Abstract
Pulmonary arterial hypertension (PAH) is a life-threatening disease characterized by progressive PAH and right ventricular failure. Despite recent advances in therapeutic approaches using prostanoids, endothelin antagonists, and so on, PAH remains a challenging condition. To develop a novel therapeutic approach, we have established a nonviral gene delivery system of poly(ethylene glycol) (PEG)-based block catiomers, which form a polyplex nanomicelle with a nanoscaled core–shell structure in the presence of DNA. The polyplex nanomicelle from PEG-b-poly{N-[N-(2-aminoethyl)-2-aminoethyl]aspartamide} (PEG-b-P[Asp(DET)]), having ethylenediamine units at the side chain, showed ~100-fold increase in luciferase transgene expression activity in mouse lung via intratracheal administration with a minimal toxicity compared with the polyplex from linear poly(ethylenimine) (LPEI). The transfection activity was highest on day 3 after administration and remained detectable until day 14. PEG-b-P[Asp(DET)] polyplex nanomicelles were formulated with a therapeutic plasmid bearing the human adrenomedullin (AM) gene and intratracheally administered to rats with monocrotaline-induced pulmonary hypertension. The right ventricular pressure significantly decreased 3 days after administration as confirmed by a notable increase of pulmonary human AM mRNA levels. Intratracheal administration of PEG-b-P[Asp-(DET)] polyplex nanomicelles showed remarkable therapeutic efficacy with PAH animal models without compromising biocompatibility.
Introduction
Idiopathic pulmonary arterial hypertension (PAH) is a rare disease characterized by a progressive increase in pulmonary vascular resistance, leading to right heart failure and death.1 Recent advances in therapeutic approaches to PAH show promising targeting pathways believed to play critical pathogenic or pathophysiologic roles;2 however, despite these findings, PAH remains a challenging condition.3
Adrenomedullin (AM), a peptide isolated from human pheochromocytoma,4 has multiple beneficial effects on cardiovascular tissues, including a powerful hypotensive effect.5 Moreover, AM is indicated for PAH because of its prodilatory effects and the abundance of AM receptors in the lung.6 Inhalation of AM was reported to ameliorate PAH in animal models7 as well as in PAH patients without inducing systemic hypotension, but this effect was transient.8 To overcome these barriers, a new, efficacious, and long-lasting AM therapy for PAH is warranted.
Gene therapy is one of the strategic approaches to continuously supply therapeutic peptides or proteins to target tissues.6 Gene delivery to the lung via inhalation can avoid many problems associated with intravenous delivery, such as immediate nuclease degradation in the blood stream and the difficulty associated with penetrating endothelial barriers. In this regard, AM-based gene therapy through intratracheal route for PAH may have a promise.7 Successful gene delivery via inhalation strongly depends on the development of advanced gene vectors to protect the therapeutic plasmid, provide site-specific targeting, and effectively release these plasmids for the desired pharmacological effect.
To develop a novel gene therapy system, we utilized our established polymeric library consisting of poly(ethylene glycol) (PEG)-based block catiomers, which form core–shell polyplex nanomicelles with core sequestration of the therapeutic plasmid.9,10 These polyplex nanomicelles are well dispersed even in aqueous media containing serum proteins and protect plasmid DNA from degradation by nuclease in vivo.11,12,13 We recently developed P[Asp(DET)], a poly(aspartamide) derivative bearing an N-(2-aminoethyl)aminoethyl group as the side chain, that showed improved transfection efficiency and biocompatibility compared to linear poly(ethylenimine) (LPEI).14 The PEG-based block catiomer with P[Asp(DET)] was applied in vivo to deliver therapeutic plasmids for a murine, skull bone defect model and a rabbit carotid artery with neointima model; its successful therapeutic efficacy with these mammalian studies provided the impetus for expanded application into the treatment of intractable diseases suited for gene therapy.15,16
