Abstract
Adult zebrafish exhibit hyperventilatory responses to absolute environmental CO2 levels as low as 0.13% ( mmHg), more than an order of magnitude lower than the typical arterial
mmHg), more than an order of magnitude lower than the typical arterial  levels (∼40 mmHg) monitored by the mammalian carotid body. The sensory basis underlying the ability of fish to detect and respond to low ambient CO2 levels is not clear. Here, we show that the neuroepithelial cells (NECs) of the zebrafish gill, known to sense O2 levels, also respond to low levels of CO2. An electrophysiological characterization of this response using both current and voltage clamp protocols revealed that for increasing CO2 levels, a background K+ channel was inhibited, resulting in a partial pressure-dependent depolarization of the NEC. To elucidate the signalling pathway underlying K+ channel inhibition, we used immunocytochemistry to show that these NECs express carbonic anhydrase (CA), an enzyme involved in CO2 sensing in the mammalian carotid body. Further, the NEC response to CO2 (magnitude of membrane depolarization and time required to achieve maximal response), under conditions of constant pH, was reduced by ∼50% by the CA-inhibitor acetazolamide. This suggests that the CO2 detection mechanism involves an intracellular sensor that is responsive to the rate of acidification associated with the hydration of CO2 and which does not require a change of extracellular pH. Because some cells that were responsive to increasing
levels (∼40 mmHg) monitored by the mammalian carotid body. The sensory basis underlying the ability of fish to detect and respond to low ambient CO2 levels is not clear. Here, we show that the neuroepithelial cells (NECs) of the zebrafish gill, known to sense O2 levels, also respond to low levels of CO2. An electrophysiological characterization of this response using both current and voltage clamp protocols revealed that for increasing CO2 levels, a background K+ channel was inhibited, resulting in a partial pressure-dependent depolarization of the NEC. To elucidate the signalling pathway underlying K+ channel inhibition, we used immunocytochemistry to show that these NECs express carbonic anhydrase (CA), an enzyme involved in CO2 sensing in the mammalian carotid body. Further, the NEC response to CO2 (magnitude of membrane depolarization and time required to achieve maximal response), under conditions of constant pH, was reduced by ∼50% by the CA-inhibitor acetazolamide. This suggests that the CO2 detection mechanism involves an intracellular sensor that is responsive to the rate of acidification associated with the hydration of CO2 and which does not require a change of extracellular pH. Because some cells that were responsive to increasing  also responded to hypoxia with membrane depolarization, the present results demonstrate that a subset of the NECs in the zebrafish gill are bimodal sensors of CO2 and O2.
also responded to hypoxia with membrane depolarization, the present results demonstrate that a subset of the NECs in the zebrafish gill are bimodal sensors of CO2 and O2.
Introduction
In a wide range of animal species, the ability to sense environmental CO2 plays an integral role in homeostasis. In mammals, CO2 sensing occurs centrally (brainstem neurons) and in the periphery (carotid body) (O’Regan & Majcherczyk, 1982; Gonzalez et al. 1992; Lahiri & Forster, 2003). The Type 1 glomus cells of the carotid body appear to be the primary sensors of CO2 in the peripheral circulation (Lahiri & Forster, 2003). Although it has been long thought that peripheral CO2 sensing in vertebrates first evolved in air-breathers, evidence is mounting that fish also sense CO2 in their environment (Burleson & Smatresk, 2000; Reid et al. 2000; Sundin et al. 2000; McKendry et al. 2001; Perry & Reid, 2002; see reviews by Milsom et al. 1999; Gilmour, 2001; Perry & Gilmour, 2002; Gilmour & Perry, 2007). The gill, which is typically the predominant site of respiratory gas transfer in fish as well as the established site of O2 chemoreception (Smith & Jones, 1978), is now also recognized as the principal site of CO2 sensing (Perry & Gilmour, 2002; Gilmour & Perry, 2007). The similarity between the gill and carotid body in the sensing of gases is consistent with the evolutionary origin of the carotid body, namely the chemoreceptor cells of the first branchial arch (Milsom, 2002; Milsom & Burleson, 2007).
Although fish, like other vertebrates, possess central and peripheral (gill) CO2 chemoreceptors, the central receptors appear to be confined to air-breathing species (Milsom, 2002). Based on the few whole animal studies that have been performed, it would appear that gill chemoreceptors are specifically sensitive to CO2 rather than H+ (Perry et al. 1999; Reid et al. 2000; Sundin et al. 2000; Perry & McKendry, 2001; McKendry & Perry, 2001; Gilmour et al. 2005). Unlike air breathers, which maintain high  levels (e.g. ∼40 mmHg in mammals) in arterial blood relative to ambient air, fish achieve arterial
levels (e.g. ∼40 mmHg in mammals) in arterial blood relative to ambient air, fish achieve arterial 
 levels which are near ambient and approximately an order of magnitude lower (∼2–3 mmHg). Thus, CO2 detection systems in fish presumably have evolved to sense deviations in
levels which are near ambient and approximately an order of magnitude lower (∼2–3 mmHg). Thus, CO2 detection systems in fish presumably have evolved to sense deviations in  from a very low set point. Moreover, because of the log-linear relationship between pH and
from a very low set point. Moreover, because of the log-linear relationship between pH and  , equivalent changes in
, equivalent changes in  will produce larger changes in arterial pH in fish (resting
will produce larger changes in arterial pH in fish (resting  mmHg) than in mammals (resting
mmHg) than in mammals (resting  mmHg). The potentially disruptive effects of small changes in
mmHg). The potentially disruptive effects of small changes in  in fish on acid–base balance reinforces the need for piscine CO2 sensors to detect small changes in
in fish on acid–base balance reinforces the need for piscine CO2 sensors to detect small changes in  deviating from a low set point. Indeed, a recent study demonstrated hyperventilatory responses in zebrafish (Danio rerio) to environmental CO2 levels of 0.13% (
deviating from a low set point. Indeed, a recent study demonstrated hyperventilatory responses in zebrafish (Danio rerio) to environmental CO2 levels of 0.13% ( mmHg; Vulesevic et al. 2006), which would have increased
mmHg; Vulesevic et al. 2006), which would have increased  by approximately 1 mmHg. Thus, studying fish provides an opportunity to compare mechanisms of CO2 sensing in animal systems that have evolved under very different constraints; the low
by approximately 1 mmHg. Thus, studying fish provides an opportunity to compare mechanisms of CO2 sensing in animal systems that have evolved under very different constraints; the low  system in fish versus the high
system in fish versus the high  system in mammals.
system in mammals.
The neuroepithelial cell (NEC), first described in the fish gill by Dunel-Erb et al. (1982), was recently identified as an O2 chemoreceptor in zebrafish (Jonz et al. 2004) and channel catfish (Ictalurus punctatus) (Burleson et al. 2006) using patch clamp techniques. The NECs are enriched with neurotransmitters (Zaccone et al. 1989, 1992; Goniakowska-Witalinska et al. 1995; Zaccone et al. 1997, 2006), are extensively innervated (Bailly et al. 1992; Jonz & Nurse, 2003; Saltys et al. 2006) and bear striking resemblance to the glomus cells of the mammalian carotid body (Gonzalez et al. 1994) that are recognized to function as both O2 and CO2 sensors in mammals (Lahiri & Forster, 2003). These similarities, as well as the proposed evolutionary relationship between the carotid body and fish gill chemorecetors, suggest that NECs might also be sensitive to both O2 and CO2. Although there are convincing data demonstrating that NECs act as O2 sensors (Jonz et al. 2004; Burleson et al. 2006), evidence implicating NECs as CO2 chemoreceptors is lacking.
Here, we use patch clamp electrophysiology to show that a subset of NECs in the zebrafish gill, previously shown to be responsive to O2, can also act as sensitive CO2 sensors. The threshold  level is an order of magnitude lower than for both mammalian carotid body glomus cells and brainstem neurons (Guyenet, 2008). Further, we show that the transduction mechanism is mediated by a carbonic anhydrase (CA)-dependent inhibition of a background K+ conductance, and can occur independently of any change in extracellular pH.
level is an order of magnitude lower than for both mammalian carotid body glomus cells and brainstem neurons (Guyenet, 2008). Further, we show that the transduction mechanism is mediated by a carbonic anhydrase (CA)-dependent inhibition of a background K+ conductance, and can occur independently of any change in extracellular pH.
Methods
Animals
Adult zebrafish (Danio rerio) were obtained from a commercial supplier (MIRDO, Montreal, Canada) and transported to the University of Ottawa Aquatic Care Facility where they were maintained in acrylic tanks (4 l) supplied with aerated, dechloraminated City of Ottawa tap water at 28°C. Fish were maintained on a constant 10: 14 light–dark photoperiod. All procedures for animal use were approved by the University of Ottawa Animal Care and Veterinary Service and carried out according to institutional guidelines and in accordance with those of the Canadian Council on Animal Care (CCAC). The procedures comply with guidelines for reporting of ethical matters in The Journal of Physiology as outlined by Drummond (2009).
