Abstract
We combine free-energy calculations and molecular dynamics to elucidate a mechanism for DNA base-pair binding and unbinding in atomic detail. Specifically, transition-path sampling is used to overcome computational limitations associated with conventional techniques to harvest many trajectories for the flipping of a terminal cytosine in a 3-bp oligomer in explicit water. Comparison with free-energy projections obtained with umbrella sampling reveals four coordinates that separate true dynamic transition states from stable reactant and product states. Unbinding proceeds via two qualitatively different pathways: one in which the flipping base breaks its intramolecular hydrogen bonds before it unstacks and another in which it ruptures both sets of interactions simultaneously. Both on- and off-pathway intermediates are observed. The relation of the results to coarse-grained models for DNA-based biosensors is discussed.
Microdevices that identify specific DNA sequences and/or mismatches are widely used in genomics applications. Many of these devices operate by detecting the hybridization of solubilized oligomers to surface-attached probe sequences (1–6). Effective design of these device platforms would be aided by models that describe the dynamics of DNA hybridization. There are a number of phenomenological and coarse-grained statistical mechanical models used in this endeavor (7–12). However, such theories require certain assumptions regarding the microscopic dynamics associated with hybridization, about which little is known. In particular, there is insufficient understanding of the mechanism underlying the fundamental event in this process, the binding/unbinding of an individual base pair. In this work we use atomistic simulations to examine the free energy and dynamics of this important reaction and thereby elucidate its mechanism.
Nucleic acids have been examined in several atomistic simulation studies. By driving the reaction along a coordinate based on the distance between bases, Norberg and Nilsson (13) calculated the free energy for unstacking three nucleotides in single-stranded RNA. The free energy for flipping an internal base in a B-form DNA dodecamer in the presence (14) and absence (15–17) of a methyltransferase enzyme has been investigated in a similar manner. Most closely related to the present work, Pohorille et al. (18) determined the free energy for unbinding of an end base pair in DNA as a function of the distance between hydrogen-bonding groups and obtained a single 0.45-ns molecular dynamics trajectory. Based on these data, they concluded that this coordinate alone was not sufficient to describe the dynamics.
Boczko and Brooks (19) have argued that multiple order parameters are required to describe complex biomolecular reactions such as those described above. Although this seems often to be true, it is important to point out that, in studies where reactive trajectories were compared directly with free-energy projections (20, 21), the order parameters that described the dynamics were found to be system-specific. Indeed, free-energy surfaces that are functions of dynamically irrelevant variables can be misleading in that rate-determining free-energy barriers can be obscured. In such a case, probability densities do not evolve on the calculated free-energy surface in a way consistent with the corresponding projection of the microscopic dynamics. To avoid this pitfall, we use transition-path sampling (TPS) (22, 23) to overcome limitations associated with conventional simulations to harvest many reactive trajectories. These explicit dynamic data enable us to demonstrate the kinetic pathways by which a base pair binds/unbinds.
We focus on the flipping of a pyrimidine end base of a 3-bp oligomer in explicit solvent. This represents the first step in the melting of short DNA chains. We identify four order parameters that provide a meaningful description of the reaction. Two monitor the interaction energy and distance between the cytosine and guanine that unbind, and two track the interaction energy and distances associated with intrastrand stacking (Fig. 1). The distances describe the overall reaction, whereas the energies give a more detailed view of the initial unbinding process. Several coordinates based on the numbers and properties of water molecules near the DNA bases were examined as well and found to be superfluous. The analysis reveals that there are two qualitatively different reactive pathways: one in which the flipping base breaks its intramolecular hydrogen bonds before it unstacks and another in which it ruptures both sets of interactions simultaneously. In both cases, transition states are found at the point when all CG interactions are reduced to the magnitude of the thermal energy. These states are associated with the rate-determining process, in which the cytosine diffuses away from the guanine into a state with increased conformational freedom. The implications of these findings for the design of better coarse-grained models and experiments are discussed.
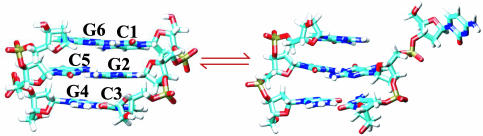
Fig. 1.
