Abstract
Obesity has become an epidemic problem in western societies, contributing to metabolic diseases, hypertension, and cardiovascular disease. Overweight and obesity are frequently associated with increased plasma levels of aldosterone. Recent evidence suggests that human fat is a highly active endocrine tissue. Therefore, we tested the hypothesis that adipocyte secretory products directly stimulate adrenocortical aldosterone secretion. Secretory products from isolated human adipocytes strongly stimulated steroidogenesis in human adrenocortical cells (NCI-H295R) with a predominant effect on mineralocorticoid secretion. Aldosterone secretion increased 7-fold during 24 h of incubation. This stimulation was comparable to maximal stimulation of these cells with forskolin (2 × 10–5 M). On the molecular level, there was a 10-fold increase in the expression of steroid acute regulatory peptide mRNA. This effect was independent of adipose angiotensin II as revealed by the stimulatory effect of fat cell-conditioned medium even in the presence of the angiotensin type 1 receptor antagonist, valsartan. None of the recently defined adipocytokines accounted for the effect. Mineralocorticoid-stimulating activity was heat sensitive and could be blunted by heating fat cell-conditioned medium to 99°C. Centrifugal filtration based on molecular mass revealed at least two releasing factors: a heat sensitive fraction (molecular mass >50 kDa) representing 60% of total activity, and an inactive fraction (molecular mass <50 kDa). However, the recovery rate increased to 92% when combining these two fractions, indicating the interaction of at least two factors. In conclusion, human adipocytes secrete potent mineralocorticoid-releasing factors, suggesting a direct link between obesity and hypertension.
Overweight and obesity are increasing at an alarming rate worldwide, with a prevalence of 60% and more in western societies (1–3). This tendency constitutes one of the major health problems because obesity has been associated with severe metabolic consequences such as type 2 diabetes, cardiovascular disease, and a higher mortality from certain cancers (4, 5).
White adipose tissue has traditionally been regarded merely as lipid and, consequently, energy storage. However, the importance of adipose tissue as a highly active endocrine organ and its involvement in the body's metabolism and homeostasis has recently been revealed. The first adipocyte-specific hormone to be characterized was leptin (6). It has an important hypothalamic role in the regulation of appetite as well as peripheral effects in liver, muscle, endothelial cells, and adrenal (7, 8). Its discovery was followed by the characterization of numerous other peptides and proteins secreted from white adipocytes with potential autocrine or paracrine effects on adipose metabolism, or with endocrine effects on distant organs (9–11). Importantly, leptin has been shown to stimulate release of tumor necrosis factor α (TNFα) and nitric oxide (NO) from epididymal fat pads in vitro and after its i.v. injections into conscious male rats, suggesting a possible important role of leptin in control of not only TNFα release but also that of NO to regulate blood flow through fat tissue (12).
Obesity is associated with several endocrine disorders (4), including adrenal malfunction. Hyperactivity of the hypothalamus–pituitary–adrenal (HPA) axis and, consequently, increased cortisol secretion and even subclinical Cushing's syndrome have been related to obesity (13–16). Because of the central role of the HPA axis in the body's homeostasis, primary importance has been attributed to HPA axis hyperactivity in the development of other obesity-related endocrine abnormalities (17). In addition, adrenal androgens are elevated in obese children and have been associated with early pubertal development in these children (18–21). Data on adrenal androgens in adults are inconclusive. Whereas adrenal C19 steroid levels in male obese subjects are reported to be reduced (22), a slight increase in dehydroepiandrosterone (DHEA) levels was observed in obese women (23). It is not clear if these abnormalities in HPA axis regulation are located centrally (24) or peripherally at the level of the adrenal itself. At the level of the adrenal, a possible hypersensitivity to ACTH has been discussed (25).
Furthermore, obesity frequently is associated with hyperaldosteronism (26–31). Aldosterone is the most potent mineralocorticoid secreted by the adrenal cortex, promoting sodium retention and elevation of arterial pressure. Hypertension is caused by primary aldosteronism (32), and increased serum aldosterone levels have been linked to the development of obesity hypertension (33), a common disorder related to obesity. It is therefore not surprising that, in a large prospective study, high sodium intake was significantly associated with increased cardiovascular disease risk in obese compared with nonobese subjects (34, 35). However, the molecular basis of the relationship between increased body fat mass and hyperaldosteronism remains unclear. Different mechanisms have been discussed such as a genetic predisposition (36), the involvement of adipose renin–angiotensin system (for review, see ref. 37), adipose receptors for atrial natriuretic peptide (38), or fatty acids transformed through hepatic oxidation (39). However, because adipocytes are highly active endocrine cells, a direct influence of adipocyte secretory products on adrenocortical steroidogenesis has to be considered.
In the present in vitro study, we tested the hypothesis that human adipocyte secretory products are responsible for obesity-associated malfunctions in adrenal steroid, especially increased adrenal mineralocorticoid secretion by directly influencing adrenal function.
Materials and Methods
Human Tissues. Tissue specimens of white human adipose tissue were obtained from healthy (20- to 35-yr-old) women undergoing surgical mammary reduction (n = 10). All women were otherwise healthy and free of metabolic or endocrine diseases. The body mass index (BMI) range of the donors was between 21.4 and 29.2 (25.4 ± 2.8, mean ± SD). Normal adrenal glands were obtained from patients who underwent nephrectomy for renal carcinoma.
Informed consent was obtained from all donors before the surgical procedure. The study was approved by the ethical committee of the Heinrich Heine University of Düsseldorf.