In this paper, we report advanced, pulmonary transfection efficiencies using intratracheally inhaled PEG-b-P[Asp(DET)] polymeric nanomicelles without compromising biocompatibility. The intratracheal administration of the AM gene by PEG-b-P[Asp-(DET)] polyplex nanomicelles reduced right ventricular pressure in PAH animal models without inducing inflammation, suggesting its suitability as a vector for translational research.
Results
Reporter gene transfer using PEG-b-P[Asp(DET)] via intratracheal administration
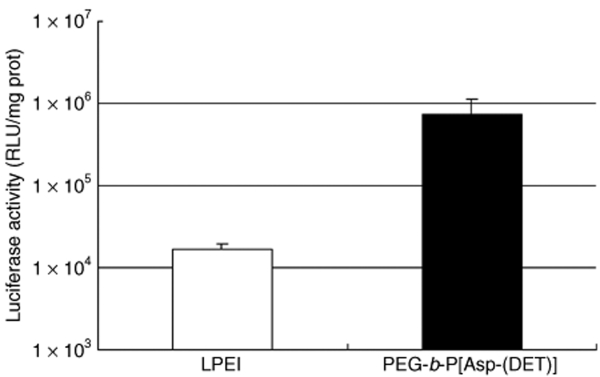
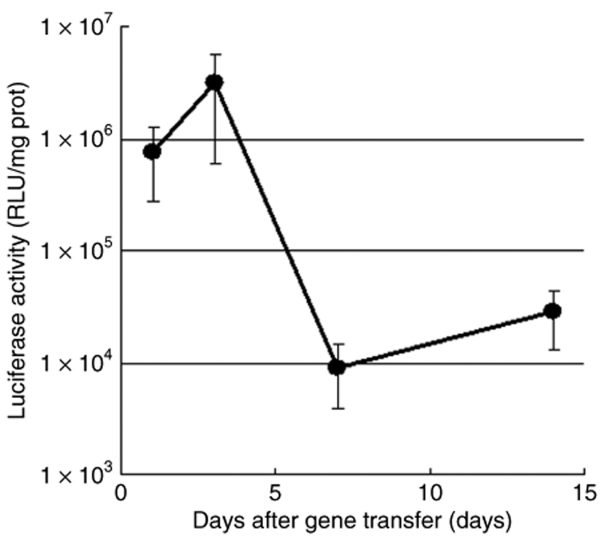
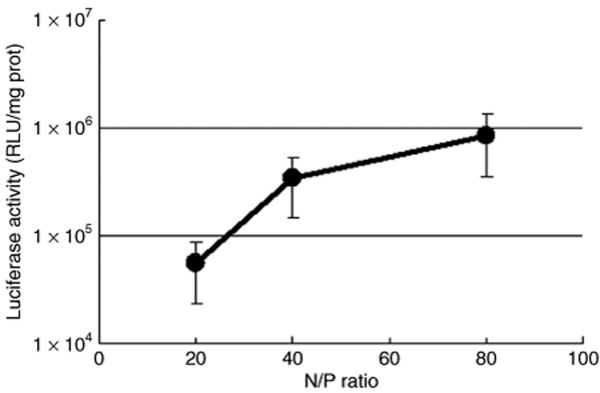
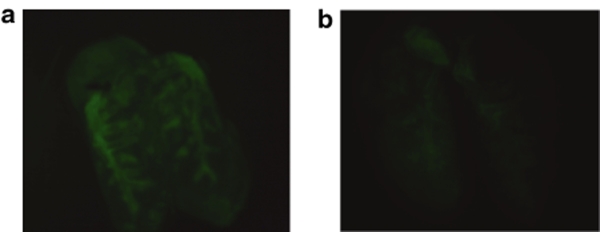
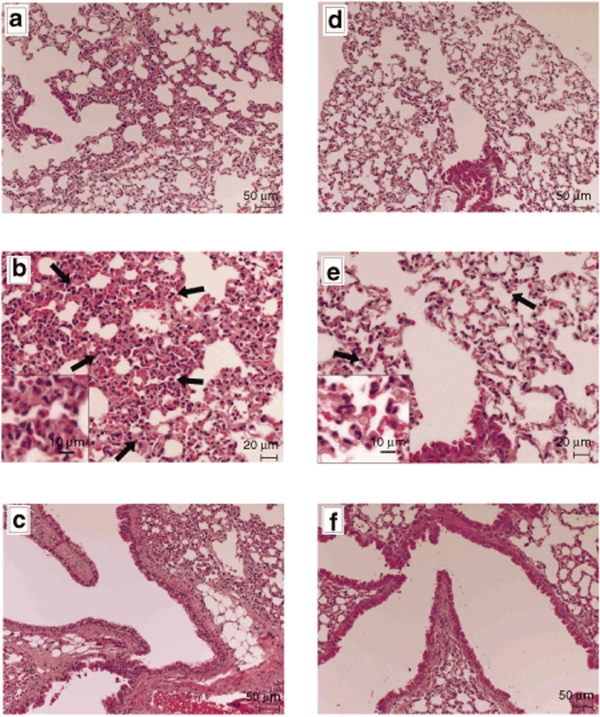
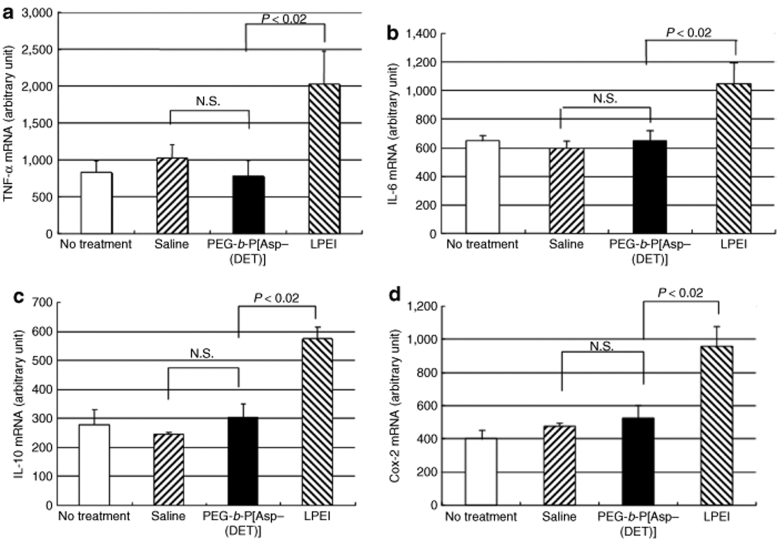
Plasmids bearing the luciferase reporter gene were formulated with PEG-b-P[Asp(DET)] (N/P = 80) and LPEI (N/P = 6) and were sprayed intratracheally into ICR mice. Here, N/P ratio refers to the unit molar ratio of the amino group in the polymer to the phosphate group in the plasmid DNA. After 1 day, the mice were killed and the pulmonary tissues were harvested to quantify luciferase activity. PEG-b-P[Asp(DET)] polyplex nanomicelles showed nearly a 100-fold increase in luciferase levels than the LPEI controls (Figure 1). Figure 2 shows the time-dependent changes of luciferase gene expression in the pulmonary tissue with PEG-b-P[Asp(DET)] polyplex nanomicelles. Luciferase activity was highest on day 3, and remained detectable until day 14. To elucidate the effect of PEG-b-P([Asp(DET)]/pLuc N/P ratios on luciferase gene expression, a series of the nanomicelles formulated under the varying N/P ratios (20, 40, and 80) were also examined. Figure 3 shows an ~50-fold increase in luciferase expression from N/P = 20 to N/P = 80 over a 3-day period. Next, PEG-b-P[Asp(DET)] polyplex nanomicelles loaded with plasmid DNA bearing the yellow fluorescence protein (YFP) gene (N/P = 80) or LPEI/pYFP polyplexes (N/P = 6) were sprayed intratracheally in ICR mice. After 1 day, the pulmonary tissue was harvested and the YFP gene expression was visualized by fluorescence microscopy (Figure 4). Significantly higher fluorescence intensity was clearly seen in the lungs treated with the PEG-b-P[Asp(DET)] polyplex nanomicelle than the LPEI polyplexes; moreover, YFP fluorescence activity was distinctly visible for the animals treated with PEG-b-P[Asp(DET)] polyplex micelles in the secondary bronchi and lower pulmonary generations. To evaluate the toxicity, immunohistochemistry was conducted on the lung tissues after the transfection of LPEI/pLuc controls (N/P = 6) (Figure 5a–c) or PEG-b-P[Asp(DET)]/pLuc (N/P = 80) (Figure 5d–f). The lung administered with LPEI/pLuc showed moderate infiltration of neutrophils at the terminal bronchiole and alveoli as indicated (Figure 5a,b). However, the lung administered with PEG-b-P[Asp(DET)]/pLuc, neutrophilic infiltration was scattered and minimal or absent (Figure 5d,e). No apparent inflammatory infiltrate was observed in the bronchus of the both groups (Figure 5c,f). The findings thereby supported increased biocompatibility with the PEG-b-P[Asp(DET)] polyplex micelle. To further evaluate the toxicity of the gene carrier systems, mRNA levels of inflammatory cytokines in the pulmonary tissue were measured using real-time reverse transcriptase (RT)-PCR. A nontreated cohort was used as a control. Proinflammatory cytokine mRNA levels did not increase for intratracheally administered naked pLuc in saline or the PEG-b-P[Asp(DET)] polyplex micelles; however, LPEI/pLuc polyplexes revealed a twofold increase in TNF-α, interleukin (IL)-6, IL-10, and Cox-2 compared to the control (Figure 6a–d). Notably, PEG-b-P[Asp(DET)]/pLuc proinflammatory levels were statistically similar to the negative control cohorts in TNF-α, IL-6, IL-10, and Cox-2 mRNA levels.
