Abstract
Background:
Oxygen exposure during delivery room (DR) resuscitation, even when brief, is potentially toxic. A practice plan (PP) was introduced for very low birth weight (VLBW) infants ⩽1500 g as follows: initial FiO2 from 0.21 to 1.0 using blenders, oxygen guided by oximetry to maintain saturation between 85% to 95% from birth.
Objective:
To determine whether the initiating FiO2 could be safely lowered, and by doing so whether the number of infants with a PaO2 >80 mm Hg could be minimized on admission, as well as lowering oxygen requirement at 24 h.
Methods:
In all, 53 infants admitted between June 2006 and June 2007 were evaluated and compared with 47 infants from 2004 managed with 100 % oxygen (historical comparison group (HC)).
Result:
Stabilization/Resuscitation included intubation (n=28) and continuous positive airway pressure (CPAP) (n=25); no cardiopulmonary resuscitation (CPR). The heart rate increased rapidly in all cases. The initiating FiO2 decreased from 0.42 to 0.28 over 12 months (P=0.00005); 14 (26%) were resuscitated with room air. Correspondingly, the pH increased from 7.24 to 7.30 (P=0.002) and PCO2 decreased from 53 to 41 (P=0.001). A comparison of infants during the PP with the HC revealed that 36/53 versus 21/47 had an initial PaO2 <80 mm Hg (P=0.02); the median PaO2, that is, 64 versus 86 and saturation, that is, 95% versus 99% on admission were significantly lower. The median FiO2 at 24 h was 0.25 versus 0.40.
Conclusion:
DR resuscitation of VLBW infants can be initiated with less oxygen even with room air without concomitant overt morbidity. This change was associated with more infants with an initial PaO2 <80 mm Hg and lower saturation values on admission as well as a lower FiO2 requirement at 24 h.
Keywords: delivery room resuscitation, supplemental oxygen, room air, premature infants, pulse oximetry
Introduction
There is increasing evidence to suggest that oxygen exposure during neonatal resuscitation is potentially more toxic than considered earlier. Given that the partial pressure of oxygen in the fetus before delivery approximates 27 mm Hg, even brief (minutes) relative hyperoxic exposure immediately after birth may trigger long-term detrimental effects. Indeed, numerous experimental and clinical studies have shown the deleterious effects of resuscitation with 100% oxygen. Initial animal studies showed that room air as compared with pure oxygen is as efficient in restoring metabolism, blood pressure, cardiac output, pulmonary blood flow and cerebral blood flow.1, 2, 3, 4, 5, 6, 7, 8, 9 However, more recent studies have shown that the effect on the pulmonary and cerebral circulations is indeed more complex with a more favorable early vascular response, that is, increased blood flow noted with supplemental oxygen use when compared with room air.10, 11, 12, 13 Clinical studies have shown that delivery room (DR) resuscitation with 100% oxygen when compared with room air is associated with a lower 5-min Apgar score, a prolonged time to first cry and breath,14, 15 increased neonatal mortality,16, 17 increased oxidative stress that persisted for at least 4 weeks after birth in one study,18, 19 increased myocardial and kidney injury 20 and also with a higher risk for childhood leukemia and cancer.21, 22 These studies were largely undertaken in underdeveloped countries and primarily involved term babies.
This led to changes by the American Heart Association (AHA) and the American Academy of Pediatrics (AAP) in the guideline recommendations for the use of oxygen during newborn resuscitation23, 24 The guidelines suggested that supplemental oxygen ranging from room air to 100%, could be used to initiate resuscitation with subsequent delivery to be guided by pulse oximetry. However, unresolved questions particularly in the premature infant, included the optimal PaO2 level as well as the desired oxygen saturation range necessary to reduce the risk of oxygen toxicity. It had been suggested that to achieve this goal, the PaO2 be maintained <80 mm Hg25, 26 and the saturations be maintained between 85% and 95%.27, 28, 29 On the basis of these new neonatal resuscitation program (NRP) guidelines, a practice plan (PP) was introduced in our program in June 2006 for infants of birth weight (BW) ⩽1500 g as follows: stabilization/ resuscitation could be initiated with an FiO2 from 0.21 to 1.0 using blenders; oxygen delivery was to be guided by pulse oximetry to maintain saturation values between 85% and 95%. The objectives of the PP were to determine (1) whether the initiating FiO2 could be safely lowered during DR resuscitation, (2) whether there would be less infants with a PaO2 >80 mm Hg on admission to the neonatal intensive care unit (NICU) and (3) whether the requirement for oxygen at 24 h of life would be reduced.