Isolation of Adipocytes and Preparation of Fat Cell-Conditioned Medium (FCCM). After surgical removal, adipose tissue samples of 20–60 g wet weight were immediately transported to the laboratory in DMEM/Nutrient Mix F12 (DMEM/F12, Life Technologies, Karlsruhe, Germany) containing 2% BSA, 100 units/ml penicillin, and 100 μg/ml streptomycin. Adipose tissue was dissected free from fibrous material and visible blood vessels, minced into small pieces, and digested in Kreb's Ringer Bicarbonate buffer (KRB) containing 2% BSA and 120 units/ml collagenase type I from clostridium histolyticum (Sigma) in a shaking water bath for 45–60 min at 37°C. The digested tissue was filtered twice through nylon gauze (250 μm) and washed with KRB containing 0.1% BSA. Two milliliters of isolated floating adipocytes was cultured in culture flasks (Becton Dickinson) containing 5 ml of cell culture medium (DMEM/F12 containing 15 mmol/liter Hepes and 2.5 mmol/liter l-glutamine, supplemented with 1.125 g/liter NaHCO3, 100 units/ml penicillin, and 100 μg/ml streptomycin) with either 10% FCS or under serum-free conditions. Cells were kept at 37°C in a humidified atmosphere of 5% CO2/95% air and cultured for 24 h. The conditioned medium was then collected, carefully avoiding the lipid floating on the top, and kept frozen at –20°C until used.
To determine a heat sensitivity of the effect, FCCM was heated to 99°C for 15 min, denaturated protein was pelleted by centrifugation (5 min, 12,000 × g), and the supernatant was collected and kept frozen at –20°C.
By using filtration devices with a cutoff of 50 kDa (Centricon YM-50, Millipore, Bedford, MA) at 5,000 × g for 20 min, we selectively fractionated FCCM. The resulting fractions were reconstituted to the original volume in culture medium.
Ammonium sulfate precipitation of protein was achieved in 70% ammonium sulfate (40). Samples were solubilized in PBS and desalted by repeated washing using filtration devices (Centricon YM-50).
NCI-H295R Cells. NCI-H295R adrenocortical cells were kindly provided by T. Skurk (German Diabetes Center, Düsseldorf, Germany). Cells were grown in DMEM/F12 supplemented with insulin (66 nM), hydrocortisone (10 nM), 17β-estradiol (10 nM), transferrin (10 μg/ml), selenite (30 nM), penicillin (100 units/ml), streptomycin (100 μg/ml), and 2% FBS. NCI-H295R cells were grown in 75-cm2 flasks (Becton Dickinson) at 37°C in a humidified atmosphere of 5% CO2/95% air. The medium was changed every 3 days, and cells were subcultured every 7 days by using Accutase (PAA Laboratories, Cölbe, Germany) for cell detachment. Cells used for experiments were subcultured from 70% confluent stock cultures into 24-well culture plates (Falcon) at a density of 70,000 cells per cm2 for 96 h. Cells were then treated as described below.
Incubation of NCI-H295R Cells. NCI-H295R cells were incubated with the respective stimulation medium for 24 h or for 3–36 h to evaluate the time dependency of the effect. FCCM was supplemented with insulin (66 nM), hydrocortisone (10 nM), β-estradiol (10 nM), transferrin (10 μg/ml), selenite (30 nM), penicillin (100 units/ml), and streptomycin (100 μg/ml). After incubation, culture medium was collected and kept frozen at –20°C until assayed.
Characterization of FCCM. Cortisol, aldosterone, and DHEA in the incubation medium were measured by direct specific RIA (Diagnostic Products, Los Angeles) according to the manufacturer's protocols. Leptin concentrations in FCCM were determined by enzyme immunoassay to human leptin (R & D Systems), adiponectin concentrations were determined by ELISA to human adiponectin (B-Bridge International, San Jose, CA), IL-6 concentrations were determined by ELISA to human IL-6 (CLB, Peli kine, Amsterdam), and TNFα concentrations were determined by ELISA to human TNFα (Pharmingen).
Triglycerides were deteremined by enzymatic colorimetric assays (Roche Diagnostics), and K+ concentrations were determined with an ion-specific electrode (Hitachi Analyzer 912).
Quantitative TaqMan PCR. To quantify expression of steroid acute regulatory peptide (StAR), we applied TaqMan PCR by using the 7700 Sequence Detector (Applied Biosystems, Foster, CA). Reaction contained 1× TaqMan Universal PCR Master mix, 900 nM of forward and reverse primers, and 200 nM of the TaqMan probes. Primers (forward: 5′-GGCTGGCATGGACACAGACT-3′, reverse: 5′-GGTGAAGCACCATGCAAGTG-3′) and probe (5′-FA M-CCGTGCTCCGCCCTGATGACATAMRA-3′) for human StAR were designed by using the primer design software primer express (Applied Biosystems). TaqMan probes were labeled with a reporter fluorescent dye, FAM (6-carboxyfluorescein), at the 5′ end and a fluorescent dye quencher, TAMRA (6-carboxy-tetramethyl-rhodamine), at the 3′ end. The specificity of PCR primers was tested under normal PCR conditions in a thermocycler.
As an internal control, we used commercial 18S rRNA primers and probe (TaqMan Ribosomal Control Reagents, Applied Biosystems). Internal control primers and probe were added at 50 nM concentration. Thermal cycling proceeded with 50 cycles at 95°C for 15 s and 60°C for 1 min. Data were analyzed by using a Sequence Detector 1.7 program (Perkin–Elmer). Input RNA amounts were calculated with a multiple comparative method for the mRNA of interest and 18S rRNA. Experiments were performed in triplicate for each data point.