Cell isolation and immunocytochemistry
Zebrafish were decapitated after a sharp blow to the head. All procedures for isolation of NECs were carried out under sterile conditions in a laminar flow hood (Jonz et al. 2004). Gill baskets were removed and rinsed in a wash solution (2% penicillin–streptomycin in phosphate-buffered saline (PBS)) for 10 min. All eight gill arches were then separated and distal filaments rich in NECs (Jonz et al. 2004) were selectively removed. Tissue was placed in 0.01% hyaluronidase for 10 min then 0.25% trypsin/EDTA (Invitrogen Corp.) for 1 h at room temperature, minced with fine forceps and triturated in a 15 ml centrifuge tube with a Pasteur pipette. The trypsin reaction was stopped with the addition of 10% fetal calf serum (FCS). Lower concentrations of trypsin or mechanical separation alone did not work well for dissociation. The cell suspension was centrifuged (140 g) for 5 min and the pellet triturated in PBS. Gill cells were centrifuged once more with PBS and suspended in Leibovitz's (L-15, with l-glutamine but without phenol red) culture medium supplemented with 1% penicillin–streptomycin and 5% FCS.
For the immunocytochemical studies of NECs in culture, cells were plated on sterile CellBIND culture dishes (Corning Inc., New York; Ryan, 2005) for 24–48 h after dissociation, followed by fixation with 4% paraformaldehyde (PFA) in PBS for 15 min at room temperature and immunolabelled using mouse monoclonal anti-5-hydroxytryptamine (5HT) pre-diluted antibodies (1: 1, Biomeda Corp., Foster City, CA, USA) in conjunction with polyclonal human CA II antibodies (1:100, Rockland Immunochemicals, Gilbertsville, PA, USA). Monoclonal 5HT antibodies were visualized with goat anti-mouse secondary antibodies conjugated with Alexa 546 (1: 400, Molecular Probes, Eugene, OR, USA). Polyclonal CA antibodies were localized with goat anti-rabbit secondary antibodies conjugated with Alexa 488 (1: 400, Molecular Probes). All antibodies were diluted with PBS containing 0.5% Triton X-100 (PBST). Fixed cells were incubated in primary antibody for 2 h, followed by immersion in secondary antibody in darkness for 1 h at room temperature. Labelled cells were washed (3 × 5 min) and mounted with media containing 4′,6′-diamidino-2-phenylindole (DAPI) to stain nuclei (Vectashield, Vector Laboratories, Inc., Burlingame, CA, USA). Cells were observed and photographed using a Zeiss Axiophot fluorescence microscope and a Hamamatsu C5985 chilled CCD camera (East Syracuse, NY, USA). Images were captured using Metamorph imaging software (v. 4.01). Observations were performed using at least 10 cell culture dishes unless stated otherwise and the results obtained were consistently reproduced. To verify the specificity of the 5HT and CA antibodies, control experiments were conducted in which NECs were incubated with PBS lacking primary antibodies or immersed in primary antibodies preabsorbed for 30 min with excess antigen (40 μg μl−1 5HT for 5HT antibody or 50 μg μl−1 bovine CA for CA antibody). The specificity of the human CA II antibody against the zebrafish orthologue (zCAc; see Gilmour & Perry, 2009) was established in preliminary experiments in which western blotting revealed that the human antibody detected single zebrafish and rat proteins of similar size (29 kDa). Furthermore, a CA antibody (Georgalis et al. 2006) developed against rainbow trout cytosolic CA (tCAc; Esbaugh et al. 2005) was equally effective at detecting a 29 kDa zebrafish protein. We opted to use the human rather than the trout antibody in the present study because it yielded higher quality (less background) immunocytochemistry images.
Electrophysiology
Whole-cell recordings (Hamill et al. 1981) were performed in vitro using zebrafish gill cell culture preparations, essentially as described previously (Jonz et al. 2004). Specialized 35 mm culture dishes (see above, Corning Inc.) were plated with dissociated NECs. After 24 and 48 h, NECs that adhered to the culture substrate were identified using 2 mg ml−1 Neutral Red (NR; Sigma), a vital marker used to identify NECs (Jonz et al. 2004). Based on observation and measurement of NECs in vitro, larger NR-positive cells (≥ 7 μm in diameter) were selected for electrophysiological recording. Later on, dishes were mounted on a fixed stage of a Zeiss inverted microscope and a perfusion insert (Warner Instruments, LLC, Hamden, CT, USA) with inlet and outlet was placed in it. Patch electrodes were fabricated from borosilicate glass with filament (1.5 mm o.d.; 0.86 mm i.d.; 10 cm length; Sutter Instrument Co., Novato, CA, USA). Glass electrodes were pulled on a horizontal pipette puller (P-2000, Sutter Instrument Co.) and filled with pipette (intracellular recording) solution containing (mm): KCl 135, NaCl 5, CaCl2 0.1, EGTA 11, Hepes 10 and Mg-ATP 2; pH adjusted to 7.2 with KOH; resulting tip resistances were typically 5–7 MΩ. Bath (extracellular) solution contained (mm): NaCl 135, KCl 5, CaCl2 2, MgCl2 2, glucose 10 and Hepes 10; pH adjusted to 7.4 with NaOH. Seal resistance was typically more than 5 GΩ and holding current was less than 5 pA at −60 mV.
Voltage-clamp protocols were performed using a MultiClamp 700B amplifier (Molecular Devices, Sunnyvale, CA, USA) interfaced with an IBM PC, data acquisition system (DigiData 1322A, Molecular Devices) and Clampex 9.2 software (Molecular Devices). Under voltage-clamp, cells were typically held at −60 mV and currents were evoked either by changing the membrane potential from −100 mV (or −90 mV) to +60 mV over a period of 1 s (ramp protocol) or by delivering a step command (+30 mV for 50 ms) every 10 s from a holding potential of −60 mV. In the latter cases, steady-state current was measured as the average current evoked between 40 and 49 ms of each 50 ms voltage step interval. Current-clamp recordings were performed using the same apparatus. All recordings were filtered at 5 kHz and digitized at 10 kHz. In some cases, acquired signals were additionally filtered using an 8-pole Bessel filter (cut off at 200 Hz) to remove high frequency noise. All data were analysed using Clampfit 9.0 software (Molecular Devices). In Fig. 4B, the Goldman–Hodgkin–Katz current equation (eqn (14.5), p. 445, Hille, 2001) was used to describe the CO2-sensitive current as a function of membrane voltage, with PK= 2.5 × 10−6, [K+]i= 135 mm, [K+]o= 5 mm.
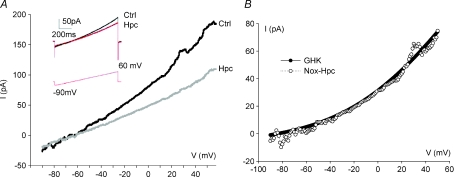
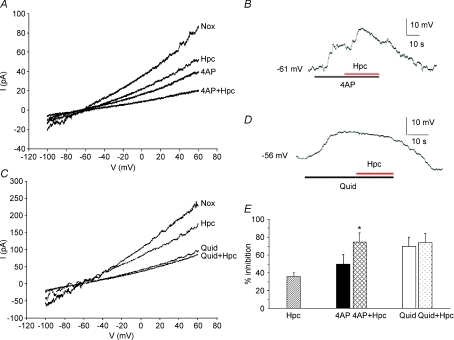
Figure 4. Current–voltage (I–V) relationship for a CO2-sensitive current in isolated cultured gill neuroepithelial cells (NECs) of zebrafish (Danio rerio).
A, mean current–voltage (I–V) relationships during exposure to normoxia (Nox) or hypercapnia (Hpc) from 12 cells. Currents were evoked by changing the voltage from −90 mV to +60 mV following a ramp protocol from a holding potential of −60 mV (inset). Inset also shows representative current traces from a single NEC. B, mean I–V relationship of the CO2-sensitive difference current (open symbols, Nox-Hpc) from 12 cells. The CO2-sensitive difference current  reverses near EK (calculated using bath and pipette K+ concentrations) and fits the corresponding Goldman–Hodgkin–Katz current equation (GHK, continuous curve; Hille, 2001), suggesting that it is predominantly carried by K+ ions.
reverses near EK (calculated using bath and pipette K+ concentrations) and fits the corresponding Goldman–Hodgkin–Katz current equation (GHK, continuous curve; Hille, 2001), suggesting that it is predominantly carried by K+ ions.