Methods
To obtain baseline information with regard to DR management at our institution, we retrospectively analyzed the medical records of 47 consecutive inborn infants of birth weight (BW) <1500 g, who required stabilization and/or resuscitation in the DR and were admitted to the NICU at New York Presbyterian Hospital between January and September 2004 (termed historical comparison group (HC)). Infants with congenital abnormalities and those who required chest compressions were excluded. The practice at the time was to use 100% oxygen for resuscitation and during transport to the NICU; pulse oximetry was not routinely used in the DR. It is noted that 39 (83%) of the infants had saturation values >95% on admission to the NICU, with an initial PaO2 of 117±88 mm Hg as compared with eight (17%) who had a saturation value <95% with an initial PaO2 of 59±17 mm Hg (P=0.02) On the basis of the retrospective analysis and consistency with the new AHA/AAP guidelines, the neonatal division reached consensus and adopted a PP for infants of estimated BW <1500 g who required stabilization and/or resuscitation. Infants with known or obvious congenital abnormalities were excluded, and those infants who received chest compressions were expected to be resuscitated with 100% FiO2. The PP included the following: (1) Provision of blended oxygen to deliver an initiating FiO2 of between 0.21 and 1.0—the initial FiO2 was left to the discretion of the provider, (2) Oxygen delivery was to be guided by postductal pulse oximetry to achieve saturation values of between 85% and 95% from birth through transport to the NICU. The one caveat was that there had to be a rapid increase in the heart rate, that is, >100 beats/min (within 30 s of initiating respiratory support) in those cases where the initial assessment of the heart rate was less than 80 beats per min.30 The goal was to achieve saturations that approximated 85% by 3 min, and if this was not achieved then the oxygen was increased incrementally until this goal was met. Blenders were placed in each DR and on the neonatal transport isolette to deliver the desired oxygen concentration. Pulse oximetry was determined using a Nellcor transport monitor (Paragon Medical, Coral Springs, Florida, USA), which does not have the capacity to record data. The probe was placed post-ductally in all cases. The transport time from the DR to the NICU is approximately 5 min. As part of standard practice, an arterial blood gas is obtained within the first 30–60 min following an admission to the NICU. Data retrieved included BW; gestational age; initial DR and admission FiO2; saturation values on NICU admission and at 24 h; initial PaO2, PCO2, pH, DR respiratory support, that is, continuous positive airway pressure (CPAP) or intubation. All data were collected in a standardized manner over 12 months, starting in June 2006 through June 2007.
As this was a major shift in clinical practice, an intensive educational program was implemented as follows: in-services were provided to all neonatal physicians and nurses, respiratory therapists, obstetrical physicians and labor and delivery nurses before implementation. The data were reviewed every 3 months by the entire neonatal division at a designated meeting, with the primary goal of identifying any potential adverse acute consequences of lowering the initiating oxygen concentration and specifically evidence for hypercarbia or acidosis. In addition, providers were given the opportunity to voice concerns and/or suggestions to improve the practice plan.
Statistical analysis
The primary outcome was a reduction in the number of infants with hyperoxia, defined as PaO2 value >80 mm Hg. The data were analyzed for the entire cohort and then subsequently for two groups based on gestational age, that is, <28 wks and ⩾28wks, using Student's t-tests, chi-square analysis, analysis of variance (ANOVA) for repeat measures where appropriate. A P-value of <0.05 was considered to be statistically significant. All values are presented as mean ±s.d., unless as stated otherwise.
The Institutional Review Board of Weill Cornell Medical College approved the PP and the collection of data.