Histological and Immunohistochemical Analysis. Paraffin-embedded sections of human adrenal tissue were immunostained as described (41) with antibodies to chromogranin A (clone DAK-A3, DAKO) and 17α-hydroxylase (courtesy of M. R. Waterman, Vanderbilt University School of Medicine, Nashville, TN) for characterization of chromaffin and cortical cells, respectively. Bound antibodies were detected by the linked streptavidin–biotin–peroxidase method (DAKO), and the enzyme reaction was visualized with 3-amino-ethylcarbazole (Dianova, Hamburg, Germany). Monoclonal mouse Ig was used as a negative control.
Results
Characterization of FCCM. Leptin, adiponectin, IL-6, triglycerides, and potassium (K+) concentrations in the FCCM are listed in Table 1. Triglycerides (4.1 × 10–4 M), free fatty acids (10–3 M), leptin (1.8 × 10–9 M), adiponectin (3.8 × 10–10 M), and IL-6 (4 × 10–10 M) were significantly increased in FCCM as compared with culture medium, whereas TNFα concentrations were slightly increased to 1.1 × 10–12 M.
Table 1. Concentrations of K+, triglycerides, leptin, adiponectin, IL-6, and TNFα in FCCM as compared to culture medium.
| Culture medium | FCCM | |
|---|---|---|
| K+, mmol/liter | 4.67 ± 0.24 | 4.78 ± 0.04 |
| Triglycerides, mmol/liter | 0.053 ± 0.023 | 0.41 ± 0.05 |
| Leptin, ng/ml | ND | 29.21 ± 2.57 (1.8 × 10-9 M) |
| Adiponectin, ng/ml | ND | 11.32 ± 2.57 (3.8 × 10-10 M) |
| IL-6, ng/ml | ND | 9.87 ± 2.35 (4 × 10-10 M) |
| TNFα, pg/ml | ND | 18.98 ± 3.91 (1.1 × 10-12 M) |
ND, not detectable; mean ± SEM, n = 4 fat cell preparations.
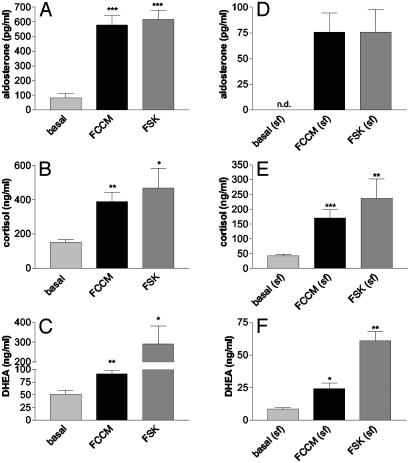
Stimulation of Steroidogenesis by FCCM. FCCM strongly stimulated the secretion of aldosterone, cortisol, and DHEA from NCI-H295R cells in medium containing 10% FBS or at serum-free conditions (Fig. 1). Secretion of aldosterone increased from basal secretion of 80.6 ± 30.7 pg/ml to 578.4 ± 65.8 pg/ml after stimulation with FCCM (P < 0.001). The FCCM-induced stimulation was comparable to stimulation with forskolin 2 × 10–5 M (617.0 ± 63.0 ng/ml; P < 0.001). Cortisol secretion was stimulated from 151.3 ± 16.0 to 389.1 ± 53.5 ng/ml by FCCM (P < 0.01) and to 468.6 ± 115.0 ng/ml by forskolin 2 × 10–5 M (P < 0.05); DHEA secretion was enhanced 1.5 times by FCCM stimulation (from 51.2 ± 7.7 to 91.1 ± 6.6 ng/ml; P < 0.01) and 5.7 times (to 290.9 ± 91.1; P < 0.05) by stimulation with forskolin (Fig. 1 A–C). Under serum-free conditions, basal and stimulated steroid secretion were reduced. Qualitatively, however, FCCM stimulated steroid secretion to the same extent as in the presence of serum, and FCCM-induced aldosterone secretion was comparable to stimulation with forskolin 2 × 10–5 M (Fig. 1 D–F).
Fig. 1.
Secretion of aldosterone (A and D), cortisol (B and E), and DHEA (C and F) from NCI-H295R cells in medium containing 10% FBS (A–C) or in serum-free conditions (D–F). Stimulation with FCCM was compared with the maximal stimulation with forskolin 2 × 10–5 M (FSK). Cells were incubated for 24 h. n.d., not detectable. Mean ± SEM, n = 4 separate fat cell preparations, four wells per experiment. Differences significant from basal secretion are indicated: ***, P < 0.001; **, P < 0.01; *, P < 0.05.
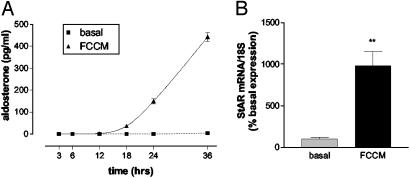
FCCM-induced aldosterone secretion was time dependent with a maximal response after 36 h of stimulation (Fig. 2A). The strong stimulation of aldosterone secretion was reflected on the molecular level. Incubation with FCCM increased the expression on StAR mRNA 9.8-fold over basal expression levels (P < 0.05; Fig. 2B).
Fig. 2.
(A) Time-dependency of the FCCM effect on aldosterone secretion from H295R cells under serum free conditions. Mean ± SEM, n = 4 wells per time point. (B) Effect of FCCM on the mRNA expression for StAR in NCI-H295R cells under serum-free conditions. FCCM significantly up-regulated the mRNA levels of StAR as detected by quantitative TaqMan PCR. Data represent mean ± SEM, n = 3; **, P < 0.01.
