) and in adhesion fibroblasts (from 1.07 μM to 4.15 μM and to 3.26 μM, p < 0.01, Figure 5).
FIGURE 5.
Lipid peroxidation levels in human normal peritoneal and adhesion fibroblasts (n=3). LPO was assessed in cell lysates from human normal peritoneal and adhesion fibroblasts before and after various PCB treatments for 24 hours. *p<0.0001, **p<0.0001, #p<0.0001, ##p<0.0001, and ###p<0.0001 compared to adhesion fibroblasts before PCBs treatment, respectively. Results are representative of the mean of three independent experiments.
Discussion
PCBs, which have been shown to increase the expression of type I collagen in human normal peritoneal and adhesion fibroblasts, are also mechanicastically linked to enhanced tissue fibrosis in men and women through a pathway that involves overproduction of O2•− [1, 29]. Therefore, scavenging excess O2•− levels by SOD3 provides a hypothetical means to prevent fibrosis and/or postoperative adhesions. Recently, we have shown that hypoxia induced the development of the adhesion phenotype through a mechanism involve superoxide production. Indeed scavenging O2•− protected against hypoxia induced adhesion phenotype development [30]. In the present study, we identified a significant reduction of SOD3 mRNA levels in response to PCB180 and PCB077 exposure, to a level even below that observed in the profibrotic adhesion fibroblasts.
Hypoxia is a crucial trigger for the function of fibroblast cells during peritoneal healing and the development of postoperative adhesions through a mechanism that involves the production of O2•− [30, 31]. Similarly, PCBs are known to be a major cause of excessive levels of O2•− through a mechanism, which involves mammalian peroxidase enzymes that convert hydroquinone (H2Q) to SQ•−, which in turn converts oxygen to O2•− [32]. Recently we have shown that MPO is differentially expressed in normal and adhesion fibroblasts established from normal peritoneal and adhesion tissues of the same patient(s) [22]. Hypoxia reduces MPO activity in normal peritoneal fibroblasts to the suppressed levels observed in adhesion fibroblasts, indicating the importance of this enzyme in the development of the adhesion phenotype. In this study we have also shown that PCB exposure significantly decreased MPO activity in normal peritoneal fibroblasts to the suppressed levels observed in adhesion fibroblasts (Figure 4).
PCBs are also known to mediate free iron release from metalloproteins [33]. Most iron inside a cell is either bound to enzyme or sequestered by an iron storage protein. Attenuation of SOD3 results an increased flux of O2•− which leads to an increase in free iron by oxidatively attacking the [Fe-S]x clusters of a number of enzymes [34]. It has been recently shown that the increased fluxes of O2•− in an SOD3-deficient E.coli strain lead to increased levels of free intracellular iron and that this is the result of O2•−-mediated release of iron from [Fe-S]x proteins such as aconitase [35, 36]. Therefore, the increased levels of O2•− after PCB exposure may mobilize other iron from iron-binding proteins such as transferrin, ferritin, and lactoferrin. In support of this hypothesis, we have recently shown that MPO may serve as a source of free iron under oxidative stress when levels of both NO and O2•− are elevated [22].
Iron-related oxidative stress can promote lipid peroxidation, DNA strand breaks, and modification or degradation of biomolecules, eventually leading to cell death [37, 38]. Our current results demonstrate that basal MPO activity levels differed between normal peritoneal and adhesion fibroblasts. Adhesion fibroblasts manifested significantly lower levels of MPO activity. Both PCBs significantly lowered MPO activity in normal peritoneal fibroblasts to levels seen in adhesion fibroblasts (Figure 4). In contrast, PCB treatment slightly reduced MPO activity in adhesion fibroblasts (Figure 4). The free iron generated by MPO destruction and other sources may induce oxidative stress and make it highly toxic as it can rapidly react with H2O2 and molecular oxygen to produce hydroxyl radicals through the Fenton reaction [26, 39]. Our current results also demonstrate that there was a difference in basal LPO levels between normal peritoneal and adhesion fibroblasts, with adhesion fibroblasts manifesting significantly higher LPO corresponding to enhanced oxidative stress (Figure 5). Both PCBs significantly increased LPO in both cell types. Our results clearly support the hypothesis that, under oxidative stress, i.e. PCBs exposure, MPO loses its activity through a mechanism that involves MPO heme depletion and iron release. Free iron is capable of stimulating the production of free radicals, which cause oxidative damage leading to LPO and stimulates the development of the adhesion phenotype resulting in postoperative adhesion development.