Reaction being investigated, which is the flipping of C1. The nomenclature used to denote the individual bases is shown in the native state (Left). (Right) Unfolded state.
Methods
Here we summarize the methods; details are given in Supporting Methods, which is published as supporting information on the PNAS web site. All calculations were performed with a version of charmm (24) c28b2 that we modified to include TPS.
System. We model the base-pairing and -unpairing at the 5′ end of a CGC DNA sequence with its complementary strand. A 3-bp oligomer is the smallest duplex system that contains interior bases and is thermodynamically stable at 300 K. CGC is more stable as a duplex than a sequence containing A or T. The oligomer was solvated in a (34 Å)3 box containing 1,345 TIP3 water molecules and four sodium (Na+) ions that were included to make the system neutral. This corresponds to a Na+ concentration of ≈0.2 M. Although salt concentration is known to influence the stability of DNA duplexes (7, 25, 26), we chose not to include additional ions for simplicity. We did not expect counterions to play a direct role in the reaction, and, consistent with this idea, the included ions were generally quite mobile in our simulations. Periodic boundary conditions were applied, and the particle mesh Ewald method (27) was used to treat the nonbonded interactions.
Obtaining Reactive Trajectories. To start TPS, it is necessary to define reactant and product basins in terms of a set of order parameters and generate at least one trajectory. The initial path need not be physical but must connect the basins. Although all our studies are conducted at 300 K, to obtain a set of such initial trajectories we performed 43 molecular dynamics simulations, each of which started in the native state and proceeded for 500 ps at high temperature (400 K). Unpairing events occurred in 21 of these trajectories (see Table 1, which is published as supporting information on the PNAS web site), of which 15 involved the bases of interest, C1 and G6 (Fig. 1). In almost all these cases, unpairing occurred by a stepwise process in which one base unstacked significantly before the other. In the remainder of the article, we concentrate on reactions in which C1 flips and define the unfolded state as that in which C1 does not interact with either G2 or G6 (see Fig. 1).
Determining a set of order parameters that reflects the dynamics of the system is a trial-and-error procedure (23). A set of order parameters that does not characterize the stable states properly is indicated by dynamical trajectories that rapidly relax from one basin to the other. As described in Supporting Methods (see Tables 2 and 3 and Figs. 6–12, which are published as supporting information on the PNAS web site), we tried many sets of order parameters, a number of which characterized properties of the solvent. We eventually found that the following four coordinates served as suitable order parameters: the distance (d16) and the interaction energy (E16) between the unpairing bases, C1 and G6, and the distance (d12) and the interaction energy (E12) between the unstacking bases, C1 and G2. The energy coordinates (E12 and E16) do not include energy associated with the backbone atoms. As explained in TPS of the End Base-Pair Unfolding in Supporting Methods, the interaction energies provide a good description of the reaction near the native state, but the distances are required to describe regions in the vicinity of the unfolded state.
The coordinates E12, E16, d12, and d16 were used as the basis for a TPS procedure. Specifically, we harvested trajectories 400 ps in length in which one endpoint was in the basin corresponding to the bound state (E12 < –4.0 and E16 < –12.0), and there was a point anywhere along the path in the unfolded basin (12.5 < d12 < 14.5 and 17.0 < d16 < 20.0). Throughout the article, values of E12 and E16 are given in kcal/mol and d12 and d16 are reported in angstroms. As discussed in Supporting Methods (see Fig. 13, which is published as supporting information on the PNAS web site), the length of the trajectories was chosen to be 400 ps to ensure that they are longer than the relaxation time across the barrier. Two types of moves were used to generate trial paths: shifting and shooting (23). Because the system is highly diffusive, no velocity perturbation was applied in the shooting moves; rather, the motions of the waters far away (>15.0 Å) from the center of the DNA molecule were simulated with a stochastic (Langevin) algorithm, and the path was updated in only one direction. Shooting in only one direction at a time significantly increased the acceptance rate, consistent with ref. 28.