The recording chamber was continuously perfused (4 ml min−1) with bath solution at room temperature (22–24°C) using a constant pressure head and a four-channel remote valve control system (ALA Scientific Instruments Inc., Farmingdale, NY, USA). Hypoxia was produced by bubbling N2 through standard extracellular recording solution in the perfusion reservoir (60 ml syringe) for at least 15 min until the desired  (15–17 mmHg) was reached. This degree of hypoxia was slightly greater than that used by Jonz et al. (2004; 25 mmHg) but consistent with estimated arterial
(15–17 mmHg) was reached. This degree of hypoxia was slightly greater than that used by Jonz et al. (2004; 25 mmHg) but consistent with estimated arterial  levels in zebrafish experiencing a maximal hyperventilatory response during acute hypoxia (Vulesevic et al. 2006). Different levels of hypercapnia were produced by bubbling commercially produced CO2 mixtures (0.25%, 0.5% or 1% CO2 balanced with air; BOC Canada Ltd) through the extracellular recording solution in the perfusion reservoir (60 ml syringe) until water
levels in zebrafish experiencing a maximal hyperventilatory response during acute hypoxia (Vulesevic et al. 2006). Different levels of hypercapnia were produced by bubbling commercially produced CO2 mixtures (0.25%, 0.5% or 1% CO2 balanced with air; BOC Canada Ltd) through the extracellular recording solution in the perfusion reservoir (60 ml syringe) until water  reached equilibrium. Water
reached equilibrium. Water  was measured using a CO2 electrode (Cameron Instrument Co., Guelph, Ontario, Canada; model E201) connected to a Cameron BGM 200 blood gas meter. Measurements of O2 were made using a fibre optic oxygen electrode (Ocean Optics Foxy AL300, Dunedin, FL, USA) and associated hardware and software (Ocean Optics SD 2000). Gas impermeable tubing (Tygon, Saint-Gobain Performance Plastics Corp., Akron, OH, USA) was used to transfer the perfusate to the recording chamber. Control experiments were performed in which the perfusate was bubbled with air.
was measured using a CO2 electrode (Cameron Instrument Co., Guelph, Ontario, Canada; model E201) connected to a Cameron BGM 200 blood gas meter. Measurements of O2 were made using a fibre optic oxygen electrode (Ocean Optics Foxy AL300, Dunedin, FL, USA) and associated hardware and software (Ocean Optics SD 2000). Gas impermeable tubing (Tygon, Saint-Gobain Performance Plastics Corp., Akron, OH, USA) was used to transfer the perfusate to the recording chamber. Control experiments were performed in which the perfusate was bubbled with air.
Two conventional blockers of K+ channels were used to characterize the CO2 sensitive current. 4-AP (2.5 mm) was used to block voltage-gated K+ channels while quinidine (0.5 mm) was used to block background K+ channels. To assess the importance of CA activity in CO2 sensing, cells were exposed to the membrane-permeant CA blocker, acetazolamide (50 μm). All drugs were purchased from Sigma and dissolved in the extracellular bath solution (pH adjusted to 7.4).
Statistical analysis
Data acquired under conditions of current or voltage clamp are presented as means ±s.e.m. Differences between treatments were analysed using Student's t test for paired data or one-way repeated measures ANOVA followed by Bonferroni's post hoc test for multiple comparisons. Significance was accepted if P < 0.05. Statistical analyses of data were performed using commercial software (SigmaStat v. 3.1; SPSS).
Results
Effects of hypercapnia or hypoxia on NEC membrane potential under current-clamp recording
NECs exhibited membrane depolarization when treated with hypoxic (Hox;  mmHg) or hypercapnic (Hpc; 1% CO2;
mmHg) or hypercapnic (Hpc; 1% CO2;  mmHg) solutions (Fig. 1). In 18 cells (of 30 cells tested), there was a significant change in membrane potential (Vm) from −54.8 ± 1.8 to −28.8 ± 4.6 mV (paired t test, P < 0.001) after bath perfusion with hypercapnic solution (Fig. 1D). The 12 unresponsive cells were not tested for their O2 sensitivity. In another set of experiments, six cells (of 18 cells tested) responded to both elevated CO2 and reduced O2 (Fig. 1B and C). NECs exposed sequentially to hypercapnia, allowed to recover and then exposed to hypoxia displayed similar increases in Vm (Fig. 1E). The change in the membrane potential outlasted the hypercapnic/hypoxic treatment to a varying degree.
mmHg) solutions (Fig. 1). In 18 cells (of 30 cells tested), there was a significant change in membrane potential (Vm) from −54.8 ± 1.8 to −28.8 ± 4.6 mV (paired t test, P < 0.001) after bath perfusion with hypercapnic solution (Fig. 1D). The 12 unresponsive cells were not tested for their O2 sensitivity. In another set of experiments, six cells (of 18 cells tested) responded to both elevated CO2 and reduced O2 (Fig. 1B and C). NECs exposed sequentially to hypercapnia, allowed to recover and then exposed to hypoxia displayed similar increases in Vm (Fig. 1E). The change in the membrane potential outlasted the hypercapnic/hypoxic treatment to a varying degree.
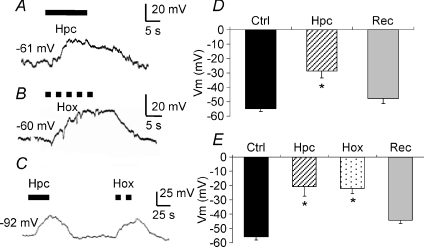
Figure 1. Effects of hypercapnia or hypoxia on membrane potential in isolated cultured gill neuroepithelial cells (NECs) of zebrafish (Danio rerio).
A and B, typical current clamp recordings showing membrane depolarization during perfusion with hypercapnic (Hpc, 1% CO2= 7.5 mmHg, continuous line) or hypoxic solutions (Hox,  mmHg, dashed line), respectively. C, current clamp recording showing depolarization during perfusion with hypercapnic (filled arrow bar) or hypoxic solution (dashed bar) measured on the same cell. D, mean ±s.e.m. (n= 18 cells) membrane potential (Vm) of NECs during perfusion with control (Ctrl) or hypercapnic (Hpc) solutions, and subsequent recovery (Rec). Vm was significantly reduced by 26 mV during hypercapnia (paired t test, *P < 0.001). E, mean ±s.e.m. (n= 6) Vm of NECs for sequential stages of control, hypercapnia, hypoxia (Hox) and recovery during recording from the same cell. Vm was significantly and reversibly reduced in the presence of hypercapnia and hypoxia relative to controls (one-way repeated measures ANOVA, *P < 0.001).
mmHg, dashed line), respectively. C, current clamp recording showing depolarization during perfusion with hypercapnic (filled arrow bar) or hypoxic solution (dashed bar) measured on the same cell. D, mean ±s.e.m. (n= 18 cells) membrane potential (Vm) of NECs during perfusion with control (Ctrl) or hypercapnic (Hpc) solutions, and subsequent recovery (Rec). Vm was significantly reduced by 26 mV during hypercapnia (paired t test, *P < 0.001). E, mean ±s.e.m. (n= 6) Vm of NECs for sequential stages of control, hypercapnia, hypoxia (Hox) and recovery during recording from the same cell. Vm was significantly and reversibly reduced in the presence of hypercapnia and hypoxia relative to controls (one-way repeated measures ANOVA, *P < 0.001).
Dose-dependent effects of hypercapnia on NECs under current-clamp recording
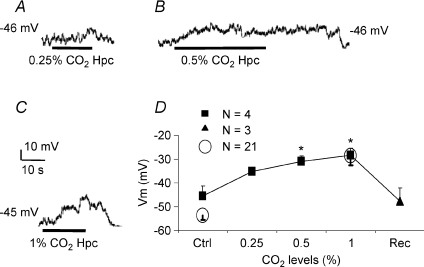
Figure 2A, B and C show typical Vm traces of the reversible depolarization recorded from a single NEC when exposed sequentially to bath perfusates equilibrated with 0.25%, 0.5% or 1% CO2. Figure 2D shows the mean values of Vm at the three different levels of  . Clearly, hypercapnic stimuli evoked NEC membrane depolarization in a dose (partial pressure)-dependent manner.
. Clearly, hypercapnic stimuli evoked NEC membrane depolarization in a dose (partial pressure)-dependent manner.
Figure 2. Dose ( )-dependent effects of hypercapnia on membrane potential in isolated cultured neuroepithelial cells (NECs) of zebrafish (Danio rerio).
)-dependent effects of hypercapnia on membrane potential in isolated cultured neuroepithelial cells (NECs) of zebrafish (Danio rerio).
