Abstract

Highly reactive rhodium (II) N-trifluoromethylsulfonyl azavinyl carbenes are formed in situ from NH-1,2,3-triazoles, triflic anhydride, and rhodium carboxylates. They rapidly and selectively react with olefins, providing cyclopropane carboxaldehydes and 2,3-dihydropyrroles in generally excellent yields and high enantio- and diastereoselectivity.
Diazo carbonyl compounds are versatile reagents which, among other applications, are often used for the generation of transition metal carbenes.1 The latter are versatile synthetic intermediates that readily and often stereoselectively add to carbon–carbon and carbon–heteroatom multiple bonds and undergo C–H insertion reactions.2 Diazo group transfer from a sulfonyl azide to an activated methylene carbon is the most common route to diazo compounds.3 However, many sulfonyl azides, as well as diazo compounds, are not shelf stable and require precautions in handling. We have recently demonstrated that 1-sulfonyl 4-substituted 1,2,3-triazoles can serve as stable and readily accessible synthetic equivalents of diazo compounds.4 They undergo facile ring-chain isomerization, and the diazoimine tautomer reacts with transition metal catalysts, resulting in the formation of azavinyl carbenes. These novel intermediates react selectively with nitriles and olefins, resulting in the formation of imidazoles and cyclopropanes, respectively. However, these reactions require elevated temperatures, and starting sulfonyl triazoles are normally prepared from sulfonyl azides.
Here we report a new and practical method for the generation of reactive rhodium azavinyl carbenes from stable and readily accessible NH-triazoles,5 which are sulfonylated in situ by triflic anhydride in the presence of a pyridine base (eq. 1). The methodology does not involve sulfonyl azides at any stage of the synthesis, and the resulting N-triflyl azavinyl carbenes exhibit exceptional reactivity toward olefins, producing cyclopropanes and 1,2-dihydropyrroles with excellent enantio- and diastereoselectivity. The ability to introduce extremely electron withdrawing groups into the triazole nucleus, thereby controlling the electrophilicity of the resulting carbene, is easily the most valuable feature of the new approach. Note that the corresponding N-sulfonylated triazoles cannot be prepared in isolated form because of their extreme susceptibility to hydrolysis.
 |
(1) |
We expected that sulfonylation of NH-triazoles would lead to a mixture of isomeric N2- and N1-sulfonylated triazole products 2 and 3.6 These two isomers exist in equilibrium, but only the N1 isomer 3 can undergo ring-chain isomerization forming the diazo imine tautomer which reacts with rhodium complexes, leading to a carbene intermediate 4.
We first examined the sulfonylation reaction of 4-phenyl-NH-1,2,3-triazole 5 by triflic anhydride in the presence of pyridine bases (eq. 2). 2,6-Di-t-butyl-4-methylpyridine 6 provided optimal results and was used in all subsequent reactions. Monitoring reaction progress by 1H NMR (from −60 °C to r.t. in 10 °C increments) revealed that the 1,5-disubstituted triazole isomer 9 was indeed formed in negligible amount, whereas the expected isomers 7 and 8 were formed in approximately 1:1 ratio.
 |
(2) |
With these conditions in hand, we turned our attention to the rhodium-catalyzed reaction of the in situ generated sulfonyl triazoles with olefins. As illustrated in Table 1, 4-phenyl-NH 1,2,3-triazole 5 was reacted with triflic anhydride in the presence of 1.2 equivalents of base 6 at −60 °C. After 10 minutes, styrene (3 eq) and 0.5 mol% of Rh2(S-NTTL)4,7 12, were added and the reaction was allowed to warm to RT over 3 hours. Cyclopropane 11 was isolated in 35% yield, 98% ee, and dr >20:1 (entry 1). Changing the solvent to 1,2-dichloroethane and increasing the starting temperature of the reaction −30 °C improved the yield to 65% while maintaining high enantioselectivity (entry 2). Changing the solvent to chloroform further increased the yield to 90% and gave the product with 99% ee (entry 3). The less sterically hindered Rh2(S-NTV)4 catalyst 13 resulted in the lower ee (76%, entry 4). The yield of cyclopropane was reduced to 81% when only 2 equiv. of styrene were used (entry 5).
Table 1.
Cyclopropanation of styrene with 4-phenyl-NH-triazole.
 | |||||
|---|---|---|---|---|---|
| entry | solvent | catalyst | equiv. of styrene | yield, % | ee, % |
| 1a | DCM | Rh2(S-NTTL)4 | 3 | 35 | 98 |
| 2 | 1,2-DCE | Rh2(S-NTTL)4 | 3 | 65 | 97 |
| 3 | CHCl3 | Rh2(S-NTTL)4 | 3 | 90 | 99 |
| 4 | CHCl3 | Rh2(S-NTV)4 | 3 | 88 | 76 |
| 5 | CHCl3 | Rh2(S-NTTL)4 | 2 | 81 | 96 |
| 6b | CHCl3 | Rh2(S-NTTL)4 | 3 | 91 | 99 |
reaction performed at −60 °C;
triazole, the catalyst, and styrene were charged to the reaction vessel at the beginning of the reaction.