Free Energy. As described above, order parameters are considered to provide a good description of the reaction if projection of the atomistic dynamics onto these coordinates is consistent with the topography of the free-energy landscape projected onto these variables. In other words, we consider these coarse-grained order parameters to be the significant slow variables in the binding–unbinding kinetics. We used umbrella sampling to obtain the free energy first as a function of E12 and E16 and then as a function of d12 and d16. Thus, in the first set of simulations, harmonic terms were used to constrain the system to be close to particular values of E12 and E16 and, in the second set, constraints were imposed in terms of d12 and d16.
Results
Overall Mechanism. In this section we project both the dynamics and free energy of the flipping of C1 (see Fig. 1) onto the energy (E12 and E16) and the distance (d12 and d16) coordinates. Stacking of bases C1 and G2 is monitored by E12 or d12, whereas the pairing of C1 and G6 is monitored by E16 or d16. It is important to note that E16 registers contributions from stacking interactions between C1 and G6 in nonnative conformations. The frequency of observing values of these coordinates during TPS dynamics is shown in Fig. 2. Fig. 2a, which shows E12 and E16, reveals that at least two coordinates are necessary to describe the reaction, because there are two qualitatively different paths between the native state (E12 ≈ –6.5 and E16 ≈ –16; d12 ≈ 4 and d16 ≈ 6) and the completely unfolded state (E12 ≈ 0 and E16 ≈ 0; d12 ≈ 13 and d16 ≈ 16).
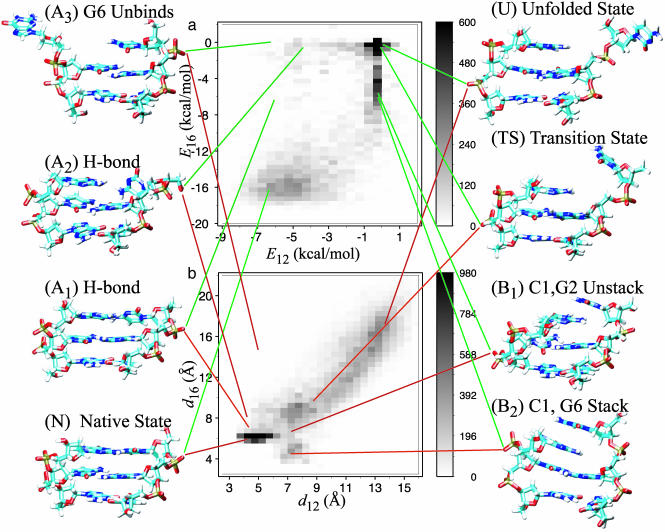
Fig. 2.
The frequency of observing values of the order parameters during 1,650 TPS trajectories. The structures shown next to the plots are snapshots taken from the TPS simulations except A3, which was taken from an umbrella sampling simulation. Note that the gray scale has been cut off at 600 (a) and 980 (b) such that regions with smaller frequencies can be seen. (a) Frequency of observing E12 and E16. (b) Frequency of observing d12 and d16.
Along the first pathway, the hydrogen bonds between C1 and G6 break first, and then C1 unstacks from G2. The hydrogen bonds break mainly through twisting motions of the bases. Presumably, twisting motions are more likely than linear separation, because the angular dependence (felt over ≈22°) of hydrogen bond energies is softer than the distance dependence. Because twisting is the dominant motion along this pathway, the distance coordinate, d16 (see Fig. 2b), does not provide as good a description of this process as E16. The process typically begins with rotation about the glycosidic bond, which weakens all three hydrogen bonds, as well as a swinging motion in the original plane of the base that causes one of the outer hydrogen bonds to break first. This is illustrated in structure A1 (E12 ≈ –6.2 and E16 ≈ –6.6; d12 ≈ 4.2 and d16 ≈ 6.6), in which the foremost hydrogen bond has broken. The rotation proceeds until all the hydrogen bonds are broken and C1 begins to unbind as shown in structure A2 (E12 ≈ –4.5 and E16 ≈ –0.5; d12 ≈ 4.2 and d16 ≈ 8.7). From this point, the path proceeds through the transition state (TS) to the unfolded state (U) as described below. The twisting leading to A1 often involves significant motion on the part of G6, which enables the hydrogen bonds to break while minimizing disruption of the stacking between C1 and G2. This can also lead to an off-pathway stable state, A3 (E12 ≈ –6.5 and E16 = 0; d12 ≈ 4.8 and d16 ≈ 15.3). In this work, we only consider trajectories that lead to C1 unbinding.