A–C, representative traces of reversible depolarization of the same NEC during perfusion (bar) with hypercapnic solutions, 0.25% ( mmHg), 0.5% (
mmHg), 0.5% ( mmHg) or 1% (
mmHg) or 1% ( mmHg) CO2. Scale bar = 10 mV/10 s in C (applies to A and B as well). D, mean ±s.e.m. values of membrane potential (Vm) during hypercapnia. Continuous line and filled squares show the dose-dependent trend of Vm during hypercapnia on the same set of NECs (n= 4; filled triangle for recovery n= 3; one-way repeated measures ANOVA versus control, *P < 0.05). Open circles represent data from NECs using control and 1% CO2 alone (n= 21).
mmHg) CO2. Scale bar = 10 mV/10 s in C (applies to A and B as well). D, mean ±s.e.m. values of membrane potential (Vm) during hypercapnia. Continuous line and filled squares show the dose-dependent trend of Vm during hypercapnia on the same set of NECs (n= 4; filled triangle for recovery n= 3; one-way repeated measures ANOVA versus control, *P < 0.05). Open circles represent data from NECs using control and 1% CO2 alone (n= 21).
Temporal effects of hypercapnia on NECs under voltage-clamp recording
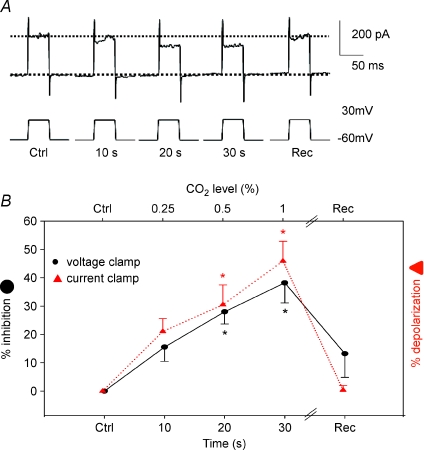
While there was a clear dose dependence of the NEC response to hypercapnia, the response onset times were variable. To address this issue directly, voltage clamp protocols were used to measure the responses of NECs before, during (10–30 s) and after the perfusion of the hypercapnic solution. Typical whole-cell currents and average values of inhibition in NECs (n= 6) are shown in Fig. 3, where currents were evoked using a voltage step protocol from −60 mV to 30 mV every 10 s. The reduction in current over time under these conditions was similar to the dose-dependent change in membrane potential under current clamp (during bath perfusion with different levels of hypercapnia, Fig. 2D). This provides further evidence of a dose response and suggests that much of the time dependence in the response onset is due to the delay in reaching the target  . The data depicted in Figs 2 and 3 may actually underestimate the cellular responses of NECs to CO2 because the need for rapid experimentation on the fragile cells meant that steady-state responses were not always achieved.
. The data depicted in Figs 2 and 3 may actually underestimate the cellular responses of NECs to CO2 because the need for rapid experimentation on the fragile cells meant that steady-state responses were not always achieved.
Figure 3. Time-series effects of hypercapnia-induced current in cultured gill neuroepithelial cells (NECs) of zebrafish (Danio rerio) under voltage-clamp (Vclamp) recording.
A, representative whole cell current traces evoked every 10 s using voltage steps from −60 to 30 mV before, at times 10–30 s of hypercapnia and during recovery, respectively. B, mean ±s.e.m. (n= 6) percentage of steady state current inhibition before, at times 10–30 s of hypercapnia and during recovery (continuous line with filled circles). For comparison, dashed line and triangles show mean ±s.e.m. values of percentage of membrane potential (Vm) depolarization (data taken from Fig. 2D) during bath perfusion with different levels of hypercapnia under current clamp (one-way repeated measures ANOVA versus control, *P < 0.05).
NECs express a CO2-sensitive outward current
Under voltage clamp, NECs expressed an outwardly rectifying current that was sensitive to changes in  . Figure 4A depicts the average current–voltage (I–V) relationship from 12 NECs (obtained using a ramp protocol, −90 to 60 mV, inset) under control and hypercapnic conditions. The inhibitory effects of hypercapnia on outward currents were apparent over a broad range of membrane potential (from −60 mV to 60 mV), and when measured at 50 mV ranged from 21 to 51% with an average inhibition of 36% (n= 12). Figure 4B shows the mean CO2-sensitive difference current (Nox – Hpc) from the same 12 cells. The CO2-sensitive current reverses near the potassium equilibrium potential, EK (calculated as −83 mV in the present conditions) and is well-described by the Goldman–Hodgkin–Katz (GHK) current equation, suggesting that it is mediated by background potassium channels (referred to here as
. Figure 4A depicts the average current–voltage (I–V) relationship from 12 NECs (obtained using a ramp protocol, −90 to 60 mV, inset) under control and hypercapnic conditions. The inhibitory effects of hypercapnia on outward currents were apparent over a broad range of membrane potential (from −60 mV to 60 mV), and when measured at 50 mV ranged from 21 to 51% with an average inhibition of 36% (n= 12). Figure 4B shows the mean CO2-sensitive difference current (Nox – Hpc) from the same 12 cells. The CO2-sensitive current reverses near the potassium equilibrium potential, EK (calculated as −83 mV in the present conditions) and is well-described by the Goldman–Hodgkin–Katz (GHK) current equation, suggesting that it is mediated by background potassium channels (referred to here as  ).
).
Pharmacological characterization of the CO2 sensitive current 
The K+ channel blockers 4-AP and quinidine were used to pharmacologically characterize the CO2 sensitive current. CO2 sensitive NECs were first identified by the occurrence of hypercapnic inhibition of the outward current evoked by a ramp protocol under voltage clamp (n= 5, Fig. 5A). A decrease in whole cell outward current was also observed with 2.5 mm 4-AP alone (indicating the presence of voltage-dependent potassium channels in NECs). However, subsequent hypercapnia caused further inhibition (proportionately similar to that observed with hypercapnia alone; compare Nox with Hpc and 4-AP with 4-AP+Hpc in Fig. 5A), suggesting that  was not carried by voltage-dependent K+ channels. This conclusion was further supported by current clamp recordings. Figure 5B depicts membrane depolarization of a NEC after application of 2.5 mm 4-AP followed by a further membrane depolarization during exposure to hypercapnia.
was not carried by voltage-dependent K+ channels. This conclusion was further supported by current clamp recordings. Figure 5B depicts membrane depolarization of a NEC after application of 2.5 mm 4-AP followed by a further membrane depolarization during exposure to hypercapnia.
Figure 5. Pharmacological characteristics of CO2 sensing in isolated cultured gill neuroepithelial cells (NECs) of zebrafish (Danio rerio).
A, average current–voltage (I–V) relationship of whole-cell voltage clamp recordings from five CO2-sensitive NECs during exposure to normoxia (Nox), hypercapnia (Hpc), 4-AP and 4-AP+Hpc showing the persistence of the hypercapnic effects in the presence of 4-AP. Currents were evoked by changing the voltage from −100 mV to +60 mV following a ramp protocol (1 s) from a holding potential of −60 mV. B, typical current clamp recording of a reversible hypercapnic depolarization following application of 2.5 mm 4-AP. C, average current–voltage (I–V) relationship of whole-cell voltage clamp recordings from six CO2-sensitive NECs during exposure to Nox, Hpc, quinidine (Quid) and Quid+Hpc; a ramp protocol was used (as in A). Hypercapnia (Hpc) reduced currents over a range of potentials, but had little effect in the presence of 0.5 mm quinidine (Quid+Hpc). D, typical current clamp recording showing the reversible depolarization following bath application of 0.5 mm quinidine, but subsequent hypercapnia having no additional affect. E, mean ±s.e.m. inhibition of whole-cell current at 50 mV using the data points in Fig. 4 as well as in panels A and C, showing hypercapnia effects alone (Hpc), hypercapnia effects in the presence of 4-AP (4AP, 4AP + Hpc, paired t test, *P < 0.05) and quinidine (Quid, Quid+Hpc) respectively.
Application of 0.5 mm quinidine reduced currents over a range of membrane potentials, but a subsequent hypercapnia had little effect (Quid+Hpc; n= 6, Fig. 5C). Again, these data are corroborated by current-clamp recordings. Figure 5D shows that in a single NEC, the prominent depolarization under bath application of 0.5 mm quinidine prevented further depolarization during hypercapnia. A summary of the  sensitivity to quinidine and insensitivity to 4-AP is shown in Fig. 5E, and suggests that background K+ channels in NECs underlie
sensitivity to quinidine and insensitivity to 4-AP is shown in Fig. 5E, and suggests that background K+ channels in NECs underlie  .
.