Results
General
The 53 infants evaluated were of BW1052±259 g, gestational age 28.3±2.5 wks, which was comparable to the HC of infants, that is, 1131±273 g and gestational age 28.9±2.2 wks. Of the 53 infants, 21 were <28 wks. Stabilization and/or resuscitation included bag-mask ventilation and intubation (n=28) and CPAP only (n=25). No infant received cardiopulmonary resuscitation (CPR). The heart rate increased rapidly in all cases with respiratory support, irrespective of the initiating FiO2 concentration.
Changes during the PP
Initiating FiO2 concentration in the delivery room
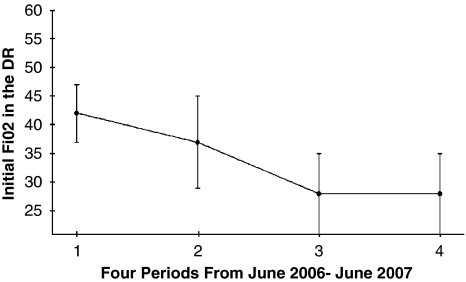
The initiating FiO2 concentration was 0.50 (n=6), 0.40 (n=16), 0.30 (n=16), 0.25 (n=1) and room air (n=14). There was a progressive decrease in the initiating FiO2 from 0.42±6 to 0.28±7 (P=0.00005) during the PP. (Figure 1). The initiating FiO2 used for stabilization/ resuscitation did not differ among VLBW infants <28 wks (n=21) versus ⩾28 wks (n=32), that is, 0.34±0.1 versus 0.31±0.1, respectively.
Figure 1.
Effect of change in practice on starting FiO2 in the delivery room (DR). The 12 months were divided into four periods: Period 1: June 2006–August 2006; Period 2: September 2006–November2006; Period 3: December 2006–February 2007; Period 4: March 2007–June 2007.
Pulse oximetry detection of heart rate and saturation in the delivery room
The heart rate was rapidly detected in all cases with pulse oximetry (within the first minute). In general, saturation values increased slowly to the desired range of 85%–95% over 5–10 min. Twenty-nine (55%) infants achieved the target range of 85%–95% during stabilization/resuscitation and transport to the NICU. Twenty-four infants had saturation values >95% eight (33%) of these infants were always in room air. The initiating FiO2 in the delivery room and the FiO2 concentration on admission to the NICU in infants with saturation values > 95% were comparable, that is, 31±8 versus 37±26 mm Hg, respectively (NS).
Initial FiO2 concentration on admission to the NICU
The admitting FiO2 concentration was lower than the initiating FiO2 in the DR in 16 (31%) infants; it remained the same in 12 (22%) infants and increased in 25 (47%) infants. Overall, 10 (19%) infants were in room air and nine (17%) in 100% oxygen on admission to the NICU. During the PP, the admitting FiO2 decreased from 0.70±0.32 to 0.38±0.21 (P=0.02).
Admission PaO2 values
Thirty-seven (70%) infants had an admitting PaO2 concentration <80 mm Hg. The initial PaO2 concentration was lower, that is, 60±16 versus 103±59 mm Hg (P=0.0001) when the saturation values were <95 versus ⩾95%, respectively. More infants with an admitting PaO2 >80 mm Hg, that is 16/30 (53%) versus 2/23 (8.6%) were likely to have saturation values ⩾95 versus <95% (P=0.0009), respectively.
FiO2 values at 24 h of age
The FiO2 at 24 h of life was higher in infants <28 weeks versus ⩾28 weeks, that is, 0.38±0.23 versus 0.24±0.05 for P=0.0007.
PCO2 and pH changes during the PP
The PCO2 decreased from 53±10 to 41±12 (P=0.001) and the pH increased from 7.24±0.07 to 7.3±0.1 (P=0.002) during the PP.