Leptin (10–11 Mto10–7 M), adiponectin (10–10 Mto10–7 M), IL-6 (10–11 Mto10–9 M), and TNFα (10–11 Mto10–8 Mng/ml) had no stimulatory effect on steroidogenesis in NCI-H295R cells. TNFα (10–7 M) and IL-6 (10–8 M) slightly stimulated aldosterone secretion. In addition, incubation of H295R cells with leptin (10–8 M), adiponectin (10–9 M), IL-6 (10–9 M), or TNFα (10–9 M) together with the >50-kDa fraction did not further enhance the stimulatory effect of the >50-kDa fraction (data not shown).
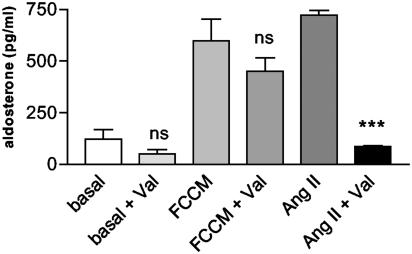
The Effect Is Not Mediated by Angiotensin II (AngII) Produced by Adipocytes. As previously shown, stimulation of aldosterone secretion from NCI-H295R cells by AngII is mediated by AngII type 1 (AT1) receptor (42). Therefore, the possible involvement of adipocyte AngII in the FCCM-stimulated aldosterone secretion was tested by specifically blocking the AngII-effect with Valsartan, an AT1 receptor antagonist (kindly provided by Novartis, Basel). AngII-stimulated aldosterone secretion was completely blocked by addition of Valsartan (10–5 M). In contrast, FCCM-stimulated aldosterone secretion was not significantly reduced by the concomitant incubation with Valsartan (Fig. 3).
Fig. 3.
Angiotensin II (10–5 M) stimulated aldosterone secretion (AngII) is significantly inhibited (***, P < 0.001) by addition of the angiotensin type 1 receptor antagonist valsartan (Val) (10–5 M), whereas it had no significant (ns) effect on basal and FCCM-stimulated aldosterone secretion. FCCM contained 10% FBS. Mean ± SEM, n = 3 separate fat cell preparations, four wells per experiment.
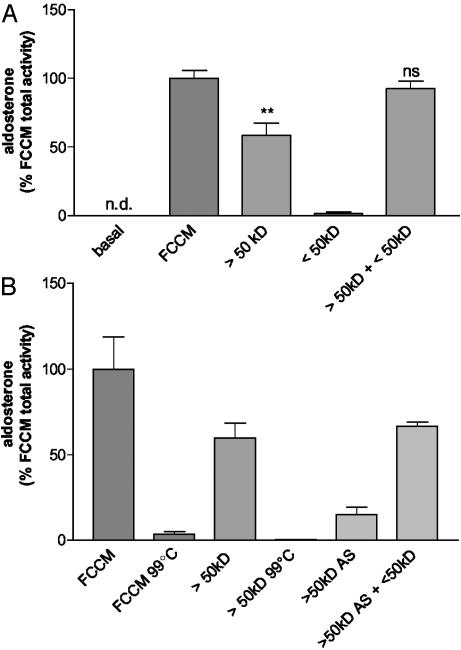
Stimulatory Effect Depends on at Least Two Factors. Centrifugal filtration of FCCM based on molecular mass showed an active fraction (molecular mass >50 kDa) representing 60% of total activity and an inactive fraction (molecular mass <50 kDa). However, when combining these two fractions, the recovery rate increased to 92%, not significantly different from FCCM total activity, indicating the interaction of least two factors in the FCCM (Fig. 4A).
Fig. 4.
(A) Fractionation of FCCM (serum free) revealed the existence of at least two releasing factors: an active (MW >50kD) and an inactive (MW <50kD) fraction. Stimulation with >50-kDa fraction alone leads to a significant decrease to 58.4 ± 9.14% of total FCCM activity (**, P < 0.005), whereas the recovery rate increased to 92.4 ± 5.5% (not significantly different from FCCM; ns) when combining these two fractions. Data represent mean ± SEM. (B) FCCM is heat-sensitive and precipitates with ammonium sulfate. Heating FCCM (FCCM 99°C) and the active >50-kDa fraction (>50kD 99°C) blunted the stimulatory effect on aldosterone secretion. After ammonium sulfate precipitation of the >50-kDa fraction (>50kD AS), 15.0% of the FCCM total activity were recovered. By adding the <50-kDa fraction (>50kD AS +<50kD), 66.7% activity were recovered. n = 3 experiments, four wells per experiment.
Steroidogenic Activity Is Heat Sensitive and Precipitates with Ammonium Sulfate. Heating FCCM for 15 min at 99°C destroyed the stimulatory effect (Fig. 4B). To avoid unspecific effects of heating, NCI-H295R cells were stimulated with heated culture medium containing forskolin (2 × 10–5 M). No significant reduction of forskolin-stimulated steroidogenesis was observed.
After ammonium sulfate precipitation of the >50-kDa FCCM-fraction, 15.0% of the FCCM total activity was recovered in the solubilized precipitate. By adding the <50-kDa FCCM fraction to the solubilized precipitate, the stimulatory effect increased to 66.7% of FCCM total activity. These physicochemical properties strongly suggest that the activity depends on protein released from the fat cells.