Most of the generated O2•− undergoes a SOD3-catalyzed reaction generating H2O2 as an end product. This reaction is a central element in the maintenance of the cellular redox balance. If O2•− is not detoxified by SOD3, during the peritoneal injury, O2•− interacts with NO at near diffusion-controlled rate yielding ONOO−, a much more toxic reagent which attacks many cellular components, reacting with thiols and iron–sulfur centers, as well as initiating LPO. It also nitrates tyrosine by a reaction catalyzed by SOD [40]. Nitrite, a major product of NO metabolism, can readily promote protein nitration via reactions with peroxidases [41, 42]. Similar process may cause alterations in protein function in peritoneal fibroblasts, thereby mediating the metabolism of several cytotoxic biological reactions. Indeed we have shown that adhesion fibroblasts to manifest higher protein nitration as compared to normal peritoneal fibroblasts [2].
Recently we have shown that MPO and iNOS are co-localized in normal peritoneal and adhesion fibroblasts established from the same patient(s) [22]. MPO not only oxidizes H2Q to SQ•−, it also up-regulates the catalytic activity of iNOS by scavenging NO, preventing feedback inhibition attributed to the formation of an iNOS-Fe-NO complex [43]. When the MPO-H2O2 system is impaired, i.e. MPO activity decreases, it could not up-regulates the catalytic activity of iNOS by scavenging NO and iNOS activity decreases accordingly. Our data also indicated that PCB exposure led to a marked reduction of iNOS in peritoneal fibroblasts (Figures 1A and 2). This process was associated with a decreased formation of nitrate, the end product of NO bioconversion.
In summary, a theoretical model has been created that shows the link between PCBs, MPO, SOD3, iNOS, and O2•− production (Figure 6). In this model, we highlight the capacity of MPO in: 1) utilizing hydroxylated PCBs as a one-electron substrate through the peroxidation cycle; and 2) serving as a source of free iron under oxidative stress where H2O2 and ONOO− are elevated. The ground state of the enzyme, MPO-Fe(III), reacts rapidly with H2O2 to form Compound I, a two electron oxidized intermediate possessing a Fe(IV)=O group and a resonance-stabilized porphyrin π cation radical. Compound I is capable of oxidizing either halides or pseudohalides through a 2 electron transition, generating MPO-Fe(III) and the corresponding hypohalous acid [44]. Compound I may oxidize PCBs or other organic and inorganic substrates through two sequential one electron steps forming Compound II and MPO-Fe(III), respectively. The conversion of Compound II to MPO-Fe(III) is the rate limiting step of the MPO catalytic cycle which may accelerated by PCB dihydroxy metabolites. PCBs are first metabolized via formation of arene oxides or by direct insertion of a hydroxy-group [45]. The intermediate arene oxide is subsequently rearranged to a hydroxylated PCB (OH-PCB) or further metabolized to a diol via secondary hydroxylation [46]. One electron oxidation of PCB dihydroxy metabolites catalyzed by MPO yields a SQ•− [47, 48]. These intermediates react with molecular O2 to give the Q and O2•− as shown by peroxidase catalyzed oxidation of PCB dihydroxy compounds [49, 50].
Collectively, our results show that PCBs promote the development of the adhesion phenotype by generating an oxidative stress environment. This is evident by lower iNOS, MPO, nitrite/nitrate and the simultaneous increase in LPO. Loss of MPO activity, possibly through a mechanism involving MPO heme depletion and free iron release, is yet another source of oxidative stress. Continuous scavenging of free radicals with the use of a dietary antioxidant supplement may have a beneficial long-term protection against not only the development of postoperative adhesions but also other similar medical conditions. This is a theoretical benefit, but that the role of dietary antioxidants on local tissue scavenging needs to be ascertained.
Acknowledgments
This study was supported in part by NIH grant number NIH 1RO1 GM069941-01A3 to G.M. Saed.