Carbonic anhydrase mediates the NEC responses to hypercapnia
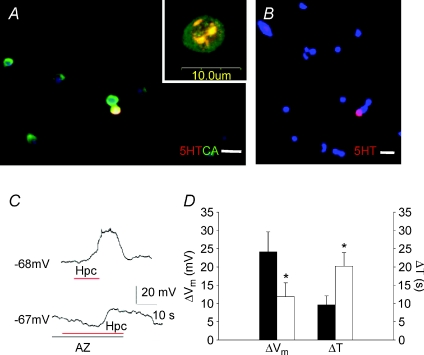
Hypercapnia is associated with decreased pH in the extracellular fluids and thus could act through a pH sensor rather than an intracellular CO2-mediated CA-dependent intracellular process. We first tested for the presence of CA using immunocytochemistry. Representative images of 5HT immunoreactivity (IR) and/or CA-IR NECs in primary cell culture are depicted in Fig. 6A. Although all 5HT-IR cells (NEC) exhibited CA-IR, some cells appeared to exclusively express CA (data not shown). If NECs were treated with both 5HT and CA primary antibodies after CA preabsorption, only 5HT-IR was observed (Fig. 6B).
Figure 6. Effects of acetazolamide (AZ) on the CO2-sensitive membrane potential in isolated cultured gill neuroepithelial cells (NECs) of zebrafish (Danio rerio).
A, representative image of 5HT-immunoreactive (IR; red) and/or CA-IR (green) NECs in primary cell culture. The cell nuclei appear as blue; yellow/orange represents co-localization of 5HT and CA. Note that all 5HT-IR cells exhibit CA-IR. In addition, some cells appeared to exclusively express CA, whereas other cells appeared to lack both 5HT and CA. Inset: confocal image of a cell colocalizing 5HT and CA. B, preabsorption of the CA antibody with excess CA followed by application of 5HT and CA antibodies yielded only 5HT-IR NECs. Scale bars = 20 μm in A (applies to B also). C, typical current-clamp recording of a reversible depolarization during perfusion with hypercapnic solution (upper trace) as well as a reduced and delayed depolarization after the administration of 50 μm acetazolamide (AZ) on the same cell (lower trace). D, mean ±s.e.m. (n= 6) membrane potential changes (ΔVm) and time to peak depolarization (ΔT) during hypercapnia alone (ctrl; filled bars) and in the presence of 50 μm acetazolamide (AZ; open bars; paired t test, *P < 0.05).
To directly examine the role of CA in the response of NECs to CO2, NECs were exposed to isohydric hypercapnia (constant pH 7.4). Figure 6C and D show that typical hypercapnia-induced responses persisted in the absence of extracellular acidosis but were inhibited by the presence of 50 μm acetazolamide (a CA inhibitor). Acetazolamide reduced the magnitude of responses to a transient CO2 stimulus by an average of 12 mV (n= 6 CO2 sensitive NECs). Acetazolamide also delayed the time to reach peak membrane depolarization by an average of 10.5 s when comparing cells before and after drug treatment.
Discussion
The results of the present study provide the first direct evidence of CO2 transduction in fish and demonstrate that the underlying mechanism involves inhibition of background K+ channels via a pathway involving carbonic anhydrase. The finding that a sub-population of gill NECs act as sensors for both O2 and CO2 provides further support for the evolutionary link between the fish gill and the carotid body in mammals. However, a major difference between the two CO2-sensing structures is that the gill NECs are able to respond to absolute partial pressures of CO2 approximately 10 times lower than the carotid body. The evolutionary significance of these different sensing thresholds is that each CO2-sensing structure is exquisitely adapted to detect deviations in arterial blood  from normal set points which in mammals and water-breathing fish are approximately 40 mmHg and 2–3 mmHg, respectively.
from normal set points which in mammals and water-breathing fish are approximately 40 mmHg and 2–3 mmHg, respectively.
Electrophysiological properties of CO2 sensitivity
Whole cell voltage and current clamp recordings, performed on isolated NECs, allowed us to investigate the electrophysiological responses to hypercapnia. Hypercapnia inhibited a CO2 sensitive channel, resulting in a decrease in whole-cell conductance. The current mediated by this channel reversed near the equilibrium potential for K+, displayed open rectification, and is well-described by the Goldman–Hodgkin–Katz current equation (Fig. 4). These findings are consistent with the effect of hypercapnia on membrane potential in rat carotid body glomus cells (Buckler & Vaughan-Jones, 1994a,b; Urena et al. 1994; Montoro et al. 1996). The involvement of a K+ current in mediating CO2 sensing is further supported by pharmacological data using K+ channel blockers (Fig. 5).
Whole cell recording of NECs under voltage clamp revealed that the CO2-dependent inhibition of outward current was not significantly affected by 4-AP; in the presence of 4-AP, hypercapnia resulted in a further 24% reduction in current. In contrast, CO2-dependent inhibition was almost completely blocked by quinidine; additional hypercapnia was unable to cause further reduction in current in the presence of quinidine. These observations were corroborated under current clamp as well (Fig. 5B and D). Interestingly, these data are similar to the hypoxia-induced responses observed in other chemoreceptor cells, such as human neuroepithelial body-derived H-146 cells (O’kelly et al. 1999), carotid body glomus cells (Buckler, 1997; Buckler et al. 2000) and other peripheral neurons such as rat glossopharyngeal neurons (Campanucci et al. 2003). Generally speaking, the CO2 sensitive current we describe possesses all the characteristics of a current mediated by background (or leak) K+ channels, which show no voltage or time dependence (Bayliss et al. 2003; Buckler, 2007). However, further experiments will be required to determine the specific type of background K+ channel involved in CO2 sensing in gill NECs. In mammalian neuroepithelial sensory receptors (e.g. carotid body glomus cells), a background K+ current causing membrane depolarization has been identified as a member of the TWIK (tandem P domain weakly inward-rectifying K+)-related acid-sensitive K+ (TASK) family (Buckler et al. 2000; Bayliss et al. 2003; Buckler, 2007). Moreover, TASK/TASK-like K+ channels have been reported in other neurons (Nattie, 1999; Putnam et al. 2004; Buckler, 2007) including (1) central chemosensory neurons mediating CO2 sensing in the brain, e.g. medullary raphe neurons (Wang et al. 2002; Washburn et al. 2002, 2003), ventral medulla neurons (Neubauer et al. 1991; Wellner-Kienitz & Shams, 1998) and locus coeruleus neurons (Bayliss et al. 2001); (2) respiratory motor neurons conveying central respiratory drive to the muscles of breathing (Nattie, 1999); and (3) other peripheral sensory neurons such as rat nociceptive cells (Cooper et al. 2004).
Carbonic anhydrase and CO2 chemoreception in NECs
This study also investigated the involvement of carbonic anhydrase (CA) on CO2 transduction. It was demonstrated that bath application of acetazolamide reversibly reduced the responses to a transient CO2 stimulus, similar to observations in the carotid body glomus cells (Buckler et al. 1991a,b; Iturriaga et al. 1991; Iturriaga & Lahiri, 1993; Iturriaga, 1993), cat laryngeal cells (Coates et al. 1996), cat ventral medulla neurons (Coates et al. 1991) and isolated rat neonatal chromaffin cells (Munoz-Cabello et al. 2005). It would appear that CA activity in the zebrafish gill NECs, as in the carotid body, is important in setting the magnitude and speed of the initial response to an elevation of  , presumably by accelerating the hydration reaction and thus the rate of intracellular acidification. The residual response of NECs to hypercapnia after CA inhibition presumably reflects intracellular acidification occurring at the uncatalysed rate of CO2 hydration. Thus, although intracellular acidification would still occur in the absence of CA, its presence in CO2-sensing cells allows a more rapid and vigorous response than otherwise might be possible.
, presumably by accelerating the hydration reaction and thus the rate of intracellular acidification. The residual response of NECs to hypercapnia after CA inhibition presumably reflects intracellular acidification occurring at the uncatalysed rate of CO2 hydration. Thus, although intracellular acidification would still occur in the absence of CA, its presence in CO2-sensing cells allows a more rapid and vigorous response than otherwise might be possible.
Because the zebrafish NECs also respond to metabolic acidosis (reduced pH at constant  ; Z. Qin, J. Lewis & S. F. Perry, unpublished data), it was necessary to assess the role of CA using isohydric hypercapnia. This protocol served to exclude the CA-independent contribution of intracellular H+not arising from the inward diffusion of CO2. It will be important to extend these results by directly measuring intracellular pH while exposing NECs to changes in
; Z. Qin, J. Lewis & S. F. Perry, unpublished data), it was necessary to assess the role of CA using isohydric hypercapnia. This protocol served to exclude the CA-independent contribution of intracellular H+not arising from the inward diffusion of CO2. It will be important to extend these results by directly measuring intracellular pH while exposing NECs to changes in  in the presence or absence of acetazolamide. Additionally, future experiments should attempt to assess the relative involvement of extracellular versus intracellular acidosis in promoting membrane depolarization when NECs are exposed to hypercapnia.
in the presence or absence of acetazolamide. Additionally, future experiments should attempt to assess the relative involvement of extracellular versus intracellular acidosis in promoting membrane depolarization when NECs are exposed to hypercapnia.