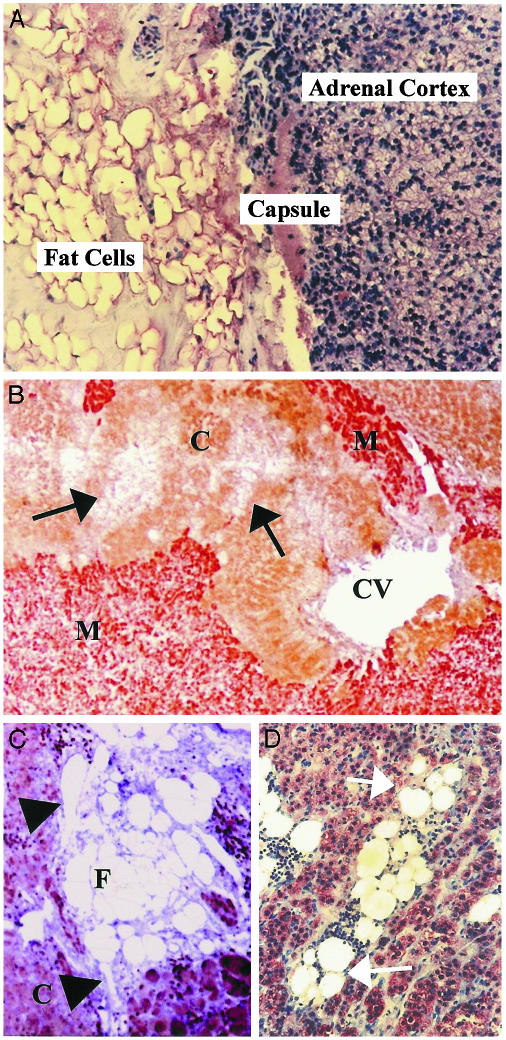
Fat Cells Are Located Within the Human Adrenal. The adrenal glands are embedded in adipose tissue (Fig. 5A). However, fat cells were also found within the adrenal gland where they were in direct contact with the steroid-producing adrenocortical cells (Fig. 5 B–D). Fat cells were frequently accompanying adrenal vessels or they occurred as clusters located with the adrenal gland.
Fig. 5.
Paraffin sections of normal human adrenal gland. (A) Human adrenals are embedded in periadrenal fat. (B–D) Adrenocortical cells are immunostained (brown) with an antibody against 17α-hydroxylase; in B, chromaffin cells are immunostained with an antibody against chromogranin A (red staining). (C) Adipose tissue may accompany adrenal vessels (arrow heads) or occur within the adrenal cortex in direct contact with adrenocortical cells (arrows in B and D). C, adrenal cortex; M, adrenal medulla; CV, central vein; arrows demonstrate clusters of fat cells.
Discussion
The present data indicate that human adipocytes secrete potent mineralocorticoid-releasing factors. Fat cell-derived secretagogues stimulated adrenocortical steroidogenesis with the most prominent effect on aldosterone release. These findings suggest a hitherto unknown direct involvement of adipose tissue in the regulation of adrenocortical function and especially mineralocorticoid secretion.
Human adipose tissue secretes a wide variety of endocrinologically active factors. In our system of isolated human adipocytes, the cells were functionally active and released adipokinins, some of which, such as leptin, IL-6, TNFα, or AngII are known to influence adrenal function either by stimulating or inhibiting steroidogenesis (43, 44). To our knowledge, no data are published on the effect of adiponectin on adrenocortical steroidogenesis. In concentrations up to 10–8 M adiponectin did not influence steroidogenesis. At a concentration of 10–7 M, cells detached and became necrotic. Plasma adiponectin levels are inversely correlated with markers of obesity (45) and with insulin levels (46), and therefore, in accordance with our data, adiponectin is unlikely to be responsible for obesity-associated hyperaldosteronism. Adipocytes express a complete renin–angiotensin system and consequently are able to produce AngII (for review, see ref. 37). AngII stimulation of aldosterone release is mediated by AT1 receptors (42). However, the AT1 receptor antagonist valsartan had no significant effect on FCCM-stimulated aldosterone release, despite blunting AngII-stimulated aldosterone release, indicating a AngII-independent stimulation. In addition to AngII, aldosterone secretion is regulated by plasma potassium levels (47). Potassium concentrations in the conditioned media were not different from concentrations in the culture medium, and therefore were not responsible for the mineralocorticoid-releasing activity of the conditioned media.
Adipokinins leptin, adiponectin, IL-6, and TNFα did not stimulate steroidogenesis when added at concentrations comparable to those in the FCCM.
Maximum stimulation of aldosterone release from NCI-H295R cells is achieved with forskolin 2 × 10–5 M by activating the protein kinase A pathway (48). Mineralocorticoid release stimulated by FCCM was identical to the maximal stimulation with forskolin. Therefore, these adipocyte-derived factors constitute an extremely strong mineralocorticoid-releasing activity. FCCM stimulated steroidogenesis when serum was present in the medium or under serum-free conditions. Therefore, serum was not responsible for the stimulatory effect of FCCM, although absolute hormone levels were higher in the presence of serum.
The human adrenocortical cell line NCI-H295R is an adherent growing subpopulation of NCI-H295 cells (48, 49). These cells are a widely accepted model for human adrenocortical studies and represent the only cell line to maintain the ability to produce all adrenocortical steroids and express the three major pathways of adrenal steroidogenesis including the main steroidogenic enzymes. They have extensively been used to characterize the regulation of steroidogenesis including the regulation of aldosterone synthesis (50, 51). Our initial data from normal human adrenal cells in primary culture have also shown the steroidogenic effects of FCCM on these cells (unpublished observation).
The steroidogenic activity is heat sensitive and can be precipitated by 70% ammonium sulfate, producing strong evidence for the involvement of protein. By fractionating FCCM using filtration devices, the centricon-50 retentate contained 60% activity. Interestingly, the activity of this heat-sensitive fraction was potentiated by coincubation with the lower molecular mass fraction (<50 kDa) that had no stimulatory effect of its own. These observations indicate the involvement of at least two factors that interact in their stimulation of aldosterone release. To determine whether one of the prominent adipocyte secretory products was responsible for the activity in the lower molecular mass fraction, cells were incubated with leptin, IL-6, adiponectin, or TNFα either alone or together with the >50-kDa fraction. However, these adipokinins had no stimulatory effect per se or in combination with the >50-kDa fraction of the FCCM. Further studies will be needed to characterize the adipose mineralocorticoid-releasing factors.
