Abstract
The terminally differentiated umbrella cells of bladder epithelium contain unique cytoplasmic organelles, the fusiform vesicles, which deliver preassembled crystalline arrays of uroplakin proteins to the apical cell surface of urothelial umbrella cells. We have investigated the possible role of Rab proteins in this delivery process, and found Rab27b to be expressed at an extraordinary high level (0.1% of total protein) in urothelium, whereas Rab27b levels were greatly reduced (to <5% of normal urothelium) in cultured urothelial cells, which synthesized only small amounts of uroplakins and failed to form fusiform vesicles. Immuno-electron microscopy showed that Rab27b was associated with the cytoplasmic face of the fusiform vesicles, but not with that of the apical plasma membrane. The association of Rab27b with fusiform vesicles and its differentiation-dependent expression suggest that this Rab protein plays a role in regulating the delivery of fusiform vesicles to the apical plasma membrane of umbrella cells.
The urinary bladder and much of the rest of the urinary tract are lined by urothelium, a stratified epithelium that serves to contain the urine and to protect the underlying tissues from its potentially harmful effects. The urothelium accomplishes this by forming a top layer of highly differentiated umbrella cells that elaborate a specialized apical asymmetric unit membrane. This unique membrane structure is composed mainly of four integral membrane proteins, the uroplakins Ia, Ib, II, and III (1–6). These proteins begin to assemble in the endoplasmic reticulum where they form specific dimers (7–9), and they assemble further in a post-Golgi compartment represented by discoidal vesicles, where they form increasingly larger crystalline arrays composed of 16-nm hexagonal particles (1, 10–13). These crystalline uroplakin arrays expand and cover almost the entire surface of mature fusiform vesicles, which ultimately fuse with the apical plasma membrane of umbrella cells (10, 11, 13–15). Uroplakin plaques at the cell surface are thought to undergo a dynamic process of internalization and reinsertion that modulates the surface area of the urothelium during the micturition cycle (11, 16–21). However, it has recently been reported that the retrieval of uroplakins from the plasma membrane is an endocytic process not followed by temporary storage for future recycling, but rather by the degradation of uroplakins in lysosomal compartments (21).
Rab proteins are key regulators of vesicular transport steps that interconnect membrane-bounded organelles, and are thought to play critical roles in the targeting, docking, and/or fusion of vesicular carriers with their specific acceptor compartments (22–26). Certain Rab proteins support functions common to all cells, whereas others are only found in some cell types where they participate in specialized functions (27). Thus, the restricted tissue distribution of certain Rab proteins is likely to reflect cell type-specific membrane traffic events in which they participate. Therefore, we hypothesized that specific Rab proteins, which may be expressed in urothelium in a differentiation-dependent fashion, may play a key role in the regulated targeting of the uroplakin-containing fusiform vesicles to the highly specialized urothelial apical surface. An experimental system that may be useful for investigating the differentiation-dependent expression of Rab proteins is cultured urothelial cells, which synthesize and transport small amounts of uroplakins to the plasma membrane, but fail to form fusiform vesicles and uroplakin crystals (28). Thus, it would be interesting to determine whether these cultured urothelial cells are deficient in certain (normal) urothelium-enriched Rab protein(s) that are required for the biogenesis and/or function of fusiform vesicles. In this article, we demonstrate that Rab27b is urothelium-enriched, differentiation-dependent, and associated with fusiform vesicles. These observations suggest that Rab27b plays a key role in the regulated delivery and/or targeting of fusiform vesicles to the apical plasma membrane of urothelial umbrella cells, and that it provides a link to the cytoskeleton of urothelial cells thus serving a dual structural and regulatory role. This links Rab27b functions in urothelium to the known functions of Rab27b and its isoform Rab27a in other cells in targeting melanosomes (29, 30), secretory granules (31, 32), and secretory lysosomes (33).
Materials and Methods
Degenerate PCR. Taking advantage of the fact that all Rab proteins share a 7-aa motif (WDTAGQE) in the so-called region 2, we designed degenerate PCR primers corresponding to this conserved region and to the more variable sequences ([A/G/V]NKXD) of region 3 (34). A cDNA preparation obtained from bovine urothelium by using oligo(dT) priming was used as template. Three groups of degenerate primers (FR1, FR2, and FR3) were used: a common forward primer F (17 nt), 5′-TGGGA(T/C)A(C/T)IGGIGGICA-3′; and the reverse primers R1 (16 nt), 5′-G(G/A)TCAC(T/A)(C/T)TTGTT(A/G)GC-3′; R2 (17 nt), 5′-A(A/G/T)(A/G)TC(A/G/C)(C/A/G)I(T/C)(T/C)T(G/A)TT(T/G)CC-3′; and R3 (17 nt), 5′-A(A/G/T)(A/G)TC(A/G/T)(C/A/G)I(T/C)TT(G/A)TT(C/A)CC-3′. The amplified fragments were cloned into the pGEM vector and sequenced.