Interestingly, the imine intermediate 10 was never observed, regardless of the method of work-up. However, basic extraction from saturated sodium bicarbonate solution allowed for the almost quantitative recovery of base 6. It was also discovered that all of the components, except for the triflic anhydride, could be charged to the reaction vessel at the beginning of the reaction, without detriment to the yield, diastereo-, or enantioselectivity (entry 6), thus further simplifying the experimental procedure. This approach also allowed a visual readout of the reaction progress: the purple solution of a complex between the NH-triazole and rhodium catalyst turned green upon the addition of triflic anhydride, indicating that all NH-triazole had been consumed in the reaction.
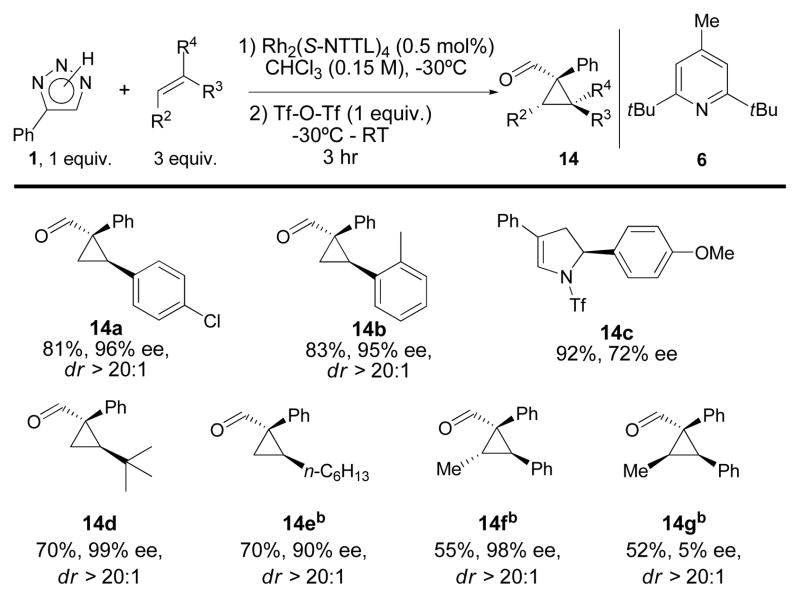
The scope of the process with respect to the alkene component was next examined (Scheme 1). 2-Methyl- and 4-chloro styrene gave good yields, as well as excellent ee and dr. However, the electron-rich 4-methoxystyrene gave only 2,3-dihydro-pyrrole 14c in 92% yield, but with a lower ee of 72%. The reaction of t-butylethene was equally high yielding and selective. Unfortunately, when the less reactive 1-octene was used in the reaction, the product cyclopropane 14e was isolated in only 35% yield, albeit with excellent ee and dr. Performing the reaction in carbon tetrachloride yielded 70% of 14e. Finally, trisubstituted alkenes gave acceptable yields of cyclopropanes only when the reactions were performed in carbon tetrachloride. (E)-Methyl styrene gave the corresponding cyclopropane 14f in moderate yield, but with excellent enantioselectivity. In contrast, (Z)-methyl styrene gave very low ee, ~5%, although diastereoselectivity was preserved (product 14g).
Scheme 1. Scope with respect to the olefin.a.
a Reactions were performed on 0.35 mmol scale (with respect to the triazole); bthese reactions were performed in CCl4.
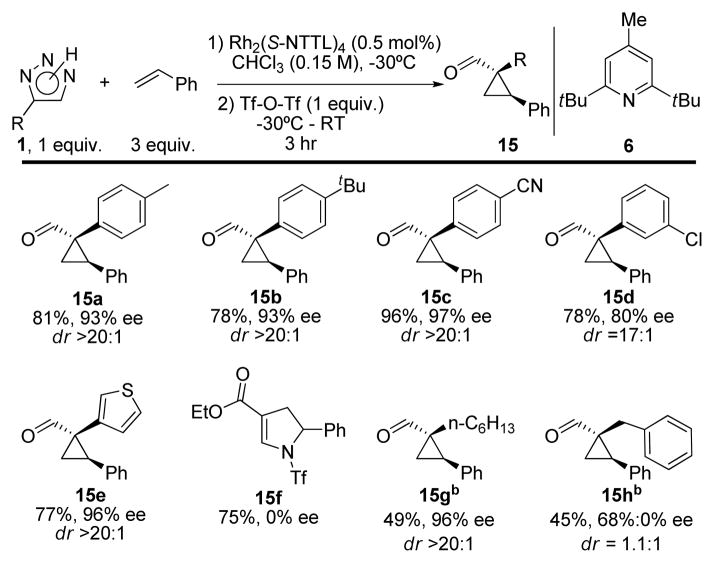
Variation of the substituent on the triazole ring revealed that the reaction proceeded well when p-methyl-, 15a, p-t-butyl-, 15b, and p-cyano-, 15c, 4-phenyl-NH-1,2,3-triazoles were used (Scheme 2). Both diastereo- and enantioselectivity were excellent. However, when the m-chlorophenyl-NH triazole was used, the dr of the product 15d eroded to 17:1, and ee was 80%. We hypothesize that this was due to unfavorable steric interaction of the meta-substituent with the catalyst. When the benzene ring was replaced by a thiophene, the reaction proceeded in a good yield, with excellent ee and dr (15e). However, introduction of an ester functionality caused the formation of 2,3-dihydropyrrole 15f in good yield, but unlike 1,2-dihydropyrrole 14c, racemic 15f was obtained in 75%, suggesting that different mechanisms are responsible for the formation of these products. 4-Alkyl-NH -1,2,3-triazole could also be used in the reaction, albeit the yields of cyclopropane were lower due to competing intramolecular 1,2-hydride shift in the carbene complex.8 Despite the lower yield, 4-octyl triazole produced cyclopropane 15g in excellent enantio- and diastereoselectivity. Unfortunately, benzyl NH-triazole yielded a 1.1:1 mixture of diastereomers, with the major diastereomer having an ee of 68%, and the minor found to be racemic (product 15h).