Interaction between hypoxia and hypercapnia on NEC chemosensitivity
An important finding of the present study is that individual NECs can respond to both hypoxia and hypercapnia. Such bimodal sensing, also seen in the carotid body, suggests that reflex cardiorespiratory responses to hypercapnia and hypoxia are mediated, at least in part, by the same chemosensory cells. Thus, the transduction pathways for both O2 and CO2 coexist within a common primary receptor cell and might converge at or after the membrane depolarization. In carotid body glomus cells, both stimuli have been shown to elicit K+ current inhibition, electrical activity, voltage-gated Ca2+ influx (Peers, 1990a,b; Peers & Green, 1991; Buckler & Vaughan-Jones, 1993, 1994a,b; Buckler, 1997; Dasso et al. 2000) and subsequent neurotransmitter release and afferent nerve activation (Lahiri & DeLaney, 1975; Zhang & Nurse, 2004). Therefore, the sensing of hypoxia and hypercapnia/acidosis involves common steps. There is also some evidence that the primary CO2/pH sensor is independent of the O2 sensor in some cases; the effects of CO2 and/or pH are not mediated through changes in the affinity of an O2 sensor in carotid body glomus cells (Dasso et al. 2000). However, there are also data suggesting that CO2 chemosensitivity may be modulated by hypoxia (Reid et al. 2005; see review by Lahiri & Forster, 2003). It is interesting that while the glomus cells of the carotid body proliferate in response to chronic hypoxia, the density of the analogous population of gill NECs (those possessing 5-HT) in zebrafish is unaffected by long-term hypoxia (Jonz et al. 2004; Vulesevic et al. 2006). On the other hand, NECs not containing 5-HT (without a proven function) do display a significant proliferation upon exposure of zebrafish to chronic hypoxia (Jonz et al. 2004). Thus, in keeping with the lack of a robust response to chronic hypoxia, it is not surprising that the numbers of gill filament NECs remain unaltered by chronic hypercapnia (Vulesevic et al. 2006).
Although evidence has shown the membrane voltage response of zebrafish NECs to hypoxia closely resembles the discharge frequency in sensory fibres of the isolated trout gill preparations (Burleson & Milsom, 1993; Jonz et al. 2004), a direct link between CO2-sensitive currents in NECs and sensory neuron discharge remains to be determined. A recent study of rat adrenal medulla chromaffin cells (Munoz-Cabello et al. 2005) which act as neonatal CO2 sensors before complete maturation of peripheral and central chemoreceptors has linked hypercapnia-induced membrane depolarization to Ca2+-dependent catecholamine secretion. Similar studies using a combination of patch clamp and amperometric techniques will be important for future chemotransduction studies in fish NECs.
Summary and perspectives
Exposure of fish to hypercapnia initiates several cardiorespiratory reflexes including hyperventilation, bradycardia, increased peripheral vascular resistance and catecholamine secretion (Perry et al. 1999; Perry & Reid, 2002; see reviews by Perry & Gilmour, 2002; Gilmour & Perry, 2007). In this study, we provide evidence that these reflexes may originate from the activation of branchial NECs that can function as bimodal CO2 and O2 chemoreceptors. The results demonstrate several similarities with mammalian peripheral chemoreceptors (i.e. carotid body). This comparison suggests a phylogenetic trend whereby CO2 chemoreception may first have arisen in the periphery to sense the external environment, and subsequently central chemoreception arose to monitor internal blood and optimize metabolism (Milsom, 2002).
While it is clear that zebrafish NECs respond to elevated CO2 with membrane depolarization, further research is required to elucidate the specific signal transduction cascade and its downstream effects. Because the genetically tractable zebrafish displays CO2 chemosensitivity at 3 days post-fertilization (L. Labonté and S. F. Perry, unpublished data), it should be feasible to assess cardiorespiratory responses in vivo and electrophysiological responses in vitro in fish (or in cells derived from fish) subjected to selective gene knockdown.
Acknowledgments
This study was financially supported by Natural Sciences and Engineering Research Council (NSERC) Discovery and Research Tools and Innovation grants to S.F.P. and J.L. J.L. was also supported by the Canadian Foundation for Innovation (CFI). We are grateful to Michael Jonz and Andrew Ochalski for invaluable assistance.
Glossary
Abbreviations
- CA
carbonic anhydrase
- NEC
neuroepithelial cell
Author contributions
All authors had similar roles in the conception and design of experiments. Z.Q. performed the laboratory experiments and analysed the data. All authors contributed equally to the writing of the manuscript and to its final approval.
References
- Bailly Y, Dunelerb S, Laurent P. The neuroepithelial cells of the fish gill filament: indolamine-immunocyto-chemistry and innervation. Anat Rec. 1992;233:143–161. doi: 10.1002/ar.1092330118. [DOI] [PubMed] [Google Scholar]
- Bayliss DA, Sirois JE, Talley EM. The TASK family: two-pore domain background K+ channels. Mol Interv. 2003;3:205–219. doi: 10.1124/mi.3.4.205. [DOI] [PubMed] [Google Scholar]
- Bayliss DA, Talley EM, Sirois JE, Lei Q. TASK-1 is a highly modulated pH-sensitive ‘leak’ K+ channel expressed in brainstem respiratory neurons. Respir Physiol. 2001;129:159–174. doi: 10.1016/s0034-5687(01)00288-2. [DOI] [PubMed] [Google Scholar]
- Buckler KJ. A novel oxygen-sensitive potassium current in rat carotid body type I cells. J Physiol. 1997;498:649–662. doi: 10.1113/jphysiol.1997.sp021890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckler KJ. TASK-like potassium channels and oxygen sensing in the carotid body. Respir Physiol Neurobiol. 2007;157:55–64. doi: 10.1016/j.resp.2007.02.013. [DOI] [PubMed] [Google Scholar]
- Buckler KJ, Williams BA, Honore E. An oxygen-, acid- and anaesthetic-sensitive TASK-like background potassium channel in rat arterial chemoreceptor cells. J Physiol. 2000;525:135–142. doi: 10.1111/j.1469-7793.2000.00135.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckler KJ, Vaughan-Jones RD. Effects of acidic stimuli on intracellular calcium in isolated type I cells of the neonatal rat carotid body. Pflugers Arch. 1993;425:22–27. doi: 10.1007/BF00374499. [DOI] [PubMed] [Google Scholar]
- Buckler KJ, Vaughan-Jones RD. Effects of hypercapnia on membrane potential and intracellular calcium in rat carotid body type I cells. J Physiol. 1994a;478:157–171. doi: 10.1113/jphysiol.1994.sp020239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckler KJ, Vaughan-Jones RD. Effects of hypoxia on membrane potential and intracellular calcium in rat neonatal carotid body type I cells. J Physiol. 1994b;476:423–428. doi: 10.1113/jphysiol.1994.sp020143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckler KJ, Vaughan-Jones RD, Peers C, Lagadic-Gossmann D, Nye PC. Effects of extracellular pH, PCO2 and HCO3− on intracellular pH in isolated type-I cells of the neonatal rat carotid body. J Physiol. 1991a;444:703–721. doi: 10.1113/jphysiol.1991.sp018902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckler KJ, Vaughan-Jones RD, Peers C, Nye PC. Intracellular pH and its regulation in isolated type I carotid body cells of the neonatal rat. J Physiol. 1991b;436:107–129. doi: 10.1113/jphysiol.1991.sp018542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burleson ML, Mercer SE, Wilk-Blaszczak MA. Isolation and characterization of putative O2 chemoreceptor cells from the gills of channel catfish (Ictalurus punctatus) Brain Res. 2006;1092:100–107. doi: 10.1016/j.brainres.2006.03.085. [DOI] [PubMed] [Google Scholar]