Comparison of the PP values to the HC
The median admitting PaO2 concentration, that is, 64 versus 86 mm Hg (P=0.01), as well as the saturation values, that is, 95% versus 99% (P=0.0001) was lower during the PP when compared with the HC. Table 1 In addition, significantly more infants managed with the PP versus the HC, that is,37 of 53 (70%) versus 21 of 47 (44%) had an initial PaO2 <80 mm Hg (P=0.02). The median FiO2 at 24 h was lower with the PP versus HC, that is, 0.25 versus 0.40 (P=0.0005) However, the FiO2 at 24 h for infants <28 wks was higher with the PP versus HC, that is, 0.32±0.10 versus 0.26±0.08 (P=0.05), but was significantly lower for infants ⩾28 wks, that is, 0.24±0.05 versus 0.28±0.05 (P=0.01), respectively.
Table 1. Impact of change in the practice plan on PaO2 saturation (Sats) values, FiO2, PCO2 and pH in comparison to the historical comparison group (2004).
| Characteristic | 2006/2007 | 2004 | P-value |
|---|---|---|---|
| PaO2 mm Hg Median | 64 | 86 | 0.01 |
| Sats upon NICU admission median | 95% | 99% | 0.0001 |
| PaO2 <80 mm Hg | 37/53 (70%) | 21/47 (44%) | 0.02 |
| All infants FiO2 at 24 h | 0.25 | 0.40 | 0.0005 |
| <28wks | 0.38±0.23 | 0.26±0.08 | P=0.0007 |
| ⩾28wks | 0.24±0.05 | 0.28±0.05 | |
| PCO2 mm Hg | 45±12 | 47±13 | NS |
| pH | 7.27±0.06 | 7.26±0.10 | NS |
Abbreviations: NICU, neonatal intensive care unit; Sats, saturation values.
Discussion
The observations contained in this report indicate that DR stabilization/resuscitation of VLBW infants can be initiated with an FiO2 of less than 100% and even with room air without concomitant early overt morbidity. Lowering the initiating FiO2 was associated with more VLBW infants with an initial PaO2 of less than 80 mm Hg and lower saturation values on admission to intensive care, as well as a lower FiO2 requirement at 24 h as compared with HC. These observations add support to two recently completed randomized studies in premature infants showing that resuscitation can be safely initiated with less than 100% oxygen.31, 32
The most recent AHA/AAP guidelines for oxygen administration in the DR were non–specific, and recommended that resuscitation could be initiated with varying oxygen concentration from room air to 100%.23 It is interesting to note that at the onset of the PP, providers elected to use an initial FiO2 concentration that approximated 40%. This gradually decreased to less than 30% after 12 months, a concentration that is similar to the starting concentration suggested by Escrig et al.31
Consistent with the new guideline recommendations, pulse oximetry was used to modulate oxygen delivery with a target goal of maintaining saturation values between 85% and 95%. Once the technique was established, providers considered pulse oximetry invaluable for several reasons. First, the heart rate was readily detected in a timely manner in all cases, that is, usually within 60 s after birth and it was easier to follow than either auscultation or palpation. This is important as a rapid increase in the heart rate above 100 beats per min is regarded as the most effective assessment of adequate ventilation and thus effective resuscitation.23, 24, 30 Second, oximetry was essential in guiding oxygen delivery decision-making. Thus, to achieve the target saturation range, the initiating FiO2 concentration was reduced or remained the same in half the cases, whereas the oxygen concentration was increased in the remaining infants. In many cases, saturation values reached the low target range of 85% slowly, usually 5–10 min after initiating resuscitation; this slow increase in saturation is consistent with earlier data in both preterm and term infants.27, 28, 29, 31, 32
As a consequence of using the target range, significantly more infants (70%) had an initial PaO2 concentration <80 mm Hg as compared with the HC (44%). Moreover, the FiO2 on admission to the NICU decreased by 45% from a mean of 0.70–0.38 over the 12 months. When the data were examined for all infants during the PP, the FiO2 on admission was not different between infants less or greater than 28 wks.
The FiO2 at 24 h for the cohort was significantly lower during the PP when compared with the HC. However, this was not a consistent finding, that is, the oxygen concentration was significantly lower in the larger premature infant >28 wks, whereas it was higher in the smallest infants. Although not entirely comparable, a similar finding was noted during the PP where the FiO2 was significantly lower at 24 h in the larger as compared with the smaller infants.