Along the second pathway, C1 unstacks (E12 ≈ –0.5) concomitantly with a decrease in E16 to ≈–6(d12 ≈ 7 and d16 ≈ 6), denoted as B1. As in the case of A1, there are two very weak hydrogen bonds remaining in B1 but also some interactions between the ring atoms in C1 and G6. At this point, some trajectories enter an off-pathway kinetic intermediate (B2) in which C1 stacks on top of G6 (E12 ≈ –0.5 and E16 ≈ –6; d12 ≈ 7 and d16 ≈ 4). Stacking interactions are the main contribution to E16 in B2, because there are no hydrogen bonds between C1 and G6. This structure sometimes persists for several hundred picoseconds. A similar structure was described in ref. 18. Further progress along this path occurs when C1 swings away and breaks the remaining hydrogen bond (B1) or the stacking interaction (B2).
The two pathways rejoin at the point (TS) where the magnitudes of the interaction energies are comparable with the thermal energy (E12 ≈ E16 ≈–0.1; d12 ≈ 9 and d16 ≈ 10). The completely unfolded state (U) is reached when C1 entirely disengages from G6 by diffusing to d12≈13 and d16≈16. As explained next, it is necessary to use the distances to describe this process because the energy coordinates approach zero and do not contain useful information about configurational degrees of freedom relaxing to the entropically stabilized free-energy minimum.
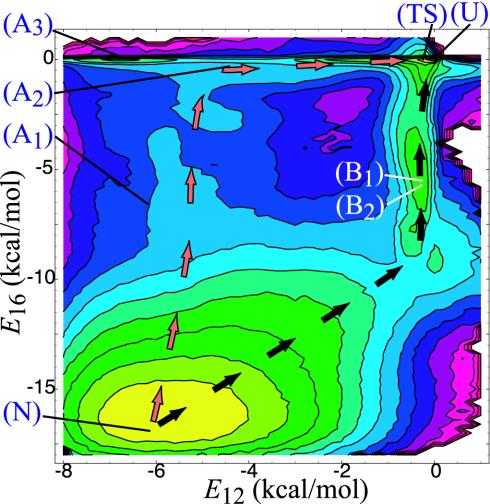
Free Energy. Because E16 and E12 appear to describe the dynamics near the native state well, we have projected the free energy onto them as shown in Figs. 3 and 4. The two paths described above correspond to low free-energy troughs that connect the two major basins. In addition, there are local minima that correspond to A3 and B2.
Fig. 3.
Projection of free energy onto E12 and E16 (contour lines are separated by 0.5 kcal/mol). The letters overlayed on the plots correspond to the location of the structures depicted in Fig. 2. The arrows were drawn by hand to indicate the two predominant paths discussed in the text.
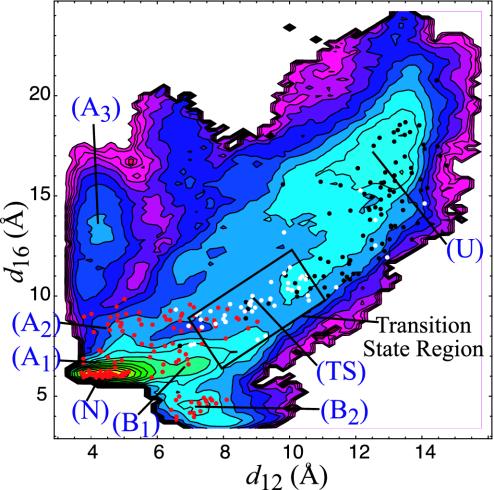
Fig. 4.