In the present study, adipocyte secretory products in concert strongly stimulated adrenal aldosterone release by using adipocyte conditioned media or cocultures of human adipocytes and NCI-H295R cells (data not shown). However, in vitro studies of isolated secretory products only insufficiently reflect the conditions in vivo and will not reveal these complex interactions. The use of adipocyte conditioned media or cocultures were the prerequisite to define this effect. In the past, similar systems proved to be extremely potent to characterize important regulatory loops. Concerning the regulation of adrenocortical steroidogenesis, we were able to define extrapituitary effects by using coculture systems and conditioned media (43, 52, 53). Recently, it could be shown in a coculture system that secretory products from human adipocytes increase insulin resistance in isolated muscle cells (54). Similarly, the characterization of pituitary releasing hormones (55) and of the effect of endothelial NO on smooth muscle cells (56) were based on similar cell culture systems.
How do adipocyte secretory products reach the adrenal cortex? Many adipocyte secretory products influence adipocyte metabolism in an auto/paracrine manner. However, several factors are released into the circulation and can be measured in plasma (9, 10, 12). Thus, mineralocorticoid-releasing factors secreted from s.c. or visceral fat into the circulation may reach the adrenal in an endocrine manner in concentrations suffcient to stimulate adrenocortical steroidogenesis.
However, fat cells are present within the adrenal in direct contact with steroid-producing adrenocortical cells. This close cellular proximity forms the prerequisite for paracrine interactions and indicates a direct stimulation of steroidogenesis by adipocyte secretory products. Recently, our group published the case of an intraadrenal myelolipoma associated with corticotropin-independent Cushing's syndrome (57) with highly intermingled myelolipomatous and adrenocortical tumor cells with ample direct cell–cell contact. It is conceivable that paracrine influences of secretory products from the myelolipoma cells were responsible for the enhanced cortisol secretion in this patient.
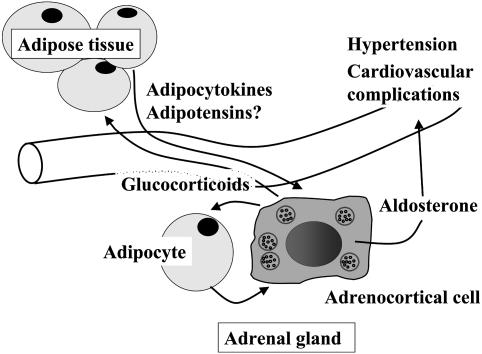
In conclusion, our data demonstrate that adipocytesecretory products regulate adrenocortical aldosterone secretion and enzyme expression. Thus, adipocytes secrete mineralocorticoid-releasing factors that may contribute to the hyperaldosteronism seen in obese individuals. This suggests a direct link between increased fat cell mass and hypertension (Fig. 6) Identification and further characterization of these factors should open new avenues for the treatment of obesity hypertension.
Fig. 6.
Adipocytes release secretagogues that stimulate adrenocortical steroidogenesis with a potent effect on mineralocorticoid secretion. Enhanced aldosterone levels may be responsible for hypertension and cardiovascular complications associated with obesity. Adrenal glucocorticoids stimulate fat cell growth and proliferation. Arrows indicate stimulation.
Acknowledgments
We thank Prof. R. R. Olbrisch and his team, Department of Plastic Surgery, Florence Nightingale Hospital Düsseldorf, for support in obtaining adipose tissue samples. This work was supported by Deutsche Forschungsgemeinschaft Grants Eh 161/2-4 (to M.E.-B. and S.R.B.) and SCHE 225/11-1 (to W.A.S. and S.R.B.).
Abbreviations: FCCM, fat cell-conditioned medium; HPA, hypothalamus–pituitary–adrenal; DHEA, dehydroepiandrosterone; TNFα, tumor necrosis factor α; StAR, steroid acute regulatory peptide; AngII, angiotensin II; AT1, AngII type 1.
