Abstract
How embryos consistently orient asymmetries of the left-right (LR) axis is an intriguing question, as no macroscopic environmental cues reliably distinguish left from right. Especially unclear are the events coordinating LR patterning with the establishment of the dorsoventral (DV) axes and midline determination in early embryos. In frog embryos, consistent physiological and molecular asymmetries manifest by the second cell cleavage; however, models based on extracellular fluid flow at the node predict correct de novo asymmetry orientation during neurulation. We addressed these issues in Xenopus embryos by manipulating the timing and location of dorsal organizer induction: the primary dorsal organizer was ablated by UV irradiation, and a new organizer was induced at various locations, either early, by mechanical rotation, or late, by injection of lithium chloride (at 32 cells) or of the transcription factor XSiamois (which functions after mid-blastula transition). These embryos were then analyzed for the position of three asymmetric organs. Whereas organizers rescued before cleavage properly oriented the LR axis 90% of the time, organizers induced in any position at any time after the 32-cell stage exhibited randomized laterality. Late organizers were unable to correctly orient the LR axis even when placed back in their endogenous location. Strikingly, conjoined twins produced by late induction of ectopic organizers did have normal asymmetry. These data reveal that although correct LR orientation must occur no later than early cleavage stages in singleton embryos, a novel instructive influence from an early organizer can impose normal asymmetry upon late organizers in the same cell field.
Keywords: Left-right asymmetry, Midline, Conjoined twins, Heterotaxia, Organizer, Siamois, Xenopus
INTRODUCTION
During development, embryos undergo an exquisitely complex biophysical program to acquire specific patterns along each of three orthogonal axes. Despite an overtly bilateral-symmetric body-plan, all vertebrates (and many invertebrates) exhibit a striking left-right (LR) asymmetry of the heart, viscera and brain that is conserved through evolution, and in all normal individuals. Abnormalities in the proper development of laterality form a class of human birth defects with significant implications for the health of the individual (Casey, 1998; Casey and Hackett, 2000). These defects occur in more than 1 in 8000 live births and often have serious medical consequences for the individual (Peeters and Devriendt, 2006; Ramsdell, 2005).
Prior work identified asymmetrically expressed genes that function together in an inductive/repressive cascade to regulate downstream morphogenesis of asymmetric organs (Levin et al., 1995; Speder et al., 2007). However, the most important and controversial questions in the LR asymmetry field concern mechanisms that set up the sidedness of the first asymmetric genes (Levin and Palmer, 2007; Tabin, 2005). One popular model proposes that asymmetry is initiated by the motion of cilia during gastrulation (McGrath and Brueckner, 2003; Tabin and Vogan, 2003; Wagner and Yost, 2000). These flow-based models are driven largely by studies in the mouse and zebrafish (Kramer-Zucker et al., 2005; Nonaka et al., 2002; Otto et al., 2003), where no consistent LR asymmetries have been reported prior to movement of cilia in the node/Kupffer's vesicle.
However, in several vertebrate and invertebrate systems (Arabidopsis, Drosophila, snail, sea urchin, Ciona, Caenorhabditis elegans and frog), consistent asymmetry is set at very early stages, long before the appearance of, or entirely without, motile cilia (Levin, 2006). While the degree of evolutionary conservation is far from clear (Levin, 2006; Okumura et al., 2008; Schlueter and Brand, 2007), these mechanisms have been worked out in the most detail in Xenopus embryos. The development of normal LR asymmetry in Xenopus embryos requires four specific ion transporters: two H+ pumps, V-ATPase (Adams et al., 2006) and H,K-ATPase (Levin et al., 2002), and two K+ channels, KvLQT-1 (Morokuma et al., 2008) and Katp (Chen and Levin, 2004). Each transporter is asymmetrically localized during the first three cleavages in a process that is dependent on a chirally oriented cytoskeletal organization (Aw et al., 2008; Danilchik et al., 2006). The asymmetric ion transporter localization in left and right blastomeres results in consistently biased differences in pH and membrane-voltage potential of cells on the left and right sides, which become transduced into differential gene expression via Ca2+- and serotonin-dependent mechanisms (Levin et al., 2006; Raya and Belmonte, 2006). Molecular-genetic equalization of these physiological asymmetries results in LR randomization of the asymmetric gene cascade and ultimately heterotaxia involving the heart and viscera.
The frog embryo data reveal candidate mechanisms (Aw and Levin, 2009) for the initial symmetry-breaking event (e.g. the chiral structures of a microtubule organizing center), as well as for amplifying intracellular asymmetries into organ laterality (asymmetric intracellular transport of bioelectric components that ultimately control asymmetric expression of the Nodal-Lefty-Pitx cassette). However, this model capitalizes on the fact that, in the context of the few large early blastomeres that cleave along the midline of the animal, asymmetric localization of ion transporter molecules across the first cleavage planes can be driven by intracellular motor protein transport, resulting in different bioelectric properties on the left and right sides (Levin and Nascone, 1997; Levin and Palmer, 2007). How would it apply to species in which the midline (and thus LR asymmetry) is not determined until much later?
Although it has been argued that even amniotes may set up axes far earlier than the disk/cylinder stages (Aw and Levin, 2008; Gardner, 2001; Plusa et al., 2002), it is generally believed that in birds and mammals asymmetry cannot rely on a Xenopus-like system. No individual cell can redistribute components across the midline of the whole embryo in a blastoderm that contains many thousands of small cells at the time when the primary axis is established; none of these small cells can set up macroscopic asymmetry by intracellular transport. Ciliary mechanisms are consistent with such a system because they do not require large blastomeres or intracellular transport; although cilia are important for asymmetry in frog embryos (Schweickert et al., 2007), it is not known whether they are a late amplification step or whether they can initiate asymmetry de novo.
Xenopus is a unique system in which to evaluate the relative importance of early versus late symmetry breaking events because it is the only system in which both cilia and early physiological mechanisms are known to operate, offering the opportunity to dissect the relationship between them. The ciliary model predicts that organizers induced after early cleavage stages (bypassing early cytoskeleton-dependent events) will give rise to embryos with normal asymmetry, since ciliary events should occur normally during neurulation. By contrast, the cytoplasmic model predicts that late-induced organizers, which did not have the benefit of early cleavage-stage mechanisms, will give rise to embryos that exhibit LR randomization.
In one important study, conjoined twins were generated by injection of the organizer-inducing transcription factor XSiamois (Nascone and Mercola, 1997). Twins are induced by this method by ectopic organizers forming only after zygotic transcription is initiated at the mid-blastula transition. When located on the left, such twins had normal asymmetry. This was widely interpreted as showing that, even in frog, late organizers could dispense with early intracellular events and initiate correct asymmetry. Such a mechanism would be of wide relevance to other types of embryos.
To understand the true relevance of the processes that occur during the first cleavages, we developed an assay in which the ability of late-induced organizers to direct normal LR patterning could be analyzed in singleton embryos, without the confounding effects of conjoined twinning and cross-twin leakage of asymmetric gene products. In this assay, the endogenous axial patterning information was ablated before organizer signaling was ‘rescued’ in a controlled manner. Previous studies demonstrated that ultraviolet (UV) irradiation of one-cell-stage embryos blocks microtubule-dependent cortical rotation (Houliston, 1994; Scharf and Gerhart, 1983), preventing the formation of Spemann's organizer (Brannon and Kimelman, 1996; Medina et al., 1997) and resulting in ‘belly piece’ embryos lacking dorsoventral (DV) axes (Bouwmeester et al., 1996; Cooke and Smith, 1987).
There are at least three methods whereby the DV axis can be rescued and restored in embryos that are ‘unpatterned’ by UV irradiation. First, the embryos can be mechanically rotated (tipped) 90 degrees during the one-cell stage (Scharf and Gerhart, 1980). When tipped, gravity can fully substitute for the cortical rotation that would typically occur during the first cell cycle, leading to the normal development of Spemann's organizer. Second, UV-irradiated embryos can be injected with XSiamois at the 16-cell stage (Fan and Sokol, 1997). The presence of XSiamois is sufficient to induce a normal DV axis; crucially, because it is a transcription factor, its effects will not occur until the mid-blastula transition, enabling characterization of late organizers. Finally, irradiated embryos can be rescued by injecting lithium chloride (LiCl) at the 32-cell stage. Unlike soaking embryos in LiCl, which causes dorsalization of the entire embryo (Kao and Elinson, 1998), injection of LiCl into a third tier cell dorsalizes the response of just that cell (and its daughter cells) to mesoderm induction signals (Kao and Elinson, 1989). Because the organizer establishes the position of the DV axis of the embryo, and the LR axis must be defined with respect to the anteroposterior (AP) and DV axes, it becomes crucial to understand whether (and when) an induced dorsal organizer can direct consistent LR asymmetry.