Projection of free energy onto d12 and d16 (contour lines are separated by 0.5 kcal/mol). The points overlayed on the plot correspond to the structures from which the committors were estimated. Color coding: red, tends not to unfold (0 ≤ Pu < 0.35); white, “transition state” (0.35 ≤ Pu ≤ 0.65); black, tends to unfold (0.65 < Pu ≤ 1.0). During the estimation, the unfolded state was defined as d12 > 10.75 and d16 > 11. Only structures that reached a stable state were counted in the estimation of Pu. The other stable states were defined as the native state (d12 < 9.0; 5.5 < d16 < 8.0), G6 unfolds (d12 < 7.0; 8.0 < d16 < 18), and the stacked state (6.0 < d12 < 9.0; d16 < 5.5). The region marked with the rectangle has been identified as the “transition state region” (see Fig. 5). The qualitative distribution of committor values and location of the transition-state region are relatively insensitive to the definitions of the stable and transition states.
In the region of phase space near the unfolded region (E12 ≈ 0 and E16 ≈ 0), large changes in spatial position correspond to small changes in the interaction energies, which results in a projection of the unfolded basin that is extremely narrow with a high degree of curvature. Thus, in this region, E12 and E16 are no longer slow variables compared with molecular time scales. Rather, the slow variables in this region are d12 and d16. Thus, we have projected the free energy onto these coordinates (see Fig. 4). On this projection, we can see that, although the unfolded basin begins at approximately d12 ≈ 10 and d16 ≈ 10, C1 has to diffuse to approximately d12 ≈ 12.5 and d16 ≈ 15 before the free energy falls to more than kBT below the saddle point, indicating that there is only a weak driving force to enter the unfolded basin. This is because there are only a few degrees of freedom contributing to the entropically stabilized basin when only one base pair unbinds.
Integration of the Boltzmann weights over the basins in Fig. 4 yields a free-energy difference of ΔG ≈ 1.5 kcal/mol for the unfolded state relative to the native state. This free-energy change is in reasonable agreement with the value one obtains from empirical nearest-neighbor potentials, ΔG ≈ 1.8 (26). However, it is important to keep in mind that empirical potentials derive from experiments in which the entire molecule melts and are not precise estimates for the unbinding of an individual base pair. In addition, integration of the basins corresponding to A3 and B2 results in free-energy differences relative to the native state of 3.3 and 3.0 kcal/mol, respectively. To characterize the basin corresponding to A3 fully, however, we would have to include a coordinate that characterizes the stacking of G6.
Transition States. To determine whether the coordinates E12 and E16 or d12 and d16 are sufficient to characterize the transition states of the reaction in addition to the stable state, we tested selected structures from the TPS trajectories for their probabilities of committing to the bound and unfolded basins (Pb and Pu, respectively). Each selected structure was subjected to 10 trials, each of which began with velocities assigned randomly from a Maxwell–Boltzmann distribution and proceeded for 200 ps. A trial contributed positively to Pu if its final structure was in the unfolded basin. We counted A3, B2, and the native state (N) as a single basin because the TPS trajectories indicate that the rate-limiting step is going from one of these states to the fully unfolded state. If it were a two-state reaction with strongly attracting basins and a true reaction coordinate were identified, we would expect the distribution of Pu for structures close to the free-energy saddle point to be sharply peaked at 0.5.
The results of the committor calculation are overlayed on the free-energy projection in Fig. 4. We note that in estimating Pu, the error in an individual commitor estimation that uses 10 trials is ≈0.3 (23, 29). Therefore, although we labeled the structures for which Pu ≈ 0.5 as “transition states” in Fig. 4 (the white points), we stress the statistical uncertainty in this designation. We present these committor estimations not to identify specific structures as transition states but to locate a transition-state region in which a large fraction of structures are near Pu ≈ 0.5. Although statistical uncertainty in the committor estimation precludes precise identification of structures that comprise the transition-state ensemble, it does not prevent us from qualitatively locating a region containing the transition states.