Abbreviations
- HAE
4-hydroxyalkenals
- DMEM
Dulbecco’s Modified Eagle Medium
- eNOS
endothelial NOS
- FBS
fetal bovine serum
- H2O2
hydrogen peroxide
- H2Q
hydroquinone
- HOCL
hypochlorous acid
- LPO
lipid peroxidation
- MDA
malondialdehyde
- MPO
myeloperoxidase
- nNOS
neuronal NOS
- NO
nitric oxide
- ONOO−
peroxynitrite
- PCBs
polychlorinated biphenyls
- ROS
reactive oxygen species
- SQ•−
semiquinone
- O2•−
superoxide
- SOD
superoxide dismutase
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Diamond MP, Wirth JJ, Saed GM. PCBs enhance collagen I expression from human peritoneal fibroblasts. Fertil Steril. 2008;90(4 Suppl):1372–5. doi: 10.1016/j.fertnstert.2007.07.1380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Saed GM, et al. Regulation of inducible nitric oxide synthase in post-operative adhesions. Hum Reprod. 2006;21(6):1605–11. doi: 10.1093/humrep/dei500. [DOI] [PubMed] [Google Scholar]
- 3.Khaitan D, Dwarakanath BS. Endogenous and induced oxidative stress in multi-cellular tumour spheroids: implications for improving tumour therapy. Indian J Biochem Biophys. 2009;46(1):16–24. [PubMed] [Google Scholar]
- 4.Hileman EO, et al. Intrinsic oxidative stress in cancer cells: a biochemical basis for therapeutic selectivity. Cancer Chemother Pharmacol. 2004;53(3):209–19. doi: 10.1007/s00280-003-0726-5. [DOI] [PubMed] [Google Scholar]
- 5.Cheng G, et al. Homologs of gp91phox: cloning and tissue expression of Nox3, Nox4, and Nox5. Gene. 2001;269(1–2):131–40. doi: 10.1016/s0378-1119(01)00449-8. [DOI] [PubMed] [Google Scholar]
- 6.Diamond MP, El-Mowafi DM. Pelvic Adhesions. Surg Technol Int. 1998;VII:273–283. [PubMed] [Google Scholar]
- 7.Diamond MP, et al. The effect of periovarian adhesions on follicular development in patients undergoing ovarian stimulation for in vitro fertilization-embryo transfer. Fertil Steril. 1988;49(1):100–3. doi: 10.1016/s0015-0282(16)59657-5. [DOI] [PubMed] [Google Scholar]
- 8.El-Mowafi DM, Diamond MP. Are pelvic adhesions preventable? Surg Technol Int. 2003;11:222–35. [PubMed] [Google Scholar]
- 9.Diamond MP, et al. Adhesion reformation and de novo adhesion formation after reproductive pelvic surgery. Fertil Steril. 1987;47(5):864–6. doi: 10.1016/s0015-0282(16)59181-x. [DOI] [PubMed] [Google Scholar]
- 10.Diamond MP, Freeman ML. Clinical implications of postsurgical adhesions. Hum Reprod Update. 2001;7(6):567–76. doi: 10.1093/humupd/7.6.567. [DOI] [PubMed] [Google Scholar]
- 11.Cheong YC, et al. Peritoneal healing and adhesion formation/reformation. Hum Reprod Update. 2001;7(6):556–66. doi: 10.1093/humupd/7.6.556. [DOI] [PubMed] [Google Scholar]
- 12.Holmdahl L, et al. Adhesions: pathogenesis and prevention-panel discussion and summary. Eur J Surg Suppl. 1997;(577):56–62. [PubMed] [Google Scholar]
- 13.diZerega GS. The peritoneum and its response to surgical injury. Prog Clin Biol Res. 1990;358:1–11. [PubMed] [Google Scholar]
- 14.Yamamoto Y, et al. Hypoxia induces production of nitric oxide and reactive oxygen species in glomus cells of rat carotid body. Cell Tissue Res. 2006;325(1):3–11. doi: 10.1007/s00441-006-0178-4. [DOI] [PubMed] [Google Scholar]
- 15.Zhu H, Bunn HF. Oxygen sensing and signaling: impact on the regulation of physiologically important genes. Respir Physiol. 1999;115(2):239–47. doi: 10.1016/s0034-5687(99)00024-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jiang ZL, et al. Nitric oxide synthase isoforms expression in fibroblasts isolated from human normal peritoneum and adhesion tissues. Fertil Steril. 2008 doi: 10.1016/j.fertnstert.2007.07.1313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Harrison JE, Schultz J. Studies on the chlorinating activity of myeloperoxidase. J Biol Chem. 1976;251(5):1371–4. [PubMed] [Google Scholar]