Library Screening and Plasmid Construction. The full-length cDNA of bovine Rab27b was isolated by screening a urothelium library (35) with a 140-bp bovine Rab27b-specific probe prepared from a cDNA fragment that had been obtained by degenerate PCR as described above. Three positive clones of different length were isolated after two rounds of screening and used to reconstitute a full-length cDNA, which was cloned into the pCDNA3.0 vector and then subcloned into the pMAL-C2 (New England Biolabs, Beverly, MA). The GenBank accession number of the bovine Rab27b sequence is AAG45503.
RNA Isolation, RT-PCR, and Northern Blot Analysis. Total RNAs were isolated, by using the TRIzol reagent (Invitrogen) and following the manufacturer's protocol, from 1–3 g of urothelium obtained by scraping bovine bladders, from cultured urothelial cells, or from the bovine tissues indicated in Fig. 1. For RT-PCR, cDNA was synthesized by using a SuperScript first-strand synthesis system (Invitrogen). Primers for PCR amplification of bovine Rab27b were: forward, 5′-ATGACCGATGGGGACTATGATTATCTG-3′, and reverse, 5′-GGAGAAGTCAGCAGAGAAGAAATGTGC-3′. For Northern blot analysis, samples containing equal amounts of total RNA (15 μg) were separated by agarose gel (1%) electrophoresis, transferred to Hybond nitrocellulose membranes (Amersham Biosciences), and then hybridized with a bovine Rab27b-specific probe labeled with 32P by using the Rediprime II DNA-labeling kt (Amersham Biosciences).
Fig. 1.
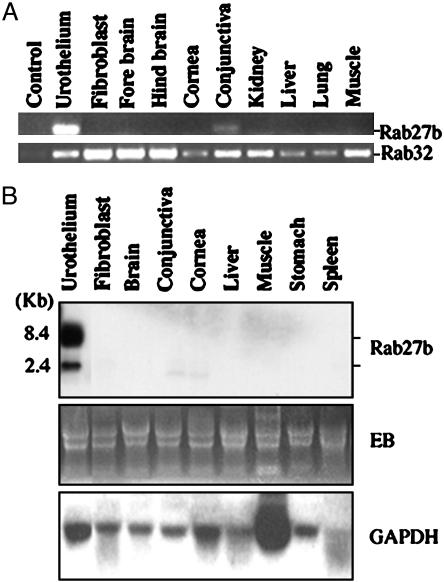
Rab27b mRNA is highly enriched in bovine bladder epithelium. (A) Rab27b (Upper) and Rab32 (Lower) obtained by RT-PCR from various bovine tissues and cells (as labeled) were analyzed by agarose gel electrophoresis. The control sample contained no cDNA. (B) Tissue distribution of bovine Rab27b as determined by Northern blot analysis. Total RNA (15 μg) isolated from bovine tissues was subjected to electrophoreis on a formaldehyde agarose (1%) gel and stained with ethidium bromide (EB, Middle), or blots were probed with a 32P-labeled Rab27b cDNA (Top)ora 32P-labeled GAPDH cDNA (Bottom). Note the presence of a major (8.4 kb) and a minor (2.4 kb) mRNA species in bladder epithelium.
Cell Culture, Antibodies, and Western Blotting. Urothelial cells isolated from bovine bladder were cultured as described (28). The mouse anti-Rab27a monoclonal antibody was purchased from BD Biosciences (Lexingon, KY). Rabbit antibodies against mouse Rab27b (32) and bovine uroplakin II (αUPII), as well as a monoclonal anti-uroplakin III (αUPIII) antibody, were generated as described (6, 9, 36). The monoclonal antibody to Rab6 was kindly provided by Anke Schiedel (New York University School of Medicine, New York) (37). FITC and Texas redlabeled secondary antibodies were from Jackson ImmunoResearch Laboratories and horseradish peroxidase-conjugated anti-rabbit or anti-mouse IgG antibodies were from Roche Diagnostics (Indianapolis, IN). Western blot analysis was performed as described (9).
Immunofluorescence Microscopy. Paraffin sections of urinary bladders from 8- to 12-week-old mice were deparaffinized, rehydrated, and microwaved to expose the antigenic sites. They were then incubated with polyclonal anti-Rab27b antibodies (1:1,000) and monoclonal anti-UPIII antibodies (1:1,000) for 1 h at 37°C, followed by washing with PBS and incubation with FITC-labeled donkey anti-mouse IgG (1:100) and Texas red-labeled donkey anti-rabbit IgG (1:100) for 45 min at room temperature. The sections were viewed with a Zeiss Axiophot fluorescence microscope (Zeiss).