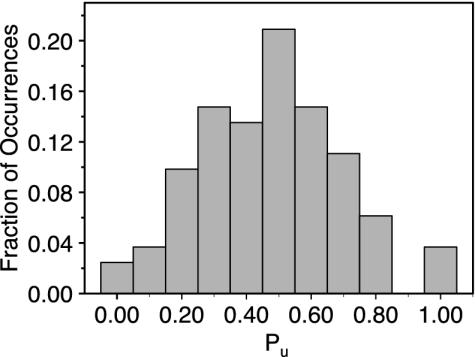
We see in Fig. 4 that the transition states primarily fall at a saddle point on this surface (d12 ≈ 9, d16 ≈ 9), although the correspondence is not perfect. The frequency of observing commitor values in this region is shown in Fig. 5; there is a broad peak centered at ≈0.5. This location can be understood as follows. The magnitudes of the interaction energies in this area are in the range 0.1 < |E1i| < 1.0 kcal/mol, where i is 2 or 6. The unfolded basin is entropically stabilized at large d12 and d16 because E12 ≈ 0 and E16 ≈ 0, which allows the greatest conformational freedom for the C1 base. In contrast, the other basins are energetically stabilized. The transition states thus fall at the crossover, where the entropy is reduced by the fact that nonzero E12 and E16 restrict the spatial location and orientations of the base but are not significantly larger than the thermal energy (kBT = 0.6 kcal/mol).
Fig. 5.
The probability of observing values of Pu in the transition-state region (boxed region in Fig. 4).
Given the complexity of the system and the small number of trial trajectories, which result in large errors in commitor estimations, the description of the transition-state region in Fig. 4 is surprisingly good. It is still possible that additional coordinates may be necessary to characterize the reaction in further detail. However, we tried a number of other order parameters (as described in Coordinates in Supporting Methods), none of which significantly improved the precision with which we are able to identify the transition-state ensemble.
Discussion
This study integrates free-energy calculations and atomistic dynamics to elucidate a detailed mechanism for base-pair binding and unbinding in DNA. We found four coordinates that describe the kinetic pathway well. Two of them monitor the interaction energies between the flipping base (C1) and its nearest neighbors in the native structure (G2 and G6). These parameters indicate that, close to the native state, the dynamics is dominated by specific intramolecular hydrogen bonds and stacking interactions. In particular, there are two pathways to unfolding at 300 K: one in which the hydrogen bonds first break via twisting of bases C1 and G6, followed by unstacking of C1 from G2, and one in which C1 simultaneously breaks the stacking interactions and the hydrogen bonds with G6. The other two coordinates that we identified are pairwise center-of-mass distances between the flipping base and its neighbors (G2 and G6). These parameters do not perfectly describe the initial exit from the native state, which involves twisting out of hydrogen bonds. However, they effectively describe the reaction outside this region, which is the location of the transition states between folded or misfolded states and the unfolded state. These states occur at the crossover between the energy- and entropy-dominated regions, where the interaction energy is about equal to the thermal energy. Given that E12 (E16) and d12 (d16) can be related in some areas of phase space, it is possible that one could reduce the number of coordinates to three or two by interpolating between the energies and distances, but we chose not to do so because the separate coordinates are physically more transparent.
The goal of this investigation is to understand the microscopic details of DNA hybridization/melting to validate or improve existing coarse-grained models of DNA. For example, Brownian dynamics simulations were used to study the kinetics of melting in a model in which each nucleotide was represented by two sites (11, 30). This model seems to capture many of the details of melting successfully. However, it is not a priori obvious that the simplified coordinates remaining in such a model are sufficient to describe all the important reaction dynamics. In particular, this model assumes that there are no important reaction coordinates associated with the solvent structure.
With these concerns in mind, we tested many coordinates related to properties of water for their ability to describe the reaction. These order parameters included the interaction energy between water and the hydrogen-bonding groups in the unpairing bases, the number of waters between stacking bases, and the hydration number of the unbinding bases. As described in Supporting Methods, however, we did not find any combination of these coordinates that was sufficient to describe the unfolded state or the location of the transition states. The values of these coordinates usually changed concomitantly with the breaking of hydrogen bonds, which is registered in E16. Coordinates that monitor water properties could be more important for describing the flipping of an interior base (as in refs. 14–17) because there is a greater change in hydration in that case.