- Burleson ML, Milsom WK. Sensory receptors in the first gill arch of rainbow trout. Respir Physiol. 1993;93:97–110. doi: 10.1016/0034-5687(93)90071-h. [DOI] [PubMed] [Google Scholar]
- Burleson ML, Smatresk NJ. Branchial chemoreceptors mediate ventilatory responses to hypercapnic acidosis in channel catfish. Comp Biochem Physiol A. 2000;125:403–414. doi: 10.1016/s1095-6433(00)00167-7. [DOI] [PubMed] [Google Scholar]
- Campanucci VA, Fearon IM, Nurse CA. A novel O2-sensing mechanism in rat glossopharyngeal neurones mediated by a halothane-inhibitable background K+ conductance. J Physiol. 2003;548:731–743. doi: 10.1113/jphysiol.2002.035998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coates EL, Li AH, Nattie EE. Acetazolamide on the ventral medulla of the cat increases phrenic output and delays the ventilatory response to CO2. J Physiol. 1991;441:433–451. doi: 10.1113/jphysiol.1991.sp018760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coates EL, Knuth SL, Bartlett DJ. Laryngeal CO2 receptors: influence of systemic PCO2 and carbonic anhydrase inhibition. Respir Physiol. 1996;104:53–61. doi: 10.1016/0034-5687(96)00009-6. [DOI] [PubMed] [Google Scholar]
- Cooper BY, Johnson RD, Rau KK. Characterization and function of TWIK-related acid sensing K+ channels in a rat nociceptive cell. Neuroscience. 2004;129:209–224. doi: 10.1016/j.neuroscience.2004.06.066. [DOI] [PubMed] [Google Scholar]
- Dasso LL, Buckler KJ, Vaughan-Jones RD. Interactions between hypoxia and hypercapnic acidosis on calcium signalling in carotid body type I cells. Am J Physiol Lung Cell Mol Physiol. 2000;279:L36–42. doi: 10.1152/ajplung.2000.279.1.L36. [DOI] [PubMed] [Google Scholar]
- Drummond GB. Reporting ethical matters in The Journal of Physiology: standards and advice. J Physiol. 2009;587:713–719. doi: 10.1113/jphysiol.2008.167387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunel-Erb S, Bailly Y, Laurent P. Neuroepithelial cells in fish gill primary lamellae. J Appl Physiol. 1982;53:1342–1353. doi: 10.1152/jappl.1982.53.6.1342. [DOI] [PubMed] [Google Scholar]
- Esbaugh A, Perry SF, Bayaa M, Georgalis T, Nickerson JG, Tufts BL, Gilmour KM. Cytoplasmic carbonic anhydrase isozymes in rainbow trout Oncorhynchus mykiss: Comparative physiology and molecular evolution. J Exp Biol. 2005;208:1951–1961. doi: 10.1242/jeb.01551. [DOI] [PubMed] [Google Scholar]
- Georgalis T, Perry SF, Gilmour KM. The role of branchial carbonic anhydrase in acid-base regulation in rainbow trout (Oncorhynchus mykiss) J Exp Biol. 2006;209:518–530. doi: 10.1242/jeb.02018. [DOI] [PubMed] [Google Scholar]
- Gilmour KM. The CO2/pH ventilatory drive in fish. Comp Biochem Physiol. 2001;130:219–240. doi: 10.1016/s1095-6433(01)00391-9. [DOI] [PubMed] [Google Scholar]
- Gilmour KM, Milsom WK, Rantin FT, Reid SG, Perry SF. Cardiorespiratory responses to hypercarbia in tambaqui (Colossoma macropomum): chemoreceptor orientation and specificity. J Exp Biol. 2005;208:1095–1107. doi: 10.1242/jeb.01480. [DOI] [PubMed] [Google Scholar]
- Gilmour KM, Perry SF. Branchial chemoreceptor regulation of cardiorespiratory function. In: Hara TJ, Zielinski B, editors. Sensory Systems Neuroscience. San Diego: Academic Press; 2007. pp. 97–151. [Google Scholar]
- Gilmour KM, Perry SF. Carbonic anhydrase and acid-base regulation in fish. J Exp Biol. 2009;212:1647–1661. doi: 10.1242/jeb.029181. [DOI] [PubMed] [Google Scholar]
- Goniakowska-Witalinska L, Zaccone G, Fasulo S, Mauceri A, Licata A, Youson J. Neuroendocrine cells in the gills of the bowfin Amia calva. An ultrastructural and immunocytochemical study. Folia Histochem Cytobiol. 1995;33:171–177. [PubMed] [Google Scholar]
- Gonzalez C, Almaraz L, Obeso A, Rigual R. Carotid body chemoreceptors: from natural stimuli to sensory discharges. Physiol Rev. 1994;74:829–898. doi: 10.1152/physrev.1994.74.4.829. [DOI] [PubMed] [Google Scholar]
- Gonzalez C, Almaraz L, Obeso A, Rigual R. Oxygen and acid chemoreception in the carotid body chemoreceptors. Trends Neurosci. 1992;15:146–153. doi: 10.1016/0166-2236(92)90357-e. [DOI] [PubMed] [Google Scholar]
- Guyenet PG. The 2008 Carl Ludwig Lecture: retrotrapezoid nucleus, CO2 homeostasis, and breathing automaticity. J Appl Physiol. 2008;105:404–416. doi: 10.1152/japplphysiol.90452.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 1981;391:85–100. doi: 10.1007/BF00656997. [DOI] [PubMed] [Google Scholar]
- Hille B. Ion Channels of Excitable Membranes. 3rd edn. Sunderland, MA, USA: Sinauer Associates; 2001. [Google Scholar]
- Iturriaga R. Carotid body chemoreception: the importance of CO2-HCO3− and carbonic anhydrase. Biol Res. 1993;26:319–329. [PubMed] [Google Scholar]
- Iturriaga R, Lahiri S. Carbonic anhydrase and carotid body chemoreception in the presence and absence of CO2-HCO3−. Adv Exp Med Biol. 1993;337:171–176. doi: 10.1007/978-1-4615-2966-8_24. [DOI] [PubMed] [Google Scholar]
- Iturriaga R, Lahiri S, Mokashi A. Carbonic anhydrase and chemoreception in the cat carotid body. Am J Physiol Cell Physiol. 1991;261:C565–573. doi: 10.1152/ajpcell.1991.261.4.C565. [DOI] [PubMed] [Google Scholar]
- Jonz MG, Fearon IM, Nurse CA. Neuroepithelial oxygen chemoreceptors of the zebrafish gill. J Physiol. 2004;560:737–752. doi: 10.1113/jphysiol.2004.069294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jonz MG, Nurse CA. Neuroepithelial cells and associated innervation of the zebrafish gill: a confocal immunofluorescence study. J Comp Neurol. 2003;461:1–17. doi: 10.1002/cne.10680. [DOI] [PubMed] [Google Scholar]
- Lahiri S, DeLaney RG. Stimulus interaction in the responses of carotid body chemoreceptor single afferent fibres. Respir Physiol. 1975;24:249–266. doi: 10.1016/0034-5687(75)90017-1. [DOI] [PubMed] [Google Scholar]
- Lahiri S, Forster RE. CO2/H+ sensing: peripheral and central chemoreception. Int J Biochem Cell Biol. 2003;35:1413–1435. doi: 10.1016/s1357-2725(03)00050-5. [DOI] [PubMed] [Google Scholar]
- McKendry JE, Milsom WK, Perry SF. Branchial CO2 receptors and cardiorespiratory adjustments during hypercarbia in Pacific spiny dogfish (Squalus acanthias) J Exp Biol. 2001;204:1519–1527. doi: 10.1242/jeb.204.8.1519. [DOI] [PubMed] [Google Scholar]
- McKendry JE, Perry SF. Cardiovascular effects of hypercapnia in rainbow trout (Oncorhynchus mykiss): A role for externally oriented chemoreceptors. J Exp Biol. 2001;204:115–125. doi: 10.1242/jeb.204.1.115. [DOI] [PubMed] [Google Scholar]
- Milsom WK. Phylogeny of CO2/H+ chemoreception in vertebrates. Respir Physiol Neurobiol. 2002;131:29–41. doi: 10.1016/s1569-9048(02)00035-6. [DOI] [PubMed] [Google Scholar]
- Milsom WK, Burleson ML. Peripheral arterial chemoreceptors and the evolution of the carotid body. Respir Physiol Neurobiol. 2007;157:4–11. doi: 10.1016/j.resp.2007.02.007. [DOI] [PubMed] [Google Scholar]
- Milsom WK, Sundin L, Reid S, Kalinin A, Rantin FT. Chemoreceptor control of cardiovascular reflexes. In: Val AL, Almeida-Val VMF, editors. Biology of Tropical Fishes. Manaus, Brazil: INPA; 1999. pp. 363–374. [Google Scholar]