In normal embryos, the plane of early cleavages coincides with the bisection that an organizer must perform when initiating the primary embryonic axis. Using the above methods, we sought to dissociate the patterning events occurring during early cleavage stages from functions of the organizer. Our experiments addressed two questions. (1) Do embryos re-patterned by moving the primary organizer have normal LR asymmetry? And (2), does the timing of the re-patterning event affect the ability to properly orient the LR axis? The data revealed that although the original observation of late-induced conjoined twins having normal asymmetry is correct, this is due to a novel and unexpected instructive effect from the primary twin (which acquired its LR information at early stages). Indeed, in singleton embryos, organizers induced at or after the 32-cell stage cannot normally orient asymmetry, which is consistent with a key importance of very early events in laterality, and argues against the primacy of events occurring at the node during gastrulation.
MATERIALS AND METHODS
Animal rearing
Xenopus laevis embryos were collected and fertilized in vitro according to standard protocols (Sive et al., 2000), in 0.1×modified Marc's Ringers (MMR; pH 7.8) with 0.1% Gentamicin. Xenopus embryos were housed at 14-18°C and staged according to Nieuwkoop and Faber (Nieuwkoop and Faber, 1967). All experiments were approved by the Tufts University Animal Research Committee in accordance with the Guide for Care and Use of Laboratory Animals.
Twin induction
For microinjection of XSiamois, 5 pg capped synthetic mRNAs (Sive et al., 2000) were dissolved in water with a lineage tracer and injected into a single ventral vegetal blastomere of 16-cell embryos in 3% ficoll. At stage 22, twin embryos were sorted for left- and right-side induced twins using fluorescence. A total of 60 left-side induced and 58 right-side induced twins were examined for organ situs. An additional 44 left-side induced and 41 right-side induced twins were examined for Xnr-1 mRNA localization by in situ hybridization.
Laterality assay
At stage 45, Xenopus embryos were analyzed for position (situs) of the heart, stomach and gall bladder (Levin and Mercola, 1998b). Heterotaxia was defined as the reversal in position of one or more organs. Only embryos with a normal dorsoanterior index (DAI=5) were scored to prevent confounding of results by randomization caused by midline defects (Danos and Yost, 1995). Percentage heterotaxia was calculated as number of heterotaxic embryos divided by the total number of scorable embryos. A Pearson χ2 test with Yates correction for increased stringency was used to compare absolute counts of heterotaxic embryos.
In situ hybridization
Whole-mount in situ hybridization was performed using standard protocols (Harland, 1991). In situ hybridization probes against XNR-1 (Sampath et al., 1997) and Chordin (Sasai et al., 1994) mRNAs were generated in vitro from linearized templates using DIG-labeling mix (Invitrogen, Carlsbad, CA, USA).
Cell lineage tracer
In control four-cell embryos, both right or both ventral blastomeres were injected with mRNA encoding β-galactosidase (β-gal) based on patterns of cell division and pigment that revealed the darker prospective ventral cells (Klein, 1987). In UV-irradiated embryos, these differences in pigmentation remained (see Fig. S1 in the supplementary material). Cells that had a ‘ventral’ appearance, i.e. darker and larger blastomeres, are referred to as UV-Ven cells, whereas cells that had a dorsal appearance, i.e. lighter and smaller blastomeres, are referred to as UV-Dors cells. Therefore, ‘right’ (i.e. UV-Rt) cells were defined by placing the UV-Dors cells towards the experimenter and then injecting the cells on the experimenter's right. At stage 45, fixed embryos were stained with X-gal.
UV irradiation for axis-alteration experiments
Experimental timing was normalized for temperature differences, such that 0NT=the time of fertilization and 1.0NT=the time of first cleavage. Unless otherwise stated, one-cell embryos were dejellied and collected at 0.4NT in 6% ficoll. (A subset of embryos, as indicated in the text, was irradiated at 0.2NT.) These time points were chosen because UV irradiation is most effective prior to 0.52NT (Scharf and Gerhart, 1983). In all cases, the vegetal sides of these embryos were exposed to UV waves for a period of 60 seconds. Only 4-7% of embryos appeared unaffected by UV irradiation and developed normally (see Fig. S2A in the supplementary material).
Late axis rescue in UV-irradiated embryos
To introduce axis information to the UV-irradiated embryos at a later stage in development, two methods were used. In method 1, UV-irradiated embryos were allowed to divide normally until they reached 16 cells. First, XSiamois was injected (8 pg) in a single ventral vegetal blastomere (Fan and Sokol, 1997) using the methods described above (n=110). Next, XSiamois injections were targeted to UV-Ven (n=168), UV-dors (n=102) or cells 90 degrees from the original organizer (UV-Lt or UV-Rt; n=81). In method 2, UV-irradiated embryos were allowed to divide normally until they reached the 32-cell stage. A total of 0.3 nl of 1.75 M LiCl was injected into a single third tier blastomere (Kao and Elinson, 1989) (n=51).
Early axis rescue in UV-irradiated embryos
Irradiated embryos were gently pipetted into an agarose grid with individual holes approximately 1.3 mm in diameter in 6% ficoll, which dehydrates the perivitelline space, preventing movement of the embryo within the vitelline membrane. The embryos were held in position by the agarose, preventing self-orienting (Scharf and Gerhart, 1980). The entire grid, containing embryos, was then tipped on an incline of 0, 20, 30, 45 or 90 degrees until the first cleavage occurred. Embryos were scored for heterotaxia at stage 45 [n=69-159 per treatment (exact numbers are shown in Table 2)]. While a tipping angle of 90 degrees has been used in previous studies, this angle was not the most effective rescue of the LR axis after UV irradiation (see Table S1 in the supplementary material); thus, the majority of our results focus on embryos tipped 30 degrees. This angle better recapitulates the degree of cortical rotation in a normal embryo and gives better rescue of dorsoanterior development after UV (Vincent et al., 1986; Vincent et al., 1987).
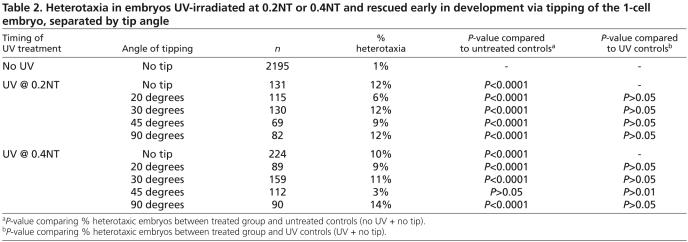
Table 2.
Heterotaxia in embryos UV-irradiated at 0.2NT or 0.4NT and rescued early in development via tipping of the 1-cell embryo, separated by tip angle
RESULTS
Rescue of UV irradiation past the 32-cell stage produces embryos that cannot properly orient the LR axis
In order to examine mechanisms of LR patterning by organizers induced at different time points, we required a method (1) whereby organizer induction could be prevented during cleavage stages and subsequently re-imposed upon the embryo, and (2) that did not result in multiple organizers or cause confounding Nodal signaling, as occurs during the induction of conjoined twins (Levin et al., 1996; Nascone and Mercola, 1997). Thus, we chose UV-irradiation of one-cell embryos as a method to remove axial patterning information, followed by its re-imposition upon these irradiated embryos at different points in development (Fan and Sokol, 1997; Kao and Elinson, 1989; Scharf and Gerhart, 1980).
We first titrated our UV transilluminator to find an effective exposure time to generate a maximum number of belly pieces and minimize the number of normal embryos (see Fig. S2A in the supplementary material). Belly pieces lack embryonic body axes and retain the simple radial symmetry of the egg with no apparent plane of bilateral symmetry (Scharf and Gerhart, 1983). The characterization of UV-irradiated embryos as radially symmetrical was confirmed by gross morphology (Fig. S2B in the supplementary material), histology (Fig. S2C) (Scharf and Gerhart, 1980; Scharf and Gerhart, 1983), and radially-symmetric expression (Fig. S2D) of dorsal markers, including Cerberus and Chordin (Bouwmeester et al., 1996). Together, these data confirm that UV-irradiated embryos lack patterning information sufficient to specify a dorsal organizer.
Microinjection of Siamois into UV-irradiated embryos at the 16-cell stage can restore normal dorsoanterior development (Fan and Sokol, 1997). Embryos were UV irradiated at 0.4NT (Scharf and Gerhart, 1983) and a single vegetal blastomere (16-cell stage) was injected with XSiamois mRNA (Fig. 1A). XSiamois injections rescued development with a 4-fold increase in the number of embryos with a normal DAI and a 6-fold decrease in the number of embryos with DAI=0, compared with UV-irradiation alone (data not shown). Only 1% of embryos treated with UV and injected with Siamois developed two axes, suggesting that a negligible number of embryos in the Siamois-rescue group escaped UV irradiation. Thus, the mRNA injections efficiently induced an organizer in irradiated embryos that was able to establish a primary axis and generate embryos with a bilateral body plan.