An interesting and unexplained observation was the decrease in PCO2 and increase in pH during the period of observation concomitant with the reduction in the initiating oxygen use in the DR. The mechanism/s accounting for this observation are unclear.
A major limitation of this report is that it was a small cohort, not randomized and thus lacked a control group. However, the findings are consistent with two recent randomized studies showing that DR resuscitation can be initiated ‘safely' with less than 100% oxygen. In the first of these studies, premature infants were assigned to either 90% or 30% oxygen to achieve a target saturation of 85% at 10 min of life. In the high group, the FiO2 was reduced to 45% and in the low group the FiO2 was increased to the same concentration to achieve the saturation goal. The authors suggest that resuscitation can be safely initiated with an FiO2 of 30%.31 In the second study, premature infants of <32 weeks were assigned to either 100% oxygen or room air with the goal of achieving saturation values of >70% by 3 min or 80 % by 5 min. All infants resuscitated with room air received supplemental oxygen with a mean concentration at 10 min of approximately 55%.32 There were no overt complications noted from starting with room air, although the sample size was small.
These cumulative observations that stabilization and/or resuscitation of premature infants in the DR can be initiated with lower oxygen concentrations, be sustained, and even reduced on admission and at 24 h, without overt untoward consequences, is exciting. However, it is essential to recognize that in half the cases the oxygen concentration had to be increased to achieve the target range and the positive impact, that is, a lower FiO2 concentration at 24 h appears to be predominantly in the larger premature infant. Moreover, initiation of this change in practice required intensive education of all providers, a team effort in the DR that included preventing heat loss, establishing effective respiratory support as indicated and placing the pulse oximeter probe to guide oxygen use in a timely and coordinated manner, that is, all within the first minute after hand-off of a VLBW infant.
What are the practical implications for the clinician? The data now strongly suggest that an FiO2 concentration of considerably less than 100% can be used to initiate resuscitation in the delivery room. It appears that 30% may be a good starting concentration, provided the heart rate is rapidly increasing or greater than 100 beats per min. The potential benefit/s of this change in delivery room practice requires careful longitudinal follow-up.
Conflict of interest
The authors declare no conflict of interest.
References
- Rootwelt T, Løberg EM, Moen A, Oyasaeter S, Saugstad OD. Hypoxemia and reoxygenation with 21 or 100% oxygen in newborn pigs: changes in blood pressure, base deficit, and hypoxanthine and brain morphology. Pediatr Res. 1992;32:107–113. doi: 10.1203/00006450-199207000-00021. [DOI] [PubMed] [Google Scholar]
- Rootwelt T, Odden JP, Hall C, Ganes T, Saugstad OD. Cerebral blood flow and evoked potentials during reoxygenation with 21 or 100% O2 in newborn pigs. J Appl Physiol. 1993;75:2054–2060. doi: 10.1152/jappl.1993.75.5.2054. [DOI] [PubMed] [Google Scholar]
- Rootwelt T, Odden JP, Hall C, Saugstad OD. Regional blood flow during severe hypoxemia and resuscitation with 21 or 100% in newborn pigs. J Perinat Med. 1996;24:227–236. doi: 10.1515/jpme.1996.24.3.227. [DOI] [PubMed] [Google Scholar]