Immunoelectron Microscopy. Mouse bladders were excised and fixed for 2 h in freshly made PHEM buffer (240 mM Pipes/100 mM Hepes/40 mM EGTA/8 mM MgCl2, pH 7.0) containing 2% paraformaldehyde and 0.2% glutaraldehyde. For single labeling, ultra-thin frozen sections were incubated with primary antibodies (1:500 for anti-UPII and 1:250 for anti-Rab27b) at 4°C overnight and then for 30 min at room temperature with protein A conjugated to 10-nm gold particles. Double labeling was performed as described with slight modifications (38) by using 10-nm protein A gold particles for Rab27b and 5-nm protein gold for uroplakin III.
Results
Rab mRNAs Expressed in Urothelial Cells. We identified the sequences of 12 Rabs or Rab-related proteins, including Rab4, -5, -8, -11, -13, -15, -27b, -28, and -32, Ras1, RhoA, and RhoC, in PCR-amplified cDNAs obtained from bovine urothelium preparations by using degenerate primers corresponding to the conserved regions of Rab proteins, followed by cloning. With the exception of Rab27b, most of these proteins were known to function in processes common to all cells, and their mRNAs are present in essentially all tissues, as illustrated for Rab32 in Fig. 1 A Lower (39). In contrast, the mRNA of Rab27b was present in large amounts in urothelium and in very small amounts in conjunctiva, but it was not detected in fibroblasts, kidney, cornea, liver, muscle, stomach, or spleen (Fig. 1 A Upper). Northern blot analysis (Fig. 1B Top) revealed that two forms of Rab27b mRNAs are expressed in bovine bladder epithelium, represented by major (8.4 kb) and minor (2.4 kb) bands that were barely detected in conjunctiva and cornea.
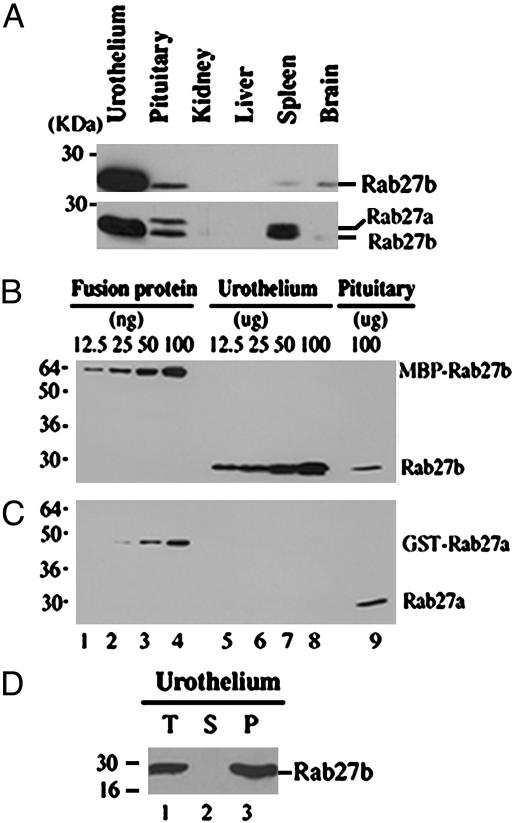
Only the b Isoform of Rab27 Is Expressed in Urothelium. Two isoforms of Rab27, designated Rab27a and Rab27b, which have a high degree of sequence similarity (81%), have been defined (40). Both isoforms were originally identified in platelets, but whereas the Rab27a isoform was expressed in many tissues, Rab27b expression has been previously detected mainly in platelets, digestive tract epithelia, and pituitary gland (41). We analyzed lysates of several mouse tissues, including kidney, liver, spleen, brain, and pituitary gland, by Western blotting using an affinity-purified rabbit antibody against mouse Rab27b. This antibody recognized Rab27b in mouse and bovine tissue. As shown in Fig. 2A Upper, urothelium expressed by far the highest levels of Rab27b, exceeding pituitary, spleen, and brain by at least 10-fold (see also ref. 32). Most strikingly, the only Rab27 isoform detected in urothelial cells was Rab27b, whereas both the a and b isoforms were present in pituitary tissue (Fig. 2 A Lower). Using recombinant fusion proteins of maltose-binding protein (MBP) with bovine Rab27b (MBP-Rab27b) or glutatione S-transferase with mouse Rab27a (GST-Rab27a) as standards, we estimated that the a and b isoforms of Rab27 constituted ≈0.1% and ≈0.02%, respectively, of the total protein of pituitary gland tissue. In contrast, Rab27b represented as much as ≈0.1% of total protein in bovine urothelium, whereas Rab27a was not detectable (Fig. 2 B and C).