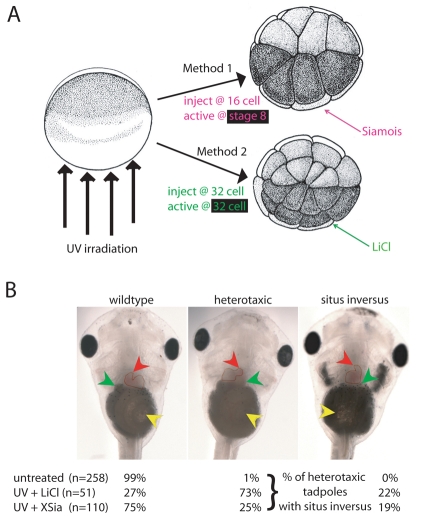
Fig. 1.
Late rescue of UV irradiation via injection of LiCl or XSiamois mRNA results in heterotaxia in embryos with a normal DAI. (A) One-cell embryos are exposed to UV irradiation from the vegetal side. Method 1: XSiamois mRNA is injected into a vegetal blastomere at the 16-cell stage. Method 2: LiCl is injected into a third tier blastomere at the 32-cell stage. (B) Organ situs of rescued embryos with DAI=5 compared with untreated embryos. All embryos were determined to be wild type or heterotaxic (inverted placement of at least one asymmetric organ). Of the heterotaxic embryos, the percentage with situs inversus (randomization of all three organs) is reported. Red arrowhead, heart loop; green arrowhead, gall bladder; yellow arrowhead, stomach.
The organizer resulting from XSiamois injection at the 16-cell stage does not have the benefit of any patterning information that is normally established prior to the fifth cell cleavage; moreover, because Siamois is a transcription factor and is not expected to be active until after mid-blastula transition initiates zygotic transcription (Hair et al., 1998), the induced organizer will appear significantly after cleavage stages. In order to determine whether XSiamois-rescued embryos can properly orient the LR axis, those with normal dorsoanterior development (DAI=5) were scored for organ situs. Twenty-five percent of these embryos were heterotaxic compared with less than 1% in untreated controls (n=110, χ2=46.4; P<0.0001; Fig. 1B). This incidence of heterotaxia is quite similar to what has been previously observed in well-established LR mutants [for instance, KiF3B mutants (Nonaka et al., 1998), cilia-related mutants (Bonnafe et al., 2004; Vierkotten et al., 2007; Zhao and Malicki, 2007), and ablation of cells at the node (Essner et al., 2005)]. We conclude that organizers induced by XSiamois do not have the ability to correctly pattern the LR axis. The variable and incomplete penetrance of almost all known perturbations of the LR pathway has not yet been explained, and may suggest multiple converging mechanisms or other poorly understood homeostatic mechanisms.
Many mutants in elements of the LR pathway have been shown to exhibit other developmental problems, including axial and midline defects (e.g. Ermakov et al., 2009; Kishimoto et al., 2008; Nonaka et al., 1998). We also examined embryos with slight defects in other axes (DAI=4). If such embryos were to be included in the analysis, 40% of XSiamois rescued embryos would be scored as heterotaxic (n=216, χ2=184.7; P<<0.0001), which is a high level of randomization and is significantly greater than that observed for many laterality syndromes or mutants. However, defects in the DV and AP axes are known to indirectly influence organ laterality (Danos and Yost, 1995); thus, although the significant incidence of heterotaxia resulting from XSiamois rescue could be increased even further, we chose not to include embryos with abnormal dorsoanterior development in further analyses, so as to maintain focus on primary LR mechanisms.
Because mid-blastula transition delay for transcription in Xenopus is not guaranteed to be absolute (Yang et al., 2002), and to further narrow down the timepoint after which LR asymmetry could not be normally established, embryos were UV irradiated at 0.4NT and a third tier blastomere (32-cell stage) was injected with 1.75 M LiCl (Fig. 1A). As shown previously (Kao and Elinson, 1989), LiCl injection rescued dorsoanterior development, with a 4.5-fold increase in embryos with a normal DAI and a 5-fold decrease in the number of embryos with DAI=0, compared with UV irradiation alone (data not shown). Less than 1% of embryos treated with UV and injected with LiCl developed two axes, suggesting that a negligible number of embryos had escaped UV irradiation (data not shown). Embryos with a normal DAI were then scored for organ situs.
Of the embryos that were UV-irradiated and rescued with LiCl injection, a striking 73% were heterotaxic, compared with only 1% of untreated embryos (n=51, χ2=113.7; P≤0.0001; Fig. 1B). It should be noted that the highest possible level of observable heterotaxia is about 87% (as, when three organs are fully and independently randomized, the organs will sometimes land in their normal positions by chance, being incorrectly scored as wild type); thus, this level of heterotaxia is close to the theoretical maximum. To test for non-specific effects of LiCl, we injected normal non-irradiated embryos with the same dose of LiCl, again targeting single third tier blastomeres at the 32-cell stage. Less than 4% of embryos with DAI=5 were heterotaxic, a non-significant increase over uninjected embryos (n=133, χ2=0.8; P=0.375). Thus, LiCl injection is not itself randomizing; however, LiCl-induced organizers are unable to properly pattern the LR axis.
We conclude that although rescue of UV irradiation, either via injection of XSiamois or LiCl, is capable of normally patterning the DV and AP axes, organizers induced after the first few cleavages cannot normally orient LR asymmetry.
The position of the induced organizer does not determine the incidence of heterotaxia
Late-induced organizers could be deficient in LR patterning because they miss the unique events occurring during early cleavage stages, which allow chiral cytoskeletal structures to be amplified into multicellular LR asymmetry. However, it is also possible that the randomization observed in late-rescued embryos was due to positioning of the organizer directly across from where the endogenous organizer would form. To distinguish these possibilities and determine whether the position of the induced organizer relative to the first two cleavage planes is a factor in its ability to orient the LR axis, we assayed laterality outcomes when organizers were induced in different locations during early cleavages. In most normal embryos, pigment differences allow the prospective ventral blastomeres (darker) to be distinguished from the prospective dorsal blastomeres (lighter). First, four-cell control embryos were injected with β-gal in the right side (Fig. 2A); 88% had detectable signal only in the right side, indicating that targeting of the right side of the embryo was successful (Fig. 2A, Table 1). A similar success rate was observed for control embryos injected with β-gal on the ventral side (Table 1). Thus, consistent with numerous studies in which one side of the embryo is targeted and the other is used as a contralateral control (e.g. Harvey and Melton, 1988; Helbling et al., 2000; Martin and Harland, 2006; Ristoratore et al., 1999; Warner et al., 1984), the large majority of normal embryos exhibited a good concordance of first cleavage plane with final embryonic midline, and a second cleavage plane that gives rise to identifiable dorsal and ventral precursor blastomeres.
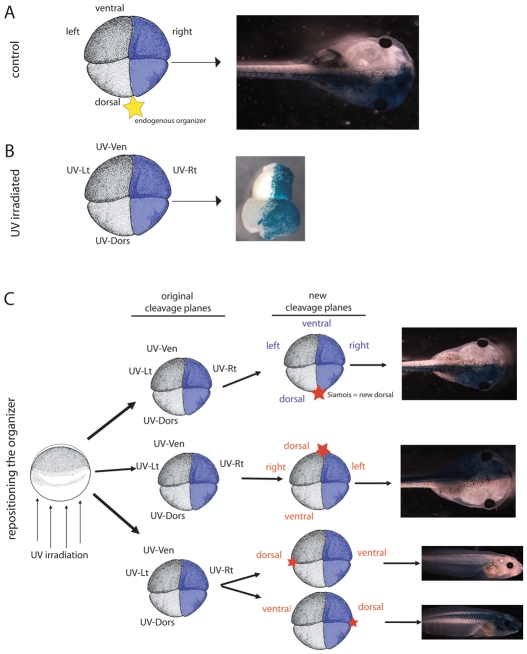
Fig. 2.
Establishment of the midline is dependent on the location of the new organizer, unless no organizer is present. (A) Both right blastomeres of control four-cell embryos were injected with β-gal. When the organizer develops on the dorsal side as expected (termed the ‘endogenous placement’, marked by a yellow star), β-gal signal remains localized to the right side of the tadpole. (B) In UV-irradiated embryos, the blastomeres no longer have ‘dorsal’ or ‘ventral’ fates, although they maintain the same appearance (i.e. cell size and pigmentation). New terms are therefore assigned to each blastomere (i.e. UV-Ven, UV-Dors, UV-Rt, UV-Lt). β-gal was injected into both UV-Rt blastomeres at the four-cell stage. If no further treatment is given, the β-gal signal localizes to one half of the resulting belly piece. (C) Both UV-Rt blastomeres of four-cell UV-irradiated embryos were injected with β-gal. Then, the position of the organizer was specifically targeted to one of three locations (marked by a red star) via injection of XSiamois at the 16-cell stage. When XSiamois is injected into the same location as the endogenous placement (UV-Dors), the cells maintain their original identities and the β-gal localizes to the right side of the rescued tadpole. When XSiamois is injected opposite the endogenous placement, the UV-Ven cells are re-defined as dorsal, and the β-gal signal localizes to the left side of rescued tadpole. Finally, if the organizer is positioned 90 degrees from the endogenous placement, β-gal is localized to either the ventral or the dorsal cells. Thus, proper organizer placement could be verified by β-gal localization in rescued tadpoles. Labels for cells that maintain their original identity are shown in blue, labels for cells that changed identity after placement of the organizer are shown in red.