Figure 1.
Luciferase gene expression by intratracheal administration of LPEI polyplex (N/P = 6) or PEG-b-P[Asp(DET)] polyplex nanomicelle (N/P = 80). Samples of the polyplex and the polyplex nanomicelle were prepared before the administration and left for 1 day. The mice (five mice per group) were anesthetized and the polyplex or the polyplex nanomicelle was administered intratracheally. At 24 hour postadministration, the lung tissues were harvested, homogenized, and measured for luciferase activity (mean ± SEM, N = 5). LPEI, linear poly(ethylenimine); PEG-b-P[Asp(DET)], PEG-b-poly{N-[N-(2-aminoethyl)-2-aminoethyl]aspartamide}.
Figure 2.
Time-dependent changes in gene expression after intratracheal administration of PEG-b-P[Asp(DET)] polyplex nanomicelle loaded with luciferase gene. The mice (five mice per group) were anesthetized and the polyplex nanomicelle was administered intratracheally. After the indicated time, the lung tissues were harvested, homogenized, and measured for luciferase activity (mean ± SEM, N = 5). PEG-b-P[Asp(DET)], PEG-b-poly{N-[N-(2-aminoethyl)-2-aminoethyl]aspartamide}.
Figure 3.
Charge-ratio-dependent changes in gene expression after intratracheal administration of the PEG-b-P[Asp(DET)] polyplex nanomicelle loaded with luciferase gene. The mice (five mice per group) were anesthetized and the polyplex nanomicelle was administered intratracheally. At 3 days postadministration, the lung tissues were harvested, homogenized, and measured for luciferase activity (mean ± SEM, N = 5). PEG-b-P[Asp(DET)], PEG-b-poly{N-[N-(2-aminoethyl)-2-aminoethyl]aspartamide}.
Figure 4.
Fluorescence photographs of lungs transfected by intratracheal administration of YFP gene using PEG-b-P[Asp(DET)] polyplex nanomicelle or LPEI polyplex. (a) PEG-b-P[Asp(DET)] polyplex nanomicelle (N/P = 80), (b) LPEI polyplex (N/P = 6). The lung specimens were observed under a fluorescence microscope (SZX12; Olympus). LPEI, linear poly(ethylenimine); PEG-b-P[Asp(DET)], PEG-b-poly{N-[N-(2-aminoethyl)-2-aminoethyl]aspartamide}.
Figure 5.
Representative photomicrographs of lung tissues 7 days postintratracheal administration of LPEI/pLuc (N/P = 6) (a–c) or PEG-b-P[Asp(DET)]/pLuc (N/P = 80) (d–f). The terminal bronchiole and alveoli of the lungs administered with LPEI polyplex (a,b) and PEG-b-P[Asp(DET)] polyplex nanomicelle (d,e) are shown. The neutrophil infiltration is indicated with arrows in the photomicrographs with higher magnification (b,e). Each inset is the picture with higher magnification. The bronchus of the lungs administered with LPEI polyplex (c) and PEG-b-P[Asp(DET)] polyplex nanomicelle (f) are also shown. LPEI, linear poly(ethylenimine); PEG-b-P[Asp(DET)], PEG-b-poly{N-[N-(2-aminoethyl)-2-aminoethyl]aspartamide}.
Figure 6.
mRNA expression of inflammatory cytokines in lung tissues 7 days postintratracheal administration of luciferase gene in saline; LPEI polyplex with luciferase gene (N/P = 6); or PEG-b-P[Asp(DET)] polyplex nanomicelle with luciferase gene (N/P = 80) (mean ± SEM, N = 4). (a) TNF-α, (b) IL-6, (c) IL-10, (d) Cox-2. IL, interleukin; LPEI, linear poly(ethylenimine); TNF, tumor necrosis factor; PEG-b-P[Asp(DET)], PEG-b-poly{N-[N-(2-aminoethyl)-2-aminoethyl]aspartamide}.
Effect of AM gene transfer by PEG-b-P[Asp(DET)] polyplex nanomicelle in a rat model of PAH
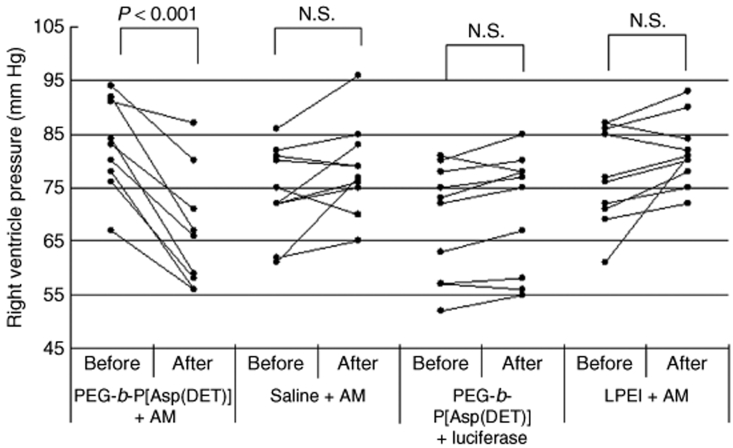
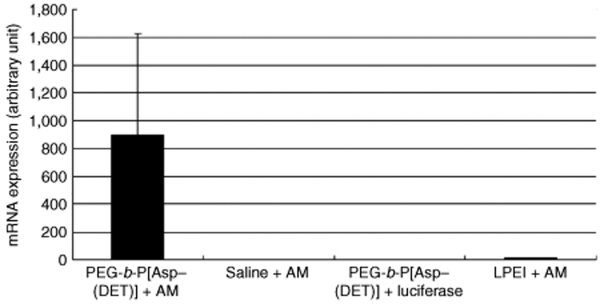
After 4 weeks of monocrotaline injection, right ventricular pressure was increased to twice of the normal value (Figure 7). Notably, right ventricular pressure was decreased significantly by an intratracheal spray of the PEG-b-P[Asp(DET)] polyplex nanomicelle loaded with the expression vector of AM (N/P = 40). On the other hand, right ventricular pressure did not change significantly after administration of naked plasmid encoding the AM gene in saline or the LPEI polyplex loaded with the AM gene, or the polyplex nanomicelle loaded with the luciferase gene. The mRNA levels of human AM in the lung were measured by real-time RT-PCR (Figure 8). The lung transfected with the polyplex nanomicelle loaded with the expression vector of AM had high levels of AM mRNA. Alternatively, the levels were much lower in the lung transfected with the LPEI polyplex loaded with the expression vector of AM. The lung transfected with the polyplex nanomicelle loaded with the luciferase gene or with the naked AM gene in saline showed no expression of human AM.