Fig. 2.
Only the b isoform of the Rab27 protein is expressed in urothelium. (A) Tissue distribution of Rab27b assessed by Western blot analysis. (Upper) Equal amounts of total protein (100 μg) from the indicated mouse tissue lysates were analyzed by SDS/17% PAGE and immunoblotted with a Rab27b antibody (1:5,000). (Lower) The same membrane was reprobed with a mixture of the monoclonal Rab27a (1:250) and polycolonal Rab27b antibodies (1:5,000). (B and C) Mouse urothelium and pituitary lysates were analyzed by immunoblotting to estimate the amounts of Rab27a and Rab27b in mouse urothelium. As indicated, different amounts of the MBP-tagged bovine Rab27b (MBP-Rab27b) and GST-tagged mouse Rab27a (GST-Rab27a) were used as standards. (D) Rab27b is associated of with the cellular membranes. A bovine urothelium homogenate (T) was fractionated by centrifugation (60 min at 100,000 × g) into cytosolic (S) and pellet (P) fractions. Equivalent amounts of the three samples were subjected to SDS/PAGE and Western blot using an anti-Rab27b antibody.
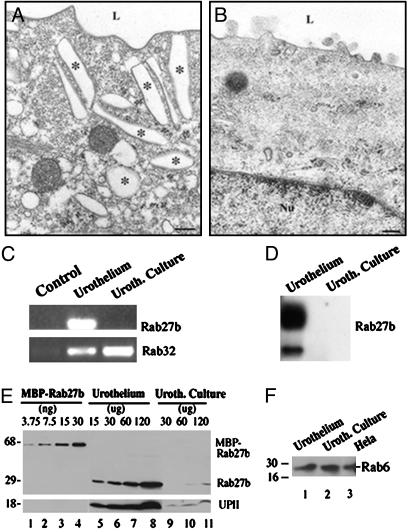
Expression of Rab27b Is Down-Regulated in Cultured Urothelial Cells. We had previously shown that cultured urothelial cells stratify and continue to synthesize small amounts of uroplakins (28) but lack fusiform vesicles (Fig. 3B). Fig. 3 C and D show that Rab27b mRNAs were undetectable in cultured urothelial cells, whereas those for ubiquitously expressed Rabs, such as Rab32 (see also Fig. 1 A), were found at levels comparable to those in normal urothelium (Fig. 3 C and D). Similarly, the protein level of Rab27b in cultured urothelial cells (Fig. 3E, lanes 9–11) was almost 20 times lower than in normal urothelium (Fig. 3E, lanes 5–8). This decrease is similar to that observed for UPII (Fig. 3E Lower, lanes 5–8). In contrast, the protein level of Rab6, a ubiquitously expressed Rab protein that functions in intra-Golgi transport (42), in cultured urothelial cells was similar to those in normal urothelium (Fig. 3F, compare lanes 1 and 2) and HeLa cells (Fig. 3F, lane 3). It is therefore clear that, as is the case with uroplakins, Rab27b is expressed in urothelial cells in a differentiation-dependent fashion.
Fig. 3.
Expression of Rab27b in urothelium correlates with the expression of uroplakins and with the formation of fusiform vesicles. (A) Electron micrograph of the apical portion of a mouse urothelial umbrella cell showing numerous fusiform vesicles as well as the typical scaloped contour of the apical surface membrane. (B) Electron micrograph of the apical cytoplasm of a cultured bovine urothelial cell. Fusiform vesicles are absent from the cytoplasm. (C) Analysis of cDNAs obtained by RT-PCR from urothelium and cultured urothelial cells. The cultured cells continue to express Rab32 but lose the expression of Rab27b. (D) Northern blot analysis demonstrates that Rab27b mRNA is abundant in urothelium but is almost undetectable in cultured urothelial cells. (E) Estimation of Rab27b levels in cultured urothelial by Western blotting (for details see legend to Fig. 2B). Expression of Rab27b and UPII are down-regulated in cultured urothelial cells. The anti-UPII antibody was used at 1:1,000 dilution. (F) Western blot analysis demonstrated that expression levels of the ubiquitously expressed Rab6 are similar in urothelium, cultured urothelial cells, and HeLa cells. Equal amounts of the respective lysates (50 μg each) were analyzed. The anti-Rab6 antibody was used at 1:200 dilution.