Table 1.
Heterotaxia in UV-irradiated embryos rescued late in development by injection of XSiamois at one of three locations
Interestingly, even embryos UV-irradiated to a degree that results in massive death or DAI=0 establish pigment differences that distinguish prospective dorsal from ventral blastomeres (see Fig. S1 in the supplementary material), although in irradiated embryos the subsequent fate of these blastomeres is identical because the organizer (and thus the DV axis) has been ablated. To indicate the distinction between the darker blastomeres in control embryos that would give rise to ventral structures and the darker blastomeres in irradiated embryos, we refer to the darker cells in UV-irradiated embryos as UV-Ven (likewise, with UV-Rt, etc.). Embryos were UV irradiated and injected with β-gal lineage tracer at the four-cell stage in either UV-Rt or UV-Ven cells; analysis of the lineage marker signal revealed that even in embryos with no DV patterning, the early cleavage planes establish domains that divide the resulting belly piece into approximately equal halves (Fig. 2B).
We used pigment differences among blastomeres in UV-irradiated embryos to selectively place the organizer at different locations. At the 16-cell stage, XSiamois was injected into one of three positions (UV-Dors, UV-Ven, or 90° from the original organizer). The embryos were examined at stage 45 for localization of the lineage marker. Of the embryos rescued with XSiamois in each of three positions, a majority of the embryos had β-gal signal localized to a position that was correct relative to the placement of the new organizer (Fig. 2C, Table 1). Similar results were also obtained with LiCl injections (data not shown). These data indicate that this technique successfully and reliably repositions the organizer to the desired locale during early cleavage stages.
Using the β-gal positioning assay to verify organizer placement, we examined embryos that were injected with XSiamois at each of three locations for organ situs. Regardless of the position of the XSiamois injection, the resulting embryos were heterotaxic (Table 1). Consistent with the known fate-equivalence of blastomeres after organizer ablation, we found no position in which a late-induced organizer could properly pattern its LR axis. We conclude that specific placement relative to early cleavage planes is insufficient to enable a late organizer to pattern its LR axis; even when placed back in its original position (UV+XSiamois injected into UV-Dors cell), organizers induced any time after the 32-cell stage are unable to break symmetry in the correct orientation.
Tipping of UV-irradiated embryos rescues LR patterning
UV-irradiated embryos rescued later in development by injection of LiCl or XSiamois cannot orient the LR axis properly. Thus, it was important to ensure that it was not the UV treatment itself that was abolishing the embryos' ability to establish correct laterality, as well as to identify an early time period for organizer induction that, unlike later stages, was sufficient for normal asymmetry. To distinguish between randomizing effects of the UV treatment per se and the timing of organizer induction, we sought to rescue LR patterning much earlier, when the new organizer should still have the benefit of the cytoskeletal and bioelectrical events that occur during early cleavages.
To determine if an early rescue of UV-irradiated embryos allows for normal orientation of the LR axis, we used tipping, a procedure long known to rescue organizer induction (Scharf and Gerhart, 1980; Scharf and Gerhart, 1983). Embryos were irradiated, placed on an incline and allowed to complete the first cleavage at this angle (Fig. 3A). Of embryos tipped at 30°, only 11% were heterotaxic (Table 2). Other angles and an earlier timing of UV treatment (0.2NT) were tested and produced similar results (Table 2). This level of heterotaxia is relatively low compared with other treatments. Analysis of embryos that were not irradiated but tipped indicated that neither the physical constraints of the tipping apparatus nor the tipping itself affects development of the axes (data not shown).
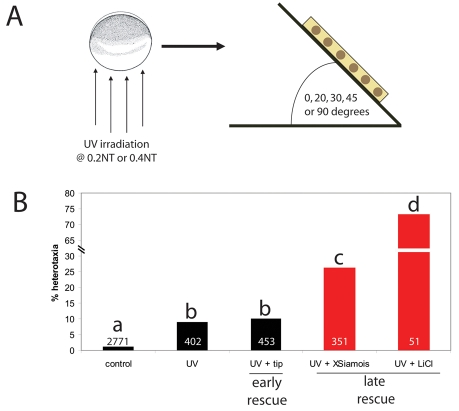
Fig. 3.
Very early rescue of UV irradiation via tipping results in embryos with normal LR asymmetry. (A) One-cell embryos were UV irradiated at either 0.2NT or 0.4NT and immediately pipetted into an agarose grid. The grid (depicted by a yellow rectangle) was then tipped at 0, 20, 30, 45 or 90 degrees for at least one cell division. (B) Statistical comparison of all embryos irradiated at 0.4NT indicates that late rescue of UV irradiation (via XSiamois or LiCl injections) produces significantly more heterotaxia than the randomizing effects of the UV irradiation itself (as indicated from the tipping rescue). Different letters indicate groups that are significantly different from each other (P<0.01). Sample sizes are indicated on the bars of the graph.
χ2 tests were performed to compare the laterality outcomes in all the treatments discussed above (UV+tipping, UV+XSiamois, UV+LiCl). While UV irradiation itself did produce a low background level of heterotaxia, late rescues of UV irradiation (XSiamois and LiCl injections) exhibited significantly more heterotaxia than the background level caused by UV irradiation (Fig. 3B). The very low level of heterotaxia observed in early rescues of UV irradiation (3-14%) might be indicative of the importance of extremely early events in establishing LR asymmetry, i.e. the establishment of a biased cytoskeleton (Danilchik et al., 2006; Qiu et al., 2005). However, it is clear that early rescue (via tipping) results in much better LR patterning than does late rescue.
Left-sided twins have normal organ situs, even when they arise from late organizer induction, if a primary, early-induced twin is present
Given our finding that late-induced singleton organizers cannot orient asymmetry correctly, we first repeated the study of Nascone and Mercola (Nascone and Mercola, 1997) to ensure that differences in technique were not responsible for differences in results. They reported that the induced twin (forming when thousands of cells are present) has normal LR asymmetry when that twin is located on the left (Nascone and Mercola, 1997). We induced conjoined twins using injections of XSiamois opposite the location of the endogenous organizer (Kessler, 1997; Medina et al., 1997). At stage 45, each twin was scored for organ situs (Fig. 4A-D). When the secondary axis (induced twin) formed on the left side, heart development in this twin was rarely inverted, whereas heart looping in the primary twin was randomized (Table 3). Additionally, the sidedness of Nodal (Xnr-1) expression was assessed in stage 22 embryos via in situ hybridization. As observed previously (Hyatt and Yost, 1998; Nascone and Mercola, 1997), randomized heart looping in twins developing on the right side correlated with the randomization of Xnr-1 expression in the twin, regardless of the side where the secondary axis develops (see Table S2 in the supplementary material).
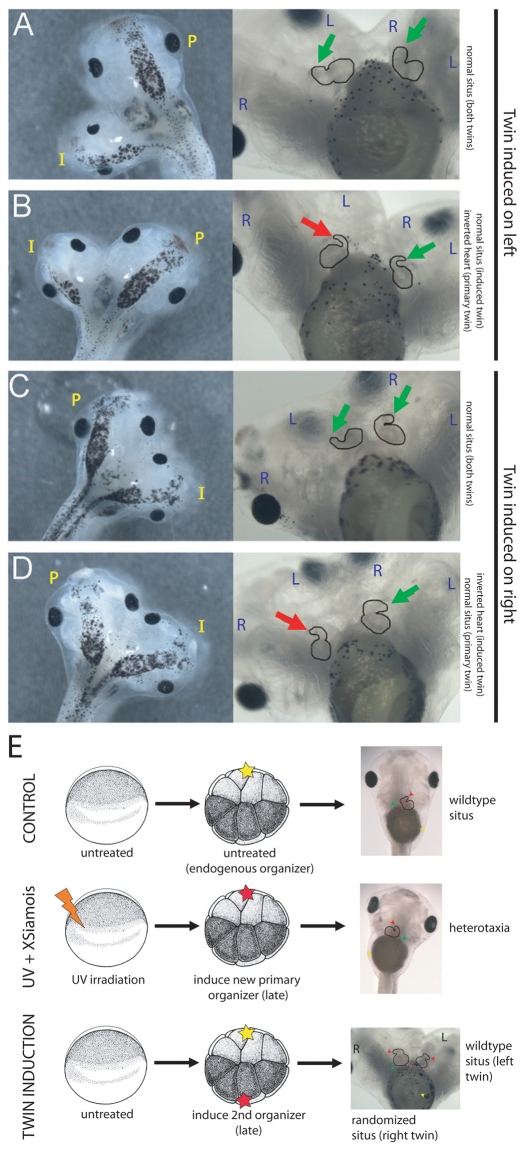
Fig. 4.