- Poulsen JP, Øyasæter S, Saugstad OD. Hypoxanthine, xanthine, and uric acid in newborn pigs during hypoxemia followed by resuscitation with room air or 100% oxygen. Crit Care Med. 1993;21:1058–1065. doi: 10.1097/00003246-199307000-00024. [DOI] [PubMed] [Google Scholar]
- Tølløfsrud PA, Solås AB, Saugstad OD. Newborn piglets with meconium aspiration resuscitated with room air or 100% oxygen. Pediatr Res. 2001;50:423–429. doi: 10.1203/00006450-200109000-00020. [DOI] [PubMed] [Google Scholar]
- Bagenholm R, Hagberg H, Kjellmer I. Impact of reoxygenation with oxygen and air on the extent of the brain damage after hypoxia-ischemia in neonatal rats. Acta Paediatr. 1996;85:1228–1231. doi: 10.1111/j.1651-2227.1996.tb18234.x. [DOI] [PubMed] [Google Scholar]
- Medbo S, Yu XQ, Asberg A, Saugstad OD. Pulmonary hemodynamics and plasma endothelin-1 during hypoxemia and reoxygenation with room air or 100% oxygen in a piglet model. Pediatr Res. 1998;44:843–849. doi: 10.1203/00006450-199812000-00004. [DOI] [PubMed] [Google Scholar]
- Feet BA, Brun NC, Hellstrom-Westas L, Svenningsen NW, Greisen G, Saugstad OD. Early cerebral metabolic and electrophysiological recovery during controlled hypoxemic resuscitation in piglets. J Appl Physiol. 1998;84:1208–1216. doi: 10.1152/jappl.1998.84.4.1208. [DOI] [PubMed] [Google Scholar]
- Børke WB, Munkeby BH, Mørkrid L, Thaulow E, Saugstad OD. Resuscitation with 100% O2 does not protect the myocardium in hypoxic newborn piglets. Arch Dis Child Fetal Neonatal Educ. 2004;89:F156–F160. doi: 10.1136/adc.2002.020594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lakshminrusimha S, Russell JA, Steinhorn RH, Ryan R, Gugino S, Morin FC, et al. Pulmonary artery contractility increases with 100% oxygen resuscitation. Pediatr Res. 2006;59:137–141. doi: 10.1203/01.pdr.0000191136.69142.8c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lakshminrusimha S, Russell JA, Steinhorn RH, Swartz DD, Ryan RM, Gugino SF, et al. Pulmonary hemodynamics in neonatal lambs resuscitated with 21, 50 and 100% oxygen. Pediatr Res. 2007;62:313–318. doi: 10.1203/PDR.0b013e3180db29fe. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solas AB, Kalous P, Saugstad OD. Resuscitation with 100 or 21% oxygen after cerebral hypoxemia- ischemia hypercapnia in newborn piglets. Biol Neonate. 2004;85:105–111. doi: 10.1159/000074966. [DOI] [PubMed] [Google Scholar]
- Presti AL, Kishkurno SV, Slinko SK, Randis TA, Ratner VI, Polin RA, et al. Reoxygenation with 100% oxygen or room air. Late neuroanatomical and neurofunctional outcome in neonatal mice with hypoxic-ischemic brain injury. Pediatr Res. 2006;60:55–59. doi: 10.1203/01.pdr.0000223766.98760.88. [DOI] [PubMed] [Google Scholar]
- Ramji S, Ahuja S, Thirupuram S, Rootwelt T, Rooth G, Saugstad OD. Resuscitation of asphyxic newborn infants with room air or 100% oxygen. Pediatr Res. 1993;34:809–812. doi: 10.1203/00006450-199312000-00023. [DOI] [PubMed] [Google Scholar]
- Saugstad OD, Rootwelt T, Aalen O. Resuscitation of asphyxiated newborn infants with room air or oxygen: An international controlled trial: The Resair 2 study. Pediatrics. 1998;102:1–7. doi: 10.1542/peds.102.1.e1. [DOI] [PubMed] [Google Scholar]
- Tan A, Schulze A, O'Donnell CFP, Davis PG.Air versus oxygen for reuscitation of newborns at birth (Cochrane Review) The Cochrane libraryIssue 3John Wiley & Sons, Ltd: Chishester UK; 2004 [Google Scholar]