Figure 7.
Effect of gene transfer on the right ventricle pressure in the PAH rat model. At 4 weeks after subcutaneous monocrotaline injection, a hemodynamic study was performed to measure the RV pressure indicated as “Before”. The PEG-b-P[Asp(DET)] polyplex nanomicelle loaded with AM expression vector; AM expression vector in saline; PEG-b-P[Asp(DET)] polyplex micelle loaded with luciferase gene; or the LPEI polyplex loaded with AM expression vector were sprayed intratracheally. Three days later, a hemodynamic study was performed again to measure the RV pressure indicated as “After”. AM, adrenomedullin; LPEI, linear poly(ethylenimine); PAH, pulmonary arterial hypertension; PEG-b-P[Asp(DET)], PEG-b-poly{N-[N-(2-aminoethyl)-2-aminoethyl]aspartamide}; RV, right ventricular.
Figure 8.
mRNA expression of human AM in the PAH rat model postintratracheal administration of PEG-b-P[Asp(DET)] polyplex nanomicelles loaded with the AM expression vector; AM expression vector in saline; PEG-b-P[Asp(DET)] polyplex nanomicelles loaded with the luciferase gene; or LPEI polyplexes loaded with the AM expression vector. Rats were transfected intratracheally at 4 weeks after subcutaneous monocrotaline injection. Three days later, lungs were harvested, homogenized, and measured for AM mRNA using real-time RT-PCR (mean ± SEM, N = 4). AM, adrenomedullin; LPEI, linear poly(ethylenimine); PAH, pulmonary arterial hypertension; PEG-b-P[Asp(DET)], PEG-b-poly{N-[N-(2-aminoethyl)-2-aminoethyl]aspartamide}.
Discussion
The large number of human diseases presenting poor prognoses and limited efficacy with current therapeutic regimens necessitates the advent of alternative approaches. PAH is such a disease without a highly efficacious therapeutic regimen.17 PAH patients are currently treated with a variety of drugs including prostacyclin, prostacyclin analogues, calcium channel blockers, nitric-oxide inhalation, angiotensin-converting enzyme inhibitors, endothelin receptor antagonists, and phosphodiesterase type 5 inhibitors; in severe cases lung transplantation and subsequent immunosuppression are necessary.17 However, promising alternative therapies for PAH have been recently reported. For example, Champion et al. reported that adenoviral gene transfer of endothelial nitric-oxide synthase to the lungs of endothelial nitric-oxide synthase knockout mice ameliorates the symptoms of PAH.18 Champion et al. also reported that adenoviral gene transfer of calcium gene-related peptide attenuates the symptoms of PAH.19 Nagaya et al. reported that transfection of human prostacyclin synthase using hemagglutinating virus of Japan–liposomes ameliorates monocrotaline-induced PAH.20 However, in these attempts, viral or viral-related vectors were used for the delivery of therapeutic genes and these gene carriers have the potential for immunogenicity and inflammatory response. In diseases where a single dose can cure or provide palliative care, viral vectors may be suitable; however, PAH therapy requires repeated administrations for efficacy, hence the utility of viral or viral-based gene therapy is contraindicated.
Alternatively, nonviral gene carriers have been recognized with several advantages over viral vectors in terms of safety, immunogenicity, and ease of manufacture. To develop a method of gene therapy suitable for clinical translation, four primary factors must be clearly addressed: (i) the gene carrier, (ii) the therapeutic gene, (iii) the route of administration, and (iv) patient compliance. In this study, we chose to further explore the promising (i) PEG-b-P[Asp(DET)] polyplex nanomicelle nonviral gene carrier system based upon our previous findings. Next, we chose (ii) AM as the therapeutic gene because of its reported effectiveness in the transient treatment of PAH. For the route of administration, we selected (iii) intratracheal administration to avoid the rapid propensity of nuclease degradation in the blood compartment and also that we might exploit the lung, based upon its enormous surface area, for use as a therapeutic bioreactor for AM production. Pulmonary administration is a promising therapeutic route of administration in the clinic for its (iv) high patient compliance with utilization of an inhaler or nebulizer.
Recently, we have demonstrated that PEG-b-P[Asp(DET)] polyplex nanomicelles achieved amplified in vitro and in vivo transfection activity with minimal cytotoxicity.14,15,16,21 With regard to the transfection mechanism, P[Asp(DET)] possesses the ethylenediamine side chain, which undergoes two-step protonation from the mono-protonated gauche form at physiological pH to di-protonated anti form at acidic pH, thereby exhibiting an effective buffering function in the acidic endosomal compartment.22 Also, we revealed the membrane destabilization effect of P[Asp(DET)] responding to acidic endosomal pH conditions by hemolysis, leakage of cytoplasmic enzyme (lactate dehydrogenase assay), and confocal laser scanning microscopic observation.22 Consequently, we observed the facilitated transport of Cy5-labeled plasmid DNA by the P[Asp(DET)] polyplexes from endo/lysosomal compartment into cytoplasm directly under the confocal laser scanning microscope in single cellular level.22 Therefore, the increased transgene expression in Figure 3 may be a result of the facilitated translocation of the polyplex nanomicelles from the endosome to the cytoplasm based on the buffering capacity (proton sponge effect) and/or endosomal membrane–destabilizing effect of P[Asp(DET)] segment. The reason why a relatively high N/P ratio was required for the efficient transfection (Figure 3) may be because the membrane-destabilizing effect of P[Asp(DET)] is dependent on the polymer concentration as previously reported. Nevertheless, the PEG-b-P[Asp(DET)] polyplex nanomicelles displayed minimal cytotoxicity even at a high N/P ratio, which may be due to the pH-sensitive properties of P[Asp(DET)] segment.22 The highly transfectable but less cytotoxic properties of PEG-b-P[Asp(DET)] polyplex nanomicelles motivated us to apply them to the gene therapy of PAH animal models through the intratracheal administration in this study.