The location of the transition states and free-energy saddle point indicate that the outcome of a trajectory is not determined by the breaking of the DNA–DNA hydrogen bonds or subsequent formation of DNA–water hydrogen bonds. It is furthermore not decided by the penetration of water between stacking bases, such as might be the case in a reaction dominated by hydrophobic forces. Rather, unfolding requires that C1 diffuse away from G6 to a distance at which the conformational freedom is maximized and the DNA–DNA bonds cannot reform easily. There is only a weak bias for the base to diffuse away due to the entropic stabilization of the unfolded state. The fate of a transition thus is decided by a random walk across a region that appears as a broad plateau on the free-energy surface (Fig. 4). The entropic stabilization is limited in this case because there is only a small increase in the number of conformations available to the DNA after the unbinding of one base. This can be contrasted with the cases in which an entire chain unfolds, such as protein unfolding (31, 32), in which there is a larger increase in available conformations and thus a much stronger entropic driving force.
The fact that transitions are determined by structures in which stacking and hydrogen bonding are mostly dissolved does not mean that the rate of unfolding is independent of the strength of these bonds. The frequency of random walks that cross the transition-state region is directly related to the probability that a trajectory surmounts the free-energy cliff leading to this region: the breaking of hydrogen bonds and stacking interactions.
The fact that the dynamics are well described by coordinates (energies and distances) that can be calculated from positions of only the DNA atoms might imply that implicit solvent models are adequate for describing nucleic acids. However, it is important to consider that both the dynamics and free-energy calculations included explicit solvent atoms, the effect of which is therefore included in the coordinates we have used. In addition, our description of the reaction is not perfect, and unidentified coordinates related to the solvent could still play a role.
Conclusions
In summary, we have projected the atomistic dynamics and the free energy of a DNA base-pair binding/unbinding onto four order parameters. This has enabled us to identify the kinetic pathways by which the reaction proceeds, several kinetic intermediates, and the approximate location of the dynamical transition states. The identification of suitable order parameters allows the determination of rate constants (23, 33, 34). These rates can serve as the basis for testing and further development of coarse-grained models.
Supplementary Material
Acknowledgments
We thank Arun Majumdar for fruitful discussions. Financial support was provided by the Defense Advanced Research Projects Agency. A.R.D. was also supported by a Burroughs Wellcome Fund Hitchings–Elion Fellowship. D.C. is supported by the National Science Foundation.
Abbreviation: TPS, transition-path sampling.
References
- 1.Demers, L. M., Mirkin, C. A., Mucic, R. C., Reynolds, R. A. I., Letsinger, R. L., Elghanian, R. & Viswanadham, G. (2000) Anal. Chem. 72, 5535–5541. [DOI] [PubMed] [Google Scholar]
- 2.Fritz, J., Baller, M. K., Lang, H. P., Rothuizen, H., Vettiger, P., Meyer, E., Guüntherodt, H., Gerber, C. & Gimzewski, J. K. (2000) Science 288, 316–318. [DOI] [PubMed] [Google Scholar]