- Montoro RJ, Urena J, Fernandez-Chacon R, Alvarez de Toledo G, Lopez-Barneo J. Oxygen sensing by ion channels and chemotransduction in single glomus cells. J Gen Physiol. 1996;107:133–143. doi: 10.1085/jgp.107.1.133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munoz-Cabello AM, Toledo-Aral JJ, Lopez-Barneo J, Echevarria M. Rat adrenal chromaffin cells are neonatal CO2 sensors. J Neurosci. 2005;25:6631–6640. doi: 10.1523/JNEUROSCI.1139-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nattie E. CO2, brainstem chemoreceptors and breathing. Prog Neurobiol. 1999;59:299–331. doi: 10.1016/s0301-0082(99)00008-8. [DOI] [PubMed] [Google Scholar]
- Neubauer JA, Gonsalves SF, Chou W, Geller HM, Edelman NH. Chemosensitivity of medullary neurons in explant tissue cultures. Neuroscience. 1991;45:701–708. doi: 10.1016/0306-4522(91)90282-s. [DOI] [PubMed] [Google Scholar]
- O’Kelly I, Stephens RH, Peers C, Kemp PJ. Potential identification of the O2-sensitive K+ current in a human neuroepithelial body-derived cell line. Am J Physiol Lung Cell Mol Physiol. 1999;276:L96–104. doi: 10.1152/ajplung.1999.276.1.L96. [DOI] [PubMed] [Google Scholar]
- O’Regan RG, Majcherczyk S. Role of peripheral chemoreceptors and central chemosensitivity in the regulation of respiration and circulation. J Exp Biol. 1982;100:23–40. doi: 10.1242/jeb.100.1.23. [DOI] [PubMed] [Google Scholar]
- Peers C, Green FK. Inhibition of Ca2+-activated K+ currents by intracellular acidosis in isolated type I cells of the neonatal rat carotid body. J Physiol. 1991;437:589–602. doi: 10.1113/jphysiol.1991.sp018613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peers C. Effect of lowered extracellular pH on Ca2+-dependent K+ currents in type I cells from the neonatal rat carotid body. J Physiol. 1990a;422:381–395. doi: 10.1113/jphysiol.1990.sp017990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peers C. Hypoxic suppression of K+ currents in type I carotid body cells: selective effect on the Ca2+-activated K+ current. Neurosci Lett. 1990b;119:253–256. doi: 10.1016/0304-3940(90)90846-2. [DOI] [PubMed] [Google Scholar]
- Perry SF, Fritsche R, Hoagland T, Duff DW, Olson KR. The control of blood pressure during external hypercapnia in the rainbow trout (Oncorhynchus mykiss) J Exp Biol. 1999;202:2177–2190. doi: 10.1242/jeb.202.16.2177. [DOI] [PubMed] [Google Scholar]
- Perry SF, Gilmour KM. Sensing and transfer of respiratory gases at the fish gill. J Exp Zool. 2002;293:249–263. doi: 10.1002/jez.10129. [DOI] [PubMed] [Google Scholar]
- Perry SF, McKendry JE. The relative roles of external and internal CO2versus H+ in eliciting the cardiorespiratory responses of Salmo salar and Squalus acanthias to hypercarbia. J Exp Biol. 2001;204:3963–3971. doi: 10.1242/jeb.204.22.3963. [DOI] [PubMed] [Google Scholar]
- Perry SF, Reid SG. Cardiorespiratory adjustments during hypercarbia in rainbow trout (Oncorhynchus mykiss) are initiated by external CO2 receptors on the first gill arch. J Exp Biol. 2002;205:3357–3365. doi: 10.1242/jeb.205.21.3357. [DOI] [PubMed] [Google Scholar]
- Putnam RW, Filosa JA, Ritucci NA. Cellular mechanisms involved in CO2 and acid signalling in chemosensitive neurons. Am J Physiol Cell Physiol. 2004;287:C1493–526. doi: 10.1152/ajpcell.00282.2004. [DOI] [PubMed] [Google Scholar]
- Reid SG, Perry SF, Gilmour KM, Milsom WK, Rantin FT. Reciprocal modulation of oxygen and carbon dioxide cardiorespiratory chemoreflexes in the tambaqui (Colossoma macropomum) Respir Physiol Neurobiol. 2005;147:175–194. doi: 10.1016/j.resp.2004.12.008. [DOI] [PubMed] [Google Scholar]
- Reid SG, Sundin L, Kalinin AL, Rantin FT, Milsom WK. Cardiovascular and respiratory reflexes in the tropical fish, traira (Hoplias malabaricus): CO2/pH chemoresponses. Respir Physiol. 2000;120:47–59. doi: 10.1016/s0034-5687(99)00100-0. [DOI] [PubMed] [Google Scholar]
- Ryan JA. Growing more cells: A simple guide to small volume cell culture scale-up. 2005. pp. 1–16. http://www.corning.com/Lifesciences/technical_information/techDocs/cc_scale_up_guide.pdf Corning Inc.
- Saltys HA, Jonz MG, Nurse CA. Comparative study of gill neuroepithelial cells and their innervation in teleosts and Xenopus tadpoles. Cell Tissue Res. 2006;323:1–10. doi: 10.1007/s00441-005-0048-5. [DOI] [PubMed] [Google Scholar]
- Smith FM, Jones DR. Localization of receptors causing hypoxic bradycardia in trout (Salmo gairdneri) Can J Zool. 1978;56:1260–1265. [Google Scholar]
- Sundin L, Reid SG, Rantin FT, Milsom WK. Branchial receptors and cardiorespiratory reflexes in the neotropical fish, Tambaqui (Colossoma macropomum) J Exp Biol. 2000;203:1225–1239. doi: 10.1242/jeb.203.7.1225. [DOI] [PubMed] [Google Scholar]
- Urena J, Fernandez-Chacon R, Benot AR, Alvarez de Toledo GA, Lopez-Barneo J. Hypoxia induces voltage-dependent Ca2+ entry and quantal dopamine secretion in carotid body glomus cells. Proc Natl Acad Sci U S A. 1994;91:10208–10211. doi: 10.1073/pnas.91.21.10208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vulesevic B, McNeill B, Perry SF. Chemoreceptor plasticity and respiratory acclimation in the zebrafish, Danio rerio. J Exp Biol. 2006;209:1261–1273. doi: 10.1242/jeb.02058. [DOI] [PubMed] [Google Scholar]
- Wang W, Bradley SR, Richerson GB. Quantification of the response of rat medullary raphe neurones to independent changes in pHo and PCO2. J Physiol. 2002;540:951–970. doi: 10.1113/jphysiol.2001.013443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wellner-Kienitz MC, Shams H. CO2-sensitive neurons in organotypic cultures of the fetal rat medulla. Respir Physiol. 1998;111:137–151. doi: 10.1016/s0034-5687(97)00124-2. [DOI] [PubMed] [Google Scholar]
- Washburn CP, Bayliss DA, Guyenet PG. Cardiorespiratory neurons of the rat ventrolateral medulla contain TASK-1 and TASK-3 channel mRNA. Respir Physiol Neurobiol. 2003;138:19–35. doi: 10.1016/s1569-9048(03)00185-x. [DOI] [PubMed] [Google Scholar]
- Washburn CP, Sirois JE, Talley EM, Guyenet PG, Bayliss DA. Serotonergic raphe neurons express TASK channel transcripts and a TASK-like pH- and halothane-sensitive K+ conductance. J Neurosci. 2002;22:1256–1265. doi: 10.1523/JNEUROSCI.22-04-01256.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaccone G, Fasulo S, Ainis L, Licata A. Paraneurons in the gills and airways of fishes. Microsc Res Tech. 1997;37:4–12. doi: 10.1002/(SICI)1097-0029(19970401)37:1<4::AID-JEMT2>3.0.CO;2-R. [DOI] [PubMed] [Google Scholar]
- Zaccone G, Lauweryns JM, Fasulo S, Tagliafierro G, Ainis L, Licata A. Immunocytochemical localization of serotonin and neuropeptides in the neuroendocrine paraneurons of teleost and lungfish gills. Acta Zool. 1992;73:177–183. [Google Scholar]
- Zaccone G, Mauceri A, Fasulo S. Neuropeptides and nitric oxide synthase in the gill and the air-breathing organs of fishes. J Exp Zoolog. 2006;305:428–439. doi: 10.1002/jez.a.267. [DOI] [PubMed] [Google Scholar]
- Zaccone G, Tagliafierro G, Goniakowska-Witalinska L, Fasulo S, Ainis L, Mauceri A. Serotonin-like immunoreactive cells in the pulmonary epithelium of ancient fish species. Histochemistry. 1989;92:61–63. doi: 10.1007/BF00495017. [DOI] [PubMed] [Google Scholar]
- Zhang M, Nurse CA. CO2/pH chemosensory signalling in co-cultures of rat carotid body receptors and petrosal neurons: role of ATP and ACh. J Neurophysiol. 2004;92:3433–3445. doi: 10.1152/jn.01099.2003. [DOI] [PubMed] [Google Scholar]