Induced twins re-pattern the LR axis information established by the primary twin but the LR axis cannot be oriented de novo late in development. (A,B) In conjoined twins where the induced twin is located on the left, the left twin typically develops with proper heart situs. However, the twin on the right has randomized heart situs. (C,D) When the induced twin is located on the right, the primary twin, forming on the left, still develops with proper heart situs. However, the right-sided twin is randomized. Green arrow, correct heart looping; red arrow, inverted heart looping. The primary (P) and induced (I) twins are indicated, and the left (L) and right (R) halves of each twin are marked. (E) Schematic detailing experiments distinguishing the LR patterning in singleton versus twinned late organizers. In untreated embryos, tadpoles develop normal organ situs. In embryos that have patterning information ablated at the one-cell stage and a single organizer introduced at late stages, the embryo is often heterotaxic. In embryos that maintain the primary axis but develop a second axis late in development, the induced twin typically has normal organ situs (if it develops on the left). Yellow star, endogenous organizer; red star, induced organizer.
Table 3.
Organ situs in conjoined twins is dependent on twin position
Confirming the results of Nascone and Mercola, we observed that late twins can orient their LR axis properly. However, our data reveal that this is not an intrinsic ability of late organizers but rather occurs only when there exists a primary conjoined twin that had the opportunity to orient its LR axis during early cleavage stages (Fig. 4E). In singleton organizers induced late, which have no opportunity to receive instructive signals from an early twin with correct laterality, asymmetry cannot be oriented consistently. Our data reveal that an organizer can orient its LR axis in two ways: from events occurring prior to the first cleavage (recapitulated by tipping), or secondarily by instruction from a primary organizer that participated in those events.
DISCUSSION
Rescue of UV irradiation enables the study of early and late LR patterning
LR patterning is defined with respect to a plane bisecting the primary embryonic axes. During normal development, the sperm entry point marks the side of the embryo that will be ventral (Scharf and Gerhart, 1980); cortical rotation then shifts dorsalizing elements from the vegetal pole to the dorsal side (Chang et al., 1996; Nagano et al., 2000). Only when this process occurs is the DV axis established, and the LR axis can then be oriented with respect to the DV and AP axes. Blocking cortical rotation with treatments that prevent the polymerization of microtubules (Elinson and Rowning, 1988; Scharf and Gerhart, 1983) prevents the formation of Spemann's organizer. Without a functional organizer, these embryos form ‘belly pieces’ lacking axes or patterning information (Bouwmeester et al., 1996; Sato and Sargent, 1990; Scharf and Gerhart, 1983).
Development of belly pieces can be prevented and proper DV and AP patterning can be achieved if UV-irradiated embryos are injected with XSiamois (Fan and Sokol, 1997) or LiCl (Kao and Elinson, 1989), or are tipped early in development (Scharf and Gerhart, 1980). Each of these rescue methods restores the Spemann's organizer at a different time point and a specific location. In normal embryos, the organizer is aligned with respect to the LR and DV planes determined by the early cleavages; however, these can be experimentally dissociated (by moving the organizing activity relative to the first few blastomeres) in order to probe the independent functions of organizer versus early events in LR determination. Thus, UV irradiation followed by organizer induction is a powerful technique with which to analyze the concordance of the three axes in early development.
Early-cleavage events are crucial for the establishment of LR asymmetry by an organizer
Establishment of consistent LR patterning in an embryo requires an organizer to set up an LR axis that is correct relative to the primary (AP/DV) axes it induces. Spemann's organizer is essential for the development and induction of DV and AP patterning in both twins and singletons (Bouwmeester, 2001; Branford et al., 2000; Nascone and Mercola, 1997; Sive, 1993). The ability to ablate the endogenous organizer via UV irradiation and then rescue it with a variety of reagents at different time points provides a means of investigating precisely when an organizer is able to coordinate the three axes. This is important for deciding among models that postulate very early, intracellular events versus late gastrulation extracellular processes as being key to LR orientation (Aw and Levin, 2009; Speder et al., 2007; Tabin, 2005). Our data reveal that even when the organizer is returned to the same location where it would normally occur (in UV-irradiated embryos rescued by injecting XSiamois into UV-Dors blastomeres), as little as a few cell divisions later than it would normally appear, the resulting embryo cannot orient the LR axis correctly. Importantly, the resulting embryos are otherwise normal, with a wild-type dorsoanterior index, which indicates that the late organizer is able to successfully pattern the AP and DV axes, but cannot originate reliable LR orientation. Moreover, this demonstrates that late organizers can bisect themselves and establish an anatomical midline and a bilateral bodyplan, but not orient the LR axis with respect to the AP and DV axes. Importantly, it is seen (Fig. 2C) that simple alignment with respect to cleavage planes is insufficient — the early timing is crucial.
It should be noted that our results could not be due to a failure to fully ablate the original organizer; failure to remove this organizer would not have resulted in the observed difference in LR properties between early- and late-rescued embryos. Lack of such a difference could have cast doubt on the UV treatment, but the converse was observed: the UV treatment was clearly able to ablate the function of the organizer enough so a change in LR orienting properties could be observed. Moreover, our results suggest that the UV treatment was very effective in erasing the primary DV patterning information because: (1) >80% of embryos developed morphologically as belly pieces (see Fig. S2 in the supplementary material); (2) gene expression data confirm that these embryos did not form an organizer (see Fig. S2); and (3) less than 1% of embryos developed two axes, as compared with our typical success rate of 60% for generating twins in non-irradiated embryos (data not shown). Because UV irradiation disrupts the cytoskeleton, an element known to be crucial for LR patterning across a wide range of phyla (Aw et al., 2008; Danilchik et al., 2006; Qiu et al., 2005; Thitamadee et al., 2002), it was not possible to rescue embryos 100%. However, early tipping was quite effective, reducing the incidence of heterotaxia significantly compared with rescue taking place after the 32-cell stage.
Thus, we propose that the requirement for early induction is due to the need for the unique events occurring during early cleavage stages, which allow chiral cytoskeletal structures to be amplified via several intracellular and physiological processes into multicellular LR asymmetry (Levin and Palmer, 2007; Raya and Belmonte, 2006). A most likely candidate for the relevant process is the cytoskeletal rearrangement that takes place during cortical rotation, which is an ideal mechanism for integrating three axes with a chiral component (Levin and Mercola, 1998a; Yost, 1991). This model is applicable even to embryos with radically different architectures, as similar cytoskeletal orientations are known to occur even in single mammalian cells in culture (Xu et al., 2007).
Our data do not rule out late nodal flow models: even though ciliary motion and density in the nodes of late-induced organizers appeared normal, it is possible that some subtle, unknown aspect of ciliary function could have been disrupted in late-induced embryos and could thus be responsible for their LR randomization. However, it is clear that our results are not predicted by models relying on nodal flow at neurulation to initiate consistent asymmetry de novo. Ciliary models predict that even a late-induced organizer that makes a competent node (and fully normal dorso-anterior axes, as we observed) would be able to orient asymmetry properly. The above data are instead more consistent with models in which primary LR asymmetry is established prior to the 32-cell stage (Levin, 2006; Yost, 1991), and can be amplified or refined by numerous downstream mechanisms.
Late organizers can properly orient LR asymmetries, if instructed by a primary twin that oriented LR early
The influential study of Nascone and Mercola (Nascone and Mercola, 1997) has been widely interpreted to suggest that, even in Xenopus where asymmetries clearly exist very early (Adams et al., 2006; Aw et al., 2008; Danilchik et al., 2006; Hyatt and Yost, 1998; Levin et al., 2002), asymmetry can be generated de novo after the mid-blastula transition. Although we have confirmed their original observation, our data reveal a different explanation: instructive interactions between organizers.
Studies of conjoined twins in experimental animals, as well as humans, have offered many insights into how tissues are organized across large distances (Kaufman, 2004; Levin, 1999). Conjoined twins often have laterality defects and cardiac abnormalities (Levin et al., 1996; Manning, 2008), revealing that primary axes in the same blastoderm interact functionally with respect to LR patterning. However, earlier data revealed only confounding cross-talk between twins at primitive streak stages. We confirmed the results reported previously that an induced twin (forming when thousands of cells are present) has normal LR asymmetry (Nascone and Mercola, 1997). This only occurs when that twin is located on the left, but the difference is due to randomization by cross-over of Shh/Activin/Nodal at late stages. Because of the confounding presence of multiple axes in the same organism, we developed a new assay that analyzed asymmetry in early- or late-organized singleton embryos. Although the UV treatment itself led to a small degree of laterality disturbance (3-14% when rescued by early tipping), this was clearly distinguishable from the near-maximal randomization (73%) that was observed following rescue even a few cell cleavages later (via LiCl).