References
- 1.World Health Organization (1998) Obesity—Preventing and Managing the Global Epidemic: Report of a WHO Consultation on Obesity (W.H.O., Geneva). [PubMed]
- 2.Flegal, K. M., Carroll, M. D., Ogden, C. L. & Johnson, C. L. (2002) J. Am. Med. Assoc. 288, 1723–1727. [DOI] [PubMed] [Google Scholar]
- 3.Cameron, A. J., Welborn, T. A., Zimmet, P. Z., Dunstan, D. W., Owen, N., Salmon, J., Dalton, M., Jolley, D. & Shaw, J. E. (2003) Med. J. Aust. 178, 427–432. [DOI] [PubMed] [Google Scholar]
- 4.Kopelman, P. G. (2000) Nature 404, 635–643. [DOI] [PubMed] [Google Scholar]
- 5.Calle, E. E., Rodriguez, C., Walker-Thurmond, K. & Thun, M. J. (2003) N. Engl. J. Med. 348, 1625–1638. [DOI] [PubMed] [Google Scholar]
- 6.Zhang, Y., Proenca, R., Maffei, M., Barone, M., Leopold, L. & Friedman, J. M. (1994) Nature 372, 425–432. [DOI] [PubMed] [Google Scholar]
- 7.Havel, P. J. (2002) Curr. Opin. Lipidol. 13, 51–59. [DOI] [PubMed] [Google Scholar]
- 8.Bornstein, S. R., Uhlmann, K., Haidan, A., Ehrhart-Bornstein, M. & Scherbaum, W. A. (1997) Diabetes 46, 1235–1238. [DOI] [PubMed] [Google Scholar]
- 9.Kim, S. & Moustaid-Moussa, N. (2000) J. Nutr. 130, 3110S–3115S. [DOI] [PubMed] [Google Scholar]
- 10.Trayhurn, P. & Beattie, J. H. (2001) Proc. Nutr. Soc. 60, 329–339. [DOI] [PubMed] [Google Scholar]
- 11.Skurk, T., Van, H., V, Lee, Y. M., Wirth, A. & Hauner, H. (2002) Horm. Metab. Res. 34, 659–663. [DOI] [PubMed] [Google Scholar]
- 12.Mastronardi, C. A., Yu, W. H. & McCann, S. M. (2002) Proc. Natl. Acad. Sci. USA 99, 5721–5726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Boushaki, F. Z., Rasio, E. & Serri, O. (1997) Clin. Endocrinol. (Oxford) 46, 461–466. [DOI] [PubMed] [Google Scholar]
- 14.Rosmond, R., Dallman, M. F. & Bjorntorp, P. (1998) J. Clin. Endocrinol. Metab 83, 1853–1859. [DOI] [PubMed] [Google Scholar]
- 15.Duclos, M., Gatta, B., Corcuff, J. B., Rashedi, M., Pehourcq, F. & Roger, P. (2001) Clin. Endocrinol. (Oxford) 55, 447–454. [DOI] [PubMed] [Google Scholar]
- 16.Rask, E., Walker, B. R., Soderberg, S., Livingstone, D. E., Eliasson, M., Johnson, O., Andrew, R. & Olsson, T. (2002) J. Clin. Endocrinol. Metab. 87, 3330–3336. [DOI] [PubMed] [Google Scholar]
- 17.Bjorntorp, P. & Rosmond, R. (2000) Int. J. Obes. Relat. Metab. Disord. 24, Suppl. 2, S80–S85. [DOI] [PubMed] [Google Scholar]
- 18.L'Allemand, D., Schmidt, S., Rousson, V., Brabant, G., Gasser, T. & Gruters, A. (2002) Eur. J. Endocrinol. 146, 537–543. [DOI] [PubMed] [Google Scholar]
- 19.Genazzani, A. R., Pintor, C. & Corda, R. (1978) J. Clin. Endocrinol. Metab. 47, 974–979. [DOI] [PubMed] [Google Scholar]
- 20.Pintor, C., Loche, S., Faedda, A., Fanni, V., Nurchi, A. M. & Corda, R. (1984) Horm. Metab Res. 16, 544–548. [DOI] [PubMed] [Google Scholar]
- 21.Katz, S. H., Hediger, M. L., Zemel, B. S. & Parks, J. S. (1986) Hypertension 8, 277–284. [DOI] [PubMed] [Google Scholar]
- 22.Tchernof, A., Despres, J. P., Belanger, A., Dupont, A., Prud'homme, D., Moorjani, S., Lupien, P. J. & Labrie, F. (1995) Metabolism 44, 513–519. [DOI] [PubMed] [Google Scholar]
- 23.MacCario, M., Mazza, E., Ramunni, J., Oleandri, S. E., Savio, P., Grottoli, S., Rossetto, R., Procopio, M., Gauna, C. & Ghigo, E. (1999) Clin. Endocrinol. (Oxford) 50, 595–600. [DOI] [PubMed] [Google Scholar]
- 24.Bjorntorp, P. (1997) J. Endocrinol. 155, 193–195. [DOI] [PubMed] [Google Scholar]
- 25.MacCario, M., Grottoli, S., Divito, L., Rossetto, R., Tassone, F., Ganzaroli, C., Oleandri, S. E., Arvat, E. & Ghigo, E. (2000) Clin. Endocrinol. (Oxford) 53, 437–444. [DOI] [PubMed] [Google Scholar]
- 26.Rocchini, A. P., Key, J., Bondie, D., Chico, R., Moorehead, C., Katch, V. & Martin, M. (1989) N. Engl. J. Med. 321, 580–585. [DOI] [PubMed] [Google Scholar]
- 27.Licata, G., Scaglione, R., Ganguzza, A., Corrao, S., Donatelli, M., Parrinello, G., Dichiara, M. A., Merlino, G. & Cecala, M. G. (1994) Am. J. Hypertens. 7, 314–320. [DOI] [PubMed] [Google Scholar]
- 28.Egan, B. M., Stepniakowski, K. & Goodfriend, T. L. (1994) Am. J. Hypertens. 7, 886–893. [DOI] [PubMed] [Google Scholar]
- 29.Goodfriend, T. L., Egan, B. M. & Kelley, D. E. (1998) Endocr. Res. 24, 789–796. [DOI] [PubMed] [Google Scholar]
- 30.Suter, P. M., Locher, R., Hasler, E. & Vetter, W. (1998) Am. J. Hypertens. 11, 1305–1311. [DOI] [PubMed] [Google Scholar]
- 31.El Gharbawy, A. H., Nadig, V. S., Kotchen, J. M., Grim, C. E., Sagar, K. B., Kaldunski, M., Hamet, P., Pausova, Z., Gaudet, D., Gossard, F., et al. (2001) Hypertension 37, 845–850. [DOI] [PubMed] [Google Scholar]
- 32.Stowasser, M., Gunasekera, T. G. & Gordon, R. D. (2001) Clin. Exp. Pharmacol. Physiol. 28, 1087–1090. [DOI] [PubMed] [Google Scholar]
- 33.Thakur, V., Richards, R. & Reisin, E. (2001) Am. J. Med. Sci. 321, 242–248. [DOI] [PubMed] [Google Scholar]
- 34.He, J., Ogden, L. G., Vupputuri, S., Bazzano, L. A., Loria, C. & Whelton, P. K. (1999) J. Am. Med. Assoc. 282, 2027–2034. [DOI] [PubMed] [Google Scholar]
- 35.He, J., Ogden, L. G., Bazzano, L. A., Vupputuri, S., Loria, C. & Whelton, P. K. (2002) Arch. Intern. Med. 162, 1619–1624. [DOI] [PubMed] [Google Scholar]
- 36.Russo, P., Siani, A., Venezia, A., Iacone, R., Russo, O., Barba, G., D'Elia, L., Cappuccio, F. P. & Strazzullo, P. (2002) J. Hypertens. 20, 1785–1792. [DOI] [PubMed] [Google Scholar]
- 37.Engeli, S. & Sharma, A. M. (2001) J. Mol. Med. 79, 21–29. [DOI] [PubMed] [Google Scholar]
- 38.Dessi-Fulgheri, P., Sarzani, R., Tamburrini, P., Moraca, A., Espinosa, E., Cola, G., Giantomassi, L. & Rappelli, A. (1997) J. Hypertens. 15, 1695–1699. [DOI] [PubMed] [Google Scholar]
- 39.Goodfriend, T. L., Ball, D. L. & Gardner, H. W. (2002) Prostaglandins Leukotrienes Essent. Fatty Acids 67, 163–167. [DOI] [PubMed] [Google Scholar]
- 40.Page, M., Baines, M. G. & Thorpe, R. (1994) in Basic Protein and Peptide Protocols, ed. Walker, J. M. (Humana, Totowa, NJ), pp. 407–432.