- Saugstad OD, Ramji S, Soll RF, Vento M. Resuscitation of newborn infants with 21 or 100% oxygen; an updated systematic review and meta-analysis. Neonatology. 2008;94:176–182. doi: 10.1159/000143397. [DOI] [PubMed] [Google Scholar]
- Vento M, Asensi M, Sastre J, García-Sala F, Pallardó FV, Vina J. Resuscitation with room air instead of 100% oxygen prevents oxidative stress in moderately asphyxiated term neonates. Pediatrics. 2001;107:642–647. doi: 10.1542/peds.107.4.642. [DOI] [PubMed] [Google Scholar]
- Vento M, Asensi M, Sastre J, Lloret A, Garcia-Sala F, Vina J. Oxidative stress in asphyxiated infants resuscitated with 100% oxygen (published correction appears in J Pediatr. 2003; 142; 616) J Pediatr. 2003;142:240–246. doi: 10.1067/mpd.2003.91. [DOI] [PubMed] [Google Scholar]
- Vento M, Sastre J, Asensi MA, Vina J. Room-air resuscitation causes less damage to heart and kidney than 100% oxygen. Am J Respir Crit Care Med. 2005;172:1393–1398. doi: 10.1164/rccm.200412-1740OC. [DOI] [PubMed] [Google Scholar]
- Namburg E, Bellocco R, Cnattingius S, Jonzon A, Ekbom A. Supplementary oxygen and risk of childhood leukemia. Acta Paediatr. 2002;91:1328–1333. doi: 10.1080/08035250216105. [DOI] [PubMed] [Google Scholar]
- Spector LG, Klebanoff MA, Feusner JH, Georgieff MK, Ross JA. Childhood cancer following neonatal oxygen supplementation. J Pediatr. 2005;147:27–31. doi: 10.1016/j.jpeds.2005.03.008. [DOI] [PubMed] [Google Scholar]
- American Heart Association Neonatal Resuscitation Guidelines. Circulation. 2005;112 suppl:IV-188–IV-195. [Google Scholar]
- International Liaison Committee on Resuscitation 2005 International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science With Treatment Recommendations. Part 7; Neonatal Resuscitation. Resuscitation. 2005;67:293–303. doi: 10.1016/j.resuscitation.2005.09.014. [DOI] [PubMed] [Google Scholar]
- Taeush HW, Ballard RA, Geason CA.Avery's Disease of the Newborn8th edn.Elsevier Saunders, Philadelphia, PA; 20051540 [Google Scholar]
- American Academy of Pediatrics, American College of Obstetricians and Gynecologists Guidelines for Perinatal care4th edn.American Academy of Pediatrics, Elk Grove Village IL; 1997 [Google Scholar]
- Kamlin COF, O'Donnell CPF, Davis PG, Morley CJ. Oxygen saturation in healthy infants immediately after birth. J Pediatr. 2006;148:585–589. doi: 10.1016/j.jpeds.2005.12.050. [DOI] [PubMed] [Google Scholar]
- O'Donnell CPF, Kamlin COF, Davis PG, Morley CJ. Feasibility of and delay in obtaining pulse oximetry during neonatal resuscitation. J Pediatr. 2005;147:698–699. doi: 10.1016/j.jpeds.2005.07.025. [DOI] [PubMed] [Google Scholar]
- Rabi Y, Yee W, Chen SY, Singhal N. Oxygen saturation trends immediately after birth. J Pediatr. 2006;148:590–594. doi: 10.1016/j.jpeds.2005.12.047. [DOI] [PubMed] [Google Scholar]
- Palme-Kilander C, Tunell R. Pulmonary gas exchange during face mask ventilation immediately after birth. Arch Dis Child. 1993;68:11–16. doi: 10.1136/adc.68.1_spec_no.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Escrig R, Arruza L, Izquierdo I, Villar G, Sáenz P, Gimeno A, et al. Achievement of targeted saturation values in extremely low gestational age neonates resuscitated with low of high oxygen concentrations: A prospective randomized trial. Pediatr. 2008;121:875–881. doi: 10.1542/peds.2007-1984. [DOI] [PubMed] [Google Scholar]
- Wang CL, Anderson C, Leone TA, Rich W, Govindaswami B, Finer NN. Resuscitation of preterm neonates by using room air or 100% oxygen. Pediatr. 2008;121:1083–1089. doi: 10.1542/peds.2007-1460. [DOI] [PubMed] [Google Scholar]