- 18.Kettle AJ, van Dalen CJ, Winterbourn CC. Peroxynitrite and myeloperoxidase leave the same footprint in protein nitration. Redox Rep. 1997;3(5–6):257–8. doi: 10.1080/13510002.1997.11747120. [DOI] [PubMed] [Google Scholar]
- 19.Weiss SJ, et al. Chlorination of taurine by human neutrophils. Evidence for hypochlorous acid generation. J Clin Invest. 1982;70(3):598–607. doi: 10.1172/JCI110652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Carlsson LM, et al. Mice lacking extracellular superoxide dismutase are more sensitive to hyperoxia. Proc Natl Acad Sci U S A. 1995;92(14):6264–8. doi: 10.1073/pnas.92.14.6264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Suliman HB, Ali M, Piantadosi CA. Superoxide dismutase-3 promotes full expression of the EPO response to hypoxia. Blood. 2004;104(1):43–50. doi: 10.1182/blood-2003-07-2240. [DOI] [PubMed] [Google Scholar]
- 22.Saed GM, et al. The role of myeloperoxidase in the pathogenesis of postoperative adhesions. Wound Repair Regen. 2009;17(4):531–9. doi: 10.1111/j.1524-475X.2009.00500.x. [DOI] [PubMed] [Google Scholar]
- 23.Galijasevic S, et al. Myeloperoxidase up-regulates the catalytic activity of inducible nitric oxide synthase by preventing nitric oxide feedback inhibition. Proc Natl Acad Sci U S A. 2003;100(25):14766–71. doi: 10.1073/pnas.2435008100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Galijasevic S, et al. Myeloperoxidase interaction with peroxynitrite: chloride deficiency and heme depletion. Free Radic Biol Med. 2009;47(4):431–9. doi: 10.1016/j.freeradbiomed.2009.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Safe SH. Polychlorinated biphenyls (PCBs): environmental impact, biochemical and toxic responses, and implications for risk assessment. Crit Rev Toxicol. 1994;24(2):87–149. doi: 10.3109/10408449409049308. [DOI] [PubMed] [Google Scholar]
- 26.Saed GM, Diamond MP. Differential expression of alpha smooth muscle cell actin in human fibroblasts isolated from intraperitoneal adhesions and normal peritoneal tissues. Fertil Steril. 2004;82(Suppl 3):1188–92. doi: 10.1016/j.fertnstert.2004.02.147. [DOI] [PubMed] [Google Scholar]
- 27.Esterbauer H, Schaur RJ, Zollner H. Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. Free Radic Biol Med. 1991;11(1):81–128. doi: 10.1016/0891-5849(91)90192-6. [DOI] [PubMed] [Google Scholar]
- 28.Saed G, et al. The role of myeloperoxidase in the pathogenesis of postoperative adhesions. Wound Repair and Regeneration. 2009 doi: 10.1111/j.1524-475X.2009.00500.x. In press. [DOI] [PubMed] [Google Scholar]
- 29.Gao F, et al. Extracellular superoxide dismutase in pulmonary fibrosis. Antioxid Redox Signal. 2008;10(2):343–54. doi: 10.1089/ars.2007.1908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fletcher NM, et al. Hypoxia-generated superoxide induces the development of the adhesion phenotype. Free Radic Biol Med. 2008;45(4):530–6. doi: 10.1016/j.freeradbiomed.2008.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Binda MM, Molinas CR, Koninckx PR. Reactive oxygen species and adhesion formation: clinical implications in adhesion prevention. Hum Reprod. 2003;18(12):2503–7. doi: 10.1093/humrep/deg481. [DOI] [PubMed] [Google Scholar]
- 32.Metodiewa D, Dunford HB. The role of myeloperoxidase in the oxidation of biologically active polyhydroxyphenols (substituted catechols) Eur J Biochem. 1990;193(2):445–8. doi: 10.1111/j.1432-1033.1990.tb19358.x. [DOI] [PubMed] [Google Scholar]
- 33.Whysner J, Wang CX. Hepatocellular iron accumulation and increased cell proliferation in polychlorinated biphenyl-exposed Sprague-Dawley rats and the development of hepatocarcinogenesis. Toxicol Sci. 2001;62(1):36–45. doi: 10.1093/toxsci/62.1.36. [DOI] [PubMed] [Google Scholar]