- 41.González-Hernández, J. A., Bornstein, S. R., Ehrhart-Bornstein, M., Geschwend, J. E., Adler, G. & Scherbaum, W. A. (1994) Cell Tissue Res. 278, 201–205. [DOI] [PubMed] [Google Scholar]
- 42.Hilbers, U., Peters, J., Bornstein, S. R., Correa, F. M. A., Jöhren, O., Saavedra, J. M. & Ehrhart-Bornstein, M. (1999) Hypertension 33, 1025–1030. [DOI] [PubMed] [Google Scholar]
- 43.Ehrhart-Bornstein, M., Hinson, J. P., Bornstein, S. R., Scherbaum, W. A. & Vinson, G. P. (1998) Endocr. Rev. 19, 101–143. [DOI] [PubMed] [Google Scholar]
- 44.Marx, C., Ehrhart-Bornstein, M., Scherbaum, W. A. & Bornstein, S. R. (1998) Horm. Metab. Res. 30, 416–420. [DOI] [PubMed] [Google Scholar]
- 45.Arita, Y., Kihara, S., Ouchi, N., Takahashi, M., Maeda, K., Miyagawa, J., Hotta, K., Shimomura, I., Nakamura, T., Miyaoka, K., et al. (1999) Biochem. Biophys. Res. Commun. 257, 79–83. [DOI] [PubMed] [Google Scholar]
- 46.Mohlig, M., Wegewitz, U., Osterhoff, M., Isken, F., Ristow, M., Pfeiffer, A. F. & Spranger, J. (2002) Horm. Metab Res. 34, 655–658. [DOI] [PubMed] [Google Scholar]
- 47.Quinn, S. J. & Williams, G. H. (1992) in The Adrenal Gland, ed. James, V. H. T. (Raven, New York), pp. 159–189.
- 48.Rainey, W. E., Bird, I. M. & Mason, J. I. (1994) Mol. Cell Endocrinol. 100, 45–50. [DOI] [PubMed] [Google Scholar]
- 49.Bird, I. M., Pasquarette, M. M., Rainey, W. E. & Mason, J. I. (1996) J. Clin. Endocrinol. Metab 81, 2171–2178. [DOI] [PubMed] [Google Scholar]
- 50.Bird, I. M., Hanley, N. A., Word, R. A., Mathis, J. M., McCarthy, J. L., Mason, J. I. & Rainey, W. E. (1993) Endocrinology 133, 1555–1561. [DOI] [PubMed] [Google Scholar]
- 51.Condon, J. C., Pezzi, V., Drummond, B. M., Yin, S. & Rainey, W. E. (2002) Endocrinology 143, 3651–3657. [DOI] [PubMed] [Google Scholar]
- 52.Haidan, A., Bornstein, S. R., Glasow, A., Uhlmann, K., Lübke, C. & Ehrhart-Bornstein, M. (1998) Endocrinology 139, 772–780. [DOI] [PubMed] [Google Scholar]
- 53.Wolkersdörfer, G. W., Lohmann, T., Marx, C., Schröder, S., Pfeiffer, R., Stahl, H.-D., Scherbaum, W. A., Chrousos, G. P. & Bornstein, S. R. (1999) J. Clin. Endocrinol. Metab. 84, 4220–4227. [DOI] [PubMed] [Google Scholar]
- 54.Dietze, D., Koenen, M., Rohrig, K., Horikoshi, H., Hauner, H. & Eckel, J. (2002) Diabetes 51, 2369–2376. [DOI] [PubMed] [Google Scholar]
- 55.Krulich, L., Dhariwal, A. P. & McCann, S. M. (1968) Endocrinology 83, 783–790. [DOI] [PubMed] [Google Scholar]
- 56.Furchgott, R. F. & Zawadzki, J. V. (1980) Nature 288, 373–376. [DOI] [PubMed] [Google Scholar]
- 57.Vrezas, I., Wentworth, P. & Bornstein, S. R. (2003) Endocr. Res. 29, 67–71. [DOI] [PubMed] [Google Scholar]