A number of nonviral vectors including polyplex and lipoplex have been applied for in vivo intratracheal transfection. Special notice for the pulmonary gene delivery via airways is that the lung has the features critically influencing the transfection efficiency, such as the presence of surfactant, alveolar macrophages, and mucocilliary clearance mechanisms. In the early 1990s, lipoplex was used by aerosol delivery or intratracheal instillation. However, cationic lipids were shown to have a decreased transfection efficiency due to the interaction with lung surfactant compared to cationic polymer like PEI.23,24 To overcome the surfactant barriers, cationic emulsion was used and showed much higher transfection activity compared with lipoplexes, such as lipofectin, lipofectamine, and DMRIE/c.25 However, even for the cationic emulsion, the luciferase activity was limited to 55 pg/mg protein, which is significantly lower than the value attained by the polyplex nanomicelles loaded with PEG-b-P[Asp(DET)] [3,000,000 relative light unit/mg protein (135 ng/mg protein)] as reported here. Polyplexes made from cationic polymer was reported to show higher transfection efficiency compared with cationic lipoplexes for pulmonary gene delivery via airways.23,24 PEI or modified PEI has been shown to be one of the most effective agents for constructing gene delivery systems available today with high levels of pulmonary gene transfer by airways.26,27 Intratracheal injection of polyplex loaded with 22 kd of LPEI (ExGen 500) showed up to 20,000–40,000 relative light unit/mg protein of luciferase activity in the lung by adjusting N/P ratio. Worth noting is that the nanomicelles achieved two orders of magnitude higher value in luciferase gene expression compared to the LPEI polyplex. Furthermore, no induction of cytokine responses is appealing for the nanomicelles over LPEI polyplex (Figure 6), which was reported to induce the activation of CD8+ and CD4+ T cells, and Fas ligand–mediated antigen-induced cell death.28,29
To ameliorate the symptoms of PAH in animal models, several genes have been identified including: endothelial nitric-oxide synthase, inducible nitric-oxide synthase, prostacyclin synthase, calcium gene-related peptide, vascular endothelial growth factor, and hepatocyte growth factor.18,19,20,30,31,32,33,34,35,36 We used AM as a therapeutic gene because of its high potency and long-term effectiveness as a vasodilator in the pulmonary vascular bed.37 The effect of pulmonary vasodilation is mediated by cyclic adenosine monophosphate–dependent and nitric oxide–dependent mechanisms.38 PAH patients have elevated plasma AM concentrations, which increase in step with the disease's severity39,40 often resulting in pulmonary hypotension. In previous studies, intravenous administration41 and inhalation8 of the AM peptide showed acute hemodynamic and hormonal efficacy in PAH patients. However, despite alternative routes of delivery, the small AM peptide was rapidly degraded in vivo displaying a poor pharmacokinetic profile with a temporal window of only 30–45 minutes. For the treatment of PAH, sustained effect of AM is required. In this report, the therapeutic indicator for successful nonviral AM delivery was a decrease in the right ventricular pressure. Indeed, for the polyplex nanomicelle/pAM formulation, the right ventricular pressure did decrease, but more importantly the persistence of AM gene expression continued within a therapeutic range for a minimum of 3 days. The results indicate that the therapeutic approach using the polyplex nanomicelle as a vector does not require chronic infusion or very frequent inhalation, which will make the therapy more clinically applicable.
In this study, we succeeded in delivering DNA to the lung via intratracheal administration using the polyplex nanomicelles, resulting in extremely improved transfection efficiency with concomitant high biocompatibility. More specifically, the lung transfected with the polyplex nanomicelle had much lower toxicity than that transfected with the LPEI polyplex, according to the histological findings and measurement of the mRNA levels of inflammatory cytokines (Figure 6a–d). We also developed an effective treatment for the PAH rat model by delivering the therapeutic AM gene with polyplex nanomicelles from PEG-b-P[Asp(DET)]. These results showed a significant increase in transfection efficiently in vivo with intratracheal administration. The PEG-b-P[Asp(DET)] polyplex nanomicelle delivery system clearly showed promising in vivo results of transgene and therapeutic AM expression, when coupled with the clear visual, localization of the polyplex nanomicelles in the pulmonary tissue and the lack of proinflammatory responses. We posit that the PEG-b-P[Asp(DET)] nonviral gene carrier clearly shows those characteristics requisite for novel and advanced therapeutic systems ideally suited for translational research.
Materials and Methods
Materials. An expression vector for YFP (RIKEN, Tokyo, Japan) was amplified in competent HB101 Escherichia coli and purified by Plasmid Giga Kits (Qiagen, Hilden, Germany). An expression vector for luciferase with a CAG promoter was provided by RIKEN. An expression vector for human AM was constructed as follows. The EcoRI/XhoI fragment of the full-length human AM complementary DNA (cDNA)42 was ligated into the EcoRI/XhoI site of pcDNA1 (Invitrogen, Carlsbad, CA). The restriction maps of expression vector for luciferase and human AM cDNA are listed in Supplementary Figure S1a,b, respectively. To confirm that pcDNA/AM encodes AM, pcDNA/AM was transfected into Chinese hamster ovary cells, and the medium and the cells were collected for the measurement of immunoreactive AM using an AM radioimmunoassay Shionogi (Cosmic, Tokyo, Japan).
Animals. Male ICR mice weighing 25–30 g were administered the reporter gene intratracheally. Male Wistar rats weighing 100–120 g were used to make a model of PAH. All protocols were performed in accordance with the guidelines of the Animal Care Ethics Committee of the National Cardiovascular Center Research Institute (Osaka, Japan).
Synthesis and characterization of PEG-b-P[Asp(DET)]. PEG-b-P[Asp(DET)] was prepared as previously described.14 Briefly, PEG-poly(β-benzyl-L-aspartate) (PEG-PBLA) diblock copolymer was synthesized by the ring-opening polymerization of β-benzyl-L-aspartate N-carboxy-anhydride from the terminal primary amino group of α-methoxy-ω-amino PEG (Mw: 12,000; Nippon Oil and Fats, Tokyo, Japan). Gel-permeation chromatography confirmed that the copolymer was unimodal with a narrow molecular weight distribution (Mw/Mn: 1.23), and the number of benzyl-L-aspartate repeating units was calculated to be 68 by 1H-NMR. The N-terminal amino group of PEG-PBLA was then acetylated using acetic anhydride in dichloromethane solution to obtain PEG-PBLA-Ac. The obtained polymer was dissolved in distilled N,N-dimethylformamide (Wako Pure Chemical Industries, Osaka, Japan) and reacted with diethylenetriamine (Tokyo Kasei Kogyo, Tokyo, Japan) for 24 hours at 40 °C in a dry argon atmosphere to undergo aminolysis of the benzyl side chain. After 24 hours, the solution was slowly dripped into a 10% acetic acid solution and dialyzed (Spectra/Por Membrane, 3,500 molecular weight cutoff; Spectrum Laboratories, Rancho Dominguez, CA) against 0.01 N HCl and subsequently against distilled water. The final solution was lyophilized to obtain PEG-b-P[Asp(DET)] as the hydrochloride salt form. 1H-NMR confirmed the complete substitution of benzyl ester of the polymer with diethylenetriamine through the aminolysis reaction, as well as the chemical structure of the obtained PEG-b-P[Asp(DET)] block copolymer.
Preparation of polyplex nanomicelles. The PEG-b-P[Asp(DET)] block copolymer and plasmid DNA were separately dissolved in 10 mmol/l HEPES buffer (pH 7.4). Both solutions were mixed at the indicated nitrogen/phosphate ratios [=(total amines in cationic segment)/(phosphates in plasmid DNA)] and incubated overnight at room temperature to make PEG-b-P[Asp-(DET)] polyplex nanomicelle. LPEI (ExGen; Cosmo Bio, Tokyo, Japan) polyplexes were prepared by mixing plasmid DNA and LPEI according to the manufacturer's protocol.