- 3.Wu, G., Ji, H., Hansen, K., Thundat, T., Datar, R., Cote, R., Hagan, M. F., Chakraborty, A. K. & Majumdar, A. (2001) Proc. Natl. Acad. Sci. USA 98, 1560–1564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stimpson, D. I., Hoijer, J. V., Hsieh, W., Jou, C., Gordon, J., Theriault, T., Gamble, R. & Baldeschwieler, J. D. (1994) Proc. Natl. Acad. Sci. USA 92, 6379–6383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Storhoff, J. J., Elghanian, R., Mucic, R. C., Mirkin, C. A. & Letsinger, R. L. (1998) J. Am. Chem. Soc. 120, 1959–1964. [Google Scholar]
- 6.Peterlinz, K. A., Georgiadis, R. M., Herne, T. M. & Tarlov, M. J. (1997) J. Am. Chem. Soc. 119, 3401–3402. [Google Scholar]
- 7.Wetmur, J. G. (1991) Crit. Rev. Biochem. Mol. Biol. 26, 227–259. [DOI] [PubMed] [Google Scholar]
- 8.Peyrard, M. & Bishop, A. R. (1989) Phys. Rev. Lett. 62, 2755–2758. [DOI] [PubMed] [Google Scholar]
- 9.Zhang, Y., Zheng, W., Liu, J. & Chen, Y. Z. (1997) Phys. Rev. E Stat. Phys. Plasmas Fluids Relat. Interdiscip. Top. 56, 7100–7115. [Google Scholar]
- 10.Campa, A. & Giansanti, A. (1999) J. Biol. Phys. 24, 141–155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Drukker, K. & Schatz, G. C. (2000) J. Phys. Chem. B 104, 6108–6111. [Google Scholar]
- 12.Jin, R., Wu, G., Li, Z., Mirkin, C. & Schatz, G. C. (2003) J. Am. Chem. Soc. 125, 1643–1654. [DOI] [PubMed] [Google Scholar]
- 13.Norberg, J. & Nilsson, L. (1996) J. Phys. Chem. 100, 2550–2554. [Google Scholar]
- 14.Huang, N., Banavali, N. K. & MacKerell, A. D. J. (2003) Proc. Natl. Acad. Sci. USA 100, 68–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Giudice, E., Varnai, P. & Lavery, R. (2001) Chem. Phys. Chem. 2, 673–677. [DOI] [PubMed] [Google Scholar]
- 16.Giudice, E., Varnai, P. & Lavery, R. (2003) Nucleic Acids Res. 31, 1434–1443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Banavali, N. K. & MacKerrel, A. D. J. (2002) J. Mol. Biol. 319, 141–160. [DOI] [PubMed] [Google Scholar]
- 18.Pohorille, A., Ross, W. S. & Tinoco, I. J. (1990) Int. J. Supercomput. Appl. 4, 81–96. [DOI] [PubMed] [Google Scholar]
- 19.Boczko, E. M. & Brooks, C. L., III (1996) Science 269, 393–396. [DOI] [PubMed] [Google Scholar]
- 20.Dinner, A. R. & Karplus, M. (1999) J. Phys. Chem. B 103, 7976–7994. [Google Scholar]
- 21.Bolhuis, P. G. (2003) Proc. Natl. Acad. Sci. USA 100, 12129–12134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bolhuis, P. G., Chandler, D., Dellago, C. & Geissler, P. L. (2002) Annu. Rev. Phys. Chem. 53, 291–318. [DOI] [PubMed] [Google Scholar]
- 23.Dellago, C., Bolhuis, P. G. & Geissler, P. L. (2001) Adv. Chem. Phys. 123, 1–78. [Google Scholar]
- 24.Brooks, B. R., Bruccoleri, R. E., Olafson, B. D., States, D. J., Swaminathan, S. & Karplus, M. (1983) J. Comput. Chem. 4, 187–217. [Google Scholar]
- 25.Wetmur, J. G. & Davidson, N. (1968) J. Mol. Biol. 31, 349–370. [DOI] [PubMed] [Google Scholar]
- 26.Bloomfield, V. A., Crothers, D. M. & Tinoco, I. J. (2000) Nucleic Acids: Structures, Properties, and Functions (University Science Books, Sausilito, CA).
- 27.Essmann, U., Perera, L., Berkowitz, M. L., Darden, T., Lee, H. & Pedersen, L. G. (1995) J. Chem. Phys. 103, 8577–8593. [Google Scholar]
- 28.Bolhuis, P. G. (2003) J. Phys. Condens. Matter 15, S113–S120. [Google Scholar]
- 29.McCormick, T. (2003) Ph.D. thesis (Univ. of California, Berkeley).
- 30.Drukker, K., Wu, G. & Schatz, G. C. (2001) J. Chem. Phys. 114, 579–590. [Google Scholar]
- 31.Bryngelson, J. D. & Wolynes, P. G. (1987) Proc. Natl. Acad. Sci. USA 84, 7524–7528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Karplus, M. & Shakhnovich, E. (1992) in Protein Folding, ed. Creighton, T. E. (Freeman, New York), pp. 127–193.
- 33.Dellago, C., Bolhuis, P. G., Csajka, F. S. & Chandler, D. (1998) J. Chem. Phys. 108, 1964–1977. [Google Scholar]
- 34.van Erp, T. S., Moroni, D. & Bolhuis, P. G. (2003) J. Chem. Phys. 118, 7762–7774. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.