These experiments revealed a striking and novel interaction between the organizers, which is schematized in Fig. 5. We showed that a singleton embryo completely lacks LR orientation when its organizer is induced after the first few cleavages (Fig. 5B). However, if the blastoderm already contains an early-induced twin, then the late-induced twin can properly orient the late-forming LR axis (Fig. 5C). We propose that the laterality of the induced twin is somehow instructed by the primary twin, which had access to the unique processes occurring during the first few cleavages. These processes include a right-biased cytoskeleton (Aw et al., 2008; Danilchik et al., 2006) that is likely to guide intracellular transport via kinesins and dyneins (Nonaka et al., 1998; Qiu et al., 2005; Supp et al., 1997), asymmetric ion flows and voltage gradients (Adams et al., 2006; Levin et al., 2002), and the resulting asymmetric redistribution of small signaling molecules (Fukumoto et al., 2005a; Fukumoto et al., 2005b).
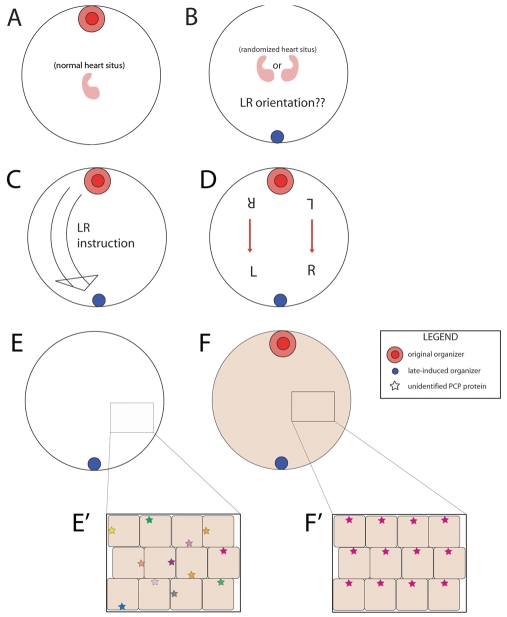
Fig. 5.
A polarity model for the instructive influence exerted by an early-induced organizer upon late-induced twin axes. (A) Schematic of a single organizer in an unperturbed, normal embryo. (B) When the primary organizer is not present and an organizer is induced late, the LR orientation is randomized. (C) If an organizer is induced late in the presence of an endogenous early organizer, LR instruction is passed from the primary organizer to the late-forming organizer. (D) The new orientation of the LR axis cannot be due to leaky signaling from the primary axis because only the left and right halves of each twin are in contact to share a LR morphogen. (E) In an embryo lacking a primary organizer with a late-induced organizer, a PCP protein (indicated by stars) that normally points all cells ‘dorsal’ towards the organizer is distributed randomly (E′). (F) Using cytoplasmic mechanisms, an embryo with a primary organizer and a late-induced organizer can orient all cells in a coherent pattern (F′) that each organizer can use, in combination with its own local AP/DV direction, to derive a consistent LR axis.
Although the first chick experiments revealed separate left and right compartments with a unique gene expression cascade (Levin et al., 1995), subsequent analysis of earlier steps revealed an obligate communication between the left and right sides (Levin and Mercola, 1998b; Levin and Mercola, 1999). A similar pattern emerges from twin:twin interactions: while conjoined twins adversely affect each others' laterality during later development by crossover of side-specific secreted signals (Branford et al., 2000; Levin et al., 1996; Nascone and Mercola, 1997), we have discovered that early twins communicate in a coordinated manner, allowing a late, ectopic twin to derive appropriate LR pattern from a primary organizer that took advantage of the unique information present at cleavage stages. How might this happen?
Simple secreted factor cross-over models do not work: Fig. 5D shows that a leakage of R- and L-specific signals would result in a secondary twin with situs inversus, as the left side of the secondary twin would be close to the right side of the primary twin and vice versa. Instead, the axis induced by the secondary organizer in these cases is correct with respect to its own AP and DV axes. Thus, we propose a different class of models, based on the recent suggestion that mechanisms of planar cell polarity can impose a LR orientation across a blastoderm (Aw and Levin, 2009).
In this model, a late-induced singleton finds itself in a blastoderm that has not been aligned properly by a primary organizer (Fig. 5E). By contrast, a late-induced twin organizer is embedded in a blastoderm that has already been aligned in the plane by early cytoskeleton-derived polarity (Fig. 5F) (see also Aw et al., 2008). We propose that each organizer makes use of this coherent orientation, in combination with its own local AP/DV direction, to derive a consistent LR axis and impose it on subsequent organ primordia. Early analyses of LR patterning (Brown and Wolpert, 1990) distinguished several steps, including generation of asymmetry, orientation with respect to AP/DV axes, and amplification across cell fields. We suggest that late organizers can orient asymmetry with respect to their own, local DV/AP direction. However, these organizers are missing the early events that establish asymmetry in the first place and the coordination needed to impose this asymmetry on multicellular cell fields (necessary for asymmetric gene induction). Planar polarity pathways oriented by early cytoskeletal events are ideal candidates for these roles. Future work will test specific mechanistic predictions of this model, and characterize the molecular-genetic nature of LR-relevant planar polarizations induced by the early organizer. It will also be important to examine the above questions in other species, particularly in amniotes (chick and rabbit) and teleosts (zebrafish).
Conclusion
Laterality is a crucial aspect of embryonic development; yet, many key questions remain about the timing of the earliest steps of LR asymmetry generation and their relationship with the origins of the primary axis and midline. Our results suggest that the LR axis in Xenopus cannot be reliably oriented de novo after the 32-cell stage, strongly supporting a model in which crucial LR asymmetry events take place shortly after fertilization. Surprisingly, we discovered that conjoined twins can be correctly re-aligned by patterning information established in the primary organizer during early cytoskeletal rearrangements. Future work to understand the fascinating molecular details of organizer orientation, cross-organizer instruction, and midline bisection of cell fields will illuminate important problems of evolutionary, cell and developmental biology, as well as an important class of birth defects.
Supplementary Material
Acknowledgements
The authors are grateful to N. Nascone-Yoder for helpful suggestions with the production of twins and the XSiamois clone, S. Moody for the Chordin clone, C. Wright for the XNr-1 clone, P. Koustubhan and A. Currier for Xenopus husbandry and general lab assistance, D. Kessler for useful discussions about UV irradiation and the loan of a quartz cuvette, D. Adams for assistance with statistics, and D. Blackiston for assistance with in situ hybridization and suggestions on the manuscript. This work was supported by American Heart Association Established Investigator Grant 0740088N and NIH grant R01GM077425 to M.L., and NRSA grant 1F32GM087107 to L.N.V. Deposited in PMC for release after 12 months.
Footnotes
Competing interests statement
The authors declare no competing financial interests.