In vivo gene delivery by intratracheal administration. ICR mice were anesthetized by intraperitoneal administration of pentobarbital (30 mg/kg) (Dainippon Sumitomo Pharma, Osaka, Japan). Tracheostomies were performed under sterile conditions for PEG-b-P[Asp-(DET)] polyplex nanomicelle or LPEI polyplex (10 µg of DNA for each mouse) in a 50 µl of solution administration by a microsprayer Model IA-1C (Penn Century, Philadelphia, PA). After the indicated time, the mice were killed by cervical dislocation and the pulmonary tissues harvested. To measure luciferase activity, the pulmonary tissues were homogenized in a lysis buffer using a polytron. The lysate was then centrifuged at 14,000g for 10 minutes at 4 °C, and 20 µl of the supernatant was analyzed for luciferase activity by a Luminous CT-9000D luminometer (Dia-Iatron, Tokyo, Japan), according to a previously described method.43 Background of luciferase activity in the lung was measured from the lung of mice after administration of saline, which was <3% of the total activity of day 14. All the data of luciferase activity were obtained by subtraction of background data. To detect YFP expression, mice were killed by cervical dislocation and the lungs harvested. Frozen sections (5-µm thick) of the lung specimens were visualized by a fluorescence microscope (SZX12; Olympus, Tokyo, Japan). To examine the histological features of the lung tissue, the specimens were also fixed in 4% paraformaldehyde and embedded in paraffin. Sections (3-µm thick) were stained with hematoxylin.
Isolation of RNA and cDNA synthesis. Total RNA was extracted using the Trizol method (Gibco BRL Life Technologies, Breda, Netherlands) according to the protocol provided by the manufacturer. The RNA was dissolved in RNAse-free water and quantified by a spectrophotometer. The cDNA was synthesized using the High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA).
Real-time RT-PCR. mRNA expression levels of TNF-α, IL-6, IL-10, Cox-2 and human AM were measured by quantitative real-time RT-PCR based on TaqMan chemistry (Applied Biosystems) using an ABI PRISM 7700 sequence detector (Applied Biosystems). The reaction mixture contained 0.5 µl of 5 µmol/l probe (final concentration, 100 nmol/l); 1 µl of 10 µmol/l forward primer and 1 µl of 10 µmol/l reverse primer (400 nmol/l final concentration of each primer); 12.5 µl of TaqMan Universal Mastermix, 5 µl of diethyl pyrocarbonate–treated water, and 5 µl of a cDNA sample. Assay controls were performed in the same TaqMan plate with no-template controls to test for the contamination of any assay reagents. The thermocycling conditions were initiated at 50 °C for 2 minutes with an enzyme activation step of 95 °C for 10 minutes followed by 40 PCR cycles of denaturation at 95 °C for 15 seconds, and anneal/extension at 60 °C for 1 minute.
Hemodynamic studies. Hemodynamic studies were performed 4 weeks after gene transfer. Rats were anesthetized with intraperitoneal pentobarbital (30 mg/kg) and placed on a heating pad to maintain body temperature 37–38 °C throughout the study. Under sterile conditions, a polyethylene catheter (PE-50; BD Biosciences, San Jose, CA) was inserted through the right jugular vein into the right ventricle to measure right ventricular pressure by a hemodynamic transducer (PowerLab 8/30; ADInstruments, Colorado Springs, CO).
Evaluation of gene transfer effect in a PAH rat model. Monocrotaline (60 mg/kg) was subcutaneously injected into male Wistar rats and left for 4 weeks to make a model of PAH. After 4 weeks, a hemodynamic study was performed to introduce a catheter into the right ventricle through the right jugular vein. The PEG-b-P[Asp(DET)] polyplex nanomicelle loaded with the AM expression vector (200 µg of DNA for each rat) in a 200 µl of solution was sprayed intratracheally. Three days later, a hemodynamic study was performed again and the gene transfer effect was evaluated. Pulmonary tissue specimens were frozen to measure AM gene expression by real-time RT-PCR.
Statistical analysis. All data are expressed as means ± SEM unless otherwise indicated. Comparisons of parameters among four groups were made by one-way analysis of variance, followed by Scheffe's multiple-comparison test. Paired t-test was applied for the comparison of the values before and after the gene transfection (Figure 7).
SUPPLEMENTARY MATERIALFigure S1. The restriction maps of expression vector for luciferase and human adrenomedullin cDNA.
Supplementary Material
The restriction maps of expression vector for luciferase and human adrenomedullin cDNA.
Acknowledgments
This work was supported by the Core Research Program for Evolutional Science and Technology from the Japan Science and Technology Corporation, by Grants-in-Aid for Scientific Research from the Japanese Ministry of Health, Labor, and Welfare (H19-Nano-012 and H20-Genomu-Ippan-008), by the Program for the Promotion of Fundamental Studies in Health Sciences of the National Institute of Biomedical Innovation of Japan, and by the Takeda Science Foundation. We thank Keiko Jinno, Shoko Obora, Hiroko Miyata, Moto Ohira, and Eri Abe and Mutsumi Goda (National Cardiovascular Center Research Institute) for their excellent technical assistance, including animal care. We also thank Hisayuki Matsuo and Hitonobu Tomoike for their helpful discussion and advice, Darin Y. Furgeson (University of Wisconsin–Madison) for proofreading of this manuscript.