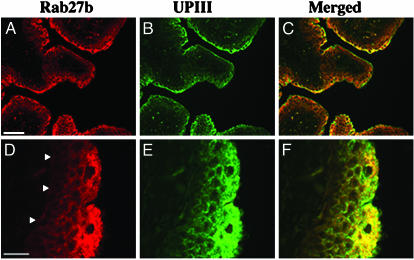
Rab27b Is Associated with the Fusiform Vesicles of Umbrella Cells. Rab proteins perform their functions when bound to membranes of specific organelles (22, 24, 25, 43). A fractionation of bovine urothelial lysates (T) by differential centrifugation (Fig. 2D) showed that Rab27b is recovered in the membrane-containing pellet fraction (P). The subcellular distribution of Rab27b in urothelium (Fig. 4 A and D) was compared with that of UPIII (Fig. 4 B and E) by double immunofluorescence microscopy on histological sections of mouse bladder (Fig. 4). UPIII served as a marker for fusiform vesicles and for the apical plasma membrane of umbrella cells. A gradient of increasing expression of both proteins was observed as basal cells differentiate into the highly specialized superficial umbrella cells. Merged images showed that Rab27b largely colocalized with the UPIII in subplasmalemmal cytoplasmic structures; however, only the latter protein seemed to be present at the apical plasma membrane, which stained only weakly for Rab27b (Fig. 4 C and F).
Fig. 4.
Rab27b and UPIII are highly expressed in umbrella cells. Sections of mouse bladder were immuno-stained with antibodies to Rab27b (A and D; red) and UPIII (B and E; green). (C and F) The merged images. High (A–C) and lowg (D–F) magnifications are shown. The arrowheads in D indicate the contour of the basement membrane. (Scale bars: A, 500 μm; D, 100 μm.)
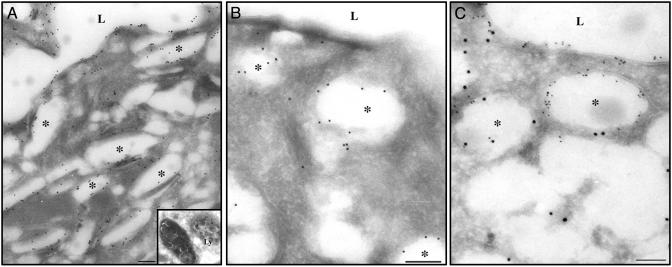
A more precise intracellular localization of Rab27b, as compared with uroplakin III, was obtained by immuno-electron microscopy on frozen sections of mouse bladder urothelium using the immuno-gold technique (Fig. 5). A mouse monoclonal antibody to UPIII stained intensely not only the apical umbrella cell membrane but also the fusiform vesicles, which are abundant in the apical portions of umbrella cell cytoplasm (Fig. 5A). Significant UPIII-labeling was also found in multivesicular bodies (Fig. 5A Inset), which are part of the endosomal–lysosomal system where endocytosed uroplakins undergo degradation (21). Antibody to Rab27b labeled the fusiform vesicles but not the apical plasma membrane (Fig. 5B). Double labeling with both antibodies (Fig. 5C; UPIII with small gold particles, and Rab27b with large gold particles) clearly showed the differential distribution of the two antigens, with UPIII associated with both the apical plasma membranes and fusiform vesicles and Rab27b associated mainly with fusiform vesicles. It should be noted that, in umbrella cells, no significant labeling for Rab27b was observed on the Golgi apparatus, mitochondria, or the nucleus. However, we could not determine whether Rab27b was also associated with the immature post-Golgi (discoidal) vesicles because they are smaller in number, and under the fixation conditions used to obtain frozen sections, both discoidal and fusiform vesicles were distorted in shape and tended to be more spherical, making it difficult to differentiate between the two biogenetically related organelles.
Fig. 5.
Localization of Rab27b to fusiform vesicles in mouse urothelial umbrella cells. Frozen sections from mouse bladder were processed for immuno-gold electron microscopy by using a mouse monoclonal antibody against UPIII (A), an affinity-purified polyclonal rabbit antibody against Rab27b (B), or both antibodies (C). UPIII (10-nm particles in A and 5-nm particles in C) is located along the apical plasma membrane facing the lumen (L), on fusiform vesicles (asterisks), and also on some lysosome-like (Ly) structures (Inset). Rab27b (10-nm particles in B and C) is associated with fusiform vesicles but not with the plasma membrane. (Scale bar, 200 nm for A and 500 nm for B and C.)