Supplementary material
Supplementary material for this article is available at http://dev.biologists.org/lookup/suppl/doi:10.1242/dev.041798/-/DC1
References
- Adams D. S., Robinson K. R., Fukumoto T., Yuan S., Albertson R. C., Yelick P., Kuo L., McSweeney M., Levin M. (2006). Early, H+-V-ATPase-dependent proton flux is necessary for consistent left-right patterning of non-mammalian vertebrates. Development 133, 1657-1671 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aw S., Levin M. (2008). What's left in asymmetry? Dev. Dyn. 237, 3453-3463 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aw S., Levin M. (2009). Is left-right asymmetry a form of planar cell polarity? Development 136, 355-366 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aw S., Adams D. S., Qiu D., Levin M. (2008). H,K-ATPase protein localization and Kir4.1 function reveal concordance of three axes during early determination of left-right asymmetry. Mech. Dev. 125, 353-372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonnafe E., Touka M., AitLounis A., Baas D., Barras E., Ucla C., Moreau A., Flamant F., Dubruille R., Couble P., et al. (2004). The transcription factor RFX3 directs nodal cilium development and left-right asymmetry specification. Mol. Cell. Biol. 24, 4417-4427 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouwmeester T. (2001). The Spemann-Mangold organizer: the control of fate specification and morphogenetic rearrangements during gastrulation in Xenopus. Int. J. Dev. Biol. 45, 251-258 [PubMed] [Google Scholar]
- Bouwmeester T., Kim S.-H., Sasai Y., Lu B., DeRobertis E. M. (1996). Cerberus is a head-inducing secreted factor expressed in the anterior endoderm of Spemann's organizer. Nature 382, 595-601 [DOI] [PubMed] [Google Scholar]
- Branford W. W., Essner J. J., Yost H. J. (2000). Regulation of gut and heart left-right asymmetry by context-dependent interactions between Xenopus lefty and BMP4 signaling. Dev. Biol. 223, 291-306 [DOI] [PubMed] [Google Scholar]
- Brannon M., Kimelman D. (1996). Activation of Siamois by the Wnt pathway. Dev. Biol. 180, 344-347 [DOI] [PubMed] [Google Scholar]
- Brown N. A., Wolpert L. (1990). The development of handedness in left/right asymmetry. Development 109, 1-9 [DOI] [PubMed] [Google Scholar]
- Casey B. (1998). Two rights make a wrong: human left-right malformations. Human Mol. Genet. 7, 1565-1571 [DOI] [PubMed] [Google Scholar]
- Casey B., Hackett B. P. (2000). Left-right axis malformations in man and mouse. Curr. Opin. Genet. Dev. 10, 257-261 [DOI] [PubMed] [Google Scholar]
- Chang P., LeGuellec K., Houliston E. (1996). Immunodetection of cytoskeletal structures and the Eg5 motor protein on deep-etch replicas of Xenopus egg cortices isolated during the cortical rotation. Biol. Cell 88, 89-98 [PubMed] [Google Scholar]
- Chen I., Levin M. (2004). The role of KATP channels in development of left-right asymmetry in Xenopus. J. Dental Res. 83, A1340 [Google Scholar]
- Cooke J., Smith J. C. (1987). The midblastula cell cycle transition and the character of mesoderm in U.V.-induced nonaxial Xenopus development. Development 99, 197-210 [DOI] [PubMed] [Google Scholar]
- Danilchik M. V., Brown E. E., Riegert K. (2006). Intrinsic chiral properties of the Xenopus egg cortex: an early indicator of left-right asymmetry? Development 133, 4517-4526 [DOI] [PubMed] [Google Scholar]
- Danos M. C., Yost H. J. (1995). Linkage of cardiac left-right asymmetry and dorsal-anterior development in Xenopus. Development 121, 1467-1474 [DOI] [PubMed] [Google Scholar]
- Elinson R. P., Rowning B. (1988). A transient array of parallel microtubules in frog eggs: potential tracks for a cytoplasmic rotation that specifies the dorso-ventral axis. Dev Biol. 128, 185-197 [DOI] [PubMed] [Google Scholar]
- Ermakov A., Stevens J. L., Whitehill E., Robson J. E., Pieles G., Brooker D., Goggolidou P., Powles-Glover N., Hacker T., Young S. R., et al. (2009). Mouse mutagenesis identifies novel roles for left-right patterning genes in pulmonary, craniofacial, ocular, and limb development. Dev. Dyn. 238, 581-594 [DOI] [PubMed] [Google Scholar]
- Essner J. J., Amack J. D., Nyholm M. K., Harris E. B., Yost H. J. (2005). Kupffer's vesicle is a ciliated organ of asymmetry in the zebrafish embryo that initiates left-right development of the brain, heart and gut. Development 132, 1247-1260 [DOI] [PubMed] [Google Scholar]
- Fan M. J., Sokol S. Y. (1997). A role for Siamois in Spemann organizer formation. Development 124, 2581-2589 [DOI] [PubMed] [Google Scholar]
- Fukumoto T., Blakely R., Levin M. (2005a). Serotonin transporter function is an early step in left-right patterning in chick and frog embryos. Dev. Neurosci. 27, 349-363 [DOI] [PubMed] [Google Scholar]
- Fukumoto T., Kema I. P., Levin M. (2005b). Serotonin signaling is a very early step in patterning of the left-right axis in chick and frog embryos. Curr. Biol. 15, 794-803 [DOI] [PubMed] [Google Scholar]
- Gardner R. L. (2001). Specification of embryonic axes begins before cleavage in normal mouse development. Development Suppl. 128, 839-847 [DOI] [PubMed] [Google Scholar]
- Hair A., Prioleau M. N., Vassetzky Y., Mechali M. (1998). Control of gene expression in Xenopus early development. Dev. Genet. 22, 122-131 [DOI] [PubMed] [Google Scholar]
- Harland R. M. (1991). In situ hybridization: an improved whole mount method for Xenopus embryos. In Xenopus laevis: Practical uses in Cell and Molecular Biology, Vol. 36 (ed. Kay B. K., Peng H. B.), pp. 685-695 San Diego: Academic Press; [DOI] [PubMed] [Google Scholar]
- Harvey R. P., Melton D. A. (1988). Microinjection of synthetic Xhox-1A homeobox mRNA disrupts somite formation in developing Xenopus embryos. Cell 53, 687-697 [DOI] [PubMed] [Google Scholar]
- Helbling P. M., Saulnier D. M., Brandli A. W. (2000). The receptor tyrosine kinase EphB4 and ephrin-B ligands restrict angiogenic growth of embryonic veins in Xenopus laevis. Development 127, 269-278 [DOI] [PubMed] [Google Scholar]
- Houliston E. (1994). Microtubule translocation and polymerisation during cortical rotation in Xenopus eggs. Development 120, 1213-1220 [Google Scholar]
- Hyatt B. A., Yost H. J. (1998). The left-right coordinator: the role of Vg1 in organizing left-right axis formation. Cell 93, 37-46 [DOI] [PubMed] [Google Scholar]
- Kao K. R., Elinson R. P. (1989). Dorsalization of mesoderm induction by lithium. Dev Biol. 132, 81-90 [DOI] [PubMed] [Google Scholar]
- Kao K. R., Elinson R. P. (1998). The legacy of lithium effects on development. Biol. Cell 90, 585-590 [PubMed] [Google Scholar]
- Kaufman M. H. (2004). The embryology of conjoined twins. Childs Nerv. Syst. 20, 508-525 [DOI] [PubMed] [Google Scholar]
- Kessler D. S. (1997). Siamois is required for formation of Spemann's organizer. Proc. Natl. Acad. Sci. USA 94, 13017-13022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kishimoto N., Cao Y., Park A., Sun Z. (2008). Cystic kidney gene seahorse regulates cilia-mediated processes and Wnt pathways. Dev. Cell 14, 954-961 [DOI] [PubMed] [Google Scholar]
- Klein S. L. (1987). The first cleavage furrow demarcates the dorsal-ventral axis in Xenopus embryos. Dev. Biol. 120, 299-304 [DOI] [PubMed] [Google Scholar]
- Kramer-Zucker A. G., Olale F., Haycraft C. J., Yoder B. K., Schier A. F., Drummond I. A. (2005). Cilia-driven fluid flow in the zebrafish pronephros, brain and Kupffer's vesicle is required for normal organogenesis. Development 132, 1907-1921 [DOI] [PubMed] [Google Scholar]
- Levin M. (1999). Twinning and embryonic left-right asymmetry. Laterality 4, 197-208 [DOI] [PubMed] [Google Scholar]
- Levin M. (2006). Is the early left-right axis like a plant, a kidney, or a neuron? The integration of physiological signals in embryonic asymmetry. Birth Defects Res. C Embryo Today 78, 191-223 [DOI] [PubMed] [Google Scholar]
- Levin M., Nascone N. (1997). Two molecular models of initial left-right asymmetry generation. Medical Hypotheses 49, 429-435 [DOI] [PubMed] [Google Scholar]
- Levin M., Mercola M. (1998a). The compulsion of chirality: toward an understanding of left-right asymmetry. Genes Dev. 12, 763-769 [DOI] [PubMed] [Google Scholar]
- Levin M., Mercola M. (1998b). Gap junctions are involved in the early generation of left-right asymmetry. Dev Biol. 203, 90-105 [DOI] [PubMed] [Google Scholar]
- Levin M., Mercola M. (1999). Gap junction-mediated transfer of left-right patterning signals in the early chick blastoderm is upstream of Shh asymmetry in the node. Development 126, 4703-4714 [DOI] [PubMed] [Google Scholar]
- Levin M., Palmer A. R. (2007). Left-right patterning from the inside out: widespread evidence for intracellular control. BioEssays 29, 271-287 [DOI] [PubMed] [Google Scholar]
- Levin M., Johnson R., Stern C., Kuehn M., Tabin C. (1995). A molecular pathway determining left-right asymmetry in chick embryogenesis. Cell 82, 803-814 [DOI] [PubMed] [Google Scholar]