Scheme 2. Scope with respect to the triazole.a.
a Reactions were performed on 0.35 mmol scale (with respect to the triazole); bthese reactions were performed in CCl4.
In conclusion, the highly reactive rhodium(II) N-triflyl azavinyl carbenes can be generated from NH-1,2,3-triazoles by the treatment of the latter with triflic anhydride in the presence of rhodium(II) carboxylate complexes. These carbene intermediates efficiently engage olefins in highly enantio- and diastereoselective transformations, providing convenient access to homochiral cyclopropane carboxaldehydes and 2,3-dihydropyrroles. Further studies of their reactivity are underway in our laboratories.
Supplementary Material
Acknowledgments
Financial support of this work by the National Institute of General Medical Sciences, National Institutes of Health (GM087620) is gratefully acknowledged.
Footnotes
Supporting Information Available: Experimental details, characterization data and NMR spectral charts. This material is available free of charge via the Internet at http://pubs.acs.org
References
- 1.Doyle MP, McKervey M, Ye T. Modern catalytic methods for organic synthesis with diazo compounds: from cyclopropanes to ylides. Wiley: New York; 1998. [Google Scholar]
- 2.Doyle MP. In: In Reactive Intermediate Chemistry. Moss RA, Platz MS, Jones M Jr, editors. Wiley: New York; 2004. pp. 561–592. [Google Scholar]; Davies HML, Manning JR. Nature. 2008;451:417. doi: 10.1038/nature06485. [DOI] [PMC free article] [PubMed] [Google Scholar]; Davies HML, Beckwith REJ. Chem Rev. 2003;103:2861. doi: 10.1021/cr0200217. [DOI] [PubMed] [Google Scholar]
- 3.Maas G. Angew Chem Int Ed. 2009;48:8186. doi: 10.1002/anie.200902785. [DOI] [PubMed] [Google Scholar]
- 4.Horneff T, Chuprakov S, Chernyak N, Gevorgyan V, Fokin VV. J Am Chem Soc. 2008;130:14972. doi: 10.1021/ja805079v. [DOI] [PMC free article] [PubMed] [Google Scholar]; Chuprakov S, Kwok SW, Zhang L, Lercher L, Fokin VV. J Am Chem Soc. 2009;131:18034. doi: 10.1021/ja908075u. and references cited therein. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tomé AC. In: In Science of Synthesis: Houben-Weyl Methods of Molecular Transformations. Storr R, Gilchrist T, editors. Vol. 13. Thieme; New York: 2003. pp. 544–545. [Google Scholar]; Kalisiak J, Sharpless KB, Fokin VV. Org Lett. 2008;10:3171. doi: 10.1021/ol8006748. [DOI] [PubMed] [Google Scholar]; Loren JC, Krasinski A, Fokin VV, Sharpless KB. Synlett. 2005:2847. [Google Scholar]
- 6.Kwok SW, Hein JE, Fokin VV, Sharpless KB. Heterocycles. 2008;76:1141. doi: 10.3987/COM-08-S(N)73. [DOI] [PMC free article] [PubMed] [Google Scholar]; Yamauchi M, Miura T, Murakami M. Heterocycles. 2010;80:177. [Google Scholar]
- 7.Müller P, Bernardinelli G, Allenbach YF, Ferri M, Flack HD. Org Lett. 2004;6:1725. doi: 10.1021/ol049554n. [DOI] [PubMed] [Google Scholar]; Müller P, Lacrampe F, Bernardinelli G. Tetrahedron: Asymmetry. 2003;14:1503. [Google Scholar]
- 8.Panne P, DeAngelis A, Fox JM. Org Lett. 2008;10:2987. doi: 10.1021/ol800983y. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.