- 34.McCormick ML, Buettner GR, Britigan BE. Endogenous superoxide dismutase levels regulate iron-dependent hydroxyl radical formation in Escherichia coli exposed to hydrogen peroxide. J Bacteriol. 1998;180(3):622–5. doi: 10.1128/jb.180.3.622-625.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Keyer K, Imlay JA. Superoxide accelerates DNA damage by elevating free-iron levels. Proc Natl Acad Sci U S A. 1996;93(24):13635–40. doi: 10.1073/pnas.93.24.13635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Keyer K, Gort AS, Imlay JA. Superoxide and the production of oxidative DNA damage. J Bacteriol. 1995;177(23):6782–90. doi: 10.1128/jb.177.23.6782-6790.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kimura S, et al. Structure of the human thyroid peroxidase gene: comparison and relationship to the human myeloperoxidase gene. Biochemistry. 1989;28(10):4481–9. doi: 10.1021/bi00436a054. [DOI] [PubMed] [Google Scholar]
- 38.Ten RM, et al. Molecular cloning of the human eosinophil peroxidase. Evidence for the existence of a peroxidase multigene family. J Exp Med. 1989;169(5):1757–69. doi: 10.1084/jem.169.5.1757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Thomas C, et al. Hydroxyl radical is produced via the Fenton reaction in submitochondrial particles under oxidative stress: implications for diseases associated with iron accumulation. Redox Rep. 2009;14(3):102–8. doi: 10.1179/135100009X392566. [DOI] [PubMed] [Google Scholar]
- 40.Turko IV, Murad F. Protein nitration in cardiovascular diseases. Pharmacol Rev. 2002;54(4):619–34. doi: 10.1124/pr.54.4.619. [DOI] [PubMed] [Google Scholar]
- 41.Hazen SL, et al. Formation of nitric oxide-derived oxidants by myeloperoxidase in monocytes: pathways for monocyte-mediated protein nitration and lipid peroxidation In vivo. Circ Res. 1999;85(10):950–8. doi: 10.1161/01.res.85.10.950. [DOI] [PubMed] [Google Scholar]
- 42.van der Vliet A, et al. Formation of reactive nitrogen species during peroxidase-catalyzed oxidation of nitrite. A potential additional mechanism of nitric oxide-dependent toxicity. J Biol Chem. 1997;272(12):7617–25. doi: 10.1074/jbc.272.12.7617. [DOI] [PubMed] [Google Scholar]
- 43.Saed GM, Diamond MP. Modulation of the expression of tissue plasminogen activator and its inhibitor by hypoxia in human peritoneal and adhesion fibroblasts. Fertil Steril. 2003;79(1):164–8. doi: 10.1016/s0015-0282(02)04557-0. [DOI] [PubMed] [Google Scholar]
- 44.Davies MJ, et al. Mammalian heme peroxidases: from molecular mechanisms to health implications. Antioxid Redox Signal. 2008;10(7):1199–234. doi: 10.1089/ars.2007.1927. [DOI] [PubMed] [Google Scholar]
- 45.Ariyoshi N, et al. Metabolism of 2,4,5,2′,4′,5′-hexachlorobiphenyl with liver microsomes of phenobarbital-treated dog; the possible formation of PCB 2,3-arene oxide intermediate. Xenobiotica. 1992;22(11):1275–90. doi: 10.3109/00498259209053156. [DOI] [PubMed] [Google Scholar]
- 46.Ariyoshi N, et al. Metabolism of 2,4,5,2′,4′,5′-hexachlorobiphenyl (PCB153) in guinea pig. Xenobiotica. 1997;27(9):973–83. doi: 10.1080/004982597240136. [DOI] [PubMed] [Google Scholar]
- 47.Hollenberg PF. Mechanisms of cytochrome P450 and peroxidase-catalyzed xenobiotic metabolism. FASEB J. 1992;6(2):686–94. doi: 10.1096/fasebj.6.2.1537457. [DOI] [PubMed] [Google Scholar]
- 48.Li Y, Trush MA. DNA damage resulting from the oxidation of hydroquinone by copper: role for a Cu(II)/Cu(I) redox cycle and reactive oxygen generation. Carcinogenesis. 1993;14(7):1303–11. doi: 10.1093/carcin/14.7.1303. [DOI] [PubMed] [Google Scholar]
- 49.Amaro AR, et al. Metabolic activation of PCBs to quinones: reactivity toward nitrogen and sulfur nucleophiles and influence of superoxide dismutase. Chem Res Toxicol. 1996;9(3):623–9. doi: 10.1021/tx950117e. [DOI] [PubMed] [Google Scholar]
- 50.Oakley GG, Robertson LW, Gupta RC. Analysis of polychlorinated biphenyl-DNA adducts by 32P-postlabeling. Carcinogenesis. 1996;17(1):109–14. doi: 10.1093/carcin/17.1.109. [DOI] [PubMed] [Google Scholar]