- Levin M., Roberts D., Holmes L., Tabin C. (1996). Laterality defects in conjoined twins. Nature 384, 321 [DOI] [PubMed] [Google Scholar]
- Levin M., Thorlin T., Robinson K. R., Nogi T., Mercola M. (2002). Asymmetries in H+/K+-ATPase and cell membrane potentials comprise a very early step in left-right patterning. Cell 111, 77-89 [DOI] [PubMed] [Google Scholar]
- Levin M., Buznikov G. A., Lauder J. M. (2006). Of minds and embryos: left-right asymmetry and the serotonergic controls of pre-neural morphogenesis. Dev. Neurosci. 28, 171-185 [DOI] [PubMed] [Google Scholar]
- Manning N. (2008). The influence of twinning on cardiac development. Early Hum. Dev. 84, 173-179 [DOI] [PubMed] [Google Scholar]
- Martin B. L., Harland R. M. (2006). A novel role for lbx1 in Xenopus hypaxial myogenesis. Development 133, 195-208 [DOI] [PubMed] [Google Scholar]
- McGrath J., Brueckner M. (2003). Cilia are at the heart of vertebrate left-right asymmetry. Curr. Opin. Genet. Dev. 13, 385-392 [DOI] [PubMed] [Google Scholar]
- Medina A., Wendler S. R., Steinbeisser H. (1997). Cortical rotation is required for the correct spatial expression of nr3, sia and gsc in Xenopus embryos. Int. J. Dev. Biol. 41, 741-745 [PubMed] [Google Scholar]
- Morokuma J., Blackiston D., Levin M. (2008). KCNQ1 and KCNE1 K+ channel components are involved in early left-right patterning in Xenopus laevis embryos. Cell. Biochem. Biophys. 21, 357-372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagano T., Ito Y., Tashiro K., Kobayakawa Y., Sakai M. (2000). Dorsal induction from dorsal vegetal cells in Xenopus occurs after mid-blastula transition. Mech. Dev. 93, 3-14 [DOI] [PubMed] [Google Scholar]
- Nascone N., Mercola M. (1997). Organizer induction determines left-right asymmetry in Xenopus. Dev. Biol. 189, 68-78 [DOI] [PubMed] [Google Scholar]
- Nieuwkoop P. D., Faber J. (1967). Normal table of Xenopus laevis (Daudin) Amsterdam: North-Holland Publishing Company; [Google Scholar]
- Nonaka S., Tanaka Y., Okada Y., Takeda S., Harada A., Kanai Y., Kido M., Hirokawa N. (1998). Randomization of left-right asymmetry due to loss of nodal cilia generating leftward flow of extraembryonic fluid in mice lacking KIF3B motor protein. Cell 95, 829-837 [DOI] [PubMed] [Google Scholar]
- Nonaka S., Shiratori H., Saijoh H., Hamada H. (2002). Determination of left-right patterning of the mouse embryo by artificial nodal flow. Nature 418, 96-99 [DOI] [PubMed] [Google Scholar]
- Okumura T., Utsuno H., Kuroda J., Gittenberger E., Asami T., Matsuno K. (2008). The development and evolution of left-right asymmetry in invertebrates: lessons from Drosophila and snails. Dev. Dyn. 237, 3497-515 [DOI] [PubMed] [Google Scholar]
- Otto E. A., Schermer B., Obara T., O'Toole J. F., Hiller K. S., Mueller A. M., Ruf R. G., Hoefele J., Beekmann F., Landau D., et al. (2003). Mutations in INVS encoding inversin cause nephronophthisis type 2, linking renal cystic disease to the function of primary cilia and left-right axis determination. Nat. Genet. 34, 413-420 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peeters H., Devriendt K. (2006). Human laterality disorders. Eur. J. Med. Genet. 49, 349-362 [DOI] [PubMed] [Google Scholar]
- Plusa B., Piotrowska K., Zernicka-Goetz M. (2002). Sperm entry position provides a surface marker for the first cleavage plane of the mouse zygote. Genesis 32, 193-198 [DOI] [PubMed] [Google Scholar]
- Qiu D., Cheng S. M., Wozniak L., McSweeney M., Perrone E., Levin M. (2005). Localization and loss-of-function implicates ciliary proteins in early, cytoplasmic roles in left-right asymmetry. Dev. Dyn. 234, 176-189 [DOI] [PubMed] [Google Scholar]
- Ramsdell A. F. (2005). Left-right asymmetry and congenital cardiac defects: getting to the heart of the matter in vertebrate left-right axis determination. Dev. Biol. 288, 1-20 [DOI] [PubMed] [Google Scholar]
- Raya A., Belmonte J. C. (2006). Left-right asymmetry in the vertebrate embryo: from early information to higher-level integration. Nat. Rev. Genet. 7, 283-293 [DOI] [PubMed] [Google Scholar]
- Ristoratore F., Carl M., Deschet K., Richard-Parpaillon L., Boujard D., Wittbrodt J., Chourrout D., Bourrat F., Joly J. S. (1999). The midbrain-hindbrain boundary genetic cascade is activated ectopically in the diencephalon in response to the widespread expression of one of its components, the medaka gene Ol-eng2. Development 126, 3769-3779 [DOI] [PubMed] [Google Scholar]
- Sampath K., Cheng A., Frisch A., Wright C. (1997). Functional differences among Xenopus nodal-related genes in left-right axis determination. Development 124, 3293-3302 [DOI] [PubMed] [Google Scholar]
- Sasai Y., Lu B., Steinbeisser H., Geissert D., Gont L. K., De Robertis E. M. (1994). Xenopus chordin: a novel dorsalizing factor activated by organizer-specific homeobox genes. Cell 79, 779-790 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato S. M., Sargent T. D. (1990). Molecular approach to dorsoanterior development in Xenopus laevis. Dev. Biol. 137, 135-141 [DOI] [PubMed] [Google Scholar]
- Scharf S. R., Gerhart J. C. (1980). Determination of the dorsal-ventral axis in eggs of Xenopus laevis: complete rescue of UV-impaired eggs by oblique orientation before first cleavage. Dev. Biol. 79, 181-198 [DOI] [PubMed] [Google Scholar]
- Scharf S. R., Gerhart J. C. (1983). Axis determination in eggs of Xenopus laevis: a critical period before first cleavage, identified by the common effects of cold, pressure and ultraviolet irradiation. Dev. Biol. 99, 75-87 [DOI] [PubMed] [Google Scholar]
- Schlueter J., Brand T. (2007). Left-right axis development: examples of similar and divergent strategies to generate asymmetric morphogenesis in chick and mouse embryos. Cytogenet. Genome Res. 117, 256-267 [DOI] [PubMed] [Google Scholar]
- Schweickert A., Weber T., Beyer T., Vick P., Bogusch S., Feistel K., Blum M. (2007). Cilia-driven leftward flow determines laterality in Xenopus. Curr. Biol. 17, 60-66 [DOI] [PubMed] [Google Scholar]
- Sive H. L. (1993). The frog prince-ss: a molecular formula for dorsoventral patterning in Xenopus. Genes Dev. 7, 1-12 [DOI] [PubMed] [Google Scholar]
- Sive H., Grainger R. M., Harland R. (2000). Early development of Xenopus laevis New York: Cold Spring Harbor Laboratory Press; [Google Scholar]
- Speder P., Petzoldt A., Suzanne M., Noselli S. (2007). Strategies to establish left/right asymmetry in vertebrates and invertebrates. Curr. Opin. Genet. Dev. 17, 351-358 [DOI] [PubMed] [Google Scholar]
- Supp D. M., Witte D. P., Potter S. S., Brueckner M. (1997). Mutation of an axonemal dynein affects left-right asymmetry in inversus viscerum mice. Nature 389, 963-966 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabin C. (2005). Do we know anything about how left-right asymmetry is first established in the vertebrate embryo? J. Mol. Histol. 36, 317-323 [DOI] [PubMed] [Google Scholar]
- Tabin C. J., Vogan K. J. (2003). A two-cilia model for vertebrate left-right axis specification. Genes Dev. 17, 1-6 [DOI] [PubMed] [Google Scholar]
- Thitamadee S., Tuchihara K., Hashimoto T. (2002). Microtubule basis for left-handed helical growth in Arabidopsis. Nature 417, 193-196 [DOI] [PubMed] [Google Scholar]
- Vierkotten J., Dildrop R., Peters T., Wang B., Ruther U. (2007). Ftm is a novel basal body protein of cilia involved in Shh signalling. Development 134, 2569-2577 [DOI] [PubMed] [Google Scholar]
- Vincent J.-P., Oster G. F., Gerhart J. C. (1986). Kinematics of gray crescent formation in Xenopus eggs: the displacement of subcortical cytoplasm relative to the egg surface. Dev. Biol. 113, 484-500 [DOI] [PubMed] [Google Scholar]
- Vincent J.-P., Scharf S. R., Gerhart J. C. (1987). Subcortical rotation in Xenopus eggs: a preliminary study of its mechanochemical basis. Cell Motil. Cytoskel. 8, 143-154 [DOI] [PubMed] [Google Scholar]
- Wagner M. K., Yost H. J. (2000). Left-right development: the roles of nodal cilia. Curr. Biol. 10, R149-R151 [DOI] [PubMed] [Google Scholar]
- Warner A. E., Guthrie S. C., Gilula N. B. (1984). Antibodies to gap-junctional protein selectively disrupt junctional communication in the early amphibian embryo. Nature 311, 127-131 [DOI] [PubMed] [Google Scholar]
- Xu J., Van Keymeulen A., Wakida N. M., Carlton P., Berns M. W., Bourne H. R. (2007). Polarity reveals intrinsic cell chirality. Proc. Natl. Acad. Sci. USA 104, 9296-9300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J., Tan C., Darken R. S., Wilson P. A., Klein P. S. (2002). Beta-catenin/Tcf-regulated transcription prior to the midblastula transition. Development 129, 5743-5752 [DOI] [PubMed] [Google Scholar]
- Yost H. J. (1991). Development of the left-right axis in amphibians. Ciba Found. Symp. 162, 165-176 [DOI] [PubMed] [Google Scholar]
- Zhao C., Malicki J. (2007). Genetic defects of pronephric cilia in zebrafish. Mech. Dev. 124, 605-616 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.