References
- Rubin LJ. Pulmonary arterial hypertension. Proc Am Thorac Soc. 2006;3:111–115. doi: 10.1513/pats.200510-112JH. [DOI] [PubMed] [Google Scholar]
- Badesch DB, Abman SH, Ahearn GS, Barst RJ, McCrory DC, Simonneau G, et al. Medical therapy for pulmonary arterial hypertension: ACCP evidence-based clinical practice guidelines Chest 200412635S–62S.1 Suppl [DOI] [PubMed] [Google Scholar]
- D'Alonzo GE, Barst RJ, Ayres SM, Bergofsky EH, Brundage BH, Detre KM, et al. Survival in patients with primary pulmonary hypertension. Results from a national prospective registry. Ann Intern Med. 1991;115:343–349. doi: 10.7326/0003-4819-115-5-343. [DOI] [PubMed] [Google Scholar]
- Kitamura K, Kangawa K, Kawamoto M, Ichiki Y, Nakamura S, Matsuo H, et al. Adrenomedullin: a novel hypotensive peptide isolated from human pheochromocytoma. Biochem Biophys Res Commun. 1993;192:553–560. doi: 10.1006/bbrc.1993.1451. [DOI] [PubMed] [Google Scholar]
- Kitamura K, Kangawa K., and , Eto T. Adrenomedullin and PAMP: discovery, structures, and cardiovascular functions. Microsc Res Tech. 2002;57:3–13. doi: 10.1002/jemt.10052. [DOI] [PubMed] [Google Scholar]
- Nagaya N, Mori H, Murakami S, Kangawa K., and , Kitamura S. Adrenomedullin: angiogenesis and gene therapy. Am J Physiol Regul Integr Comp Physiol. 2005;288:R1432–R1437. doi: 10.1152/ajpregu.00662.2004. [DOI] [PubMed] [Google Scholar]
- Nagaya N, Okumura H, Uematsu M, Shimizu W, Ono F, Shirai M, et al. Repeated inhalation of adrenomedullin ameliorates pulmonary hypertension and survival in monocrotaline rats. Am J Physiol Heart Circ Physiol. 2003;285:H2125–H2131. doi: 10.1152/ajpheart.00548.2002. [DOI] [PubMed] [Google Scholar]
- Nagaya N, Kyotani S, Uematsu M, Ueno K, Oya H, Nakanishi N, et al. Effects of adrenomedullin inhalation on hemodynamics and exercise capacity in patients with idiopathic pulmonary arterial hypertension. Circulation. 2004;109:351–356. doi: 10.1161/01.CIR.0000109493.05849.14. [DOI] [PubMed] [Google Scholar]
- Katayose S., and , Kataoka K. Water-soluble polyion complex associates of DNA and poly(ethylene glycol)-poly(L-lysine) block copolymer. Bioconjug Chem. 1997;8:702–707. doi: 10.1021/bc9701306. [DOI] [PubMed] [Google Scholar]
- Kakizawa Y., and , Kataoka K. Block copolymer micelles for delivery of gene and related compounds. Adv Drug Deliv Rev. 2002;54:203–222. doi: 10.1016/s0169-409x(02)00017-0. [DOI] [PubMed] [Google Scholar]
- Katayose S., and , Kataoka K. Remarkable increase in nuclease resistance of plasmid DNA through supramolecular assembly with poly(ethylene glycol)-poly(L-lysine) block copolymer. J Pharm Sci. 1998;87:160–163. doi: 10.1021/js970304s. [DOI] [PubMed] [Google Scholar]
- Itaka K, Yamauchi K, Harada A, Nakamura K, Kawaguchi H., and , Kataoka K. Polyion complex micelles from plasmid DNA and poly(ethylene glycol)-poly(L-lysine) block copolymer as serum-tolerable polyplex system: physicochemical properties of micelles relevant to gene transfection efficiency. Biomaterials. 2003;24:4495–4506. doi: 10.1016/s0142-9612(03)00347-8. [DOI] [PubMed] [Google Scholar]
- Harada-Shiba M, Yamauchi K, Harada A, Takamisawa I, Shimokado K., and , Kataoka K. Polyion complex micelles as vectors in gene therapy—pharmacokinetics and in vivo gene transfer. Gene Ther. 2002;9:407–414. doi: 10.1038/sj.gt.3301665. [DOI] [PubMed] [Google Scholar]
- Kanayama N, Fukushima S, Nishiyama N, Itaka K, Jang WD, Miyata K, et al. A PEG-based biocompatible block catiomer with high buffering capacity for the construction of polyplex micelles showing efficient gene transfer toward primary cells. ChemMedChem. 2006;1:439–444. doi: 10.1002/cmdc.200600008. [DOI] [PubMed] [Google Scholar]
- Itaka K, Ohba S, Miyata K, Kawaguchi H, Nakamura K, Takato T, et al. Bone regeneration by regulated in vivo gene transfer using biocompatible polyplex nanomicelles. Mol Ther. 2007;15:1655–1662. doi: 10.1038/sj.mt.6300218. [DOI] [PubMed] [Google Scholar]
- Akagi D, Oba M, Koyama H, Nishiyama N, Fukushima S, Miyata T, et al. Biocompatible micellar nanovectors achieve efficient gene transfer to vascular lesions without cytotoxicity and thrombus formation. Gene Ther. 2007;14:1029–1038. doi: 10.1038/sj.gt.3302945. [DOI] [PubMed] [Google Scholar]
- Nossaman BD, Gur S., and , Kadowitz PJ. Gene and stem cell therapy in the treatment of erectile dysfunction and pulmonary hypertension; potential treatments for the common problem of endothelial dysfunction. Curr Gene Ther. 2007;7:131–153. doi: 10.2174/156652307780363161. [DOI] [PubMed] [Google Scholar]
- Champion HC, Bivalacqua TJ, Greenberg SS, Giles TD, Hyman AL, Kadowitz PJ. Adenoviral gene transfer of endothelial nitric-oxide synthase (eNOS) partially restores normal pulmonary arterial pressure in eNOS-deficient mice. Proc Natl Acad Sci USA. 2002;99:13248–13253. doi: 10.1073/pnas.182225899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Champion HC, Bivalacqua TJ, Toyoda K, Heistad DD, Hyman AL., and , Kadowitz PJ. In vivo gene transfer of prepro-calcitonin gene-related peptide to the lung attenuates chronic hypoxia-induced pulmonary hypertension in the mouse. Circulation. 2000;101:923–930. doi: 10.1161/01.cir.101.8.923. [DOI] [PubMed] [Google Scholar]
- Nagaya N, Yokoyama C, Kyotani S, Shimonishi M, Morishita R, Uematsu M, et al. Gene transfer of human prostacyclin synthase ameliorates monocrotaline-induced pulmonary hypertension in rats. Circulation. 2000;102:2005–2010. doi: 10.1161/01.cir.102.16.2005. [DOI] [PubMed] [Google Scholar]
- Masago K, Itaka K, Nishiyama N, Chung UI., and , Kataoka K. Gene delivery with biocompatible cationic polymer: Pharmacogenomic analysis on cell bioactivity. Biomaterials. 2007;28:5169–5175. doi: 10.1016/j.biomaterials.2007.07.019. [DOI] [PubMed] [Google Scholar]