Discussion
Urothelial umbrella cells developed unique cytoplasmic organelles, the fusiform vesicles, whose limiting membranes are characterized by the presence of plaques that consist of planar crystalline arrays composed mainly of the four uroplakin polypeptides. Instead of being vehicles for the delivery of soluble content proteins to the bladder luminal space, fusiform vesicles represent storage organelles that serve to expand the plasma membrane by the regulated delivery of their membranes to the apical cell surface (10, 11, 16, 18). Available evidence suggests that fusiform vesicles are formed from immature discoidal precursors that receive uroplakin oligomers from the trans-Golgi network (TGN) (10, 12, 16). As uroplakin complexes accumulate and progressively assemble into crystalline arrays, conversion into mature fusiform vesicles takes place (1, 10–13). It seems likely that, analogous to secretory granules (44, 45), the maturation of discoidal vesicles into fusiform vesicles may involve their homotypic fusion, as well as the continuous removal of membrane regions devoid of uroplakin plaques, which are recycled back to the TGN or to late endosomes. Membrane trafficking events, such as those described for the formation of fusiform vesicles and their ultimate delivery to the cell surface, are almost certainly controlled by specific Rab proteins. Our studies demonstrate that Rab27b, but not Rab27a, is expressed in urothelial umbrella cells at very high levels (Figs. 1 and 2). Biochemical measurements show that Rab27b amounts to ≈0.1% of the total urothelial protein (Fig. 2B). Because this value represents an average for all urothelial cell layers, including the Rab27b-negative cells in the basal and some intermediate cell layers (Fig. 4D), the Rab27b concentration in umbrella cells is likely to be 2- to 5-fold higher.
Even though the two members of the Rab27 subfamily may be functionally equivalent (29), our observation that Rab27b is the only isoform expressed in urothelium explains the absence of urological manifestations in patients with Griscelli syndrome or in ashen mice, in which a defective RAB27A gene leads to pigmentation abnormalities and bleeding disorders. Although no natural Rab27b mutations exist, we predict that malfunction of the RAB27B gene would have pathological manifestations in the urinary tract.
The role we propose for Rab27b in urothelial cells is similar to that assigned to the Rab27a isoforms in melanocytes. Our immunolocalization studies show that Rab27b is associated with fusiform vesicles and not with the apical membrane of umbrella cells or the Golgi apparatus (Figs. 4 and 5). Given current concepts about Rab protein function, these findings support the argument against a role of Rab27b in the retrieval of plasma membrane domains by endocytosis or in the transport of partially assembled uroplakins from the trans-Golgi network to the discoidal vesicles, where the crystalline uroplakin assemblies are formed. Instead, they are compatible with a function of Rab27b in the transport, targeting, or delivery of fusiform vesicles to the apical surface of umbrella cells.
In melanocytes, Rab27a colocalized with melanosomes and was shown to associate via the granuphilin-related bridging molecule, melanophilin, with myosin Va, with which it also colocalized immunocytochemically (30, 46–49). Thus, Rab27a may provide a regulatable molecular link that tethers melanosomes to the cortical actin network close to the plasma membrane (24, 46–49). Although we have not been able to detect granuphilin by Western blotting in mouse urothelial lysates (unpublished data), it seems likely that umbrella cells contain granuphilin-related proteins or other Rab27b effectors that mediate similar cytoskeleton-anchoring functions for fusiform vesicles. By mediating the tethering of fusiform vesicles to the cortical actin network, Rab27b would ensure that those organelles that stay in close apposition to the apical domain of the umbrella cell plasma membrane can serve as readily mobilizable uroplakin reservoirs. Such a tethering function for Rab27b, rather than one that requires its rapid turnover as a GTPase, would explain the high concentrations of Rab27b in umbrella cells (Fig. 2B). Although the precise function of Rab27b in the highly differentiated umbrella cells remains to be established, its closeness to the apical plasma membrane suggests that, in addition to a tethering function, this Rab protein, like other Rab proteins, may also be able to activate N-ethylmaleimide-sensitive factor attachment protein receptor (SNARE)-mediated fusion events (50). Future identification of specific Rab27b regulators and effectors should shed further light on the mechanistic details of the processes by which the delivery of uroplakin assemblies to the cell surface is regulated.
Acknowledgments
We thank Ms. Ivona Gumper for expert immuno-electron microscopy, and Drs. Milton Adesnik, Xue-Ru Wu, and Xiangpeng Kong for careful reading of the manuscript and valuable suggestions. This work was supported by National Institutes of Health Grants DK-52206 (to G.K.), GM-43583 (to D.D.S.), and DK-39753, 52206, and 57269 (to T.-T.S.).
Abbreviation: MBP, maltose-binding protein.
Data deposition: The sequence for the bovine Rab27b reported in this paper has been deposited in the GenBank database (accession no. AY010899).
References
- 1.Wu, X. R., Manabe, M., Yu, J. & Sun, T. T. (1990) J. Biol. Chem. 265, 19170–19179. [PubMed] [Google Scholar]
- 2.Yu, J., Manabe, M., Wu, X. R., Xu, C., Surya, B. & Sun, T. T. (1990) J. Cell Biol. 111, 1207–1216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wu, X. R. & Sun, T. T. (1993) J. Cell Sci. 106, 31–43. [DOI] [PubMed] [Google Scholar]
- 4.Wu, X. R., Lin, J. H., Walz, T., Haner, M., Yu, J., Aebi, U. & Sun, T. T. (1994) J. Biol. Chem. 269, 13716–13724. [PubMed] [Google Scholar]
- 5.Yu, J., Lin, J. H., Wu, X. R. & Sun, T. T. (1994) J. Cell Biol. 125, 171–182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lin, J. H., Wu, X. R., Kreibich, G. & Sun, T. T. (1994) J. Biol. Chem. 269, 1775–1784. [PubMed] [Google Scholar]
- 7.Wu, X. R., Medina, J. J. & Sun, T. T. (1995) J. Biol. Chem. 270, 29752–29759. [DOI] [PubMed] [Google Scholar]
- 8.Liang, F. X., Riedel, I., Deng, F. M., Zhou, G., Xu, C., Wu, X. R., Kong, X. P., Moll, R. & Sun, T. T. (2001) Biochem. J. 355, 13–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tu, L., Sun, T. T. & Kreibich, G. (2002) Mol. Biol. Cell 13, 4221–4230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hicks, R. M. (1965) J. Cell Biol. 26, 25–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Porter, K. R., Kenyon, K. & Badenhausen, S. (1967) Protoplasma 63, 262–274. [PubMed] [Google Scholar]
- 12.Staehelin, L. A., Chlapowski, F. J. & Bonneville, M. A. (1972) J. Cell Biol. 53, 73–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kachar, B., Liang, F., Lins, U., Ding, M., Wu, X. R., Stoffler, D., Aebi, U. & Sun, T. T. (1999) J. Mol. Biol. 285, 595–608. [DOI] [PubMed] [Google Scholar]
- 14.Hu, P., Deng, F. M., Liang, F. X., Hu, C. M., Auerbach, A. B., Shapiro, E., Wu, X. R., Kachar, B. & Sun, T. T. (2000) J. Cell Biol. 151, 961–972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hu, P., Meyers, S., Liang, F. X., Deng, F. M., Kachar, B., Zeidel, M. L. & Sun, T. T. (2002) Am. J. Physiol. Renal Physiol. 283, F1200–F1207. [DOI] [PubMed] [Google Scholar]
- 16.Hicks, R. M. (1966) J. Cell Biol. 28, 21–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Minsky, B. D. & Chlapowski, F. J. (1978) J. Cell Biol. 77, 685–697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lewis, S. A. & de Moura, J. L. (1982) Nature 297, 685–688. [DOI] [PubMed] [Google Scholar]
- 19.Sarikas, S. N. & Chlapowski, F. J. (1989) Cell Tissue Res. 258, 393–401. [DOI] [PubMed] [Google Scholar]
- 20.Truschel, S. T., Ruiz, W. G., Shulman, T., Pilewski, J., Sun, T. T., Zeidel, M. L. & Apodaca, G. (1999) J. Biol. Chem. 274, 15020–15029. [DOI] [PubMed] [Google Scholar]
- 21.Truschel, S. T., Wang, E., Ruiz, W. G., Leung, S. M., Rojas, R., Lavelle, J., Zeidel, M., Stoffer, D. & Apodaca, G. (2002) Mol. Biol. Cell 13, 830–846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Simons, K. & Zerial, M. (1993) Neuron 11, 789–799. [DOI] [PubMed] [Google Scholar]
- 23.Pfeffer, S. R., Soldati, T., Geissler, H., Rancano, C. & Dirac-Svejstrup, B. (1995) Cold Spring Harbor Symp. Quant. Biol. 60, 221–227. [DOI] [PubMed] [Google Scholar]
- 24.Pfeffer, S. R. (1999) Nat. Cell Biol. 1, E17–E22. [DOI] [PubMed] [Google Scholar]
- 25.Pfeffer, S. R. (2001) Trends Cell Biol. 11, 487–491. [DOI] [PubMed] [Google Scholar]
- 26.Segev, N. (2001) Sci. STKE 2001, RE11. [DOI] [PubMed] [Google Scholar]
- 27.Zerial, M. & McBride, H. (2001) Nat. Rev. Mol. Cell. Biol. 2, 107–117. [DOI] [PubMed] [Google Scholar]