- Miyata K, Oba M, Nakanishi M, Fukushima S, Yamasaki Y, Koyama H, et al. Polyplexes from poly(aspartamide) bearing 1,2-diaminoethane side chains induce pH-selective, endosomal membrane destabilization with amplified transfection and negligible cytotoxicity. J Am Chem Soc. 2008;130:16287–16294. doi: 10.1021/ja804561g. [DOI] [PubMed] [Google Scholar]
- Bragonzi A, Dina G, Villa A, Calori G, Biffi A, Bordignon C, et al. Biodistribution and transgene expression with nonviral cationic vector/DNA complexes in the lungs. Gene Ther. 2000;7:1753–1760. doi: 10.1038/sj.gt.3301282. [DOI] [PubMed] [Google Scholar]
- Wiseman JW, Goddard CA, McLelland D., and , Colledge WH. A comparison of linear and branched polyethylenimine (PEI) with DCChol/DOPE liposomes for gene delivery to epithelial cells in vitro and in vivo. Gene Ther. 2003;10:1654–1662. doi: 10.1038/sj.gt.3302050. [DOI] [PubMed] [Google Scholar]
- Kim TW, Chung H, Kwon IC, Sung HC, Shin BC., and , Jeong SY. Airway gene transfer using cationic emulsion as a mucosal gene carrier. J Gene Med. 2005;7:749–758. doi: 10.1002/jgm.711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Densmore CL. Advances in noninvasive pulmonary gene therapy. Curr Drug Deliv. 2006;3:55–63. doi: 10.2174/156720106775197547. [DOI] [PubMed] [Google Scholar]
- Furgeson DY, Chan WS, Yockman JW., and , Kim SW. Modified linear polyethylenimine-cholesterol conjugates for DNA complexation. Bioconjug Chem. 2003;14:840–847. doi: 10.1021/bc0340565. [DOI] [PubMed] [Google Scholar]
- Gautam A, Densmore CL., and , Waldrep JC. Pulmonary cytokine responses associated with PEI-DNA aerosol gene therapy. Gene Ther. 2001;8:254–257. doi: 10.1038/sj.gt.3301369. [DOI] [PubMed] [Google Scholar]
- Regnstrom K, Ragnarsson EG, Koping-Hoggard M, Torstensson E, Nyblom H., and , Artursson P. PEI—a potent, but not harmless, mucosal immuno-stimulator of mixed T-helper cell response and FasL-mediated cell death in mice. Gene Ther. 2003;10:1575–1583. doi: 10.1038/sj.gt.3302054. [DOI] [PubMed] [Google Scholar]
- Budts W, Pokreisz P, Nong Z, Van Pelt N, Gillijns H, Gerard R, et al. Aerosol gene transfer with inducible nitric oxide synthase reduces hypoxic pulmonary hypertension and pulmonary vascular remodeling in rats. Circulation. 2000;102:2880–2885. doi: 10.1161/01.cir.102.23.2880. [DOI] [PubMed] [Google Scholar]
- Chicoine LG, Tzeng E, Bryan R, Saenz S, Paffett ML, Jones J, et al. Intratracheal adenoviral-mediated delivery of iNOS decreases pulmonary vasoconstrictor responses in rats. J Appl Physiol. 2004;97:1814–1822. doi: 10.1152/japplphysiol.00193.2004. [DOI] [PubMed] [Google Scholar]
- Campbell AI, Zhao Y, Sandhu R., and , Stewart DJ. Cell-based gene transfer of vascular endothelial growth factor attenuates monocrotaline-induced pulmonary hypertension. Circulation. 2001;104:2242–2248. doi: 10.1161/hc4201.097838. [DOI] [PubMed] [Google Scholar]
- Gong F, Tang H, Lin Y, Gu W, Wang W., and , Kang M. Gene transfer of vascular endothelial growth factor reduces bleomycin-induced pulmonary hypertension in immature rabbits. Pediatr Int. 2005;47:242–247. doi: 10.1111/j.1442-200x.2005.02060.x. [DOI] [PubMed] [Google Scholar]
- Suhara H, Sawa Y, Fukushima N, Kagisaki K, Yokoyama C, Tanabe T, et al. Gene transfer of human prostacyclin synthase into the liver is effective for the treatment of pulmonary hypertension in rats. J Thorac Cardiovasc Surg. 2002;123:855–861. doi: 10.1067/mtc.2002.118687. [DOI] [PubMed] [Google Scholar]
- Ono M, Sawa Y, Fukushima N, Suhara H, Nakamura T, Yokoyama C, et al. Gene transfer of hepatocyte growth factor with prostacyclin synthase in severe pulmonary hypertension of rats. Eur J Cardiothorac Surg. 2004;26:1092–1097. doi: 10.1016/j.ejcts.2004.08.031. [DOI] [PubMed] [Google Scholar]
- Ono M, Sawa Y, Mizuno S, Fukushima N, Ichikawa H, Bessho K, et al. Hepatocyte growth factor suppresses vascular medial hyperplasia and matrix accumulation in advanced pulmonary hypertension of rats. Circulation. 2004;110:2896–2902. doi: 10.1161/01.CIR.0000146342.30470.30. [DOI] [PubMed] [Google Scholar]
- Lippton H, Chang JK, Hao Q, Summer W., and , Hyman AL. Adrenomedullin dilates the pulmonary vascular bed in vivo. J Appl Physiol. 1994;76:2154–2156. doi: 10.1152/jappl.1994.76.5.2154. [DOI] [PubMed] [Google Scholar]
- Ishizaka Y, Ishizaka Y, Tanaka M, Kitamura K, Kangawa K, Minamino N, et al. Adrenomedullin stimulates cyclic AMP formation in rat vascular smooth muscle cells. Biochem Biophys Res Commun. 1994;200:642–646. doi: 10.1006/bbrc.1994.1496. [DOI] [PubMed] [Google Scholar]
- Kakishita M, Nishikimi T, Okano Y, Satoh T, Kyotani S, Nagaya N, et al. Increased plasma levels of adrenomedullin in patients with pulmonary hypertension. Clin Sci (Lond) 1999;96:33–39. [PubMed] [Google Scholar]
- Yoshibayashi M, Kamiya T, Kitamura K, Saito Y, Kangawa K, Nishikimi T, et al. Plasma levels of adrenomedullin in primary and secondary pulmonary hypertension in patients <20 years of age. Am J Cardiol. 1997;79:1556–1558. doi: 10.1016/s0002-9149(97)00195-1. [DOI] [PubMed] [Google Scholar]
- Nagaya N, Nishikimi T, Uematsu M, Satoh T, Oya H, Kyotani S, et al. Haemodynamic and hormonal effects of adrenomedullin in patients with pulmonary hypertension. Heart. 2000;84:653–658. doi: 10.1136/heart.84.6.653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kitamura K, Sakata J, Kangawa K, Kojima M, Matsuo H., and , Eto T. Cloning and characterization of cDNA encoding a precursor for human adrenomedullin. Biochem Biophys Res Commun. 1993;194:720–725. doi: 10.1006/bbrc.1993.1881. [DOI] [PubMed] [Google Scholar]
- de Wet JR, Wood KV, DeLuca M, Helinski DR., and , Subramani S. Firefly luciferase gene: structure and expression in mammalian cells. Mol Cell Biol. 1987;7:725–737. doi: 10.1128/mcb.7.2.725. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The restriction maps of expression vector for luciferase and human adrenomedullin cDNA.