- 28.Surya, B., Yu, J., Manabe, M. & Sun, T. T. (1990) J. Cell Sci. 97, 419–432. [DOI] [PubMed] [Google Scholar]
- 29.Barral, D. C., Ramalho, J. S., Anders, R., Hume, A. N., Knapton, H. J., Tolmachova, T., Collinson, L. M., Goulding, D., Authi, K. S. & Seabra, M. C. (2002) J. Clin. Invest. 110, 247–257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wu, X., Wang, F., Rao, K., Sellers, J. R. & Hammer, J. A., III (2002) Mol. Biol. Cell 13, 1735–1749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yi, Z., Yokota, H., Torii, S., Aoki, T., Hosaka, M., Zhao, S., Takata, K., Takeuchi, T. & Izumi, T. (2002) Mol. Cell. Biol. 22, 1858–1867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhao, S., Torii, S., Yokota-Hashimoto, H., Takeuchi, T. & Izumi, T. (2002) Endocrinology 143, 1817–1824. [DOI] [PubMed] [Google Scholar]
- 33.Stinchcombe, J. C., Barral, D. C., Mules, E. H., Booth, S., Hume, A. N., Machesky, L. M., Seabra, M. C. & Griffiths, G. M. (2001) J. Cell Biol. 152, 825–834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bourne, H. R., Sanders, D. A. & McCormick, F. (1991) Nature 349, 117–127. [DOI] [PubMed] [Google Scholar]
- 35.Deng, F. M., Liang, F. X., Tu, L., Resing, K. A., Hu, P., Supino, M., Hu, C. C., Zhou, G., Ding, M., Kreibich, G. & Sun, T. T. (2002) J. Cell Biol. 159, 685–694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Riedel, I., Czernobilsky, B., Lifschitz-Mercer, B., Roth, L. M., Wu, X. R., Sun, T. T. & Moll, R. (2001) Virchows Arch 438, 181–191. [DOI] [PubMed] [Google Scholar]
- 37.Elazar, Z., Mayer, T. & Rothman, J. E. (1994) J. Biol. Chem. 269, 794–797. [PubMed] [Google Scholar]
- 38.Rindler, M. J., Ivanov, I. E., Plesken, H., Rodriguez-Boulan, E. & Sabatini, D. D. (1984) J. Cell Biol. 98, 1304–1319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bao, X., Faris, A. E., Jang, E. K. & Haslam, R. J. (2002) Eur. J. Biochem. 269, 259–271. [DOI] [PubMed] [Google Scholar]
- 40.Chen, D., Guo, J., Miki, T., Tachibana, M. & Gahl, W. A. (1997) Biochem. Mol. Med. 60, 27–37. [DOI] [PubMed] [Google Scholar]
- 41.Tiwari, S., Italiano, J. E., Barral, D. C., Mules, E. H., Novak, E. K., Swank, R. T., Seabra, M. C. & Shivdasani, R. A. (2003) Blood, in press. [DOI] [PubMed]
- 42.Martinez, O., Schmidt, A., Salamero, J., Hoflack, B., Roa, M. & Goud, B. (1994) J. Cell Biol. 127, 1575–1588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Gonzalez, L., Jr., & Scheller, R. H. (1999) Cell 96, 755–758. [DOI] [PubMed] [Google Scholar]
- 44.Rindler, M. J. (1992) Curr. Opin. Cell Biol. 4, 616–622. [DOI] [PubMed] [Google Scholar]
- 45.Tooze, S. A., Martens, G. J. & Huttner, W. B. (2001) Trends Cell Biol. 11, 116–122. [DOI] [PubMed] [Google Scholar]
- 46.Fukuda, M., Kuroda, T. S. & Mikoshiba, K. (2002) J. Biol. Chem. 277, 12432–12436. [DOI] [PubMed] [Google Scholar]
- 47.Strom, M., Hume, A. N., Tarafder, A. K., Barkagianni, E. & Seabra, M. C. (2002) J. Biol. Chem. 277, 25423–25430. [DOI] [PubMed] [Google Scholar]
- 48.Chen, Y., Samaraweera, P., Sun, T. T., Kreibich, G. & Orlow, S. J. (2002) J. Invest. Dermatol. 118, 933–940. [DOI] [PubMed] [Google Scholar]
- 49.Nagashima, K., Torii, S., Yi, Z., Igarashi, M., Okamoto, K., Takeuchi, T. & Izumi, T. (2002) FEBS Lett. 517, 233–238. [DOI] [PubMed] [Google Scholar]
- 50.Solimena, M. & Gerdes, H. H. (2003) Trends Cell Biol. 13, 399–402. [DOI] [PubMed] [Google Scholar]